Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Study Objectives and Novelty
2. Materials and Methods
2.1. Overview of Data Sources and Study Area
2.2. Data Integration and Preparation
2.3. Statistical and Spatial Analyses
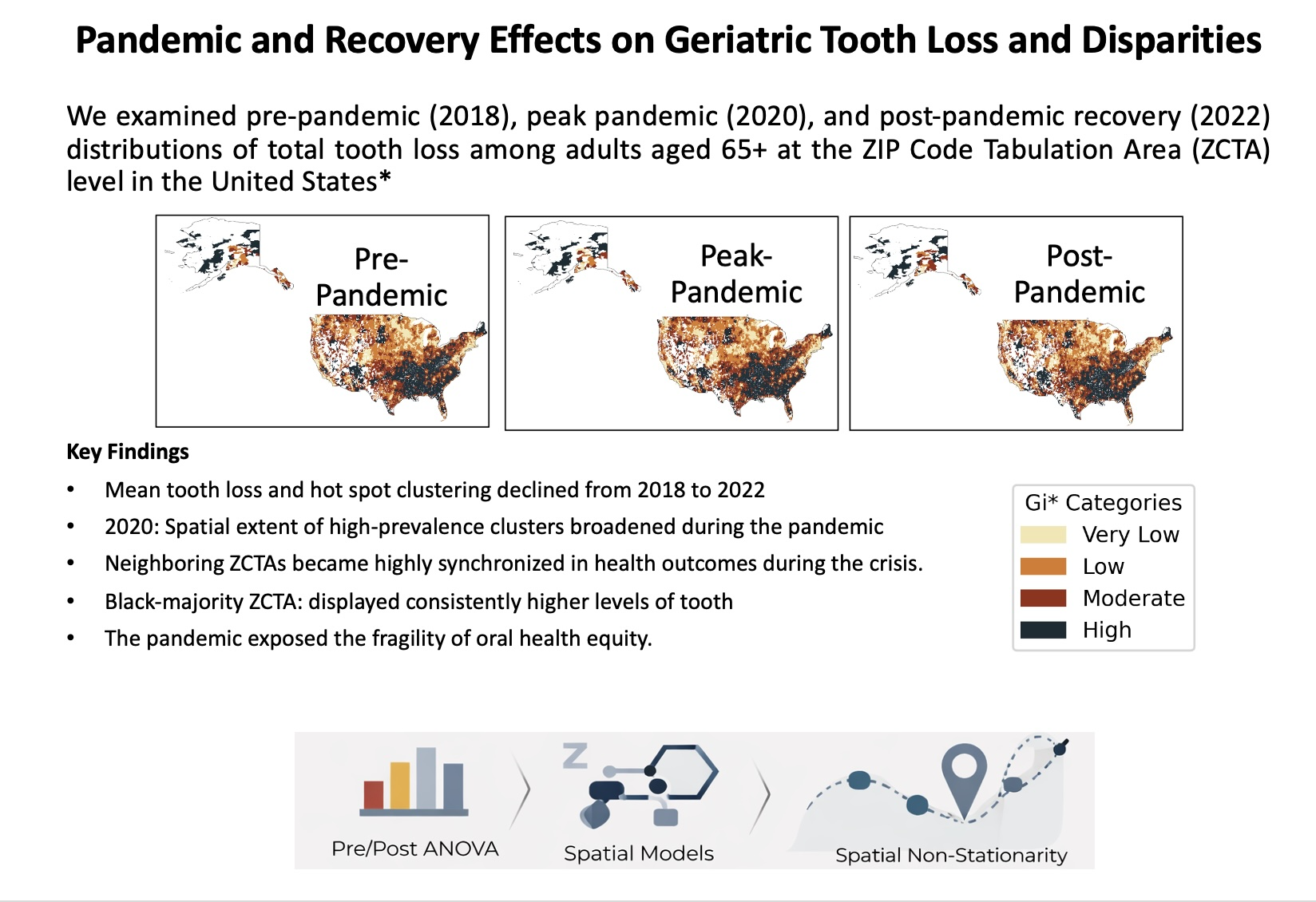
3. Results
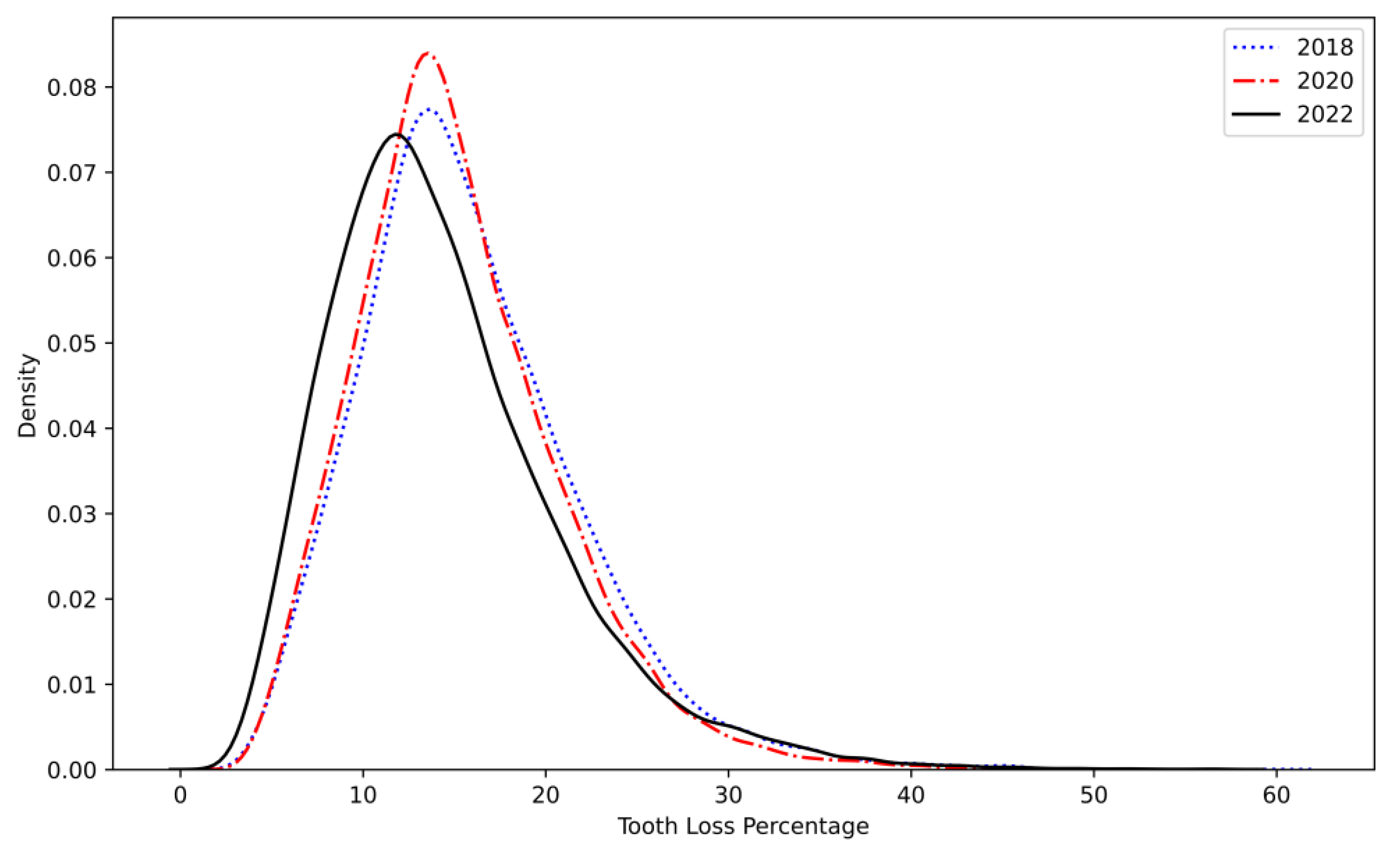
3.1. Descriptive Statistics: Tooth Loss Prevalence
3.2. Descriptive Statistics: Covariates
3.3. ANOVA and Pairwise Comparisons
3.4. Ordinary Least Squares (OLS) Regression: Without Interaction Terms
3.5. OLS Regression with Year × Race/Ethnicity-Majority Interactions
3.6. Spatial Autocorrelation (Global Moran’s I)
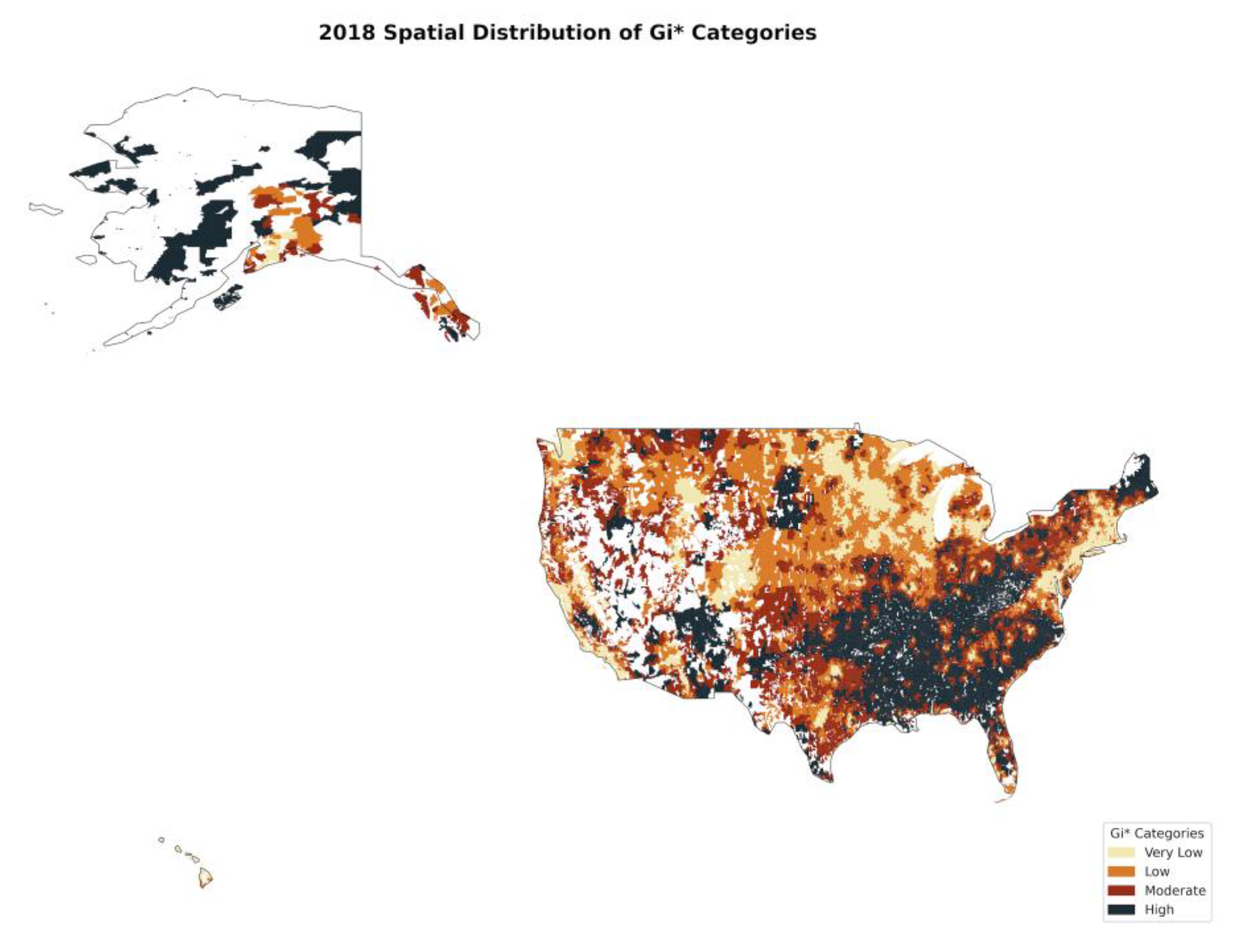
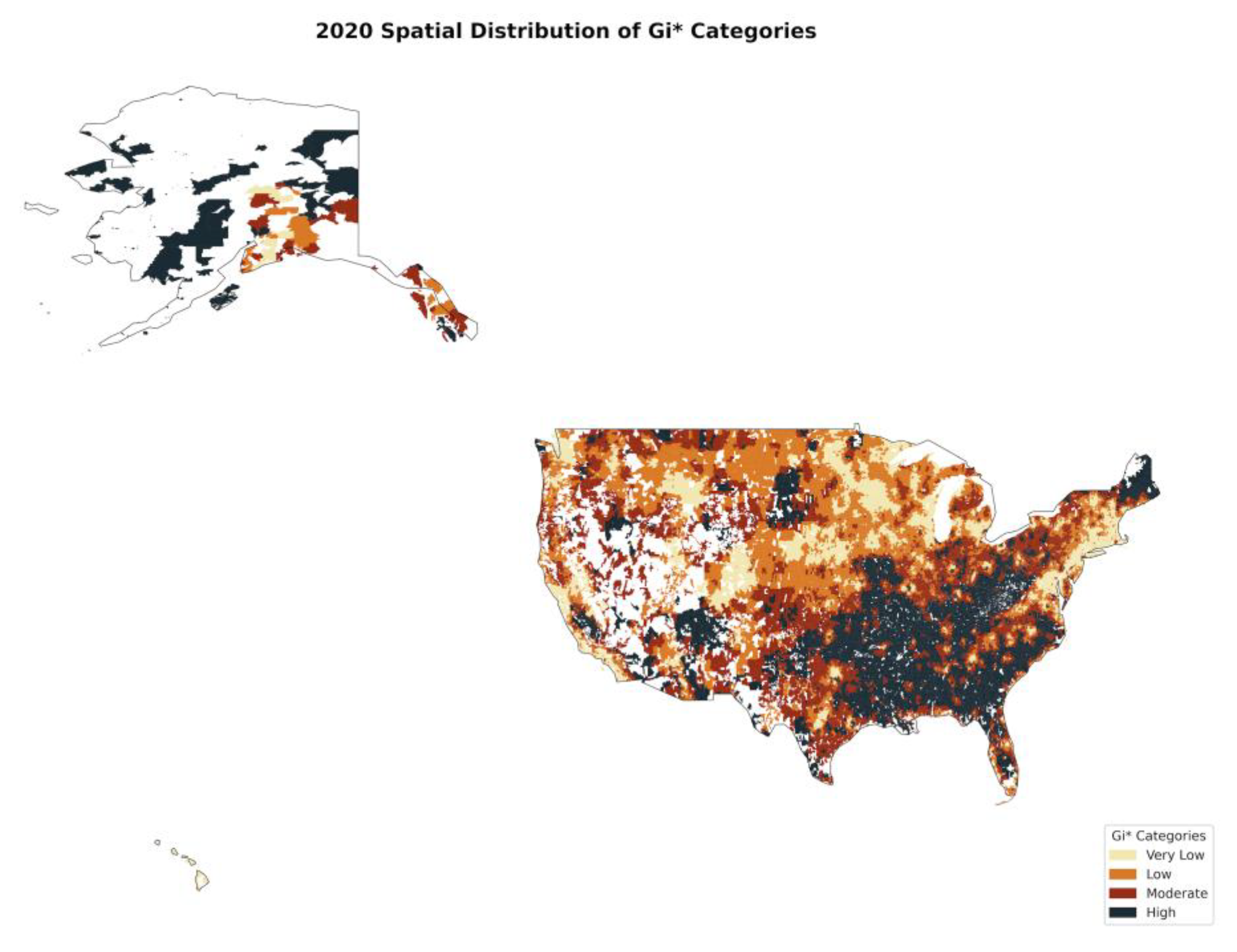
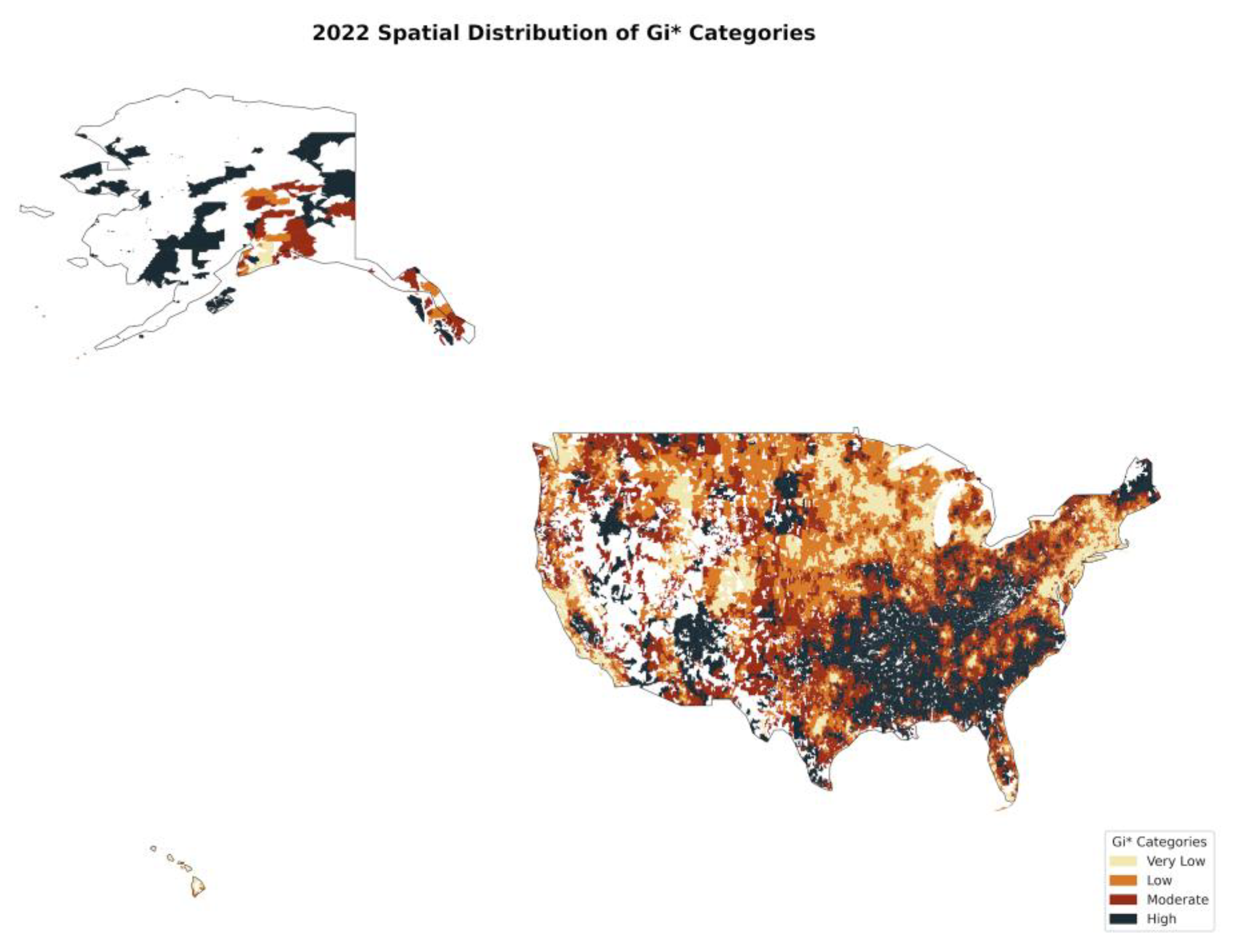
3.7. Hot Spot Analysis (Getis-Ord Gi)*
3.8. Spatial Lag Model (SLM)
3.9. Spatial Error Model (SEM)
3.10. Geographically Weighted Regression (GWR)
4. Discussion
4.1. Overall Trends and Persistent Disparities
4.2. Socioeconomic and Demographic Factors: Elaborating on Mechanisms
4.3. Racial Disparities: A Contradiction and Exacerbation
4.4. Spatial Dependence and Heterogeneity: Localized Interventions
4.5. Hot Spot Analysis (Getis-Ord Gi): Localized Clusters of Tooth Loss
4.6. Limitations
4.7. Future Research
4.8. Policy Implications (Prioritized)
- Targeted Interventions in Hotspots: The findings from the GWR and hotspot analysis highlight the value of prioritizing resources for ZCTAs that exhibit consistent high levels of dental attrition and significant correlations with socioeconomic factors such as poverty, inadequate educational achievement, and insufficient insurance coverage. This observation is consistent with the advocacy for spatially focused interventions as posited by several researchers [70,74,75].
- Expand Access to Affordable Care: The expansion of Medicare oral health coverage [65,66,67] and the correction of 'dental coverage disparities' [76] are of critical significance, especially in the context of the unequal effects of inadequate insurance on marginalized populations, which is supported by empirical findings underscoring the importance of insurance coverage [40,41,42].
- Address Social Determinants: Policies that target poverty alleviation, housing insecurity, and food insecurity are anticipated to yield considerable indirect advantages for oral health, as evidenced by empirical studies concerning the social determinants of health [77].
- Community-Based Programs: Initiatives involving community health workers [78,79], mobile dental units [80], and culturally customized oral health education [81,82] have the potential to enhance accessibility and encourage preventive practices, especially within marginalized populations, thus building upon well-established research regarding effective community-based interventions.
5. Conclusions
Declaration of Generative AI and AI-assisted technologies
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ZCTA | ZIP Code Tabulation Area |
| GWR | Geographically Weighted Regression |
| SLM | Spatial lag Model |
| SEM | Spatial Error Model |
| ACS | American Community Survey |
| REGARDS | REasons for Geographic and Racial Differences in Stroke |
| OLS | Ordinary Least Squares |
| ANOVA | Analysis of Variance |
| KDE | Kernel Density Estimate |
Appendix A
Appendix A.1
| 2018 | 2020 | 2022 | |
| Mean | 15.87 | 15.25 | 14.44 |
| Standard Deviation | 6.09 | 5.64 | 6.41 |
| Min Value | 3.00 | 3.30 | 1.90 |
| 25% | 11.70 | 11.40 | 9.90 |
| Median value | 15.00 | 14.50 | 13.40 |
| 75% | 19.20 | 18.40 | 17.70 |
| Max Value | 59.90 | 53.90 | 56.80 |
Appendix A.2
| Mean | Median | Standard Deviation | |||||||
| 2018 | 2020 | 2022 | 2018 | 2020 | 2022 | 2018 | 2020 | 2022 | |
| Percent Uninsured (>=65 years) | 0.620 | 0.620 | 0.630 | 0.000 | 0.000 | 0.000 | 2.130 | 2.320 | 2.210 |
| High school graduate or higher (>=65 Years) | 61.40 | 62.24 | 62.77 | 62.80 | 63.70 | 64.30 | 15.18 | 15.97 | 15.97 |
| Bachelor's degree or higher (6>=5 Years) | 21.19 | 22.64 | 23.71 | 17.60 | 19.00 | 20.10 | 15.74 | 16.47 | 16.60 |
| Population Percentage (>=65 years) | 18.63 | 19.53 | 20.16 | 17.39 | 18.09 | 18.69 | 8.33 | 8.94 | 9.11 |
| Number of individuals (>=65 years) per housing unit | 0.470 | 0.490 | 0.500 | 0.450 | 0.470 | 0.480 | 0.170 | 0.180 | 0.190 |
| Monthly Housing Costs in ZCTA | 926 | 962 | 1088 | 792 | 820 | 926 | 472 | 504 | 568 |
| Median income in ZCTA | 59188 | 63673 | 73352 | 54079 | 58042 | 67140 | 24898 | 26988 | 30807 |
| Percent below poverty level (>=65 years) | 9.37 | 9.40 | 10.04 | 7.60 | 7.40 | 8.10 | 8.54 | 8.97 | 8.94 |
| Female Percentage (>=65 years) | 53.65 | 53.43 | 53.15 | 54.32 | 54.22 | 53.90 | 9.00 | 9.69 | 9.49 |
Appendix A.3
| Group1 | Group2 | Mean difference | p-adj | lower | upper | reject |
| Total tooth loss (>=65 years) in 2018 | Total tooth loss (>=65 years) in 2020 | -0.62 | 0.00 | -0.74 | -0.50 | TRUE |
| Total tooth loss (>=65 years) in 2018 | Total tooth loss (>=65 years) in 2022 | -1.43 | 0.00 | -1.55 | -1.31 | TRUE |
| Total tooth loss (>=65 years) in 2020 | Total tooth loss (>=65 years) in 2022 | -0.81 | 0.00 | -0.93 | -0.69 | TRUE |
Appendix A.4
| Metric | SLM (2018) | SEM (2018) | SLM (2020) | SEM (2020) | SLM (2022) | SEM (2022) | Trend & Interpretation |
| AIC | 150789 | 142360 | 143483 | 144176 | 153664 | 154568 | SLM had a better fit in 2020, but SEM was better in 2018 and 2022. |
| Pseudo R² | 0.61 | 0.61 | 0.79 | 0.59 | 0.71 | 0.55 | SLM explained more variance than SEM, particularly in 2020 and 2022. |
| Log Likelihood | -71200 | -71170 | -72050 | -72078 | -77220 | -77275 | SLM had a stronger likelihood in 2020 and 2022, indicating significant spillover effects. |
Appendix A.5
| Metric | Lambda (SEM 2018) | Rho (SLM 2018) | Lambda (SEM 2020) | Rho (SLM 2020) | Lambda (SEM 2022) | Rho (SLM 2022) | Trend & Interpretation |
| Spatial Dependence | 0.80 | 0.10 | 0.80 | 0.60 | 0.70 | 0.60 | In 2018, SEM (lambda) was stronger, but in 2020 & 2022, SLM (rho) was stronger. |
Appendix A.6
| Variable | SLM (2018) | SEM (2018) | SLM (2020) | SEM (2020) | SLM (2022) | SEM (2022) | Trend & Interpretation |
| Percent Uninsured (>=65 years) | 0.03 | 0.05 | 0.11 | 0.04 | 0.05 | 0.06 | Uninsured status had a larger spillover effect in 2020 (SLM), but by 2022, it was more localized (SEM). |
| High School Graduate (>=65 years) | -0.08 | -0.05 | -0.75 | -0.04 | -0.06 | -0.06 | Education had a stronger impact in SLM for 2020, suggesting regional clusters of educated areas influencing neighbors. |
| Bachelor’s Degree or Higher (>=65 years) | -0.10 | -0.07 | -1.04 | -0.06 | -0.08 | -0.09 | SLM shows education-related spillover effects were highest in 2020. |
| Population (>=65 years) | -0.15 | -0.15 | -0.74 | -0.09 | -0.12 | -0.13 | Aging populations influenced neighboring regions more in 2020 (SLM stronger). |
| Individuals per Housing Unit (>=65 years) | 5.38 | 5.41 | 0.50 | 2.85 | 4.10 | 4.13 | Housing crowding effects were strongest in 2018, declined in 2020, then partially rebounded in 2022. |
| Median Income in ZCTA | 0.00 | 0.00 | -1.38 | 0.00 | -2.61 | 0.00 | In 2020 and 2022, income had stronger spillover effects (SLM). |
| Percent Below Poverty (>=65 years) | 0.07 | 0.06 | 0.43 | 0.04 | 0.07 | 0.06 | Poverty had stronger spillover effects in 2020, but returned to local effects in 2022. |
References
- Pedro, REL; Bugone, É; Dogenski, LC; Cardoso, MZ; Da Silva, AH; Linden, MSS; et al. Relationship between dentition, anthropometric measurements, and metabolic syndrome in the elderly. Revista De Odontologia Da UNESP 2019, 48. [Google Scholar] [CrossRef]
- Gerritsen, AE; Allen, PF; Witter, DJ; Bronkhorst, EM; Creugers, NH. Tooth loss and oral health-related quality of life: a systematic review and meta-analysis. Health and Quality of Life Outcomes 2010, 8, 126. [Google Scholar] [CrossRef] [PubMed]
- Vettore, MV; Vieira, JMR; Gomes, JFF; Martins, NMO; Freitas, YNL; De a Lamarca, G; et al. Individual- and City-Level Socioeconomic Factors and Tooth Loss among Elderly People: A Cross-Level Multilevel Analysis. International Journal of Environmental Research and Public Health 2020, 17, 2345. [Google Scholar] [CrossRef] [PubMed]
- Da Veiga Pessoa, DM; Roncalli, AG; De Lima, KC. Economic and sociodemographic inequalities in complete denture need among older Brazilian adults: a cross-sectional population-based study. BMC Oral Health 2016, 17. [Google Scholar] [CrossRef]
- Lyu, Y; Chen, S; Li, A; Zhang, T; Zeng, X; Sooranna, SR. Socioeconomic status and tooth loss impact on Oral Health–Related Quality of life in Chinese elderly. International Dental Journal 2023, 74, 268–75. [Google Scholar] [CrossRef]
- Luo, J; Wu, B; Zhao, Q; Guo, Q; Meng, H; Yu, L; et al. Association between Tooth Loss and Cognitive Function among 3063 Chinese Older Adults: A Community-Based Study. PLoS ONE 2015, 10, e0120986. [Google Scholar] [CrossRef]
- Kim, JH; Oh, JK; Wee, JH; Kim, YH; Byun, S-H; Choi, HG. Association between Tooth Loss and Alzheimer’s Disease in a Nested Case–Control Study Based on a National Health Screening Cohort. Journal of Clinical Medicine 2021, 10, 3763. [Google Scholar] [CrossRef]
- Oziegbe, EO; Schepartz, LA. Is parity a cause of tooth loss? Perceptions of northern Nigerian Hausa women. PLoS ONE 2019, 14, e0226158. [Google Scholar] [CrossRef]
- De Aguiar, AD; De Oliveira, ERA; De Barros Miotto, MHM. Tooth loss, sociodemographic conditions and Oral Health-Related Quality of life in the elderly. Pesquisa Brasileira Em Odontopediatria E Clínica Integrada 2022, 22. [Google Scholar] [CrossRef]
- Friedman, PK; Lamster, IB. Tooth loss as a predictor of shortened longevity: exploring the hypothesis. Periodontology 2000 2016, 72, 142–52. [Google Scholar] [CrossRef]
- Jiang, Y; Okoro, CA; Oh, J; Fuller, DL. Sociodemographic and Health-Related Risk Factors Associated with Tooth Loss Among Adults in Rhode Island. Preventing Chronic Disease 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Ando, A; Ohsawa, M; Yaegashi, Y; Sakata, K; Tanno, K; Onoda, T; et al. Factors related to tooth loss among Community-Dwelling middle-aged and elderly Japanese men. Journal of Epidemiology 2013, 23, 301–6. [Google Scholar] [CrossRef] [PubMed]
- Bomfim, RA; Schneider, IJC; De Andrade, FB; Lima-Costa, MF; Corrêa, VP; Frazão, P; et al. Racial inequities in tooth loss among older Brazilian adults: A decomposition analysis. Community Dentistry and Oral Epidemiology 2020, 49, 119–27. [Google Scholar] [CrossRef] [PubMed]
- Barbato, PR; Peres, KG. Contextual socioeconomic determinants of tooth loss in adults and elderly: a systematic review. Revista Brasileira De Epidemiologia 2015, 18, 357–71. [Google Scholar] [CrossRef]
- Lee, H; Kim, D; Jung, A; Chae, W. Ethnicity, Social, and Clinical Risk Factors to Tooth Loss among Older Adults in the U.S., NHANES 2011–2018. International Journal of Environmental Research and Public Health 2022, 19, 2382. [Google Scholar] [CrossRef]
- Singhal, A; Jackson, JW. Perceived racial discrimination partially mediates racial-ethnic disparities in dental utilization and oral health. Journal of Public Health Dentistry 2022, 82, 63–72. [Google Scholar] [CrossRef]
- Gabiec, K; Bagińska, J; Łaguna, W; Rodakowska, E; Kamińska, I; Stachurska, Z; et al. Factors Associated with Tooth Loss in General Population of Bialystok, Poland. International Journal of Environmental Research and Public Health 2022, 19, 2369. [Google Scholar] [CrossRef]
- Machida, D. Trends of health and dietary disparities by economic status among elderly individuals in Japan from 2004 to 2014: A repeated cross-sectional survey. In medRxiv (Cold Spring Harbor Laboratory); 2023. [Google Scholar] [CrossRef]
- Wong, FMF; Ng, YTY; Leung, WK. Oral Health and Its Associated Factors Among Older Institutionalized Residents—A Systematic Review. International Journal of Environmental Research and Public Health 2019, 16, 4132. [Google Scholar] [CrossRef]
- Musau, MM; Mwakio, S; Amadi, D; Nyaguara, A; Bejon, P; Berkley, JA; et al. Spatial heterogeneity of low-birthweight deliveries on the Kenyan coast. BMC Pregnancy and Childbirth 2023, 23. [Google Scholar] [CrossRef]
- Veginadu, P; Gussy, M; Calache, H; Masood, M. Disparities in spatial accessibility to public dental services relative to estimated need for oral health care among refugee populations in Victoria. Community Dentistry and Oral Epidemiology 2022, 51, 565–74. [Google Scholar] [CrossRef]
- Broomhead, T; Ballas, D; Baker, SR. Application of geographic information systems and simulation modelling to dental public health: Where next? Community Dentistry and Oral Epidemiology 2018, 47, 1–11. [Google Scholar] [CrossRef]
- Fadli M; Am R; Muhamad H; Prof A. SPATIAL ACCESSIBILITY OF PRIMARY HEALTHCARE IN RURAL POPULATION: a REVIEW. International Journal of Public Health and Clinical Sciences 2018, 5. [Google Scholar] [CrossRef]
- Yun, S; Ogawa, N; Izutsu, M; Yuki, M. The association between social isolation and oral health of community-dwelling older adults—A systematic review. Japan Journal of Nursing Science 2023, 20. [Google Scholar] [CrossRef] [PubMed]
- Letarte, L; Pomerleau, S; Tchernof, A; Biertho, L; Waygood, EOD; Lebel, A. Neighbourhood effects on obesity: scoping review of time-varying outcomes and exposures in longitudinal designs. BMJ Open 2020, 10, e034690. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, L; Hirsh, AT; Maupomé, G; Stewart, JC. Patient–Provider Language Concordance and Health Outcomes: A Systematic review, evidence map, and research agenda. Medical Care Research and Review 2019, 78, 3–23. [Google Scholar] [CrossRef]
- Aldosari, M; Archer, HR; Almutairi, FT; Alzuhair, SH; Aldosari, MA; Kennedy, E. Utilization of dental care and dentate status in diabetic and nondiabetic patients across US states: An analysis using the 2020 Behavioral Risk Factor Surveillance System. Journal of Public Health Dentistry 2024, 84, 187–97. [Google Scholar] [CrossRef]
- Ferreira, RC; Senna, MIB; Rodrigues, LG; Campos, FL; Martins, AEBL; Kawachi, I. Education and income-based inequality in tooth loss among Brazilian adults: does the place you live make a difference? BMC Oral Health 2020, 20. [Google Scholar] [CrossRef]
- Sopianah, Y; Murdiastuti, K; Amalia, R; Taftazani, RZ; Lestari, AR. Factors of dental caries, tooth mobility, and periodontal pockets on the occupation of tooth loss in the elderly. (A study in Karikil Village, Mangkubumi District, Tasikmalaya City). Open Access Macedonian Journal of Medical Sciences 2022, 10, 251–4. [Google Scholar] [CrossRef]
- Colussi, PRG; Hugo, FN; Muniz, FWMG; Rösing, CK. Tooth loss and associated factors in adolescents – impact of extractions for orthodontic reason. Brazilian Journal of Oral Sciences/Brazilian Journal of Oral Sciences 2018, 17, 1–12. [Google Scholar] [CrossRef]
- Zhang, Y; Leveille, SG; Shi, L. Multiple chronic diseases associated with tooth loss among the US adult population. Frontiers in Big Data 2022, 5. [Google Scholar] [CrossRef]
- Oue, H; Hatakeyama, R; Ishida, E; Yokoi, M; Tsuga, K. Experimental tooth loss affects spatial learning function and blood–brain barrier of mice. Oral Diseases 2022, 29, 2907–16. [Google Scholar] [CrossRef]
- Chen, Y; Li, Y; Guo, L; Hong, J; Zhao, W; Hu, X; et al. Bibliometric analysis of the inflammasome and pyroptosis in brain. Frontiers in Pharmacology 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Fushida, S; Kosaka, T; Kida, M; Kokubo, Y; Watanabe, M; Higashiyama, A; et al. Decrease in posterior occlusal support area can accelerate tooth loss: The Suita study. Journal of Prosthodontic Research 2020, 65, 321–6. [Google Scholar] [CrossRef] [PubMed]
- Guarnizo-Herreño, CC; Wehby, GL. Explaining Racial/Ethnic Disparities in Children’s Dental Health: A Decomposition analysis. American Journal of Public Health 2012, 102, 859–66. [Google Scholar] [CrossRef] [PubMed]
- Bernabé, E; Suominen, AL; Nordblad, A; Vehkalahti, MM; Hausen, H; Knuuttila, M; et al. Education level and oral health in Finnish adults: evidence from different lifecourse models. Journal of Clinical Periodontology 2010, 38, 25–32. [Google Scholar] [CrossRef]
- Mohammadi, TM; Malekmohammadi, M; Hajizamani, HR; Mahani, SA. Oral health literacy and its determinants among adults in Southeast Iran. European Journal of Dentistry 2018, 12, 439–42. [Google Scholar] [CrossRef]
- Guo, Y; Logan, HL; Dodd, VJ; Muller, KE; Marks, JG; Riley, JL. Health Literacy: A pathway to better Oral health. American Journal of Public Health 2014, 104, e85–91. [Google Scholar] [CrossRef]
- Shelley, D; Russell, S; Parikh, NS; Fahs, M. Ethnic Disparities in Self-Reported Oral Health Status and Access to Care among Older Adults in NYC. Journal of Urban Health 2011, 88, 651–62. [Google Scholar] [CrossRef]
- Naavaal, S; Griffin, SO; Jones, JA. Impact of making dental care affordable on quality of life in adults aged 45 years and older. Journal of Aging and Health 2019, 32, 861–70. [Google Scholar] [CrossRef]
- Thompson, B; Cooney, P; Lawrence, H; Ravaghi, V; Quiñonez, C. The potential oral health impact of cost barriers to dental care: findings from a Canadian population-based study. BMC Oral Health 2014, 14. [Google Scholar] [CrossRef]
- Dye, BA; Weatherspoon, DJ; Mitnik, GL. Tooth loss among older adults according to poverty status in the United States from 1999 through 2004 and 2009 through 2014. The Journal of the American Dental Association 2018, 150, 9–23.e3. [Google Scholar] [CrossRef] [PubMed]
- Williams, DR; Lawrence, JA; Davis, BA; Vu, C. Understanding how discrimination can affect health. Health Services Research 2019, 54, 1374–88. [Google Scholar] [CrossRef] [PubMed]
- Phelan, JC; Link, BG. Is racism a fundamental cause of inequalities in health? Annual Review of Sociology 2015, 41, 311–30. [Google Scholar] [CrossRef]
- Borrell, LN; Reynolds, JC; Fleming, E; Shah, PD. Access to dental insurance and oral health inequities in the United States. Community Dentistry and Oral Epidemiology 2023, 51, 615–20. [Google Scholar] [CrossRef]
- Stennett, M; Tsakos, G. The impact of the COVID-19 pandemic on oral health inequalities and access to oral healthcare in England. BDJ 2022, 232, 109–14. [Google Scholar] [CrossRef]
- Milner, A; Franz, B; Braddock, JH. We need to talk about Racism—In all of its Forms—To understand COVID-19 disparities. Health Equity 2020, 4, 397–402. [Google Scholar] [CrossRef]
- Kranz, AM; Gahlon, G; Dick, AW; Stein, BD. Characteristics of US adults delaying dental care due to the COVID-19 pandemic. JDR Clinical & Translational Research 2020, 6, 8–14. [Google Scholar] [CrossRef]
- Dickson-Swift, V; Kangutkar, T; Knevel, R; Down, S. The impact of COVID-19 on individual oral health: a scoping review. BMC Oral Health 2022, 22. [Google Scholar] [CrossRef]
- Jiang, CM; Duangthip, D; Auychai, P; Chiba, M; Folayan, MO; Hamama, HHH; et al. Changes in oral health policies and guidelines during the COVID-19 pandemic. Frontiers in Oral Health 2021, 2. [Google Scholar] [CrossRef]
- Oberoi, SS; Grover, S; Sachdeva, S. Oral health disparities in the time of COVID-19: An indication for focus on public health care strengthening and preventive care. Journal of Dental Specialities 2021, 9, 42–3. [Google Scholar] [CrossRef]
- Yuan, S; Zheng, Y; Sun, Z; Humphris, G. Does fear of infection affect people’s dental attendance during COVID-19? A Chinese example to examine the association between COVID anxiety and dental anxiety. Frontiers in Oral Health 2023, 4. [Google Scholar] [CrossRef]
- Goldstein, A; Matalon, S; Fridenberg, N; Slutzky, H. Seeking dental health care in the context of COVID-19 pandemic: A study examining the Health Belief Model. Innovation in Aging 2024, 8. [Google Scholar] [CrossRef]
- Jiang, Y; Zilioli, S; Balzarini, RN; Zoppolat, G; Slatcher, RB. Education, financial stress, and trajectory of mental health during the COVID-19 pandemic. Clinical Psychological Science 2021, 10, 662–74. [Google Scholar] [CrossRef]
- Wani, F; Rather, R; Ahmad, M. Self-reported unmet healthcare needs during coronavirus disease-19 pandemic lockdown. International Journal of Medical Science and Public Health 2020, 1. [Google Scholar] [CrossRef]
- Mularczyk-Tomczewska, P; Zarnowski, A; Gujski, M; Jankowski, M; Bojar, I; Wdowiak, A; et al. Barriers to accessing health services during the COVID-19 pandemic in Poland: A nationwide cross-sectional survey among 109,928 adults in Poland. Frontiers in Public Health 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Burgette, JM; Weyant, RJ; Ettinger, AK; Miller, E; Ray, KN. What is the association between income loss during the COVID-19 pandemic and children’s dental care? The Journal of the American Dental Association 2021, 152, 369–76. [Google Scholar] [CrossRef] [PubMed]
- Schnell, P; Bandyopadhyay, D; Reich, BJ; Nunn, M. A marginal cure rate Proportional hazards model for spatial survival data. Journal of the Royal Statistical Society Series C (Applied Statistics) 2015, 64, 673–91. [Google Scholar] [CrossRef]
- Feng, X; Sambamoorthi, U; Wiener, RC. Dental workforce availability and dental services utilization in Appalachia: a geospatial analysis. Community Dentistry and Oral Epidemiology 2016, 45, 145–52. [Google Scholar] [CrossRef]
- Jia, W; Liu, L; Wang, Z; Peng, G. Analysis of the impact of public services on residents’ health: A spatial econometric analysis of Chinese provinces. International Journal of Public Health 2023, 68. [Google Scholar] [CrossRef]
- Cheng, J; Cui, Y; Wang, X; Wang, Y; Feng, R. Spatial characteristics of health outcomes and geographical detection of its influencing factors in Beijing. Frontiers in Public Health 2024, 12. [Google Scholar] [CrossRef]
- Hl, Lima; Em, Costa; Ld, Andrade; Eb, Thomaz. Spatial-temporal analysis of hospitalizations with death caused by oral cancer in Brazil and its correlation with the expansion of healthcare coverage. Medicina Oral, Patología Oral Y Cirugía Bucal 2022, e1–8. [Google Scholar] [CrossRef]
- Tsai, P-J; Perng, C-H. Spatial autocorrelation analysis of 13 leading malignant neoplasms in Taiwan: a comparison between the 1995-1998 and 2005-2008 periods. Health 2011, 03, 712–31. [Google Scholar] [CrossRef]
- Zeng, M; Niu, L. Exploring spatiotemporal trends and impacts of health resources and services on under-5 mortality in West African countries, 2010–2019: a spatial data analysis. Frontiers in Public Health 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Wolownik, G; Cohen, SS. Dental coverage for Medicare beneficiaries. Policy Politics & Nursing Practice 2024, 25, 205–15. [Google Scholar] [CrossRef]
- Simon, L; Song, Z; Barnett, ML. The new Medicare Dental Benefit—Small but Mighty. JAMA Internal Medicine 2024. [Google Scholar] [CrossRef]
- Elani, HW; Sommers, BD; Yuan, D; Kawachi, I; Rosenthal, MB; Tipirneni, R. Dental coverage and care when transitioning from Medicaid to Medicare. JAMA Health Forum 2024, 5, e244165. [Google Scholar] [CrossRef]
- Hipp, JA; Chalise, N. Spatial Analysis and correlates of County-Level Diabetes Prevalence, 2009–2010. In Preventing Chronic Disease; 2015; p. 12. [Google Scholar] [CrossRef]
- Davis, J; Liu, M; Kao, D; Gu, X; Cherry-Peppers, G. Using GIS to analyze inequality in access to dental care in the District of Columbia. The AMA Journal of Ethic 2022, 24, E41-47. [Google Scholar] [CrossRef]
- Liu, M; Kao, D; Gu, X; Holland, W; Cherry-Peppers, G. Oral health service access in Racial/Ethnic Minority Neighborhoods: A geospatial analysis in Washington, DC, USA. International Journal of Environmental Research and Public Health 2022, 19, 4988. [Google Scholar] [CrossRef]
- Huang, DL; Park, M. Socioeconomic and racial/ethnic oral health disparities among US older adults: oral health quality of life and dentition. Journal of Public Health Dentistry 2014, 75, 85–92. [Google Scholar] [CrossRef]
- Fisher-Owens, SA; Isong, IA; Soobader, M; Gansky, SA; Weintraub, JA; Platt, LJ; et al. An examination of racial/ethnic disparities in children’s oral health in the United States. Journal of Public Health Dentistry 2012, 73, 166–74. [Google Scholar] [CrossRef]
- Flores, G; Lin, H. Trends in racial/ethnic disparities in medical and oral health, access to care, and use of services in US children: has anything changed over the years? International Journal for Equity in Health 2013, 12, 10. [Google Scholar] [CrossRef]
- Pereira, SM; Ambrosano, GMB; Cortellazzi, KL; Tagliaferro, EPS; Vettorazzi, CA; Ferraz, SFB; et al. Geographic Information Systems (GIS) in assessing dental health. International Journal of Environmental Research and Public Health 2010, 7, 2423–36. [Google Scholar] [CrossRef]
- Razdan, M; Degenholtz, H; Rubin, R. Oral Health Outreach Programs - Can they Address the Disparities in Access to Dental Care? Journal of Oral Health and Community Dentistry 2016, 10, 14–9. [Google Scholar] [CrossRef]
- Roberts, ET; Mellor, JM; McInerny, MP; Sabik, LM. Effects of a Medicaid dental coverage “cliff” on dental care access among low-income Medicare beneficiaries. Health Services Research 2022, 58, 589–98. [Google Scholar] [CrossRef]
- Moeller, J; Starkel, R; Quiñonez, C; Vujicic, M. Income inequality in the United States and its potential effect on oral health. The Journal of the American Dental Association 2017, 148, 361–8. [Google Scholar] [CrossRef] [PubMed]
- Northridge, ME; Wu, Y; Troxel, AB; Min, D; Liu, R; Liang, LJ; et al. Acceptability of a community health worker intervention to improve the oral health of older Chinese Americans: A pilot study. Gerodontology 2020, 38, 117–22. [Google Scholar] [CrossRef] [PubMed]
- Northridge, ME; Estrada, I; Schrimshaw, EW; Greenblatt, AP; Metcalf, SS; Kunzel, C. 34. Racial/Ethnic Minority Older Adults’ Perspectives on Proposed Medicaid Reforms’ Effects on Dental Care Access; American Public Health Association eBooks, 2019. [Google Scholar] [CrossRef]
- Weatherspoon, DJ; McQuistan, MR; Taylor, GW; Mays, KA. Editorial: Helping meet oral health needs in underserved communities. Frontiers in Oral Health 2023, 4. [Google Scholar] [CrossRef] [PubMed]
- Ki, J-Y; Jo, S-R; Cho, K-S; Park, J-E; Cho, J-W; Jang, J-H. Effect of Oral Health Education using a mobile app (OHEMA) on the Oral Health and Swallowing-Related Quality of Life in Community-Based Integrated Care of the Elderly: a randomized clinical trial. International Journal of Environmental Research and Public Health 2021, 18, 11679. [Google Scholar] [CrossRef]
- Khanagar, S; Kumar, A; Rajanna, V; Badiyani, B; Jathanna, V; Kini, P. Oral health care education and its effect on caregivers′ knowledge, attitudes, and practices: A randomized controlled trial. Journal of International Society of Preventive and Community Dentistry 2014, 4, 122. [Google Scholar] [CrossRef]
- Zhu, Y; Zhou, X; Wu, J; Su, J; Zhang, G. Risk factors and prevalence ofHelicobacter pyloriInfection in persistent high incidence area of gastric carcinoma in Yangzhong City. Gastroenterology Research and Practice 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Goh, K; Chan, W; Shiota, S; Yamaoka, Y. Epidemiology of Helicobacter pylori Infection and Public Health Implications. Helicobacter 2011, 16, 1–9. [Google Scholar] [CrossRef]
- Bashir, SK; Khan, MB. Overview of Helicobacter pylori Infection, Prevalence, Risk Factors, and Its Prevention. Advanced Gut & Microbiome Research 2023, 2023, 1–9. [Google Scholar] [CrossRef]
- Schmidt, TS; Hayward, MR; Coelho, LP; Li, SS; Costea, PI; Voigt, AY; et al. Extensive transmission of microbes along the gastrointestinal tract. eLife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Liu, J; Spencer, N; Utter, DR; Grossman, AS; Lei, L; Santos, NCD; et al. Persistent enrichment of multidrug-resistant Klebsiella in oral and nasal communities during long-term starvation. Microbiome 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Elani, HW; Harper, S; Thomson, WM; Espinoza, IL; Mejia, GC; Ju, X; et al. Social inequalities in tooth loss: A multinational comparison. Community Dentistry and Oral Epidemiology 2017, 45, 266–74. [Google Scholar] [CrossRef]
- Bernabé, E; Marcenes, W. Income inequality and tooth loss in the United States. Journal of Dental Research 2011, 90, 724–9. [Google Scholar] [CrossRef]
- You, Z; Cushman, M; Jenny, NS; Howard, G. Tooth loss, systemic inflammation, and prevalent stroke among participants in the reasons for geographic and racial difference in stroke (REGARDS) study. Atherosclerosis 2008, 203, 615–9. [Google Scholar] [CrossRef]
- Di Spirito, F; Amato, A; Di Palo, MP; Ferraro, GA; Baroni, A; Serpico, R; et al. COVID-19 Related Information on Pediatric Dental Care including the Use of Teledentistry: A Narrative Review. Children 2022, 9, 1942. [Google Scholar] [CrossRef]
- Bambra, C; Riordan, R; Ford, J; Matthews, F. The COVID-19 pandemic and health inequalities. Journal of Epidemiology & Community Health 2020, 74, 964–8. [Google Scholar] [CrossRef]
- Wibowo, AA. Analisis Autokorelasi Spasial Lisa menggunakan Sistem Informasi Geografis terhadap Angka Tumpatan Gigi di Kabupaten Ciamis. LaGeografia 2022, 21, 105. [Google Scholar] [CrossRef]
- Khan, G; Qin, X; Noyce, DA. Spatial analysis of weather crash patterns. Journal of Transportation Engineering 2008, 134, 191–202. [Google Scholar] [CrossRef]
- De Assis, IS; Berra, TZ; Alves, LS; Ramos, ACV; Arroyo, LH; Santos, DTD; et al. Leprosy in urban space, areas of risk for disability and worsening of this health condition in Foz Do Iguaçu, the border region between Brazil, Paraguay and Argentina. BMC Public Health 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Liang, X; Baker, J; DellaPosta, D; Andris, C. Is your neighbor your friend? Scan methods for spatial social network hotspot detection. Transactions in GIS 2023, 27, 607–25. [Google Scholar] [CrossRef]
- De Lucena, EHG; Freire, AR; Freire, DEWG; De Araújo, ECF; Lira, GNW; Brito, ACM; et al. Offer and use of oral health in primary care before and after the beginning of the COVID-19 pandemic in Brazil. Pesquisa Brasileira Em Odontopediatria E Clínica Integrada 2020, 20. [Google Scholar] [CrossRef]
- Çelik, Z; Ata, GD. The impact of the COVID-19 pandemic and vaccination on dental restorative practices in the geriatric population. Experimed 2022, 12, 61–5. [Google Scholar] [CrossRef]
- Wu, B; Liang, J; Plassman, BL; Remle, RC; Bai, L. Oral health among white, black, and Mexican-American elders: an examination of edentulism and dental caries. Journal of Public Health Dentistry 2011, 71, 308–17. [Google Scholar] [CrossRef]
- Hybels, CF; Wu, B; Landerman, LR; Liang, J; Bennett, JM; Plassman, BL. Trends in decayed teeth among middle-aged and older adults in the United States: socioeconomic disparities persist over time. Journal of Public Health Dentistry 2016, 76, 287–94. [Google Scholar] [CrossRef]
- Tiwari, T; Scarbro, S; Bryant, LL; Puma, J. Factors Associated with Tooth Loss in Older Adults in Rural Colorado. Journal of Community Health 2015, 41, 476–81. [Google Scholar] [CrossRef]
- Rahman, MdS; Blossom, JC; Kawachi, I; Tipirneni, R; Elani, HW. Dental clinic deserts in the US: Spatial Accessibility analysis. JAMA Network Open 2024, 7, e2451625. [Google Scholar] [CrossRef]
- Estai, M; Kanagasingam, Y; Tennant, M; Bunt, S. A systematic review of the research evidence for the benefits of teledentistry. Journal of Telemedicine and Telecare 2017, 24, 147–56. [Google Scholar] [CrossRef]
- Tulimiero, M; Garcia, M; Rodriguez, M; Cheney, AM. Overcoming barriers to health care access in rural Latino communities: an innovative model in the eastern Coachella Valley. The Journal of Rural Health 2020, 37, 635–44. [Google Scholar] [CrossRef]
- Evans, C. The principles, competencies, and curriculum for educating dental therapists: a report of the American Association of Public Health Dentistry Panel. Journal of Public Health Dentistry 2011, 71. [Google Scholar] [CrossRef]
| Model Fit Statistics | Value | |||||
| Dependent variable | Total tooth loss (≥65 years, %) | |||||
| Number of observations | 89380 | |||||
| R2 | 0.609 | |||||
| Adjusted R2 | 0.609 | |||||
| F-statistic | 9943 | |||||
| Prob (F-statistic) | <0.001 | |||||
| Log-likelihood | -246200 | |||||
| AIC | 492437 | |||||
| BIC | 492578 | |||||
| Durbin–Watson statistic | 1.414 | |||||
| Breusch–Godfrey LM statistic | 7841 | |||||
| Breusch–Godfrey p-value | <0.001 | |||||
| Variable | Coef | Std Err | t | P>|t| | 95% CI | VIF |
| Contstant | 28.545 | 0.145 | 196.304 | <0.001 | [28.26, 28.83] | 162.213 |
| Percent Uninsured (>=65 years) | 0.042 | 0.006 | 7.033 | <0.001 | [0.03, 0.054] | 1.079 |
| High school graduate or higher (>=65 Years) | -0.112 | 0.001 | -87.952 | <0.001 | [-0.114, -0.109] | 2.475 |
| Bachelor's degree or higher (6>=5 Years) | -0.131 | 0.001 | -93.914 | <0.001 | [-0.134, -0.128] | 3.196 |
| Population Percentage (>=65 years) | -0.149 | 0.003 | -44.157 | <0.001 | [-0.155, -0.142] | 5.435 |
| Number of individuals (>=65 years) per housing unit | 5.522 | 0.161 | 34.383 | <0.001 | [5.207, 5.837] | 5.141 |
| Monthly Housing Costs in ZCTA | -0.0030 | 0.000 | -62.706 | <0.001 | [-0.003, -0.003] | 3.888 |
| Median income in ZCTA | -0.0000656 | 0.000 | -73.018 | <0.001 | [-0.0000674, -0.0000638] | 3.989 |
| Percent below poverty level (>=65 years) | 0.072 | 0.002 | 43.841 | <0.001 | [0.069, 0.076] | 1.307 |
| Female Percentage (>=65 years) | 0.00140 | 0.001 | 0.990 | 0.322 | [-0.001, 0.004] | 1.036 |
| Year_Dummy_2020 (referenced 2018) | 0.093 | 0.031 | 2.980 | 0.003 | [0.032, 0.155] | 1.345 |
| Year_Dummy_2022 (referenced 2018) | 0.452 | 0.033 | 13.880 | <0.001 | [0.388, 0.515] | 1.447 |
| White_Majority Zipcode (>=65 years) | 2.882 | 0.089 | 32.349 | <0.001 | [2.707, 3.056] | 4.474 |
| Black_Majority Zipcode (>=65 years) | 7.481 | 0.105 | 70.929 | <0.001 | [7.274, 7.687] | 3.020 |
| Other_Majority Zipcode (>=65 years) | 7.302 | 0.124 | 58.764 | <0.001 | [7.058, 7.545] | 2.386 |
| Model Fit Statistics | Value | ||||||
| Dependent variable | Total tooth loss (≥65 years, %) | ||||||
| Number of observations | 89380 | ||||||
| R2 | 0.596 | ||||||
| Adjusted R2 | 0.596 | ||||||
| F-statistic | 7769 | ||||||
| Prob (F-statistic) | <0.001 | ||||||
| Log-likelihood | -247910 | ||||||
| AIC | 495268 | ||||||
| BIC | 495437 | ||||||
| Durbin–Watson statistic | 1.402 | ||||||
| Breusch–Godfrey LM statistic | 8128 | ||||||
| Breusch–Godfrey p-value | <0.001 | ||||||
| Variable | Coef | Std Err | t | P>|t| | 95% CI | VIF | |
| Constant | 31.62 | 0.14 | 229.90 | <0.001 | [31.35, 31.89] | 113.31 | |
| Percent Uninsured (>=65 years) | 0.04 | 0.01 | 6.98 | <0.001 | [0.03, 0.05] | 1.07 | |
| High school graduate or higher (>=65 Years) | -0.11 | 0.00 | -89.60 | <0.001 | [-0.12, -0.11] | 2.36 | |
| Bachelor's degree or higher (6>=5 Years) | -0.13 | 0.00 | -94.14 | <0.001 | [-0.13, -0.13] | 3.10 | |
| Population Percentage (>=65 years) | -0.15 | 0.00 | -44.75 | <0.001 | [-0.16, -0.15] | 5.41 | |
| Number of individuals (>=65 years) per housing unit | 5.60 | 0.16 | 34.35 | <0.001 | [5.28, 5.92] | 5.12 | |
| Monthly Housing Costs in ZCTA | -0.003 | 0.00 | -60.81 | <0.001 | [-0.003, -0.004] | 3.86 | |
| Median income in ZCTA | -0.0000673 | 0.00 | -74.02 | <0.001 | [-0.0000691, -0.0000656] | 3.96 | |
| Percent below poverty level (>=65 years) | 0.08 | 0.00 | 46.30 | <0.001 | [0.07, 0.08] | 1.30 | |
| Female Percentage (>=65 years) | 0.0033 | 0.00 | 2.38 | 0.020 | [0.001, 0.006] | 1.03 | |
| Year_Dummy_2020 (referenced 2018) | -3.08 | 0.15 | -21.16 | <0.001 | [-3.37, -2.8] | 28.25 | |
| Year_Dummy_2022 (referenced 2018) | -2.26 | 0.14 | -15.95 | <0.001 | [-2.54, -1.98] | 26.54 | |
| Interactive_year_2020_with_White_Majority (referenced to 2018, Hispanic) | 3.06 | 0.15 | 20.83 | <0.001 | [2.77, 3.35] | 27.41 | |
| Interactive_year_2020_with_Black_Majority (referenced to 2018, Hispanic) | 7.11 | 0.18 | 38.89 | <0.001 | [6.75, 7.47] | 2.50 | |
| Interactive_year_2020_with_Other_Majority (referenced to 2018, Hispanic) | 6.10 | 0.22 | 28.20 | <0.001 | [5.67, 6.52] | 1.74 | |
| Interactive_year_2022_with_White_Majority (referenced to 2018, Hispanic) | 2.51 | 0.14 | 17.60 | <0.001 | [2.23, 2.79] | 25.75 | |
| Interactive_year_2022_with_Black_Majority (referenced to 2018, Hispanic) | 7.53 | 0.180 | 41.92 | <0.001 | [7.18, 7.89] | 2.41 | |
| Interactive_year_2022_with_Other_Majority (referenced to 2018, Hispanic) | 7.45 | 0.21 | 34.99 | <0.001 | [7.03, 7.87] | 1.71 | |
| Year | Moran’s I | p-value | Z-score | Interpretation |
| 2018 | 0.71 | 0.00 | 205.27 | Strong spatial clustering |
| 2020 | 0.71 | 0.00 | 212.74 | Strong spatial clustering (slightly lower than 2018) |
| 2022 | 0.63 | 0.00 | 184.59 | Strong but weaker clustering than previous years |
| Model Fit Statistics | Value | |||
| AIC: | 150788.770 | |||
| Schwarz criterion | 150871.537 | |||
| Pseudo R-squared | 0.719 | |||
| Spatial Pseudo R-squared | 0.613 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 22.596 | 0.205 | 110.017 | <0.001 |
| Percent Uninsured (>=65 years) | 0.029 | 0.010 | 2.991 | 0.003 |
| High school graduate or higher (>=65 Years) | -0.083 | 0.002 | -43.711 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -0.097 | 0.002 | -45.893 | <0.001 |
| Percentage of Population (>=65 years) | -0.151 | 0.006 | -25.314 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 5.383 | 0.286 | 18.832 | <0.001 |
| Monthly Housing Costs in ZCTA | -0.001 | 0.000 | -12.215 | <0.001 |
| Median income in ZCTA | 0.000 | 0.000 | -52.622 | <0.001 |
| Percent below poverty level (>=65 years) | 0.071 | 0.003 | 25.968 | <0.001 |
| Coefficient for the spatially lag | 0.062 | 0.001 | 89.047 | <0.001 |
| Variable | Direct | Indirect | Total | |
| Percent Uninsured (>=65 years) | 0.029 | 0.002 | 0.031 | |
| High school graduate or higher (>=65 Years) | -0.083 | -0.006 | -0.089 | |
| Bachelor's degree or higher (>=65 Years) | -0.097 | -0.006 | -0.104 | |
| Percentage of Population (>=65 years) | -0.151 | -0.010 | -0.161 | |
| Number of individuals (>=65 years) per housing unit | 5.383 | 0.354 | 5.737 | |
| Monthly Housing Costs in ZCTA | -0.001 | 0.000 | -0.001 | |
| Median income in ZCTA | 0.000 | 0.000 | 0.000 | |
| Percent below poverty level (>=65 years) | 0.071 | 0.005 | 0.076 |
| Model Fit Statistics | Value | |||
| AIC: | 143482.792 | |||
| Schwarz criterion | 143565.765 | |||
| Pseudo R-squared | 0.787 | |||
| Spatial Pseudo R-squared | 0.636 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 5.415 | 0.067 | 80.647 | <0.001 |
| Percent Uninsured (>=65 years) | 0.114 | 0.016 | 7.323 | <0.001 |
| High school graduate or higher (>=65 Years) | -0.752 | 0.023 | -32.559 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -1.041 | 0.027 | -39.201 | <0.001 |
| Percentage of Population (>=65 years) | -0.742 | 0.036 | -20.776 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 0.496 | 0.035 | 14.342 | <0.001 |
| Monthly Housing Costs in ZCTA | -0.195 | 0.030 | -6.450 | <0.001 |
| Median income in ZCTA | -1.384 | 0.029 | -47.980 | <0.001 |
| Percent below poverty level (>=65 years) | 0.426 | 0.017 | 25.027 | <0.001 |
| Coefficient for the spatially lag | 0.645 | 0.004 | 150.326 | <0.001 |
| Variable | Direct | Indirect | Total | |
| Percent Uninsured (>=65 years) | 0.114 | 0.207 | 0.321 | |
| High school graduate or higher (>=65 Years) | -0.752 | -1.367 | -2.118 | |
| Bachelor's degree or higher (>=65 Years) | -1.041 | -1.893 | -2.935 | |
| Percentage of Population (>=65 years) | -0.742 | -1.350 | -2.092 | |
| Number of individuals (>=65 years) per housing unit | 0.497 | 0.903 | 1.399 | |
| Monthly Housing Costs in ZCTA | -0.195 | -0.355 | -0.550 | |
| Median income in ZCTA | -1.384 | -2.516 | -3.900 | |
| Percent below poverty level (>=65 years) | 0.426 | 0.774 | 1.200 |
| Model Fit Statistics | Value | |||
| AIC: | 153663.559 | |||
| Schwarz criterion | 153746.171 | |||
| Pseudo R-squared | 0.707 | |||
| Spatial Pseudo R-squared | 0.587 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 16.038 | 0.248 | 64.733 | <0.001 |
| Percent Uninsured (>=65 years) | 0.045 | 0.010 | 4.536 | <0.001 |
| High school graduate or higher (>=65 Years) | -0.062 | 0.002 | -28.541 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -0.079 | 0.002 | -33.381 | <0.001 |
| Percentage of Population (>=65 years) | -0.119 | 0.006 | -20.977 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 4.096 | 0.269 | 15.252 | <0.001 |
| Monthly Housing Costs in ZCTA | 0.000 | 0.000 | -2.259 | 0.024 |
| Median income in ZCTA | 0.000 | 0.000 | -44.291 | <0.001 |
| Percent below poverty level (>=65 years) | 0.071 | 0.003 | 25.845 | <0.001 |
| Coefficient for the spatially lag | 0.571 | 0.005 | 110.270 | <0.001 |
| Variable | Direct | Indirect | Total | |
| Percent Uninsured (>=65 years) | 0.045 | 0.060 | 0.105 | |
| High school graduate or higher (>=65 Years) | -0.062 | -0.082 | -0.144 | |
| Bachelor's degree or higher (>=65 Years) | -0.079 | -0.105 | -0.184 | |
| Percentage of Population (>=65 years) | -0.119 | -0.158 | -0.277 | |
| Number of individuals (>=65 years) per housing unit | 4.096 | 5.455 | 9.552 | |
| Monthly Housing Costs in ZCTA | 0.000 | 0.000 | 0.000 | |
| Median income in ZCTA | 0.000 | 0.000 | 0.000 | |
| Percent below poverty level (>=65 years) | 0.071 | 0.094 | 0.165 |
| Model Fit Statistics | Value | |||
| AIC: | 142360 | |||
| Schwarz criterion | 142434 | |||
| Mean dependent var | 16 | |||
| S.D. dependent var | 6 | |||
| Pseudo R-squared | 1 | |||
| Log likelihood | -71171 | |||
| Sigma-square ML | 7 | |||
| S.E of regression | 3 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 26.707 | 0.174 | 153.386 | <0.001 |
| Percent Uninsured (>=65 years) | 0.049 | 0.008 | 6.174 | <0.001 |
| High school graduate or higher (>=65 Years) | -0.053 | 0.002 | -30.723 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -0.073 | 0.002 | -36.992 | <0.001 |
| Percentage of Population (>=65 years) | -0.149 | 0.005 | -29.225 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 5.408 | 0.247 | 21.866 | <0.001 |
| Monthly Housing Costs in ZCTA | -0.002 | 0.000 | -20.553 | <0.001 |
| Median income in ZCTA | 0.000 | 0.000 | -50.086 | <0.001 |
| Percent below poverty level (>=65 years) | 0.058 | 0.002 | 25.983 | <0.001 |
| Lambda | 0.795 | 0.004 | 178.992 | <0.001 |
| Model Fit Statistics | Value | |||
| AIC: | 144176 | |||
| Schwarz criterion | 144251 | |||
| Mean dependent var | 15 | |||
| S.D. dependent var | 6 | |||
| Pseudo R-squared | 1 | |||
| Log likelihood | -72079 | |||
| Sigma-square ML | 7 | |||
| S.E of regression | 3 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 24.999 | 0.161 | 155.488 | <0.001 |
| Percent Uninsured (>=65 years) | 0.041 | 0.007 | 6.289 | <0.001 |
| High school graduate or higher (>=65 Years) | -0.041 | 0.002 | -26.779 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -0.063 | 0.002 | -35.792 | <0.001 |
| Percentage of Population (>=65 years) | -0.093 | 0.004 | -22.931 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 2.852 | 0.197 | 14.501 | <0.001 |
| Monthly Housing Costs in ZCTA | -0.003 | 0.000 | -30.549 | <0.001 |
| Median income in ZCTA | 0.000 | 0.000 | -43.063 | <0.001 |
| Percent below poverty level (>=65 years) | 0.040 | 0.002 | 21.512 | <0.001 |
| Lambda | 0.788 | 0.004 | 175.568 | <0.001 |
| Model Fit Statistics | Value | |||
| AIC: | 154568 | |||
| Schwarz criterion | 154642 | |||
| Mean dependent var | 14 | |||
| S.D. dependent var | 6 | |||
| Pseudo R-squared | 1 | |||
| Log likelihood | -77275 | |||
| Sigma-square ML | 12 | |||
| S.E of regression | 3 | |||
| Variable | Coefficient | Std Err | z-Statistic | Probability |
| Constant | 26.843 | 0.230 | 116.531 | <0.001 |
| Percent Uninsured (>=65 years) | 0.055 | 0.010 | 5.551 | <0.001 |
| High school graduate or higher (>=65 Years) | -0.059 | 0.002 | -24.919 | <0.001 |
| Bachelor's degree or higher (>=65 Years) | -0.085 | 0.003 | -32.351 | <0.001 |
| Percentage of Population (>=65 years) | -0.132 | 0.006 | -22.589 | <0.001 |
| Number of individuals (>=65 years) per housing unit | 4.134 | 0.279 | 14.844 | <0.001 |
| Monthly Housing Costs in ZCTA | -0.002 | 0.000 | -20.115 | <0.001 |
| Median income in ZCTA | 0.000 | 0.000 | -41.864 | <0.001 |
| Percent below poverty level (>=65 years) | 0.058 | 0.003 | 21.287 | <0.001 |
| Lambda | 0.683 | 0.006 | 118.410 | <0.001 |
| Model Fit Statistics | Global Regression | GWR | |||
| AIC: | 158960 | 132322 | |||
| AICc: | 158962 | 135334 | |||
| BIC: | 106130 | 181177 | |||
| R2: | 0.618 | 0.898 | |||
| Adjusted R2 | 0.618 | 0.872 | |||
| Variable | Mean | STD | Min | Median | Max |
| Intercept | 14.808 | 2.281 | 0.829 | 14.877 | 29.156 |
| Percent Uninsured (>=65 years) | 0.168 | 0.929 | -14.623 | 0.106 | 7.144 |
| High school graduate or higher (>=65 Years) | -0.991 | 1.096 | -6.024 | -0.832 | 3.758 |
| Bachelor's degree or higher (>=65 Years) | -1.328 | 1.247 | -6.487 | -1.191 | 3.990 |
| Percentage of Population (>=65 years) | -2.076 | 2.183 | -15.626 | -1.661 | 7.510 |
| Number of individuals (>=65 years) per housing unit | 1.546 | 1.870 | -7.237 | 1.247 | 10.639 |
| Monthly Housing Costs in ZCTA | -1.402 | 2.226 | -17.471 | -1.269 | 8.653 |
| Median income in ZCTA | -2.651 | 1.803 | -11.835 | -2.351 | 4.888 |
| Percent below poverty level (>=65 years) | 0.643 | 0.779 | -2.827 | 0.523 | 4.540 |
| Model Fit Statistics | Global Regression | GWR | |||
| AIC: | 159588 | 136157 | |||
| AICc: | 159590 | 139076 | |||
| BIC: | 72008 | 185014 | |||
| R2: | 0.598 | 0.877 | |||
| Adjusted R2 | 0.598 | 0.847 | |||
| Variable | Mean | STD | Min | Median | Max |
| Intercept | 14.526 | 2.169 | 2.727 | 14.478 | 26.317 |
| Percent Uninsured (>=65 years) | 0.255 | 1.142 | -19.818 | 0.134 | 17.001 |
| High school graduate or higher (>=65 Years) | -0.992 | 1.184 | -8.729 | -0.802 | 2.588 |
| Bachelor's degree or higher (>=65 Years) | -1.346 | 1.329 | -9.424 | -1.215 | 3.077 |
| Percentage of Population (>=65 years) | -1.578 | 1.964 | -14.644 | -1.241 | 4.887 |
| Number of individuals (>=65 years) per housing unit | 1.083 | 1.651 | -4.494 | 0.809 | 10.064 |
| Monthly Housing Costs in ZCTA | -1.634 | 1.864 | -18.698 | -1.456 | 5.306 |
| Median income in ZCTA | -2.040 | 1.502 | -10.418 | -1.794 | 4.730 |
| Percent below poverty level (>=65 years) | 0.602 | 0.793 | -1.867 | 0.456 | 4.270 |
| Model Fit Statistics | Global Regression | GWR | |||
| AIC: | 163614 | 147509 | |||
| AICc: | 163616 | 149226 | |||
| BIC: | 217169 | 185050 | |||
| R2: | 0.555 | 0.816 | |||
| Adjusted R2 | 0.555 | 0.781 | |||
| Variable | Mean | STD | Min | Median | Max |
| Intercept | 13.360 | 2.012 | 6.224 | 13.390 | 25.118 |
| Percent Uninsured (>=65 years) | 0.208 | 0.845 | -5.720 | 0.154 | 7.092 |
| High school graduate or higher (>=65 Years) | -1.088 | 1.261 | -7.173 | -0.947 | 4.091 |
| Bachelor's degree or higher (>=65 Years) | -1.488 | 1.399 | -8.192 | -1.407 | 5.952 |
| Percentage of Population (>=65 years) | -1.916 | 2.090 | -13.552 | -1.567 | 4.230 |
| Number of individuals (>=65 years) per housing unit | 1.358 | 1.832 | -6.463 | 1.134 | 11.637 |
| Monthly Housing Costs in ZCTA | -1.521 | 2.147 | -12.023 | -1.334 | 6.770 |
| Median income in ZCTA | -2.612 | 1.868 | -10.920 | -2.234 | 3.068 |
| Percent below poverty level (>=65 years) | 0.721 | 0.856 | -1.712 | 0.614 | 4.334 |
| Variable | 2018 | 2020 | 2022 | Trend & Interpretation |
| Pseudo R² | 0.719 | 0.787 | 0.707 | Best model fit in 2020, slight decline in 2022. |
| Spatial Pseudo R² | 0.613 | 0.636 | 0.587 | Spatial dependence strongest in 2020, weaker in 2022. |
| AIC (Model Fit) | 150789 | 143483 | 153664 | Best model fit in 2020, worsened in 2022. |
| Spatial Lag Coefficient (ρ) | 0.062 | 0.645 | 0.571 | Spatial dependence peaked in 2020, then weakened. |
| Percent Uninsured (>=65 years) | 0.031 | 0.321 | 0.105 | Huge impact in 2020 due to COVID-related healthcare access issues. Decreased in 2022 but still significant. |
| High School Graduate (>=65 years) | -0.089 | -2.118 | -0.144 | Biggest impact in 2020, possibly due to disparities in access to dental care. Stabilized in 2022. |
| Bachelor’s Degree or Higher (>=65 years) | -0.104 | -2.935 | -0.184 | Strongest effect in 2020, weakened in 2022 but still significant. |
| Population (>=65 years) | -0.161 | -2.092 | -0.277 | Aging population had the most impact in 2020, but spatial spillover reduced in 2022. |
| Individuals per Housing Unit (>=65 years) | 5.737 | 1.399 | 9.552 | Strongest impact in 2022, suggesting household crowding effects worsened post-COVID. |
| Monthly Housing Costs | -0.001 | -0.550 | 0.000 | Had a stronger effect in 2020, possibly due to economic hardships during the pandemic. |
| Median Income in ZCTA | 0.000 | -3.900 | 0.000 | Income disparities peaked in 2020 but had little impact in 2022, suggesting a shift in determinants of tooth loss. |
| Percent Below Poverty (>=65 years) | 0.076 | 1.200 | 0.165 | Poverty had the highest effect in 2020, but still strongly contributes to disparities in 2022. |
| Variable | 2018 SEM | 2020 SEM | 2022 SEM | Trend & Interpretation |
| AIC | 142360 | 144176 | 154568 | Model fit worsened in 2022, suggesting new unobserved influences on tooth loss. |
| Schwarz Criterion (BIC) | 142434 | 144251 | 154642 | Consistent trend. |
| Pseudo R² | 0.610 | 0.587 | 0.546 | Explanatory power declined, suggesting stronger spatial heterogeneity over time. |
| Log Likelihood | -71171 | -72079 | -77275 | Model fit degraded over time, likely due to shifting regional disparities. |
| Sigma-Square ML | 6.753 | 6.522 | 11.746 | Error variance increased significantly in 2022, meaning more unexplained variation. |
| S.E. of Regression | 2.599 | 2.554 | 3.427 | Greater residual variability in 2022, meaning larger differences in regional effects. |
| Percent Uninsured (>=65 years) | 0.049 | 0.041 | 0.055 | Rebounded in 2022, suggesting access to healthcare still varies regionally. |
| High School Graduate (>=65 years) | -0.053 | -0.041 | -0.059 | Education’s impact increased in 2022, reinforcing its importance in oral health. |
| Bachelor’s Degree or Higher (>=65 years) | -0.073 | -0.063 | -0.085 | Higher education had the strongest effect in 2022, likely due to better self-care and healthcare access. |
| Population (>=65 years) | -0.149 | -0.093 | -0.132 | Stronger protective effect in 2018, weakened in 2020, but partially rebounded in 2022. |
| Individuals per Housing Unit (>=65 years) | 5.408 | 2.852 | 4.134 | Crowding had the strongest effect in 2018, was weaker in 2020, and rebounded in 2022 as economic stressors persisted. |
| Monthly Housing Costs in ZCTA | -0.002 | -0.003 | -0.002 | Weaker effect in 2022, suggesting housing affordability had a diminished role. |
| Median Income in ZCTA | 0.000 | 0.000 | 0.000 | Stable effect, meaning income remained a key protective factor. |
| Percent Below Poverty (>=65 years) | 0.058 | 0.040 | 0.058 | Worsened in 2022, meaning economic disparities continued to drive tooth loss inequalities. |
| Lambda (Spatial Error Term) | 0.795 | 0.788 | 0.683 | Spatial autocorrelation weakened in 2022, meaning unobserved geographic factors are less dominant now. |
| Metric | 2018 GWR | 2020 GWR | 2022 GWR | Trend & Interpretation |
| R² | 0.898 | 0.877 | 0.816 | Model fit was highest in 2018, but declined over time, likely due to external disruptions (e.g., COVID-19, policy shifts). |
| Adjusted R² | 0.872 | 0.847 | 0.781 | Suggests increasing external influences affecting disparities. (e.g., policy changes, economic shifts). |
| AIC | 132322 | 136157 | 147509 | Best model fit in 2018, worsened over time, indicating changing patterns in disparities. |
| BIC | 181177 | 185014 | 185050 | Model complexity increased slightly, especially post-2020. |
| Percent Uninsured (>=65 years) | 0.168 | 0.255 | 0.208 | Uninsured seniors had the strongest impact on tooth loss in 2020 (COVID-19 impact). The effect weakened in 2022 as healthcare access recovered. |
| High School Graduate (>=65 years) | -0.991 | -0.992 | -1.088 | Consistently reduces tooth loss, with a slightly stronger effect in 2022. Education remained a key protective factor. |
| Bachelor’s Degree or Higher (>=65 years) | -1.328 | -1.346 | -1.488 | Higher education consistently reduced tooth loss, with the strongest effect in 2022. Likely due to greater awareness and access to dental care. |
| Population (>=65 years) | -2.076 | -1.578 | -1.916 | Aging population showed the strongest protective effect in 2018, weakened in 2020, but slightly recovered in 2022. |
| Individuals per Housing Unit (>=65 years) | 1.546 | 1.083 | 1.358 | Crowding became a weaker predictor in 2020 but rebounded in 2022, suggesting persistent economic stress in some areas. |
| Monthly Housing Costs in ZCTA | -1.402 | -1.634 | -1.521 | Stronger effect in 2020, indicating economic pressures during COVID-19, slightly weaker in 2022. |
| Median Income in ZCTA | -2.651 | -2.040 | -2.612 | Income had the strongest impact in 2018, weakened in 2020 (possibly due to pandemic shocks), then partially recovered in 2022. |
| Percent Below Poverty (>=65 years) | 0.643 | 0.602 | 0.721 | Poverty consistently worsened tooth loss, with the strongest effect in 2022. Suggests long-term economic disparities worsened post-pandemic. |
| Year | Hot Spots (High Tooth Loss) | Cold Spots (Low Tooth Loss) | Interpretation |
| 2018 | Concentrated in the Southeast, Appalachia, and Midwest. | Strongest in the West Coast, Northeast, and urban centers. | Structural disparities were already in place. |
| 2020 | Expanded into Midwest, Texas, and urban areas. | Shrunk significantly, especially in previously stable regions. | Pandemic worsened disparities and disrupted dental care access. |
| 2022 | Remained strong in Southern and rural regions. | Some recovery in urban and high-income areas. | Post-pandemic, disparities remained, with long-term factors driving the trends. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




