Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Methods
Data Sources and Study Cohort
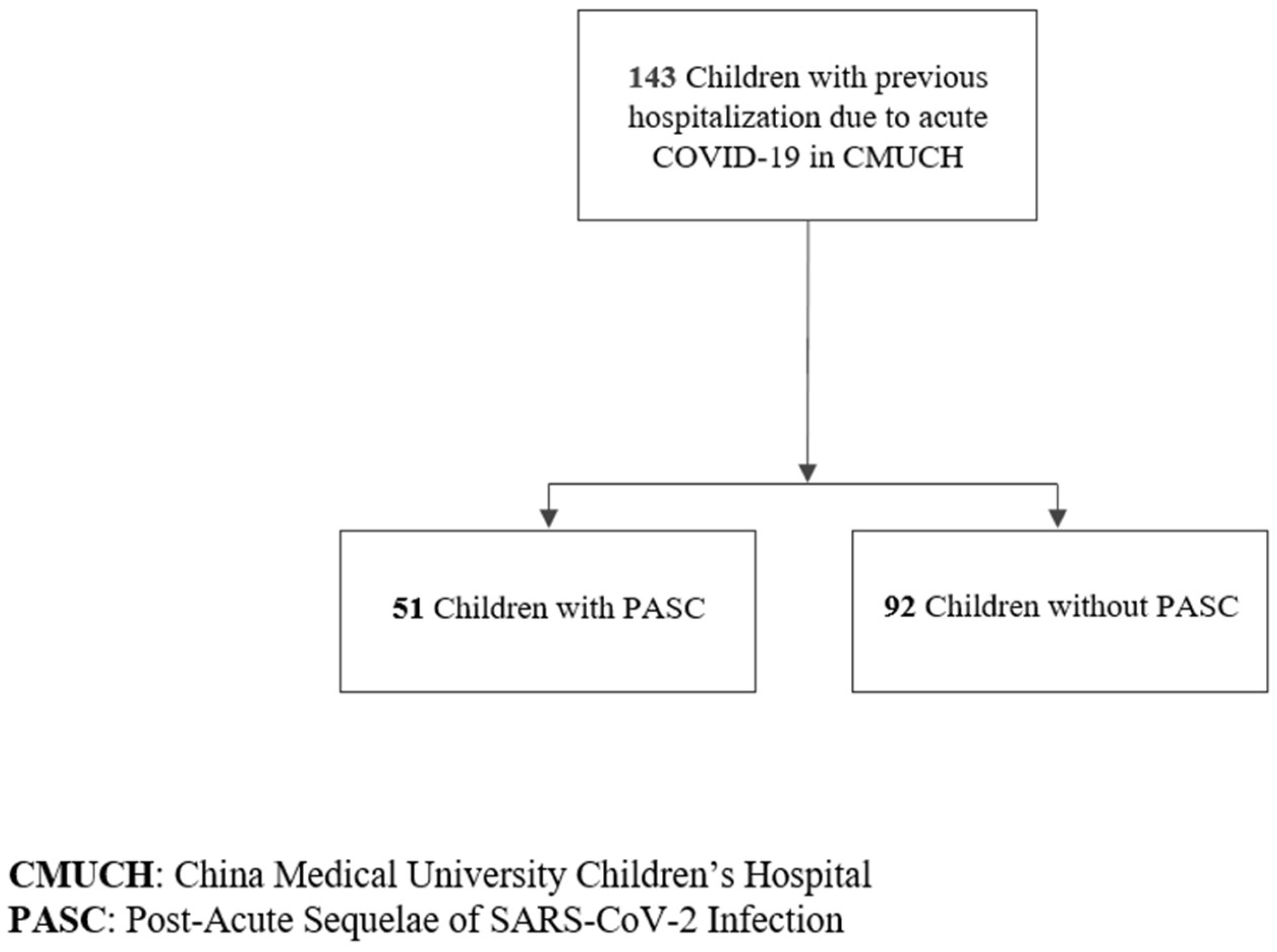
Study Outcome
Clinical Score Assignment
| Clinical category | Score |
| Respiratory system | 20 |
| Cough | 4 |
| Rhinorrhea | 4 |
| Sore throat | 4 |
| Sputum | 4 |
| Dyspnea | 4 |
| Gastrointestinal system | 20 |
| Vomiting | 6.67 |
| Diarrhea | 6.67 |
| Abdominal pain | 6.67 |
| Neurologic system | 20 |
| Neurological symptoms | 20 |
| General appearance | 20 |
| Anorexia | 5 |
| Fatigue | 5 |
| Myalgia | 5 |
| Fever | 5 |
| Medication | 20 |
| Antibiotic use | 20 |
| Total score | 100 |
Statistical Analysis
Results
|
Total (N=143) |
Cases (N=51) |
Control (N=92) | P | |
| ICU, n (%), day | 0.52 | |||
| 0 | 128 (89.5) | 44 (86.3) | 84 (91.3) | |
| 1−7 | 12 (8.4) | 5 (9.8) | 7 (7.6) | |
| 8−14 | 1 (0.7) | 1 (2.0) | 0 (0.0) | |
| ≥ 15 | 2 (1.4) | 1 (2.0) | 1 (1.1) | |
| HFNC oxygen, n (%) | 0.10 | |||
| No | 136 (95.1) | 46 (90.2) | 90 (97.8) | |
| Yes | 7 (4.9) | 5 (9.8) | 2 (2.2) | |
| Oxygen use, n (%) | 0.33 | |||
| No | 133 (93.0) | 46 (90.2) | 87 (94.6) | |
| Yes | 10 (7.0) | 5 (9.8) | 5 (5.4) | |
| Antibiotic use, n (%) | 0.15 | |||
| No | 95 (66.4) | 30 (58.8) | 65 (70.7) | |
| Yes | 48 (33.6) | 21 (41.2) | 27 (29.3) | |
| Bacteria co-infection, n (%) | 0.42 | |||
| No | 136 (95.1) | 50 (98.0) | 86 (93.5) | |
| Yes | 7 (4.9) | 1 (2.0) | 6 (6.5) | |
| Dexamethasone, n (%) | 1.00 | |||
| No | 136 (95.1) | 49 (96.1) | 87 (94.6) | |
| Yes | 7 (4.9) | 2 (3.9) | 5 (5.4) | |
| IVIG, n (%) | 0.13 | |||
| No | 139 (97.2) | 48 (94.1) | 91 (98.9) | |
| Yes | 4 (2.8) | 3 (5.9) | 1 (1.1) | |
| Antiviral agents,n (%) | ||||
| Remdesivir | 13 (9.1%) | 8 (15.7%) | 5 (5.4%) | 0.08 |
| Paxlovid | 3 (2.1%) | 2 (3.9%) | 1 (1.1%) | 0.60 |
| Remdesivir or Paxlovid | 16 (11.2%) | 10 (19.6%) | 6 (6.5%) | 0.02 |
| Symptoms |
Total (N=143) |
Cases (N=51) |
Controls (N=92) |
P |
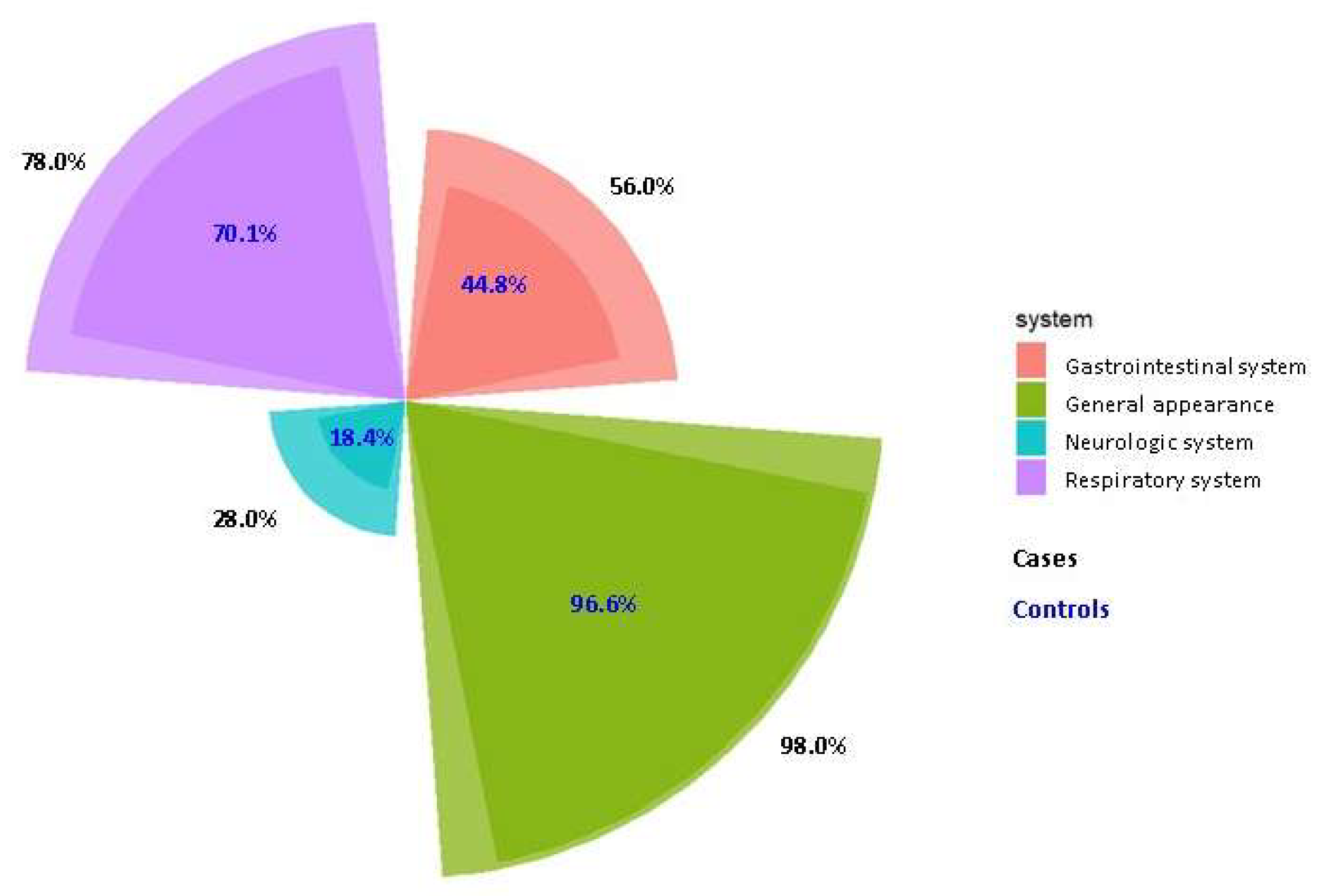
| Cough, n (%) | 93 (65.0) | 35 (68.6) | 58 (63.0) | 0.50 |
| Rhinorrhea, n (%) | 60 (42.0) | 21 (41.2) | 39 (42.4) | 0.89 |
| Sore throat, n (%) | 39 (27.3) | 15 (29.4) | 24 (26.1) | 0.67 |
| Sputum, n (%) | 39 (27.3) | 16 (31.4) | 23 (25.0) | 0.41 |
| Dyspnea, n (%) | 16 (11.2) | 9 (17.6) | 7 (7.6) | 0.07 |
| Vomiting, n (%) | 45 (31.5) | 17 (33.3) | 28 (30.4) | 0.72 |
| Diarrhea, n (%) | 18 (12.6) | 7 (13.7) | 11 (12.0) | 0.76 |
| Abdominal pain, n (%) | 31 (21.7) | 14 (27.5) | 17 (18.5) | 0.21 |
| Anorexia, n (%) | 74 (51.8) | 27 (52.9) | 47 (51.1) | 0.83 |
| Fatigue, n (%) | 66 (46.2) | 26 (51.0) | 40 (43.5) | 0.39 |
| Neurological symptoms, n (%) | 40 (28.0) | 15 (29.4) | 25 (27.2) | 0.78 |
| Seizure, n (%) | 23 (16.1) | 7 (13.7) | 16 (17.4) | 0.57 |
| Myalgia, n (%) | 27 (18.9) | 10 (19.6) | 17 (18.5) | 0.87 |
|
Total (N=143) |
Cases (N=51) |
Controls (N=92) |
P | |
| Mean ± SD | ||||
| WBC, per μL | 7150.9 ± 4285.7 | 7326.1 ± 3353.7 | 7052.7 ± 4743.4 | 0.69 |
| Neutrophil | 66.3 ± 19.9 | 69.2 ± 18.9 | 64.7 ± 20.4 | 0.21 |
| Lymphocyte | 21.7 ± 18.0 | 19.4 ± 16.5 | 23.0 ± 18.7 | 0.26 |
| Monocyte | 9.9 ± 4.5 | 9.7 ± 4.1 | 10.0 ± 4.7 | 0.69 |
| Eosinophil | 1.1 ± 2.2 | 1.1 ± 2.8 | 1.1 ± 1.9 | 0.94 |
| Basophil | 0.4 ± 0.3 | 0.3 ± 0.2 | 0.4 ± 0.4 | 0.43 |
| Bandneutrophil | 0.5 ± 2.0 | 0.3 ± 1.8 | 0.6 ± 2.2 | 0.45 |
| MDW | 24.8 ± 4.8 | 24.0 ± 5.2 | 25.2 ± 4.6 | 0.20 |
| Platelet, per μL | 211635.9 ± 87300.2 | 224396.1 ± 83649.0 | 204484.6 ± 88931.4 | 0.19 |
| MPV | 7.7 ± 0.9 | 7.6 ± 0.9 | 7.7 ± 0.9 | 0.51 |
| Hb, g/dL | 12.9 ± 1.6 | 12.9 ± 1.2 | 12.9 ± 1.8 | 0.95 |
| RBC, x106 per μL | 4.6 ± 0.5 | 4.6 ± 0.4 | 4.6 ± 0.5 | 0.56 |
| Hct | 37.2 ± 3.6 | 37.5 (3.6) | 37.1 (3.6) | 0.49 |
| hsCRP, mg/dL | 1.8 ± 3.7 | 1.7 ± 3.4 | 1.9 ± 3.9 | 0.76 |
| ALT, U/L | 23.9 ± 61.4 | 19.2 ± 40.0 | 26.6 ± 71.0 | 0.44 |
| Cr, mg/dL | 0.4 ± 0.2 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.13 |
| GFR, mL/min/1.73m2 | 123.9 ± 22.2 | 121.8 ± 23.1 | 125.1 ± 21.7 | 0.42 |
| Glucose, mg/dL | 103.1 ± 26.5 | 108.1 ± 33.9 | 100.3 ± 20.9 | 0.15 |
| Na, mmol/L | 137.8 ± 2.6 | 138.0 ± 2.7 | 137.8 ± 2.6 | 0.67 |
| K, mmol/L | 3.9 ± 0.3 | 3.9 ± 0.3 | 3.9 ± 0.3 | 0.95 |
| Clinical categories | Cases | Controls | P* |
| mean ± SD | |||
| Respiratory system | 6.85±5.52 | 7.68±5.98 | 0.41 |
| Gastrointestinal system | 4.22±5.49 | 5.07±5.31 | 0.38 |
| Neurologic system | 3.68±7.79 | 5.60±9.07 | 0.19 |
| General appearance | 10.98±5.33 | 11.10±5.92 | 0.90 |
| Medication | 5.75±9.10 | 8.40±9.97 | 0.12 |
| Total score | 31.47±15.34 | 37.85±15.50 | 0.02 |
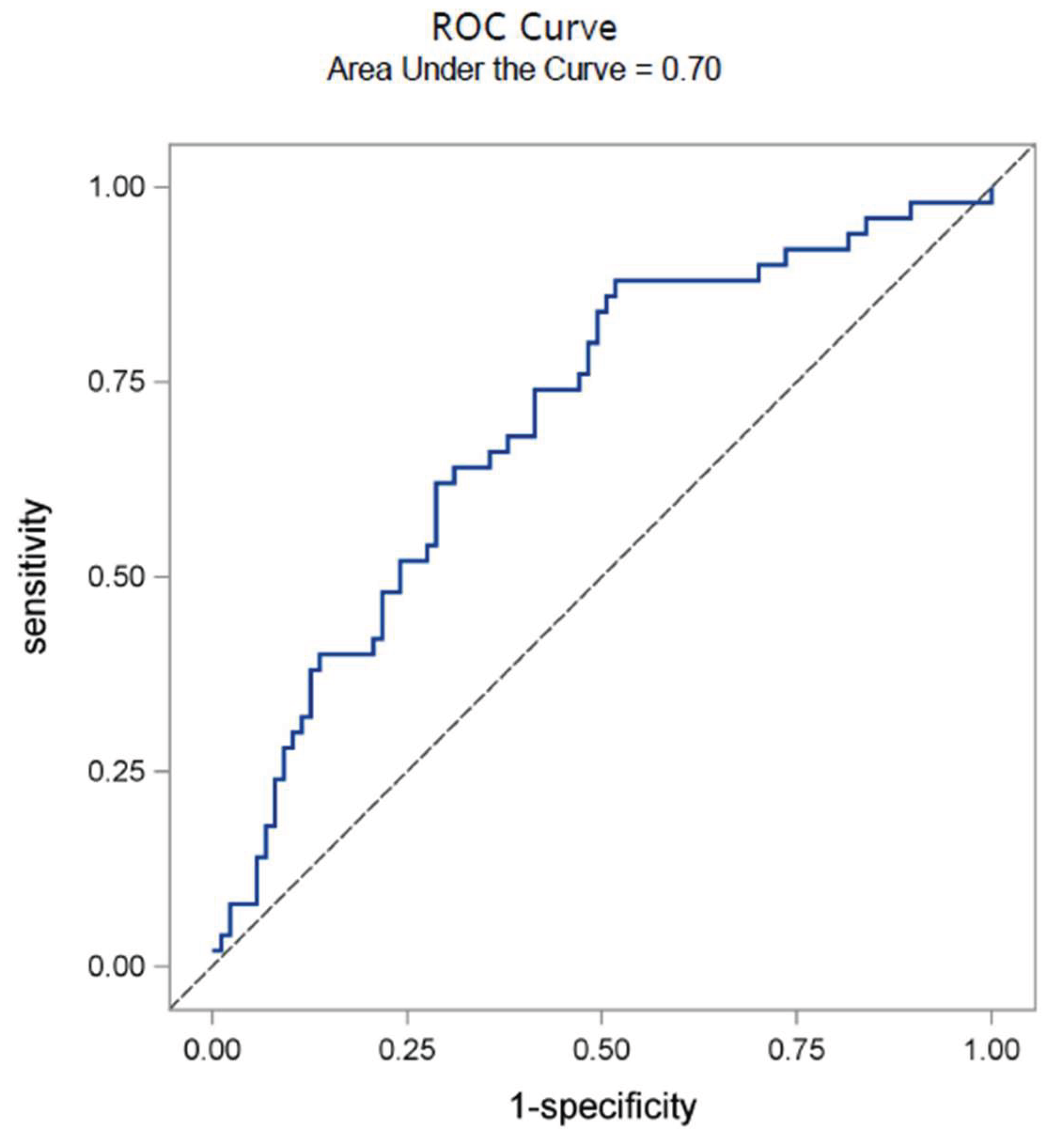
| Effect | Odds ratio (95% CI) | P |
| Total clinical scores | 1.02 (1.00- 1.05) | 0.06 |
| Age | 1.08 (0.96- 1.23) | 0.20 |
| BMI | 1.07 (0.94- 1.22) | 0.30 |
| Fever duration | 1.17 (0.95- 1.43) | 0.14 |
| c-statistic (95% CI) | 0.7 (0.61- 0.79) |
Discussion
Strengths and Limitations
Conclusions
Ethics approval and consent to participate
Clinical Trial
Consent for publication
Availability of data and material
Competing interests
Authors' contributions
Funding
Acknowledgments
References
- Hu, B; Guo, H; Zhou, P; Shi, ZL. : Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol 2021, 19(3), 141–154. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, P; Pittet, LF; Curtis, N. How Common is Long COVID in Children and Adolescents? Pediatr Infect Dis J 2021, 40(12), e482–e487. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Leon, S; Wegman-Ostrosky, T; Ayuzo Del Valle, NC; Perelman, C; Sepulveda, R; Rebolledo, PA; Cuapio, A; Villapol, S. Long-COVID in children and adolescents: a systematic review and meta-analyses. Sci Rep 2022, 12(1), 9950. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, T; Allin, B; Nugawela, MD; Rojas, N; Dalrymple, E; Pinto Pereira, S; Soni, M; Knight, M; Cheung, EY; Heyman, I. Long COVID (post-COVID-19 condition) in children: a modified Delphi process. Arch Dis Child 2022, 107(7), 674–680. [Google Scholar] [CrossRef]
- Osmanov, IM; Spiridonova, E; Bobkova, P; Gamirova, A; Shikhaleva, A; Andreeva, M; Blyuss, O; El-Taravi, Y; DunnGalvin, A; Comberiati, P. Risk factors for post-COVID-19 condition in previously hospitalised children using the ISARIC Global follow-up protocol: a prospective cohort study. Eur Respir J 2022, 59(2). [Google Scholar] [CrossRef]
- Chaichana, U; Man, KKC; Chen, A; Wong, ICK; George, J; Wilson, P; Wei, L. Definition of Post-COVID-19 Condition Among Published Research Studies. JAMA Netw Open 2023, 6(4), e235856. [Google Scholar] [CrossRef]
- Tsampasian, V; Elghazaly, H; Chattopadhyay, R; Debski, M; Naing, TKP; Garg, P; Clark, A; Ntatsaki, E; Vassiliou, VS. Risk Factors Associated With Post-COVID-19 Condition: A Systematic Review and Meta-analysis. JAMA Intern Med 2023, 183(6), 566–580. [Google Scholar] [CrossRef]
- Buonsenso, D; Munblit, D; De Rose, C; Sinatti, D; Ricchiuto, A; Carfi, A; Valentini, P. Preliminary evidence on long COVID in children. Acta Paediatr 2021, 110(7), 2208–2211. [Google Scholar] [CrossRef]
- Ludvigsson, JF. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID-19. Acta Paediatr 2021, 110(3), 914–921. [Google Scholar] [CrossRef]
- Funk, AL; Kuppermann, N; Florin, TA; Tancredi, DJ; Xie, J; Kim, K; Finkelstein, Y; Neuman, MI; Salvadori, MI; Yock-Corrales, A. Post-COVID-19 Conditions Among Children 90 Days After SARS-CoV-2 Infection. JAMA Netw Open 2022, 5(7), e2223253. [Google Scholar] [CrossRef]
- Maddux, AB; Berbert, L; Young, CC; Feldstein, LR; Zambrano, LD; Kucukak, S; Newhams, MM; Miller, K; FitzGerald, MM; He, J. Health Impairments in Children and Adolescents After Hospitalization for Acute COVID-19 or MIS-C. Pediatrics 2022, 150(3). [Google Scholar] [CrossRef] [PubMed]
- Rao, S; Lee, GM; Razzaghi, H; Lorman, V; Mejias, A; Pajor, NM; Thacker, D; Webb, R; Dickinson, K; Bailey, LC. Clinical Features and Burden of Postacute Sequelae of SARS-CoV-2 Infection in Children and Adolescents. JAMA Pediatr 2022, 176(10), 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Asadi-Pooya, AA; Nemati, H; Shahisavandi, M; Akbari, A; Emami, A; Lotfi, M; Rostamihosseinkhani, M; Barzegar, Z; Kabiri, M; Zeraatpisheh, Z. Long COVID in children and adolescents. World J Pediatr 2021, 17(5), 495–499. [Google Scholar] [CrossRef] [PubMed]
- Molteni, E; Sudre, CH; Canas, LS; Bhopal, SS; Hughes, RC; Antonelli, M; Murray, B; Klaser, K; Kerfoot, E; Chen, L. Illness duration and symptom profile in symptomatic UK school-aged children tested for SARS-CoV-2. Lancet Child Adolesc Health 2021, 5(10), 708–718. [Google Scholar] [CrossRef]
- Toepfner, N; Brinkmann, F; Augustin, S; Stojanov, S; Behrends, U. Long COVID in pediatrics-epidemiology, diagnosis, and management. Eur J Pediatr 2024, 183(4), 1543–1553. [Google Scholar] [CrossRef]
- Akobeng, AK. Understanding diagnostic tests 3: Receiver operating characteristic curves. Acta Paediatr 2007, 96(5), 644–647. [Google Scholar] [CrossRef]
- Babicki, M; Lejawa, M; Osadnik, T; Kapusta, J; Banach, M; Jankowski, P; Mastalerz-Migas, A; Kaluzinska-Kolat, Z; Kolat, D; Chudzik, M. LC risk score - development and evaluation of a scale for assessing the risk of developing long COVID. Arch Med Sci 2024, 21(1), 121–130. [Google Scholar] [CrossRef]
- Baumkotter, R; Yilmaz, S; Chalabi, J; Ten Cate, V; Mamoor Alam, AS; Golriz Khatami, S; Zahn, D; Hettich-Damm, N; Prochaska, JH; Schmidtmann, I. Risk tools for predicting long-term sequelae based on symptom profiles after known and undetected SARS-CoV-2 infections in the population. Eur J Epidemiol 2025, 40(7), 789–801. [Google Scholar] [CrossRef]
- Sun, X; Di Fusco, M; Lupton, LL; Yehoshua, A; Alvarez, MB; Allen, KE; Puzniak, L; Lopez, SMC; Cappelleri, JC. Predictors of Long COVID Among Symptomatic US Adults Testing Positive for SARS-CoV-2 at a National Retail Pharmacy. Healthcare (Basel) 2024, 12(23), 2321. [Google Scholar] [CrossRef]
- Honchar, O; Ashcheulova, T; Chumachenko, T; Chumachenko, D. Early prediction of long COVID-19 syndrome persistence at 12 months after hospitalisation: a prospective observational study from Ukraine. BMJ Open 2025, 15(1), e084311. [Google Scholar] [CrossRef]
- Morello, R; Mariani, F; Mastrantoni, L; De Rose, C; Zampino, G; Munblit, D; Sigfrid, L; Valentini, P; Buonsenso, D. Risk factors for post-COVID-19 condition (Long Covid) in children: a prospective cohort study. EClinicalMedicine 2023, 59, 101961. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, A; Sehgal, K; Gupta, A; Madhavan, MV; McGroder, C; Stevens, JS; Cook, JR; Nordvig, AS; Shalev, D; Sehrawat, TS. Post-acute COVID-19 syndrome. Nat Med 2021, 27(4), 601–615. [Google Scholar] [CrossRef] [PubMed]
- Trougakos, IP; Stamatelopoulos, K; Terpos, E; Tsitsilonis, OE; Aivalioti, E; Paraskevis, D; Kastritis, E; Pavlakis, GN; Dimopoulos, MA. Insights to SARS-CoV-2 life cycle, pathophysiology, and rationalized treatments that target COVID-19 clinical complications. J Biomed Sci 2021, 28(1), 9. [Google Scholar] [CrossRef] [PubMed]
- Bohn, MK; Hall, A; Sepiashvili, L; Jung, B; Steele, S; Adeli, K. Pathophysiology of COVID-19: Mechanisms Underlying Disease Severity and Progression. Physiology (Bethesda) 2020, 35(5), 288–301. [Google Scholar] [CrossRef]
- Kumar, S; Veldhuis, A; Malhotra, T. Neuropsychiatric and Cognitive Sequelae of COVID-19. Front Psychol 2021, 12, 577529. [Google Scholar] [CrossRef]
- Davis, HE; McCorkell, L; Vogel, JM. Topol EJ: Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol 2023, 21(3), 133–146. [Google Scholar] [CrossRef]
- Wang, C; Ramasamy, A; Verduzco-Gutierrez, M; Brode, WM; Melamed, E. Acute and post-acute sequelae of SARS-CoV-2 infection: a review of risk factors and social determinants. Virol J 2023, 20(1), 124. [Google Scholar] [CrossRef]
|
Total (N=143) |
Cases (N=51) |
Controls (N=92) |
P* | |
| Demographic data | ||||
| Age, mean ± SD, year | 6.8 ± 3.8 | 7.6 ± 4.3 | 6.3 ± 3.4 | 0.05 |
| Age, n (%), year | 0.10 | |||
| 3−6 | 87 (60.8) | 27 (52.9) | 60 (65.2) | |
| 7−12 | 41 (28.7) | 15 (29.4) | 26 (28.3) | |
| 13−18 | 15 (10.5) | 9 (17.7) | 6 (6.5) | |
| Sex, n (%) | 0.91 | |||
| Male | 85 (59.4) | 30 (58.8) | 55 (59.8) | |
| Female | 58 (40.6) | 21 (41.2) | 37 (40.2) | |
| Height, mean ± SD, cm | 120.0 ± 20.7 | 124.3 ± 23.0 | 117.6 ± 19.1 | 0.06 |
| Weight, mean ± SD, kg | 25.6 ± 14.4 | 29.1 ± 16.4 | 23.7 ± 12.8 | 0.05 |
| BMI, mean ± SD | 16.5 ± 3.5 | 17.3 ± 3.7 | 16.1 ± 3.4 | 0.06 |
| Clinical data | ||||
| Vaccine, n (%) | 0.24 | |||
| No | 56 (39.7) | 17 (33.3) | 39 (43.3) | |
| Yes | 85 (60.3) | 34 (66.7) | 51 (56.7) | |
| Comorbidity, n (%) | 0.20 | |||
| No | 94 (65.7) | 30 (58.8) | 64 (69.6) | |
| Yes | 49 (34.3) | 21 (41.2) | 28 (30.4) | |
| Duration of hospitalization, mean ± SD, day | 3.5 ± 3.0 | 3.9 ± 3.0 | 3.3 ± 3.1 | 0.34 |
| Duration of hospitalization, n (%), day | 0.25 | |||
| 1−3 | 101 (70.6) | 33 (64.7) | 68 (73.9) | |
| ≥ 4 | 42 (29.4) | 18 (35.3) | 24 (26.1) | |
| Fever temperature, mean ± SD, °C | 39.5 ± 0.8 | 39.5 ± 0.8 | 39.5 ± 0.8 | 0.96 |
| Fever temperature, n (%), °C | 0.86 | |||
| 37.8−38.9 | 29 (24.0) | 12 (26.7) | 17 (22.4) | |
| 39.0−39.9 | 51 (42.1) | 18 (40.0) | 33 (43.4) | |
| ≥ 40.0 | 41 (33.9) | 15 (33.3) | 26 (34.2) | |
| Duration of fever day, mean ± SD | 2.9 ± 1.9 | 3.2 ± 1.9 | 2.6 ± 1.9 | 0.07 |
| Duration of fever day, n (%) | 0.29 | |||
| 0 | 6 (4.4) | 2 (4.0) | 4 (4.6) | |
| 1−3 | 97 (70.8) | 31 (62.0) | 66 (75.9) | |
| 4−6 | 27 (19.7) | 13 (26.0) | 14 (16.1) | |
| ≥ 7 | 7 (5.1) | 4 (8.0) | 3 (3.4) | |
| RT-PCR Ct value, mean ± SD | 15.4 ± 5.1 | 17.1 ± 6.1 | 14.2 ± 3.8 | 0.04 |
| Antigen rapid test (ART) positive, n (%) | 77 (53.8) | 23 (45.1) | 54 (58.7) | 0.12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




