Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials
- ▪
- Indicators of vaccinometrics and mathematical formulation of proposed age-adjusted vaccine exposure index (VEI)
- ○
- Monthly vaccine exposure in infants (MVE)
- ○
- Average Age of infants during vaccination (AA)
- ○
- Average number of vaccines (AV)
- ○
- Average number of vaccine doses (AD)
- ○
- Lower VEI indicates a lower concentration of vaccines in childhood immunization schedule of country
- ○
- Higher VEI indicates a higher concentration of vaccines in childhood immunization schedule of country
- ▪
- Data and empirical evidence to validate age-adjusted vaccine exposure index (VEI)
- −
- Sample of countries for empirical evidence
- −
- Data and Sources for Statistical Analysis
- −
- Statistical analysis procedure to validate proposed age-adjusted vaccine exposure index (VEI)
- ○
- Higher Autism rate countries: Singapore, South Korea, Japan, Australia, Canada, USA (according to data in World Population Review, 2025).
- ○
- Lower Autism rate countries: Sweden, Denmark, Norway, Finland, Italy, UK (according to data in World Population Review, 2025).
- ○
- countries that administer vaccines at birth versus other that start later
- ○
- countries that administer vaccines for varicella, hepatitis B and Japanese encephalitis versus other countries that do not consider these vaccines in the childhood vaccination schedule.
- ○
- Firstly, the variables in Table 1 are analyzed with descriptive statistics given by arithmetic mean, standard deviation, skewness and kurtosis to assess the distributions and their normality. Variables with non-normal distribution are transformed into log-scale, to have a normal distribution for appropriate and robust parametric analyses.
3. Results
Sensitivity Analysis and Robustness
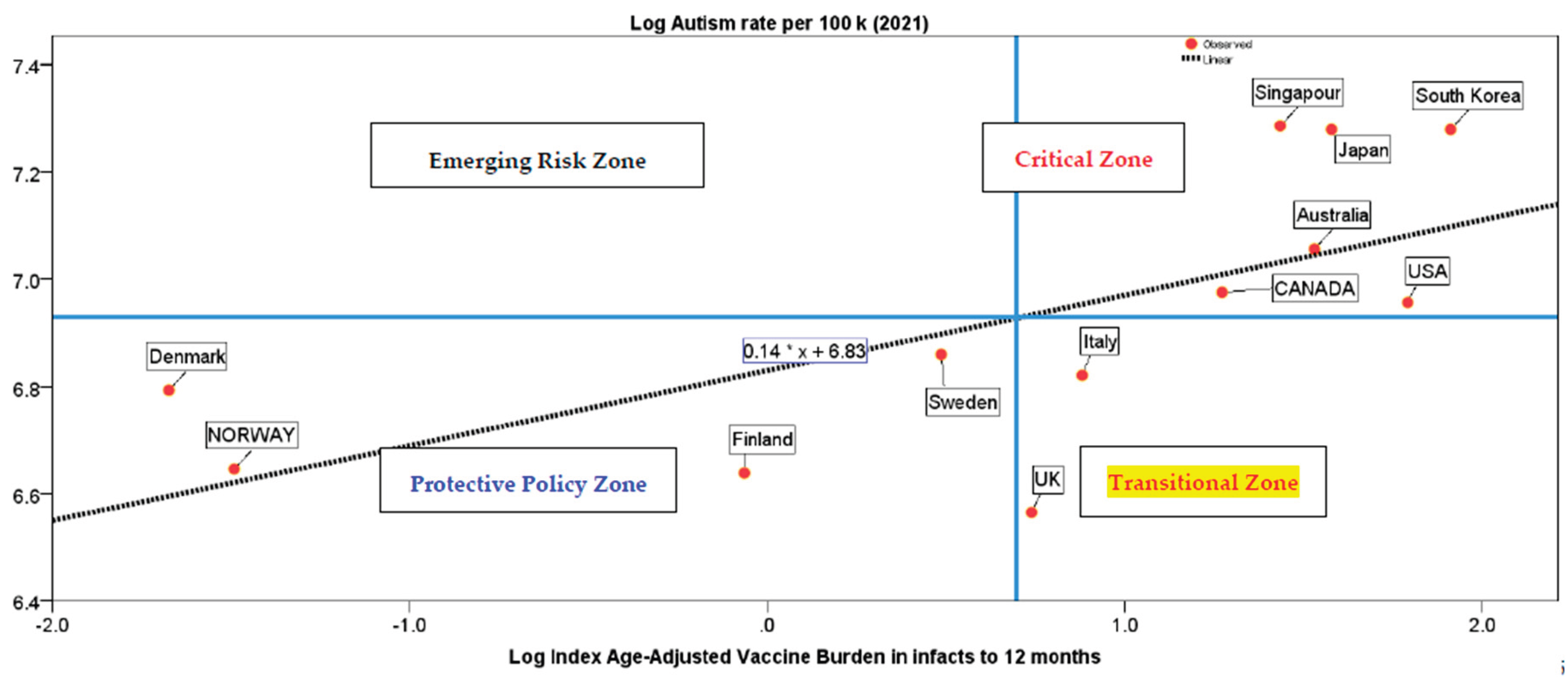
- Bottom-Left Quadrant (Low VEI, Low Autism Rate) includes countries such as Norway, Denmark, Finland, and Sweden. These nations administer fewer vaccine doses in the first year of life and exhibit lower autism rates. These Scandinavian countries with lower VEI scores demonstrate that high immunization coverage can be achieved without early and intensive vaccine schedules. These nations delay non-critical vaccines, avoid vaccination at birth, and exclude certain vaccines like varicella and hepatitis B from the first months of life. In short, these countries demonstrate that it is possible to maintain high vaccination coverage while minimizing early-life vaccine exposure. Their policies reflect a cautious approach that prioritizes long-term neurological health alongside disease prevention. Their schedules could inform revisions in countries with higher VEI scores.
- Top-Right Quadrant (High VEI, High Autism Rate). Countries including Australia, Canada, the United States, Singapore, Japan, and South Korea are positioned here. These nations administer a higher number of vaccine doses to infants under one year, often starting at birth, and show above-average autism rates. Their schedules tend to be more intensive, with early and frequent vaccinations (Frenkel, 2021). These countries with high VEI scores often administer multiple vaccines to 0-6 months and maintain dense schedules through the first year. While these programs aim to maximize protection, they may inadvertently generate adverse effects, particularly in vulnerable populations. In the U.S., autism prevalence rose from 1 in 150 children in 2000 to 1 in 31 by 2022 (CDC, 2025a). During this period, the infant immunization schedule expanded from approximately 11 vaccines to 15 before age one (CDC, 2025b). This intensification of early-life vaccine exposure may contribute to neurodevelopmental stress. International trends mirror this trajectory. Australia’s infant schedule in national immunization program increased from about 9 vaccines in 1994 to 14 in 2025, especially for Aboriginal and Torres Strait Islander children (Australian Government, 2025). Japan’s 2025 schedule includes early administration of BCG, DPT-IPV-Hib, and pneumococcal vaccines, with multiple doses before 6 months (Japan Pediatric Society, 2025; Japan, 2025). While these programs aim to maximize protection against infectious diseases, the elevated VEI scores suggest a greater early-life vaccine exposure, which may warrant further investigation into potential risks, especially in infants. Health policy reassessment in these nations should consider delaying non-essential vaccines, reducing dose intensity, and tailoring schedules for at-risk infants, such as those born preterm or with a family history of neurodevelopmental disorders (cf., (Mawson and Jacob, 2025). The VEI enables cross-country comparisons for optimizing vaccine schedules to balance efficacy and safety. Hence, by integrating VEI into national health planning, policymakers can identify high-vaccine exposure schedules, monitor trends, and design interventions that reduce early-life vaccine exposure. As Doja and Roberts (2006) argue, evidence-based policy must be grounded in rigorous epidemiological analysis—precisely what the VEI enables. Ultimately, the VEI supports a shift from one-size-fits-all immunization toward personalized, developmentally informed vaccination strategies, improving both safety and public trust (Coccia, 2023, 2022, 2022a).
- Bottom-Right Quadrant (High VEI, Low Autism Rate). Italy and the United Kingdom occupy this quadrant. Although their autism rates are below the average, their VEI scores are relatively high. In particular, Italy’s 2025 schedule shows early and intensive vaccine administration, including hexavalent vaccines and MenB within the first months. Vaccines in national immunization program schedules of these countries have considerably increased compared to 1980s-2000s period (Italy, 2025). This positioning may indicate a transitional phase in vaccine policy, with tendencies toward more intensive schedules and critical zone in space of figure 1. These countries could benefit from re-evaluating their immunization strategies, potentially aligning more closely with the Nordic model to reduce early-life exposure without compromising coverage. A Surveillance enhancement health policy can be integrating VEI into existing pharmacovigilance systems to improve early detection of adverse trends and support evidence-based policy reform.
- Top-Left Quadrant (Low VEI, High Autism Rate). Notably, no countries in the study fall into this quadrant. This absence reinforces the hypothesis-generating findings that higher vaccine exposure may be associated with higher autism rates, although causality cannot be inferred.
4. Discussion
5. Conclusions
- ▪
- Health policy implications of this study
- ▪
- Limitations and future prospects
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| VEI | Age-adjusted Vaccine exposure Index |
| 1 | The term risk here is intended as statistical measure of frequency also considering that in the fields of pharmacovigilance and epidemiology, risk is defined as the probability or likelihood that a specific disease, adverse reaction, or untoward medical event will occur in an individual within a defined timeframe following the administration of a drug. |
References
- Adams, K.; Weber, K. S.; Johnson, S. M. Exposome and immunity training: how pathogen exposure order influences innate immune cell lineage commitment and function. International Journal of Molecular Sciences 2020, 21(22), 8462. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5-TR (5th edition, text revision); Washington, DC; American Psychiatric Association Publishing, 2022. [Google Scholar]
- Anderson, L. N.; Hoyt, C. T.; Zucker, J. D.; McNaughton, A. D.; Teuton, J. R.; Karis, K.; Kumar, N. Computational tools and data integration to accelerate vaccine development: challenges, opportunities, and future directions. Frontiers in Immunology 2025, 16, 1502484. [Google Scholar] [CrossRef]
- Andersson, NW; Bech Svalgaard, I; Hoffmann, SS; et al. Aluminum-adsorbed vaccines and chronic diseases in childhood. A nationwide cohort study. Ann Intern Med 2025. Epub ahead of print. [Google Scholar] [CrossRef]
- Australia (2025) Australian Government, Department of Health, Disability and Ageing, National Immunization Program for children. https://www.health.gov.au/topics/immunisation/when-to-get-vaccinated/national-immunisation-program-schedule, https://www.health.gov.au/sites/default/files/2025-09/national-immunisation-program-schedule_0.pdf.
- Australian Government (1999) Department of Health and Aged Care, Communicable Diseases Intelligence;n. 6 - 10 June 1999, Immunisation coverage in Australian children: a systematic review 1990-1998 – Appendix. Volume 23. Available online: https://www1.health.gov.au/internet/main/publishing.nsf/Content/cda-pubs-cdi-1999-cdi2306-cdi2306a7.htm (accessed on October 2025).
- Bailey, A; Le Couteur, A; Gottesman, I; Bolton, P; Simonoff, E; Yuzda, E; Rutter, M. Autism as a strongly genetic disorder: evidence from a British twin study. Psychol Med. 1995, 25(1), 63–77. [Google Scholar] [CrossRef]
- Baron-Cohen, S; Hammer, J. Parents of Children with Asperger Syndrome: What is the Cognitive Phenotype? J Cogn Neurosci. 1997, 9(4), 548–54. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S. The hyper-systemizing, assortative mating theory of autism. Prog Neuropsychopharmacol Biol Psychiatry 2006, 30(5), 865–72. [Google Scholar] [CrossRef] [PubMed]
- Benati, I.; Coccia, M. Global analysis of timely COVID-19 vaccinations: improving governance to reinforce response policies for pandemic crises; International Journal of Health Governance, 2022; vol. 27, No. 3, pp. 240–253. [Google Scholar] [CrossRef]
- Blauvelt, C. A.; Zeme, M.; Natarajan, A.; Epstein, A.; Roh, M. E.; Morales, A.; Gaw, S. L. Respiratory syncytial virus vaccine and nirsevimab uptake among pregnant people and their neonates. JAMA network open 2025, 8(2), e2460735–e2460735. [Google Scholar] [CrossRef]
- Boretti, A. Reviewing the association between aluminum adjuvants in the vaccines and autism spectrum disorder. Journal of trace elements in medicine and biology: organ of the Society for Minerals and Trace Elements (GMS) 2021, 66, 126764. [Google Scholar] [CrossRef]
- Brophy-Williams, S.; Fidanza, M.; Marchant, A.; Way, S.; Kollmann, T. R. One vaccine for life: Lessons from immune ontogeny. Journal of paediatrics and child health 2021, 57(6), 782–785. [Google Scholar] [CrossRef]
- Buttery, J. P.; Clothier, H. Information systems for vaccine safety surveillance. Human Vaccines & Immunotherapeutics 2022, 18(6), 2100173. [Google Scholar] [CrossRef]
- Canada. Government of Canada, Recommended immunization schedules: Canadian Immunization Guide. 2025. Available online: https://www.canada.ca/en/public-health/services/publications/healthy-living/canadian-immunization-guide-part-1-key-immunization-information/page-13-recommended-immunization-schedules.html#p1c12a2.
- Benzaken, Casey L.; Miller, Joshua D.; Onono, Maricianah; Young, Sera L. Development of a cumulative metric of vaccination adherence behavior and its application among a cohort of 12-month-olds in western Kenya. Vaccine, Volume 2020, 38(Issue 18), 3429–3435. [Google Scholar] [CrossRef]
- Çatlı, Nermin Eylül; Özyurt, Gonca. The relationship between autism and autism spectrum disorders and vaccination: review of the current literature. Trends in Pediatrics. Review. Open Access 2025. [Google Scholar] [CrossRef]
- CDC. Data and Statistics on Autism Spectrum Disorder. 27 May 2025. Available online: https://www.cdc.gov/autism/data-research/index.html#cdc_data_surveillance_section_3-cdcs-addm-network.
- CDC. Autism Spectrum Disorder (ASD). 2025a. Available online: https://www.cdc.gov/autism/data-research/index.html#cdc_data_surveillance_section_3-cdcs-addm-network (accessed on October 2025).
- CDC. Recommended Childhood Immunization Schedule -- United States, 2000. 2025b. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4902a4.htm (accessed on October 2025).
- Chinese center for disease control and prevention (2021). Available online: https://en.chinacdc.cn/health_topics/immunization/202203/t20220302_257317.html.
- Coccia, M. Problem-driven innovations in drug discovery: co-evolution of the patterns of radical innovation with the evolution of problems; Health Policy and Technology, 2016; vol. 5, n. 2, pp. 143–155. [Google Scholar] [CrossRef]
- Coccia, M. Effects of human progress driven by technological change on physical and mental health; STUDI DI SOCIOLOGIA, 2021; N. 2; pp. 113–132. [Google Scholar] [CrossRef]
- Coccia, M. COVID-19 in society between 2020 (without vaccinations) and 2021 (with vaccinations): A case study; Journal of Social and Administrative Sciences - J. Adm. Soc. Sci. - JSAS, 2022; Vol. 9, n. 2, pp. 113–130. [Google Scholar] [CrossRef]
- Coccia, M. Improving preparedness for next pandemics: Max level of COVID-19 vaccinations without social impositions to design effective health policy and avoid flawed democracies; Environmental Research, 2022; Volume 213, p. 113566. [Google Scholar] [CrossRef]
- Coccia, M. COVID-19 pandemic over 2020 (with lockdowns) and 2021 (with vaccinations): similar effects for seasonality and environmental factors. Environmental Research 2022a, Volume 208, n. 112711. [Google Scholar] [CrossRef]
- Coccia, M. Optimal levels of vaccination to reduce COVID-19 infected individuals and deaths: A global analysis; Environmental Research, 2022a; vol. 204, Part C, p. 112314. [Google Scholar] [CrossRef]
- Coccia, M. Relation between COVID-19 vaccination and economic development of countries; Turkish Economic Review, 2022b; vol. 9, n. 2, pp. 121–139. [Google Scholar] [CrossRef]
- Coccia, M. COVID-19 Pandemic Crisis: Analysis of origins, diffusive factors and problems of lockdowns and vaccinations to design best policy responses Vol.2; KSP Books; Kadikoy, Istanbul, Turkey, 2023; ISBN 978-625-7813-54-9. (e-Book). [Google Scholar]
- Coccia, M. COVID-19 Vaccination is not a Sufficient Public Policy to face Crisis Management of next Pandemic Threats. Public Organiz Rev 2023, 23, 1353–1367. [Google Scholar] [CrossRef]
- Coccia, M.; Bellitto, M. Human progress and its socioeconomic effects in society; Journal of Economic and Social Thought, 2018; vol. 5, n. 2, pp. 160–178. [Google Scholar] [CrossRef]
- Conklin, L.; Hviid, A.; Orenstein, W. A.; Pollard, A. J.; Wharton, M.; Zuber, P. Vaccine safety issues at the turn of the 21st century. BMJ Global Health 2021, 6 (Suppl 2), e004898. [Google Scholar] [CrossRef]
- Davis, RL; Rubanowice, D; Shinefield, HR; et al. Immunization Levels Among Premature and Low-Birth-Weight Infants and Risk Factors for Delayed Up-to-Date Immunization Status. JAMA 1999, 282(6), 547–553. [Google Scholar] [CrossRef]
- Decouttere, C.; De Boeck, K.; Vandaele, N. Advancing sustainable development goals through immunization: a literature review. Globalization and Health 2021, 17(1), 95. [Google Scholar] [CrossRef]
- Denmark. Statens Serum Institut. Childhood vaccination programme. 2025. Available online: https://en.ssi.dk/vaccination/the-danish-childhood-vaccination-programme.
- Doja, A; Roberts, W. Immunizations and autism: a review of the literature. Can J Neurol Sci 2006, 33(4), 341–6. [Google Scholar] [CrossRef] [PubMed]
- Eicher, T.; Quackenbush, J.; Ne'eman, A. Challenging Claims of an Autism Epidemic - Misconceptions and a Path Forward. The New England journal of medicine 2026, 394(4), 313–315. [Google Scholar] [CrossRef]
- Elfatimi, E.; Lekbach, Y.; Prakash, S.; BenMohamed, L. Artificial intelligence and machine learning in the development of vaccines and immunotherapeutics—yesterday, today, and tomorrow. Frontiers in Artificial Intelligence 2025, 8, 1620572. [Google Scholar] [CrossRef]
- Estes, A; Munson, J; Rogers, SJ; Greenson, J; Winter, J; Dawson, G. Long-term outcomes of early intervention in 6-year-old children with autism spectrum disorder. J Am Acad Child Adolescent Psych 2015, 54, 580–87. [Google Scholar] [CrossRef]
- Finland. Finnish Institute for Health and Welfare. Vaccination programme for children and adults. 2025. Available online: https://thl.fi/en/topics/infectious-diseases-and-vaccinations/information-about-vaccinations/vaccination-programme-for-children-and-adults.
- Fortmann, MI; Dirks, J; Goedicke-Fritz, S; Liese, J; Zemlin, M; Morbach, H; Härtel, C. Immunization of preterm infants: current evidence and future strategies to individualized approaches. Semin Immunopathol 2022, 44(6), 767–784. [Google Scholar] [CrossRef]
- Frenkel, LD. The global burden of vaccine-preventable infectious diseases in children less than 5 years of age: Implications for COVID-19 vaccination. How can we do better? Allergy Asthma Proc. 2021, 42(5), 378–385. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Fogeda, I.; Besbassi, H.; Lariviere, Y.; Ogunjimi, B.; Abrams, S.; Hens, N. Within-host modeling to measure dynamics of antibody responses after natural infection or vaccination: A systematic review. Vaccine 2023, 41(25), 3701–3709. [Google Scholar] [CrossRef]
- GBD 2023 Vaccine Coverage Collaborators. Global, regional, and national trends in routine childhood vaccination coverage from 1980 to 2023 with forecasts to 2030: a systematic analysis for the Global Burden of Disease Study 2023. Lancet 2025, 406(10500), 235–260. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Geier, D. A.; Kern, J. K.; Homme, K. G.; Geier, M. R. Abnormal Brain Connectivity Spectrum Disorders Following Thimerosal Administration: A Prospective Longitudinal Case-Control Assessment of Medical Records in the Vaccine Safety Datalink. Dose-response: a publication of International Hormesis Society 2017, 15(1), 1559325817690849. [Google Scholar] [CrossRef]
- Global Burden of Disease Study 2021 Autism Spectrum Collaborators. The global epidemiology and health burden of the autism spectrum: findings from the Global Burden of Disease Study 2021. Lancet Psychiatry 2025, 12(2), 111–121. [CrossRef]
- Grosvenor, LP; Croen, LA; Lynch, FL; Marafino, BJ; Maye, M; Penfold, RB; Simon, GE; Ames, JL. Autism Diagnosis Among US Children and Adults, 2011-2022. JAMA Netw Open 2024, 7(10), e2442218. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S; Sharawat, IK; Panda, PK; Kothare, SV. The vaccine-autism connection: No link, still debate, and we are failing to learn the lessons. Autism 2025, 29(7), 1639–1645. [Google Scholar] [CrossRef]
- Haeuser, Emily; et al. Global, regional, and national trends in routine childhood vaccination coverage from 1980 to 2023 with forecasts to 2030: a systematic analysis for the Global Burden of Disease Study 2023. The Lancet 2025, 406(Issue 10500), 235–260. [Google Scholar] [CrossRef] [PubMed]
- Howlin, P; Magiati, I. Autism spectrum disorder: outcomes in adulthood. Curr Opin Psychiatry 2017, 30, 69–76. [Google Scholar] [CrossRef]
- Hughes, MM; Katz, J; Englund, JA; Khatry, SK; Shrestha, L; LeClerq, SC; Steinhoff, M; Tielsch, JM. Infant vaccination timing: Beyond traditional coverage metrics for maximizing impact of vaccine programs, an example from southern Nepal. Vaccine 2016, 34(7), 933–41. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Immunization Safety Review: Vaccines and Autism; Washington, DC; The National Academies Press, 2004. [Google Scholar] [CrossRef]
- Institute of Medicine. Adverse Effects of Vaccines: Evidence and Causality; Washington, DC; The National Academies Press, 2012. [Google Scholar] [CrossRef]
- Institute of Medicine. The Childhood Immunization Schedule and Safety: Stakeholder Concerns, Scientific Evidence, and Future Studies; Washington, DC; The National Academies Press, 2013. [Google Scholar] [CrossRef]
- Issac, A; Halemani, K; Shetty, A; Thimmappa, L; Vijay, VR; Koni, K; Mishra, P; Kapoor, V. The global prevalence of autism spectrum disorder in children: a systematic review and meta-analysis. Osong Public Health Res Perspect 2025, 16(1), 3–27. [Google Scholar] [CrossRef]
- Italy (2025), Regione Lombardia, Calendario Vaccinale. Available online: https://www.wikivaccini.regione.lombardia.it/wps/portal/site/wikivaccini/DettaglioRedazionale/vaccinazioni-disponibili/calendario-vaccinale.
- Japan. Changes in the Immunization Schedule Recommended by the Japan Pediatric Society, May 19, 2025. 2025. Available online: https://www.jpeds.or.jp/uploads/files/20250205_Immunization_Schedule_english.pdf.
- Japanese Pediatric Society. Changes in the Immunization Schedule Recommended. 2025. Available online: https://www.jpeds.or.jp/uploads/files/20250205_Immunization_Schedule_english.pdf (accessed on 22 October 2025).
- Kargı, B.; Coccia, M.; Uçkaç, B. C. How does the wealth level of nations affect their COVID19 vaccination plans? Economics, Management and Sustainability 2023, 8(2), 6–19. [Google Scholar] [CrossRef]
- Kargı, B.; Coccia, M. Rethinking the Role of Vaccinations in Mitigating COVID-19 Mortality: A Cross-National Analysis. KMÜ Sosyal ve Ekonomik Araştırmalar Dergisi, KMU Journal of Social and Economic Research 2024, 26(47), 1173–1192. [Google Scholar] [CrossRef]
- Lai, JKY; Weiss, JA. Priority service needs and receipt across the lifespan for individuals with autism spectrum disorder. Autism Res 2017, 10, 1436–47. [Google Scholar] [CrossRef]
- Lord, C; Charman, T; Havdahl, A; et al. The Lancet Commission on the future of care and clinical research in autism. Lancet 2022, 399, 271–334. [Google Scholar] [CrossRef]
- Magazzino, C.; Mele, M.; Coccia, M. A machine learning algorithm to analyze the effects of vaccination on COVID-19 mortality; Epidemiology and infection, 2022; pp. 1–24. [Google Scholar] [CrossRef]
- Mawson, AR; Jacob, B. Vaccination and Neurodevelopmental Disorders: A Study of Nine-Year-Old Children Enrolled in Medicaid. Science, Public Health Policy and the Law 2025, 6, 55–61. Available online: https://publichealthpolicyjournal.com/vaccination-and-neurodevelopmental-disorders-a-study-of-nine-year-old-children-enrolled-in-medicaid/ (accessed on December 7, 2025).
- Metcalf, C. J. E.; Graham, A. L.; Yates, A. J.; Cummings, D. A. Convergence and divergence of individual immune responses over the life course. Science 2025, 389(6760), 604–609. [Google Scholar] [CrossRef]
- Ministry of health; family welfare. Government of India, National Health Mission, National Immunization Schedule. 2018. Available online: https://nhm.gov.in/New_Updates_2018/NHM_Components/Immunization/report/National_%20Immunization_Schedule.pdf.
- Miravalle, AA; Schreiner, T. Neurologic complications of vaccinations. Handb Clin Neurol 2014, 121, 1549–57. [Google Scholar] [CrossRef] [PubMed]
- Morrocchi, E.; van Haren, S.; Palma, P.; Levy, O. Modeling human immune responses to vaccination in vitro. Trends in immunology 2024, 45(1), 32–47. [Google Scholar] [CrossRef]
- Nair, KS; George, RL; Remya, VR; A R, P; Saji, CV; Mullasseril, RR; Shenoi, RA; Nair, J; Krishna, R; N K, K; Thomas, A; Varughese, D; Chandra, G; Mohanakumar, KP; Rajamma, U. Prevalence Estimates of Neurodevelopmental Disorders (NDD) in a South Indian Population. Ann Neurosci 2025, 09727531251348188. [Google Scholar] [CrossRef] [PubMed]
- Nath, A. Neurologic Complications With Vaccines: What We Know, What We Don't, and What We Should Do. Neurology 2023, 101(14), 621–626. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine 2025. Statement on CDC’s Changes to Guidance on Vaccines and Autism. 2025. Available online: https://www.nationalacademies.org/news/statement-on-cdc-s-changes-to-guidance-on-vaccines-and-autism?utm_source=NASEM+News+and+Publications&utm_campaign=b32b20d912-NAP+Mail+New+2025.11.24&utm_medium=email&utm_term=0_-1c0f2fa5e6-112916397&mc_cid=b32b20d912&mc_eid=ddceeca657 (accessed on December 2025).
- National Academies of Sciences; Engineering; and Medicine. Guidance on Routine Childhood Immunizations; Washington, DC; The National Academies Press, 2024. [Google Scholar] [CrossRef]
- National Academies of Sciences; Engineering; and Medicine. The Comprehensive Autism Care Demonstration: Solutions for Military Families; Washington, DC; The National Academies Press, 2025. [Google Scholar] [CrossRef]
- National Academies of Sciences; Engineering; and Medicine. Vaccine Risk Monitoring and Evaluation at the Centers for Disease Control and Prevention; Washington, DC; The National Academies Press, 2025. [Google Scholar] [CrossRef]
- Norway (2025). Norwegian Institute of Public Health. Vaccination of children in Norway. [Internet]. Oslo: The Norwegian Directorate of Health; updated Thursday, April 4, 2024 [retrieved Friday, October 3, 2025]. Available from: https://www.helsenorge.no/en/information-in-english/vaccination-of-children/ and https://www.fhi.no/en/va/childhood-immunisation-programme/when-will-your-child-be-offered-vaccines/.
- Nouri, N.; Cao, R.G.; Bunsow, E.; et al. Young infants display heterogeneous serological responses and extensive but reversible transcriptional changes following initial immunizations. Nat Commun 2023, 14, 7976. [Google Scholar] [CrossRef]
- O'Leary, K. Antibiotics reduce vaccine responses in infants; Nat Med, 30 Apr 2025. [Google Scholar] [CrossRef]
- Pasetti, M. F.; Ackerman, M. E.; Hoen, A. G.; Alter, G.; Tsang, J. S.; Marchant, A. Maternal determinants of infant immunity: Implications for effective immunization and maternal-child health. Vaccine 2020, 38(28), 4491. [Google Scholar] [CrossRef] [PubMed]
- Pichichero, ME. Challenges in vaccination of neonates, infants and young children. Vaccine 2014, 32(31), 3886–94, Epub. [Google Scholar] [CrossRef]
- Pickering, L. K.; Meissner, H. C.; Orenstein, W. A.; Cohn, A. C. Principles of vaccine licensure, approval, and recommendations for use. In Mayo Clinic Proceedings; Elsevier, March 2020; Vol. 95, No. 3, pp. 600–608. [Google Scholar]
- Pollard, A. J.; Bijker, E. M. A guide to vaccinology: from basic principles to new developments. Nature Reviews Immunology 2021, 21(2), 83–100. [Google Scholar] [CrossRef]
- Principi, N; Esposito, S. Aluminum in vaccines: Does it create a safety problem? Vaccine 2018, 36(39), 5825–5831. [Google Scholar] [CrossRef] [PubMed]
- Quincer, E. M.; Cranmer, L. M.; Kamidani, S. Prenatal maternal immunization for infant protection: a review of the vaccines recommended, infant immunity and future research directions. Pathogens 2024, 13(3), 200. [Google Scholar] [CrossRef]
- Ramachandran, P; Grose, C. Serious neurological adverse events in immunocompetent children and adolescents caused by viral reactivation in the years following varicella vaccination. Rev Med Virol 2024, 34(3), e2538. [Google Scholar] [CrossRef]
- Reuters. Autism rates: Why are they on the rise. By Nancy Lapid, April 17. 2025; Available online: https://www.reuters.com/business/healthcare-pharmaceuticals/why-are-autism-rates-rising-2025-01-14/.
- Rodewald, L; Maes, E; Stevenson, J; Lyons, B; Stokley, S; Szilagyi, P. Immunization performance measurement in a changing immunization environment. Pediatrics 2023, 103 4 Pt 2, 889–97. [Google Scholar] [CrossRef]
- Roper, L.; Hall, M. A. K.; Cohn, A. Overview of the United States’ immunization program. The Journal of infectious diseases 2021, 224 (Supplement_4), S443–S451. [Google Scholar] [CrossRef]
- Sadarangani, M.; Kollmann, T.; Bjornson, G.; Heath, P.; Clarke, E.; Marchant, A.; Munoz, F. M. The Fifth International Neonatal and Maternal Immunization Symposium (INMIS 2019): securing protection for the next generation. Msphere 2021, 6(1), 10–1128. [Google Scholar] [CrossRef]
- Sadeck, LDSR; Kfouri, RÁ. An update on vaccination in preterm infants. J Pediatr (Rio J) 2023, 99 Suppl 1(Suppl 1), S81–S86. [Google Scholar] [CrossRef]
- Sandbank, M; Bottema-Beutel, K; Crowley LaPoint, S; et al. Autism intervention meta-analysis of early childhood studies (Project AIM): updated systematic review and secondary analysis. BMJ 2023, 383, e076733. [Google Scholar] [CrossRef]
- Santilli, V.; Sgrulletti, M.; Costagliola, G.; Beni, A.; Mastrototaro, M. F.; Montin, D.; Moschese, V. Maternal Immunization: Current Evidence, Progress, and Challenges. Vaccines 2025, 13(5), 450. [Google Scholar] [CrossRef]
- Schmitt, C.; Goedicke-Fritz, Sybelle; Fortmann, Ingmar; Zemlin, Michael. Vaccinations in preterm infants: Which and when? Seminars in Fetal and Neonatal Medicine 2025, 101670. [Google Scholar] [CrossRef]
- Scientific American. The Real Reasons Autism Rates Are Up in the U.S. By Jessica Wright & Spectrum. 3 March 2017. Available online: https://www.scientificamerican.com/article/the-real-reasons-autism-rates-are-up-in-the-u-s/#.
- Scott, J. Why Denmark’s vaccine schedule works for Denmark — but not for the United States. The two countries have fundamentally different health care systems and disease burdens. STAT10. 2025. Available online: https://www.statnews.com/2025/12/19/denmark-vaccine-schedule-vs-us/.
- Semmes, EC; Chen, JL; Goswami, R; Burt, TD; Permar, SR; Fouda, GG. Understanding Early-Life Adaptive Immunity to Guide Interventions for Pediatric Health. Front Immunol 2021, 11, 595297. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shaaban, F. L.; Groenendijk, R. W.; Baral, R.; Caballero, M. T.; Crowe, J. E.; Englund, J. A.; Bont, L. J. The path to equitable respiratory syncytial virus prevention for infants: challenges and opportunities for global implementation; The Lancet Global Health, 2025. [Google Scholar]
- Shapiro, G. K.; Kaufman, J.; Brewer, N. T.; Wiley, K.; Menning, L.; Leask, J.; BeSD Working Group. A critical review of measures of childhood vaccine confidence. Current opinion in immunology 2021, 71, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Shattock, Andrew J; et al. Contribution of vaccination to improved survival and health: modelling 50 years of the Expanded Programme on Immunization. The Lancet 2024, 403(Issue 10441), 2307–2316. [Google Scholar] [CrossRef] [PubMed]
- Shaw, KA; Williams, S; Patrick, ME; Valencia-Prado, M; Durkin, MS; Howerton, EM; Ladd-Acosta, CM; Pas, ET; Bakian, AV; Bartholomew, P; Nieves-Muñoz, N; Sidwell, K; Alford, A; Bilder, DA; DiRienzo, M; Fitzgerald, RT; Furnier, SM; Hudson, AE; Pokoski, OM; Shea, L; Tinker, SC; Warren, Z; Zahorodny, W; Agosto-Rosa, H; Anbar, J; Chavez, KY; Esler, A; Forkner, A; Grzybowski, A; Agib, AH; Hallas, L; Lopez, M; Magaña, S; Nguyen, RHN; Parker, J; Pierce, K; Protho, T; Torres, H; Vanegas, SB; Vehorn, A; Zhang, M; Andrews, J; Greer, F; Hall-Lande, J; McArthur, D; Mitamura, M; Montes, AJ; Pettygrove, S; Shenouda, J; Skowyra, C; Washington, A; Maenner, MJ. Prevalence and Early Identification of Autism Spectrum Disorder Among Children Aged 4 and 8 Years - Autism and Developmental Disabilities Monitoring Network, 16 Sites, United States, 2022. MMWR Surveill Summ 2025, 74(2), 1–22. [Google Scholar] [CrossRef] [PubMed]
- Singapour (2025) Government of Singapore, Communicable Diseases Agency, Vaccinations, Table 1: National Childhood Immunisation Schedule. https://www.cda.gov.sg/public/vaccinations and https://isomer-user-content.by.gov.sg/18/00b7fdea-24a1-4c26-b67e-d6618f31d6cb/NCIS_Sept%202025.pdf.
- South Korea (2025). Vaccination for infants, Yangcheon-Gu Office, Seul. https://www.yangcheon.go.kr/english/english/04/10402030000002016110903.jsp and https://www.gunsan.go.kr/_cms/board/eFileDownload/434/1449916/12b2c60c49317fc21ea41eb435a79159.
- Strunecka, A.; Blaylock, R. L.; Patocka, J.; Strunecky, O. Immunoexcitotoxicity as the central mechanism of etiopathology and treatment of autism spectrum disorders: A possible role of fluoride and aluminum. Surgical neurology international 2018, 9, 74. [Google Scholar] [CrossRef]
- Sweden. Vaccination programmes. The Public Health Agency of Sweden. 2025. Available online: https://www.folkhalsomyndigheten.se/the-public-health-agency-of-sweden/communicable-disease-control/vaccinations/vaccination-programmes/ (accessed on October, 2025).
- Swedish Council on Health Technology Assessment. Vaccines to Children: Protective Effect and Adverse Events: A Systematic Review [Internet]. SBU Yellow Report No. 191; Stockholm; Swedish Council on Health Technology Assessment (SBU), Feb 2009. [PubMed]
- Talantseva, OI; Romanova, RS; Shurdova, EM; Dolgorukova, TA; Sologub, PS; Titova, OS; Kleeva, DF; Grigorenko, EL. The global prevalence of autism spectrum disorder: A three-level meta-analysis. Front. Psychiatry 2023, 14, 1071181. [Google Scholar] [CrossRef]
- Tomljenovic, L; Shaw, CA. Do aluminum vaccine adjuvants contribute to the rising prevalence of autism? J Inorg Biochem. 2011, 105(11), 1489–99. [Google Scholar] [CrossRef] [PubMed]
- Tregoning G. 2022. Neonatal Immunology, Category: Immune Development, British Society for immunology. https://www.immunology.org/public-information/bitesized-immunology/immune-development/neonatal-immunology; https://www.immunology.org/sites/default/files/2022-08/Neonatal%20Immunology.pdf (accessed 12 January 2026).
- UK. NHS vaccinations and when to have them. 2025. Available online: https://www.nhs.uk/vaccinations/nhs-vaccinations-and-when-to-have-them/ (accessed on October, 2025).
- USA (2025). CDC, Vaccines & Immunizations , Child and Adolescent Immunization Schedule by Age (Addendum updated August 7, 2025). https://www.cdc.gov/vaccines/hcp/imz-schedules/child-adolescent-catch-up.html; https://www.cdc.gov/vaccines/hcp/imz-schedules/downloads/child/0-18yrs-child-combined-schedule.pdf (Accessed October, 2025).
- Vajdy, M.; Rath, B. A.; Talaat, K. R. Immunological aspects of vaccine safety. Frontiers in immunology 2023, 14, 1212148. [Google Scholar] [CrossRef]
- Van Tilbeurgh, M.; Lemdani, K.; Beignon, A. S.; Chapon, C.; Tchitchek, N.; Cheraitia, L.; Manet, C. Predictive markers of immunogenicity and efficacy for human vaccines. Vaccines 2021, 9(6), 579. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, JA; Rao, ASRS. 2025 Immunizations, Autism, and Statistical Analysis. Am J Med 2025, 138(9), 1175–1176. [Google Scholar] [CrossRef] [PubMed]
- Vekemans, J.; Hasso-Agopsowicz, M.; Kang, G.; Hausdorff, W. P.; Fiore, A.; Tayler, E.; Lipsitch, M. Leveraging vaccines to reduce antibiotic use and prevent antimicrobial resistance: a World Health Organization action framework. Clinical Infectious Diseases 2021, 73(4), e1011–e1017. [Google Scholar] [CrossRef] [PubMed]
- World Population Review (2025), Autism Rates by Country 2025. Available online: https://worldpopulationreview.com/country-rankings/autism-rates-by-country.
- World Population Review (2025a), Vaccination Rates by Country 2025. Available online: https://worldpopulationreview.com/country-rankings/vaccination-rates-by-country.
| Variables and sources | Description |
| Period under study | Year 2020, 2021 (last year available) in dataset |
| Countries. Source: World Population Review (2025), https://worldpopulationreview.com/country-rankings/autism-rates-by-country | Countries and related autism rate in World Population Review (2025): Singapore, South Korea, Japan, Australia, Canada, USA, Sweden, Denmark, Norway, Finland, Italy, and the UK |
| Autism rate 2021 per 100k people in countries under study, Source: World Population Review (2025), https://worldpopulationreview.com/country-rankings/autism-rates-by-country | Normalized rates indicate the number of children out of 100,000 who have been diagnosed at any age with autism, year 2021. |
| Months of infants (having ≤ 1 year) in immunization dates | Month of infants (having ≤ 1 year) to which vaccines are administered, set according to the country's recommended childhood vaccination schedule and designed by public health bodies. |
| Total number of vaccines to infants from birth to 6 months and from birth to 12 months | Total number of vaccines administered to infants having less than 1 year (recommended or compulsory) from national childhood immunization schedule in sample of countries under study. Sources of data in Table S1. |
| Number of doses of vaccines to infants ≤ 1 year from birth to 6 months and from birth to 12 months | Total number of doses of vaccines administered to infants having less than 1 year (recommended or compulsory) from national childhood immunization schedule in sample of countries under study. Sources of data in Table S1. |
| Vaccination Rates (%), Source: World Population Review (2025a), Vaccination Rates by Country 2020, https://worldpopulationreview.com/country-rankings/vaccination-rates-by-country | Average Percentage of Vaccination Rates 2020, Vaccinations address the following diseases: DTP3 - Diphtheria, Tetanus, and Pertussis; MCV1 - Measles and POL3 – Polio; Data estimates from WHO/UNICEF Estimates of National Immunization Coverage |
| Details for National Immunization Program Schedules for children | See Table S1 in Supplemental Information |
| Groups of countries | Vacc Rate % | Autism Rate |
Average Age infants 12 months |
Average Age infants 6 months | Total Vacc. to 12 months | Total Vacc. to 6 months | Total Doses. to 12 months | Total Doses. to 6 months | Vaccine exposure to 12 months | |
| LOWER Autism rate | M | 94.56 | 834.33 | 5.90 | 3.42 | 8.00 | 7.17 | 9.33 | 6.50 | 83.50 |
| SD | 2.40 | 98.78 | 0.63 | 0.70 | 2.83 | 1.72 | 5.32 | 3.73 | 62.22 | |
| HIGHER Autism rate | M | 95.00 | 1273.33 | 4.90 | 3.09 | 14.83 | 9.83 | 19.67 | 14.83 | 291.50 |
| SD | 2.89 | 200.67 | 0.45 | 0.39 | 0.98 | 1.60 | 3.67 | 3.06 | 56.94 | |
| NO Varicella vaccine | M | 94.80 | 817.80 | 5.91 | 3.63 | 7.20 | 6.60 | 8.40 | 6.00 | 66.60 |
| SD | 2.60 | 100.73 | 0.70 | 0.50 | 2.28 | 1.14 | 5.37 | 3.94 | 51.94 | |
| Varicella Vaccine | M | 94.76 | 1222.43 | 5.03 | 2.98 | 14.43 | 9.86 | 18.86 | 14.00 | 273.86 |
| SD | 2.71 | 227.37 | 0.54 | 0.45 | 1.40 | 1.46 | 3.98 | 3.56 | 69.86 | |
| NO Hep. B Vaccine | M | 94.25 | 829.75 | 5.73 | 3.54 | 7.50 | 6.75 | 9.75 | 7.00 | 78.75 |
| SD | 2.64 | 112.14 | 0.65 | 0.53 | 2.52 | 1.26 | 5.12 | 3.74 | 51.11 | |
| Hepatitis B Vaccine | M | 95.04 | 1165.88 | 5.24 | 3.11 | 13.38 | 9.38 | 16.88 | 12.50 | 241.88 |
| SD | 2.63 | 264.38 | 0.76 | 0.55 | 3.25 | 1.92 | 6.71 | 5.37 | 111.20 | |
| NO Birth Vaccines | M | 94.80 | 817.80 | 5.91 | 3.63 | 7.20 | 6.60 | 8.40 | 6.00 | 66.60 |
| SD | 2.60 | 100.73 | 0.70 | 0.50 | 2.28 | 1.14 | 5.37 | 3.94 | 51.94 | |
| Birth Vaccine | M | 94.67 | 1238.00 | 4.75 | 2.96 | 14.80 | 9.60 | 19.20 | 14.60 | 283.80 |
| SD | 3.10 | 202.41 | 0.28 | 0.24 | 1.10 | 1.67 | 3.90 | 3.36 | 60.06 | |
| Unclear | M | 95.00 | 1183.50 | 5.74 | 3.04 | 13.50 | 10.50 | 18.00 | 12.50 | 249.00 |
| SD | 2.36 | 376.89 | 0.09 | 1.00 | 2.12 | 0.71 | 5.66 | 4.95 | 114.55 | |
| Groups of countries | Vaccine exposure to 6 months | Average vaccines by 12 months | Average vaccines by 6 months | Average Doses by 12 months |
Average Doses by 6 months |
Monthly vaccine exposure to 12 months | Monthly vaccine exposure to 6 months | Vaccine exposure Index to 12 months | Vaccine exposure Index to 6 months | |
| LOWER Autism rate | M | 50.33 | 0.67 | 1.19 | 0.78 | 1.08 | 6.96 | 8.39 | 1.25 | 2.83 |
| SD | 33.38 | 0.24 | 0.29 | 0.44 | 0.62 | 5.19 | 5.56 | 0.95 | 2.32 | |
| HIGHER Autism rate | M | 145.00 | 1.24 | 1.64 | 1.64 | 2.47 | 24.29 | 24.17 | 5.00 | 7.91 |
| SD | 34.77 | 0.08 | 0.27 | 0.31 | 0.51 | 4.74 | 5.80 | 1.18 | 2.18 | |
| NO Varicella vaccine | M | 42.40 | 0.60 | 1.10 | 0.70 | 1.00 | 5.55 | 7.07 | 1.01 | 2.11 |
| SD | 30.34 | 0.19 | 0.19 | 0.45 | 0.66 | 4.33 | 5.06 | 0.84 | 1.68 | |
| Varicella Vaccine | M | 137.14 | 1.20 | 1.64 | 1.57 | 2.33 | 22.82 | 22.86 | 4.63 | 7.70 |
| SD | 37.95 | 0.12 | 0.24 | 0.33 | 0.59 | 5.82 | 6.32 | 1.46 | 2.07 | |
| NO Hep. B Vaccine | M | 50.00 | 0.63 | 1.13 | 0.81 | 1.17 | 6.56 | 8.33 | 1.21 | 2.52 |
| SD | 29.02 | 0.21 | 0.21 | 0.43 | 0.62 | 4.26 | 4.84 | 0.83 | 1.64 | |
| Hepatitis B Vaccine | M | 121.50 | 1.11 | 1.56 | 1.41 | 2.08 | 20.16 | 20.25 | 4.08 | 6.80 |
| SD | 56.50 | 0.27 | 0.32 | 0.56 | 0.90 | 9.27 | 9.42 | 2.06 | 3.18 | |
| NO Birth Vaccines | M | 42.40 | 0.60 | 1.10 | 0.70 | 1.00 | 5.55 | 7.07 | 1.01 | 2.11 |
| SD | 30.34 | 0.19 | 0.19 | 0.45 | 0.66 | 4.33 | 5.06 | 0.84 | 1.68 | |
| Birth Vaccine | M | 138.80 | 1.23 | 1.60 | 1.60 | 2.43 | 23.65 | 23.13 | 5.03 | 7.93 |
| SD | 34.97 | 0.09 | 0.28 | 0.32 | 0.56 | 5.01 | 5.83 | 1.32 | 2.44 | |
| Unclear | M | 133.00 | 1.13 | 1.75 | 1.50 | 2.08 | 20.75 | 22.17 | 3.63 | 7.13 |
| SD | 60.81 | 0.18 | 0.12 | 0.47 | 0.82 | 9.55 | 10.14 | 1.72 | 0.98 |
| Levene’s Test for Equality of Variances | t-test for Equality of Means | |||||
| Variables | Equal Variances | F | Sig. | t-test | Degrees of Freedom | Sig. (2-tailed) |
| Vaccination Rate Total |
assumed | 0.394 | 0.544 | -0.282 | 10 | 0.784 |
| not assumed | -0.282 | 9.679 | 0.784 | |||
| Autism rate 100k | assumed | 3.81 | 0.079 | -5.133 | 10 | 0.001 |
| not assumed | -5.133 | 9.246 | 0.001 | |||
| Average Age Infants vaccinated to 12 months | assumed | 0.871 | 0.373 | 3.224 | 10 | 0.009 |
| not assumed | 3.224 | 9.822 | 0.009 | |||
| Average Age Infants vaccinated to 6 months | assumed | 2.962 | 0.116 | 0.854 | 10 | 0.413 |
| not assumed | 0.854 | 7.807 | 0.418 | |||
| Total Vaccines done to 12 months (V12) | assumed | 10.742 | 0.008 | -4.654 | 10 | 0.001 |
| not assumed | -4.654 | 5.4 | 0.005 | |||
| Total Vaccines done to 6 months (V6) | assumed | 0.214 | 0.654 | -2.78 | 10 | 0.019 |
| not assumed | -2.78 | 8.933 | 0.022 | |||
| Total Doses done to 12 months (D12) | assumed | 13.241 | 0.005 | -2.927 | 10 | 0.015 |
| not assumed | -2.927 | 5.704 | 0.028 | |||
| Total Doses done to 6 months (D6) | assumed | 13.914 | 0.004 | -3.09 | 10 | 0.011 |
| not assumed | -3.09 | 5.817 | 0.022 | |||
| Vaccine exposure done to 12 months (V12×D12) | assumed | 12.262 | 0.006 | -3.691 | 10 | 0.004 |
| not assumed | -3.691 | 5.391 | 0.012 | |||
| Vaccine exposure done to 6 months (V6×D6) | assumed | 14.469 | 0.003 | -3.282 | 10 | 0.008 |
| not assumed | -3.282 | 5.641 | 0.018 | |||
| Average Vaccines done by 12 months | assumed | 10.742 | 0.008 | -4.654 | 10 | 0.001 |
| not assumed | -4.654 | 5.4 | 0.005 | |||
| Average Vaccines done by 6 months | assumed | 0.214 | 0.654 | -2.78 | 10 | 0.019 |
| not assumed | -2.78 | 8.933 | 0.022 | |||
| Average Doses done by 12 months | assumed | 13.241 | 0.005 | -2.927 | 10 | 0.015 |
| not assumed | -2.927 | 5.704 | 0.028 | |||
| Average Doses done by 6 months | assumed | 13.914 | 0.004 | -3.09 | 10 | 0.011 |
| not assumed | -3.09 | 5.817 | 0.022 | |||
| Monthly vaccine exposure to 12 months | assumed | 12.262 | 0.006 | -3.691 | 10 | 0.004 |
| not assumed | -3.691 | 5.391 | 0.012 | |||
| Monthly vaccine exposure to 6 months | assumed | 14.469 | 0.003 | -3.282 | 10 | 0.008 |
| not assumed | -3.282 | 5.641 | 0.018 | |||
| Age-Adjusted Vaccine exposure Index to 12 months | assumed | 13.406 | 0.004 | -3.767 | 10 | 0.004 |
| not assumed | -3.767 | 5.426 | 0.011 | |||
| Age-Adjusted Vaccine exposure Index to 6 months | assumed | 11.263 | 0.007 | -2.975 | 10 | 0.014 |
| not assumed | -2.975 | 5.541 | 0.027 | |||
|
Control Variables: General vaccination rate in population |
A | B | C | D | E | F | G | H | I | L | M | ||
| Autism Rate | A | r | 1 | -0.71 | -0.39 | 0.87 | 0.77 | 0.79 | 0.80 | 0.85 | 0.83 | 0.85 | 0.80 |
| sig. | . | 0.007 | 0.117 | 0 | 0.003 | 0.002 | 0.001 | 0 | 0.001 | 0.001 | 0.001 | ||
| Average Age infants by 12 months | B | r | 1 | 0.60 | -0.78 | -0.57 | -0.84 | -0.84 | -0.85 | -0.81 | -0.88 | -0.82 | |
| sig. | 0.025 | 0.002 | 0.033 | 0.001 | 0.001 | 0.001 | 0.001 | 0 | 0.001 | ||||
| Average Age infants by 6 months | C | r | 1 | -0.62 | -0.52 | -0.64 | -0.60 | -0.65 | -0.60 | -0.66 | -0.70 | ||
| sig. | 0.022 | 0.052 | 0.017 | 0.026 | 0.015 | 0.025 | 0.014 | 0.008 | |||||
| Average Vaccines infants by 12 months | D | r | 1 | 0.91 | 0.86 | 0.85 | 0.94 | 0.90 | 0.94 | 0.90 | |||
| sig. | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||||||
| Average Vaccines infants by 6 months | E | r | 1 | 0.81 | 0.80 | 0.88 | 0.88 | 0.85 | 0.87 | ||||
| sig. | 0.001 | 0.002 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||
| Average Doses infants by 12 months | F | r | 1 | 1.00 | 0.98 | 0.99 | 0.98 | 0.99 | |||||
| sig. | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | ||||||||
| Average Doses infants by 6 months | G | r | 1 | 0.97 | 0.99 | 0.97 | 0.98 | ||||||
| sig. | 0.00 | 0.00 | 0.00 | 0.00 | |||||||||
| Vaccine exposure to 12 months | H | r | 1 | 0.99 | 1.00 | 0.99 | |||||||
| sig. | 0.00 | 0.00 | 0.00 | ||||||||||
| Vaccine exposure to 6 months | I | r | 1 | 0.98 | 0.99 | ||||||||
| sig. | 0.00 | 0.00 | |||||||||||
| Age-Adjusted Vaccine exposure Index to 12 months | L | r | 1 | 0.99 | |||||||||
| sig. | 0.00 | ||||||||||||
| Age-Adjusted Vaccine exposure Index to 6 months | M | r | 1 | ||||||||||
| sig. | . |
| Simple regression | Explanatory variable: Monthly vaccine exposure in infants to 1 year | ||||
| Dependent variable | Constant, α | Coefficient, β1 | Stand. Coeff. Beta | R2 | F |
| Autism rate 2021 per 100k | 6.16*** | 0.16* | 0.68 | 0.46 | 8.38* |
| Explanatory variable: Index vaccine exposure (VEI12) in infants to 1 year | |||||
| Constant, α | Coefficient, β1 | Stand. Coeff. Beta | R2 | F | |
| Autism rate 2021 per 100k | 6.83*** | 0.14* | 0.67 | 0.46 | 8.34* |
| Multivariate regression | Response variable: Autism rate 2021 per 100k | ||||
| Predictors | |||||
| Constant | −18.24* | ||||
| Vaccine exposure index in infants to 1 year (VEI12) | 0.16*** | ||||
| Vaccination rate for Diphtheria, Tetanus, and Pertussis, Measles and Polio diseases | 5.51** | ||||
| R2 | 0.78 | ||||
| F | 16.31*** | ||||
| Bootstrap for Coefficients | Bootstrap a) | BCa 95% Confidence Interval | |||||
| B | Bias | Std. Error | Sig. (2-tailed) |
Lower | Upper | ||
| (Constant) | 6.83 | -0.032 | 0.101 | <.001 | 6.631 | 6.902 | |
| Index vaccine exposure (VEI12) in infants to 1 year | 0.143 | 0.021 | 0.075 | 0.048 | 0.058 | 0.469 | |
| China | India | |
| Total Vaccines | 10 | 14 |
| Total Doses | 12 | 19 |
| Vaccination Coverage DTP3, POL3 | 97% | 91% |
| Vaccination Coverage MCV1 | 97% | 93% |
| Autism rate 2023 per 100k | 484 | 509 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




