Submitted:
01 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
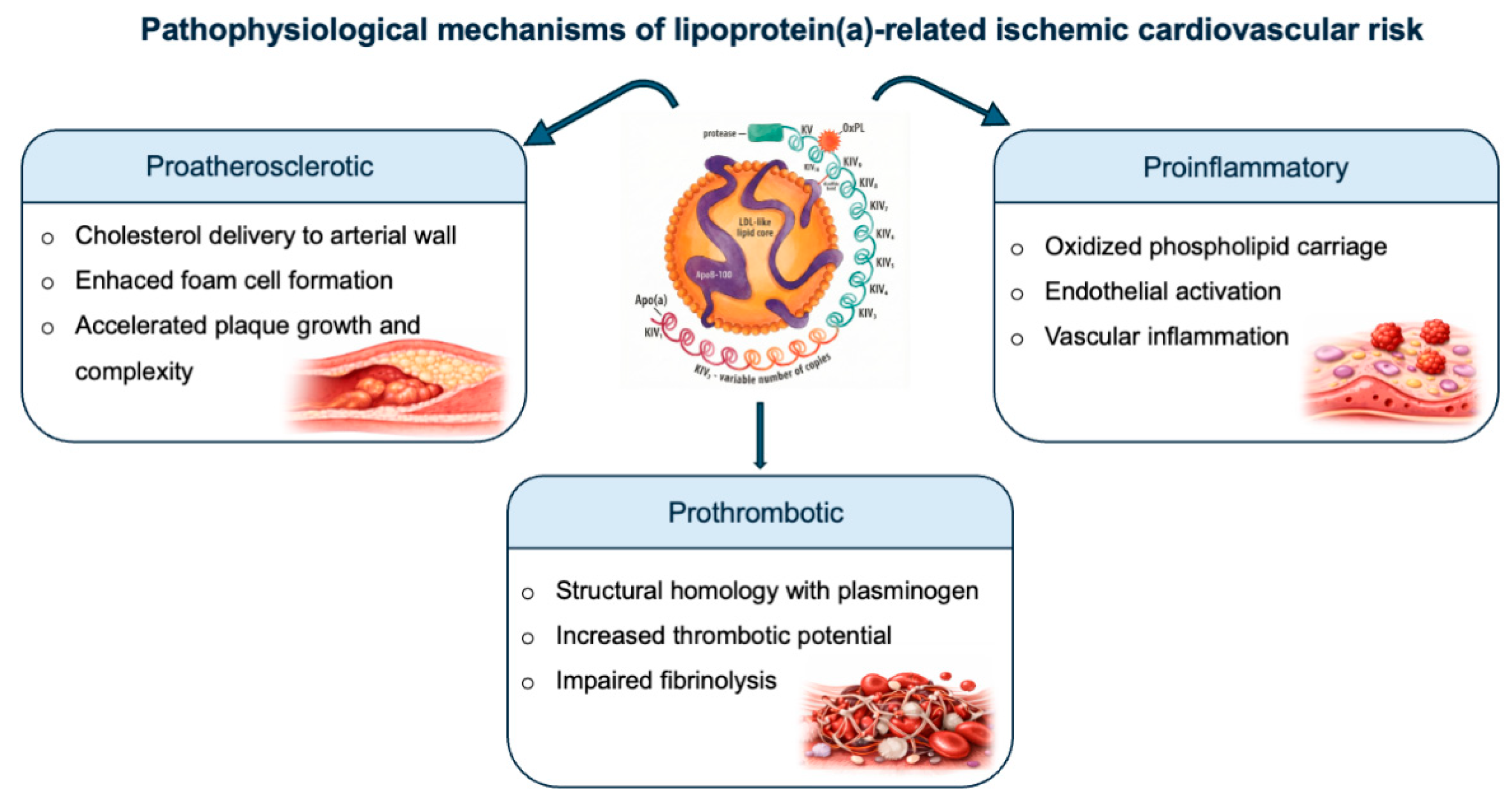
2. Lipoprotein(a): Structure, Metabolism and Pathophysiological Mechanisms
3. Genetic Determinants and Variability of Lipoprotein(a)
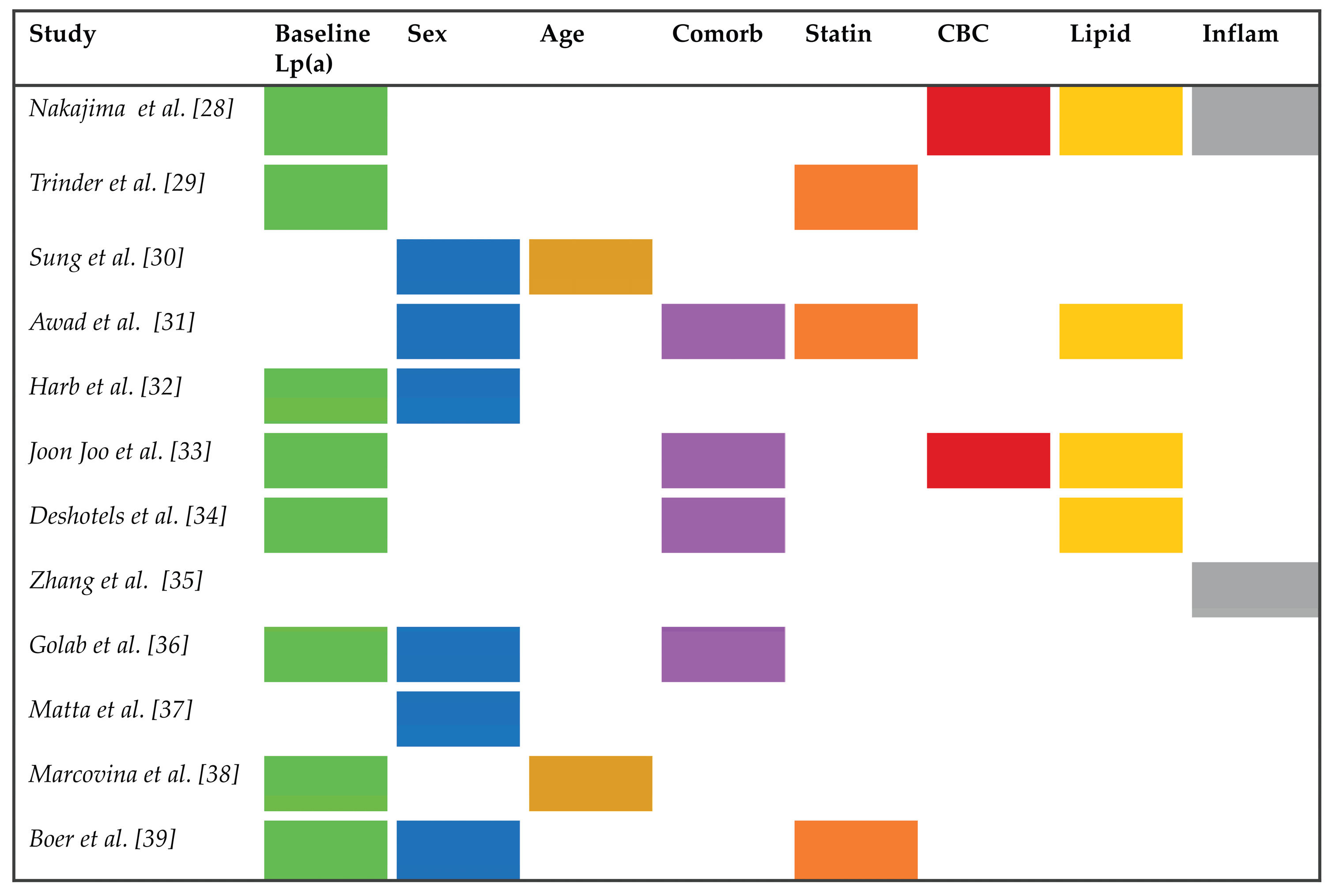
3.1. Intra-Individual Variability
4. Association Between Elevated Lipoprotein(a) Levels and Atherosclerotic Cardiovascular Disease
5. Lipoprotein(a) and Residual Cardiovascular Risk
5.1. Lipoprotein(a) as a Driver of Residual Cardiovascular Risk
5.2. Optimal Timing for Lipoprotein(a) Assessment After Acute Coronary Syndrome
6. Therapeutic Strategies Targeting Lipoprotein(a)
6.1. Current Management Strategies
6.2. Emerging Targeted Therapies
7. Clinical Practice Guidelines and Consensus Recommendations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACS | Acute Coronary Syndrome |
| MACE | Major Adverse Cardiovascular Events |
| LDL-C | Low-Density Lipoprotein Cholesterol |
| Lp(a) | Lipoprotein(a) |
| ASCVD | Atherosclerotic Cardiovascular Disease |
| RCT | Randomized Clinical Trial |
| PCSK9 | Proprotein Convertase Subtilisin/Kexin Type 9 |
| CAVD | Calcific Aortic Valvular Disease |
| ESC | European Society of Cardiology |
| EAS | European Atherosclerosis Society |
| AHA | American Heart Association |
| ASO | Antisense Oligonucleotide |
| siRNA | Small Interfering RNA |
| ApoB-100 | Apolipoprotein B-100 |
| Apo(a) | Apolipoprotein(a) |
| KIV | Kringle IV |
| CAD | Coronary Artery Disease |
| CNV | Copy Number Variation |
| HDL-C | High-Density Lipoprotein Cholesterol |
| HsCRP | High-Sensitivity C-Reactive Protein |
| HR | Hazard Ratio |
| CI | Confidence Interval |
| TVD | Three-Vessel Disease |
| MI | Myocardial Infarction |
| TG | Triglycerides |
| PCI | Percutaneous Coronary Intervention |
| MALE | Major Adverse Limb Events |
| STEMI | ST-Segment Elevation Myocardial Infarction |
| CKD | Chronic Kidney Disease |
| mRNA | Messenger RNA |
| PAD | Peripheral Artery Disease |
| RISC | RNA-Induced Silencing Complex |
References
- Steen, D.L.; Khan, I.; Andrade, K.; Koumas, A.; Giugliano, R.P. Event rates and risk factors for recurrent cardiovascular events and mortality in a contemporary post-acute coronary syndrome population representing 239,234 patients during 2005 to 2018 in the United States. J. Am. Heart Assoc. 2022, 11, e024200. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Steg, P.G.; Szarek, M.; Bhatt, D.L.; Bittner, V.A.; Diaz, R.; Edelberg, J.M.; Leiter, L.A.; Kim, J.H.; Kuder, J.F.; et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N. Engl. J. Med. 2018, 379, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- Lawler, P.R.; Bhatt, D.L.; Godoy, L.C.; Lüscher, T.F.; Bonow, R.O.; Verma, S.; Ridker, P.M. Targeting cardiovascular inflammation: next steps in clinical translation. Eur. Heart J. 2021, 42, 113–131. [Google Scholar] [CrossRef] [PubMed]
- Sabouret, P.; Angoulvant, D.; Pathak, A.; Fysekidis, M.; Laterra, G.; Costa, F.; Angelini, F.; Bocchino, P.P.; Montalescot, G.; Biondi-Zoccai, G. How to fill the GAPS-I in secondary prevention: Application of a strategy based on GLP1 analogues, antithrombotic agents, PCSK9 inhibitors, SGLT2 inhibitors and immunomodulators. Panminerva Med. 2022, 64, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G.; Langsted, A. Lipoprotein(a) and cardiovascular disease. Lancet 2024, 404, 1255–1264. [Google Scholar] [CrossRef]
- Tsioulos, G.; Kounatidis, D.; Vallianou, N.G.; Poulaki, A.; Kotsi, E.; Christodoulatos, G.S.; Tsilingiris, D.; Karampela, I.; Skourtis, A.; Dalamaga, M. Lipoprotein(a) and Atherosclerotic Cardiovascular Disease: Where Do We Stand? Int. J. Mol. Sci. 2024, 25, 3537. [Google Scholar] [CrossRef]
- Duarte Lau, F.; Giugliano, R.P. Lipoprotein(a) and its significance in cardiovascular disease: a review. JAMA Cardiol. 2022, 7, 760–769. [Google Scholar] [CrossRef]
- Mach, F.; Koskinas, K.C.; Roeters van Lennep, J.E.; Tokgözoğlu, L.; Badimon, L.; Baigent, C.; Binder, C.J.; Catapano, A.L.; De Backer, G.; Delgado, V.; et al. 2025 focused update of the 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur. Heart J. 2025, 46, 4359–4378. [Google Scholar] [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC Guideline on the management of blood cholesterol. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef]
- Mojahedi, A.; Chen, O.; Skopicki, H.A.; Rahman, T.; Sadeghian, A. Lipoprotein(a) as a predictor of cardiovascular risk in acute coronary syndrome patients undergoing percutaneous coronary intervention: a systematic review. Rev. Cardiovasc. Med. 2025, 26, 42784. [Google Scholar] [CrossRef]
- Mansoor, T.; Ismayl, M.; Parikh, S.; Nambi, V.; Virani, S.S.; Mehta, A.; Jia, X.; Minhas, A.M.K. Emerging pharmacological strategies in lipoprotein(a) reduction. Proc. (Baylor Univ. Med. Cent.) 2025, 38, 783–786. [Google Scholar] [CrossRef] [PubMed]
- Berg, K. A new serum type system in man—the LP system. Acta Pathol. Microbiol. Scand. 1963, 59, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.; Mohr, J. Genetics of the LP system. Acta Genet. Stat. Med. 1963, 13, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, K. Structure, function, and metabolism of lipoprotein(a). J. Lipid Res. 2016, 57, 1339–1359. [Google Scholar] [CrossRef]
- Kratzin, H.; Armstrong, V.W.; Niehaus, M. Structural relationship of an apolipoprotein(a) phenotype (570 kDa) to plasminogen: homologous kringle domains are linked by carbohydrate-rich regions. Biol. Chem. Hoppe Seyler 1987, 368, 1533–1544. [Google Scholar] [CrossRef]
- Boffa, M.B. Beyond fibrinolysis: the confounding role of Lp(a) in thrombosis. Atherosclerosis 2022, 349, 72–81. [Google Scholar] [CrossRef]
- Tsimikas, S.; Hall, J.L. Lipoprotein(a) as a Potential Causal Genetic Risk Factor of Cardiovascular Disease: A Rationale for Increased Efforts to Understand its Pathophysiology and Develop Targeted Therapies. J. Am. Coll. Cardiol. 2012, 60, 716–721. [Google Scholar] [CrossRef]
- Koutsogianni, A.D.; Liberopoulos, E.; Tellis, K.; Tselepis, A.D. Oxidized phospholipids and lipoprotein(a): An update. Eur. J. Clin. Invest. 2022, 52, e13710. [Google Scholar] [CrossRef]
- Boffa, M.B.; Koschinsky, M.L. Lipoprotein(a): truly a direct prothrombotic factor in cardiovascular disease? J. Lipid Res. 2016, 57, 745–757. [Google Scholar] [CrossRef]
- Tasdighi, E.; Adhikari, R.; Almaadawy, O.; Leucker, T.M.; Blaha, M.J. Lp(a): Structure, Genetics, Associated Cardiovascular Risk, and Emerging Therapeutics. Annu. Rev. Pharmacol. Toxicol. 2024, 64, 135–157. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Chapman, M.J.; Ray, K.; Borén, J.; Andreotti, F.; Watts, G.F.; Ginsberg, H.; Amarenco, P.; Catapano, A.; Descamps, O.S.; et al. Lipoprotein(a) as a cardiovascular risk factor: current status. Eur. Heart J. 2010, 31, 2844–2853. [Google Scholar] [CrossRef] [PubMed]
- Kamstrup, P.R.; Tybjærg-Hansen, A.; Nordestgaard, B.G. Elevated Lipoprotein(a) and Risk of Aortic Valve Stenosis in the General Population. J. Am. Coll. Cardiol. 2014, 63, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Volgman, A.S.; Koschinsky, M.L.; Mehta, A.; Rosenson, R.S. Genetics and Pathophysiological Mechanisms of Lipoprotein(a)-Associated Cardiovascular Risk. J. Am. Heart Assoc. 2024, 13, e033654. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Soffer, G.; Ginsberg, H.N.; Berglund, L.; Duell, P.B.; Heffron, S.P.; Kamstrup, P.R.; Lloyd-Jones, D.M.; Marcovina, S.M.; Yeang, C.; Koschinsky, M.L. Lipoprotein(a): A Genetically Determined, Causal, and Prevalent Risk Factor for Atherosclerotic Cardiovascular Disease: A Scientific Statement From the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 2022, 42, e48–e60. [Google Scholar] [CrossRef]
- Nakajima, K.; Hata, Y. Intraindividual Variations in Lipoprotein(a) Levels and Factors Related to These Changes. J. Atheroscler. Thromb. 1996, 2, 96–106. [Google Scholar] [CrossRef]
- Trinder, M.; Paruchuri, K.; Haidermota, S.; Bernardo, R.; Zekavat, S.M.; Gilliland, T.; Januzzi, J.L., Jr.; Natarajan, P. Repeat Measures of Lipoprotein(a) Molar Concentration and Cardiovascular Risk. J. Am. Coll. Cardiol. 2022, 79, 617–628. [Google Scholar] [CrossRef]
- Sung, D.E.; Lee, M.Y.; Kwon, M.J.; Sung, K.C. Longitudinal changes and borderline reclassification of Lipoprotein(a) compared with conventional lipids in over 230,000 adults. Atherosclerosis. 2025, 410, 120524. [Google Scholar] [CrossRef]
- Awad, K.; Mahmoud, A.K.; Abbas, M.T.; Alsidawi, S.; Ayoub, C.; Arsanjani, R.; Farina, J.M. Intra-individual variability in lipoprotein(a) levels: findings from a large academic health system population. Eur. J. Prev. Cardiol. 2025, 32, 716–721. [Google Scholar] [CrossRef]
- Harb, T.; Ziogos, E.; Blumenthal, R.S.; Gerstenblith, G.; Leucker, T.M. Intra-individual variability in lipoprotein(a): the value of a repeat measure for reclassifying individuals at intermediate risk. Eur. Heart J. Open. 2024, 4, oeae064. [Google Scholar] [CrossRef]
- Joo, H.J.; Yun, S.G.; Park, J.H.; Hong, S.J.; Yu, C.W.; Shin, S.Y.; Kim, E.J. Predictors of lipoprotein(a) variability in clinical practice and their impact on cardiovascular risk. Lipids Health Dis. 2025, 24, 250. [Google Scholar] [CrossRef]
- Deshotels, M.R.; Sun, C.; Nambi, V.; Virani, S.S.; Matsushita, K.; Yu, B.; Ballantyne, C.M.; Hoogeveen, R.C. Temporal Trends in Lipoprotein(a) Concentrations: The Atherosclerosis Risk in Communities Study. J. Am. Heart Assoc. 2022, 11, e026762. [Google Scholar] [CrossRef]
- Zhang, S.S.; Hu, W.Y.; Li, Y.J.; Yu, J.; Sang, S.; Alsalman, Z.M.; Xie, D.Q. Lipoprotein(a) variability is associated with mean follow-up C-reactive protein in patients with coronary artery disease following percutaneous coronary intervention. World J. Clin. Cases. 2022, 10, 12909–12919. [Google Scholar] [CrossRef] [PubMed]
- Gołąb, A.M.; Konieczyńska, M.; Ząbczyk, M.; Undas, A.; Natorska, J. Intraindividual variability of lipoprotein(a) in the middle-aged population: the impact of smoking. Pol. Arch. Intern. Med. 2024, 134, 16889. [Google Scholar] [CrossRef] [PubMed]
- Matta, M.G.; Schreier, L.; Lavalle-Cobo, A.; Garcia-Zamora, S.; Ferraresi, A.; Madsen, A.; Bellini, S.; Ramos, G.; Roubicek, P.; Corral, P. Temporal variability of Lp(a) in clinically stable patients: implications for cardiovascular risk assessment. Med. Clin. (Barc). 2024, 163, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Marcovina, S.M.; Viney, N.J.; Hughes, S.G.; Xia, S.; Witztum, J.L.; Tsimikas, S. Temporal variability in lipoprotein(a) levels in patients enrolled in the placebo arms of IONIS-APO(a)Rx and IONIS-APO(a)-LRx antisense oligonucleotide clinical trials. J. Clin. Lipidol. 2018, 12, 122–129.e2. [Google Scholar] [CrossRef]
- de Boer, L.M.; Hof, M.H.; Wiegman, A.; Stroobants, A.K.; Kastelein, J.J.P.; Hutten, B.A. Lipoprotein(a) levels from childhood to adulthood: data in nearly 3,000 children who visited a pediatric lipid clinic. Atherosclerosis 2022, 349, 227–232. [Google Scholar] [CrossRef]
- Patel, A.P.; Wang, M.; Pirruccello, J.P.; Ellinor, P.T.; Ng, K.; Kathiresan, S.; Khera, A.V. Lp(a) (Lipoprotein[a]) concentrations and incident atherosclerotic cardiovascular disease: New insights from a large national biobank. Arterioscler. Thromb. Vasc. Biol. 2021, 41, 465–474. [Google Scholar] [CrossRef]
- Amiri, M.; Raeisi-Dehkordi, H.; Verkaar, A.J.C.F.; Wu, Y.; van Westing, A.C.; Berk, K.A.; Bramer, W.M.; Aune, D.; Voortman, T. Circulating lipoprotein(a) and all-cause and cause-specific mortality: A systematic review and dose–response meta-analysis. Eur. J. Epidemiol. 2023, 38, 485–499. [Google Scholar] [CrossRef]
- Bhatia, H.S.; Wandel, S.; Willeit, P.; Lesogor, A.; Bailey, K.; Ridker, P.M.; Nestel, P.; Simes, J.; Tonkin, A.; Schwartz, G.G.; et al. Independence of lipoprotein(a) and low-density lipoprotein cholesterol–mediated cardiovascular risk: A participant-level meta-analysis. Circulation 2025, 151, 312–321. [Google Scholar] [CrossRef]
- Tian, X.; Zhang, N.; Tse, G.; Li, G.; Sun, Y.; Liu, T. Association between lipoprotein(a) and premature atherosclerotic cardiovascular disease: A systematic review and meta-analysis. Eur. Heart J. Open. 2024, 4, oeae031. [Google Scholar] [CrossRef]
- Kronenberg, F.; Mora, S.; Stroes, E.S.G.; Ference, B.A.; Arsenault, B.J.; Berglund, L.; Dweck, M.R.; Koschinsky, M.; Lambert, G.; Mach, F.; et al. Lipoprotein(a) in atherosclerotic cardiovascular disease and aortic stenosis: A European Atherosclerosis Society consensus statement. Eur. Heart J. 2022, 43, 3925–3946. [Google Scholar] [CrossRef]
- Langsted, A.; Kamstrup, P.R.; Nordestgaard, B.G. High lipoprotein(a) and high risk of mortality. Eur. Heart J. 2019, 40, 2760–2770. [Google Scholar] [CrossRef] [PubMed]
- Arnold, N.; Blaum, C.; Goßling, A.; Brunner, F.J.; Bay, B.; Zeller, T.; Ferrario, M.M.; Brambilla, P.; Cesana, G.; Leoni, V.; et al. Impact of lipoprotein(a) level on low-density lipoprotein cholesterol– or apolipoprotein B–related risk of coronary heart disease. J. Am. Coll. Cardiol. 2024, 84, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Björnson, E.; Adiels, M.; Taskinen, M.R.; Burgess, S.; Chapman, M.J.; Packard, C.J.; Borén, J. Lipoprotein(a) is markedly more atherogenic than LDL: an apolipoprotein B-based genetic analysis. J. Am. Coll. Cardiol. 2024, 83, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Wang, Z.; Dong, J.; Yu, M.; Zhou, Y. Lipoprotein(a) and panvascular disease. Lipids Health Dis. 2025, 24, 186. [Google Scholar] [CrossRef]
- Wang, Z.; Xiao, S.; Liu, N. Association of lipoprotein(a) with coronary severity in patients with new-onset acute myocardial infarction: A large cross-sectional study. Clin. Chim. Acta. 2023, 540, 117220. [Google Scholar] [CrossRef]
- Kozieł-Siołkowska, M.; Mitręga, K.; Podolecki, T.; Olma, A.; Kalarus, Z.; Streb, W. Lipoprotein(a) as an independent predictor of elevated SYNTAX score. J. Clin. Med. 2024, 13, 7109. [Google Scholar] [CrossRef]
- Rallidis, L.S.; Pavlakis, G.; Foscolou, A.; Kotakos, C.; Katsimardos, A.; Drosatos, A.; Zolindaki, M.; Panagiotakos, D.B. High levels of lipoprotein(a) and premature acute coronary syndrome. Atherosclerosis. 2018, 269, 29–34. [Google Scholar] [CrossRef]
- Fan, W.; Wu, C.; Wong, N.D. Lipoprotein(a) atherosclerotic cardiovascular disease risk score development and prediction in primary prevention from real-world data. Circ. Genom. Precis. Med. 2025, 18, e004631. [Google Scholar] [CrossRef]
- Cordero, A.; Salinas, J.M.; Quintanilla, M.A.; López-Ayala, J.M.; Blasco, Á.; Flores, E. Reclassification of low or intermediate cardiovascular risk by determining lipoprotein(a) levels. Biomedicines. 2025, 13, 2648. [Google Scholar] [CrossRef]
- Patel, K.V.; Pandey, A.; de Lemos, J.A. Conceptual framework for addressing residual atherosclerotic cardiovascular disease risk in the era of precision medicine. Circulation. 2018, 137, 2551–2553. [Google Scholar] [CrossRef]
- Hoogeveen, R.C.; Ballantyne, C.M. Residual cardiovascular risk at low LDL: Remnants, lipoprotein(a), and inflammation. Clin. Chem. 2021, 67, 143–153. [Google Scholar] [CrossRef]
- Katsi, V.; Argyriou, N.; Fragoulis, C.; Tsioufis, K. The Role of Non-HDL Cholesterol and Apolipoprotein B in Cardiovascular Disease: A Comprehensive Review. J. Cardiovasc. Dev. Dis. 2025, 12, 256. [Google Scholar] [CrossRef] [PubMed]
- Willeit, P.; Ridker, P.M.; Nestel, P.J.; Simes, J.; Tonkin, A.M.; Pedersen, T.R.; Schwartz, G.G.; Olsson, A.G.; Colhoun, H.M.; Kronenberg, F.; et al. Baseline and on-statin treatment lipoprotein(a) levels for prediction of cardiovascular events: Individual patient-data meta-analysis of statin outcome trials. Lancet. 2018, 392, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Liu, H.; Yu, X.; Tong, L.; Chen, X.; Qi, L.; Cui, C.; Cheng, L.; Cai, L.; et al. Elevated lipoprotein(a) levels as an independent predictor of long-term recurrent events in patients with acute coronary syndrome: an observational, retrospective cohort study. Coron. Artery Dis. 2022, 33, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Dai, K.; Shiode, N.; Yoshii, K.; Kimura, Y.; Matsuo, K.; Jyuri, Y.; Tomomori, S.; Higaki, T.; Oi, K.; Kawase, T.; et al. Impact of Lipoprotein(a) on Long-Term Outcomes in Patients With Acute Myocardial Infarction. Circ. J. 2023, 87, 1356–1361. [Google Scholar] [CrossRef]
- Miñana, G.; Cordero, A.; Fácila, L.; Company, M.; Fernández-Cisnal, A.; Valero, E.; Carratalá, A.; Navarro, J.; Llergo, J.T.; Fernández-Olmo, R.; et al. Lipoprotein(a) and long-term recurrent infarction after an acute myocardial infarction. Am. J. Cardiol. 2024, 211, 9–16. [Google Scholar] [CrossRef]
- Takahashi, D.; Wada, H.; Ogita, M.; Yasuda, K.; Nishio, R.; Takeuchi, M.; Shitara, J.; Tsuboi, S.; Dohi, T.; Suwa, S.; et al. Impact of Lipoprotein(a) as a Residual Risk Factor in Long-Term Cardiovascular Outcomes in Patients With Acute Coronary Syndrome Treated With Statins. Am. J. Cardiol. 2022, 168, 11–16. [Google Scholar] [CrossRef]
- Steg, P.G.; Szarek, M.; Jukema, J.W.; Bhatt, D.L.; Bittner, V.A.; Diaz, R.; Fazio, S.; Garon, G.; Goodman, S.G.; Harrington, R.A.; et al. Relation of Low-Density Lipoprotein Cholesterol, High-Sensitivity C-Reactive Protein, and Lipoprotein(a) Each to Future Cardiovascular Events and Death After Acute Coronary Syndrome on High-Intensity Statin Therapy: An Analysis of the Placebo Arm of ODYSSEY OUTCOMES. Circulation 2025, 151, 1047–1050. [Google Scholar] [CrossRef]
- Schwartz, G.G.; Ballantyne, C.M.; Barter, P.J.; Davidson, M.H.; Kallend, D.; Leiter, L.A.; Sisk, C.; Tershakovec, A.M.; Wright, R.S.; et al. Association of Lipoprotein(a) With Risk of Recurrent Ischemic Events Following Acute Coronary Syndrome: Analysis of the dal-Outcomes Randomized Clinical Trial. JAMA Cardiol. 2018, 3, 164–168. [Google Scholar] [CrossRef]
- Park, J.S.; Cho, K.H.; Hong, Y.J.; Kim, M.C.; Sim, D.S.; Kim, J.H.; Ahn, Y.; Jeong, M.H.; et al. Baseline Lipoprotein(a) Levels and Long-Term Cardiovascular Outcomes After Acute Myocardial Infarction. J. Korean Med. Sci. 2023, 38, e102. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, H.H.; Jin, J.L.; Yan, X.N.; Dong, Q.; Li, J.J.; Wang, Y.; Chen, L.; Zhao, H.; Li, X.; et al. Lipoprotein(a) and cardiovascular death in oldest-old (≥80 years) patients with acute myocardial infarction: A prospective cohort study. Atherosclerosis 2020, 312, 54–59. [Google Scholar] [CrossRef]
- Li, N.; Zhou, J.; Chen, R.; Zhao, X.; Li, J.; Zhou, P.; Liu, C.; Chen, Y.; Wang, Y.; Song, L.; et al. Prognostic impacts of diabetes status and lipoprotein(a) levels in patients with ST-segment elevation myocardial infarction: a prospective cohort study. Cardiovasc. Diabetol. 2023, 22, 151. [Google Scholar] [CrossRef]
- Bittner, V.A.; Szarek, M.; Aylward, P.E.; Bhatt, D.L.; Diaz, R.; Edelberg, J.M.; Fras, Z.; Goodman, S.G.; Halvorsen, S.; Hanotin, C.; et al. Effect of Alirocumab on Lipoprotein(a) and Cardiovascular Risk After Acute Coronary Syndrome. J. Am. Coll. Cardiol. 2020, 75, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Xia, M.; Liang, C.; Pu, F.; Liu, S.; Jia, D.; Zhang, Y.; Li, H.; Chen, X.; Zhao, Q.; et al. Prognostic value of elevated lipoprotein(a) in patients with acute coronary syndromes: a systematic review and meta-analysis. Front. Cardiovasc. Med. 2024, 11, 1362893. [Google Scholar] [CrossRef] [PubMed]
- Rigattieri, S.; Cristiano, E.; Tempestini, F.; Lo Monaco, M.; Cava, F.; Bongiovanni, M.; Tifi, P.; Berni, A.; Volpe, M.; Rossi, L.; et al. Lipoprotein(a) and the risk of recurrent events in patients with acute myocardial infarction treated by percutaneous coronary intervention. Minerva Cardiol. Angiol. 2023, 71, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Jian, S.; Zhou, W.; Zhou, Q.; Xiang, J.; Zhu, Y.; Xiang, Z.; Yang, H.; Liu, G.; Luo, S.; et al. Associations of Lipoprotein(a) With Coronary Atherosclerotic Burden and All-Cause Mortality in Patients With ST-Segment Elevation Myocardial Infarction Treated With Primary Percutaneous Coronary Intervention. Front. Cardiovasc. Med. 2021, 8, 638679. [Google Scholar] [CrossRef]
- Ray, K.K.; Szarek, M.; Bhatt, D.L.; Bittner, V.A.; Fazio, S.; Goodman, S.G.; Harrington, R.A.; Jukema, J.W.; White, H.D.; Steg, P.G.; et al. Lipoprotein(a) Levels and Time to Cardiovascular Benefit With Alirocumab in Patients With Recent Acute Coronary Syndrome. J. Am. Coll. Cardiol. 2025, 86, 2071–2076. [Google Scholar] [CrossRef]
- Gencer, B.; Rigamonti, F.; Nanchen, D.; Vuilleumier, N.; Kern, I.; Aghlmandi, S.; Klingenberg, R.; Räber, L.; Auer, R.; Carballo, D.; et al. Prognostic value of elevated lipoprotein(a) in patients with acute coronary syndromes. Eur. J. Clin. Invest. 2019, 49, e13117. [Google Scholar] [CrossRef]
- Roth, C.; Krychtiuk, K.A.; Gangl, C.; Schrutka, L.; Distelmaier, K.; Wojta, J.; Hengstenberg, C.; Berger, R.; Speidl, W.S.; Müller, P.; et al. Lipoprotein(a) plasma levels are not associated with survival after acute coronary syndromes: An observational cohort study. PLoS One 2020, 15, e0227054. [Google Scholar] [CrossRef]
- Hao, Y.; Yang, Y.; Wang, Y.; Li, J. Relationship between lipoprotein(a) and revascularization after percutaneous coronary intervention in type 2 diabetes mellitus patients with acute coronary syndrome. Curr. Med. Res. Opin. 2022, 38, 1663–1672. [Google Scholar] [CrossRef]
- Silverio, A.; Cancro, F.P.; Di Maio, M.; Bellino, M.; Esposito, L.; Centore, M.; Carrizzo, A.; Di Pietro, P.; Borrelli, A.; De Luca, G.; et al. Lipoprotein(a) levels and risk of adverse events after myocardial infarction in patients with and without diabetes. J. Thromb. Thrombolysis 2022, 54, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chen, Y.; Yu, L.; Zhu, L.; Wang, Z.; Jiao, S.; Wu, C.; Tu, Y.; Wu, Y.; Guo, Z.; et al. The relationship between lipoprotein(a) and cardiovascular events in acute coronary syndrome patients with and without chronic kidney disease. Atherosclerosis 2022, 349, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Xu, S.; Shen, J.; Sun, F. The nonlinear association between lipoprotein(a) and major adverse cardiovascular events in acute coronary syndrome patients with three-vessel disease. Sci. Rep. 2025, 15, 1720. [Google Scholar] [CrossRef] [PubMed]
- Wohlfahrt, P.; Jenča, D.; Melenovský, V.; Franeková, J.; Jabor, A.; Šramko, M.; Staněk, V.; Želízko, M.; Poledne, R.; Piťha, J.; et al. Very low lipoprotein(a) and increased mortality risk after myocardial infarction. Eur. J. Intern. Med. 2021, 91, 33–39. [Google Scholar] [CrossRef]
- Ziogos, E.; Vavuranakis, M.A.; Harb, T.; Foran, P.L.; Blaha, M.J.; Jones, S.R.; Lai, S.; Gerstenblith, G.; Leucker, T.M. Lipoprotein(a) concentrations in acute myocardial infarction patients are not indicative of levels at six month follow-up. Eur. Heart J. Open 2023, 3, oead035. [Google Scholar] [CrossRef]
- Vavuranakis, M.A.; Jones, S.R.; Ziogos, E.; Blaha, M.J.; Williams, M.S.; Foran, P.; Schindler, T.H.; Lai, S.; Schulman, S.P.; Gerstenblith, G.; et al. The trajectory of lipoprotein(a) during the peri- and early postinfarction period and the impact of proprotein convertase subtilisin/kexin type 9 inhibition. Am. J. Cardiol. 2022, 171, 1–6. [Google Scholar] [CrossRef]
- Saeki, Y.; Sawaguchi, J.; Akita, S.; Takamura, T.A.; Fujibayashi, K.; Wakasa, M.; Akao, H.; Kitayama, M.; Kawai, Y.; Kajinami, K. Initial decrease in the lipoprotein(a) level is a novel prognostic biomarker in patients with acute coronary syndrome. World J. Cardiol. 2024, 16, 329–338. [Google Scholar] [CrossRef]
- Greco, A.; Finocchiaro, S.; Spagnolo, M.; Faro, D.C.; Mauro, M.S.; Raffo, C.; Sangiorgio, G.; Imbesi, A.; Laudani, C.; Mazzone, P.M.; et al. Lipoprotein(a) as a Pharmacological Target: Premises, Promises, and Prospects. Circulation 2025, 151, 400–415. [Google Scholar] [CrossRef]
- Tsimikas, S.; Gordts, P.L.S.M.; Nora, C.; Yeang, C.; Witztum, J.L. Statin therapy increases lipoprotein(a) levels. Eur. Heart J. 2020, 41, 2275–2284. [Google Scholar] [CrossRef]
- de Boer, L.M.; Oorthuys, A.O.J.; Wiegman, A.; Langendam, M.W.; Kroon, J.; Spijker, R.; et al. Statin therapy and lipoprotein(a) levels: a systematic review and meta-analysis. Eur. J. Prev. Cardiol. 2022, 29, 779–792. [Google Scholar] [CrossRef]
- Ridker, P.M.; Lei, L.; Ray, K.K.; Ballantyne, C.M.; Bradwin, G.; Rifai, N. Effects of bempedoic acid on CRP, IL-6, fibrinogen and lipoprotein(a) in patients with residual inflammatory risk: a secondary analysis of the CLEAR harmony trial. J. Clin. Lipidol. 2023, 17, 167–175. [Google Scholar] [CrossRef]
- O’Donoghue, M.L.; Fazio, S.; Giugliano, R.P.; Stroes, E.S.G.; Kanevsky, E.; Gouni-Berthold, I.; Im, K.; Lira Pineda, A.; Wasserman, S.M.; Češka, R.; et al. Lipoprotein(a), PCSK9 inhibition, and cardiovascular risk: insights from the FOURIER trial. Circulation 2019, 139, 1483–1492. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, D.; Watts, G.F.; Robinson, J.G.; Minini, P.; Sasiela, W.J.; Edelberg, J.; Louie, M.J.; Raal, F.J. Effect of Alirocumab on Lipoprotein(a) Over ≥1.5 Years (from the Phase 3 ODYSSEY Program). Am. J. Cardiol. 2017, 119, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Khalil, I.; Rahman, M.T.; Hossain, I. The Impact of Inclisiran on Lipid Profiles in Adults with Hyperlipidemia: A Meta-Analysis and Meta-Regression of Randomized Controlled Trials. Am. J. Cardiol. 2025, 250, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, M.I.; Safiullah, M.; Farhan, K.; Ali, A.; Abdullah, S.; Ali, A.; Ahmed, R. Obicetrapib and its impact on lipid parameters: a comprehensive meta-analysis of the latest evidence. J. Clin. Lipidol. 2025, 19, 1536–1549. [Google Scholar] [CrossRef]
- Roeseler, E.; Julius, U.; Heigl, F.; Spitthoever, R.; Heutling, D.; Breitenberger, P.; Leebmann, J.; Klingel, R.; Hafner, G.; Pöss, J.; et al. Lipoprotein apheresis for lipoprotein(a)-associated cardiovascular disease: prospective 5 years of follow-up and apolipoprotein(a) characterization. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2019–2027. [Google Scholar] [CrossRef]
- Lacaze, P.; Bakshi, A.; Riaz, M.; Polekhina, G.; Owen, A.; Bhatia, H.S.; Natarajan, P.; Wolfe, R.; Beilin, L.; Nicholls, S.J.; et al. Aspirin for primary prevention of cardiovascular events in relation to lipoprotein(a) genotypes. J. Am. Coll. Cardiol. 2022, 80, 1287–1298. [Google Scholar] [CrossRef]
- Bhatia, H.S.; Trainor, P.; Carlisle, S.; Tsai, M.Y.; Criqui, M.H.; DeFilippis, A.; Tsimikas, S. Aspirin and cardiovascular risk in individuals with elevated lipoprotein(a): the Multi-Ethnic Study of Atherosclerosis. J. Am. Heart Assoc. 2024, 13, e033562. [Google Scholar] [CrossRef]
- Cho, L.; Nicholls, S.J.; Nordestgaard, B.G.; Landmesser, U.; Tsimikas, S.; Blaha, M.J.; Leitersdorf, E.; Lincoff, A.M.; Lesogor, A.; Manning, B.; et al. Design and rationale of Lp(a)HORIZON trial: assessing the effect of lipoprotein(a) lowering with pelacarsen on major cardiovascular events in patients with CVD and elevated Lp(a). Am. Heart J. 2025, 287, 1–9. [Google Scholar] [CrossRef]
- ACCLAIM-Lp(a) Investigators. A Study to Investigate the Effect of Lepodisiran on the Reduction of Major Adverse Cardiovascular Events in Adults With Elevated Lipoprotein(a) (ACCLAIM-Lp(a)). ClinicalTrials.gov 2025, NCT06292013. Available online: https://clinicaltrials.gov/ct2/show/NCT06292013 (accessed on 7 January 2026).
- MOVE-Lp(a) Investigators. Assessing the Impact of Muvalaplin on Major Cardiovascular Events in Adults With Elevated Lipoprotein(a) (MOVE-Lp(a)). ClinicalTrials.gov 2025, NCT07157774. Available online: https://clinicaltrials.gov/ct2/show/NCT07157774 (accessed on 7 January 2026).
- OCEAN(a) Investigators. Olpasiran Trials of Cardiovascular Events and Lipoprotein(a) Reduction (OCEAN(a)) - Outcomes Trial. ClinicalTrials.gov 2025, NCT05581303. Sponsor: Amgen. Available online: https://clinicaltrials.gov/ct2/show/NCT05581303 (accessed on 7 January 2026).
- OCEAN(a)-PreEvent Investigators. OCEAN(a)-PreEvent: Olpasiran Trials of Cardiovascular Events And LipoproteiN(a) Reduction to Prevent First Major Cardiovascular Events. ClinicalTrials.gov 2025, NCT07136012. Available online: https://clinicaltrials.gov/ct2/show/NCT07136012 (accessed on 7 January 2026).
- ADD-VANTAGE Investigators. Lp(a) Lowering Study of Pelacarsen (TQJ230) With Background Inclisiran in Participants With Elevated Lp(a) and Established ASCVD (ADD-VANTAGE). ClinicalTrials.gov 2025, NCT06813911. Available online: https://clinicaltrials.gov/ct2/show/NCT06813911 (accessed on 20 January 2026).
- SRSD216 Investigators. A Study of SRSD216 in Patients With Elevated Lipoprotein(a). ClinicalTrials.gov. 2025, NCT07172646. Available online: https://clinicaltrials.gov/study/NCT07172646 (accessed on 7 January 2026).
- Tsimikas, S.; Karwatowska-Prokopczuk, E.; Gouni-Berthold, I.; Tardif, J.C.; Baum, S.J.; Steinhagen-Thiessen, E.; Shapiro, M.D.; Stroes, E.S.G.; Moriarty, P.M.; Nordestgaard, B.G.; et al. Lipoprotein(a) reduction in persons with cardiovascular disease. N. Engl. J. Med. 2020, 382, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Niman, S.; Goldfaden, R.F.; Ashchi, M.; Bisharat, M.; Huston, J.; Hartmann, H.; Choksi, R. A Review of the Clinical Pharmacology of Pelacarsen: A Lipoprotein(a)-Lowering Agent. Am. J. Cardiovasc. Drugs 2022, 22, 47–54. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, M.L.; Rosenson, R.S.; Gencer, B.; López, J.A.G.; Lepor, N.E.; Baum, S.J.; Stout, E.; Gaudet, D.; Knusel, B.; Kuder, J.F.; et al. Small interfering RNA to reduce lipoprotein(a) in cardiovascular disease. N. Engl. J. Med. 2022, 387, 1855–1864. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, M.L.; Rosenson, R.S.; López, J.A.G.; Lepor, N.E.; Baum, S.J.; Stout, E.; Gaudet, D.; Knusel, B.; Kuder, J.F.; Murphy, S.A.; et al. The off-treatment effects of olpasiran on lipoprotein(a) lowering: OCEAN(a)-DOSE extension period results. J. Am. Coll. Cardiol. 2024, 84, 790–797. [Google Scholar] [CrossRef]
- Nissen, S.E.; Wolski, K.; Watts, G.F.; Koren, M.J.; Fok, H.; Nicholls, S.J.; Rider, D.A.; Cho, L.; Romano, S.; Melgaard, C.; et al. Zerlasiran, a short interfering RNA targeting lipoprotein(a): a phase 2 randomized clinical trial. JAMA 2024, 331, 1215–1224. [Google Scholar] [CrossRef]
- Nissen, S.E.; Linnebjerg, H.; Shen, X.; Wolski, K.; Ma, X.; Lim, S.; Michael, L.F.; Ruotolo, G.; Gribble, G.; Navar, A.M.; et al. Lepodisiran, an extended-duration short interfering RNA targeting lipoprotein(a): a randomized dose-escalation clinical trial. JAMA 2023, 330, 2075–2083. [Google Scholar] [CrossRef]
- Faisal, A.; Basit, A.; Iftikhar, A.; Saifullah, M.; Rehmaan, M.K.U.; Basil, A.M. Lepodisiran: from genetic targeting to cardiovascular promise: a detailed narrative review of the literature. World J. Cardiol. 2025, 17, 109657. [Google Scholar] [CrossRef]
- Nicholls, S.J.; Ni, W.; Rhodes, G.M.; Nissen, S.E.; Navar, A.M.; Michael, L.F.; Haupt, A.; Krege, J.H. Oral muvalaplin for lowering of lipoprotein(a): a randomized clinical trial. JAMA. 2025, 222–231. [Google Scholar] [CrossRef]
- Gurevitz, C.; Bajaj, A.; Khera, A.V.; Do, R.; Schunkert, H.; Musunuru, K.; Rosenson, R.S. Gene therapy and genome editing for lipoprotein disorders. Eur. Heart J. 2025, 46, 3420–3433. [Google Scholar] [CrossRef]
- Rao, S.V.; O’Donoghue, M.L.; Ruel, M.; Rab, T.; TamisHolland, J.E.; Alexander, J.H.; Baber, U.; Baker, H.; Cohen, M.G.; CruzRuiz, M.; et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI guideline for the management of patients with acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2025, 85, 2135–2237. [Google Scholar] [CrossRef]
| Study (year) |
Study design |
Study population |
STEMI | Mean of Lp(a) (mg/dL) |
Lp(a) threshold (mg/dL) |
Median follow-up | Outcome | Conclusion | |
|
Yang et al. (2022) [55] |
Retrospective multicenter cohort | 765 | 431 (56.3%) | 13.41 | ≥30 | 17 months | Composite of all-cause death, nonfatal MI, nonfatal stroke and unplanned revascularization | Each 1-SD increase in Lp(a) predicted MACE (HR 1.29, 95% CI 1.11–1.48, p=0.001) and revascularization (HR 1.59, 95% CI 1.31-1.93, p=0.001). | |
|
Dai et al. (2023) [56] |
Prospective cohort | 262 | NR | 21 | ≥32 | 4.6 years | Composite of CV death, nonfatal MI, and readmission for heart failure | Lp(a) ≥32 mg/dL independently predicted MACE (HR 2.84, 95% CI l 1.25–6.60, p=0.01). | |
|
Miñana et al. (2024) [57] |
Retrospective cohort | 1,223 | 417 (34.1%) | 28.8 | Q1 ≤8.9, Q2 9-21.6, Q3 21.6-40, Q4 40-74.6, Q5 74.6-305 | 9.9 years | MI | Lp(a) ≥50 mg/dL nonlinearly associated with reinfarction (p=0.016); not associated with long-term all-cause mortality. | |
|
Takahashi et al. (2022) [58] |
Prospective cohort | 1,758 | 872 (77.1%) | 15 | ≥15 | 2.2 years | Composite of all-cause death and MI | Risk of MACE was higher in high Lp(a) group (≥15mg/dL). | |
|
Steg et al. (2025) [59] |
Post-hoc placebo-group RCT (ODYSSEY-OUTCOMES) | 9,149 | 3,235 (35.4%) | 21.4 | NR | 2.8 years | Composite of CAD death, nonfatal MI, fatal or nonfatal stroke, or hospitalization for UA | Lp(a) below 21.4mg/dL robust predicted MACE. | |
|
Schwartz et al. (2018) [60] |
Post-hoc nested cohort analysis (dal-OUTCOMES) | 4,139 | NR | 12.3 | NR | 29 months | Composite of CAD death, a major nonfatal MI, hospitalization for UA, or resuscitated cardiac arrest, or a fatal or nonfatal stroke | Lp(a) no associated with ischemic cardiovascular events. | |
|
Park et al. (2023) [61] |
Prospective cohort | 1,908 | 695 (36.4%) | 17 | I<30, II 30–49, III ≥50 | 3 years | Composite of nonfatal MI, nonfatal stroke, and CV death |
Lp(a) no associated with MACE. |
|
|
Zhang et al. (2020) [62] |
Prospective cohort | 1,008 (≥80 years) | NR | 13 | ≤10, 10-30, >30 | 32.26 months | CV death | Lp(a) >30 mg/dL linked to CV death (HR 1.52, 95% CI 1.08–2.13, p=0.016) and lower event-free survival. | |
|
Li et al. (2023) [63] |
Prospective cohort | 1,543 (677 DM, 866 non-DM) | 100% | 16.9 DM, 17.3 non-DM | ≥30 | 3.96 years | Composite of all-cause death, recurrence of MI, and stroke | MACE risk increased linearly above Lp(a) 16.9 mg/dL in DM; no effect in non-DM. | |
|
Bittner et al. (2020) [64] |
Pre-specified analysis of RCT (ODYSSEY Outcomes) |
18,924 | 6,536 (34.5%) | 21.2 | Q1 <6.7, Q2 6.7-21.2, Q3 21.2-59.6, Q4 >59.6 | 2.8 years | Composite of CV death, nonfatal MI, stroke, or hospitalization for UA | Alirocumab lowered Lp(a) by 5 mg/dL and reduced MACE risk (HR 0.85, 95% CI 0.78–0.93, p<0.001) | |
|
Wang et al. (2024) [65] |
Meta-analysis | 18,168 | NR | NR | From 12.5 to 60 | From 2.93 to 66 months | Composite of all-cause death, stroke, non-fatal MI, and coronary revascularization | High Lp(a) associated with increased MACE risk (HR 1.26, 95% CI 1.17–1.35, p< 0.001) and all-cause mortality (HR 1.36, 95% CI: 1.05–1.76, p = 0.02). |
| Molecule | NCT Number / Trial Name | Design | Inclusion Criteria | Primary Outcomes | Study Period | ||
| Pelacarsen [90] |
NCT04023552 Lp(a)HORIZON |
Phase 3 Double-blind Placebo-controlled |
Lp(a) ≥175 nmol/L + MI ≥3m and ≤10y/ischemic stroke ≥3m and ≤10y/PAD | Time to first MACE with Lp(a)≥70 mg/dL or ≥90 mg/dL | Dic 2019 – Feb 2026 | ||
| Lepodisiran [91] |
NCT06292013 ACCLAIM-Lp(a) |
Phase 3 Double-blind Placebo-controlled |
Lp(a) ≥175 nmol/L + Age ≥18y with ASCVD or ≥55y with high CV risk | Time to MACE-4 (CV death, nonfatal MI, nonfatal ischemic stroke, and urgent coronary revascularization) | Mar 2024 – Mar 2029 | ||
| Muvalaplin [92] |
NCT07157774 MOVE-Lp(a) |
Phase 3 Double-blind Placebo-controlled |
Lp(a) ≥175 nmol/L + ASCVD or high CV risk |
Time to MACE-4 (CV death, nonfatal MI, nonfatal ischemic stroke, and urgent coronary revascularization) | Sep 2025 – Mar 2031 |
||
| Olpasiran [93] |
NCT05581303 OCEAN(a)-Outcomes |
Phase 3 Double-blind Placebo-controlled |
Age 18-85y + Lp(a) ≥200 nmol/L + ASCVD | Time to first CHD death, MI, or urgent coronary revascularization |
Dec 2022 – Dec 2026 | ||
| Olpasiran [94] |
NCT07136012 OCEAN(a)-PreEvent |
Phase 3 Double-blind Placebo-controlled |
Age ≥50y + Lp(a) ≥200 nmol/L + multiple CV risk factors |
Time to CHD death, MI, or urgent revascularization | Aug 2025 – Oct 2031 | ||
| Pelacarsen + Inclisiran vs Placebo + Inclisiran [95] |
NCT06813911 ADD-VANTAGE |
Phase 3 Double-blind Placebo-controlled |
Age 18-80y + ASCVD + Lp(a) ≥175 nmol/L + LDL-C >70 mg/dL despite statins + Run-in period of inclisiran | Change from baseline Lp(a) and LDL-C | Apr 2025 – Dec 2027 | ||
| SRSD216 [96] |
NCT07172646 SRSD216 Study |
Phase I/II Double-blind Placebo-controlled |
BMI 18-40 kg/m2 |
Safety, tolerability, and adverse events | Apr 2025 – Apr 2027 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




