Submitted:
31 January 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Study Selection
2.4. Data Extraction
2.5. Risk of Bias Assessment
2.6. Certainty of Evidence
3. Results
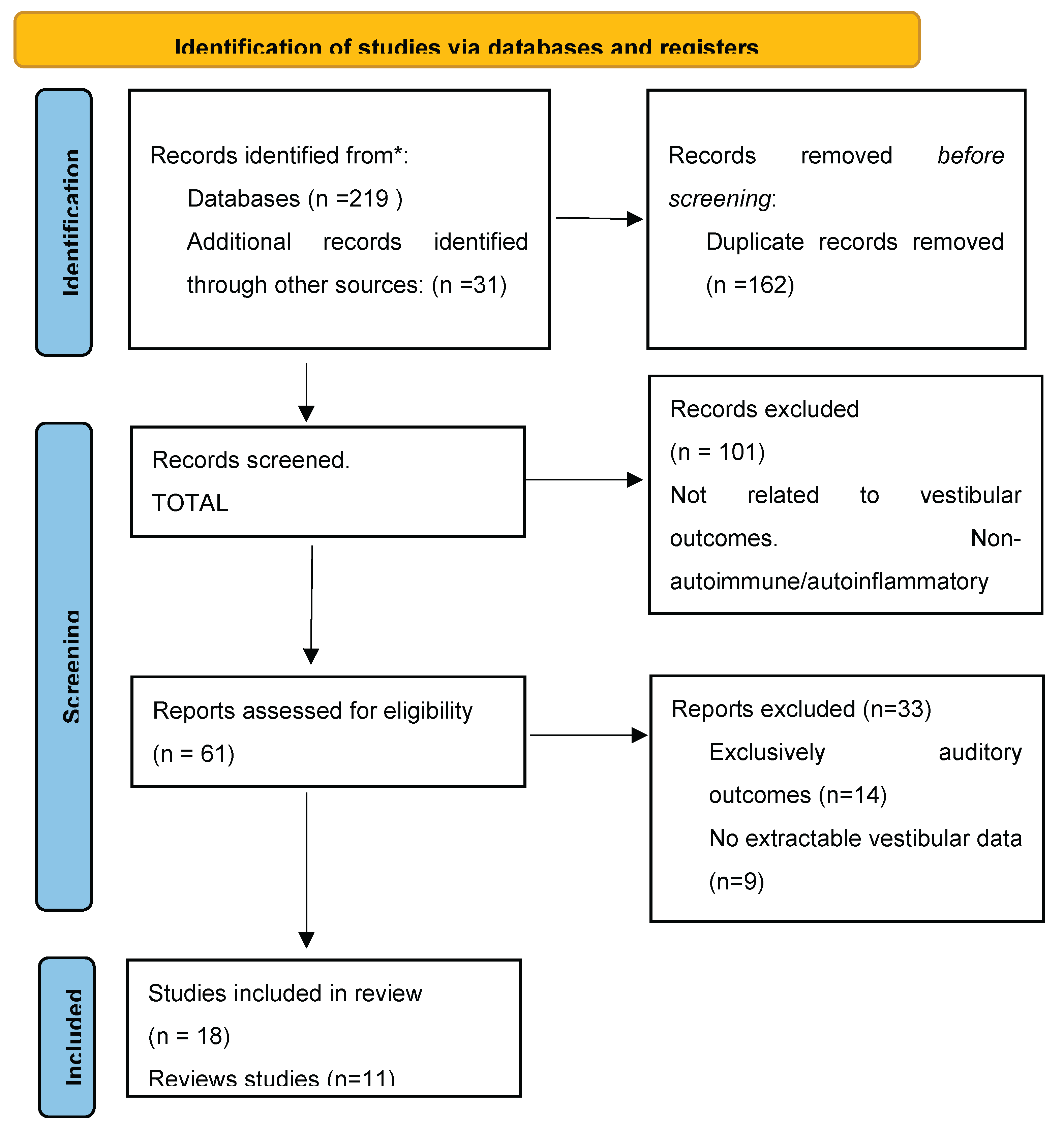
3.1. Study Selection
3.2. Characteristics of Included Studies
3.3. Vestibular Involvement Across Autoimmune and Rheumatologic Diseases
3.4. Certainty of Evidence (GRADE Assessment)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ralli, M.; Greco, A.; Turchetta, R.; Altissimi, G.; de Vincentiis, M. Audiovestibular symptoms in systemic autoimmune diseases. J. Immunol. Res. 2018, 2018, 5798103. [Google Scholar] [CrossRef] [PubMed]
- Girasoli, L.; Cazzador, D.; Padoan, R.; Delogu, M.; Zanoletti, E.; Martini, A. Update on vertigo in autoimmune disorders: From diagnosis to treatment. J. Immunol. Res. 2018, 2018, 5072582. [Google Scholar] [CrossRef]
- Bovo, R.; Aimoni, C.; Martini, A. Immune-mediated inner ear disease. Acta Otolaryngol. 2006, 126, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Athanasopoulos, D.; Papathanasiou, M.; Tsivgoulis, G. Inner ear involvement in autoimmune and autoinflammatory diseases. Autoimmun. Rev. 2024, 23, 103368. [Google Scholar] [CrossRef]
- Breslin, N.K.; Varadarajan, V.V.; Suresh, K.; et al. Autoimmune inner ear disease: A systematic review of management. Otolaryngol. Head Neck Surg. 2020, 162, 463–472. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Arias-Núñez, M.C.; Miranda-Filloy, J.A.; González-Juanatey, C.; Llorca, J.; González-Gay, M.A. Audiovestibular manifestations in patients with limited systemic sclerosis and centromere protein-B antibodies. Medicine (Baltimore) 2008, 87, 131–141. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Arias-Núñez, M.C.; Gómez-Acebo, I.; Llorca, J.; González-Gay, M.A. Benign paroxysmal positional vertigo and clinical test of sensory interaction and balance in systemic sclerosis. Otol. Neurotol. 2008, 29, 1155–1161. [Google Scholar] [CrossRef]
- Salvador, C.; Martínez, C.; Riera, A.; et al. Audiovestibular involvement in systemic sclerosis: A systematic review and meta-analysis. In Eur. Arch. Otorhinolaryngol.; 2025. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Llorca, J.; Costa-Ribas, C.; García-Porrúa, C.; González-Gay, M.A. Giant cell arteritis: A new association with benign paroxysmal positional vertigo. Laryngoscope 2004, 114, 1420–1425. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Vázquez-Rodríguez, T.R.; et al. Audiovestibular manifestations in patients with ankylosing spondylitis. Medicine (Baltimore) 2011, 90, 99–109. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Piña, T.; et al. Investigations into audiovestibular manifestations in patients with psoriatic arthritis. J. Rheumatol. 2014, 41, 2018–2026. [Google Scholar] [CrossRef]
- Ertugrul, O.; Mutlu, A.; Zindanci, I.; et al. Audiological and vestibular measurements in Behçet’s disease. Eur. Arch. Otorhinolaryngol. 2019, 276, 1625–1632. [Google Scholar] [CrossRef]
- Ulusoy, B.; Limon, M.; Yılmaz, S.; et al. Effects of primary Sjögren’s syndrome on hearing and vestibular systems. J. Laryngol. Otol. 2022, 136, 923–930. [Google Scholar] [CrossRef]
- Özkırış, M.; Kapusuz, Z.; Günaydın, İ.; et al. Does rheumatoid arthritis have an effect on audiovestibular tests? Eur. Arch. Otorhinolaryngol. 2014, 271, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Karataş, M.; Onat, A.M.; Durmuş, O.; et al. Vestibular dysfunction in patients with systemic lupus erythematosus. J. Neurol. Sci. 2007, 254, 6–10. [Google Scholar] [CrossRef]
- Huppert, D.; Strupp, M.; Möller, A.; Brandt, T. Recurrent vertigo in patients with antiphospholipid syndrome. J. Neurol. 2013, 260, 1829–1833. [Google Scholar] [CrossRef]
- Kim, J.; Jung, J.Y.; Park, H.J.; et al. Vestibular dysfunction in patients with ANCA-associated vasculitis. Clin. Exp. Rheumatol. 2012, 30, S41–S45. [Google Scholar]
- Greco, A.; Fusconi, M.; Gallo, A.; et al. Audiovestibular involvement in Cogan’s syndrome. Autoimmun. Rev. 2016, 15, 347–353. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Llorca, J.; García-Porrúa, C.; et al. Audiovestibular manifestations in giant cell arteritis. Medicine (Baltimore) 2003, 82, 13–26. [Google Scholar] [CrossRef]
- Gázquez, I.; Soto-Varela, A.; Aran, I.; et al. High prevalence of systemic autoimmune diseases in patients with Ménière’s disease. PLoS ONE 2011, 6, e26759. [Google Scholar] [CrossRef]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Regueiro-Villarín, S.; González-Gay, M.A. Audiovestibular manifestations in systemic vasculitis. Acta Otorrinolaringol. Esp. 2009, 60, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Vázquez-Rodríguez, T.R.; et al. Benign paroxysmal positional vertigo and clinical test of sensory interaction and balance in ankylosing spondylitis. Otol. Neurotol. 2011, 32, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Amor-Dorado, J.C.; Barreira-Fernández, M.P.; Llorca, J.; González-Gay, M.A. Oculographic and posturographic findings in psoriatic arthritis. Otol. Neurotol. 2017, 38, 448–453. [Google Scholar] [CrossRef] [PubMed]
- Yazici, H.; Tüzün, Y.; Pazarli, H.; et al. Vestibular involvement in Behçet disease. Acta Otolaryngol. 2001, 121, 224–228. [Google Scholar]
- Ziavra, N.; Politi, E.N.; Kastanioudakis, I.; et al. Hearing loss and vestibular dysfunction in rheumatoid arthritis. Clin. Rheumatol. 2010, 29, 279–284. [Google Scholar] [CrossRef]
- Kariya, S.; Cureoglu, S.; Fukushima, H.; et al. Vestibular involvement in sarcoidosis. Acta Otolaryngol. 2014, 134, 296–301. [Google Scholar] [CrossRef]
- Chen, J.J.; Hsu, C.W.; Chen, Y.W.; et al. Audiovestibular dysfunction related to antiphospholipid syndrome. Diagnostics 2024, 14, 2522. [Google Scholar] [CrossRef]
- Li, X.; Zhang, Y.; Wang, Y.; et al. Immune microenvironment and autoimmune inner ear disease. Front. Immunol. 2025, 30. [Google Scholar]
- Miwa, T.; Okano, H. Macrophages in autoimmune inner ear disease. Int. J. Mol. Sci. 2022, 23, 10458. [Google Scholar] [CrossRef]
- Gomaa, N.A.; Jimoh, Z.; Campbell, S.; Zenke, J.K.; Szczepek, A.J. Biomarkers for inner ear disorders: Scoping review on the role of biomarkers in hearing and balance disorders. Diagnostics 2021, 11, 42. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, P.; Li, Q.; Kong, C. Investigating the process of autoimmune inner ear disease: Unveiling the intricacies of pathogenesis and therapeutic strategies. Int. J. Med. Sci. 2025, 22, 179–187. [Google Scholar] [CrossRef]
| Author (Year) | Disease | Study Design | Participants (Patients/Controls) | Vestibular Assessment | Vestibular Outcomes | Main Vestibular Findings |
|---|---|---|---|---|---|---|
| Amor-Dorado et al. (2003) | Giant cell arteritis | Prospective cohort | 44 / 44 | Positional tests, caloric test | Vertigo, peripheral vestibulopathy | Vestibular dysfunction significantly more frequent than in controls |
| Amor-Dorado et al. (2004) | Giant cell arteritis | Case–control | 44 / 44 | Dix–Hallpike maneuver | BPPV | Strong association between GCA and BPPV |
| Amor-Dorado et al. (2008a) | Limited systemic sclerosis | Case–control | 35 / 59 | Caloric test, CTSIB, oculography | Chronic imbalance | Objective vestibular abnormalities significantly more frequent |
| Amor-Dorado et al. (2008b) | Systemic sclerosis | Case–control | 42 / 74 | Dix–Hallpike, CTSIB | BPPV, postural instability | Increased prevalence of BPPV and abnormal sensory integration |
| Amor-Dorado et al. (2011a) | Ankylosing spondylitis | Case–control | 59 / 46 | Caloric test, CTSIB | Dizziness, imbalance | Peripheral vestibular hypofunction and impaired balance integration |
| Amor-Dorado et al. (2011b) | Ankylosing spondylitis | Case–control | 59 / 46 | Dix–Hallpike, CTSIB | BPPV | Higher BPPV prevalence than in controls |
| Amor-Dorado et al. (2014) | Psoriatic arthritis | Case–control | 60 / 60 | Caloric test, CTSIB | Dizziness, imbalance | Significant vestibular dysfunction compared with controls |
| Amor-Dorado et al. (2017) | Psoriatic arthritis | Case–control | 60 / 60 | Oculography, CTSIB, CDP | Postural instability | Objective abnormalities in balance control |
| Gázquez et al. (2011) | Ménière’s disease + autoimmune disease | Multicenter cohort | >600 / – | Clinical criteria (AAO–HNS) | Episodic vertigo | Autoimmune comorbidity associated with more persistent vertigo |
| Karataş et al. (2007) | Systemic lupus erythematosus | Observational | 28 / – | Clinical vestibular assessment | Vertigo, dizziness | High frequency of vestibular symptoms |
| Ertugrul et al. (2019) | Behçet disease | Prospective case–control | 31 / 31 | vHIT, head-shake test, DHI | Peripheral vestibular dysfunction | Horizontal canal dysfunction and head-shake–induced nystagmus significantly more frequent |
| Yazici et al. (2001) | Behçet disease | Observational | 29 / – | Clinical assessment | Vertigo | Vestibular symptoms reported as part of systemic involvement |
| Ziavra et al. (2010) | Rheumatoid arthritis | Case–control | 25 / 20 | Caloric test | Dizziness | Peripheral vestibular hypofunction more frequent than in controls |
| Özkırış et al. (2014) | Rheumatoid arthritis | Prospective case–control | 81 / 81 | VNG, caloric test, positional tests | Central and peripheral vestibular dysfunction | VNG abnormalities in 38.3%; canal paresis in 13.6% |
| Ulusoy et al. (2022) | Primary Sjögren syndrome | Case–control | 35 / 35 | vHIT, cVEMP, oVEMP | Subclinical vestibular dysfunction | Reduced vHIT gain and abnormal VEMP latencies |
| Kim et al. (2012) | ANCA-associated vasculitis | Cohort | 32 / – | Caloric test | Imbalance | Vestibular dysfunction associated with disease activity |
| Kariya et al. (2014) | Sarcoidosis | Observational | 18 / – | Clinical ± VNG | Vertigo | Peripheral inflammatory vestibular involvement |
| Greco et al. (2016) | Cogan syndrome | Cohort | 25 / – | VNG | Severe vertigo | Immune-mediated vestibulopathy |
| Huppert et al. (2013) | Antiphospholipid syndrome | Case–control | 22 / 22 | Clinical assessment | Recurrent vertigo | Vestibular symptoms more frequent than in controls |
| Author (Year) | Type of Review | Diseases Included | Vestibular Scope | Main Contribution |
|---|---|---|---|---|
| Ralli et al. (2018) | Narrative clinical review | Systemic autoimmune diseases (SLE, RA, Sjögren, Behçet, vasculitis, Cogan syndrome, sarcoidosis) | Clinical vestibular symptoms | Highlights frequency and underrecognition of vestibular symptoms in systemic autoimmune diseases |
| Girasoli et al. (2018) | Narrative clinical review | Autoimmune disorders | Clinical vestibular syndromes | Provides an overview of immune-mediated vertigo and diagnostic considerations |
| Bovo et al. (2006) | Narrative clinical review | Autoimmune inner ear disease (AIED) | Vertigo, imbalance, episodic and positional vertigo | Classical clinical description of AIED, emphasizing frequent vestibular symptoms and coexistence with systemic autoimmune diseases |
| Amor-Dorado et al. (2009) | Narrative clinical review | Systemic vasculitides (GCA, Takayasu arteritis, PAN, ANCA-associated vasculitis, Behçet disease, Cogan syndrome, RA, SSc, SLE, Sjögren syndrome) | Vertigo, nystagmus, BPPV | Early comprehensive synthesis linking vasculitis and vestibular involvement, particularly in giant cell arteritis |
| Breslin et al. (2020) | Systematic review | Autoimmune inner ear disease | Vestibular outcomes (secondary) | Focused mainly on hearing outcomes; vestibular data inconsistently reported |
| Gázquez et al. (2011) | Narrative review / cohort synthesis | Ménière’s disease with autoimmune comorbidity | Recurrent vertigo | Demonstrates increased prevalence of systemic autoimmune diseases in Ménière’s disease |
| Athanasopoulos et al. (2024) | Narrative mechanistic review | Autoimmune and autoinflammatory diseases | Mechanistic (non-clinical) | Discusses immune-inflammatory and vascular mechanisms affecting the inner ear |
| Miwa & Okano (2022) | Narrative mechanistic review | Autoimmune inner ear disease | Experimental / translational (non-clinical) | Demonstrates macrophage-mediated immune mechanisms in the inner ear; supports biological plausibility of vestibular involvement |
| Li et al. (2025) | Narrative mechanistic review | Autoimmune inner ear disorders | Mechanistic (non-clinical) | Reviews immune microenvironment and inflammatory pathways in inner ear disease |
| Salvador et al. (2025) | Systematic review and meta-analysis | Systemic sclerosis | Clinical vestibular outcomes | Reports increased prevalence of vertigo in systemic sclerosis; objective vestibular findings highly heterogeneous |
| Chen et al. (2024) | Systematic review | Antiphospholipid syndrome | Clinical vestibular outcomes | Summarizes evidence linking APS with recurrent vertigo and vestibular dysfunction |
| Disease | Vestibular Outcomes | Evidence Base | Main Limitations | Certainty of Evidence (GRADE) |
|---|---|---|---|---|
| Systemic sclerosis | Vertigo, balance impairment, BPPV | Multiple observational case–control studies; systematic review and meta-analysis | Observational designs; heterogeneous vestibular testing; lack of longitudinal data | Moderate at best, within observational limitations |
| Giant cell arteritis | BPPV, vertigo | Observational cohort and case–control studies | Small sample sizes; limited objective vestibular testing | Moderate at best, within observational limitations |
| Ankylosing spondylitis | Balance impairment, BPPV | Observational case–control studies | Non-specific balance endpoints; heterogeneous methods | Low |
| Psoriatic arthritis | Balance impairment | Observational case–control studies | Small cohorts; limited vestibular specificity | Low |
| Behçet disease | Vestibular hypofunction, abnormal vHIT | Observational studies | Limited number of studies; inconsistent outcomes | Low |
| Primary Sjögren syndrome | Subclinical vestibular abnormalities | Single observational study | Small sample size; lack of replication | Low |
| Rheumatoid arthritis | Peripheral and central vestibular abnormalities | Observational studies | Conflicting findings; heterogeneous testing | Low |
| Systemic lupus erythematosus | Vertigo, vestibular dysfunction | Observational studies | Symptom-based outcomes; limited objective data | Low |
| Antiphospholipid syndrome | Recurrent vertigo | Observational studies; systematic review | Sparse primary data; indirect vestibular outcomes | Low |
| ANCA-associated vasculitis | Vestibular dysfunction | Small observational series | Very limited evidence | Very low |
| Sarcoidosis | Vestibular involvement | Case series | Rare condition; non-comparative data | Very low |
| Cogan syndrome | Vestibular dysfunction | Observational studies | Rare disease; mixed audiovestibular outcomes | Very low |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




