Submitted:
01 February 2026
Posted:
03 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
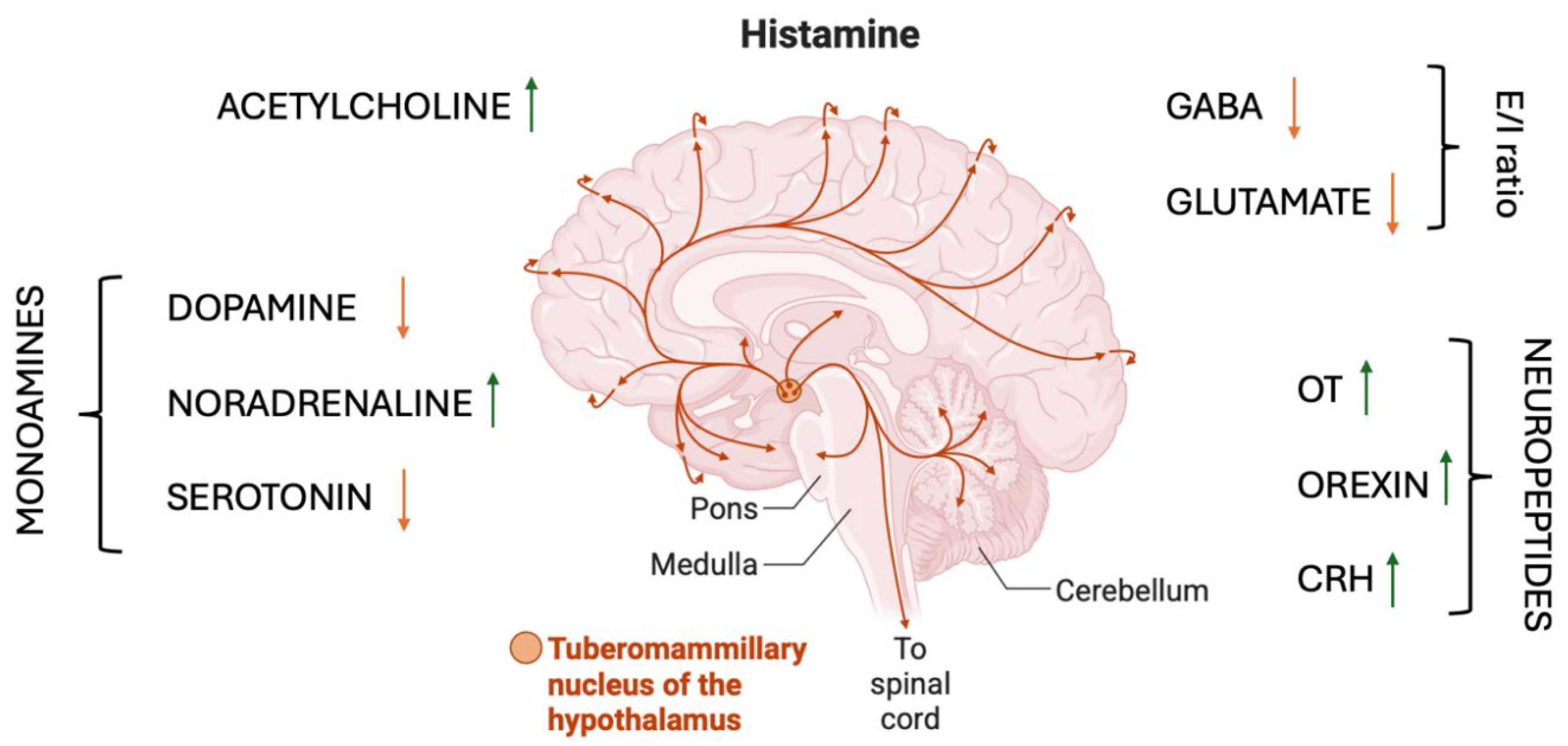
2. Anatomical and Receptor Architecture of the Histaminergic System
3. Histamine–Monoamine Interactions: Coordinating Dopamine, Serotonin, and Noradrenaline
3.1. Dopamine
3.2. Serotonin
3.3. Noradrenaline
3.4. A Coordinated Monoaminergic Framework
4. Histamine and Acetylcholine: Cortical Activation, Attention, and Learning Readiness
5. Regulation of Excitation–Inhibition Balance: Histamine, Glutamate, and GABA
6. Histamine, Neuropeptides, and the Regulation of Arousal, Motivation, and Social Salience
6.1. Orexin/Hypocretin: Sustained Wakefulness and Motivational Drive
6.2. Stress-Related Neuropeptides: CRH and Arousal Bias
6.3. Oxytocin and Social Neuropeptides: Contextual Modulation Rather than Valence
6.4. Integrating Neuropeptide Signals into Global Brain State
7. Five Avenues for Future Research
Conclusion
Acknowledgments
References
- Marder, E. Neuromodulation of neuronal circuits: back to the future. Neuron 2012, 76, 1–11. [Google Scholar] [CrossRef]
- Robbins, T. W. Chemistry of the mind: neurochemical modulation of prefrontal cortical function. J Comp Neurol 2005, 493, 140–146. [Google Scholar] [CrossRef]
- Haas, H.; Panula, P. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat Rev Neurosci 2003, 4, 121–130. [Google Scholar] [CrossRef]
- Brown, R. E.; Stevens, D. R.; Haas, H. L. The physiology of brain histamine. Prog Neurobiol 2001, 63, 637–672. [Google Scholar] [CrossRef]
- Wada, H.; Inagaki, N.; Itowi, N.; Yamatodani, A. Histaminergic neuron system in the brain: distribution and possible functions. Brain Res Bull 1991, 27, 367–370. [Google Scholar] [CrossRef]
- Ellenbroek, B. A. Histamine H(3) receptors, the complex interaction with dopamine and its implications for addiction. Br J Pharmacol 2013, 170, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Hersey, M.; et al. A tale of two transmitters: serotonin and histamine as in vivo biomarkers of chronic stress in mice. J Neuroinflammation 2022, 19, 167. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, N. J.; Stock, M. J.; Wyllie, M. G. Effects of histamine antagonists on noradrenaline-stimulated blood flow and oxygen consumption of brown adipose tissue in the rat. Pflugers Arch 1984, 402, 325–329. [Google Scholar] [CrossRef]
- Prast, H.; et al. Histaminergic neurons modulate acetylcholine release in the ventral striatum: role of H3 histamine receptors. Naunyn Schmiedebergs Arch Pharmacol 1999, 360, 558–564. [Google Scholar] [CrossRef]
- Karpati, A.; et al. Histamine elicits glutamate release from cultured astrocytes. J Pharmacol Sci 2018, 137, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; et al. Wakefulness Is Governed by GABA and Histamine Cotransmission. Neuron 2015, 87, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Panula, P.; Nuutinen, S. The histaminergic network in the brain: basic organization and role in disease. Nat Rev Neurosci 2013, 14, 472–487. [Google Scholar] [CrossRef]
- Li, B.; et al. Histamine Increases Neuronal Excitability and Sensitivity of the Lateral Vestibular Nucleus and Promotes Motor Behaviors via HCN Channel Coupled to H2 Receptor. Front Cell Neurosci 2016, 10, 300. [Google Scholar] [CrossRef]
- Green, M. Histamine in the central nervous system. Proc West Pharmacol Soc 1978, 21, 337–339. [Google Scholar] [PubMed]
- Thangam, E. B.; et al. The Role of Histamine and Histamine Receptors in Mast Cell-Mediated Allergy and Inflammation: The Hunt for New Therapeutic Targets. Front Immunol 2018, 9, 1873. [Google Scholar] [CrossRef] [PubMed]
- Mizuguchi, H.; et al. Signaling Pathway of Histamine H(1) Receptor-Mediated Histamine H(1) Receptor Gene Upregulation Induced by Histamine in U-373 MG Cells. Curr Issues Mol Biol 2021, 43, 1243–1254. [Google Scholar] [CrossRef]
- Arrang, J. M.; Devaux, B.; Chodkiewicz, J. P.; Schwartz, J. C. H3-receptors control histamine release in human brain. J Neurochem 1988, 51, 105–108. [Google Scholar] [CrossRef]
- Arrang, J. M.; Garbarg, M.; Schwartz, J. C. Auto-inhibition of brain histamine release mediated by a novel class (H3) of histamine receptor. Nature 1983, 302, 832–837. [Google Scholar] [CrossRef]
- Lin, J. S.; Sergeeva, O. A.; Haas, H. L. Histamine H3 receptors and sleep-wake regulation. J Pharmacol Exp Ther 2011, 336, 17–23. [Google Scholar] [CrossRef]
- Schlicker, E.; Kathmann, M. Role of the Histamine H(3) Receptor in the Central Nervous System. Handb Exp Pharmacol 2017, 241, 277–299. [Google Scholar] [CrossRef]
- Bargmann, C. I. Beyond the connectome: how neuromodulators shape neural circuits. Bioessays 2012, 34, 458–465. [Google Scholar] [CrossRef]
- Llinas, R. R. Intrinsic electrical properties of mammalian neurons and CNS function: a historical perspective. Front Cell Neurosci 2014, 8, 320. [Google Scholar] [CrossRef]
- Alfaro-Rodriguez, A.; et al. Histamine H3 receptor activation prevents dopamine D1 receptor-mediated inhibition of dopamine release in the rat striatum: a microdialysis study. Neurosci Lett 2013, 552, 5–9. [Google Scholar] [CrossRef]
- Bolam, J. P.; Ellender, T. J. Histamine and the striatum. Neuropharmacology 2016, 106, 74–84. [Google Scholar] [CrossRef]
- Kononoff Vanhanen, J.; Nuutinen, S.; Tuominen, M.; Panula, P. Histamine H3 Receptor Regulates Sensorimotor Gating and Dopaminergic Signaling in the Striatum. J Pharmacol Exp Ther 2016, 357, 264–272. [Google Scholar] [CrossRef]
- Rapanelli, M.; et al. Histamine H3R receptor activation in the dorsal striatum triggers stereotypies in a mouse model of tic disorders. Transl Psychiatry 2017, 7, e1013. [Google Scholar] [CrossRef] [PubMed]
- Schlicker, E.; Betz, R.; Gothert, M. Histamine H3 receptor-mediated inhibition of serotonin release in the rat brain cortex. Naunyn Schmiedebergs Arch Pharmacol 1988, 337, 588–590. [Google Scholar] [CrossRef]
- Threlfell, S.; et al. Histamine H3 receptors inhibit serotonin release in substantia nigra pars reticulata. J Neurosci 2004, 24, 8704–8710. [Google Scholar] [CrossRef] [PubMed]
- Alguacil, L. F.; Perez-Garcia, C. Histamine H3 receptor: a potential drug target for the treatment of central nervous system disorders. Curr Drug Targets CNS Neurol Disord 2003, 2, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Dayan, P.; Huys, Q. J. Serotonin, inhibition, and negative mood. PLoS Comput Biol 2008, 4, e4. [Google Scholar] [CrossRef]
- Cools, R.; Roberts, A. C.; Robbins, T. W. Serotoninergic regulation of emotional and behavioural control processes. Trends Cogn Sci 2008, 12, 31–40. [Google Scholar] [CrossRef]
- Aston-Jones, G.; Cohen, J. D. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu Rev Neurosci 2005, 28, 403–450. [Google Scholar] [CrossRef] [PubMed]
- Young, C. S.; Mason, R.; Hill, S. J. Studies on the mechanism of histamine-induced release of noradrenaline and 5-hydroxytryptamine from slices of rat cerebral cortex. Biochem Pharmacol 1988, 37, 2799–2805. [Google Scholar] [CrossRef] [PubMed]
- Hasselmo, M. E. The role of acetylcholine in learning and memory. Curr Opin Neurobiol 2006, 16, 710–715. [Google Scholar] [CrossRef]
- Goard, M.; Dan, Y. Basal forebrain activation enhances cortical coding of natural scenes. Nat Neurosci 2009, 12, 1444–1449. [Google Scholar] [CrossRef]
- Sarter, M.; Hasselmo, M. E.; Bruno, J. P.; Givens, B. Unraveling the attentional functions of cortical cholinergic inputs: interactions between signal-driven and cognitive modulation of signal detection. Brain Res Brain Res Rev 2005, 48, 98–111. [Google Scholar] [CrossRef]
- Cecchi, M.; Passani, M. B.; Bacciottini, L.; Mannaioni, P. F.; Blandina, P. Cortical acetylcholine release elicited by stimulation of histamine H1 receptors in the nucleus basalis magnocellularis: a dual-probe microdialysis study in the freely moving rat. Eur J Neurosci 2001, 13, 68–78. [Google Scholar] [CrossRef]
- Khateb, A.; Fort, P.; Pegna, A.; Jones, B. E.; Muhlethaler, M. Cholinergic nucleus basalis neurons are excited by histamine in vitro. Neuroscience 1995, 69, 495–506. [Google Scholar] [CrossRef]
- Thakkar, M. M. Histamine in the regulation of wakefulness. Sleep Med Rev 2011, 15, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Blandina, P.; et al. Inhibition of cortical acetylcholine release and cognitive performance by histamine H3 receptor activation in rats. Br J Pharmacol 1996, 119, 1656–1664. [Google Scholar] [CrossRef]
- Kay, G. G. The effects of antihistamines on cognition and performance. J Allergy Clin Immunol 2000, 105, S622–627. [Google Scholar] [CrossRef]
- Esbenshade, T. A.; et al. The histamine H3 receptor: an attractive target for the treatment of cognitive disorders. Br J Pharmacol 2008, 154, 1166–1181. [Google Scholar] [CrossRef]
- Yizhar, O.; et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature 2011, 477, 171–178. [Google Scholar] [CrossRef]
- Carcea, I.; Froemke, R. C. Cortical plasticity, excitatory-inhibitory balance, and sensory perception. Prog Brain Res 2013, 207, 65–90. [Google Scholar] [CrossRef]
- Froemke, R. C. Plasticity of cortical excitatory-inhibitory balance. Annu Rev Neurosci 2015, 38, 195–219. [Google Scholar] [CrossRef] [PubMed]
- Takei, H.; Yamamoto, K.; Bae, Y. C.; Shirakawa, T.; Kobayashi, M. Histamine H(3) Heteroreceptors Suppress Glutamatergic and GABAergic Synaptic Transmission in the Rat Insular Cortex. Front Neural Circuits 2017, 11, 85. [Google Scholar] [CrossRef]
- Chavez, H.; Vega, R.; Soto, E. Histamine (H3) receptors modulate the excitatory amino acid receptor response of the vestibular afferents. Brain Res 2005, 1064, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schlicker, E.; Malinowska, B.; Kathmann, M.; Gothert, M. Modulation of neurotransmitter release via histamine H3 heteroreceptors. Fundam Clin Pharmacol 1994, 8, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Vyazovskiy, V. V.; Harris, K. D. Sleep and the single neuron: the role of global slow oscillations in individual cell rest. Nat Rev Neurosci 2013, 14, 443–451. [Google Scholar] [CrossRef]
- Huang, Z. L.; et al. Arousal effect of orexin A depends on activation of the histaminergic system. Proc Natl Acad Sci U S A 2001, 98, 9965–9970. [Google Scholar] [CrossRef]
- Burgess, C. R. Histamine and orexin in the control of arousal, locomotion, and motivation. J Neurosci 2010, 30, 2810–2811. [Google Scholar] [CrossRef]
- Saper, C. B.; Fuller, P. M. Wake-sleep circuitry: an overview. Curr Opin Neurobiol 2017, 44, 186–192. [Google Scholar] [CrossRef]
- Kjaer, A.; Larsen, P. J.; Knigge, U.; Jorgensen, H.; Warberg, J. Neuronal histamine and expression of corticotropin-releasing hormone, vasopressin and oxytocin in the hypothalamus: relative importance of H1 and H2 receptors. Eur J Endocrinol 1998, 139, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Fleckenstein, A. E.; Lookingland, K. J.; Moore, K. E. Histaminergic neurons mediate restraint stress-induced activation of central 5-hydroxytryptaminergic neurons in the rat. Eur J Pharmacol 1994, 264, 163–167. [Google Scholar] [CrossRef]
- Provensi, G.; Passani, M. B.; Costa, A.; Izquierdo, I.; Blandina, P. Neuronal histamine and the memory of emotionally salient events. Br J Pharmacol 2020, 177, 557–569. [Google Scholar] [CrossRef]
- Arnsten, A. F. Stress signalling pathways that impair prefrontal cortex structure and function. Nat Rev Neurosci 2009, 10, 410–422. [Google Scholar] [CrossRef]
- Kjaer, A.; Knigge, U.; Warberg, J. Involvement of oxytocin in histamine- and stress-induced ACTH and prolactin secretion. Neuroendocrinology 1995, 61, 704–713. [Google Scholar] [CrossRef]
- Baettig, L.; et al. The awareness of the scared - context dependent influence of oxytocin on brain function. Brain Imaging Behav 2020, 14, 2073–2083. [Google Scholar] [CrossRef] [PubMed]
- Shamay-Tsoory, S. G.; Abu-Akel, A. The Social Salience Hypothesis of Oxytocin. Biol Psychiatry 2016, 79, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Leong, I. L.; Wang, X.; Wu, H. Oxytocin's impact on the social brain: Individual differences and context shape a core amygdala-mediated mechanism. Neurosci Biobehav Rev 2026, 183, 106566. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



