4. Discussion
The present study aimed to evaluate urinary tubular biomarkers (uGGT, uALP, uCyst and uNAG) in clinically stable dogs with MMVD to determine their potential as early indicators of CvRDCH and to assess the prevalence of tubular alterations across different disease stages.
CvRD, analogous to cardiorenal syndrome in human medicine [
28], are defined as structural or functional kidney and/or cardiovascular abnormalities triggered by disease, toxins, or drugs that disrupt the physiological interplay between both organs, leading to dysfunction in one or both [
4]. Within this framework, MMVD is considered part of the CvRD spectrum as a chronic cardiovascular condition that secondarily affects renal function [
29]. An increased prevalence of CKD has been reported in dogs with MMVD, which rises as the disease progresses [
30,
31]. Moreover, MMVD has been identified as a risk factor for CKD progression in dogs affected by both conditions [
32].
In human medicine, CKD is a negative prognostic factor in patients with mitral regurgitation [
23], and in veterinary medicine, elevations in renal biomarkers such as urea, sCr, and sCyst have been associated with worse outcomes in MMVD [
33,
34]. Conversely, the development of azotaemia has not been shown to significantly impact survival in dogs with CHF—whether of mixed aetiologies or specifically due to MMVD—at 1 month [
35] and 3 months [
36], respectively, after the onset of congestive heart failure. Similarly, another study reported no differences in sCr between dogs with MMVD that survived or died within 6 months after sampling [
37]. Taken together, these findings remain inconsistent and do not yet support a definitive conclusion regarding the prognostic value of conventional renal biomarkers.
Anaemia plays an important role in CvRD, as it can induce tissue hypoxia, leading to cellular injury at both renal and cardiac levels [
29]. In human medicine, the concept of cardiorenal-anaemia syndrome has been proposed, underscoring its clinical relevance [
38]. While anaemia is common in dogs with CKD [
39], it is not typically observed in MMVD [
40,
41].
In the present study, although the prevalence of anaemia (HGB < 13.1 g/dL) was low, —0, 11, and 11.5% in groups B1, B2, and C+D, respectively—, a significant reduction in HGB, RBC, and HCT was noted in clinical stages. This may suggest a tendency toward anaemia without overt haematologic changes. This observation aligns with previous findings, which have been associated with a correlation between markers of iron deficiency and echocardiographic measurements in MMVD, suggesting that anaemia may develop over time despite initially normal haematologic profiles [
41]. Another study also reported differences in HGB and HCT between advanced and early stages; with higher anaemia prevalence in late-stage disease compared with our findings [
40]. However, other authors have found no significant differences in RBC, HCT [
37,
42] or HGB across MMVD stages [
37], indicating heterogeneity in the literature.
Moreover, anaemia has been described as a negative prognostic factor, correlating with increased sCr and clinical stage [
40], highlighting its importance within the CvRD. In contrast, our study revealed only a moderate negative correlation between HGB and both sCyst and uCystc, suggesting that, although MMVD may predispose to anaemia and renal injury, the relationship between these processes was limited. Furthermore, a recent study reported no differences in these parameters between dogs that survived or had died within 6 months [
37].
Finally, more than half of the dogs in group C+D were receiving benazepril, and all those with decreased HGB were on this treatment. As an angiotensin-converting enzyme inhibitor, benazepril reduces angiotensin II, which promotes erythropoiesis, likely via increased erythropoietin levels [
43]. Inhibition of this pathway has been linked to anaemia in humans and cats [
44]. Therefore, benazepril may represent one of the factors underlying the trend toward anaemia observed in dogs with MMVD.
Regarding leukocyte profiles, as previously reported, a trend toward a stress leukogram was observed, with increased total leukocytes [
9,
45,
46], neutrophils and monocytes [
45,
46] and decreased lymphocytes [
45], more pronounced in clinical stages. Eosinophils reduction had not been previously documented and adds to the stress leukogram [
47]. Guglielmini et al. reported lower WBC counts in dogs that survived than in those that died within 6 months [
37], suggesting a potential prognostic impact. Thrombocytosis has also been reported [
45] and, together with these changes, reflects inflammatory haematologic alterations in dogs with MMVD, supporting the role of inflammation in the pathogenesis of the disease [
48].
Assessment of renal function in MMVD has relied on blood parameters such as sCr, SDMA, and sCyst, which reflect changes in functional renal mass and indirectly estimate glomerular filtration rate (GFR) [
14,
26,
49]. These markers typically increase in the clinical stages [
5,
7,
8,
9,
31,
50], consistent with our findings for serum sCr, SDMA, and sCyst suggesting reduced GFR in advanced disease. Given patients´ age, some changes may reflect pre-existing CKD, whose prevalence rises with age [
51]. However, subgroup ages did not differ significantly, and CKD is more prevalent in dogs with MMVD than in those without cardiac disease [
31], suggesting the changes are more likely attributable to MMVD and CvRD than age alone.
Urinalysis revealed a significant increase in UPC across all MMVD stages, consistent with reports in advanced stages [
7]. However, no stage-dependent differences were observed, contrasting with earlier findings [
5]. Only 15% of MMVD dogs were proteinuric (UPC > 0.5), which may relate to patient age, as similar proteinuria prevalence has been documented in apparently healthy geriatric dogs [
52]. Importantly, proteinuria did not worsen with disease progression in our cohort.
FE Na, FE K, and FE Cl increased significantly in clinical stages, consistent with furosemide inhibition of the Na⁺-K⁺-2Cl⁻ cotransporter in the loop of Henle, reducing electrolyte reabsorption and enhancing urinary excretion [
53]. FE values are influenced by the interval between furosemide administration and sample collection. A recent study demonstrated higher FE Na, FE Cl, and FE K shortly after drug administration compared with later intervals, with morning samples showing the highest values [
54]. As our patients were evaluated in the morning, our FE values match those reported for the morning group in that study.
Few studies have assessed urinary biomarkers in MMVD, and only one tubular damage biomarker, urinary neutrophil gelatinase-associated lipocalin (uNGAL), has been reported [
5]. uNGAL is a protein normally filtered by glomerulus and reabsorbed by the tubule, but proximal or distal tubular injury increases its synthesis and secretion, elevating urinary concentrations [
55]. Additional CvRD-related biomarkers should be investigated to improve understanding of tubular pathophysiology in MMVD [
4].
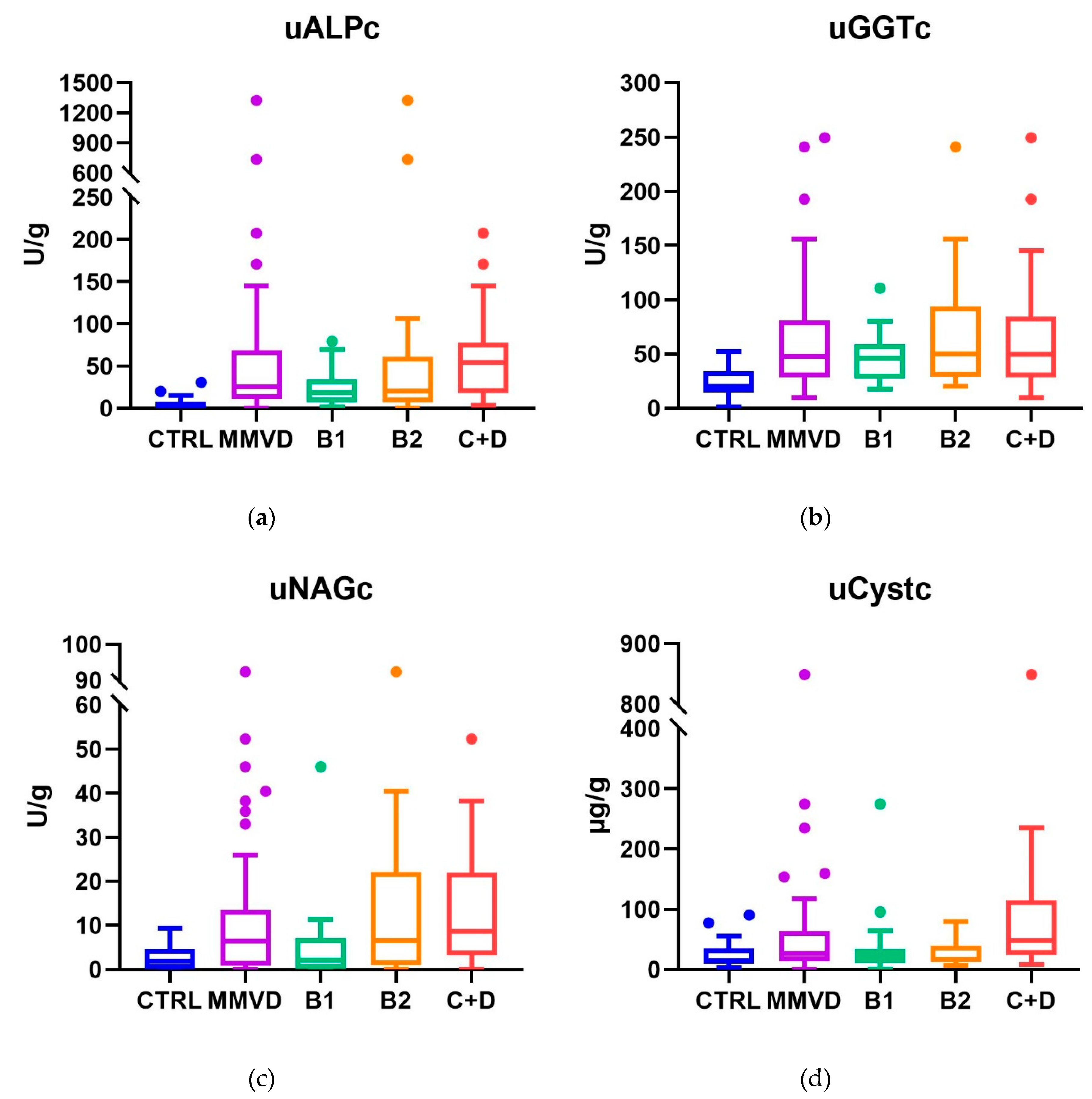
uGGT and uALP are brush-border enzymes released into urine after loss of cytoplasmic membrane integrity, indicating proximal tubular injury [
56]. Both biomarkers can be measured using standard serum/plasma techniques, making them inexpensive and readily applicable in clinical practice [
55]. uNAG is a lysosomal enzyme present in proximal tubular cells normally excreted via lysosomal fusion with the cell membrane. Increased urinary NAG may reflect lysosomal system activation [
57] or tubular damage leading to lysosomal release [
56], and it is a mixed-type marker. Proximal tubular cells are metabolically very active and prone to early damage [
58]. In the absence of tubular injury, urinary concentrations of these enzymes remain very low [
56].
Increases in these three biomarkers are mainly reported in AKI [
13,
18,
59] and they have been correlated with proximal tubular histopathological changes in bitches with pyometra confirmed by renal biopsy [
60]. uNAG and uGGT have also been described in CKD, with early elevations in canine leishmaniosis [
61,
62]. However, Nivy et al. did not detect differences in uGGT and uALP between healthy dogs and those with CKD [
63], and Smets et al. reported substantial overlap in uNAG values between dogs with and without CKD [
64]. This may reflect the fact that not all CKD cases exhibit persistent tubular involvement over time. Following an insult that leads to CKD, once tubular injury subsides, these biomarkers may return to baseline [
65].
In canine MMVD, tubular damage is present from the earliest stages of disease, as demonstrated by increases in uNGAL [
5]. This aligns with our uGGTc and uALPc findings, which showed elevations from stage B1 and exhibited good discriminatory ability between dogs with MMVD and healthy controls, based on ROC curve performance. Furthermore, a strong positive correlation was observed between uGGTc and uALPc, consistent with their shared localization in tubular cells [
56] and similar interpretation of their increase. Notably, uALPc not only increased in early stages but also demonstrated a significant additional rise in advanced stages compared with initial stages, indicating disease progression, similar to uNGAL [
5]. The moderate positive correlations between UPC and both uALPc and uGGTc are consistent with previous findings on uNGAL in dogs with MMVD [
5] and may relate to low-grade proteinuria secondary to impaired proximal tubular reabsorption [
66]. It is unlikely that uALP and uGGT originate from inappropriate passage across the glomerular barrier, given their large molecular size [
56].
Conversely, uNAGc was significantly increased in stages associated with cardiomegaly, a distinction not observed for the other biomarkers in this study nor for uNGAL in Troia et al [
5]. This is supported by its moderate ability to discriminate dogs with cardiomegaly from those without. These findings suggest tubular injury in stage B1 is less severe than in B2, as lysosomal enzymes (uNAG) indicate more pronounced damage than brush-border enzymes (uGGT and uALP) due to their intracellular localization [
66]. This supports the presence of disease progression, with tubular damage in stage B1 and both tubular damage and dysfunction in stages B2, C and D, becoming progressively more pronounced across stages. Additionally, uNAGc correlated with all three others, reinforcing its role in tubular damage and dysfunction [
56,
57].
In humans, increases in uNAG and uNGAL have been reported in stable chronic systolic heart failure. Unlike uNGAL, uNAG provided prognostic information independent of GFR, underscoring the relevance of tubular injury itself in this population [
22]. More recently, uNAG was found to correlate with CHF stage, outcome, HGB and echocardiographic parameters in patients with CHF and mitral regurgitation [
23], supporting uNAG as an important biomarker in cardiorenal syndrome. In contrast, we did not observe a correlation between uNAGc and haemoglobin in our study.
The observed increases in urinary biomarkers without concurrent changes in serum markers during early disease stages highlight their superior ability to detect early renal alterations [
55]. In human AKI, tubular injury may occur without sufficient damage to alter GFR [
67]. This is relevant in CvRD, where renal impairment may result from intermittent cardiac decompensations causing repeated renal insults or from sustained renal injury driven by persistent cardiac dysfunction [
10].
Cystatin C is a protein synthesized at a constant rate by most nucleated cells. It freely passes through the glomerular barrier and is reabsorbed and metabolized by tubular cells. Thus, its urine increase has been associated with tubular dysfunction [
68]. It has been studied mainly in CKD, with increases from early stages of canine leishmaniosis [
62] and proposed as an early CKD indicator, even in non-azotemic stages [
69]. In our study, uCystc showed significant elevation only in clinical stages, possibly reflecting an incipient reduction in GFR already detected by serum markers. This is supported by its moderate ability to discriminate MMVD dogs with stable CHF from those without it. Human patients with acute heart failure and higher proteinuria exhibited greater mortality and higher uCyst concentrations, suggesting an indirect association between uCyst elevation and worse prognosis, likely reflecting more advanced renal dysfunction within the cardiorenal context [
70].
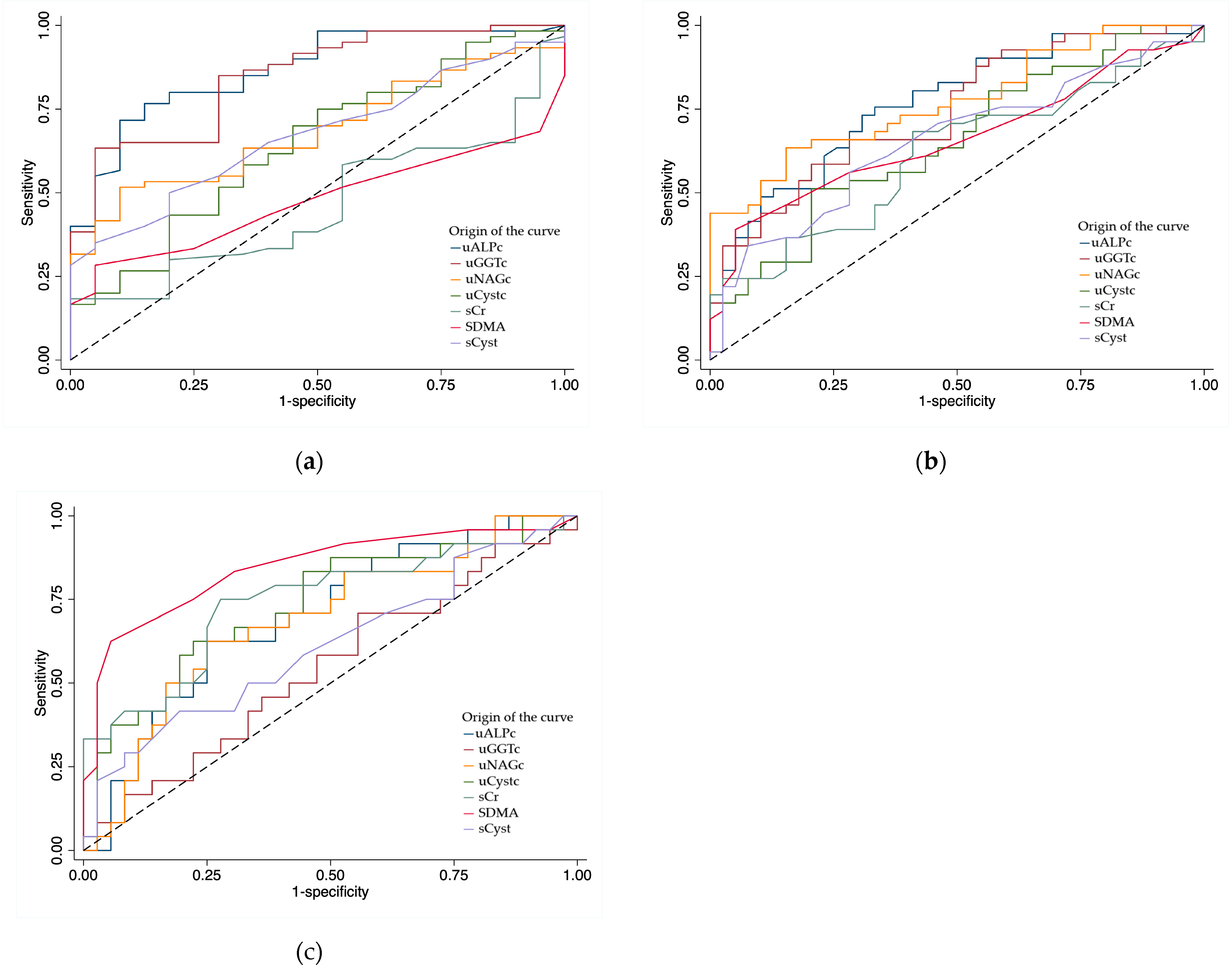
Considering optimal cut-off points, serum markers showed minimal variation across the three successive stages of disease progression (MMVD, cardiomegaly and CHF), with SDMA remaining unchanged. In contrast, urinary biomarkers exhibited a clear upward trend in optimal values, indicating progressive tubular alteration as disease advances. Overall AUC values were higher for urinary than serum biomarkers, and they performed better in the preclinical stages, consistent with previous evidence that tubular biomarkers outperform serum biomarkers for early kidney injury detection [
16,
55]. Serum biomarkers correlated among themselves, as did tubular biomarkers, but no correlations were found between serum and tubular markers, underscoring their distinct clinical significance.
Several mechanisms may contribute to tubular alteration described in CvRD
CH. First, hemodynamic factors such as venous congestion and renal hypoperfusion have been implicated [
4,
28,
71]. Venous congestion, assessed indirectly by echocardiography in human and veterinary medicine, has been associated with increased tubular biomarkers in patients with cardiac disease [
23,
72]. However, in the present study, no differences in SBP were observed between groups, which contrasts with previous findings reporting significant SBP reductions in advanced stages [
73], suggesting renal hypoperfusion as a potential contributor. Although human studies have not demonstrated a correlation between tubular biomarkers and SBP, they have shown associations with reduced ejection fraction [
23], suggesting renal hypoperfusion secondary to episodic, subclinical cardiac events may occur intermittently [
10]. Neurohormonal activation plays a key role, particularly through overactivation of the renin-angiotensin-aldosterone system (RAAS) and sympathetic nervous system [
4,
28,
71]. Current guidelines for canine MMVD, although with a weak level of evidence, strongly recommend angiotensin-converting enzyme inhibitors and spironolactone in clinical stages to inhibit RAAS [
1]. Nevertheless, a recent study found no survival benefit in dogs with MMVD receiving these drugs compared to untreated dogs [
74], indicating that the role of RAAS in this disease remains under debate. Finally, oxidative stress and inflammation should also be considered [
4,
28,
71], the latter being consistent with our leukogram findings. These mechanisms may be present from the early stages and progress alongside disease severity [
10].
Another factor described in CvRD
CH is the administration of diuretics during clinical stages, which may induce hypovolemia and stimulate the RAAS, potentially causing or exacerbating renal injury [
4,
28]. Histopathological changes have been documented in animal models treated with furosemide [
75]. However, in the present study, none of the urinary biomarkers showed a significant association with the daily furosemide dose, suggesting that diuretic therapy is unlikely to play a primary role in CvRD
CH, consistent with previous reports on uNGAL [
5]. Interestingly, human studies have reported that withdrawal of diuretic therapy in patients with CHF led to increases in uNAG, which normalized upon reintroduction of treatment [
76]. These observations further support that the biomarker elevations observed in clinical stages are more likely attributable to the aforementioned pathophysiological mechanisms rather than diuretic use.
The present study has several limitations. The control group was not matched for age and body weight with the dogs with MMVD. Because stage D MMVD has low prevalence and poor life expectancy, only a small number of dogs were included and were pooled with stage C, preventing us from determining whether these subgroups behave similarly. The C+D group received not homogenous treatments. Another constraint is the lack of blinding regarding clinical assessments and laboratory analyses. Knowledge of the dogs' group allocation (control or MMVD) may have biased the findings. Finally, despite evaluating several tubular biomarkers, no complementary tests such as urinary protein electrophoresis were performed, which would have allowed us to determine whether the proteinuria reported had a tubular, glomerular, or mixed origin. Likewise, GFR was not directly measured and renal biopsies were not performed to accurately characterize renal status; mainly due to the invasive nature of these procedures, which were probably declined by most owners.