Submitted:
26 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents and Solvents
2.2. Electrochemical Synthesis of PPy
2.3. UV–Visible Spectroscopy Coupled with Electrochemical Measurements
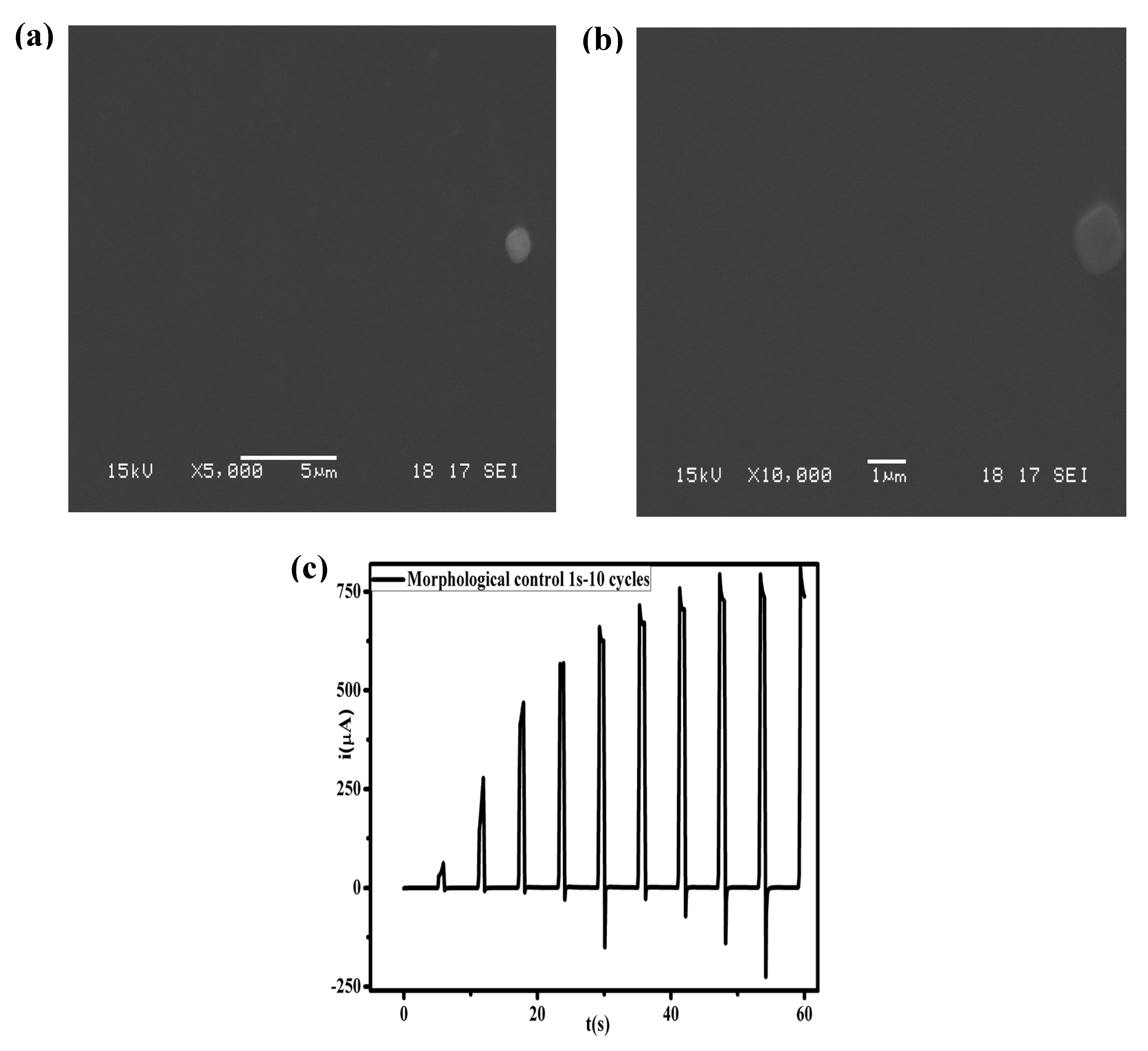
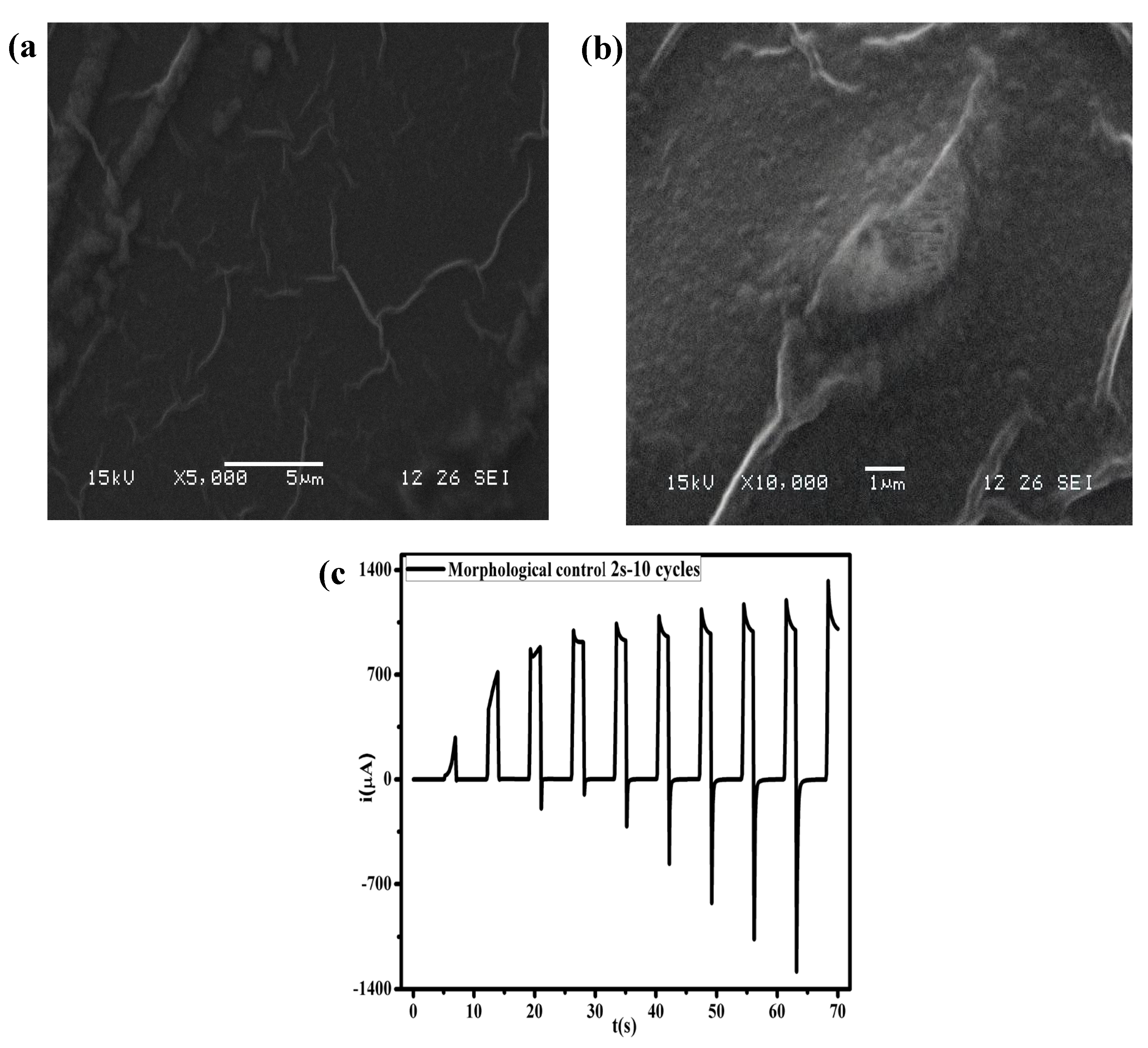
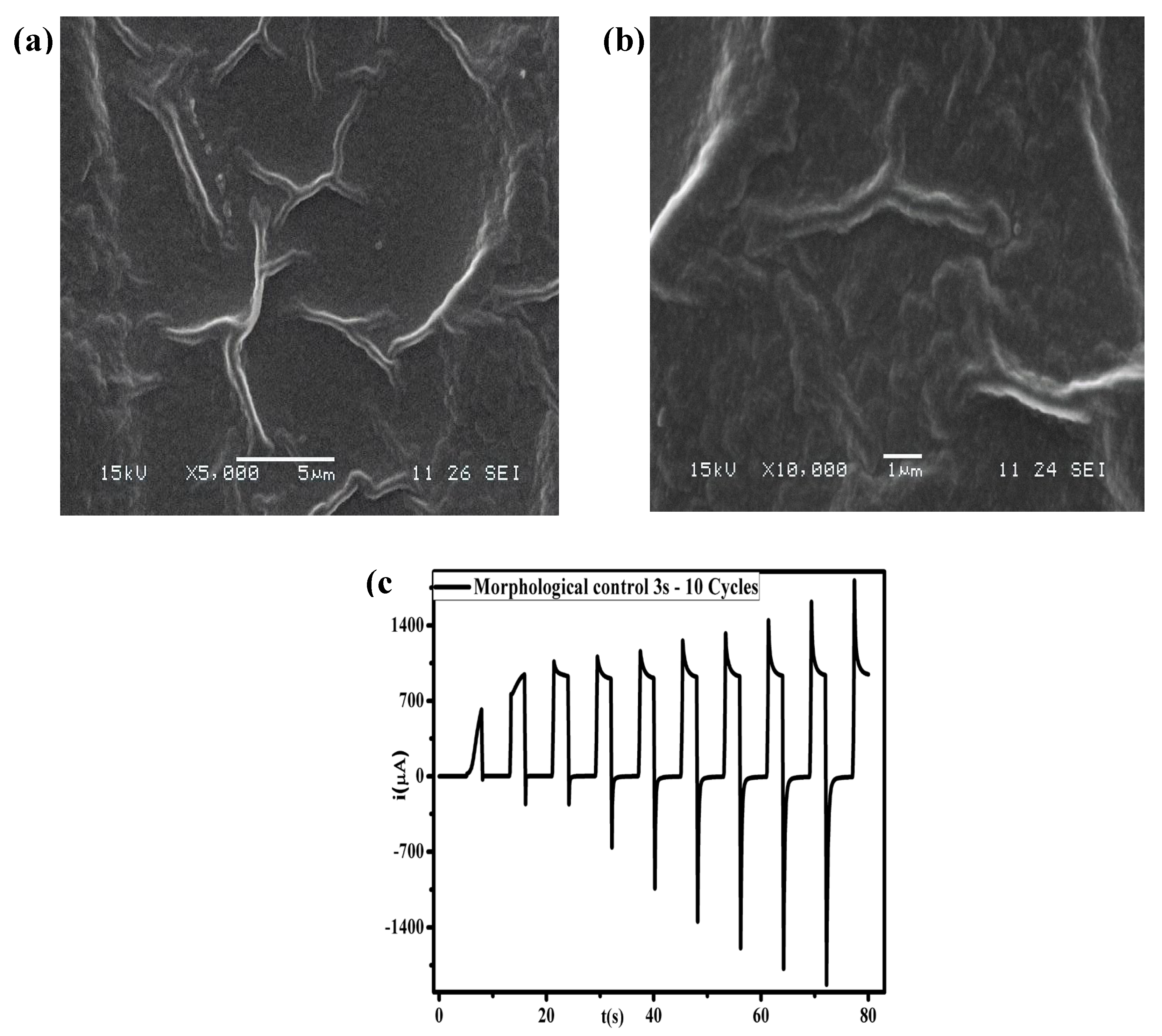
2.4. Scanning Electron Microscopy (SEM)
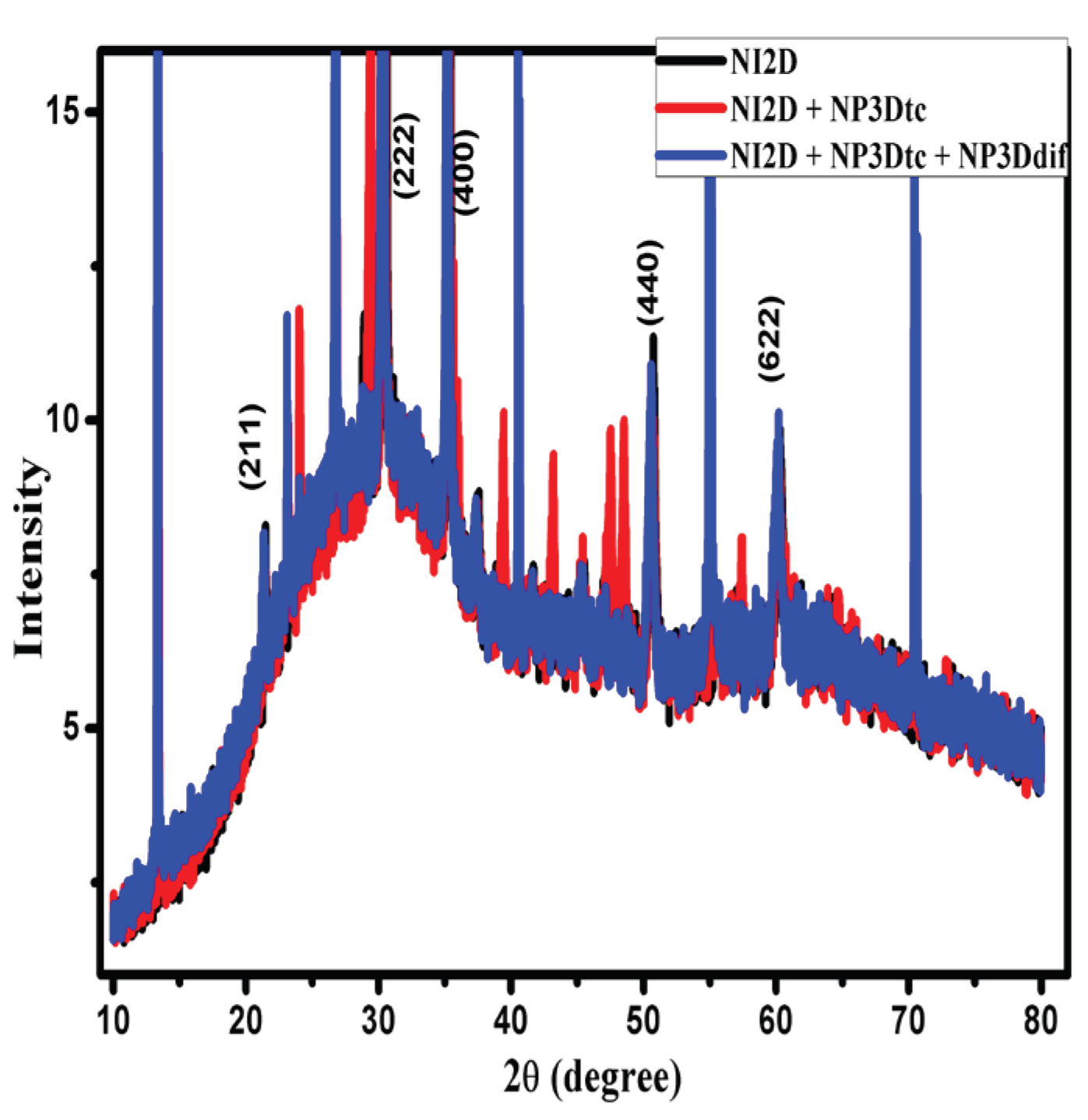
2.5. X-Ray Diffraction Characterization
3. Results
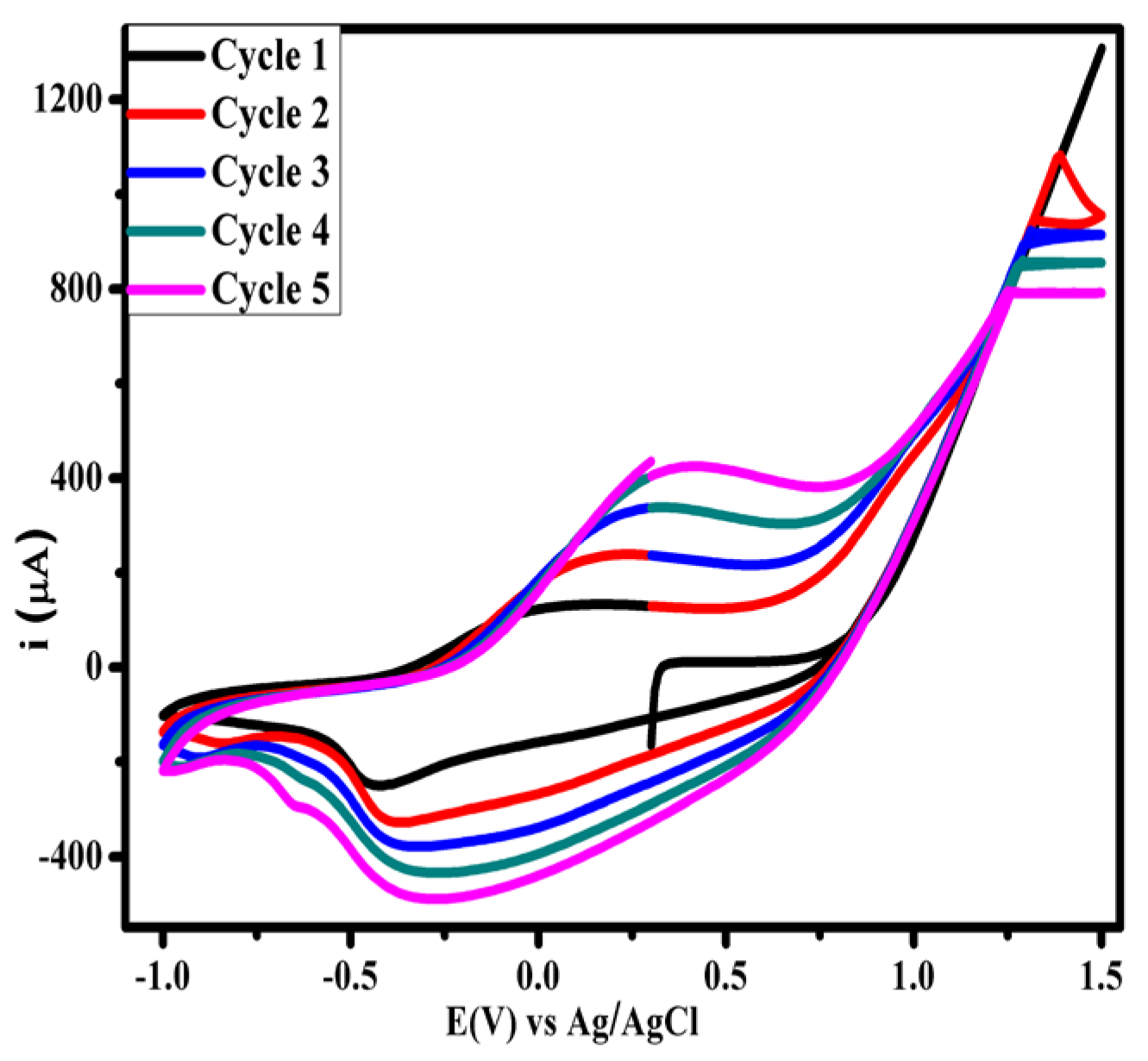
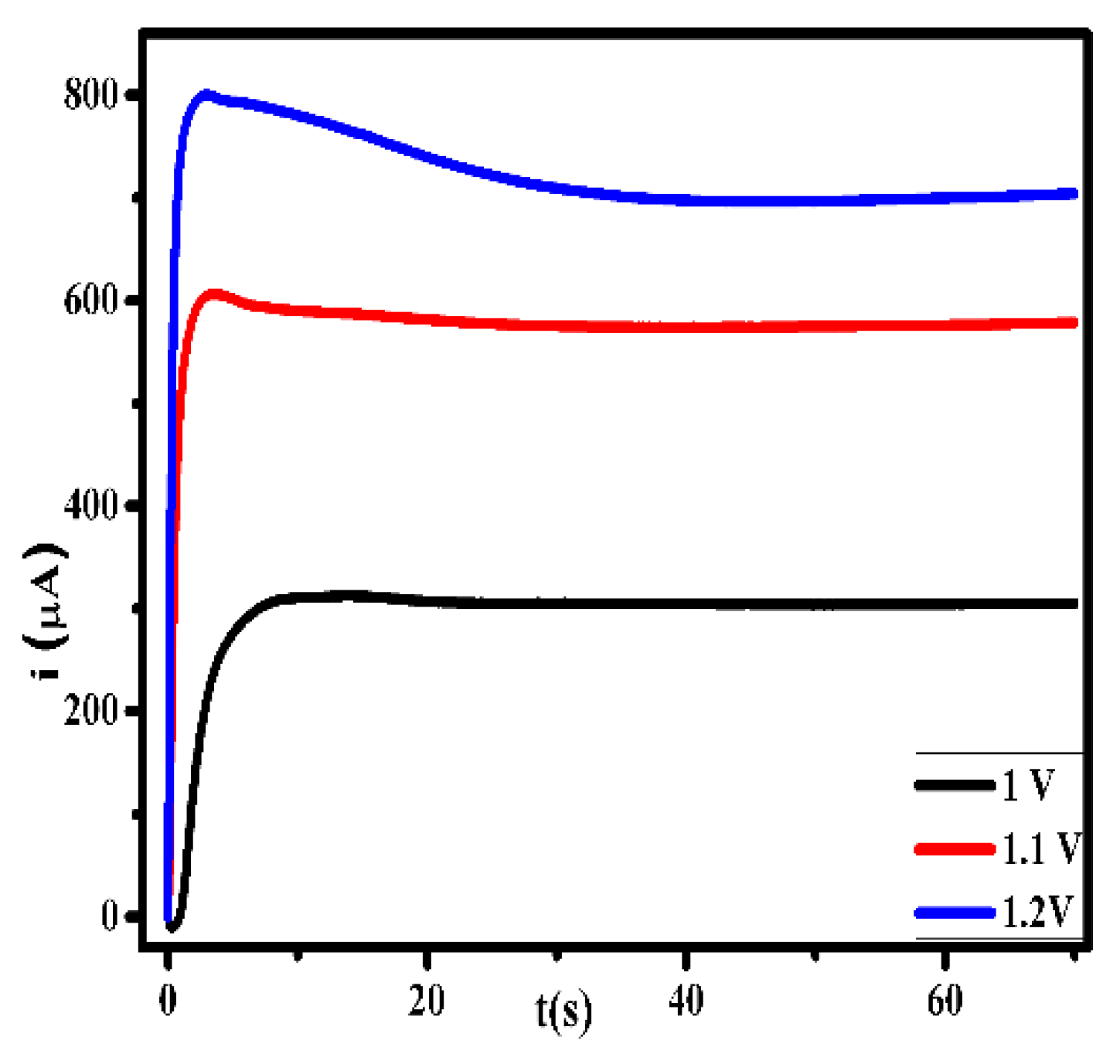
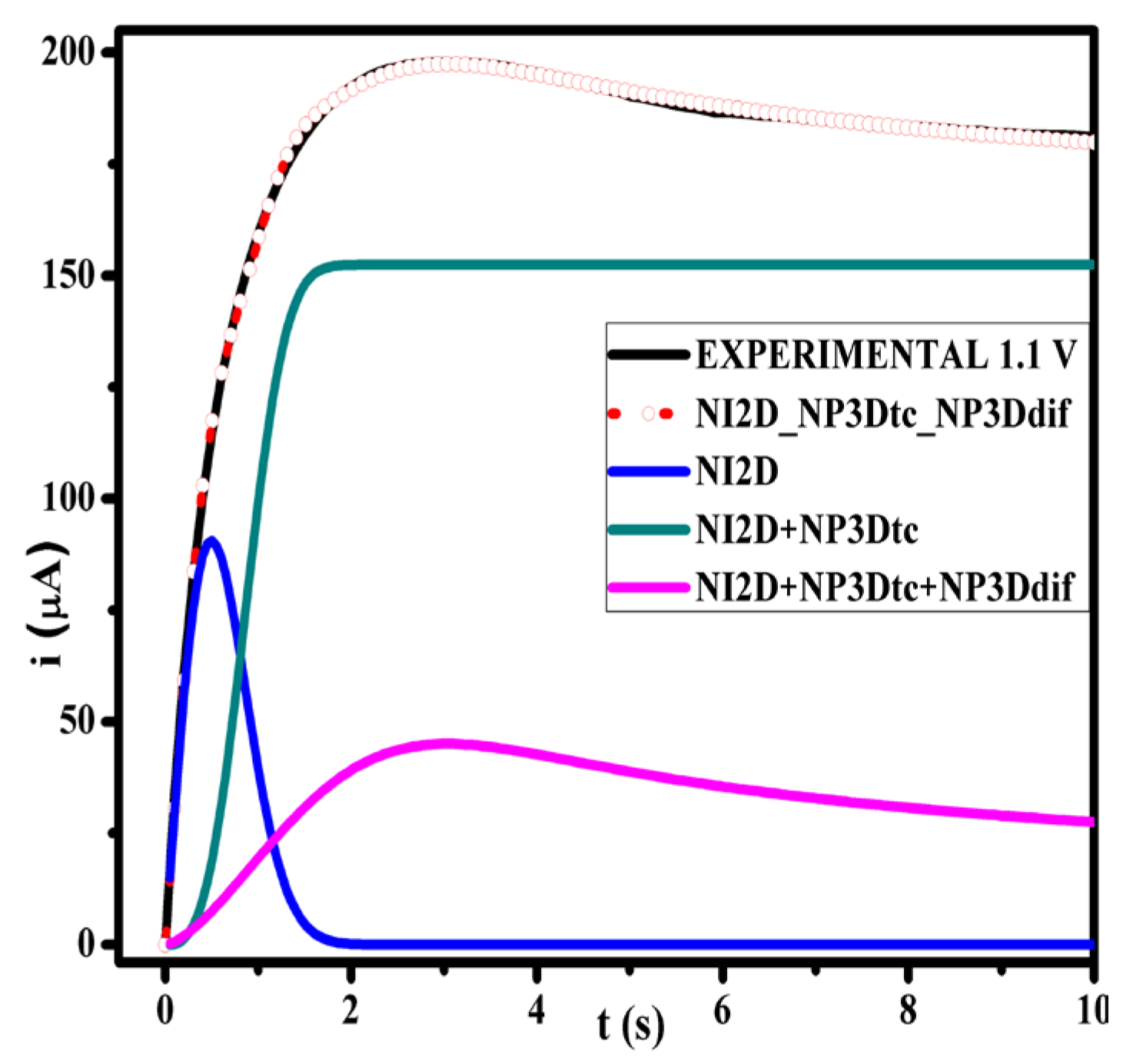
3.1. Optimization of Electropolymerization
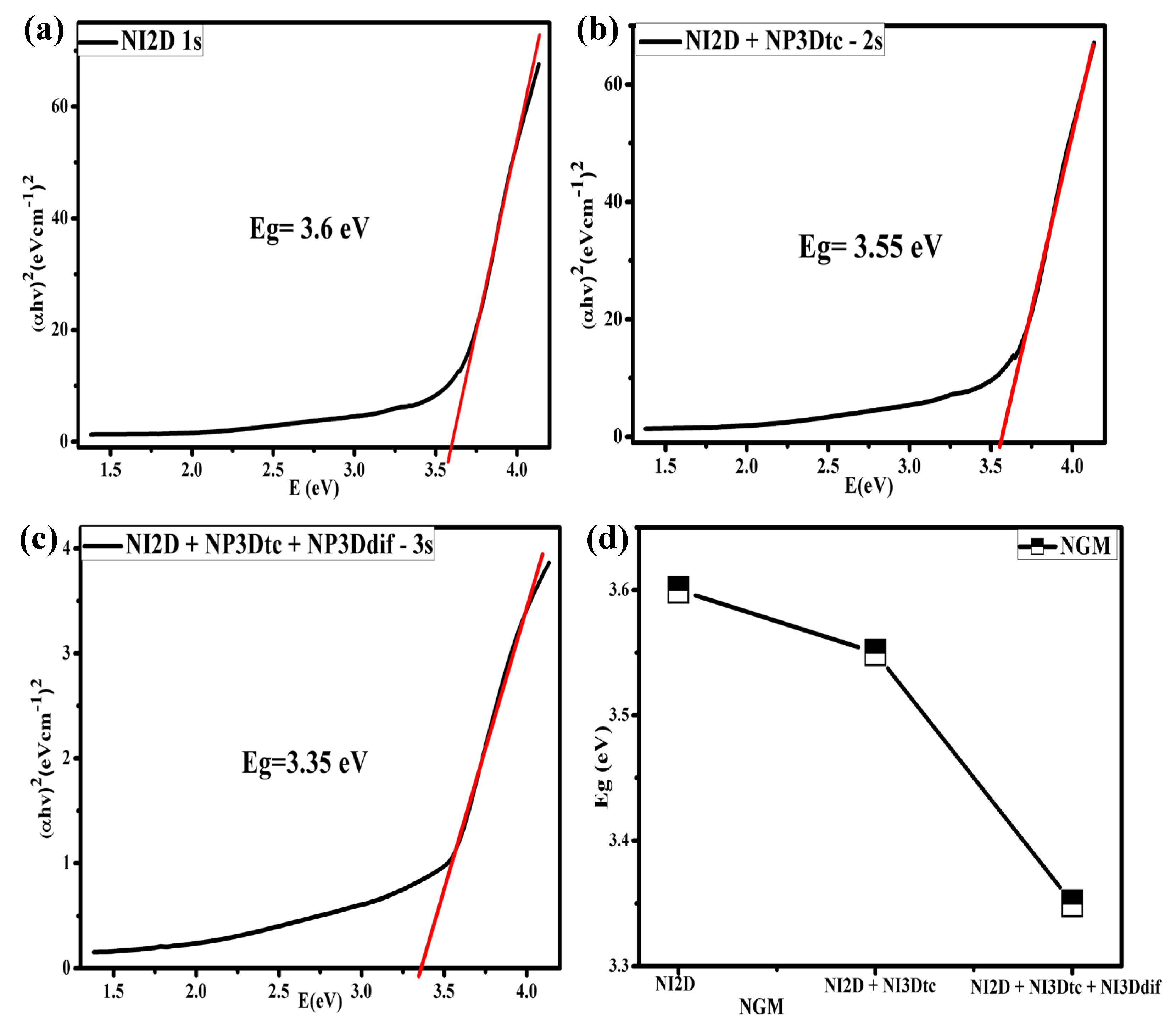
3.2. Optical Band-Gap Calculations by the Tauc Method
3.2.1. Controlled Synthesis
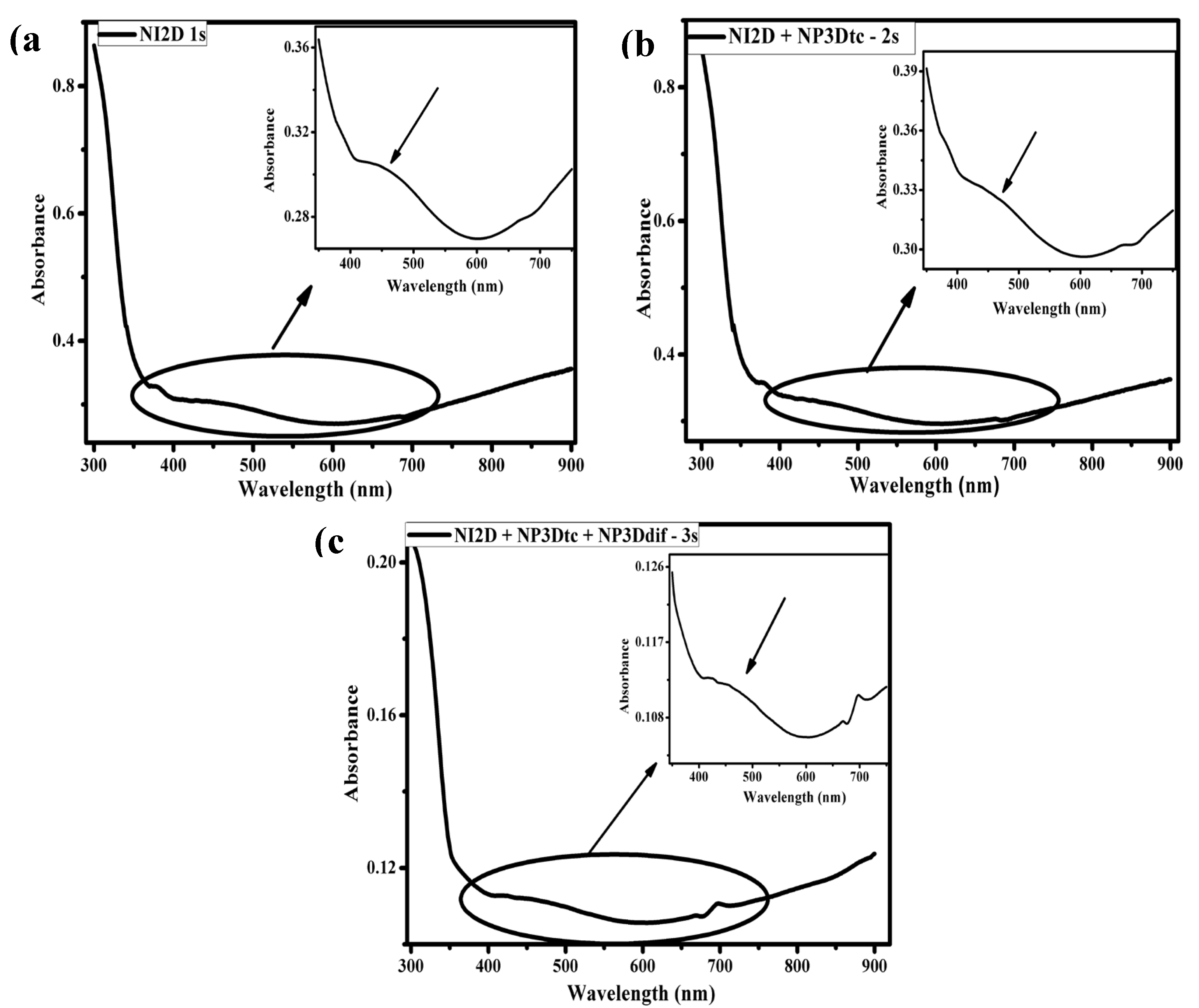
3.2.2. UV–Vis Characterization
3.3. X-Ray Diffraction (XRD) Characterization
4. Conclusions
Funding
Author Contributions
Data Availability Statement
References
- Pratap Khare, K.; Kathal, R.; Shukla, N.; Srivastava, R.; Srivastava, A. Orientation dependent DFT analysis of aniline and pyrrole based copolymer. Materials Today: Proceedings 2020, 47, 6934–6937. [Google Scholar] [CrossRef]
- Balint, R., Cassidy, N. J., & Cartmell, S. H. (2014). Conductive polymers: Towards a smart biomaterial for tissue engineering. In Acta Biomaterialia (Vol. 10, Issue 6, pp. 2341–2353). Elsevier Ltd. [CrossRef]
- Solis, A. B. M. (2019). Síntesis y caracterización de polímeros conductores para su aplicación en dispositivos electrocrómicos duales.
- Duvenhage, M. M.; Ntwaeaborwa, M.; Visser, H. G.; Swarts, P. J.; Swarts, J. C.; Swart, H. C. Determination of the optical band gap of Alq3 and its derivatives for the use in two-layer OLEDs. Optical Materials 2015, 42, 193–198. [Google Scholar] [CrossRef]
- Puerres, J.; Ortiz, P.; Cortés, M. T. Effect of electrosynthesis potential on nucleation, growth, adhesion, and electronic properties of polypyrrole thin films on fluorine-doped tin oxide (FTO). Polymers 2021, 13(15). [Google Scholar] [CrossRef] [PubMed]
- Hwang, B. J.; Santhanam, R.; Lin, Y. L. Nucleation and growth mechanism of electroformation of polypyrrole on a heat-treated gold/highly oriented pyrolytic graphite. Electrochimica acta 2001, 46(18), 2843–2853. [Google Scholar] [CrossRef]
- Roldán, L. Determination of the band gap in semiconductor materials using UV-Vis spectroscopy. Journal of Applied Chemistry 2014, 11(3), 56–62. [Google Scholar]
- Bayat, M.; Izadan, H.; Santiago, S.; Estrany, F.; Dinari, M.; Semnani, D.; Alemán, C.; Guirado, G. Study on the electrochromic properties of polypyrrole layers doped with different dye molecules. Journal of Electroanalytical Chemistry 2021a, 886. [Google Scholar] [CrossRef]
- Marín, T. A.; Isaza, F. J.; Calderón, J. A. Electrodeposición de Películas de polipirrol/platino. Portugaliae Electrochimica Acta 2009, 27(3), 397–407. [Google Scholar] [CrossRef]
- Sadki, S.; Schottland, P.; Brodie, N.; Sabouraud, G. The mechanisms of pyrrole electropolymerization. Chemical Society Reviews 2000, 29(5), 283–293. [Google Scholar] [CrossRef]
- Monk, P., Mortimer, R. y Rosseinsky, D. (2007). Electrocromismo y dispositivos electrocrómicos . Cambridge University Press.
- Díaz, F. R.; Del Valle, M. A.; Núñez, C.; Godoy, A.; Mondaca, J. L.; Toro-Labbé, A.; Bernède, J. C. Synthesis, characterization, electropolymerization, and theoretical study of 2,3-di-(2-thienyl)quinoxaline. Polymer Bulletin 2006, 56(2–3), 155–162. [Google Scholar] [CrossRef]
- Ávalos Huarte, E. (2020). Mechanism and kinetics of nucleation and electrochemical growth of polypyrrole from deep eutectic solvents. Retrieved from: https://hdl.handle.net/11191/7479.
- Morávková, Z.; Taboubi, O.; Minisy, I. M.; Bober, P. The evolution of the molecular structure of polypyrrole during chemical polymerization. Synthetic Metals 2021, 271. [Google Scholar] [CrossRef]
- Yun, S. R.; Kim, G. O.; Lee, C. W.; Jo, N. J.; Kang, Y.; Ryu, K. S. Synthesis and control of the shell thickness of polyaniline and polypyrrole half hollow spheres using the polystyrene cores. Journal of Nanomaterials 2012. [Google Scholar] [CrossRef]
- Viezbicke, B. D.; Patel, S.; Davis, B. E.; Birnie, D. P. Evaluation of the Tauc method for optical absorption edge determination: ZnO thin films as a model system. Physica Status Solidi (B) Basic Research 2015, 252(8), 1700–1710. [Google Scholar] [CrossRef]
- Samwang, T.; Watanabe, N. M.; Okamoto, Y.; Srinives, S.; Umakoshi, H. Study of Chemical Polymerization of Polypyrrole with SDS Soft Template: Physical, Chemical, and Electrical Properties. ACS Omega 2023, 8(51), 48946–48957. [Google Scholar] [CrossRef] [PubMed]
- Castro-Beltrán, A.; Domínguez, C.; Bahena-Uribe, D.; Sepúlveda-Guzmán, S.; Cruz-Silva, R. Efecto de aditivos no electroactivos en la electropolimerización de pirrol en etapa temprana sobre electrodos de óxido de indio y estaño. Thin Solid Films 2014, 566, 23–31. [Google Scholar]
- Arjomandi, J.; Raoufi, D.; Ghamari, F. Surface Characterization and Morphology of Conducting Polypyrrole Thin Films during Polymer Growth on ITO Glass Electrode. Journal of Physical Chemistry C 2016, 120(32), 18055–18065. [Google Scholar] [CrossRef]
- Alsultany, F. H.; Ahmed, N. M.; Matjafri, M. Z. Effects of CW CO<sub>2</sub> Laser Annealing on Indium Tin Oxide Thin Films Characteristics. Soft Nanoscience Letters 2014, 04(04), 83–89. [Google Scholar] [CrossRef]
- Eshaghi, A.; Graeli, A. Optical and electrical properties of indium tin oxide (ITO) nanostructured thin films deposited on polycarbonate substrates thickness effect. Optik 2014, 125(3), 1478–1481. [Google Scholar] [CrossRef]
- Chougule, M. A.; Pawar, S. G.; Godse, P. R.; Mulik, R. N.; Sen, S.; Patil, V. B. Synthesis and Characterization of Polypyrrole (PPy) Thin Films. Soft Nanoscience Letters 2011, 01(01), 6–10. [Google Scholar] [CrossRef]
- Kaur Sidhu, G.; Kumar, R. Study the Structural and Optical behaviour of Conducting Polymer based nanocomposites: ZrO2-Polypyrrole Nanocomposites. IOP Conference Series: Materials Science and Engineering 2018, 360(1). [Google Scholar] [CrossRef]
- Mitchell, G. R., Geris, A., Rg6, S. J. J., & Uk, T. 2af. (1987). Molecular organisation of electrochemically prepared conducting polypyrrole films Printed in the UK. In J. Phys. D: Appl. Phys (Vol. 20). http://iopscience.iop.org/0022-3727/20/11/002.
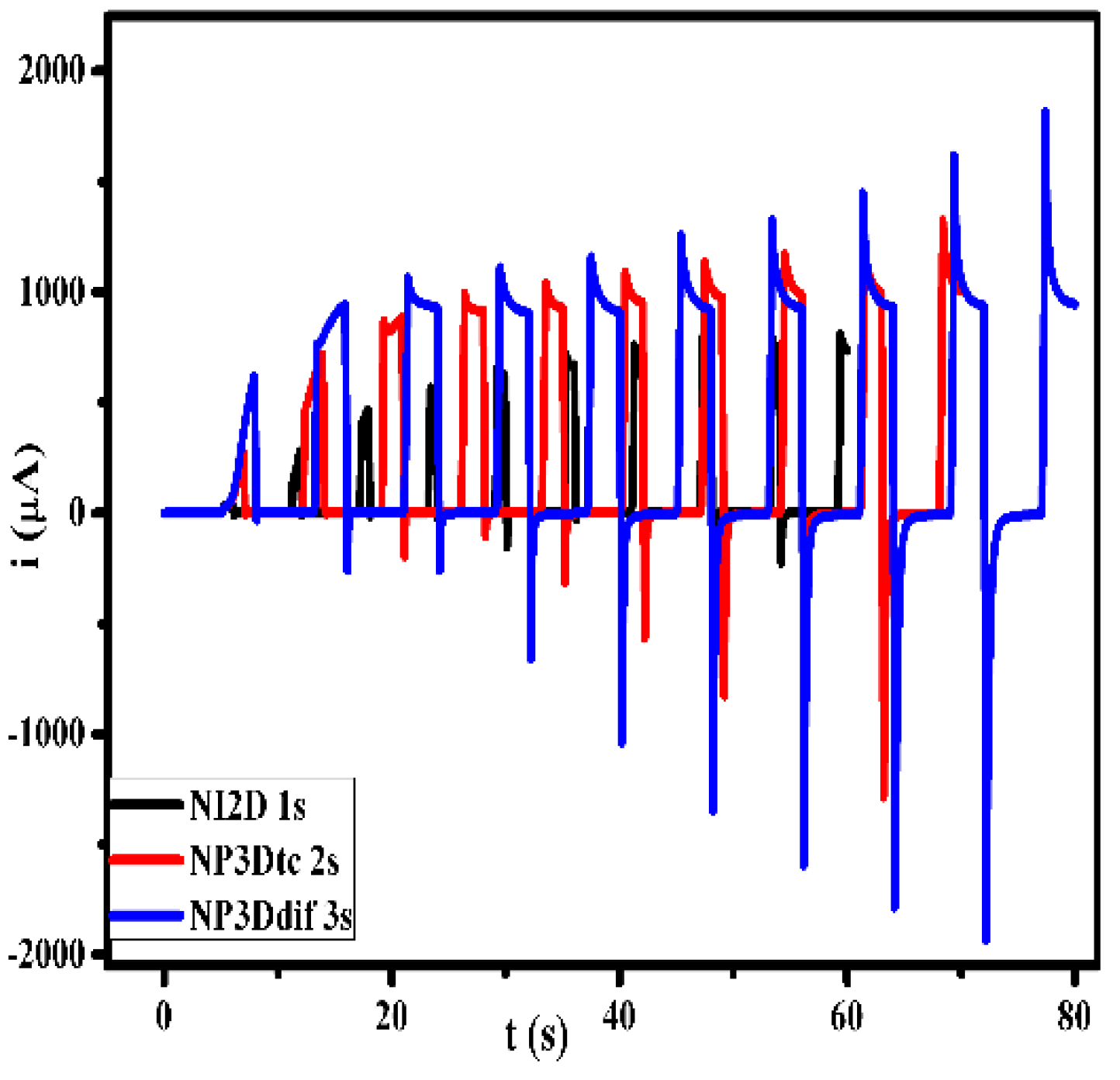

| Nucleation | Geometry | Mathematical expression |
| NI2D |  |
|
| NP2D |  |
|
| NI3Dtc |  |
|
| NP3Dtc |  |
|
| NI3Ddif |  |
|
| NP3Ddif |
| Polímero | E(V) | P1 | P2 | P3 | P4 | P5 | P6 | R2 |
| PPy | 1.1 | 300.761 | 2.028 | 152.403 | 1.0495 | 86.920 | 0.2529 | 0.999 |
| P1 | P2 | P3 | P4 | P5 | P6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




