Submitted:
22 January 2026
Posted:
26 January 2026
You are already at the latest version
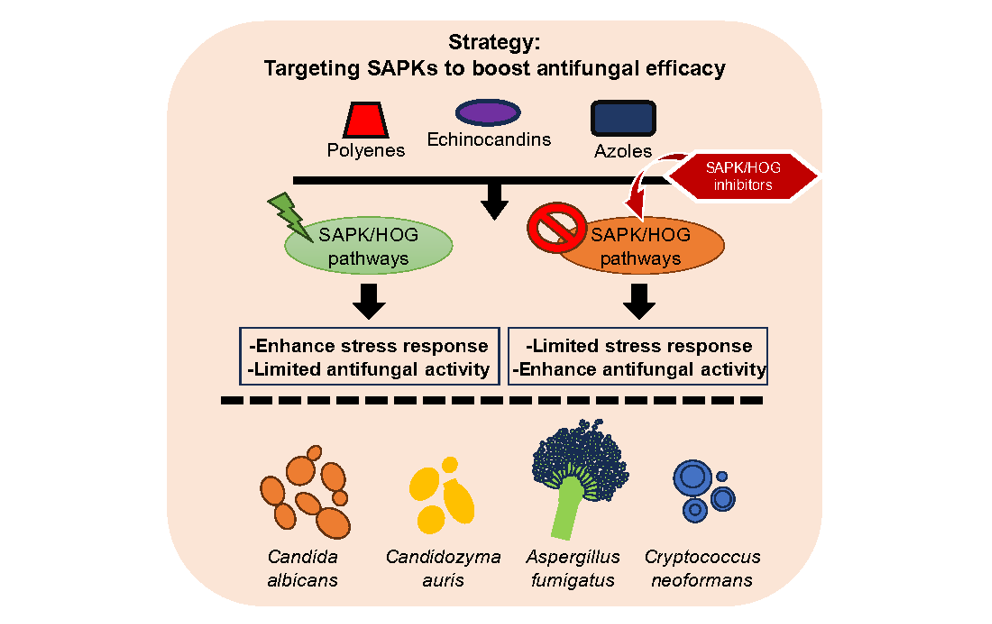
Abstract
Keywords:
1. Introduction
1.1. Polyenes
1.2. Azoles
1.3. Echinocandins
2. The Search for New Antifungal Targets Is a Therapeutic Need
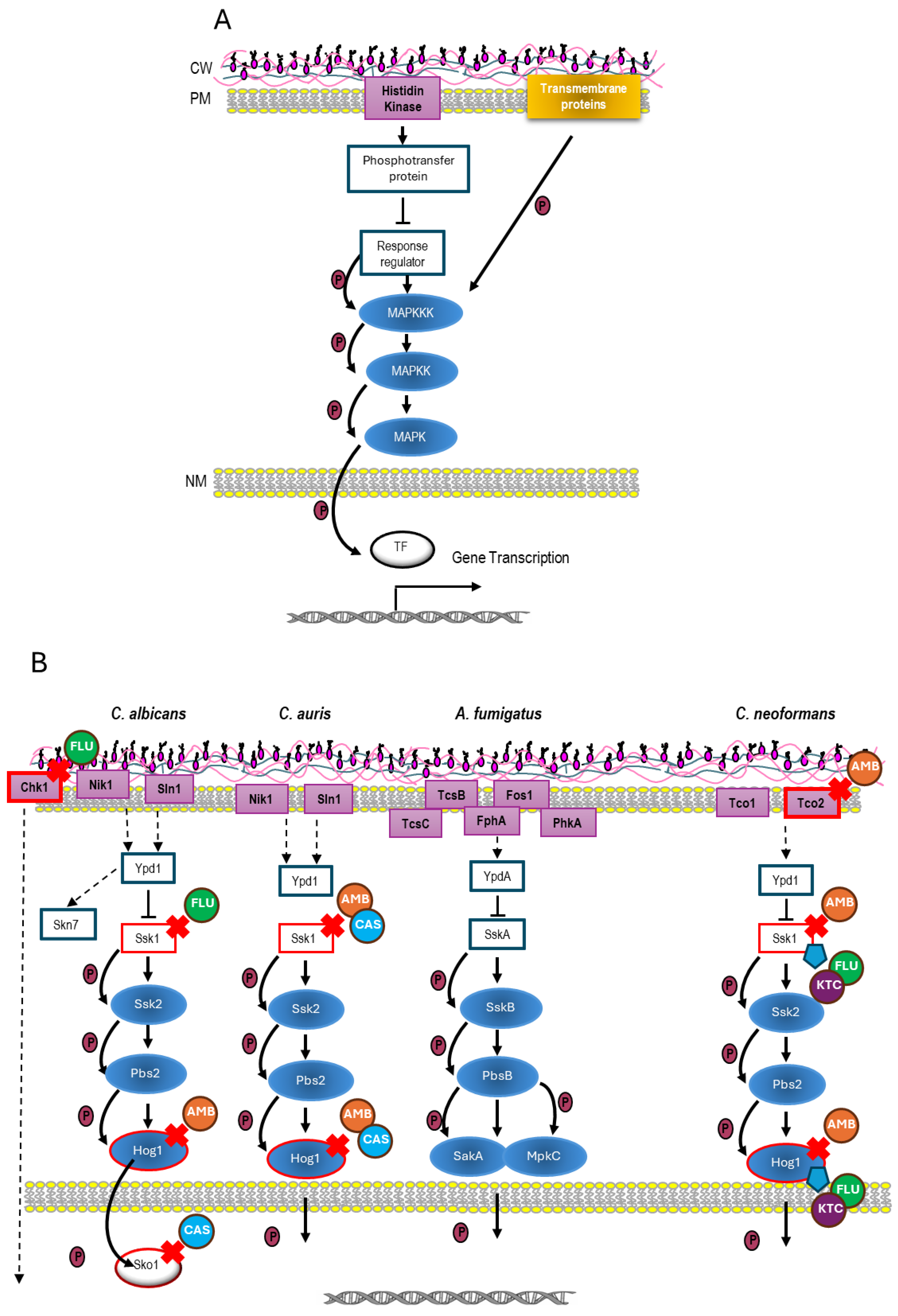
3. Sensing Antifungals by SAPK Pathways in WHO High-Priority Fungal Pathogens
3.1. SAPK Pathway in C. albicans: The HOG Pathway
3.2. SAPK Pathway in Aspergillus fumigatus: The SakA and MpkC Mediated Pathway
3.3. SAPK Pathway in Cryptococcus neoformans: The HOG Pathway
4. Histidine Kinase Inhibitors Under Study
5. Conclusions and Perspectives.
Funding
Acknowledgments
Conflicts of Interest
Abbreviations:
References
- Kainz, K.; Bauer, M.A.; Madeo, F.; Carmona-Gutierrez, D. Fungal infections in humans: The silent crisis. Microb Cell 2020, 7, 143–145. [Google Scholar] [CrossRef]
- Denning, D.W. Global incidence and mortality of severe fungal disease. Lancet Infect Dis 2024, 24, e428–e438. [Google Scholar] [CrossRef] [PubMed]
- Organization, W.H. World Health Organization (WHO) Report. WHO fungal priority pathogens list to guide research, development and public health action. 2022. [Google Scholar]
- Arguelles, J.C.; Sanchez-Fresneda, R.; Arguelles, A.; Solano, F. Natural Substances as Valuable Alternative for Improving Conventional Antifungal Chemotherapy: Lights and Shadows. J Fungi 2024, 10, 334. [Google Scholar] [CrossRef]
- Brown, G.D.; Denning, D.W.; Gow, N.A.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci Transl Med 2012, 4, 165rv113. [Google Scholar] [CrossRef]
- Jacobs, S.E.; Jacobs, J.L.; Dennis, E.K.; Taimur, S.; Rana, M.; Patel, D.; Gitman, M.; Patel, G.; Schaefer, S.; Iyer, K.; et al. Candida auris Pan-Drug-Resistant to Four Classes of Antifungal Agents. Antimicrob Agents Chemother 2022, 66, e0005322. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Barat, S.A.; Borroto-Esoda, K.; Angulo, D.; Chaturvedi, S.; Chaturvedi, V. Pan-resistant Candida auris isolates from the outbreak in New York are susceptible to ibrexafungerp (a glucan synthase inhibitor). Int J Antimicrob Agents 2020, 55, 105922. [Google Scholar] [CrossRef]
- Parsons, M.G.; Diekema, D.J. What Is New in Fungal Infections? Mod Pathol 2023, 36, 100187. [Google Scholar] [CrossRef]
- Pfaller, M.A.; Diekema, D.J. Epidemiology of invasive candidiasis: A persistent public health problem. Clinical Microbiological Reviews 2007, 20, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Pfaller, M.A.; Messer, S.A.; Deshpande, L.M.; Rhomberg, P.R.; Utt, E.A.; Castanheira, M. Antifungal drugs work together to treat germs causing fungal infections. Future Microbiol 2021, 16, 765–768. [Google Scholar] [CrossRef] [PubMed]
- Perlin, D.S.; Wiederhold, N.P. Culture-Independent Molecular Methods for Detection of Antifungal Resistance Mechanisms and Fungal Identification. J Infect Dis 2017, 216, S458–S465. [Google Scholar] [CrossRef]
- Perlin, D.S.; Rautemaa-Richardson, R.; Alastruey-Izquierdo, A. The global problem of antifungal resistance: Prevalence, mechanisms, and management. Lancet Infect Dis 2017, 17, E383–E392. [Google Scholar] [CrossRef]
- Vanreppelen, G.; Wuyts, J.; Van Dijck, P.; Vandecruys, P. Sources of Antifungal Drugs. J Fungi 2023, 9, 171. [Google Scholar] [CrossRef] [PubMed]
- Campoy, S.; Adrio, J.L. Antifungals. Biochem Pharmacol 2017, 133, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, M.; Ciric, A.; Stojkovic, D. Emerging Antifungal Targets and Strategies. Int J Mol Sci 2022, 23, 2756. [Google Scholar] [CrossRef]
- Vincent, B.M.; Lancaster, A.K.; Scherz-Shouval, R.; Whitesell, L.; Lindquist, S. Fitness Trade-offs Restrict the Evolution of Resistance to Amphotericin B. PLoS Biology 2013, 11, e1001692. [Google Scholar] [CrossRef]
- Cohen, B.E. Amphotericin B membrane action: Role for two types of ion channels in eliciting cell survival and lethal effects. J Membr Biol 2010, 238, 1–20. [Google Scholar] [CrossRef]
- Ermishkin, L.N.; Kasumov, K.M.; Potseluyev, V.M. Properties of amphotericin B channels in a lipid bilayer. Biochim Biophys Acta 1977, 470, 357–367. [Google Scholar] [CrossRef]
- Ermishkin, L.N.; Kasumov, K.M.; Potzeluyev, V.M. Single ionic channels induced in lipid bilayers by polyene antibiotics amphotericin B and nystatine. Nature 1976, 262, 698–699. [Google Scholar] [CrossRef]
- Gray, K.C.; Palacios, D.S.; Dailey, I.; Endo, M.M.; Uno, B.E.; Wilcock, B.C.; Burke, M.D. Amphotericin primarily kills yeast by simply binding ergosterol. Proc Natl Acad Sci U S A 2012, 109, 2234–2239. [Google Scholar] [CrossRef] [PubMed]
- Denning, D.W. Antifungal drug resistance: An update. Eur J Hosp Pharm 2022, 29, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Francois, I.E.; Aerts, A.M.; Cammue, B.P.; Thevissen, K. Currently used antimycotics: Spectrum, mode of action and resistance occurrence. Curr.Drug Targets 2005, 6, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Vazquez, J.A. An evaluation of Rezafungin: The latest treatment option for adults with candidemia and invasive candidiasis. Expert Opin Pharmacother 2024, 25, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Perlin, D.S. Mechanisms of echinocandin antifungal drug resistance. Ann Ny Acad Sci 2015, 1354, 1–11. [Google Scholar] [CrossRef]
- Suwunnakorn, S.; Wakabayashi, H.; Kordalewska, M.; Perlin, D.S.; Rustchenko, E. FKS2 and FKS3 Genes of Opportunistic Human Pathogen Candida albicans Influence Echinocandin Susceptibility. Antimicrob Agents Chemother 2018, 62. [Google Scholar] [CrossRef]
- Fisher, M.C.; Alastruey-Izquierdo, A.; Berman, J.; Bicanic, T.; Bignell, E.M.; Bowyer, P.; Bromley, M.; Bruggemann, R.; Garber, G.; Cornely, O.A.; et al. Tackling the emerging threat of antifungal resistance to human health. Nat Rev Microbiol 2022, 20, 557–571. [Google Scholar] [CrossRef]
- Lu, X.; Zhou, J.; Ming, Y.; Wang, Y.; He, R.; Li, Y.; Feng, L.; Zeng, B.; Du, Y.; Wang, C. Next-generation antifungal drugs: Mechanisms, efficacy, and clinical prospects. Acta Pharm Sin B 2025, 15, 3852–3887. [Google Scholar] [CrossRef]
- Lim, W.; Nyuykonge, B.; Eadie, K.; Konings, M.; Smeets, J.; Fahal, A.; Bonifaz, A.; Todd, M.; Perry, B.; Samby, K.; et al. Screening the pandemic response box identified benzimidazole carbamates, Olorofim and ravuconazole as promising drug candidates for the treatment of eumycetoma. PLoS Negl Trop Dis 2022, 16, e0010159. [Google Scholar] [CrossRef]
- McKenna, M. This ‘super gonorrhoea’ drug holds a lesson for avoiding microbial armageddon. Nature 2024, 626, 942–944. [Google Scholar] [CrossRef]
- Chen, R.E.; Thorner, J. Function and regulation in MAPK signaling pathways: Lessons learned from the yeast Saccharomyces cerevisiae. Biochim Biophys Acta 2007, 1773, 1311–1340. [Google Scholar] [CrossRef]
- Ma, Y.; Nicolet, J. Specificity models in MAPK cascade signaling. FEBS Open Bio 2023, 13, 1177–1192. [Google Scholar] [CrossRef]
- Widmann, C.; Gibson, S.; Jarpe, M.B.; Johnson, G.L. Mitogen-activated protein kinase: Conservation of a three-kinase module from yeast to human. Physiol Rev. 1999, 79, 143–180. [Google Scholar] [CrossRef]
- Alonso-Monge, R.; Román, E.; Pla, J.; Nombela, C. A host view of the fungal cell wall. In Evolutionary biology of bacterial and fungal pathogens; Baquero Mochales, F., Nombela, C., Cassel, G.H., Gutiérrez-Fuentes, J.A., Eds.; ASM Press: Washington, D.C., 2009; pp. 105–115. [Google Scholar]
- Day, A.M.; Quinn, J. Stress-Activated Protein Kinases in Human Fungal Pathogens. Front Cell Infect Microbiol 2019, 9, 261. [Google Scholar] [CrossRef]
- Kultz, D.; Burg, M. Evolution of osmotic stress signaling via MAP kinase cascades. J.Exp.Biol. 1998, 201, 3015–3021. [Google Scholar] [CrossRef]
- Underhill, D.M.; Iliev, I.D. The mycobiota: Interactions between commensal fungi and the host immune system. Nat Rev Immunol 2014, 14, 405–416. [Google Scholar] [CrossRef]
- Calera, J.A.; Zhao, X.J.; Calderone, R. Defective hyphal development and avirulence caused by a deletion of the SSK1 response regulator gene inCandida albicans. Infection and Immunity 2000, 68, 518–525. [Google Scholar] [CrossRef]
- Calera, J.A.; Calderone, R. Histidine kinase, two-component signal transduction proteins ofCandida albicans and the pathogenesis of candidosis. Mycoses 1999, 42, 49–53. [Google Scholar] [CrossRef]
- Chauhan, N.; Inglis, D.; Román, E.; Pla, J.; Li, D.; Calera, J.A.; Calderone, R. Candida albicans response regulator gene SSK1 regulates a subset of genes whose functions are associated with cell wall biosynthesis and adaptation to oxidative stress. Eukaryotic Cell 2003, 2, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Monge, R.; Navarro-García, F.; Molero, G.; Díez-Orejas, R.; Gustin, M.; Pla, J.; Sánchez, M.; Nombela, C. Role of the mitogen-activated protein kinase Hog1p in morphogenesis and virulence of Candida albicans. Journal of Bacteriology 1999, 181, 3058–3068. [Google Scholar] [CrossRef] [PubMed]
- Arana, D.M.; Alonso-Monge, R.; Du, C.; Calderone, R.; Pla, J. Differential susceptibility of mitogen-activated protein kinase pathway mutants to oxidative-mediated killing by phagocytes in the fungal pathogen Candida albicans. Cellular Microbiology 2007, 9, 1647–1659. [Google Scholar] [CrossRef] [PubMed]
- Vylkova, S.; Jang, W.S.; Li, W.; Nayyar, N.; Edgerton, M. Histatin 5 initiates osmotic stress response in Candida albicans via activation of the Hog1 mitogen-activated protein kinase pathway. Eukaryot Cell 2007, 6, 1876–1888. [Google Scholar] [CrossRef]
- Day, A.M.; Smith, D.A.; Ikeh, M.A.; Haider, M.; Herrero-de-Dios, C.M.; Brown, A.J.; Morgan, B.A.; Erwig, L.P.; MacCallum, D.M.; Quinn, J. Blocking two-component signalling enhances Candida albicans virulence and reveals adaptive mechanisms that counteract sustained SAPK activation. PLoS Pathog 2017, 13, e1006131. [Google Scholar] [CrossRef]
- Guirao-Abad, J.P.; Sanchez-Fresneda, R.; Roman, E.; Pla, J.; Arguelles, J.C.; Alonso-Monge, R. The MAPK Hog1 mediates the response to amphotericin B in Candida albicans. Fungal Genet Biol 2020, 136, 103302. [Google Scholar] [CrossRef]
- Kelly, J.; Rowan, R.; McCann, M.; Kavanagh, K. Exposure to caspofungin activates Cap and Hog pathways in Candida albicans. Med.Mycol 2009, 47, 697–706. [Google Scholar] [CrossRef]
- Alonso-Monge, R.; Guirao-Abad, J.P.; Sanchez-Fresneda, R.; Pla, J.; Yague, G.; Arguelles, J.C. The Fungicidal Action of Micafungin is Independent on Both Oxidative Stress Generation and HOG Pathway Signaling in Candida albicans. Microorganisms 2020, 8, 1867. [Google Scholar] [CrossRef]
- Guirao-Abad, J.P.; Sanchez-Fresneda, R.; Alburquerque, B.; Hernandez, J.A.; Arguelles, J.C. ROS formation is a differential contributory factor to the fungicidal action of Amphotericin B and Micafungin in Candida albicans. Int J Med Microbiol 2017, 307, 241–248. [Google Scholar] [CrossRef]
- Heredia, M.Y.; Gunasekaran, D.; Ikeh, M.A.C.; Nobile, C.J.; Rauceo, J.M. Transcriptional regulation of the caspofungin-induced cell wall damage response in Candida albicans. Curr Genet 2020, 66, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, N.; Kruppa, M.; Calderone, R. The Ssk1p response regulator and Chk1p histidine kinase mutants of Candida albicans are hypersensitive to fluconazole and voriconazole. Antimicrob Agents Chemother 2007, 51, 3747–3751. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Effron, G.; Dilger, A.; Alcazar-Fuoli, L.; Park, S.; Mellado, E.; Perlin, D.S. Rapid detection of triazole antifungal resistance in Aspergillu fumigatus. J Clin Microbiol 2008, 46, 1200–1206. [Google Scholar] [CrossRef] [PubMed]
- Perlin, D.S.; Mellado, E. Antifungal Mechanisms of Action and Resistance. Aspergillus Fumigatus and Aspergillosis 2009, 457–466. [Google Scholar]
- Bruder Nascimento, A.C.; Dos Reis, T.F.; de Castro, P.A.; Hori, J.I.; Bom, V.L.; de Assis, L.J.; Ramalho, L.N.; Rocha, M.C.; Malavazi, I.; Brown, N.A.; et al. Mitogen activated protein kinases SakA(HOG1) and MpkC collaborate for Aspergillus fumigatus virulence. Mol Microbiol 2016, 100, 841–859. [Google Scholar] [CrossRef]
- Fuller, K.K.; Ringelberg, C.S.; Loros, J.J.; Dunlap, J.C. The fungal pathogen Aspergillus fumigatus regulates growth, metabolism, and stress resistance in response to light. mBio 2013, 4. [Google Scholar] [CrossRef]
- Silva, L.P.; Frawley, D.; Assis, L.J.; Tierney, C.; Fleming, A.B.; Bayram, O.; Goldman, G.H. Putative Membrane Receptors Contribute to Activation and Efficient Signaling of Mitogen-Activated Protein Kinase Cascades during Adaptation of Aspergillus fumigatus to Different Stressors and Carbon Sources. mSphere 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Mattos, E.C.; Silva, L.P.; Valero, C.; de Castro, P.A.; Dos Reis, T.F.; Ribeiro, L.F.C.; Marten, M.R.; Silva-Rocha, R.; Westmann, C.; da Silva, C.; et al. The Aspergillus fumigatus Phosphoproteome Reveals Roles of High-Osmolarity Glycerol Mitogen-Activated Protein Kinases in Promoting Cell Wall Damage and Caspofungin Tolerance. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Mattos, E.C.; Palmisano, G.; Goldman, G.H. Phosphoproteomics of Aspergillus fumigatus Exposed to the Antifungal Drug Caspofungin. mSphere 2020, 5. [Google Scholar] [CrossRef]
- Hokken, M.W.J.; Zoll, J.; Coolen, J.P.M.; Zwaan, B.J.; Verweij, P.E.; Melchers, W.J.G. Phenotypic plasticity and the evolution of azole resistance in Aspergillus fumigatus; an expression profile of clinical isolates upon exposure to itraconazole. BMC Genomics 2019, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.J.; Yu, Y.M.; Kim, G.B.; Lee, G.W.; Maeng, P.J.; Kim, S.; Floyd, A.; Heitman, J.; Bahn, Y.S. Remodeling of global transcription patterns of Cryptococcus neoformans genes mediated by the stress-activated HOG signaling pathways. Eukaryot Cell 2009, 8, 1197–1217. [Google Scholar] [CrossRef]
- Bahn, Y.S.; Kojima, K.; Cox, G.M.; Heitman, J. Specialization of the HOG pathway and its impact on differentiation and virulence of Cryptococcus neoformans. Molecular Biology of the Cell 2005, 16, 2285–2300. [Google Scholar] [CrossRef]
- Bahn, Y.S.; Kojima, K.; Cox, G.M.; Heitman, J. A Unique Fungal Two-Component System Regulates Stress Responses, Drug Sensitivity, Sexual Development, and Virulence of Cryptococcus neoformans. Molecular Biology of the Cell 2006, 17, 3122–3135. [Google Scholar] [CrossRef]
- Barrett, J.F.; Hoch, J.A. Two-component signal transduction as a target for microbial anti-infective therapy. Antimicrob Agents Chemother 1998, 42, 1529–1536. [Google Scholar] [CrossRef]
- Barrett, J.F.; Goldschmidt, R.M.; Lawrence, L.E.; Foleno, B.; Chen, R.; Demers, J.P.; Johnson, S.; Kanojia, R.; Fernandez, J.; Bernstein, J.; et al. Antibacterial agents that inhibit two-component signal transduction systems. Proc Natl Acad Sci U S A 1998, 95, 5317–5322. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yu, C.; Wu, H.; Li, G.; Li, C.; Hong, W.; Yang, X.; Wang, H.; You, X. Recent Advances in Histidine Kinase-Targeted Antimicrobial Agents. Front Chem 2022, 10, 866392. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



