Submitted:
22 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patient Population Characteristics
2.2. Fluoroenzyme Immunoassay (FEIA)
2.3. Indirect Immunofluorescence (IIF)
2.4. Immunoblot (IB) Analysis
2.5. Data Processing and Statistical Analysis
3. Results
3.1. Anti-dsDNA Antibody Level
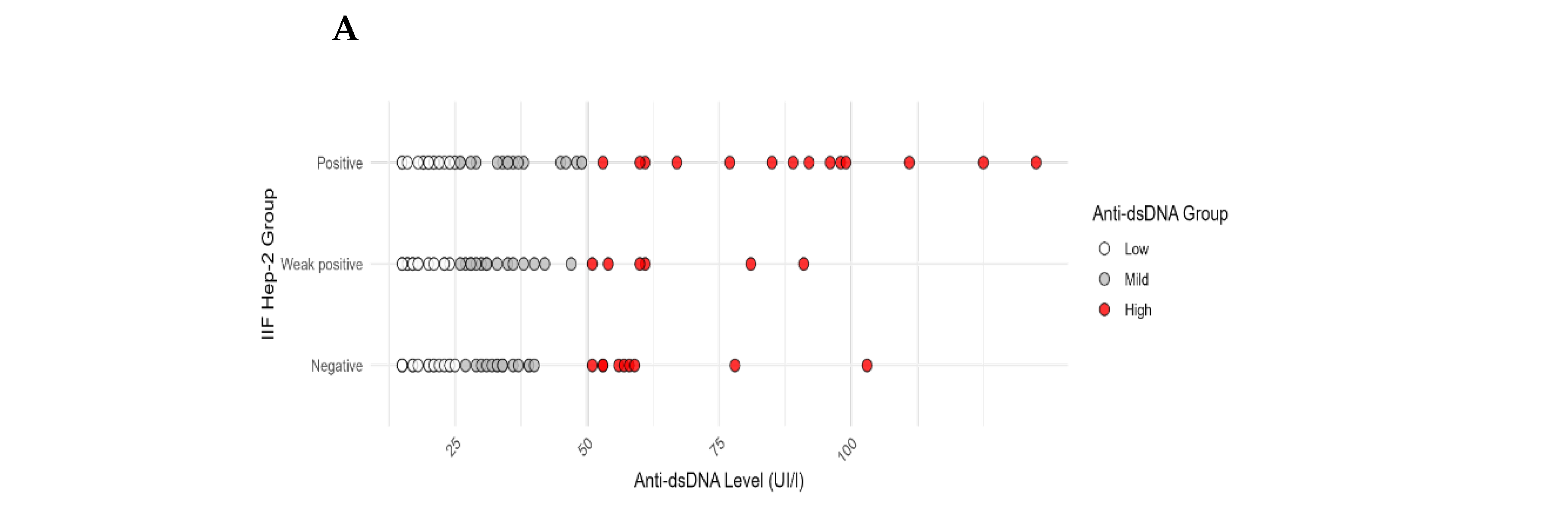
3.2. Correlation Between Hep-2 Fluorescence Titers and Plasma Levels of Anti-dsDNA Antibodies
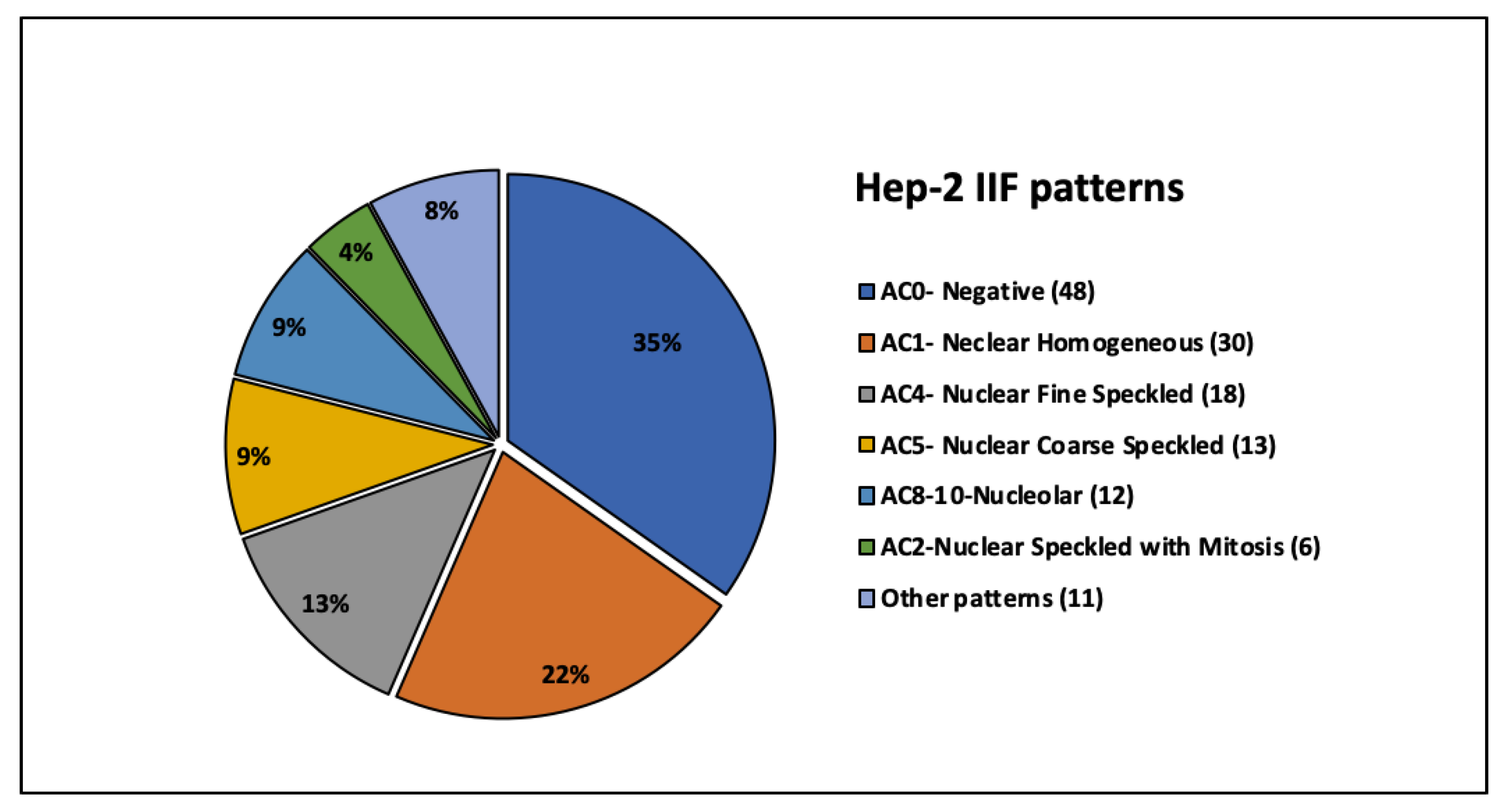
3.3. Correlation Between Anti-dsDNA Antibodies and HEp-2 IIF Patterns
3.4. Comparison of Positivity Rates Across Different Anti-dsDNA Detection Methods
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aringer M, Costenbader K, Daikh D, Brinks R, Mosca M, Ramsey-Goldman R, et al. 2019 European League AgainstRheumatism/American College of Rheumatologyclassificationcriteria for systemic lupus erythematosus. AnnRheumDis.2019 Sep;78(9):1151-1159. Epub 2019 Aug 5. PMID: 31383717. [CrossRef]
- Petri M, Orbai AM, Alarcón GS, Gordon C, Merrill JT, Fortin PR, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. ArthritisRheum. 2012 Aug;64(8):2677-86. PMID: 22553077; PMCID: PMC3409311. [CrossRef]
- Arbuckle MR, McClain MT, Rubertone MV, Scofield RH, Dennis GJ, James JA, Harley JB. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med. 2003 Oct 16;349(16):1526-33. PMID: 14561795. [CrossRef]
- Andrejevic S, Jeremic I, Sefik-Bukilica M, Nikolic M, Stojimirovic B, Bonaci-Nikolic B. Immunoserological parameters in SLE: high-avidity anti-dsDNA detected by ELISA are the most closely associated with the disease activity. ClinRheumatol. 2013 Nov;32(11):1619-26. Epub 2013 Jul 16. PMID: 23857662. [CrossRef]
- Swaak T, Smeenk R. Clinical significance of antibodies to double stranded DNA (dsDNA) for systemic lupus erythematosus (SLE). ClinRheumatol. 1987 Jun;6 Suppl 1:56-73. PMID: 3304800. [CrossRef]
- Fillatreau S, Manfroi B, Dörner T. Toll-like receptor signalling in B cells during systemic lupus erythematosus. Nat Rev Rheumatol.2021 Feb;17(2):98-108. Epub 2020 Dec 18. PMID: 33339987; PMCID: PMC7747191. [CrossRef]
- Compagno M, Jacobsen S, Rekvig OP, Truedsson L, Heegaard NH, Nossent J, Jönsen A, Jacobsen RS, Eilertsen GØ, Sturfelt G, Bengtsson AA. Low diagnostic and predictive value of anti-dsDNA antibodies in unselected patients with recent onset of rheumatic symptoms: results from a long-term follow-up Scandinavian multicentre study. Scand J Rheumatol. 2013;42(4):311-6. Epub 2013 Mar 16. PMID: 23496224. [CrossRef]
- Agmon-Levin N, Damoiseaux J, Kallenberg C, Sack U, Witte T, Herold M, et al. International recommendations for the assessment of autoantibodies to cellular antigens referred to as anti-nuclear antibodies. Ann Rheum Dis. 2014 Jan;73(1):17-23. Epub 2013 Oct 14. PMID: 24126457. [CrossRef]
- Ghirardello A, Villalta D, Morozzi G, Afeltra A, Galeazzi M, Gerli R, et al. Forum Interdisciplinare per la Ricerca sulle Malattie Autoimmuni(FIRMA) study group. Diagnostic accuracy of currently available anti-double-stranded DNA antibody assays. An Italian multicentre study. ClinExpRheumatol. 2011 Jan-Feb;29(1):50-6. Epub 2011 Feb 23. PMID: 21345292.
- Albrecht K, Troll W, Callhoff J, Strangfeld A, Ohrndorf S, Mucke J. Sex- and gender-related differences in systemic lupus erythematosus: a scoping review. Rheumatol Int. 2025 Jun 27;45(7):160. PMID: 40576834; PMCID: PMC12204902. [CrossRef]
- Somers EC, Marder W, Cagnoli P, Lewis EE, et al. Population-based incidence and prevalence of systemic lupus erythematosus: the Michigan Lupus Epidemiology and Surveillance program. Arthritis Rheumatol. 2014 Feb;66(2):369-78. PMID: 24504809; PMCID: PMC4198147. [CrossRef]
- Pan N, Amigues I, Lyman S, Duculan R, Aziz F, Crow MK, Kirou KA. A surge in anti-dsDNA titer predicts a severe lupus flare within six months. Lupus. 2014 Mar;23(3):293-8. Epub 2013 Dec 6. PMID: 24316605. [CrossRef]
- Narayanan K, Marwaha V, Shanmuganandan K, Shankar S. Correlation between Systemic Lupus Erythematosus Disease Activity Index, C3, C4 and Anti-dsDNA Antibodies. Med J Armed Forces India. 2010 Apr;66(2):102-7. Epub 2011 Jul 21. PMID: 27365721; PMCID: PMC4920905. [CrossRef]
- Olson SW, Lee JJ, Prince LK, Baker TP, Papadopoulos P, Edison J, Abbott KC. Elevated subclinical double-stranded DNA antibodies and future proliferative lupus nephritis. Clin J Am SocNephrol. 2013 Oct;8(10):1702-8. Epub 2013 Jul 5. PMID: 23833315; PMCID: PMC3789337. [CrossRef]
- Abozaid HSM, Hefny HM, Abualfadl EM, Ismail MA, Noreldin AK, Eldin ANN, Goda AM, Ali AH. Negative ANA-IIF in SLE patients: what is beyond? Clin Rheumatol. 2023 Jul;42(7):1819-1826. Epub 2023 Apr 4. PMID: 37016193; PMCID: PMC10267001. [CrossRef]
- Uzun GS, Apsley E, Isenberg D. Identification of patients with ANA-negative and double-stranded DNA positive: what is the significance in daily rheumatology practice? Clin Rheumatol. 2025 Feb;44(2):863-865. Epub 2024 Dec 24. PMID: 39718671. [CrossRef]
- Robier C, Haas M, Quehenberger F. The clinical relevance of anti-dsDNA antibodies determined by the Elia™ dsDNA assay in patients with negative indirect immunofluorescence on the HEp-2 cell. ClinChem Lab Med. 2020 Oct 16;59(3):541-546. PMID: 33064668. [CrossRef]
- Trujillo Aguilera A, Bernardo Serrano R, Navas A, Alcaide Molina J, Alvarez Romero P, Jurado Roger A. Longitudinal study of patients with discrepant results in CLIFT and a solid-phase dsDNA antibody assay: does a gold standard dsDNA assay exist? Lupus Sci Med. 2023 Oct;10(2):e000984. PMID: 37903589; PMCID: PMC10618974. [CrossRef]
- Cuomo L, Vitillo M, Della Rocca M, Trivedi P. Comparative analysis of three methods in anti-dsDNA antibodies detection: implications for Systemic Lupus Erythematosus diagnosis. Scand J Immunol. 2022 Feb;95(2):e13123. Epub 2021 Dec 12. PMID: 34865261. [CrossRef]
- Infantino M, Palterer B, Previtali G, Alessio MG, Villalta D, Carbone T, et al. Comparison of current methods for anti-dsDNA antibody detection and reshaping diagnostic strategies. Scand J Immunol. 2022 Dec;96(6):e13220. Epub 2022 Oct 10. PMID: 36373656. [CrossRef]
- Bizzaro N, Villalta D, Giavarina D, Tozzoli R. Are anti-nucleosome antibodies a better diagnostic marker than anti-dsDNA antibodies for systemic lupus erythematosus? A systematic review and a study of metanalysis. Autoimmun Rev. 2012 Dec;12(2):97-106. Epub 2012 Jul 15. PMID: 22810055. [CrossRef]

| Anti-dsDNAgroup | N | Anti-dsDNA (IU/mL) | Age Mean ± SD |
| Negative (≤14 IU/mL) | 29 | 8.6 ± 2.7 | 59.4 ± 14.9 |
| Low (15–25 IU/mL) | 63 | 19.1 ± 3.4 | 57.8 ± 17.4 |
| Mild (26–49 IU/mL) | 46 | 34.8 ± 6.4 | 53.3 ± 16.4 |
| High (≥50 IU/mL) | 29 | 76.3 ± 23.7 | 50.8 ± 14.1 |
| Anova | p=0.105 |
| Hep-2 IIF group | Tot | Anti-dsDNA Low | Anti-dsDNA Mild | Anti-dsDNA High |
| (15-25 UI/mL) | (26-49 UI/mL) | (over 50 UI/mL) | ||
| N (%) | N (%) | N (%) | ||
| Negative <1:80 | 48 | 24 (50) | 15 (31) | 9 (19) |
| Weak positive (1:80-1:160) | 34 | 13 (38) | 15 (44) | 6 (18) |
| Positive (1:320-1:1280) | 56 | 26 (46) | 16 (29) | 14 (25) |
| Tot | 138 | 63 (46) | 46 (33) | 29 (21) |
| A | ||||
| Marker | N | Positive (%) | Negative (%) | NA |
| Cell pattern (IIF-Hep2) | 30 | 22 (73) | 8 (27) | -- |
| dsDNA (FEIA) | 30 | 28 (93) | 2 (7) | -- |
| dsDNA (Immunoblot) | 29 | 23 (79) | 6 (21) | 1 |
| dsDNA (CLIFT) | 25 | 6 (24) | 19 (76) | 5 |
| Nucleosome (Immunoblot) | 29 | 3 (10.3) | 26 (89.7) | 1 |
| Histone (Immunoblot) | 29 | 0 (0) | 100 (100) | 1 |
| B | ||||
| Anti-dsDNApositivity concordance (%) | ||||
| FEIA vs IB (agreement %) | FEIA vs CLIFT (agreement %) |
IB vs CLIFT (agreement %) |
||
| IIF-Hep2 negative | 100 | 40 | 40 | |
| IIF-Hep2 positive | 81 | 30 | 30 | |
| IIF-Hep2 (all) | 86 | 32 | 32 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




