Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Background and Objectives: Procalcitonin (PCT) kinetics are increasingly used as prognostic markers in sepsis, but their interpretation is confounded by dynamic changes in renal function during acute illness. This study evaluated the prognostic value of ΔPCT for 30-day mortality in critically ill patients with sepsis or septic shock by incorporating serial kinetic eGFR measurements and renal function–adjusted ΔPCT cut-off values based on the mean kinetic eGFR during the first 48-72 hours of ICU admission. Materials and Methods: This retrospective cohort study included 106 adult ICU patients with sepsis or septic shock. Serial procalcitonin measurements were used to calculate ΔPCT as ratio of follow-up to baseline values, while renal function was assessed using mean kinetic eGFR over the first 72 hours of ICU admission. Results: Thirty-day mortality was 43.4%. ΔPCT was a strong independent predictor of mortality across all models. At 48 hours, ΔPCT2 was independently associated with 30-day mortality in the overall cohort (AUC 0.793) and retained independent prognostic significance only in patients with preserved renal function (GFR ≥30 mL/min/1.73 m²). The optimal ΔPCT2 cut-off corresponded to a 56% reduction in procalcitonin levels. At 72 hours, ΔPCT3 emerged as an independent predictor of mortality regardless of renal function. ROC analysis identified an optimal ΔPCT3 cut-off corresponding to 62% procalcitonin reduction in the overall cohort, with renal function–specific thresholds of ~50% for patients with GFR <30 mL/min/1.73 m² and ~73% for those with preserved renal function. The combination of APACHE II score and ΔPCT3 demonstrated the highest discriminative performance for mortality prediction (AUC 0.948). Conclusions: Procalcitonin kinetics provide clinically meaningful prognostic information in sepsis when interpreted alongside dynamic renal function. While 48-hour procalcitonin kinetics offer prognostic value primarily in patients with preserved renal function, 72-hour ΔPCT provides renal function–independent and superior mortality discrimination. Integrating serial kinetic eGFR measurements enables renal function–adapted ΔPCT threshold determination and may improve risk stratification in critically ill septic patients.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
| Group | Time point | ΔPCT cut-off (log10) | Ratio (10^ΔPCT) | Required PCT decline (%) |
|---|---|---|---|---|
| All patients | 48 h (ΔPCT2) | −0.36 | 0.44 | 56.0% decrease |
| All patients | 72 h (ΔPCT3) | −0.42 | 0.38 | 62.0% decrease |
| GFR <30 mL/min/1.73 m² | 72 h (ΔPCT3) | −0.30 | 0.50 | 50.0% decrease |
| GFR ≥30 mL/min/1.73 m² | 48 h (ΔPCT2) | −0.35 | 0.45 | 55.0% decrease |
| GFR ≥30 mL/min/1.73 m² | 72 h (ΔPCT3) | −0.57 | 0.27 | 73.0% decrease |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Abbreviation | Definition |
| AKI | Acute kidney injury |
| APACHE II | Acute Physiology and Chronic Health Evaluation II |
| AUC | Area under the curve |
| CI | Confidence interval |
| CKD | Chronic kidney disease |
| COPD | Chronic obstructive pulmonary disease |
| CRRT | Continuous renal replacement therapy |
| ΔPCT | Delta procalcitonin ((log₁₀[PCT_follow-up / PCT_baseline]) |
| eGFR | Estimated glomerular filtration rate |
| ESRD | End-stage renal disease |
| GFR | Glomerular filtration rate |
| ICU | Intensive care unit |
| IHD | Intermittent hemodialysis |
| kGFR | Kinetic estimated glomerular filtration rate |
| OR | Odds ratio |
| PCT | Procalcitonin |
| ROC | Receiver operating characteristic |
| SD | Standard deviation |
| SOFA | Sequential Organ Failure Assessment |
References
- Becker, KL; Nylén, ES; White, JC; Müller, B; Snider, RH. Procalcitonin and the Calcitonin Gene Family of Peptides in Inflammation, Infection, and Sepsis: A Journey from Calcitonin Back to Its Precursors. The Journal of Clinical Endocrinology & Metabolism 2004, 89(4), 1512–1525. [Google Scholar] [CrossRef] [PubMed]
- Assicot, M; Bohuon, C; Gendrel, D; Raymond, J; Carsin, H; Guilbaud, J. High serum procalcitonin concentrations in patients with sepsis and infection. The Lancet 1993, 341(8844), 515–518. [Google Scholar] [CrossRef] [PubMed]
- Gregoriano, C; Heilmann, E; Molitor, A; Schuetz, P. Role of procalcitonin use in the management of sepsis. J Thorac Dis. 2020, 12(S1), S5–S15. [Google Scholar] [CrossRef]
- Singer, M; Deutschman, CS; Seymour, CW; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315(8), 801. [Google Scholar] [CrossRef]
- Samsudin, I; Vasikaran, SD. Clinical Utility and Measurement of Procalcitonin. Clin Biochem Rev. 2017, 38(2), 59–68. [Google Scholar] [PubMed]
- Wu, SC; Liang, CX; Zhang, YL; Hu, WP. Elevated serum procalcitonin level in patients with chronic kidney disease without infection: A case-control study. J Clin Lab Anal. 2020, 34(2), e23065. [Google Scholar] [CrossRef] [PubMed]
- Foulon, N; Haeger, SM; Okamura, K; et al. Procalcitonin levels in septic and nonseptic subjects with AKI and ESKD prior to and during continuous kidney replacement therapy (CKRT). Crit Care 2025, 29(1), 171. [Google Scholar] [CrossRef] [PubMed]
- Chen, S. Kinetic Glomerular Filtration Rate in Routine Clinical Practice—Applications and Possibilities. Advances in Chronic Kidney Disease 2018, 25(1), 105–114. [Google Scholar] [CrossRef] [PubMed]
- Dark, P; Hossain, A; McAuley, DF; et al. Biomarker-Guided Antibiotic Duration for Hospitalized Patients With Suspected Sepsis: The ADAPT-Sepsis Randomized Clinical Trial. JAMA 2025, 333(8), 682. [Google Scholar] [CrossRef] [PubMed]
- Inker, LA; Eneanya, ND; Coresh, J; et al. New Creatinine- and Cystatin C–Based Equations to Estimate GFR without Race. N Engl J Med. 2021, 385(19), 1737–1749. [Google Scholar] [CrossRef] [PubMed]
- Park, AK; Nakasone, TS; Kaushal, A; Renault, C. Quantifying the Relationship Between Renal Function and Procalcitonin: A Study of 14 431 Blood Cultures. Open Forum Infectious Diseases 2025, 12(11), ofaf654. [Google Scholar] [CrossRef] [PubMed]
- Özger, HS; Çorbacıoglu, SK; Boyacı-Dündar, N; et al. Changes of Procalcitonin Kinetics According to Renal Clearance in Critically Ill Patients with Primary Gram-Negative Bloodstream Infections. Infect Dis Clin Microbiol. 2024, 6(3), 206–215. [Google Scholar] [CrossRef] [PubMed]
| Variable | Categories | n (%) |
|---|---|---|
| Sex | Female | 38 (35.8) |
| Male | 68 (64.2) | |
| Sepsis category | Sepsis | 76 (71.7) |
| Septic shock | 30 (28.3) | |
| Diabetes mellitus | Present | 52 (49.1) |
| Absent | 54 (50.9) | |
| Hypertension | Present | 65 (61.3) |
| Absent | 41 (38.7) | |
| COPD | Present | 33 (31.1) |
| Absent | 73 (68.9) | |
| Chronic kidney disease | Present | 41 (38.7) |
| Absent | 65 (61.3) | |
| Coronary artery disease | Present | 34 (32.1) |
| Absent | 72 (67.9) | |
| Source of infection | Community-acquired | 42 (39.6) |
| Hospital-acquired | 64 (60.4) | |
| Blood culture result | No growth | 57 (53.8) |
| Growth present | 42 (39.6) | |
| Culture not obtained | 7 (6.6) | |
| Pathogen type | No pathogen detected | 62 (58.5) |
| Gram-negative | 23 (21.7) | |
| Gram-positive | 12 (11.3) | |
| Fungal | 8 (7.5) | |
| Viral | 1 (0.9) | |
| Empiric antibiotic adequacy | Adequate | 34 (32.1) |
| Inadequate | 9 (8.5) | |
| Unknown | 62 (58.5) | |
| RRT modality | None | 75 (70.8) |
| IHD | 13 (12.3) | |
| CRRT | 7 (6.6) | |
| Chronic HD patient | 11 (10.4) | |
| AKI stage | No AKI | 29 (27.4) |
| Stage 1 | 13 (12.3) | |
| Stage 2 | 10 (9.4) | |
| Stage 3 | 42 (39.6) | |
| Chronic HD | 12 (11.3) | |
| AKI outcome | No AKI | 31 (29.2) |
| Complete recovery | 27 (25.5) | |
| Partial recovery | 7 (6.6) | |
| ESRD | 9 (8.5) | |
| AKI-related death | 32 (30.2) | |
| 30-day mortality | Survived | 60 (56.6) |
| Died | 46 (43.4) |
| Variable | Survivors Mean ± SD | Non-survivors Mean ± SD | p-value |
|---|---|---|---|
| Age (years) | 68.92 ± 16.82 | 73.17 ± 12.87 | 0.431 |
| APACHE II | 21.15 ± 7.22 | 37.54 ± 11.58 | <0.001 |
| SOFA | 6.60 ± 2.94 | 8.71 ± 3.50 | 0.001 |
| Procalcitonin (baseline) | 24.00 ± 32.24 | 18.56 ± 32.83 | 0.027 |
| Creatinine (baseline) | 2.18 ± 1.86 | 2.61 ± 1.89 | 0.086 |
| Estimated GFR (baseline) | 52.33 ± 33.79 | 37.33 ± 29.66 | 0.086 |
| MeanGFR (kinetic) | 56.71 ± 32.43 | 38.30 ± 27.04 | 0.007 |
| DeltaPCT1 (log10) | −0.1668 ± 0.1948 | −0.0103 ± 0.2636 | 0.002 |
| DeltaPCT2 (log10) | −0.4157 ± 0.2868 | 0.0080 ± 0.5158 | <0.001 |
| DeltaPCT3 (log10) | –0.7684 ± 0.4349 | 0.0943 ± 0.7413 | <0.001 |
| Albumin (baseline) | 30.38 ± 6.21 | 28.36 ± 5.54 | 0.092 |
| Days until death | 2.37 ± 10.33 | 9.41 ± 6.58 | 0.608 |
| Model | p-value | OR (Exp B) | 95% CI | Accuracy (%) |
|---|---|---|---|---|
| Model 1 (APACHE II + ΔPCT2) | <0.001 | 20.48 | 2.92–143.70 | 82.1 |
| Model 2 (SOFA + ΔPCT2) | <0.001 | 22.72 | 4.18–123.59 | 78.3 |
| Model 1 (APACHE II + ΔPCT3) | <0.001 | 1.202 | 1.10–1.32 | 84 |
| Model 2 (SOFA + ΔPCT3) | <0.001 | 42.718 | 9.04–201.89 | 84 |
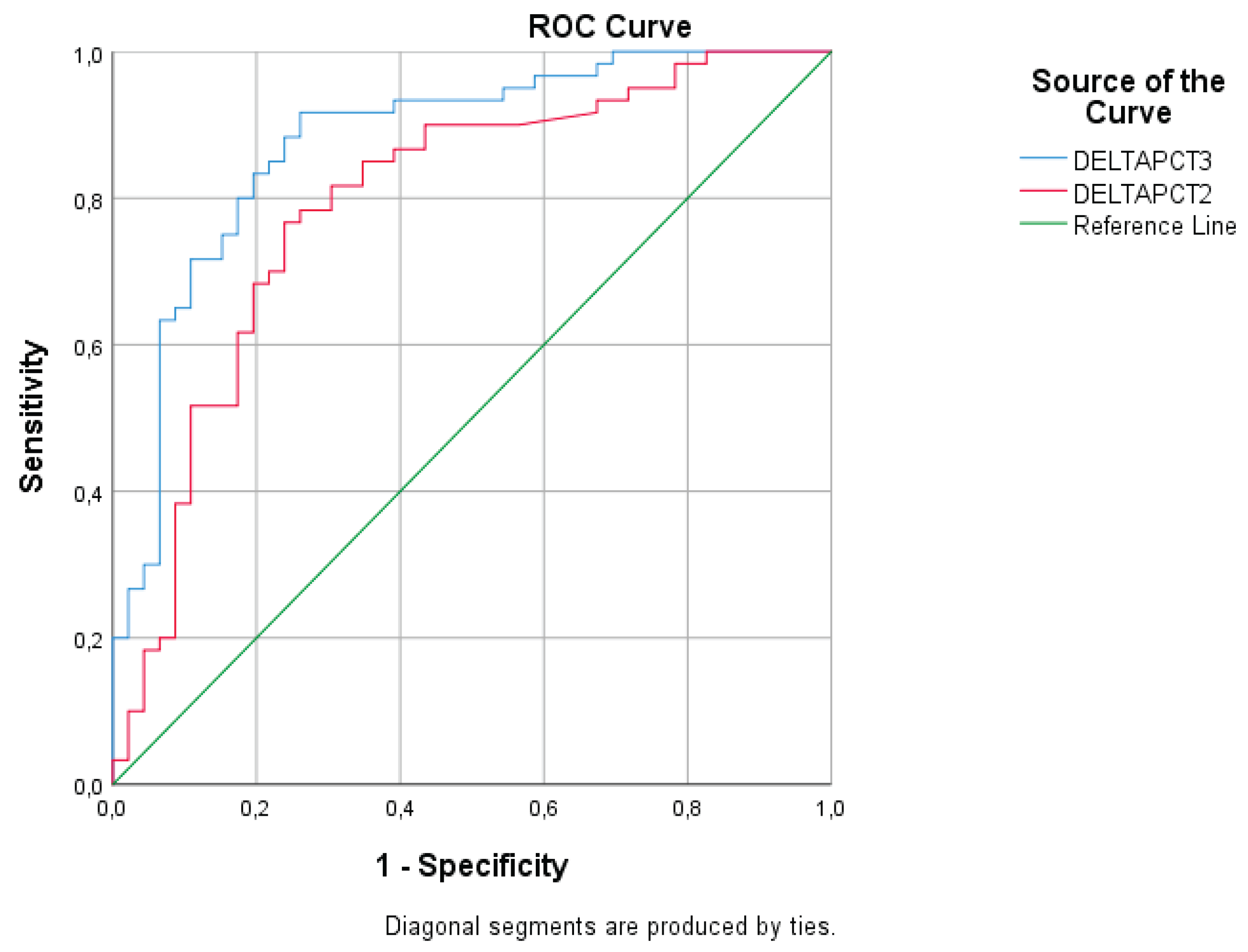
| Predictor | AUC | Std Error | p-value | 95% CI |
|---|---|---|---|---|
| APACHE II + ΔPCT2 | 0.904 | 0.029 | <0.001 | 0.846–0.961 |
| SOFA + ΔPCT2 | 0.814 | 0.044 | <0.001 | 0.728–0.900 |
| ΔPCT2 | 0.793 | 0.046 | <0.001 | 0.703–0.883 |
| APACHE II | 0.877 | 0.037 | <0.001 | 0.804–0.950 |
| SOFA | 0.676 | 0.054 | 0.002 | 0.569–0.782 |
| APACHE II + ΔPCT3 | 0.948 | 0.018 | <0.001 | 0.912–0.984 |
| SOFA + ΔPCT3 | 0.885 | 0.032 | <0.001 | 0.821–0.948 |
| ΔPCT3 | 0.878 | 0.035 | <0.001 | 0.810–0.946 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




