4. Discussion
The present study provides descriptive information on 68 cases that were diagnosed as FIP by qualified general practitioner veterinarians, during and after the dosumented 2023 outbreak in Cyprus [
38]. These cases were documented through a standardized, structured questionnaire voluntarily completed by practicing veterinarians in real time as the outbreak evolved. This study presents field-based observations from cases clinically considered compatible with FIP during the outbreak period, reflecting how the condition was perceived, approached, and managed in general practice settings across Cyprus. It is crucial to clarify that this study is not a product of a systematic surveillance program, and it should not be perceived as such. These findings also illustrate the usefulness of structured questionnaires as a practical epidemiological tool for capturing real-time clinical impressions and trends during suspected emerging infectious disease events.
The response rate of the study was 21.0% (22 clinics answered the questionnaire out of 105 contacted). Although modest, this response rate is comparable to those reported in questionnaire-based studies involving veterinary professionals, particularly in companion animal infectious disease research requiring detailed clinical data. Reported response rates in the veterinary literature vary widely depending on study design and target population [
44,
45,
46,
47], reflecting the practical challenges of achieving high participation in voluntary clinic-based surveys. Non-response bias cannot be excluded and should be considered when interpreting the results.
Most cases were recorded in Limassol and Nicosia with the most probable explanation for this being the higher number of veterinary facilities in those two areas. However, this distribution may also reflect the population density of cats, with higher numbers of feeding stations, and greater interaction between owned and free-roaming cats in those two cities of Cyprus [
36].
The typical signalment of cats with FIP includes young, non-pedigree cats, with a reported male predominance [
30,
31,
33,
48,
49]. In the present dataset, the mean age of the 68 cats reported was 3.9 years. This is higher than the 1–2 years commonly reported in earlier literature [
32,
33] although age distributions have varied across the literature [
10,
11]. One possible explanation for the relatively higher mean age observed in these 68 cases, particularly in the cases that coincide with the early phase of the outbreak, may relate to population-level susceptibility. At the onset of widespread viral circulation, cats across multiple age groups may have been immunologically naïve and therefore susceptible to developing clinical disease. Over time, shifts in exposure patterns or acquired immunity at the population level may have influenced the age distribution of reported cases, with proportionally more cases occurring in younger cats as the outbreak progressed. This interpretation remains speculative and would require longitudinal epidemiological and immunological data for confirmation. In contrast to reports indicating that pedigree cats account for up to 70% of typically described FIP cases [
30,
32,
33] few pedigree cats were represented in the current dataset. This finding may reflect the demographic structure of the cat population in Cyprus, where large numbers of free-roaming and formerly unowned cats exist [
36], and where the majority of owned cats are domestic shorthaired or domestic longhaired animals adopted from the streets [
50]. Therefore, the breed distribution observed during the outbreak period is likely influenced by the underlying population structure rather than breed predisposition alone.
The overwhelming presence of cats with prior contact with other cats (95.6%) reinforces the recognized role of social and environmental transmission in FCoV maintenance [
51], and also supports the direct transmission dynamics of FCoV-23 which caused the epizootic [
10,
11,
12]. An additional noteworthy observation was that 14/68 cats were reported to have experienced a stressor preceding the onset of clinical signs. This finding supports the recognized association between stress and the development of FIP. The identification of stress prior to disease manifestation aligns with existing knowledge regarding risk factors that may contribute to the development of FIP in susceptible individuals [
52,
53,
54].
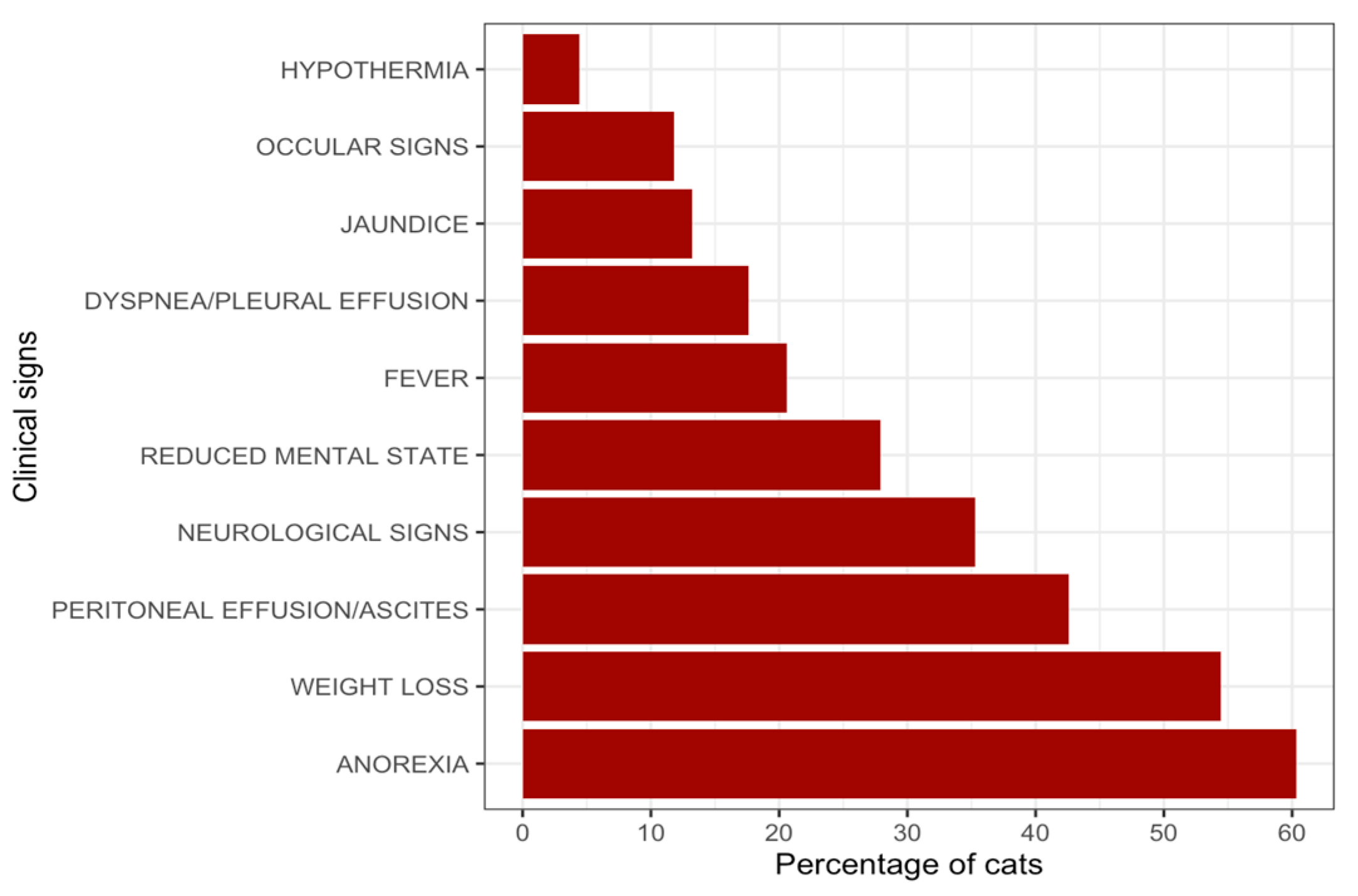
The clinical manifestations recorded in this study were largely consistent with previous descriptions of FIP, including anorexia, weight loss, lethargy, and effusive peritoneal disease [
30,
31,
33,
55]. The notable rate of reported neurological signs (35.3%) deserves attention. This proportion exceeds the 10–30% range reported in earlier large population studies [
30,
32,
33]. The neurological manifestation reported for this study reflects the diagnosis reached by participating veterinarians for each respective case, absent definitive confirmation. While this observation may indicate a change in clinical phenotype during the outbreak period, such conclusions remain conjectural. None of the neurological cases included in this dataset were verified through RT-PCR or immunohistochemistry; as such, no direct virological association can be determined at the individual case level. Concurrent molecular analyses conducted during the same epizootic period identified the presence of a recombinant feline coronavirus strain (FCoV-23) circulating in Cyprus [
10,
11,
12].All RT-PCR-confirmed cases analyzed in those studies were attributed to that recombinant strain. While experimental and genomic analyses from those investigations have suggested biological features that could potentially influence tissue tropism [
56,
57,
58,
59,
60,
61]. The present study does not provide direct evidence linking the increased proportion of neurological presentations to a specific viral variant, especially in the absence of molecular data. Accordingly, the higher frequency of neurological involvement observed here should be interpreted as a descriptive field finding during the outbreak period, rather than as proof of altered neurotropism. Further virological and clinicopathological studies would be required to clarify any causal relationship between viral genotype and clinical phenotype. Diagnostic approaches reported by the veterinary practitioners on Cyprus reflected the practical challenges of field conditions. Only 22.1% (15/68) of cases were RT-PCR-confirmed as FIP, with most diagnoses being based on the combination of clinical signs and in-house laboratory test findings. A number of 14 out of the 15 RT-PCR confirmed FIP cases reported in this study were confirmed FCoV-23 through a second dedicated RT-PCR. The 15
th sample was not available for further testing. Three of these cases belong to the sample pool reported by Attipa et al [
11,
38], the publication that introduced FCoV-23.
These findings are consistent with patterns reported in several large clinical studies, where the cost and accessibility of molecular tests limit their use [
30,
48,
62]. This situation is particularly evident in countries where companion animal health insurance systems are not established and where large populations of stray animals exist, such as in Cyprus[
36,
50], resulting in financial and logistical limitations that restrict the routine use of advanced diagnostic methods [
63,
64,
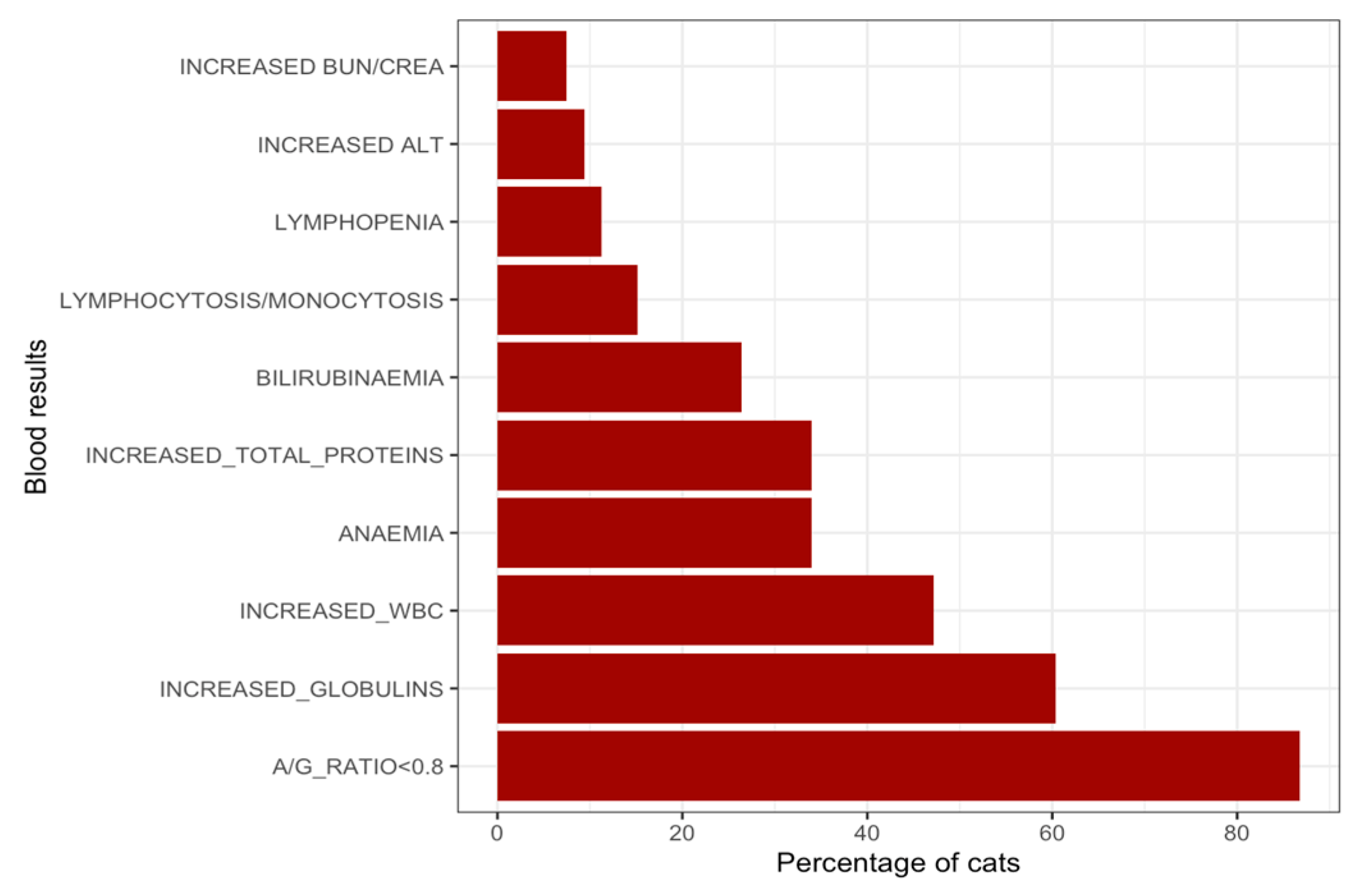
65]. Despite this, most cases (55/68; 80.9%) included in-house analyzer blood examinations, reflecting the standard of animal care in Cyprus. The high prevalence of hyperglobulinemia and low serum albumin-to-globulin ratios (A/G <0.8) mirror typically observed FIP profiles and supports their continued diagnostic value [
48,
52]. A raised serum gamma-globulin concentration was the most common protein electrophoretic pattern (77.4%), consistent with the immunopathogenic nature of the disease [
68,
69]. Also, in line with the current literature is the finding that hyperglobulinemia and A/G ratio are not dependent on the FIP form[
30,
32]. An additional noteworthy observation is that 14 out of 15 RT-PCR confirmed samples were identified as FCoV23. This supports Attipa et al [
11]claim that FCoV23 constitutes stable recombination which caused an epizootic. The consistent detection of FCoV23 over a span of more than two years also indicates its endemic presence within the feline population of Cyprus. This argument has to be supported by more evidence.
Therapeutically, Cyprus was the first European Country that approved three different options for the treatment of FIP in cats, injectable Remdesivir (BOVA UK Ltd, United Kingdom) and GS441524 50mg tablets (BOVA UK Ltd, United Kingdom) and molnupiravir (Lagevrio™, Merck Sharp & Dohme B.V.) [
68]. The study confirms the widespread adoption of these antiviral drugs for the treatment of FIP, as 92% of the cases received one of these agents, or a combination, with an overall reported clinical improvement rate of 88.9%. These figures are consistent with published efficacy data of the antiviral drugs on FIP [
29,
33,
69,
70,
71]. Treatment response rates differed across diagnostic categories. Notably, all Category A cases demonstrated a positive treatment response (9/9; 100%). In Category B, a high proportion of cases responded positively to antiviral therapy (34/38; 89.5%), while Category C also showed a substantial rate of positive response (13/15; 86.7%). Overall, the majority of treated cases (56/62; 90.3%) exhibited favorable outcomes, supporting the clinical judgement of the veterinarians involved. These findings reinforce the assertion that the 43 cases from Categories A and B, despite lacking confirmatory RT-PCR testing, were correctly diagnosed as FIP.
Since 68.3% of the cats were given unlicensed preparations, concerns have to be raise regarding the legal and ethical issues surrounding the administration of such preparations [
72,
73]. Despite an expanding market for licensed treatments, unlicensed products are still widely used for FIP treatment [
74]. While this study focuses on 68 specific cases, it is important to highlight that during the first six months of the outbreak in Cyprus, animal carers had no treatment options available. During this period, online providers of unlicensed products established an efficient distribution network, making these drugs easily accessible and limiting the use of licensed therapies, when the latter became available. This led the government of Cyprus to significantly reduce the cost of licensed molnupiravir once legal treatment options became available[
68]. The information gathered for the licensed molnupiravir preparation referenced in the study (Lagevrio™, Merck Sharp & Dohme B.V.) is useful for the knowledge of the use of this medication for the treatment of FIP in cats which was specifically permitted for this use in Cyprus [
68]. Administration to 25 cases, four also administered with remdesivir, resulted in an 84% success rate. This outcome is consistent with studies showing high survival rates (78–92%) [
75,
76,
77,
78,
79,
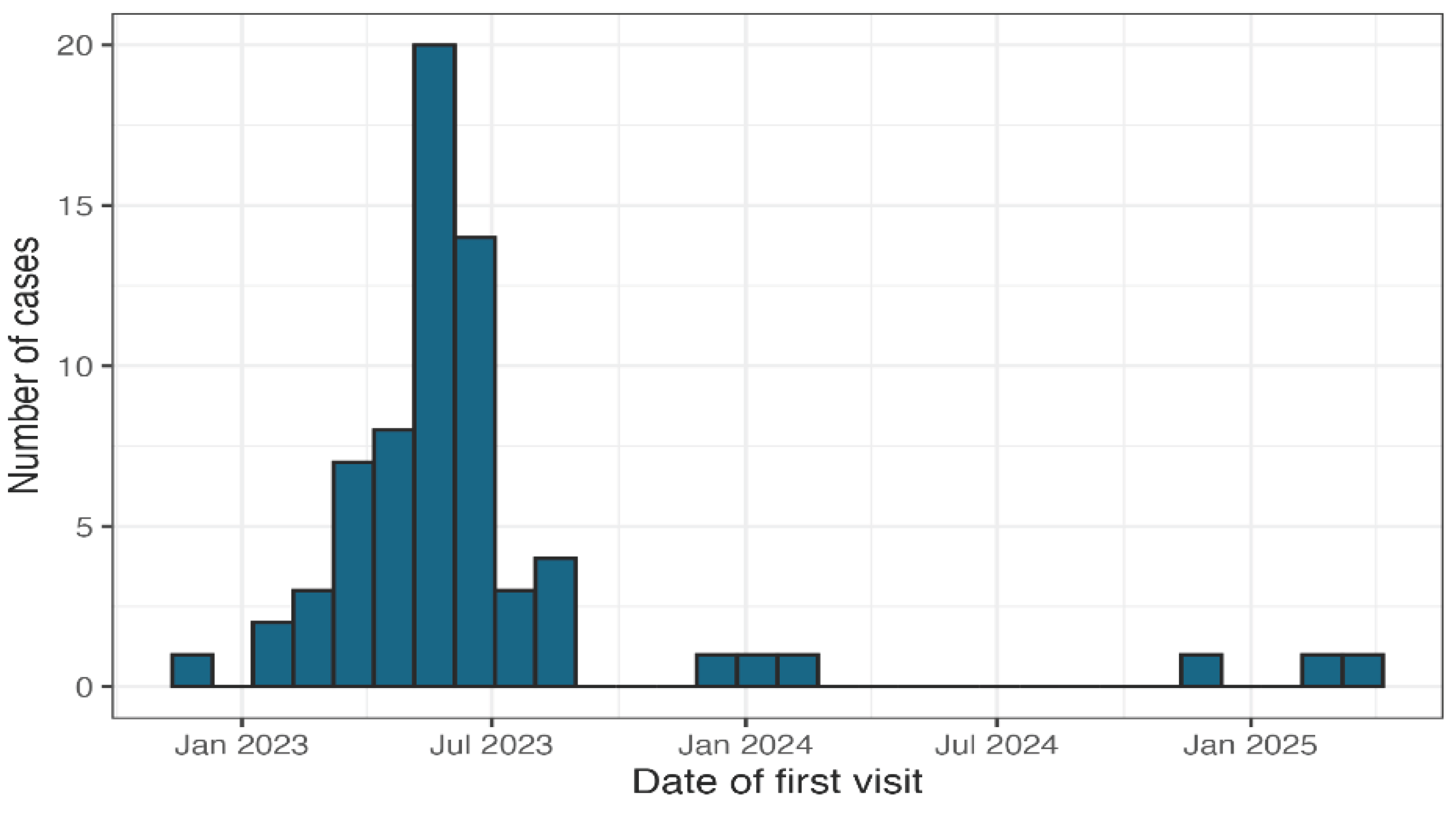
80] for cats with FIP treated with generic molnupiravir, underscoring the drug’s effectiveness. The successful treatment of cats with FIP in Cyprus demonstrates the rapid dissemination of treatment protocols among veterinary practitioners during the outbreak. It exemplifies effective professional communication and adaptability in response to crisis conditions as well as the crucial role that the PVA played in this crisis-management. A major strength of this work lies in its prospective design, with almost real-time data collection. Once the outbreak was discovered, the authors, with the help of the PVA, asked veterinary practitioners on Cyprus to complete questionnaires for cases of FIP they had seen since the beginning of 2023. They were then asked to prospectively collect data for all new cases while the outbreak was ongoing; this minimized recall bias and allowed for a more accurate reflection of temporal and geographic trends. Unlike fully retrospective or laboratory-based studies, this approach provides a ground-level view of the dynamics in clinical practice. Moreover, the island-wide coverage achieved through voluntary participation of veterinarians across all districts was something that occurred for the first time in Cyprus in the field of companion animals. Another key strength is the integration of clinical, epidemiological, and therapeutic data in one dataset. This allows holistic interpretation of how the outbreak manifested, evolved, and was managed. Finally, this study demonstrates the feasibility and value of a questionnaire-based veterinary reporting model, which could be replicated for future similar events in companion animal medicine, especially where a systematic surveillance program does not exist.
Despite these strengths, several limitations must be acknowledged. The voluntary and self-reported nature of the questionnaire may have introduced selection bias: clinics that experienced more cases or were more aware of the outbreak may have been more likely to participate. If their diagnostic protocols differed from under-represented clinics, this could have biased the data. Second, diagnostic heterogeneity represents a significant limitation. The reliance on clinical and in-house diagnostic findings rather than molecular confirmation in most cases means that misclassification cannot be excluded. Although clinical patterns were strongly consistent with FIP, supported by positive response to FIP treatment in the majority of cases, some overlap with other systemic diseases is possible. It is very crucial to determine that the findings of this study represent practitioner reported cases and not in any way a comprehensive surveillance.