Submitted:
30 January 2026
Posted:
02 February 2026
You are already at the latest version
Abstract
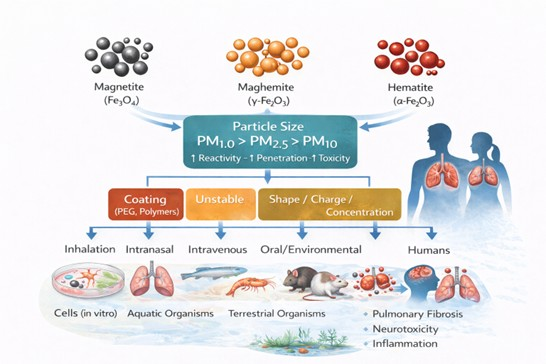
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Iron Oxides and Biological Damage Across Animal Models
3.1.1. Rats
3.1.2. Cellular Models
3.1.3. Humans
3.1.4. Mice
3.1.5. Fish
3.1.6. Amphibians
3.2. Magnetite and Damage Induced in Biological Models
3.2.1. Rats
3.2.2. Cell Models
3.2.3. Humans
3.2.4. Mice
3.2.5. Aquatic and Invertebrate Models
3.3. Maghemite and Adverse Effects Induced by Exposure in Living Organisms
3.3.1. Rats
3.3.2. Cell Models
3.3.3. Humans
3.3.4. Mice
3.3.5. Fish
3.3.6. Primates
3.3.7. Mollusks
3.4. Hematite and Its Biological Effects Following Exposure
3.4.1. Rats
3.4.2. Cells
3.4.3. Amphibians
3.4.4. Humans
3.4.5. Mice
3.4.6. Fish
4. Discussion
4.1. Cellular Models
4.2. Rat Models
4.3. Mouse Models
4.4. Human Evidence
4.5. Knowledge Gaps and Current Limitations
5. Conclusions
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campos, E.A.; Pinto, D.V.B.S.; Oliveira, J.I.S.D.; Mattos, E.D.C.; Dutra, R.D.C.L. Synthesis, characterization and applications of iron oxide nanoparticles: A short review. J. Aerosp. Technol. Manag. 2015, 7, 267–276. [Google Scholar] [CrossRef]
- Benhammada, A.; Trache, D.; Kesraoui, M.; Tarchoun, A.F.; Chelouche, S.; Mezroua, A. Synthesis and characterization of α-Fe2O3 nanoparticles from different precursors and their catalytic effect on the thermal decomposition of nitrocellulose. Thermochim. Acta 2020, 686, 178570. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Teja, A.S.; Koh, P.Y. Synthesis, properties, and applications of magnetic iron oxide nanoparticles. Prog. Cryst. Growth Charact. Mater. 2009, 55, 22–45. [Google Scholar] [CrossRef]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef] [PubMed]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knüchel, R.; Kiessling, F.; Lammers, T. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef]
- Ogbezode, J.E.; Ezealigo, U.S.; Bello, A.; Anye, V.C.; Onwualu, A.P. A narrative review of the synthesis, characterization, and applications of iron oxide nanoparticles. Discover Nano 2023, 18, 125. [Google Scholar] [CrossRef]
- Wang, F.; Zhou, L.; Mu, D.; Zhang, H.; Zhang, G.; Huang, X.; Xiong, P. Current research on ecotoxicity of metal-based nanoparticles: From exposure pathways, ecotoxicological effects to toxicity mechanisms. Front. Public Health 2024, 12, 1390099. [Google Scholar] [CrossRef] [PubMed]
- Zwozdziak, A.; Sówka, I.; Willak-Janc, E.; Zwozdziak, J.; Kwiecińska, K.; Balińska-Miśkiewicz, W. Influence of PM1 and PM2.5 on lung function parameters in healthy schoolchildren—A panel study. Environ. Sci. Pollut. Res. 2016, 23, 23892–23901. [Google Scholar] [CrossRef]
- Rahman, M.M.; Khan, S.B.; Jamal, A.; Faisal, M.; Aisiri, A.M. Iron oxide nanoparticles. Nanomaterials 2011, 3, 43–67. [Google Scholar]
- Shoudho, K.N.; Uddin, S.; Rumon, M.M.H.; Shakil, M.S. Influence of physicochemical properties of iron oxide nanoparticles on their antibacterial activity. ACS Omega 2024, 9, 33303–33334. [Google Scholar] [CrossRef]
- Von Der Heyden, B.; Roychoudhury, A.; Myneni, S. Iron-rich nanoparticles in natural aquatic environments. Minerals 2019, 9, 287. [Google Scholar] [CrossRef]
- Yoshida, A.; Ohata, S.; Moteki, N.; Adachi, K.; Mori, T.; Koike, M.; Takami, A. Abundance and emission flux of the anthropogenic iron oxide aerosols from the East Asian continental outflow. J. Geophys. Res. Atmos. 2018, 123, 11194–11211. [Google Scholar] [CrossRef]
- Maher, B.A.; González-Maciel, A.; Reynoso-Robles, R.; Torres-Jardón, R.; Calderón-Garcidueñas, L. Iron-rich air pollution nanoparticles: An unrecognised environmental risk factor for myocardial mitochondrial dysfunction and cardiac oxidative stress. Environ. Res. 2020, 188, 109816. [Google Scholar] [CrossRef]
- Garry, S.; Nesslany, F.; Aliouat, E.; Haguenoer, J.M.; Marzin, D. Hematite (Fe2O3) acts by oxidative stress and potentiates benzo[a]pyrene genotoxicity. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2004, 563, 117–129. [Google Scholar] [CrossRef]
- Auffan, M.; Decome, L.; Rose, J.; Orsiere, T.; De Meo, M.; Briois, V.; Bottero, J.-Y. In vitro interactions between DMSA-coated maghemite nanoparticles and human fibroblasts: A physicochemical and cyto-genotoxic study. Environ. Sci. Technol. 2006, 40, 4367–4373. [Google Scholar] [CrossRef]
- Chen, C.; Hu, J.; Shao, D.; Li, J.; Wang, X. Adsorption behavior of multiwall carbon nanotube/iron oxide magnetic composites for Ni(II) and Sr(II). J. Hazard. Mater. 2009, 164, 923–928. [Google Scholar] [CrossRef]
- Tada, Y.; Yano, N.; Takahashi, H.; Yuzawa, K.; Ando, H.; Kubo, Y.; Nakae, D. Acute phase pulmonary responses to a single intratracheal spray instillation of magnetite (Fe3O4) nanoparticles in Fischer 344 rats. J. Toxicol. Pathol. 2012, 25, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Pease, C.; Rücker, T.; Birk, T. Review of the evidence from epidemiology, toxicology, and lung bioavailability on the carcinogenicity of inhaled iron oxide particulates. Chem. Res. Toxicol. 2016, 29, 237–254. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wen, W.; Wang, X.; Huang, D.; Cao, J.; Qi, X.; Shen, S. Ultrasmall iron oxide nanoparticles cause significant toxicity by specifically inducing acute oxidative stress to multiple organs. Part. Fibre Toxicol. 2022, 19, 24. [Google Scholar] [CrossRef]
- Ferraz, F.S.; de PF Dantas, G.; Coimbra, J.L.; López, J.L.; Lacerda, S.M.; Dos Santos, M.L.; Costa, G.M. Effects of superparamagnetic iron oxide nanoparticles (SPIONs) testicular injection on Leydig cell function and sperm production in a murine model. Reprod. Toxicol. 2024, 126, 108584. [Google Scholar] [CrossRef] [PubMed]
- Imam, S.Z.; Lantz-McPeak, S.M.; Cuevas, E.; Rosas-Hernandez, H.; Liachenko, S.; Zhang, Y.; Gracia, E.; Sarkar, S.; Binienda, Z.K. Iron oxide nanoparticles induce dopaminergic damage: In vitro pathways and in vivo imaging reveal mechanism of neuronal damage. Molecular Neurobiology 2015, 52, 913–926. [Google Scholar] [CrossRef]
- Patten, K.T.; Valenzuela, A.E.; Wallis, C.; Berg, E.L.; Silverman, J.L.; Bein, K.J.; Lein, P.J. The effects of chronic exposure to ambient traffic-related air pollution on Alzheimer’s disease phenotypes in wildtype and genetically predisposed male and female rats. Environmental Health Perspectives 2021, 129, 057005. [Google Scholar] [CrossRef]
- Abakumov, M.A.; Semkina, A.S.; Skorikov, A.S.; Vishnevskiy, D.A.; Ivanova, A.V.; Mironova, E.; Chekhonin, V.P. Toxicity of iron oxide nanoparticles: Size and coating effects. J. Biochem. Mol. Toxicol. 2018, 32, e22225. [Google Scholar] [CrossRef] [PubMed]
- Caro, C.; Egea-Benavente, D.; Polvillo, R.; Royo, J.L.; Leal, M.P.; García-Martín, M.L. Comprehensive toxicity assessment of PEGylated magnetic nanoparticles for in vivo applications. Colloids Surf. B Biointerfaces 2019, 177, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Bourgkard, E.; Wild, P.; Courcot, B.; Diss, M.; Ettlinger, J.; Goutet, P.; Moulin, J.-J. Lung cancer mortality and iron oxide exposure in a French steel-producing factory. Occup. Environ. Med. 2009, 66, 175–181. [Google Scholar] [CrossRef]
- Marchini, T.; Magnani, N.; D’Annunzio, V.; Tasat, D.; Gelpi, R.J.; Alvarez, S.; Evelson, P. Impaired cardiac mitochondrial function and contractile reserve following an acute exposure to environmental particulate matter. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 2545–2552. [Google Scholar] [CrossRef]
- Marchini, T.; Magnani, N.D.; Paz, M.L.; Vanasco, V.; Tasat, D.; Maglio, D.G.; et al. Time course of systemic oxidative stress and inflammatory response induced by an acute exposure to residual oil fly ash. Toxicol Appl Pharmacol. 2014, 274, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Radu, M.; Din, I.M.; Hermenean, A.; Cinteză, O.L.; Burlacu, R.; Ardelean, A.; et al. Exposure to iron oxide nanoparticles coated with phospholipid-based polymeric micelles induces biochemical and histopathological pulmonary changes in mice. Int J Mol Sci. 2015, 16, 29417–29435. [Google Scholar] [CrossRef]
- Di Bona, K.R.; Xu, Y.; Gray, M.; Fair, D.; Hayles, H.; Milad, L.; et al. Short- and long-term effects of prenatal exposure to iron oxide nanoparticles: Influence of surface charge and dose on developmental and reproductive toxicity. Int J Mol Sci. 2015, 16, 30251–30268. [Google Scholar] [CrossRef]
- Dos Santos, T.M.; Righetti, R.F.; do Nascimento Camargo, L.; Leick, E.A.; Fukuzaki, S.; de Campos, E.C.; et al. Effect of VAChT reduction on lung alterations induced by exposure to iron particles in an asthma model. J Inflamm. 2024, 21, 24. [Google Scholar] [CrossRef]
- Murthy, M.K.; Khandayataray, P.; Mohanty, C.S.; Pattanayak, R. Investigating the toxic mechanism of iron oxide nanoparticles-induced oxidative stress in tadpole (Duttaphrynus melanostictus): A combined biochemical and molecular study. Environ Toxicol Pharmacol. 2024, 107, 104432. [Google Scholar] [CrossRef]
- Pauluhn, J. Retrospective analysis of 4-week inhalation studies in rats with focus on fate and pulmonary toxicity of two nanosized aluminum oxyhydroxides (boehmite) and pigment-grade iron oxide (magnetite): The key metric of dose is particle mass and not particle surface area. Toxicology 2009, 259, 140–148. [Google Scholar] [CrossRef]
- Pauluhn, J. Subchronic inhalation toxicity of iron oxide (magnetite, Fe3O4) in rats: Pulmonary toxicity is determined by the particle kinetics typical of poorly soluble particles. J. Appl. Toxicol. 2012, 32, 488–504. [Google Scholar] [CrossRef] [PubMed]
- Szalay, B.; Tátrai, E.; Nyírő, G.; Vezér, T.; Dura, G. Potential toxic effects of iron oxide nanoparticles in in vivo and in vitro experiments. J Appl Toxicol. 2012, 32, 446–453. [Google Scholar] [CrossRef]
- Jarockyte, G.; Daugelaite, E.; Stasys, M.; Statkute, U.; Poderys, V.; Tseng, T.C.; et al. Accumulation and toxicity of superparamagnetic iron oxide nanoparticles in cells and experimental animals. Int J Mol Sci. 2016, 17, 1193. [Google Scholar] [CrossRef]
- Matusiak, K.; Skoczen, A.; Setkowicz, Z.; Kubala-Kukus, A.; Stabrawa, I.; Ciarach, M.; et al. The elemental changes occurring in the rat liver after exposure to PEG-coated iron oxide nanoparticles: total reflection X-ray fluorescence (TXRF) spectroscopy study. Nanotoxicology 2017, 11, 1225–1236. [Google Scholar] [CrossRef]
- Könczöl, M.; Ebeling, S.; Goldenberg, E.; Treude, F.; Gminski, R.; Gieré, R.; Mersch-Sundermann, V. Cytotoxicity and genotoxicity of size-fractionated iron oxide (magnetite) in A549 human lung epithelial cells: Role of ROS, JNK, and NF-κB. Chem. Res. Toxicol. 2011, 24, 1460–1475. [Google Scholar] [CrossRef]
- Ramesh, V.; Ravichandran, P.; Copeland, C.L.; Gopikrishnan, R.; Biradar, S.; Goornavar, V.; Hall, J.C. Magnetite induces oxidative stress and apoptosis in lung epithelial cells. Mol. Cell. Biochem. 2012, 363, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Alhadlaq, H.A.; Alam, J.; Majeed, K.; Ali, D.; Alarafi, S. Iron oxide nanoparticle-induced oxidative stress and genotoxicity in human skin epithelial and lung epithelial cell lines. Curr. Pharm. Des. 2013, 19, 6681–6690. [Google Scholar] [CrossRef] [PubMed]
- Könczöl, M.; Weiss, A.; Stangenberg, E.; Gminski, R.; Garcia-Käufer, M.; Gieré, R.; Mersch-Sundermann, V. Cell-cycle changes and oxidative stress response to magnetite in A549 human lung cells. Chem. Res. Toxicol. 2013, 26, 693–702. [Google Scholar] [CrossRef]
- Gokduman, K.; Bestepe, F.; Li, L.; Yarmush, M.L.; Usta, O.B. Dose-, treatment- and time-dependent toxicity of superparamagnetic iron oxide nanoparticles on primary rat hepatocytes. Nanomedicine 2018, 13, 1267–1284. [Google Scholar] [CrossRef]
- Kirschvink, J.L.; Kobayashi-Kirschvink, A.; Woodford, B.J. Magnetite biomineralization in the human brain. Proc Natl Acad Sci U S A 1992, 89, 7683–7687. [Google Scholar] [CrossRef] [PubMed]
- Totsuka, Y.; Ishino, K.; Kato, T.; Goto, S.; Tada, Y.; Nakae, D.; et al. Magnetite nanoparticles induce genotoxicity in the lungs of mice via inflammatory response. Nanomaterials 2014, 4, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Orel, V.; Shevchenko, A.; Romanov, A.; Tselepi, M.; Mitrelias, T.; Barnes, C.H.; et al. Magnetic properties and antitumor effect of nanocomplexes of iron oxide and doxorubicin. Nanomedicine (Lond) 2015, 11, 47–55. [Google Scholar] [CrossRef]
- Özgür, M.E.; Ulu, A.; Balcıoğlu, S.; Özcan, I.; Köytepe, S.; Ateş, B. The toxicity assessment of iron oxide (Fe3O4) nanoparticles on physical and biochemical quality of rainbow trout spermatozoon. Toxics 2018, 6, 62. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Xu, B.; Huang, X.; Zhang, Y.; Yu, M.; Han, X.; et al. Metabolomics reveals the role of acetyl-L-carnitine metabolism in γ-Fe2O3 nanoparticle-induced embryonic development toxicity via mitochondria damage. Nanotoxicology 2019, 13, 204–220. [Google Scholar] [CrossRef]
- Kaloyianni, M.; Dimitriadi, A.; Ovezik, M.; Stamkopoulou, D.; Feidantsis, K.; Kastrinaki, G.; et al. Magnetite nanoparticles effects on adverse responses of aquatic and terrestrial animal models. Journal of Hazardous Materials 2020, 383, 121204. [Google Scholar] [CrossRef]
- Fascineli, M.L.; Cáceres-Vélez, P.R.; Pinheiro, W.O.; Chaves, S.B.; Sousa, M.H.; Peternella, W.S.; et al. Lack of genotoxicity of iron oxide maghemite (γ-Fe2O3) and magnetite (Fe3O4) nanoparticles to Oreochromis niloticus after acute exposures. Genetics and Molecular Biology 2024, 47, e20230330. [Google Scholar] [CrossRef]
- Silva, A.C.; Dos Santos, A.G.R.; Pieretti, J.C.; Rolim, W.R.; Seabra, A.B.; Ávila, D.S. Iron oxide/silver hybrid nanoparticles impair the cholinergic system and cause reprotoxicity in Caenorhabditis elegans. Food and Chemical Toxicology 2023, 179, 113945. [Google Scholar] [CrossRef]
- Basso Abraham, A.L.R.; Rockenbach de Ávila, M.; Torres, R.; Diz, V.E. Magnetite nanoparticles as a promising non contaminant method to control populations of fruit flies (Diptera: Tephritidae). J Appl Biotechnol Bioeng. 2021, 8, 112–117. [Google Scholar] [CrossRef]
- Hanini, A.; Schmitt, A.; Kacem, K.; Chau, F.; Ammar, S.; Gavard, J. Evaluation of iron oxide nanoparticle biocompatibility. International Journal of Nanomedicine 2011, 6, 787–794. [Google Scholar] [CrossRef]
- Safi, M.; Sarrouj, H.; Sandre, O.; Mignet, N.; Berret, J.-F. Interactions between sub-10-nm iron and cerium oxide nanoparticles and 3T3 fibroblasts: The role of the coating and aggregation state. Nanotechnology 2010, 21, 145103. [Google Scholar] [CrossRef]
- Caldeira, D.F.; Paulini, F.; Silva, R.C.; Azevedo, R.B.D.; Lucci, C.M. In vitro exposure of bull sperm cells to DMSA-coated maghemite nanoparticles does not affect cell functionality or structure. Int. J. Hyperthermia 2018, 34, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Iversen, N.K.; Frische, S.; Thomsen, K.; Laustsen, C.; Pedersen, M.; Hansen, P.B.; Wang, T. Superparamagnetic iron oxide polyacrylic acid coated γ-Fe2O3 nanoparticles do not affect kidney function but cause acute effect on the cardiovascular function in healthy mice. Toxicol. Appl. Pharmacol. 2013, 266, 276–288. [Google Scholar] [CrossRef]
- Qualhato, G.; Rocha, T.L.; de Oliveira Lima, E.C.; Silva DMe Cardoso, J.R.; Grisolia, C.K.; de Sabóia-Morais, S.M.T. Genotoxic and mutagenic assessment of iron oxide (maghemite-γ-Fe2O3) nanoparticle in the guppy Poecilia reticulata. Chemosphere 2017, 183, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Qualhato, G.; de Sabóia-Morais, S.M.T.; Silva, L.D.; Rocha, T.L. Melanomacrophage response and hepatic histopathologic biomarkers in the guppy Poecilia reticulata exposed to iron oxide (maghemite) nanoparticles. Aquat. Toxicol. 2018, 198, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Chun, H.S.; Moon, J.Y.; Choi, J.S.; Park, D.; Lee, Y.C. Correlation of α/γ-Fe2O3 nanoparticles with the toxicity of particulate matter originating from subway tunnels in Seoul stations, Korea. J. Hazard. Mater. 2020, 382, 121175. [Google Scholar]
- Gürkan, M.; Gürkan, S.E.; Yılmaz, S.; Ateş, M. Comparative toxicity of alpha and gamma iron oxide nanoparticles in rainbow trout: Histopathology, hematology, accumulation, and oxidative stress. Water Air Soil Pollut. 2021, 232, 37. [Google Scholar] [CrossRef]
- Gonçalves, B.B.; Dias, F.C.; de Souza Trigueiro, N.S.; Marques, E.; Rodrigues, C.C.; Madureira, I.B.; Rocha, T.L. Chronic exposure to iron oxide nanoparticles (γ-Fe2O3) induces gonadal histopathology on male guppies (Poecilia reticulata). Environ. Nanotechnol. Monit. Manag. 2021, 16, 100522. [Google Scholar] [CrossRef]
- Monge-Fuentes, V.; Garcia, M.P.; Tavares, M.C.H.; Valois, C.R.; Lima, E.C.; Teixeira, D.S.; et al. Biodistribution and biocompatibility of DMSA-stabilized maghemite magnetic nanoparticles in nonhuman primates (Cebus spp.). Nanomedicine 2011, 6, 1529–1544. [Google Scholar] [CrossRef]
- Garry, S.; Nesslany, F.; Aliouat, E.; Haguenoer, J.M.; Marzin, D. Hematite (Fe2O3) enhances benzo[a]pyrene genotoxicity in endotracheally treated rat, as determined by comet assay. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2003, 538, 19–29. [Google Scholar] [CrossRef]
- Gaharwar, U.S.; Paulraj, R. Iron oxide nanoparticles induced oxidative damage in peripheral blood cells of rat. J. Biomed. Sci. Eng. 2015, 8, 274. [Google Scholar] [CrossRef]
- Askri, D.; Cunin, V.; Ouni, S.; Béal, D.; Rachidi, W.; Sakly, M.; Sève, M. Effects of iron oxide nanoparticles (γ-Fe2O3) on liver, lung and brain proteomes following sub-acute intranasal exposure: A new toxicological assessment in rat model using iTRAQ-based quantitative proteomics. Int. J. Mol. Sci. 2019, 20, 5186. [Google Scholar] [CrossRef] [PubMed]
- Smital, K.; Niharika, S.; Mansee, T. Sub-acute toxicity assessment of green synthesized hematite nanoparticles (α-Fe2O3 NPs) using Wistar rat. Res. J. Biotechnol. 2020, 15, 4. [Google Scholar]
- Warshawsky, D.; Reilman, R.; Cheu, J.; Radike, M.; Rice, C. Influence of particle dose on the cytotoxicity of hamster and rat pulmonary alveolar macrophage in vitro. J. Toxicol. Environ. Health A 1994, 42, 407–421. [Google Scholar] [CrossRef]
- Bhattacharya, K.; Hoffmann, E.; Schins, R.F.; Boertz, J.; Prantl, E.M.; Alink, G.M.; et al. Comparison of micro- and nanoscale Fe3+-containing (hematite) particles for their toxicological properties in human lung cells in vitro. Toxicological Sciences 2012, 126, 173–182. [Google Scholar] [CrossRef]
- Freyria, F.S.; Bonelli, B.; Tomatis, M.; Ghiazza, M.; Gazzano, E.; Ghigo, D.; et al. Hematite nanoparticles larger than 90 nm show no sign of toxicity in terms of lactate dehydrogenase release, nitric oxide generation, apoptosis, and comet assay in murine alveolar macrophages and human lung epithelial cells. Chemical Research in Toxicology 2012, 25, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Kalive, M.; Zhang, W.; Chen, Y.; Capco, D.G. Human intestinal epithelial cells exhibit a cellular response indicating a potential toxicity upon exposure to hematite nanoparticles. Cell Biology and Toxicology 2012, 28, 343–368. [Google Scholar] [CrossRef]
- Faust, J.J.; Zhang, W.; Chen, Y.; Capco, D.G. Alpha-Fe2O3 elicits diameter-dependent effects during exposure to an in vitro model of the human placenta. Cell Biology and Toxicology 2014, 30, 31–53. [Google Scholar] [CrossRef]
- Cardillo, D.; Tehei, M.; Hossain, M.S.; Islam, M.M.; Bogusz, K.; Shi, D.; et al. Synthesis-dependent surface defects and morphology of hematite nanoparticles and their effect on cytotoxicity in vitro. ACS Applied Materials & Interfaces 2016, 8, 5867–5876. [Google Scholar] [CrossRef]
- Lewis, C.S.; Torres, L.; Miyauchi, J.T.; Rastegar, C.; Patete, J.M.; Smith, J.M.; et al. Absence of cytotoxicity towards microglia of iron oxide (α-Fe2O3) nanorhombohedra. Toxicology Research 2016, 5, 836–847. [Google Scholar] [CrossRef] [PubMed]
- Nations, S.; Wages, M.; Cañas, J.E.; Maul, J.; Theodorakis, C.; Cobb, G.P. Acute effects of Fe2O3, TiO2, ZnO and CuO nanomaterials on Xenopus laevis. Chemosphere 2011, 83, 1053–1061. [Google Scholar] [CrossRef]
- Oliveira, V.R.; Uriarte, J.J.; Falcones, B.; Jorba, I.; Zin, W.A.; Farré, R.; Almendros, I. Biomechanical response of lung epithelial cells to iron oxide and titanium dioxide nanoparticles. Frontiers in Physiology 2019, 10, 1047. [Google Scholar] [CrossRef]
- Rafieepour, A.; Azari, M.R.; Khodagholi, F.; Jaktaji, J.P.; Mehrabi, Y.; Peirovi, H. Interactive toxicity effect of combined exposure to hematite and amorphous silicon dioxide nanoparticles in human A549 cell line. Toxicology and Industrial Health 2021, 37, 289–302. [Google Scholar] [CrossRef]
- Goadby, K.W. Fibrosis of the lung in iron miners. Journal of the Royal Microscopical Society 1925, 45, 432–437. [Google Scholar] [CrossRef]
- Lawler, A.B.; Mandel, J.S.; Schuman, L.M.; Lubin, J.H. A retrospective cohort mortality study of iron ore (hematite) miners in Minnesota. Journal of Occupational Medicine 1985, 27, 507–517. [Google Scholar]
- Wang, B.; Feng, W.Y.; Wang, M.; Shi, J.W.; Zhang, F.; Ouyang, H.; et al. Transport of intranasally instilled fine Fe2O3 particles into the brain: micro-distribution, chemical states, and histopathological observation. Biological Trace Element Research 2007, 118, 233–243. [Google Scholar] [CrossRef]
- Rajendran, K.; Pujari, L.; Krishnamoorthy, M.; Sen, S.; Dharmaraj, D.; Karuppiah, K.; Ethiraj, K. Toxicological evaluation of biosynthesised hematite nanoparticles in vivo. Colloids and Surfaces B: Biointerfaces 2021, 198, 111475. [Google Scholar] [CrossRef] [PubMed]
- Carleton, H.M. The effects produced by the inhalation of haematite and iron dusts in guinea-pigs. Epidemiol. Infect. 1927, 26, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, A.K.; Rahman, Q.; Viswanathan, P.N. Enzymatic changes in guinea pig lungs due to haematite dust. Toxicology Letters 1979, 3, 51–54. [Google Scholar] [CrossRef]
- Das, B.; Khatoon, N.; Srivastava, R.C.; Viswanathan, P.N.; Rahman, Q. Biochemical studies on the toxicity of hematite dust. Environmental Research 1983, 32, 372–381. [Google Scholar] [CrossRef] [PubMed]
| Authors | Biological Model | Exposure Route/Particle Type | Main Biological Effects Observed |
| Imam et al., 2015 | Sprague–Dawley rat | Intravenous injection (10 nm iron oxide NPs) | Brain penetration and retention; decreased T2 relaxation values; reduced striatal dopamine levels; neuronal, dopaminergic, and cerebral vascular damage |
| Patten et al., 2021 | TgF344-AD and WT rats (♂/♀) | Chronic inhalation of traffic-related air pollution (TRAP) | Increased cerebral particle burden; behavioral impairments; Alzheimer-like phenotype (↑β-amyloid, ↑phosphorylated tau, glial activation), dependent on age, sex, and genotype |
| Marchini et al., 2013 | Female Swiss mouse | Intranasal instillation (ROFA, 1 mg/kg) | Reduced cardiac oxygen consumption; mitochondrial dysfunction; decreased ATP production |
| Marchini et al., 2014 | Female Swiss mouse | Intranasal instillation (ROFA) | Systemic oxidative damage; redox imbalance (↓GSH, ↑GSSG); systemic inflammatory response (↑TNF-α, ↑IL-6) |
| Radu et al., 2015 | Male CD-1 mouse | Intravenous injection (IONPs–PM, 5–15 mg Fe/kg) | Pulmonary oxidative stress; activation of apoptotic pathways; dose-dependent lung injury |
| Di Bona et al., 2015 | Pregnant CD-1 mouse | Intraperitoneal injection (Fe2O3 nanoparticles) | Fetal loss; surface charge–dependent reproductive toxicity; transgenerational effects |
| Caro et al., 2019 | Balb/c mouse | Intravenous injection (PEGylated iron oxide NPs) | Rapid hepatic uptake without histopathological damage; absence of acute systemic toxicity |
| Dos Santos et al., 2024 | WT and VAChT KD mice | Passive environmental inhalation (iron-rich PM10/PM2.5) | Pulmonary inflammation; airway hyperresponsiveness; oxidative stress; exacerbated effects under cholinergic deficiency |
| Authors | Cell Model | Exposure Conditions | Main Cellular Effects Observed |
| Imam et al., 2015 | Human neuronal cells (SH-SY5Y) | Direct exposure (10–30 nm; 2.5–10 μg/mL, 24 h) | Dopaminergic dysfunction; increased ROS production; mitochondrial impairment; activation of pro-apoptotic pathways |
| Imam et al., 2015 | Rat brain microvascular endothelial cells (rBMVECs) | Direct exposure (24 h) | Increased ROS levels; reduced transendothelial electrical resistance (TEER); increased permeability; functional impairment of the blood–brain barrier |
| Abakumov et al., 2018 | Human fibroblasts (HF) and glioblastoma cells (U251) | Direct exposure (24–48 h) | Size- and time-dependent cytotoxicity and genotoxicity; increased ROS generation; DNA fragmentation |
| Caro et al., 2019 | Murine microglial cells (N13) | Direct exposure (0.1–100 μg/mL, 24 h) | No detectable cytotoxicity; preserved cell viability; high biocompatibility |
| Authors | Biological model | Route of administration | Damage induced/Observed effects |
| Pauluhn, 2009 | Rattus norvegicus | Nose-only inhalation | Dose-dependent pulmonary accumulation; reduced pulmonary clearance; lung inflammation (↑ LDH, proteins, PMNs in BAL); cellular infiltration; prolonged particle retention |
| Pauluhn, 2012 | Wistar rat | Nose-only inhalation, subchronic | Pulmonary inflammation; ↑ neutrophils in BAL; histopathological changes in the respiratory tract; ↑ lung and lung-associated lymph node weights; increased septal collagen; NOAEL 4.7 mg/m3 |
| Szalay et al., 2012 | Male Wistar rat | Single intratracheal instillation | Focal pulmonary inflammation; mild pulmonary fibrosis; reduced body-weight gain; decreased relative weights of lung, liver, and kidney |
| Tada et al., 2012 | Fischer 344 rat | Single intratracheal instillation | Increased lung weight; accumulation in alveolar macrophages; inflammatory infiltration; multinucleated cells; pulmonary granulomas; reactive epithelial changes |
| Jarockyte et al., 2016 | Wistar rat | Intramuscular injection | Dose-dependent tissue retention; slow clearance; persistence of nanoparticles at high doses |
| Matusiak et al., 2017 | Male Wistar rat | Intravenous injection | Transient increase in liver mass; elevated hepatic Fe levels; persistent alterations in Ca, Cu, and Zn; no significant changes in body weight |
| Totsuka et al., 2014 | Mus musculus (ICR and gpt delta) | Intratracheal instillation (single and repeated) | Pulmonary DNA damage; ↑ oxidative adducts (8-oxodG, HεdG/HεdC); mutagenesis; pulmonary inflammation; granuloma formation |
| Orel et al., 2015 | Male C57BL/6 mouse | Intravenous injection | Mitochondrial alterations in tumor cells; ↑ oxidative stress; ↓ ATP production; inhibition of tumor growth (controlled therapeutic application) |
| Wu et al., 2022 | Male ICR mouse | Intravenous injection | Size-dependent acute and lethal toxicity; cardiotoxicity; ↑ ROS and ·OH; elevated ALT/AST; systemic tissue damage |
| Authors | Cell model | Exposure type | Induced damage/Observed effects |
| Könczöl et al., 2011 | Human pulmonary epithelial cells (A549) | Direct exposure in culture | Oxidative stress (↑ ROS), mitochondrial membrane potential disruption, DNA damage (Comet assay), micronucleus formation, sustained JNK activation and NF-κB modulation; ROS-dependent genotoxicity |
| Ramesh et al., 2012 | Rat pulmonary epithelial cells (RL-65) | Direct exposure in culture | Dose-dependent inhibition of proliferation, ↑ ROS and lipid peroxidation, ↓ GSH and SOD, activation of caspases-3/-8, DNA fragmentation, and apoptosis |
| Ahamed et al., 2013 | Human epithelial cells A431 (skin) and A549 (lung) | Direct exposure in culture | Dose-dependent cytotoxicity, ↑ LDH, ↑ ROS and MDA, ↓ GSH, DNA damage, and caspase-3/-9-mediated apoptosis |
| Könczöl et al., 2013 | Human cells A549 and H1299 | Direct exposure in culture | Size-dependent superoxide radical generation, ↓ GSH, ↑ CAT activity, cell-cycle alteration (↑ sub-G1 population), p21 activation without apoptosis induction |
| Jarockyte et al., 2016 | Mouse embryonic fibroblasts (NIH3T3) | Direct exposure in culture | Perinuclear endocytic internalization, minimal cytotoxicity, mild morphological changes; relatively high biocompatibility |
| Gokduman et al., 2018 | Primary hepatocytes from Lewis rats | Direct exposure in culture (single and cumulative dosing) | Reduced viability, ↑ ROS, impaired hepatic functions (albumin and urea production), time- and accumulation-dependent increase in cell death |
| Wu et al., 2022 | MCF-7 cells (human breast carcinoma) | Direct exposure in culture | Size-dependent toxicity (<5 nm), ·OH generation, ↑ ROS, marked reduction in viability, nuclear entry of ultrasmall nanoparticles, and acute cellular damage |
| Authors | Biological model | Route of administration | Induced damage/Observed effects |
| Hanini et al., 2011 | Wistar rats | Single intravenous injection | ~50% increase in leukocyte count; inflammatory infiltration and cellular damage in lung, liver, and kidney; no changes in body weight or severe clinical signs |
| Iversen et al., 2013 | BALB/cJ mice | Intravenous injection | Significant but transient decrease in mean arterial pressure (12–24 h) and reversible reduction in arterial contractility; no renal damage |
| Huang et al., 2019 | Pregnant ICR female mice | Oral (gastrointestinal) exposure | At high doses: fetal resorption and embryonic death, reduced placental weight, placental histological alterations, decreased Crat expression, reduced mitochondrial ATP production, and oxidative stress–related alterations |
| Monge-Fuentes et al., 2011 | Primate (Cebus spp.) | Intravenous injection | Mild long-term hepatic alterations; particle presence in alveolar macrophages, hepatocytes, and renal tubules; no necrosis, hemorrhage, or hematological or behavioral alterations |
| Authors | Biological model (cells) | Exposure type | Induced damage/Observed effects |
| Auffan et al., 2006 | Human dermal fibroblasts | In vitro exposure | Mild reduction in cell viability at 10−6–10−3 g/L (2–24 h); absence of genotoxicity (Comet assay); increased mitochondrial activity at higher concentrations; no detectable genetic damage |
| Safi et al., 2010 | Murine fibroblasts (NIH/3T3) | In vitro exposure | No cytotoxic effects observed (≈100% viability); cellular uptake dependent on surface coating: high uptake with citrate (~250 pg Fe/cell) and low uptake with PAA2K (<30 pg/cell); no associated cellular damage |
| Hanini et al., 2011 | Human endothelial cells (HUVEC, Ea.hy 926) | In vitro exposure | Significant reduction in viability at 48–72 h; increased cytotoxicity from 24 h; elevated ROS production; absence of caspase-3 activation, suggesting predominantly necrotic cell death |
| Caldeira et al., 2018 | Bovine spermatozoa | In vitro exposure | No alterations in motility, membrane integrity, acrosomal reaction, or ultrastructure; presence of extracellular aggregates without internalization; absence of toxic effects |
| Author (year) | Biological model | Route of administration | Induced damage/Observed effects |
| Carleton (1927) | Guinea pig (Cavia porcellus) | Inhalation of fine dust in a closed chamber | Mild–moderate pulmonary inflammatory response (alveolar epithelial proliferation, transient bronchitis); dust migration to subpleural regions and lymph nodes; progressive pulmonary clearance without fibrosis; limited systemic iron accumulation, mainly in spleen |
| Jaiswal et al. (1979) | Guinea pig (Cavia porcellus) | Intratracheal instillation | Persistent pulmonary metabolic alterations: increased fructose-1,6-bisphosphatase, decreased G6PD, marked reduction of mitochondrial succinate dehydrogenase and carbonic anhydrase; mitochondrial dysfunction without overt fibrosis |
| Das et al. (1983) | Guinea pig (Cavia porcellus) | Intratracheal instillation | Systemic iron mobilization to liver, spleen, bone marrow, kidney, and heart; pulmonary mitochondrial accumulation; increased cytochrome c oxidase and altered mitochondrial permeability, without marked pulmonary fibrosis |
| Wang et al. (2007) | Male CD-ICR mouse | Intranasal instillation | Brain iron accumulation in olfactory bulb and brainstem; increased Fe(III); neuronal degeneration in hippocampal CA3; evidence supporting transport via olfactory and trigeminal pathways |
| Lewis et al. (2016) | MacGreen transgenic mouse | Bilateral stereotactic injection into dorsal hippocampus | Uptake by hippocampal microglia without inflammatory activation or apparent structural alterations at 24 h |
| Rajendran et al. (2021) | Swiss albino mouse (Mus musculus) | Oral gavage administration | No mortality or acute toxicity; hematological, biochemical, and histopathological parameters largely normal, with a slight increase in bilirubin |
| Garry et al. (2003) | Male Sprague–Dawley rat | Endotracheal instillation | Hematite alone showed no genotoxicity; B[a]P and B[a]P-coated hematite induced significant DNA damage, indicating enhancement under co-exposure |
| Gaharwar & Paulraj (2015) | Male Wistar rat | Intravenous injection | Dose- and time-dependent hematological and inflammatory alterations; systemic oxidative stress without detectable genotoxic damage |
| Author (year) | Biological model | Exposure type | Induced damage/Observed effects |
| Warshawsky et al. (1994) | Alveolar macrophages | Direct in vitro exposure | Efficient particle phagocytosis; preserved viability at most concentrations; moderate viability reduction (~67%) only at the highest dose |
| Garry et al. (2004) | Alveolar macrophages, pulmonary cells, hepatocytes, and peripheral lymphocytes from Sprague–Dawley rats | In vitro exposure | Hematite alone: no genotoxicity; B[a]P alone: DNA damage; B[a]P adsorbed onto Fe2O3: marked enhancement of genotoxic damage in a cell-type–dependent manner, demonstrating a vector effect of hematite |
| Bhattacharya et al. (2012) | Human lung cells (BEAS-2B and IMR-90) | In vitro exposure | Intracellular internalization; reduced viability and increased ROS at ≥50 µg/mL, more pronounced for nanoparticles; toxicity strongly modulated by agglomeration and biomolecule interactions |
| Freyria et al. (2012) | Murine alveolar macrophages (MH-S) and human pulmonary epithelial cells (A549) | In vitro exposure | No cytotoxicity, apoptosis/necrosis, DNA damage, or nitric oxide production; low acute toxicity under the evaluated conditions |
| Kalive et al. (2012) | Human intestinal epithelial cells (Caco-2) | Single exposure | Size-dependent disruption of epithelial barrier integrity; 17 nm particles reduced TEER, altered cell junctions and epithelial architecture without inducing early cell death |
| Faust et al. (2014) | Human placental epithelial cells (BeWo b30) | In vitro exposure | Progressive and irreversible TEER reduction with 50 and 78 nm particles; increased ROS, tight junction disruption, and apoptotic-related alterations |
| Cardillo et al. (2016) | Canine renal epithelial cells (MDCK) | In vitro exposure | Morphology-dependent internalization; elongated particles showed higher uptake; significant ROS increase observed only for particles with specific surface characteristics |
| Lewis et al. (2016) | Primary microglia from MacGreen mice and N9 microglial cell line | In vitro exposure | Size-dependent internalization; overall low cytotoxicity, with significant effects only at the highest concentration and longer exposure times |
| Lee et al. (2020) | Human HeLa cells | In vitro exposure | Minimal effects at ≤50 µg/mL; reduced viability, oxidative stress, and cell death observed at 100 µg/mL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




