Submitted:
13 January 2026
Posted:
14 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell line and Culture Conditions
2.2. Experimental Design and Treatment Conditions
2.3. Plate Layout and Controls
- Vehicle (F0; “CTR”), on every plate.
- Positive ROS control: H₂O₂ 100 μM for 60 min (plate-end spike) where indicated.
- Antioxidant control: N-acetyl-L-cysteine (NAC) 0.5 mM maintained throughout the 48 h exposure (where indicated).
- ΔΨm depolarization control (for JC-1): CCCP/FCCP 50 μM for 5 min before reading; plates kept protected from light.
| Condition | Progesterone (P4) [nM]* | Estradiol (E2) [nM]* | Dienogest (DNG) [nM]* | Ethinylestradiol (EE) [nM]* | Description |
|---|---|---|---|---|---|
| F0 | 0 | 0 | 0 | 0 | Vehicle (control) |
| F1 | 1.87 | 0.28 | 0 | 0 | Follicular-like background |
| F2 | 40.0 | 0.50 | 0 | 0 | Luteal-like background |
| F3 | 1.87 | 0.28 | 160 | 0.30 | F1 + DNG/EE |
| F4 | 40.0 | 0.50 | 160 | 0.30 | F2 + DNG/EE |
| F5 | 0 | 0 | 160 | 0.30 | DNG/EE only |
2.4. Fluorimetric Assays
2.5. JC-1 (Mitochondrial Membrane Potential, ΔΨm)
- Green (monomer): Ex 485–490 nm / Em ~530 nm
- Red (aggregate): Ex 525–540 nm / Em 590–610 nm
- The red/green ratio was used as the primary ΔΨm endpoint.
2.6. H₂DCFDA/DCF (ROS)
2.7. DAF-FM Diacetate (Nitric Oxide, NO)
2.8. Hoechst 33342 (Nuclear Content/Proportionality)
2.9. Data Processing and Normalization
- Primary endpoints: JC-1 red/green ratio (ΔΨm), DCF (ROS), DAF-FM (NO), and Hoechst (nuclear signal).
- Normalization: In addition to raw values, we report Hoechst-normalized endpoints to correct for cell-number bias:
- Aggregation: Technical replicates (n = 8) were summarized per condition/plate before inferential testing.
2.10. Quality Control and Statistics
- Across-condition comparisons: Kruskal–Wallis.
- Pairwise contrasts (e.g., F1–F5 vs F0; F1 vs F2; background effects F3 vs F1, F4 vs F2): Mann–Whitney tests, with Benjamini–Hochberg FDR control (q = 0.05).
- Effect sizes: median differences and rank-biserial correlation; 95% CIs by bootstrap where applicable.
- All analyses were run in MATLAB (The MathWorks, Natick MA) with custom scripts (provided as Supplementary)
2.11. Reagents and Solutions
3. Results
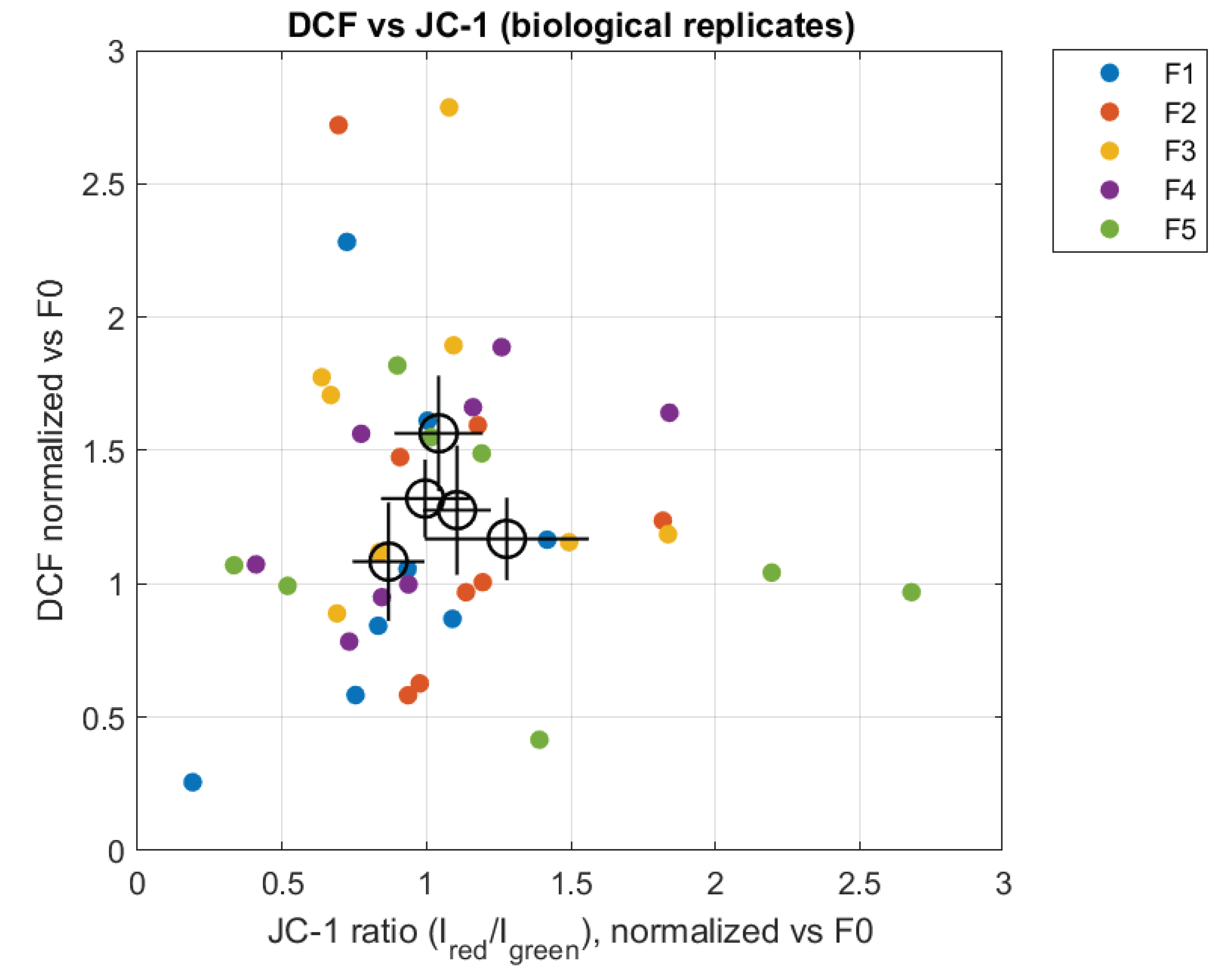
3.1. Data Quality, Replicates, and Normalization
3.2. Proliferation (Hoechst 33342)
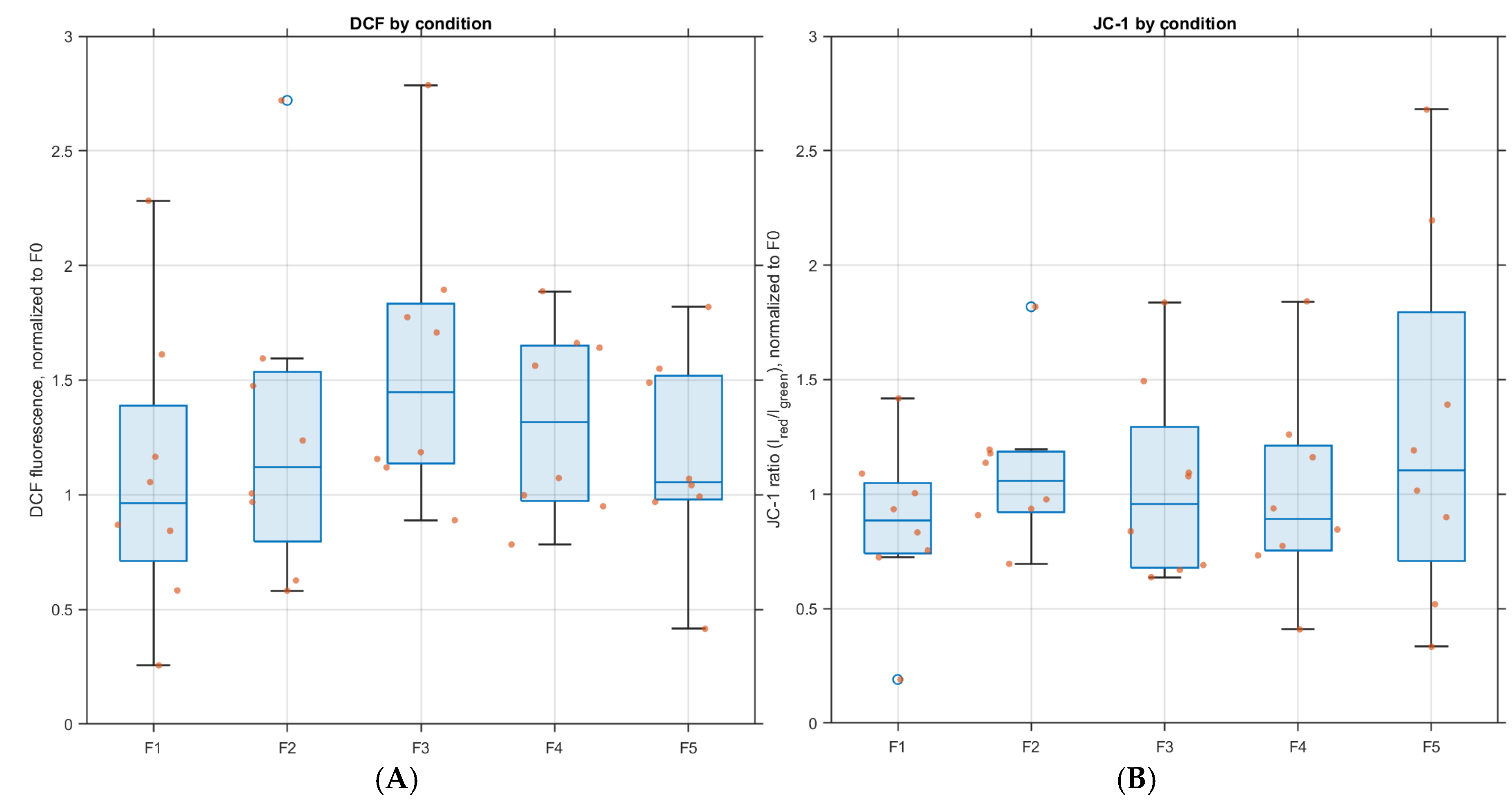
3.3. ROS (DCF)
3.4. Mitochondrial Membrane Potential (JC-1 Ratio)
3.5. Nitric Oxide (DAF-FM)
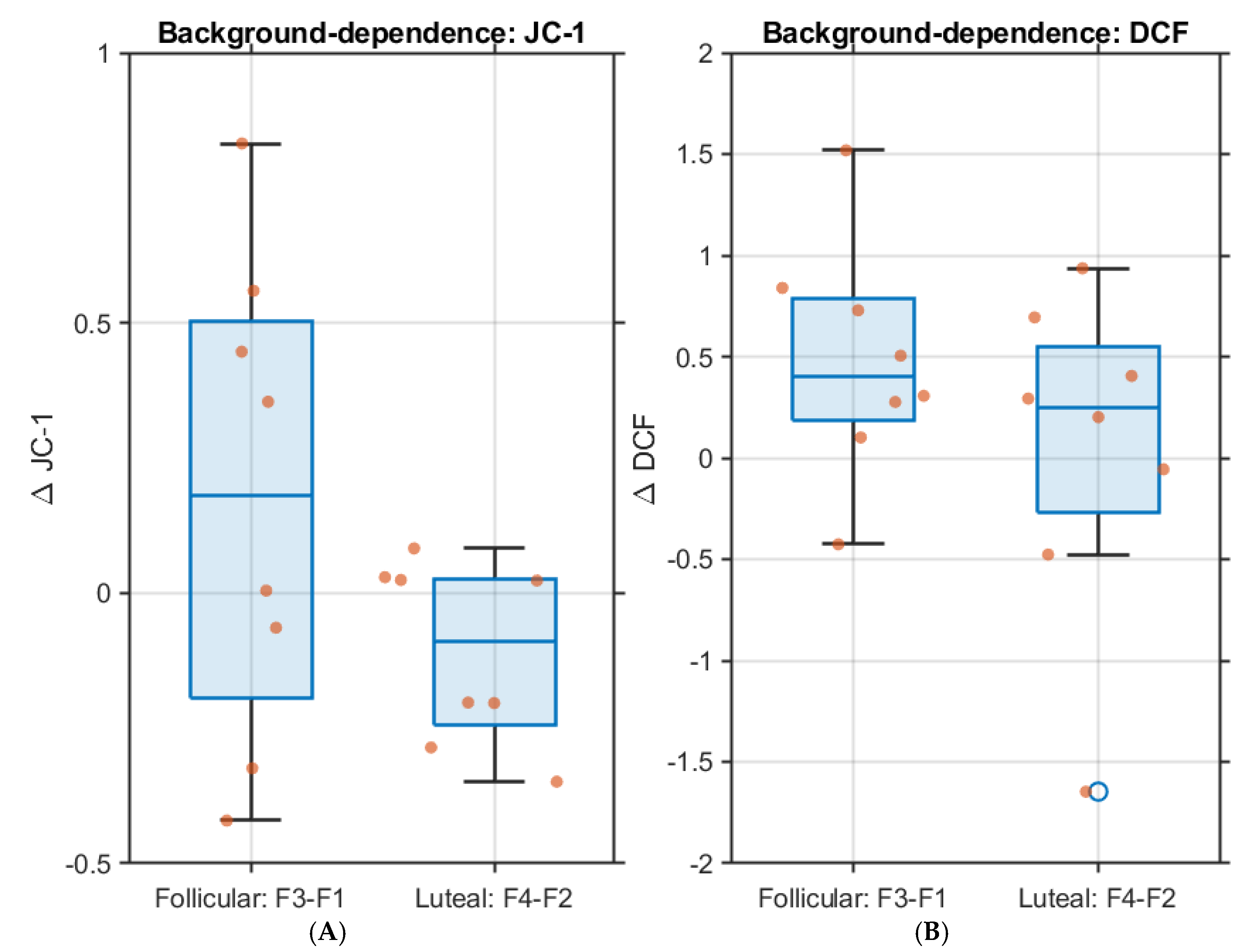
3.6. Background-Dependence (Follicular vs Luteal Milieus)
- ROS (DCF): the largest ROS rise occurred on the follicular-like background (F3 > F4).
- ΔΨm (JC-1): the strongest depolarization occurred on the luteal-like background (F4 > F3).
- A simple interaction contrast (F4–F2) vs (F3–F1) was directional for both endpoints, indicating that phase-like hormonal context primes mitochondrial responses to COC steroids at 48 h (Figure 2 A and B).
3.7. Sensitivity Analyses and Controls
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Klinge, C.M. Estrogenic control of mitochondrial function. Redox Biol 2020, 31, 101435. [Google Scholar] [CrossRef]
- Brinton, R.D. The healthy cell bias of estrogen action: mitochondrial bioenergetics and neurological implications. Trends Neurosci 2008, 31, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Guennoun, R. Progesterone in the Brain: Hormone, Neurosteroid and Neuroprotectant. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Bassani, T.B.; Bartolomeo, C.S.; Oliveira, R.B.; Ureshino, R.P. Progestogen-Mediated Neuroprotection in Central Nervous System Disorders. Neuroendocrinology 2023, 113, 14–35. [Google Scholar] [CrossRef] [PubMed]
- Finco, A.; Belcaro, G.; Cesarone, M.R. Evaluation of oxidative stress after treatment with low estrogen contraceptive either alone or associated with specific antioxidant therapy. Contraception 2012, 85, 503–508. [Google Scholar] [CrossRef]
- Kowalska, K.; Milnerowicz, H. Pro/antioxidant status in young healthy women using oral contraceptives. Environ Toxicol Pharmacol 2016, 43, 1–6. [Google Scholar] [CrossRef]
- Quinn, K.M.; Roberts, L.; Cox, A.J.; Borg, D.N.; Pennell, E.N.; McKeating, D.R.; Fisher, J.J.; Perkins, A.V.; Minahan, C. Blood oxidative stress biomarkers in women: influence of oral contraception, exercise, and N-acetylcysteine. Eur J Appl Physiol 2022, 122, 1949–1964. [Google Scholar] [CrossRef]
- Xicoy, H.; Wieringa, B.; Martens, G.J. The SH-SY5Y cell line in Parkinson's disease research: a systematic review. Mol Neurodegener 2017, 12, 10. [Google Scholar] [CrossRef]
- Gustafsson, H.; Runesson, J.; Lundqvist, J.; Lindegren, H.; Axelsson, V.; Forsby, A. Neurofunctional endpoints assessed in human neuroblastoma SH-SY5Y cells for estimation of acute systemic toxicity. Toxicol Appl Pharmacol 2010, 245, 191–202. [Google Scholar] [CrossRef]
- Sivandzade, F.; Bhalerao, A.; Cucullo, L. Analysis of the Mitochondrial Membrane Potential Using the Cationic JC-1 Dye as a Sensitive Fluorescent Probe. Bio Protoc 2019, 9. [Google Scholar] [CrossRef]
- Kalyanaraman, B.; Darley-Usmar, V.; Davies, K.J.; Dennery, P.A.; Forman, H.J.; Grisham, M.B.; Mann, G.E.; Moore, K.; Roberts, L.J.; Ischiropoulos, H. Measuring reactive oxygen and nitrogen species with fluorescent probes: challenges and limitations. Free Radic Biol Med 2012, 52, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P.; Bayir, H.; Belousov, V.; Chang, C.J.; Davies, K.J.A.; Davies, M.J.; Dick, T.P.; Finkel, T.; Forman, H.J.; Janssen-Heininger, Y.; et al. Guidelines for measuring reactive oxygen species and oxidative damage in cells and in vivo. Nat Metab 2022, 4, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci Rep 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed]
- Humphries, M.P.; Maxwell, P.; Salto-Tellez, M. QuPath: The global impact of an open source digital pathology system. Comput Struct Biotechnol J 2021, 19, 852–859. [Google Scholar] [CrossRef]
- Siegfried, L.G.; Bilik, S.M.; Burgess, J.L.; Catanuto, P.; Jozic, I.; Pastar, I.; Stone, R.C.; Tomic-Canic, M. An Optimized and Advanced Algorithm for the Quantification of Immunohistochemical Biomarkers in Keratinocytes. JID Innov 2024, 4, 100270. [Google Scholar] [CrossRef]
- Anckaert, E.; Jank, A.; Petzold, J.; Rohsmann, F.; Paris, R.; Renggli, M.; Schönfeld, K.; Schiettecatte, J.; Kriner, M. Extensive monitoring of the natural menstrual cycle using the serum biomarkers estradiol, luteinizing hormone and progesterone. Pract Lab Med 2021, 25, e00211. [Google Scholar] [CrossRef]
- Usala, S.J.; Vineyard, D.D.; Kastis, M.; Trindade, A.A.; Gill, H.S. Comparison of Day-Specific Serum LH, Estradiol, and Progesterone with Mira. Medicina (Kaunas) 2024, 60. [Google Scholar] [CrossRef]
- Huysseune, A.; Larsen, U.G.; Larionova, D.; Matthiesen, C.L.; Petersen, S.V.; Muller, M.; Witten, P.E. Bone Formation in Zebrafish: The Significance of DAF-FM DA Staining for Nitric Oxide Detection. Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Namin, S.M.; Nofallah, S.; Joshi, M.S.; Kavallieratos, K.; Tsoukias, N.M. Kinetic analysis of DAF-FM activation by NO: toward calibration of a NO-sensitive fluorescent dye. Nitric Oxide 2013, 28, 39–46. [Google Scholar] [CrossRef]
- Sacks, D.; Baxter, B.; Campbell, B.C.V.; Carpenter, J.S.; Cognard, C.; Dippel, D.; Eesa, M.; Fischer, U.; Hausegger, K.; Hirsch, J.A.; et al. Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. Int J Stroke 2018, 13, 612–632. [Google Scholar] [CrossRef]
- Gaignard, P.; Liere, P.; Thérond, P.; Schumacher, M.; Slama, A.; Guennoun, R. Role of Sex Hormones on Brain Mitochondrial Function, with Special Reference to Aging and Neurodegenerative Diseases. Front Aging Neurosci 2017, 9, 406. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Luo, M.; Wang, R.; Ye, J.; Wang, X. Mitochondria in Sex Hormone-Induced Disorder of Energy Metabolism in Males and Females. Front Endocrinol (Lausanne) 2021, 12, 749451. [Google Scholar] [CrossRef]
- Chang, N.S.; Schultz, L.; Hsu, L.J.; Lewis, J.; Su, M.; Sze, C.I. 17beta-Estradiol upregulates and activates WOX1/WWOXv1 and WOX2/WWOXv2 in vitro: potential role in cancerous progression of breast and prostate to a premetastatic state in vivo. Oncogene 2005, 24, 714–723. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Shu, C.; Chen, L.; Yao, B. Impact of sex, body mass index and initial pathologic diagnosis age on the incidence and prognosis of different types of cancer. Oncol Rep 2018, 40, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Liu, Y.H. Sex difference, proteostasis and mitochondrial function impact stroke-related sarcopenia-A systematic review and meta-analysis. Ageing Res Rev 2024, 101, 102484. [Google Scholar] [CrossRef]
- Huang, Y.; Li, H.; Liang, R.; Chen, J.; Tang, Q. The influence of sex-specific factors on biological transformations and health outcomes in aging processes. Biogerontology 2024, 25, 775–791. [Google Scholar] [CrossRef]
- Huang, J.; Liu, Y.; Zou, L.; Zhu, C.; Xia, W. Ameliorative effects of Gly[14]-humanin on cyclophosphamide-induced premature ovarian insufficiency and underlying mechanisms. Reprod Biomed Online 2025, 51, 104901. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, C.; Cui, H.; Sun, G.; Qi, X.; Yao, X. Mitochondrial dysfunction in age-related sarcopenia: mechanistic insights, diagnostic advances, and therapeutic prospects. Front Cell Dev Biol 2025, 13, 1590524. [Google Scholar] [CrossRef]
- de Assis, G.G.; de Sousa, M.B.C.; Murawska-Ciałowicz, E. Sex Steroids and Brain-Derived Neurotrophic Factor Interactions in the Nervous System: A Comprehensive Review of Scientific Data. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef]
- Traiffort, E.; Kassoussi, A.; Zahaf, A. Revisiting the role of sexual hormones in the demyelinated central nervous system. Front Neuroendocrinol 2025, 76, 101172. [Google Scholar] [CrossRef]
- Prossnitz, E.R.; Barton, M. The G protein-coupled oestrogen receptor GPER in health and disease: an update. Nat Rev Endocrinol 2023, 19, 407–424. [Google Scholar] [CrossRef]
- Głombik, K.; Detka, J.; Budziszewska, B. Hormonal Regulation of Oxidative Phosphorylation in the Brain in Health and Disease. Cells 2021, 10. [Google Scholar] [CrossRef]
- Shaw, G.A. Mitochondria as the target for disease related hormonal dysregulation. Brain Behav Immun Health 2021, 18, 100350. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




