1. Introduction
Lymphatic filariasis (LF) is a mosquito-borne neglected tropical disease (NTD) caused by three species of filarial worms:
Wuchereria bancrofti, Brugia malayi, and
B. timori, and transmitted to humans by mosquito vectors, including Aedes, Culex, and Anopheles species [
1]. Globally,
W. bancrofti accounts for about 90% of infections [
2]. Adult worms live in the lymphatic system, where untreated infections can lead to progressive and irreversible lymphatic damage. The clinical consequences, such as severe lymphoedema, skin thickening and hydrocele, are associated with pain, disability, and significant social and economic exclusion [
3,
4].
In 2018, an estimated 51 million people were living with LF, mainly in Africa and South Asia. This indicates a 74% reduction in prevalence since the World Health Organization’s (WHO) Global Programme to Eliminate Lymphatic Filariasis (GPELF) was launched in 2000 [
5]. The decline is widely attributed to sustained national efforts to interrupt transmission and reduce disease burden through mass drug administration (MDA) and morbidity management [
6]. Nevertheless, despite this notable progress, the remaining burden and the risk of resurgence in settings that have eliminated LF call for renewed focus on surveillance and long-term programme sustainability [
7,
8].
WHO has established a formal validation process for countries that meet LF elimination thresholds. The 2011 guideline criteria (used to date but updated in 2025 [yet to be applied]) defined the LF elimination threshold as <2% Ag prevalence in areas in which
W. bancrofti is endemic and
Anopheles or
Culex is the vector, <1% Ag prevalence in regions in which W. bancrofti is endemic and
Aedes is the primary vector, and <2% antibody (Ab) prevalence in areas were Brugia spp. are endemic [
9,
10]. To improve sensitivity, the 2025 LF surveillance guidelines [
10] suggest that <1%
Ag (
W. bancrofti) and
Ab (
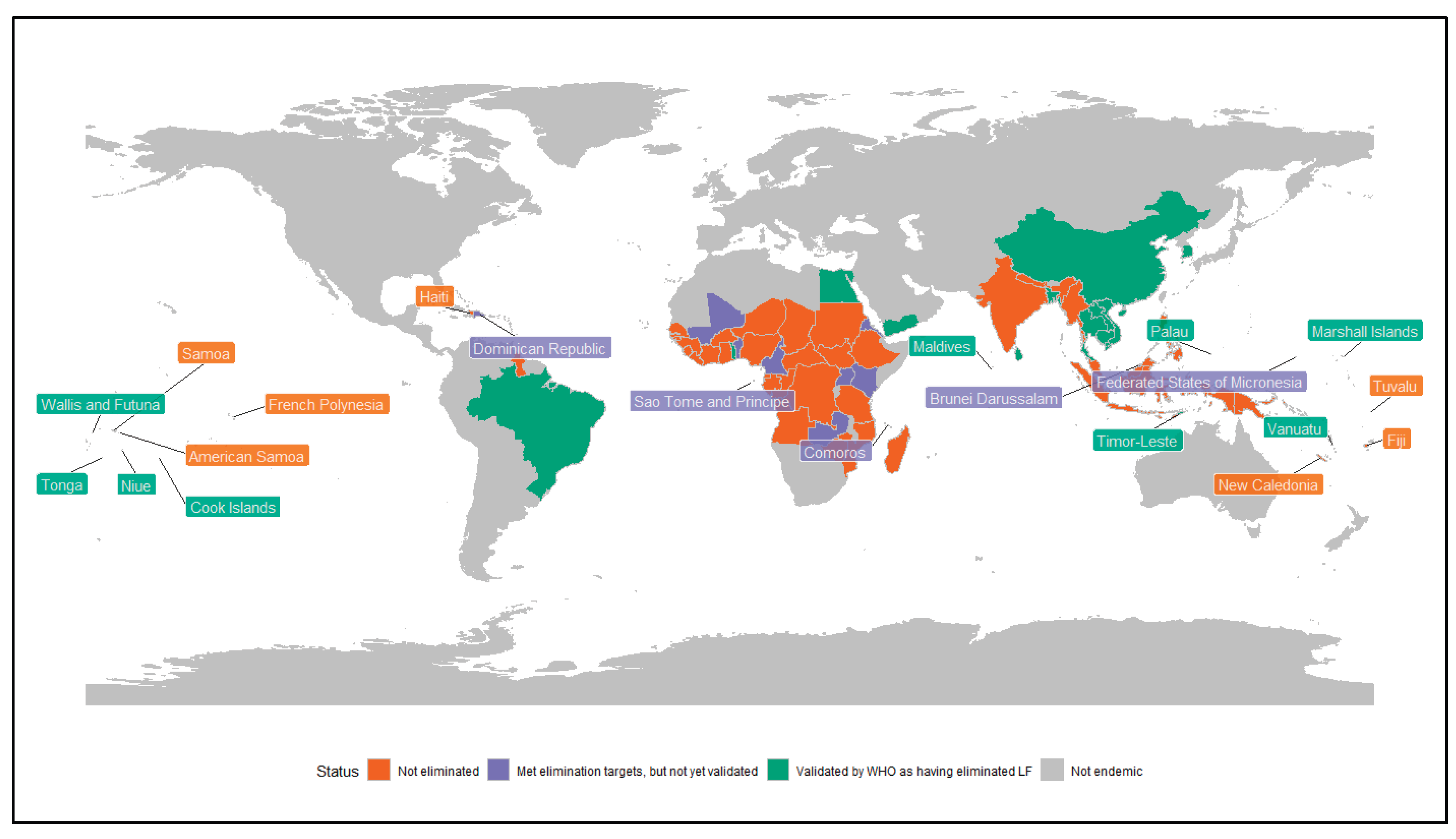
Brugia spp.) prevalence target threshold for all vector and parasite species. Countries seeking validation of having eliminated the disease as a public health problem submit a dossier of evidence to WHO, including results of epidemiological surveys, plans for morbidity management, and a commitment to implement post-validation surveillance (PVS). As of September 2025, 21 (of 72) LF-endemic countries have received WHO validation for elimination as a public health problem, with a further 14 having completed mass drug administration (MDA) and are under surveillance but have not yet been validated. 38 other LF endemic countries continue to implement MDA to reduce the prevalence of the disease, and one has yet to start MDA [
11] (
Figure 1).
Sixteen of the 22 Pacific Island Countries and Territories (PICTs) have reported LF transmission. Among these, eight (Cook Islands, Kiribati, Niue, the Republic of the Marshall Islands, Palau, Tonga, Vanuatu, and Wallis and Futuna) have gained WHO validation for eliminating LF as a public health problem. Although each of these countries submitted PVS plans as part of the validation process, their implementation remains limited. The reasons behind this inaction are not well understood; without urgent efforts to close this knowledge gap, creating demand for PVS will be challenging, risking decades of elimination work at risk.
In 2025, the WHO issued new guidelines for the design and implementation of LF surveillance [
10]. These guidelines mark a significant shift towards clearer global expectations for the surveillance activities that countries working towards sustained LF elimination should implement. This paper directly responds to this development. It aims to identify the core barriers to PVS implementation in the PICT context and to gather implementer-led solutions that can drive policy and operational changes. The insights here are grounded in the perspectives of national program managers and field staff. They are relevant not only to the Pacific but also to countries currently implementing or preparing to implement PVS. Building on our earlier conceptual work, [
12] this study offers practice-oriented evidence to inform the next phase of global LF elimination efforts.
2. Materials and Methods
Study Design and Research Questions
This study used two complementary qualitative methods to examine the views of national program managers and frontline staff on the systemic barriers and enablers to implement PVS for LF in PICTs. The methods included a Nominal Group Technique (NGT)-based consultation in 2024, and an expert workshop held in 2025.
Nominal Group Technique
A range of consensus-based and participatory methods exist to elicit stakeholder priorities, including surveys, focus groups, citizen juries, discrete choice experiments, and Delphi techniques [
13,
14]. Among these, consensus group methods are especially valuable when empirical evidence is limited or contested. Widely used across sectors such as education, engineering, management, and health, these methods have informed the development of guidelines, diagnostic criteria, and program planning [
15,
16]. The NGT, developed by Delbecq and Van de Ven in 1971 [
17], is a structured approach designed to facilitate idea generation, discussion, and prioritisation. NGT enables comparison of priorities across individuals and groups, highlighting areas of agreement and divergence. By design, the NGT promotes equal participation and minimises the influence of dominant individuals, thereby reducing measurement bias [
16,
18].
The NGT followed seven sequential steps, described in detail in a companion methods paper [
19], and summarised here: (i) presentation of the nominal question; (ii) silent generation of individual ideas; (iii) round-robin generation of ideas; (iv) group discussion to clarify and elaborate; (v) individual scoring and rank of ideas; (vi) aggregation and presentation of collective rankings; (vii) open discussion to reflect on the results.
The NGT was held during the 2024 ‘Coalition for Operational Research on Neglected Tropical Diseases (COR-NTD) Pacific Islands’ meeting held in Brisbane, Australia in 25-26 September 2024. The nominal question, developed by two authors (AC and HL) following guidance from Mullen et al [
20], was, ‘What do you consider to be the most significant challenges you face, or anticipate facing, in implementing PVS for LF in your country or territory?’ For the scoring step (step 5), participants were allocated ten votes to distribute across identified challenges as they saw fit. Votes could be concentrated on a single issue or spread across multiple priorities. All notes and individual scoring sheets were collected and transcribed. Facilitators kept detailed records of the discussion, capturing participants’ views, insights, and supporting arguments.
Expert Workshop
A follow-up expert workshop was held during the ‘Voices and Visions: Building Partnerships for Integrated Surveillance of Lymphatic Filariasis and Other Infectious Diseases in the Pacific Islands’ meeting in Brisbane, Australia, from 8-10 July 2025. The workshop gathered 50 participants, including 15 from 14 PICTs and Timor-Leste. The workshop focused on advancing the implementation of PVS in the region. It featured themed discussions on aligning LF surveillance with PICTs’ context, operationalising LF PVS, and responding to PVS-generated surveillance signals. Sessions were audio-recorded and transcribed for later analysis.
Data Analysis
Quantitative ranking data generated during the NGT were summarised descriptively. Qualitative data from both the NGT and the expert workshop were analysed using thematic analysis with an inductive approach [
21]. Transcripts and facilitator notes were reviewed to identify recurring concepts and patterns in participants’ contributions. Themes emerging from the NGT were compared with those from the expert workshop to explore areas of convergence and divergence across the two data sources.
Results
Participant Characteristics
Eight individuals (one female and seven males) from six PICTs participated in the NGT, and 12 individuals (seven females and five males) from nine PICTs and Timor-Leste took part in the expert workshop. Three people were involved in both the NGT and the expert workshop. Collectively, participants had a range of expertise, including medical practice, nursing, epidemiology, surveillance, environmental health, program management, and academia.
Identification and Prioritisation of Challenges
The nominal question sought to identify the most significant challenges in implementing PVS of LF in the Pacific Island context. Participants identified 70 distinct obstacles, highlighting the breadth and complexity of challenges faced in PVS implementation across the region.
The 70 challenges identified were condensed into ten broad themes.
Table 1 presents these themes and the participants’ voting results. The ranking exercise demonstrated a strong consensus in opinions. It is important to note that all participants ranked the top three themes within their highest tier. The main challenges reported in implementing PVS were a lack of understanding or commitment to PVS among national leaders, the irregular nature of technical and financial support for PVS activities, and the absence of clear guidance for selecting and applying epidemiologically and contextually suitable PVS strategies. These and other themes are explored below.
National Leadership Understanding and Commitment to PVS
Participants in the NGT were unanimous in identifying limited awareness and engagement among national leaders as the most pressing barrier to PVS implementation; this was reiterated during the expert workshop discussion. The importance of this issue was underscored by the fact that it received more than twice as many votes as the next-highest-ranked challenge during the NGT and was a recurring theme throughout the workshop. Several participants explained that while technical discussions around prevalence thresholds, transmission modelling, and cost-effectiveness are essential, they are unlikely to result in meaningful action unless senior officials, including ministers, permanent secretaries, and health directors, understand the ongoing risk of LF resurgence and the rationale for continued surveillance.
A common theme in the discussions was that the term “elimination” is frequently misunderstood by those not intimately involved in LF work, leading to the interpretation that LF is no longer a threat. This misconception, often equating elimination with eradication, was cited as a key reason why PVS does not receive political or budgetary attention. In some contexts, the problem is compounded by translation challenges. Some participants noted that in their languages there is no clear distinction between the terms “eliminate” and “eradicate,” making effective communication and advocacy even more difficult.
Discussions during the NGT and subsequent expert workshop raised the possibility that international partners may need to adjust their support strategies. Rather than focusing on technical implementation or field activities, participants suggested the need for evidence that directly addresses policymakers’ information needs and presents an economic argument for investment in LF PVS. Participants suggested that to get ‘traction’ on LF PVS it needs to be raised and endorsed by the regional health governance bodies: namely, at the Pacific Public Health Surveillance Network (PPHSN) and Pacific Heads of Health meetings. Given their secretariat functions, meeting participants felt the WHO and Pacific Community (SPC) are well-positioned to advocate for this.
Resource Mobilisation for PVS
Participants identified resource mobilisation as the second most critical barrier to effective PVS implementation. They described a noticeable shift from routine activities and funding under the Pacific Program to Eliminate LF (PacELF) toward sporadic, externally driven, short-term projects. This change was interpreted as a decline in donor (and perhaps national leaders) interest in LF after countries achieve elimination targets.
Several participants emphasised that, without predictable financing, it was challenging to advocate and embed PVS activities within broader health system planning. Instead, it was said that programs were often left reliant on externally funded research projects that tended to be short-term and piecemeal in approach. As one participant explained, “Without reliable domestic or partner financing, PVS cannot be planned and hence stops.” Another noted that this situation risks making PVS a reactive or opportunistic activity, rather than a routine and integrated part of public health systems.
Participants made clear that resourcing extends beyond money alone. It includes access to diagnostic kits, the availability of fieldwork equipment, including vehicles for transportation, and technical advice. What is most needed, they said, is predictability and stability to allow national health programs to organise and implement LF PVS.
In response to these concerns, participants proposed exploring ways to align PVS with established, well-funded and complementary programs, such as vaccination programs, and concepts, such as health systems strengthening and universal health coverage. Support from technical partners in developing cost-effectiveness arguments and investment cases was viewed as beneficial in encouraging both domestic and international donor financing.
Access to Context-Appropriate Guidance for PVS Implementation
Participants consistently emphasised that practical context-relevant guidance to support the implementation of PVS was critical. While global frameworks and recommendations exist, such as those from WHO, these were viewed as too general and not sufficiently aligned to the realities of small island settings. Specifically, participants noted gaps in advice related to designing and selecting surveillance strategies, how to advocate (domestically and internationally) for LF resourcing effectively, and determining triggers for public health action. In the absence of such guidance, participants expressed reluctance to advocate for PVS within their health systems. As one participant put it, “We don’t have anything to back up our requests… we don’t know if what we think is the best thing to do, and there is no one who can advise.” This uncertainty is reported to have contributed to program managers feeling under-supported in making technical decisions.
Participants at the 2025 expert workshop, where the new WHO guidelines for LF PVS [
10] were introduced, acknowledged that the document addressed some previously identified needs. However, echoing the 2024 NGT findings, they emphasised the importance of guidance that reflects the practical realities of Pacific Island contexts. They recommended developing Pacific-specific tools, training materials, and case studies from other small island settings to translate global recommendations into more feasible strategies and to use these to advocate (domestically and with development partners and donors) for increased financial and technical support.
National Capacity for PVS Implementation
Participants described a reliance on external agencies to fund and implement PVS, contrasting this with earlier successes under the Pacific Programme for the Elimination of Lymphatic Filariasis (PacELF) program [
22] where strong national leadership and sustained engagement contributed to the effective State-led implementation of LF elimination activities. The participants noted that the PacELF model of ownership should be extended to PVS; however, reduced funding, shifting leadership priorities, and competing demands across the health system have made it difficult to maintain domestic momentum, leading to greater reliance on external agencies.
To address these challenges, participants called for investment in national workforce development. Participants recommended that capacity-building be long-term and embedded within national systems, rather than delivered through one-off external workshops. Approaches such as mentoring, mirroring, and South-South exchange, supported by regional partners, were seen as helpful in building practical, applied expertise. Development partners were encouraged to continue their support, but in ways that support capacity building and the transition to country-led implementation.
Mandates for PVS Implementation
Participants highlighted the lack of formal mandates or strict accountability measures as reasons for the deprioritisation of PVS within national health systems. It was noted that “without clearly defined requirements, LF PVS activities are seen as optional, and often overlooked in favour of other urgent health issues.” This observation echoes lessons from other disease elimination programs, where activities cease once formal monitoring is reduced.
To strengthen accountability, participants recommended that the need for PVS be more clearly articulated and that compliance monitoring and evaluation be integrated into routine health system indicator sets, with results reported in public health service delivery reports. Participants suggest that development partners, such as WHO, play an important advocacy role by encouraging and supporting countries to include PVS as part of health service performance monitoring frameworks. Some also propose that countries consider codifying PVS-related responsibilities in national legislation or strategic plans, which could help secure long-term political and financial support. Others suggested adding LF as a national notifiable disease to elevate its importance and to impose a legal obligation to take public health action upon receipt of a surveillance signal.
Discussion
This study provides insights into the practical and systemic challenges of implementing LF PVS in PICTs. By combining structured prioritisation through an NGT with broader perspectives from an expert workshop, we identified not only recurring barriers but also strategies to improve the feasibility, sustainability, and acceptability of PVS.
A consistent theme across both engagements was that the success or failure of PVS cannot be reduced to a single issue. Instead, implementation reflects the interplay of context-specific, structural, and political influences. This complexity necessitates moving beyond simple linear problem–solution frameworks toward more systems-based approaches and highlights the need for settings-specific design of PVS strategies, a point reflected in the new (2025) second edition of the WHO Monitoring and epidemiological assessment of mass drug administration in the GPELF manual [
10]. Developing user-friendly tools and operational guidance to support context-sensitive planning, such as decision trees or costing calculators, could enable program managers to design appropriate PVS strategies.
Both the NGT and workshop underscored the misalignment between global expectations for sustained PVS, and the operational realities faced by PICTs. Despite strong encouragement, PVS implementation has often been ad hoc, constrained by resource scarcity, geographic dispersion, and the absence of formal mandates or dedicated funding. Countries that achieve elimination frequently experience a paradox: success is followed not by sustained investment, but by declining political attention. Raising awareness among senior decision-makers of the risks of LF resurgence is therefore critical. Suggested strategies include high-level advocacy that situates PVS within the broader agenda of regional health security, public recognition of national leadership, and research that directly addresses decision-makers’ priorities, including cost and budget-impact analyses that clarify the health system implications of conducting, or not conducting, PVS.
At the global and regional level, aligning LF PVS with frameworks such as the WHO NTD Roadmap [
25], WHO Guidelines on Verification of Measles Elimination in the Western Pacific Region [
26], and embedding advocacy and regional planning for PVS implementation within the established governance architecture of the Pacific through the Pacific Public Health Surveillance Network [
27] will likely foster commitment, sustainability, and accountability. Workshop discussions also highlighted that the release of the new WHO guidelines [
28], although welcomed, will require accompanying context-relevant guidance and technical assistance in order for them to be adopted as intended. Co-development of PICT-specific supporting material and support from advisors who “understand the Pacific context” was called for.
Findings from both engagements emphasised that the absence of national or regional mandates has left PVS vulnerable to donor cycles, individual champions, or ad hoc opportunities. Embedding surveillance within established frameworks, such as the Pacific Healthy Islands Monitoring Framework [
29], or re-envisioning the successful PacELF model to suit current epidemiological realities could create programmatic continuity and attract donor support.
Integration emerged as a pragmatic approach in settings facing financial and logistical constraints. Embedding PVS into established service platforms, such as NCD screening, workplace health checks, or maternal health services, can reduce marginal costs and increase acceptability. Recent Pacific examples demonstrate feasibility and offer models for replication.
Community engagement remains a pivotal factor. Misunderstandings among leaders and communities regarding the distinction between elimination and eradication, coupled with survey fatigue, were identified as barriers. Conversely, leveraging trusted networks, such as churches, offers opportunities to enhance participation. Importantly, community engagement should be positioned as a central design feature of PVS, with risk communication that emphasises the value of surveillance rather than relying on fear-based messaging.
Operational bottlenecks, such as lengthy procurement processes, limited forecasting, and stock shortages, were also identified. These challenges are solvable through systematic planning and pooled procurement mechanisms, supported by WHO and partners.
Conclusion
The small population sizes of PICTs present both challenges and opportunities. Regional collaboration offers a means to address systemic barriers, pool resources, and sustain LF elimination. A programmatic platform for LF PVS across the region, supported by national authorities, donors, and academia, could strengthen efficiency and impact. Lessons from early PICT adopters, such as Tonga, Wallis and Futuna, and Niue, should inform broader practice across the islands. Such evidence is likely relevant to those seeking solutions to PVS implementation challenges globally.
Author Contributions
Conceptualisation, ATC, CLL, and HLSL; methodology, ATC and HLSL; validation, all authors; formal analysis, ATC.; resources, ATC and CLL; data curation, ATC.; writing—original draft preparation, ATC; writing—review and editing, all authors; visualization, ATC; supervision, CLL.; project administration, ATC; funding acquisition, CLL. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Operational Research and Decision Support for Infectious Diseases (ODeSI) program, which is funded by The University of Queensland’s Health Research Accelerator (HERA) initiative (2021-2028).
Institutional Review Board Statement
The study was approved by The University of Queensland’s Human Research Ethics Committee (Project #2024/HE000224).
Data Availability Statement
Data are available from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| GPELF |
Global Programme to Eliminate Lymphatic Filariasis |
| NGT |
Nominal Group Technique |
| NTD |
Neglected-tropical disease |
| LF |
Lymphatic filariasis |
| MDA |
Mass drug administration |
| NCD |
Non-communicable disease |
| PacELF |
Pacific Programme to Eliminate Lymphatic Filariasis |
| PICTs |
Pacific Island Countries and Territories |
| PPHSN |
Pacific Public Health Surveillance Network |
| PVS |
Post-validation surveillance |
| SPC |
[Secretariat of the] Pacific Community |
| STEPs and STEPwise |
“STEPs” and “STEPwise” refers to the step-by-step structure of the WHO NCD risk factor survey methodology rather than being an acronym |
| WHO |
World Health Organization |
References
- de Souza, D.K.; Bockarie, M.J. Current perspectives in the epidemiology and control of lymphatic filariasis. Clinical Microbiology Reviews 2025, 38, e00126-23. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.A.; Jones, M.K.; McManus, D.P. The History of Bancroftian Lymphatic Filariasis in Australasia and Oceania: Is There a Threat of Re-Occurrence in Mainland Australia? Tropical Medicine and Infectious Disease 2018, 3, 58. [Google Scholar] [CrossRef] [PubMed]
- Hofstraat, K.; van Brakel, W.H. Social stigma towards neglected tropical diseases: a systematic review. Int Health 2016, 8, i53–i70. [Google Scholar] [CrossRef] [PubMed]
- Partono, F. The spectrum of disease in lymphatic filariasis. In Ciba Foundation Symposium 127-Filariasis: Filariasis: Ciba Foundation Symposium 127: 2007; Wiley Online Library, 2007; pp. 15–31. [Google Scholar]
- World Health Organization. Global programme to eliminate lymphatic filariasis: progress report, 2019. Weekly Epidemiological Record 2020, 95, 509–523. [Google Scholar]
- World Health Organization. Lymphatic filariasis: monitoring and epidemiological assessment of mass drug administration: a manual for national elimination programmes; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Couteaux, C.; Demaneuf, T.; Bien, L.; Munoz, M.; Worms, B.; Chésimar, S.; Takala, G.; Lie, A.; Jessop, V.; Selemago, M.K. Postelimination Cluster of Lymphatic Filariasis, Futuna, 2024. Emerging Infectious Diseases 2025, 31, 488. [Google Scholar] [CrossRef] [PubMed]
- Lawford, H.L.S.; Tukia, O.; Takai, J.; Sheridan, S.; Ward, S.; Jian, H.; Martin, B.M.; Ofanoa, R.; Lau, C.L. Persistent lymphatic filariasis transmission seven years after validation of elimination as a public health problem: a cross-sectional study in Tonga. The Lancet Regional Health – Western Pacific 2025, 57. [Google Scholar] [CrossRef] [PubMed]
- Collyer, B.S.; Irvine, M.A.; Hollingsworth, T.D.; Bradley, M.; Anderson, R.M. Defining a prevalence level to describe the elimination of Lymphatic Filariasis (LF) transmission and designing monitoring & evaluating (M&E) programmes post the cessation of mass drug administration (MDA). PLOS Neglected Tropical Diseases 2020, 14, e0008644. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Monitoring and epidemiological assessment of mass drug administration in the global programme to eliminate lymphatic fi lariasis: a manual for national elimination programmes, 2nd ed.; WHO: Geneva, 2025. [Google Scholar]
- World Health Organization. Global programme to eliminate lymphatic filariasis: progress report, 2023. Weekly Epidemiological Record 2024, 99, 565–576. [Google Scholar]
- Lawford, H.; Jian, H.; Tukia, O.; Takai, J.; Couteaux, C.; Thein, C.; Jetton, K.; Tabunga, T.; Bauro, T.; Nehemia, R.; Ave, C.; Mokoia, G.; Fetaui, P.; Taleo, F.; Udui, C.-A.; Lau, C.L.; Craig, A.T. Barriers and Facilitators to Implementing Post-Validation Surveillance of Lymphatic Filariasis in Pacific Island Countries and Territories: A Conceptual Framework Developed from Qualitative Data. 2025. [Google Scholar] [CrossRef]
- Grill, C. Involving stakeholders in research priority setting: a scoping review. Research Involvement and Engagement 2021, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Shelton, K.; Haynes, C.A.; Creghan, K.A. Fundamentals of Delphi research methodology. In Research Anthology on Innovative Research Methodologies and Utilization Across Multiple Disciplines; IGI Global Scientific Publishing, 2022; pp. 473–497. [Google Scholar]
- Murphy, M.; Black, N.; Lamping, D.; McKee, C.; Sanderson, C.; Askham, J.; Marteau, T. Consensus development methods, and their use in clinical guideline development. Health Technology Assessment 1998, 2, i-88. [Google Scholar] [CrossRef]
- Jones, J.; Hunter, D. Consensus methods for medical and health services research. BMJ 1995, 311, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Delbecq, A.L.; Van de Ven, A.H. A group process model for problem identification and program planning. The journal of applied behavioral science 1971, 7, 466–492. [Google Scholar] [CrossRef]
- Harvey, N.; Holmes, C.A. Nominal group technique: an effective method for obtaining group consensus. Int J Nurs Pract 2012, 18, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.T.; Lawford, H.L.S.; Viali, S.; Tuitama, G.; Lau, C.L. Perspectives on the implementation of post-validation surveillance for lymphatic filariasis in the Pacific Islands: A nominal group technique-based study protocol. PLoS One 2024, 19, e0313957. [Google Scholar] [CrossRef] [PubMed]
- Mullen, R.; Kydd, A.; Fleming, A.; McMillan, L. A practical guide to the systematic application of nominal group technique. Nurse Res 2021, 29, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Terry, G.; Hayfield, Nikki; Clarke, Victoria; Braun, Virginia. Thematic Analysis. In The SAGE Handbook of qualitative research in psychology; Willig, C.R.W., Ed.; SAGE Publications Ltd: London, 2017; pp. 17–36. [Google Scholar]
- Ichimori, K.; Graves, P.M. Overview of PacELF—the Pacific Programme for the Elimination of Lymphatic Filariasis. Tropical medicine and health 2017, 45, 34. [Google Scholar] [CrossRef] [PubMed]
- Craig, A.T.; Lawford, H.; Mokoia, G.; Ikimau, M.; Fetaui, P.; Marqardt, T.; Lau, C.L. Integrating post-validation surveillance of lymphatic filariasis with the WHO STEPwise approach to non-communicable disease risk factor surveillance in Niue, a study protocol. PLoS ONE 2025, 20, e0315625. [Google Scholar] [CrossRef] [PubMed]
- Lawford, H.; Tukia; Takai, J.; Sheridan, S.; Lau, C.L. Operational research to inform post-validation surveillance of lymphatic filariasis in Tonga study protocol: History of lymphatic filariasis elimination, rational, objectives, and design. PLoS ONE 2024, 19, e0307331. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Ending the neglect to attain the Sustainable Development Goals: a rationale for continued investment in tackling neglected tropical diseases 2021–2030; WHO: Geneva, Switzerland, 2022. [Google Scholar]
- World Health Organization. Guidelines on verification of measles elimination in the Western Pacific Region, 2nd ed.; WHO: Manila, Philippines, 2020. [Google Scholar]
- Sheel, M.; Tinessia, A.; Hall, J.; Abdi, I.; Craig, A.; Zimmerman, P-A.; Rockett, R.; Nadeeka, S. Review of the Pacific Public Health Surveillance Network; SPC: Neumea, New Caledonia, 2023. [Google Scholar]
- World Health Organization. Monitoring and epidemiological assessment of mass drug administration in the global programme to eliminate lymphatic filariasis: a manual for national elimination programmes, second edition, 2nd ed.; WHO: Geneva, Switzerland, 2025. [Google Scholar]
- World Health Organization. Monitoring progress towards the vision of healthy islands in the Pacific: second progress report 2019; WHO: Manila, Philippines, 2020. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |