Submitted:
07 January 2026
Posted:
08 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Patients
Response Definitions
Statistical Analysis
Results
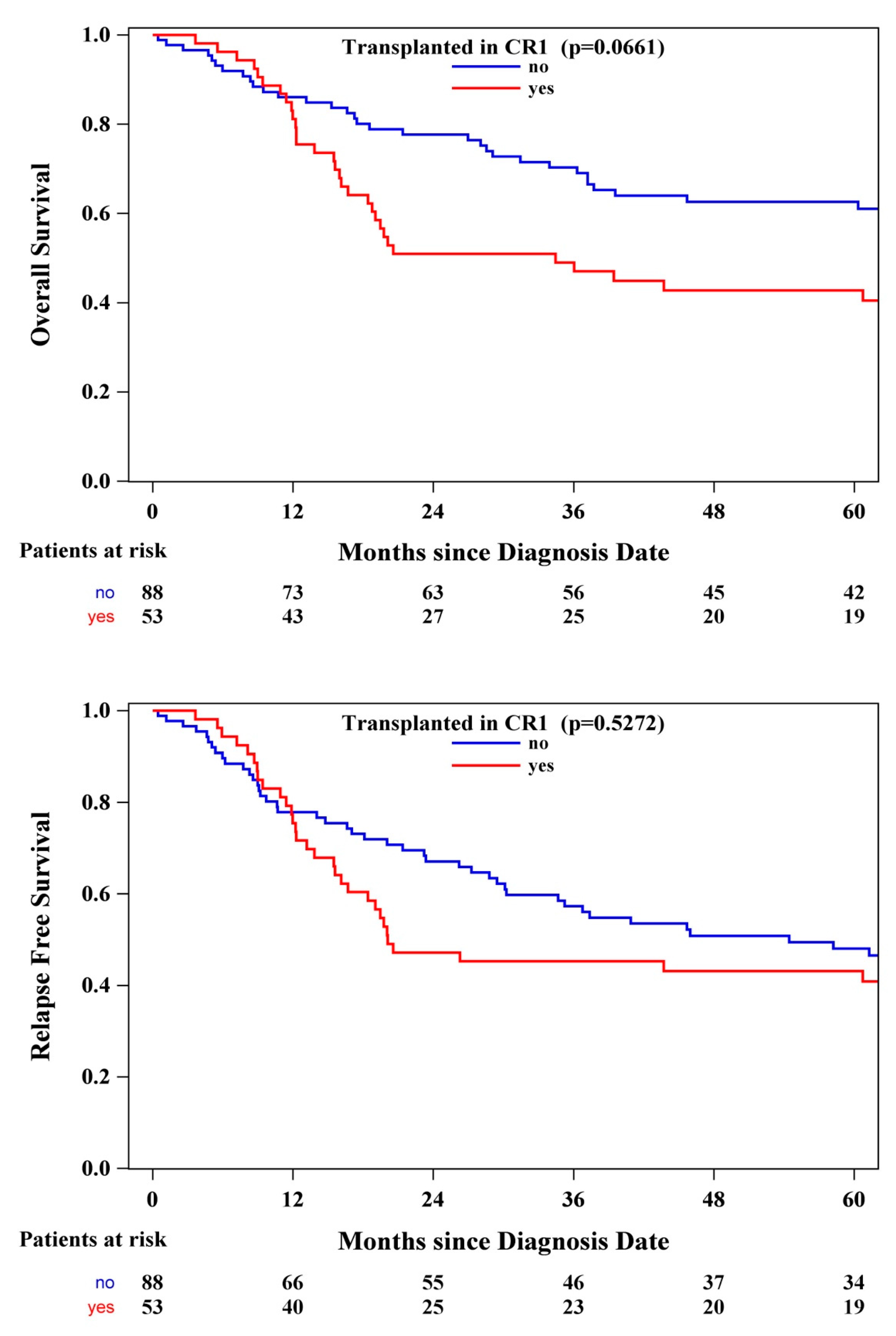
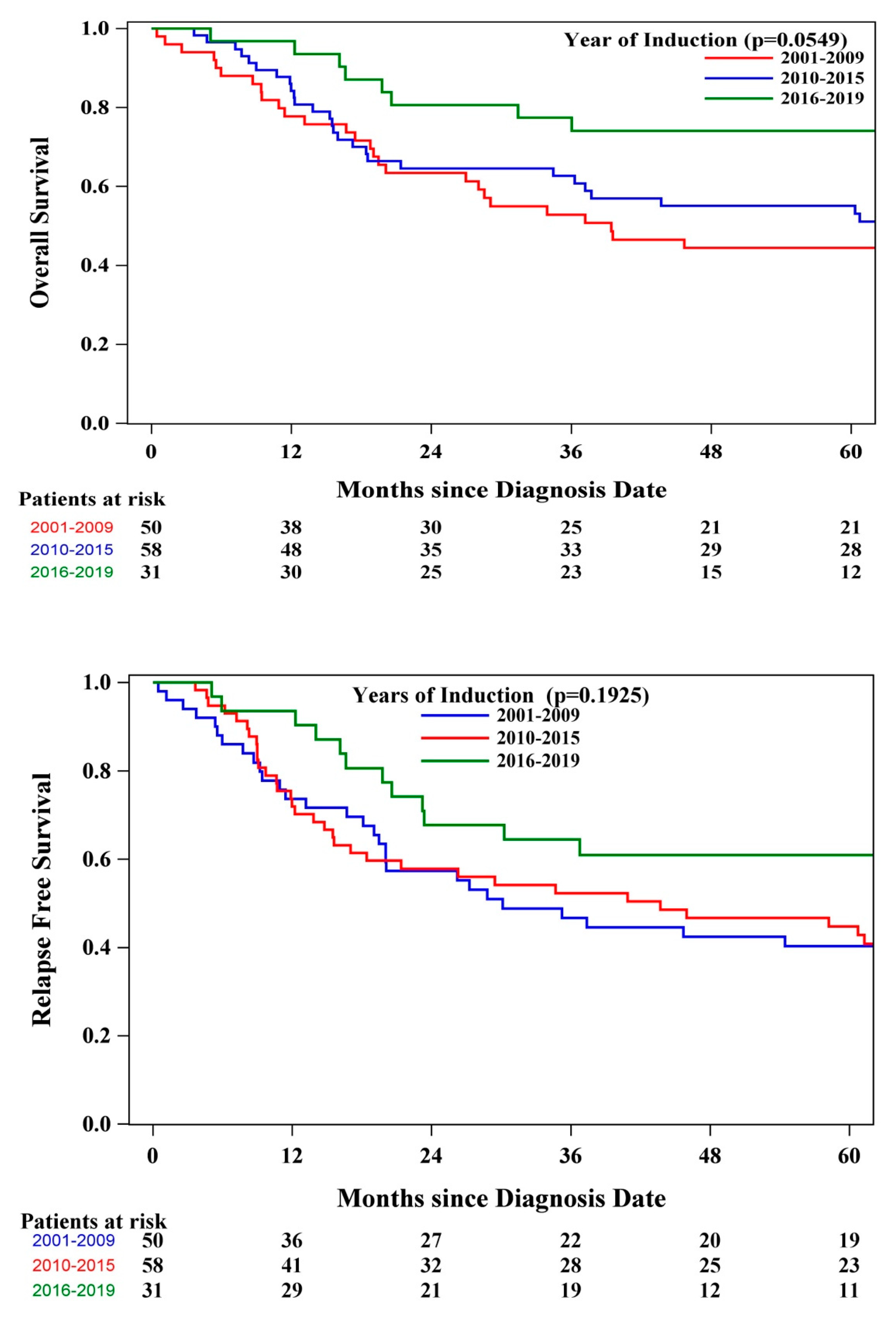
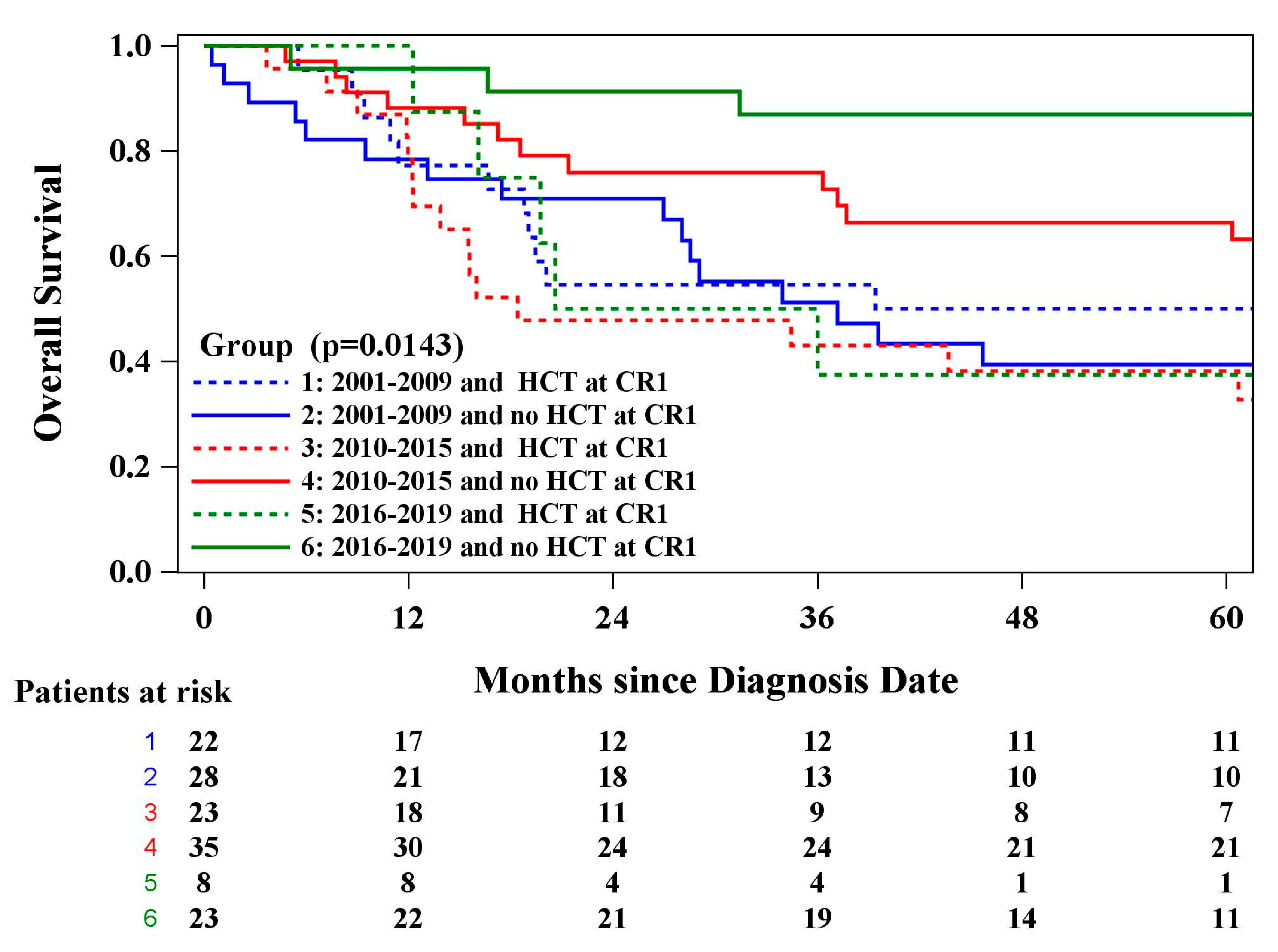
Post-Induction Status and Survival
Univariate and Multivariable Analyses
Discussion
Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Badar, T.; Alkhateeb, H.; Aljurf, M.; Kharfan-Dabaja, M.A. Management of Philadelphia chromosome positive acute lymphoblastic leukemia in the current era. Curr Res Transl Med. 2023a, 71(2), 103392. [Google Scholar] [CrossRef]
- Badar, T.; Narra, R.; Mims, A.; Shallis, R.M.; De Camargo Correia, G.S.; Hunter, C.; Kota, V.K.; Desai, S.; Patel, A.A.; Duvall, A.S.; Palmisiano, N.; Curran, E.K.; Zulfa, O.; Advani, A.S.; Atallah, E.L.; Litzow, M.R. Achievement of Undetectable BCR::ABL1 (uBCR::ABL1) Is Predictive of Improved Survival in Philadelphia Chromosome Positive (Ph+ve) Acute Lymphoblastic Leukemia (ALL) Patients Not Receiving Allogeneic Stem Cell Transplantation. Blood 2023b, 142 Supplement 1, 1464. [Google Scholar] [CrossRef]
- Bassan, R.; Rossi, G.; Pogliani, E.M.; Di Bona, E.; Angelucci, E.; Cavattoni, I.; Lambertenghi-Deliliers, G.; Mannelli, F.; Levis, A.; Ciceri, F.; Mattei, D.; Borlenghi, E.; Terruzzi, E.; Borghero, C.; Romani, C.; Spinelli, O.; Tosi, M.; Oldani, E.; Intermesoli, T; Rambaldi, A. Chemotherapy-phased imatinib pulses improve long-term outcome of adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia: Northern Italy Leukemia Group protocol 09/00. J Clin Oncol 2010, 28(22), 3644–52. [Google Scholar] [CrossRef]
- Bazarbachi, A.; Labopin, M.; Dalle, I.A.; Yakoub-Agha, I.; Socié, G.; Schroeder, T.; Blaise, D.; Poiré, X.; Balsat, M.; Salmenniemi, U.; Kröger, N.; Kulagin, A.; Wagner-Drouet, E.M.; Wu, D.; Brissot, E.; Nagler, A.; Giebel, S.; Ciceri, F.; Mohty, M. Improved post-transplant outcomes since 2000 for Ph-positive acute lymphoblastic leukemia in first remission: A study from the EBMT Acute Leukemia Working Party. Hemasphere 2025, 9(4), e70117. [Google Scholar] [CrossRef] [PubMed]
- Chalandon, Y.; Thomas, X.; Hayette, S.; Cayuela, J.M.; Abbal, C.; Huguet, F.; Raffoux, E.; Leguay, T.; Rousselot, P.; Lepretre, S.; Escoffre-Barbe, M.; Maury, S.; Berthon, C.; Tavernier, E.; Lambert, J.F.; Lafage-Pochitaloff, M.; Lhéritier, V.; Chevret, S.; Ifrah, N.; Dombret, H.; Group for Research on Adult Acute Lymphoblastic Leukemia (GRAALL). Randomized study of reduced-intensity chemotherapy combined with imatinib in adults with Ph-positive acute lymphoblastic leukemia. Blood 2015, 125(24), 3711–9. [Google Scholar] [CrossRef]
- Daver, N.; Thomas, D.; Ravandi, F.; Cortes, J.; Garris, R.; Jabbour, E.; Garcia-Manero, G.; Borthakur, G.; Kadia, T.; Rytting, M.; Konopleva, M.; Kantarjian, H.; O’Brien, S. Final report of a phase II study of imatinib mesylate with hyper-CVAD for the front-line treatment of adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia. Haematologica 2015, 100(5), 653–61. [Google Scholar] [CrossRef] [PubMed]
- DeFilipp, Z.; Advani, A.S.; Bachanova, V.; Cassaday, R.D.; Deangelo, D.J.; Kebriaei, P.; Rowe, J.M.; Seftel, M.D.; Stock, W.; Tallman, M.S.; Fanning, S.; Inamoto, Y.; Kansagra, A.; Johnston, L.; Nagler, A.; Sauter, C.S.; Savani, B.N.; Perales, M.A.; Carpenter, P.A.; Larson, R.A.; Weisdorf, D. Hematopoietic Cell Transplantation in the Treatment of Adult Acute Lymphoblastic Leukemia: Updated 2019 Evidence-Based Review from the American Society for Transplantation and Cellular Therapy. Biol Blood Marrow Transplant 2019, 25(11), 2113–2123. [Google Scholar] [CrossRef]
- Fielding, A.K.; Rowe, J.M.; Buck, G.; Foroni, L.; Gerrard, G.; Litzow, M.R.; Lazarus, H.; Luger, S.M.; Marks, D.I.; McMillan, A.K.; Moorman, A.V.; Patel, B.; Paietta, E.; Tallman, M.S.; Goldstone, A.H. UKALLXII/ECOG2993: addition of imatinib to a standard treatment regimen enhances long-term outcomes in Philadelphia positive acute lymphoblastic leukemia. Blood 2014, 123(6), 843–50. [Google Scholar] [CrossRef]
- Foà, R.; Bassan, R.; Vitale, A.; Elia, L.; Piciocchi, A.; Puzzolo, M.C.; Canichella, M.; Viero, P.; Ferrara, F.; Lunghi, M.; Fabbiano, F.; Bonifacio, M.; Fracchiolla, N.; Di Bartolomeo, P.; Mancino, A.; De Propris, M.S.; Vignetti, M.; Guarini, A.; Rambaldi, A.; Chiaretti, S. GIMEMA Investigators. Dasatinib-Blinatumomab for Ph-Positive Acute Lymphoblastic Leukemia in Adults. N Engl J Med. 2020, 383(17), 1613–1623. [Google Scholar] [CrossRef]
- Foà, R.; Bassan, R.; Elia, L.; Piciocchi, A.; Soddu, S.; Messina, M.; Ferrara, F.; Lunghi, M.; Mulè, A.; Bonifacio, M.; Fracchiolla, N.; Salutari, P.; Fazi, P.; Guarini, A.; Rambaldi, A.; Chiaretti, S. Long-Term Results of the Dasatinib-Blinatumomab Protocol for Adult Philadelphia-Positive ALL. J Clin Oncol 2024, 42(8), 881–885. [Google Scholar] [CrossRef]
- Jabbour, E.; DerSarkissian, M.; Duh, M.S.; McCormick, N.; Cheng, W.Y.; McGarry, L.J.; Souroutzidis, A.; Huang, H.; O’Brien, S.; Ravandi, F.; Kantarjian, H.M. Efficacy of Ponatinib Versus Earlier Generation Tyrosine Kinase Inhibitors for Front-line Treatment of Newly Diagnosed Philadelphia-positive Acute Lymphoblastic Leukemia. Clin Lymphoma Myeloma Leuk 2018, 18(4), 257–265. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, E.; Short, N.J.; Jain, N.; Huang, X.; Montalban-Bravo, G.; Banerjee, P.; Rezvani, K.; Jiang, X.; Kim, K.H.; Kanagal-Shamanna, R.; Khoury, J.D.; Patel, K.; Kadia, T.M.; Daver, N.; Chien, K.; Alvarado, Y.; Garcia-Manero, G.; Issa, G.C.; Haddad, F.G.; Kwari, M.; Thankachan, J.; Delumpa, R.; Macaron, W.; Garris, R.; Konopleva, M.; Ravandi, F.; Kantarjian, H. Ponatinib and blinatumomab for Philadelphia chromosome-positive acute lymphoblastic leukaemia: a US, single-centre, single-arm, phase 2 trial. Lancet Haematol 2023, 10(1), e24–e34. [Google Scholar] [CrossRef] [PubMed]
- Kanfar, S.S.; Chan, S.M.; Gupta, V.; Schimmer, A.D.; Schuh, A.C.; Sibai, H.; Yee, K.W.L.; Minden, M.D. Outcomes of Adult Philadelphia Positive Acute Lymphoblastic Leukemia Patients Treated with Pediatric Multi-Agent Chemotherapy and Imatinib and the Impact of Residual Disease Monitoring on Survival. Blood 2016, 128(22), 3976. [Google Scholar] [CrossRef]
- Lee, K.H.; Lee, J.H.; Choi, S.J.; Lee, J.H.; Seol, M.; Lee, Y.S.; Kim, W.K.; Lee, J.S.; Seo, E.J.; Jang, S.; Park, C.J.; Chi, H.S. Clinical effect of imatinib added to intensive combination chemotherapy for newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia 2005, 19(9), 1509–16. [Google Scholar] [CrossRef]
- Martell, M.P.; Atenafu, E.G.; Minden, M.D.; Schuh, A.C.; Yee, K.W.; Schimmer, A.D.; Gupta, V.; Brandwein, J.M. Treatment of elderly patients with acute lymphoblastic leukaemia using a paediatric-based protocol. Br J Haematol 2013, 163(4), 458–64. [Google Scholar] [CrossRef]
- Martinelli, G.; Iacobucci, I.; Storlazzi, C.T.; Vignetti, M.; Paoloni, F.; Cilloni, D.; Soverini, S.; Vitale, A.; Chiaretti, S.; Cimino, G.; Papayannidis, C.; Paolini, S.; Elia, L.; Fazi, P.; Meloni, G.; Amadori, S.; Saglio, G.; Pane, F.; Baccarani, M.; Foà, R. IKZF1 (Ikaros) deletions in BCR-ABL1-positive acute lymphoblastic leukemia are associated with short disease-free survival and high rate of cumulative incidence of relapse: a GIMEMA AL WP report. J Clin Oncol 2009, 27(31), 5202–7. [Google Scholar] [CrossRef]
- Patel, B.; Kirkwood, A.A.; Dey, A.; Marks, D.I.; McMillan, A.K.; Menne, T.F.; Micklewright, L.; Patrick, P.; Purnell, S.; Rowntree, C.J.; Smith, P.; Fielding, A.K. Pegylated-asparaginase during induction therapy for adult acute lymphoblastic leukaemia: toxicity data from the UKALL14 trial. Leukemia 2017, 31(1), 58–64. [Google Scholar] [CrossRef]
- Ravandi, F.; Jorgensen, J.L.; Thomas, D.A.; O’Brien, S.; Garris, R.; Faderl, S.; Huang, X.; Wen, S.; Burger, J.A.; Ferrajoli, A.; Kebriaei, P.; Champlin, R.E.; Estrov, Z.; Challagundla, P.; Wang, S.A.; Luthra, R.; Cortes, J.E.; Kantarjian, H.M. Detection of MRD may predict the outcome of patients with Philadelphia chromosome-positive ALL treated with tyrosine kinase inhibitors plus chemotherapy. Blood 2013, 122(7), 1214–21. [Google Scholar] [CrossRef]
- Ribera, J.M.; Ribera, J.; Genescà, E. The role of stem cell transplantation in the management of Philadelphia chromosome-positive acute lymphoblastic leukemia. Ther Adv Hematol 2018, 9(12), 357–368. [Google Scholar] [CrossRef]
- Saleh, K.; Fernandez, A.; Pasquier, F. Treatment of Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia in Adults. Cancers (Basel) 2022, 14(7), 1805. [Google Scholar] [CrossRef]
- Shah, B.; Mattison, R.J.; Abboud, R.; Abdelmessieh, P.; Aldoss, I.; Burke, P.W.; DeAngelo, D.J.; Dinner, S.; Fathi, A.T.; Gauthier, J.; Haddadin, M.; Jain, N.; Jonas, B.; Kirby, S.; Liedtke, M.; Litzow, M.; Logan, A.; Long, M.; Luger, S.; Mangan, J.K.; Massaro, S.; May, W.; Oluwole, O.; Park, J.; Przespolewski, A.; Rangaraju, S.; Saygin, C.; Schwartz, M.; Shami, P.; Tomlinson, B.; Webster, J.; Awotiwon, A.; Stehman, K. Acute Lymphoblastic Leukemia, Version 2.2024, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2024, 22(8), 563–576. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Jabbour, E.; Sasaki, K.; Patel, K.; O’Brien, S.M.; Cortes, J.E.; Garris, R.; Issa, G.C.; Garcia-Manero, G.; Luthra, R.; Thomas, D.; Kantarjian, H.; Ravandi, F. Impact of complete molecular response on survival in patients with Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood 2016, 128(4), 504–7. [Google Scholar] [CrossRef]
- Snowden, J.A.; Sánchez-Ortega, I.; Corbacioglu, S.; Basak, G.W.; Chabannon, C.; de la Camara, R.; Dolstra, H.; Duarte, R.F.; Glass, B.; Greco, R.; Lankester, A.C.; Mohty, M.; Neven, B.; de Latour, R.P.; Pedrazzoli, P.; Peric, Z.; Yakoub-Agha, I.; Sureda, A.; Kröger, N.; European Society for Blood and Marrow Transplantation (EBMT). Indications for haematopoietic cell transplantation for haematological diseases, solid tumours and immune disorders: current practice in Europe, 2022. Bone Marrow Transplant 2022, 57(8), 1217–1239. [Google Scholar] [CrossRef]
- Thyagu, S.; Minden, M.D.; Gupta, V.; Yee, K.W.; Schimmer, A.D.; Schuh, A.C.; Lipton, J.H.; Messner, H.A.; Xu, W.; Brandwein, J.M. Treatment of Philadelphia chromosome-positive acute lymphoblastic leukaemia with imatinib combined with a paediatric-based protocol. Br J Haematol 2012, 158(4), 506–14. [Google Scholar] [CrossRef] [PubMed]
- Yanada, M.; Takeuchi, J.; Sugiura, I.; Akiyama, H.; Usui, N.; Yagasaki, F.; Kobayashi, T.; Ueda, Y.; Takeuchi, M.; Miyawaki, S.; Maruta, A.; Emi, N.; Miyazaki, Y.; Ohtake, S.; Jinnai, I.; Matsuo, K.; Naoe, T.; Ohno, R. Japan Adult Leukemia Study Group. High complete remission rate and promising outcome by combination of imatinib and chemotherapy for newly diagnosed BCR-ABL-positive acute lymphoblastic leukemia: a phase II study by the Japan Adult Leukemia Study Group. J Clin Oncol 2006, 24(3), 460–6. [Google Scholar] [CrossRef] [PubMed]
| Parameter | All Patients N=141 |
|---|---|
| Median age, year (range) | 50.00 (18.0-79.1) |
| Age ≥60, n (%) | 39 (27.66) |
| Male, n (%) | 77 (54.61) |
| Female, n (%) | 64 (45.39) |
| Median WBC count, x109/L (range) | 19.20 (0.80-272.0) |
| Extramedullary disease, n (%) | 14 (9.93) |
| CNS involvement at diagnosis, n (%) | 15 (10.64) |
| Prior cancer history, n (%) | 13 (9.63) |
| Chemotherapy, n (%) | |
| Pediatric-inspired multiagent protocol | 140 (99.29) |
| Other | 1 (0.71) |
| Asparaginase, n (%) | 35 (24.82) |
| TKI, n (%) | |
| Imatinib | 138 (98.57) |
| Dasatinib | 2 (1.43) |
| Transplant in CR1, n (%) | 53 (37.59) |
|
Variable |
OS | RFS | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | |
| Age (>60 years) | 0.91 | (0.54, 1.54) | 0.7255 | 1.06 | (0.65, 1.72) | 0.8198 |
| Sex (female) | 1.52 | (0.95, 2.43) | 0.0824 | 1.42 | (0.92, 2.20) | 0.1162 |
| WBC (>30 x109/L) | 1.27 | (0.78, 2.10) | 0.3389 | 1.24 | (0.78, 1.97 | 0.3696 |
| Extramedullary disease + CNS involvement (present) | 1.20 | (0.67, 2.17) | 0.5434 | 0.95 | (0.53, 1.70) | 0.8690 |
| Prior cancer (present) | 1.51 | (0.72, 3.18) | 0.2720 | 1.69 | (0.84, 3.41) | 0.1384 |
| Asparaginase (yes) | 1.10 | (0.65, 1.87) | 0.7245 | 0.99 | (0.60, 1.65) | 0.9824 |
| Year of induction (ref=2001-2009) |
0.87 |
(0.53, 1.44) |
0.0651 0.5970 |
1.01 |
(0.62, 1.63) |
0.2008 0.9747 |
| 2010-2015 | ||||||
| 2016-2019 | 0.39 | (0.18, 0.86) | 0.0201 | 0.57 | (0.29, 1.12) | 0.1012 |
| HCTCR1t (time varying) | 2.19 | (1.35, 3.57) | 0.0016 | 1.73 | (1.09, 2.74) | 0.0204 |
| BCR::ABL1 3/6 months (>3-log reduction) | 1.09 | (0.51, 2.32) | 0.8329 | 1.02 | (0.51, 2.03) | 0.9557 |
| BCR::ABL1 9/12 months (>3-log reduction) | 0.35 | (0.16, 0.79) | 0.0115 | 0.38 | (0.18, 0.81) | 0.0120 |
|
Variable |
OS | RFS | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | |
| Sex (female) | 1.66 | (0.90, 3.06) | 0.1016 | 1.53 | (0.87, 2.69) | 0.1371 |
| Prior cancer (present) | 1.35 | (0.52, 3.56) | 0.5385 | 1.56 | (0.65, 3.74) | 0.3215 |
| BCR::ABL1 9/12 months (>3-log reduction) | 0.37 | (0.15, 0.94) | 0.0368 | 0.36 | (0.15, 0.86) | 0.0220 |
| Year of induction (ref=2001-2009) | 0.2876 | 0.2870 | ||||
| 2010-2015 | 1.24 | (0.63, 2.45) | 0.5350 | 1.39 | (0.74, 2.62) | 0.3021 |
| 2016-2019 | 0.47 | (0.13, 1.73) | 0.2583 | 0.71 | (0.25, 2.03) | 0.5211 |
| HCTCR1t (time varying) | 2.26 | (1.18, 4.33) | 0.0139 | 1.88 | (1.01, 3.48) | 0.0452 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




