1. Introduction
Obstructive sleep apnea (OSA) is a prevalent sleep disorder characterized by recurrent episodes of partial or complete upper airway obstruction during sleep, leading to intermittent hypoxia, sympathetic activation, and sleep fragmentation [
1,
2]. In recent years, there has been growing interest in the prevalence and consequences of OSA in patients with chronic pulmonary diseases, particularly those with bronchiectasis, a condition marked by permanent dilation of bronchi, impaired mucociliary clearance, and chronic airway inflammation [
3,
4].
Among bronchiectasis subtypes, both cystic fibrosis (CF)-related and non-CF bronchiectasis have been associated with an increased risk of sleep-disordered breathing (SDB) [
5]. Structural lung damage, mucus plugging, and airflow obstruction can compromise nocturnal ventilation and gas exchange, predisposing patients to more severe desaturation events during sleep [
6,
7]. Prior studies have demonstrated a high prevalence of OSA in these populations, often exceeding that in the general population [
8,
9].
Furthermore, the pathophysiological burden of OSA may be exacerbated by comorbid systemic conditions such as anemia, which is frequently observed in bronchiectasis patients due to chronic inflammation, recurrent infections, nutritional deficiencies, and in the case of CF, malabsorption syndromes [
10,
11]. Anemia reduces the blood’s oxygen carrying capacity, which may compound the deleterious effects of OSA-related intermittent hypoxia [
10,
12,
13].
Despite this reasonable pathophysiological synergy, the interaction between anemia and OSA severity on nocturnal hypoxemia in bronchiectasis remains underexplored. Most existing studies have examined these factors in isolation, without evaluating their combined impact on sleep-related oxygenation. Understanding this relationship is crucial, as it may help refine risk stratification and identify novel therapeutic targets to improve sleep and respiratory outcomes in this vulnerable patient group.
This study aims to investigate whether anemia modifies the relationship between OSA and sleep-related hypoxemia in patients with bronchiectasis, hypothesizing that the presence of anemia amplifies nocturnal oxygen desaturation in those with coexisting OSA.
2. Materials and Methods
2.1. Study Design and Participants
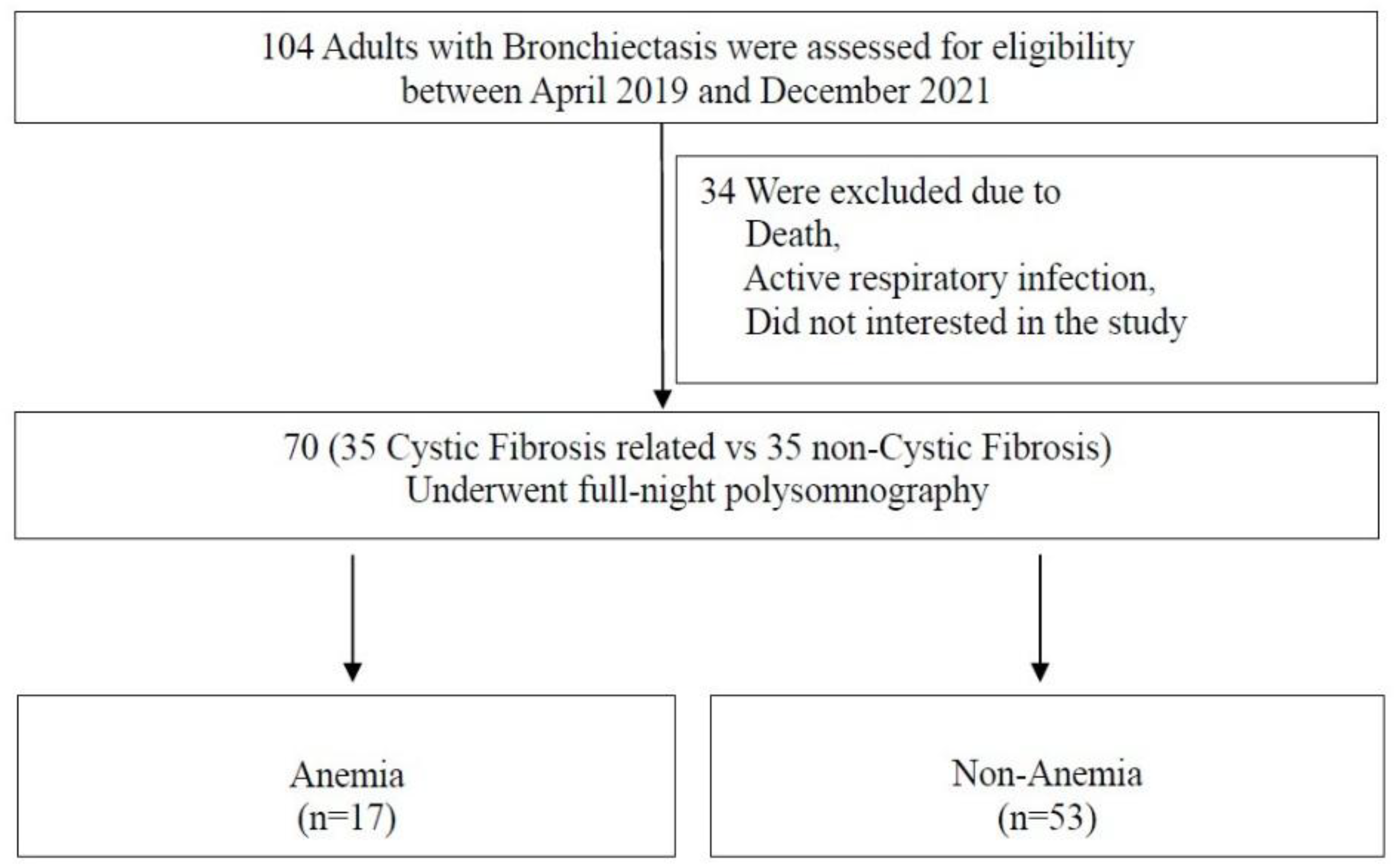
This prospective, cross-sectional investigation was carried out between April 2019 and December 2021. Adult patients (aged ≥18 years) diagnosed with bronchiectasis, either CF-related or non-CF—were recruited from the Adult Chest Diseases Outpatient Clinic at Marmara University Pendik Training and Research Hospital (
Figure 1). Of the 104 initially eligible individuals, 34 were excluded due to lack of consent for further evaluation, death, or active respiratory infection during the follow-up period. The final study cohort included 70 participants, evenly divided between CF (n=35) and non-CF (n=35) bronchiectasis cases. Anemia was defined using standard hemoglobin thresholds: <13 g/dL for men and <12 g/dL for women. The study received approval from the Marmara University Institutional Ethics Committee (protocol number: 09.2019.503) and adhered to the ethical standards outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants.
2.2. Polysomnographic Evaluation
All subjects underwent overnight attended polysomnography (PSG) using the Embletta device (Natus Medical Incorporated, Orlando, USA). The PSG recordings included data on sleep architecture, total sleep duration, nasal airflow, thoracoabdominal movements, body posture, heart rate, oxygen saturation (SpO₂), leg muscle activity, periodic limb movements, and snoring events. In cases where total sleep time was less than 240 minutes, a repeat PSG was offered. Scoring and interpretation were performed according to the American Academy of Sleep Medicine (AASM) criteria by a single trained physician (B.B) to ensure consistency across assessments [
14].
2.3. Participant Stratification
As shown in
Figure 1, 104 adult patients were screened for eligibility. Following exclusion criteria, 70 patients were included in the analysis, of whom 17 were identified as anemic. All included patients completed an in-laboratory full-night PSG, and only those with a total sleep time exceeding 4 hours were retained for final analysis.
2.4. Assessment Tools
2.4.1. Epworth Sleepiness Scale (ESS)
Daytime sleepiness was evaluated using the validated Turkish version of the ESS, a self-report questionnaire composed of eight items scored from 0 to 3. The cumulative score ranges from 0 to 24, with scores ≥11 indicating excessive daytime sleepiness (EDS) [
15].
2.4.2. Charlson Comorbidity Index
This scoring system was applied to quantify each participant's comorbid burden, serving as a predictor of 1-year mortality risk. Higher scores represent more severe comorbid conditions [
16].
2.4.3. Modified Medical Research Council (mMRC) Dyspnea Scale
Participants' perceptions of breathlessness were assessed using the mMRC scale, a 5-point tool ranging from 0 (no breathlessness) to 4 (severe dyspnea during minimal exertion) [
17].
2.4.4. Zung Self-Rating Depression Scale (SDS)
The presence of depressive symptoms was determined at baseline using the Turkish version of the Zung SDS [
18]. This self-administered instrument contains 20 items, each rated on a 4-point scale from 1 ("a little of the time") to 4 ("most of the time"). Reverse scoring was applied to items 2, 5, 6, 11, 12, 14, 16, 17, 18, and 20. Scores ≥40 were considered indicative of depression. All responses were entered into the study database by a designated investigator.
2.5. Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp., Armonk, NY, USA). Continuous variables were summarized as mean ± standard deviation (SD) if normally distributed or as median with interquartile range (IQR) if not. Categorical data were expressed as counts and percentages, along with 95% confidence intervals (CI). Comparisons between groups were conducted using the t-test for normally distributed continuous variables, the Mann–Whitney U test for non-normally distributed data, and the chi-square test (or Fisher’s exact test when appropriate) for categorical variables. Logistic regression models were employed to examine associations between sleep apnea and clinical variables, with results reported as odds ratios (ORs) and 95% confidence intervals. All tests were two-tailed, and statistical significance was defined as a p-value <0.05.
3. Results
3.1. Demographics, Clinical, and Sleep Related Characteristics
As shown in
Table 1, 17 participants had anemia, while 53 did not. Notably, the proportion of female participants was significantly higher in the anemia group compared to the non-anemia group (38.5% vs. 6.5%; p = 0.002). Furthermore, nutritional problems were more frequently reported by patients with anemia (47.1% vs. 20.8%; p = 0.034). The prevalence of OSA was also significantly greater in the anemia group (94.1% vs. 54.7%; p = 0.003). Additionally, hospitalization rates per year were markedly higher among anemic patients (1.41 ± 0.41 vs. 0.43 ± 0.10; p = 0.002). In contrast, the mean age was nearly identical between the two groups, and there was no significant difference in body mass index (BMI) or smoking status. Comorbidities such as diabetes, pancreatic disease, cardiac disease, and oxygen supplementation use were similar between groups. Measures of daytime sleepiness also did not differ significantly; although the anemia group had a higher mean ESS score, this difference did not reach statistical significance, and subjective sleepiness was also comparable. There were no significant group differences in dyspnea severity MMRC, Charlson Comorbidity Index, or disease duration. Although patients with anemia experienced more frequent exacerbations per year, the difference was not statistically significant. Similarly, depressive symptoms measured by the SDS score were higher in the anemia group but not significantly so. Polysomnographic parameters, including total sleep time, sleep efficiency, sleep latency, and REM latency were not significantly different between the groups. Similarly, no significant AHI, ODI, average and lowest oxygen saturation, or the duration and percentage of time spent with SpO₂ < 90%. Finally, although the average heart rate tended to be higher in the anemia group this did not reach statistical significance.
3.2. Factors Associated with Anemia in Adults with Bronchiectasis
As presented in
Table 2, In the univariate analysis, female gender was strongly associated with anemia (OR: 9.06; 95% CI: 1.88–43.62; p = 0.006). In addition, hospitalization frequency was significantly associated with anemia (OR:1.99; 95% CI: 1.21–3.23; p = 0.006). Nutritional support also showed a significant association (OR: 3.39; 95% CI: 1.06–10.83; p = 0.039). Lastly, OSA was significantly associated with anemia (OR: 3.42; 95% CI: 1.64–7.21; p = 0.015). Several other variables were not significantly associated with anemia in either model. These included age, BMI, and smoking status. Neither CF status nor pancreatic disease showed significant associations. Disease duration, MMRC dyspnea score, Charlson Comorbidity Index, and comorbidities such as diabetes mellitus and oxygen therapy use were also not significantly linked to anemia. Although ESS score and average heart rate showed a trend toward association (ESS: OR: 1.14; p = 0.080; heart rate: OR: 1.05; p = 0.095), these did not reach statistical significance. Additionally, no significant associations were found for polysomnographic parameters, including total sleep time, sleep efficiency, sleep latency, AHI, ODI, average or lowest oxygen saturation, or duration of desaturation below 90%. In the multivariate analysis, after adjusting for potential confounders, female gender remained a independent predictor of anemia (OR: 12.32; 95% CI: 3.12–45.96; p = 0.002). Similarly, OSA continued to be significantly associated with anemia (OR: 4.70; 95% CI: 2.67–45.29; p = 0.007). Hospitalization and nutritional support, while significant in univariate analysis, did not retain significance after adjustment.
3.3. Factors Associated with Anemia in Participants with Cystic Fibrosis
As demonstrated in
Table 3, In the univariate analysis, OSA was found to be significantly associated with anemia among CF participants (OR: 1.24; 95% CI: 1.88–12.25; p = 0.045). Female gender was related with anemia, but did not reach statistical significance. Hospitalization frequency (OR: 1.68; p = 0.093) and Zung SDS score for depressive symptoms (OR: 1.06; p = 0.094) also showed borderline associations. Other clinical and demographic characteristics such as age, BMI, smoking status, disease duration, MMRC score, Charlson index, diabetes, and nutritional support were not significantly associated with anemia. Likewise, subjective sleepiness and ESS score itself did not reach statistical significance. Polysomnographic parameters, including total sleep time, sleep efficiency, sleep latency, AHI, ODI, and oxygen saturation metrics, did not show meaningful associations with anemia. In the multivariate model, no variables remained statistically significant. OSA lost its significance (OR: 1.03; 95% CI: 0.98–1.13; p = 0.660), and neither female gender (OR: 1.01; p = 0.217) nor age (OR: 1.27; p = 0.740) independently predicted anemia among CF patients.
3.4. Factors Associated with Anemia in Bronchiectasis Patients (Excluding Cystic Fibrosis)
As shown in
Table 4, In the univariate analysis, hospitalization frequency, and OSA (OR: 4.50; 95% CI: 0.47–42.5; p = 0.049) was significantly associated with anemia (OR: 2.85; 95% CI: 1.01–8.01; p = 0.046). Other variables, including age, female gender, and BMI were not significantly associated with anemia. Additional clinical parameters such as smoking status, disease duration, MMRC score, Charlson index, and diabetes mellitus did not show meaningful associations. While nutritional support showed a trend toward increased anemia risk (OR: 3.80; p = 0.091), this did not reach statistical significance. Subjective sleep-related variables like ESS score, and mood symptoms measured via Zung SDS score, were not significantly linked to anemia. Similarly, polysomnographic measures such as total sleep time, sleep efficiency, sleep latency, AHI, ODI, and oxygen saturation metrics did not differ significantly between those with and without anemia. In the multivariate model, after adjusting for age and OSA, hospitalization remained an independent predictor of anemia (OR: 3.69; 95% CI: 1.06–12.84; p = 0.040), OSA did not retain significance (OR: 1.12; 95% CI: 0.96–1.31; p = 0.136).
4. Discussion
This study reveals important associations between anemia and clinical, nutritional, and sleep-related characteristics in a bronchiectasis cohort. Our findings suggest that anemia may be linked to several adverse outcomes, including a higher prevalence of obstructive sleep apnea (OSA), increased hospitalization rates, and more frequent nutritional challenges.
The significantly higher proportion of female participants in the anemia group aligns with well-established epidemiological data showing that anemia is more prevalent among women due to factors such as menstruation, pregnancy, and iron deficiency [
19,
20]. This sex-based disparity may also reflect broader biological and sociocultural influences that affect healthcare access and nutritional status. Nutritional deficiencies were more commonly reported among patients with anemia, consistent with the known etiological relationship between inadequate intake or absorption of micronutrients—particularly iron, folate, and vitamin B12—and the development of anemia [
21]. In bronchiectasis patients, systemic inflammation and malabsorption may further exacerbate these deficiencies.
One of the most striking findings of our study is the significantly higher prevalence of OSA among patients with anemia and significant association OSA with anemia. This association aligns with prior research, for example, Khan et al. noted that in elderly patients, OSA treatment significantly changed hemoglobin and hematocrit levels, supporting a model where OSA-related hypoxia and inflammation influence erythropoiesis and anemia pathogenesis [
22]. Another study demonstrated that serum ferritin—an inflammatory and iron-storage biomarker—correlated positively with OSA severity, independent of BMI, suggesting systemic inflammation and iron dysregulation as meeting points between OSA and anemia [
23]. Further, a large NHANES cohort study found a U-shaped association between ferritin levels and OSA risk, supporting the view that both iron deficiency and inflammatory excess contribute to disease interplay in overweight/obese individuals [
24]. Finally, emergent research highlights that anemia and OSA may interact synergistically in influencing comorbid conditions like hypertension, rather than a simple unidirectional effect [
25]. Together, these lines of evidence support the hypothesis that OSA and anemia are interconnected via mechanisms such as hypoxia-induced erythropoietin alterations, systemic inflammation, and iron metabolism disruption, potentially amplifying each other’s clinical impact, all of which can contribute to impaired red blood cell production and anemia [
22,
26]. Although hospitalization frequency and nutritional support were significantly associated with anemia in univariate analysis, these associations did not persist in the adjusted model. This attenuation may reflect confounding by OSA or sex, or it may indicate that hospitalization and malnutrition act as mediators or downstream consequences rather than independent predictors. Nonetheless, their initial significance highlights the broader systemic impact of anemia in chronic lung disease and warrants further investigation. No significant associations were found between anemia and other clinical parameters as well as polysomnographic indices such as AHI, ODI, or nocturnal desaturation, suggesting that the presence of OSA itself—rather than its severity—may be a more relevant predictor of anemia risk. This supports a potential threshold effect, where even mild OSA may trigger inflammatory and hypoxic processes sufficient to alter erythropoiesis [
27].
Among CF participants, OSA was significantly associated with anemia in univariate analysis (OR: 1.24; p = 0.045). While this represents the only univariate predictor reaching significance, it aligns with evidence showing high prevalence of anemia and iron deficiency in adult CF patients—often related to malabsorption and vitamin deficiency (e.g., iron, folate, B₁₂). One study reported anemia in ~33% of CF adults, with 90% of vitamin-deficient patients being anemic (p=0.02), and iron deficiency common in evaluated cases [
28]. These findings suggest a potential but not definitive link between OSA and anemia in CF, underscoring the need for larger studies to clarify this relationship.
In non-CF bronchiectasis, both OSA (OR: 2.85; p = 0.046) and hospitalization frequency (OR: 4.50; p = 0.049) were significantly associated with anemia in univariate analysis. However, only hospitalizations remained an independent predictor in the multivariate model (OR: 3.69; p = 0.040), while OSA lost significance. This supports previous observations that anemia in bronchiectasis is linked with greater disease severity and healthcare utilization—studies report anemia prevalence in bronchiectasis up to ~65%, with associated low serum iron levels and inflammatory markers in affected patients [
29,
30]. Taken together, increased hospitalizations may serve as a robust indicator of anemia risk in non-CF bronchiectasis populations.
Limitations
Several limitations should be acknowledged. First, the cross-sectional design of the study precludes any causal inference regarding the relationship between anemia and OSA. Longitudinal studies are needed to clarify temporal and directional associations. Second, the sample size—particularly in subgroup analyses—was relatively small, which may have limited the statistical power to detect associations and led to wide confidence intervals in multivariate models. Third, anemia etiology was not specifically stratified (e.g., iron deficiency vs. anemia of chronic disease), and no direct laboratory markers such as ferritin, transferrin saturation, or vitamin B12 were included, which could have strengthened mechanistic interpretation. Additionally, although polysomnographic scoring was conducted by a single trained physician to minimize variability, there remains the possibility of scoring bias. Finally, generalizability may be limited, as all participants were recruited from a single tertiary referral center with advanced bronchiectasis care, possibly reflecting more severe phenotypes.
5. Conclusions
This study provides new insights into the relationship between anemia and obstructive sleep apnea (OSA) in patients with bronchiectasis, including both cystic fibrosis (CF)-related and non-CF etiologies. Anemia was significantly more prevalent in patients with OSA and was independently associated with female sex and OSA in the overall bronchiectasis cohort. In subgroup analyses, OSA was also associated with anemia in patients with CF, while in non-CF bronchiectasis, hospitalization frequency emerged as the strongest independent predictor. These findings highlight the multifactorial nature of anemia in bronchiectasis and underscore the potential contribution of sleep-disordered breathing to its pathogenesis. Clinically, the results suggest that evaluating and addressing OSA may have implications beyond sleep quality, potentially influencing systemic comorbidities such as anemia. Future longitudinal and interventional studies are warranted to determine whether targeted management of OSA or anemia can yield improvements in respiratory, hematologic, and overall functional outcomes in this population.
Author Contributions
Conceptualization, B.B., D.V. and B.C.; methodology, B.B.; software, B.B., Y.C.; validation, B.B., B.C. and D.V.; formal analysis, B.B., Y.C.; investigation, D.V.; resources, D.V., C.C.; data curation, B.B.; writing—original draft preparation, B.B.; writing—review and editing, B.B., D.V.; visualization, R.O.; supervision, B.C.; project administration, D.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Ethics Committee of Marmara University Institutional Ethics Committee, protocol number: 09.2019.503.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| AASM |
American Academy of Sleep Medicine |
| AHI |
Apnea–Hypopnea Index |
| BMI |
Body Mass Index |
| CF |
Cystic Fibrosis |
| CI |
Confidence Interval |
| EDS |
Excessive Daytime Sleepiness |
| ESS |
Epworth Sleepiness Scale |
| g/dL |
grams per deciliter |
| IBM |
International Business Machines |
| IQR |
Interquartile Range |
| mMRC |
Modified Medical Research Council |
| NHANES |
National Health and Nutrition Examination Survey |
| ODI |
Oxygen Desaturation Index |
| OR |
Odds Ratio |
| OSA |
Obstructive Sleep Apnea |
| PSG |
Polysomnography |
| REM |
Rapid Eye Movement |
| SDB |
Sleep-Disordered Breathing |
| SD |
Standard Deviation |
| SDS |
Zung Self-Rating Depression Scale |
| SpO₂ |
Peripheral Oxygen Saturation |
References
- Bradley, T.D.; Phillipson, E.A. Pathogenesis and pathophysiology of the obstructive sleep apnea syndrome. Med Clin North Am 1985, 69(6), 1169–85. [Google Scholar] [CrossRef] [PubMed]
- Jordan, A.S.; McSharry, D.G.; Malhotra, A. Adult obstructive sleep apnoea. Lancet 2014, 383(9918), 736–47. [Google Scholar] [CrossRef] [PubMed]
- Pasteur, M.C.; Bilton, D.; Hill, A.T. British Thoracic Society guideline for non-CF bronchiectasis. Thorax 2010, 65 Suppl 1, i1–58. [Google Scholar] [CrossRef]
- McShane, P.J. Non-cystic fibrosis bronchiectasis. Am J Respir Crit Care Med 2013, 188(6), 647–56. [Google Scholar] [CrossRef]
- Elborn, J.S. Cystic fibrosis. Lancet 2016, 388(10059), 2519–2531. [Google Scholar] [CrossRef] [PubMed]
- Panchabhai, T.S. Plugs of the Air Passages: A Clinicopathologic Review. Chest 2016, 150(5), 1141–1157. [Google Scholar] [CrossRef]
- Mikami, Y. Chronic airway epithelial hypoxia exacerbates injury in muco-obstructive lung disease through mucus hyperconcentration. Sci Transl Med 2023, 15(699), p. eabo7728. [Google Scholar] [CrossRef]
- Locke, B.W.; Lee, J.J.; Sundar, K.M. OSA and Chronic Respiratory Disease: Mechanisms and Epidemiology. Int J Environ Res Public Health 2022, 19(9). [Google Scholar] [CrossRef]
- Sunwoo, B.Y.; Malhotra, A. Mechanical Interactions Between the Upper Airway and the Lungs that Affect the Propensity to Obstructive Sleep Apnea in Health and Chronic Lung Disease. Sleep Med Clin 2024, 19(2), 211–218. [Google Scholar] [CrossRef]
- Bliss, S. Anemia and Oxygen Delivery. Vet Clin North Am Small Anim Pract 2015, 45(5), 917–30. [Google Scholar] [CrossRef]
- Zoroddu, S. The relationship between haemoglobin concentrations and obstructive sleep apnea syndrome: A systematic review and meta-analysis. Sleep Med 2024, 121, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z. The association of hemocyte profile and obstructive sleep apnea. J Clin Lab Anal 2019, 33(2), p. e22680. [Google Scholar]
- Pittman, R.N. Integrated Systems Physiology: From Molecule to Function to Disease, in Regulation of Tissue Oxygenation; Morgan & Claypool Life Sciences Copyright © 2011 by Morgan & Claypool Life Sciences.: San Rafael (CA), 2011. [Google Scholar]
- Sateia, M.J. International classification of sleep disorders-third edition: highlights and modifications. Chest 2014, 146(5), 1387–1394. [Google Scholar] [CrossRef]
- Johns, M.W. Daytime sleepiness, snoring, and obstructive sleep apnea. The Epworth Sleepiness Scale. Chest 1993, 103(1), 30–6. [Google Scholar] [CrossRef]
- Charlson, M.E. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987, 40(5), 373–83. [Google Scholar] [CrossRef]
- Fletcher, C.M. The significance of respiratory symptoms and the diagnosis of chronic bronchitis in a working population. Br Med J 1959, 2(5147), 257–66. [Google Scholar]
- Zung, W.W.; Richards, C.B.; Short, M.J. Self-rating depression scale in an outpatient clinic. Further validation of the SDS. Arch Gen Psychiatry 1965, 13(6), 508–15. [Google Scholar] [CrossRef] [PubMed]
- McLean, E. Worldwide prevalence of anaemia, WHO Vitamin and Mineral Nutrition Information System, 1993-2005. Public Health Nutr 2009, 12(4), 444–54. [Google Scholar]
- Camaschella, C. Iron-deficiency anemia. N Engl J Med 2015, 372(19), 1832–43. [Google Scholar] [CrossRef]
- Badireddy, M.; Baradhi, K.M. Chronic Anemia, in StatPearls. In StatPearls Publishing Copyright © 2025; StatPearls Publishing LLC.: Treasure Island (FL), 2025. [Google Scholar]
- Khan, A.M. Anemia of aging and obstructive sleep apnea. Sleep Breath 2011, 15(1), 29–34. [Google Scholar] [CrossRef] [PubMed]
- Seifen, C. Serum Ferritin Levels in Severe Obstructive Sleep Apnea. Diagnostics 2023, 13(6), 1154. [Google Scholar] [CrossRef]
- Zhang, Y. Association of Serum Ferritin Levels With Obstructive Sleep Apnea in Overweight/Obese US Populations: A Population-Based Study From the NHANES. Nat Sci Sleep 2025, 17, 223–238. [Google Scholar] [CrossRef]
- Yao, J. Synergistic effect of anemia and obstructive sleep apnea on hypertension: National Health and Nutrition Examination Survey 2015-2018. J Hypertens 2025, 43(4), 598–605. [Google Scholar] [CrossRef] [PubMed]
- Lavie, L. Oxidative stress inflammation and endothelial dysfunction in obstructive sleep apnea. Front Biosci (Elite Ed) 2012, 4(4), 1391–403. [Google Scholar] [CrossRef]
- Unruh, M.L. Sleep apnea in patients on conventional thrice-weekly hemodialysis: comparison with matched controls from the Sleep Heart Health Study. J Am Soc Nephrol 2006, 17(12), 3503–9. [Google Scholar] [CrossRef] [PubMed]
- von Drygalski, A.; Biller, J. Anemia in cystic fibrosis: incidence, mechanisms, and association with pulmonary function and vitamin deficiency. Nutr Clin Pract 2008, 23(5), 557–63. [Google Scholar] [CrossRef] [PubMed]
- Abdelaziz, A.O. Prevalence and impact of anemia in patients with chronic respiratory diseases. The Egyptian Journal of Chest Diseases and Tuberculosis 2018, 67(4), 461–470. [Google Scholar]
- Sarkar, M.; Rajta, P.N.; Khatana, J. Anemia in Chronic obstructive pulmonary disease: Prevalence, pathogenesis, and potential impact. Lung India 2015, 32(2), 142–51. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).