Submitted:
04 January 2026
Posted:
05 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Study Design and Study Population
- Group 1 (Standard rFSH Group): Cycles stimulated exclusively with conventional recombinant follicle-stimulating hormone (rFSH), initiated on cycle day 2 or 3.
- Group 2 (Letrozole + Late rFSH Group): Cycles stimulated with oral letrozole administered early in the follicular phase (cycle days 3–7), followed by late-onset gonadotropin stimulation initiated on cycle day 6 or later.
Inclusion and Exclusion Criteria
Ovarian Stimulation Protocols
Cycle Monitoring and Data Collection
- Duration of stimulation (days)
- Total gonadotropin dose (IU)
- Number of follicles measuring ≥17 mm
- Number of follicles measuring ≥14-17 mm
- Endometrial thickness on the day of ovulation trigger (mm)
- Serum estradiol (E2) concentration on the trigger day (pg/mL)
Ovulation Trigger, Insemination Procedure, and Luteal Phase Support
Outcome Measures
Statistical Analysis
Descriptive and Univariate Analyses
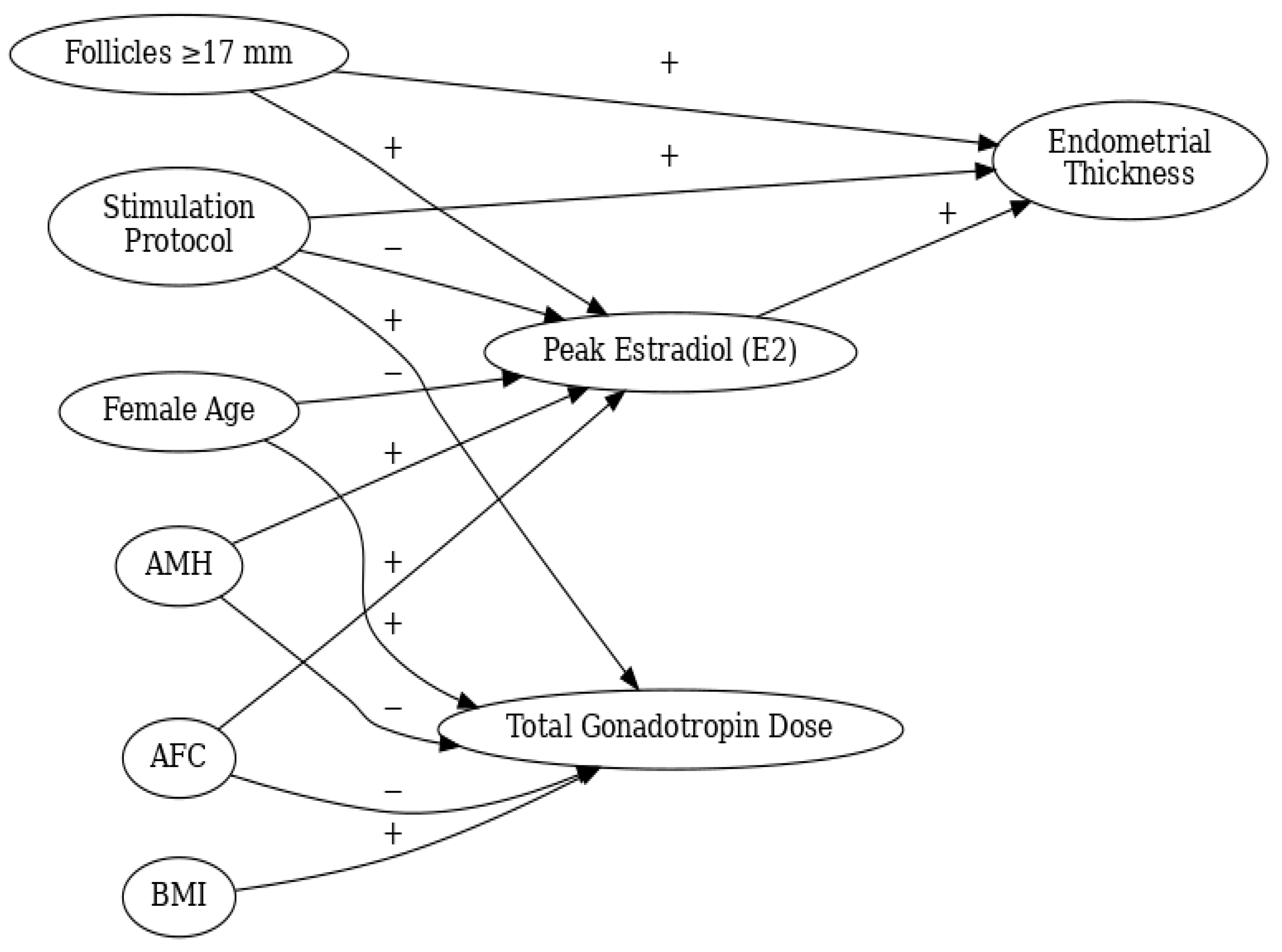
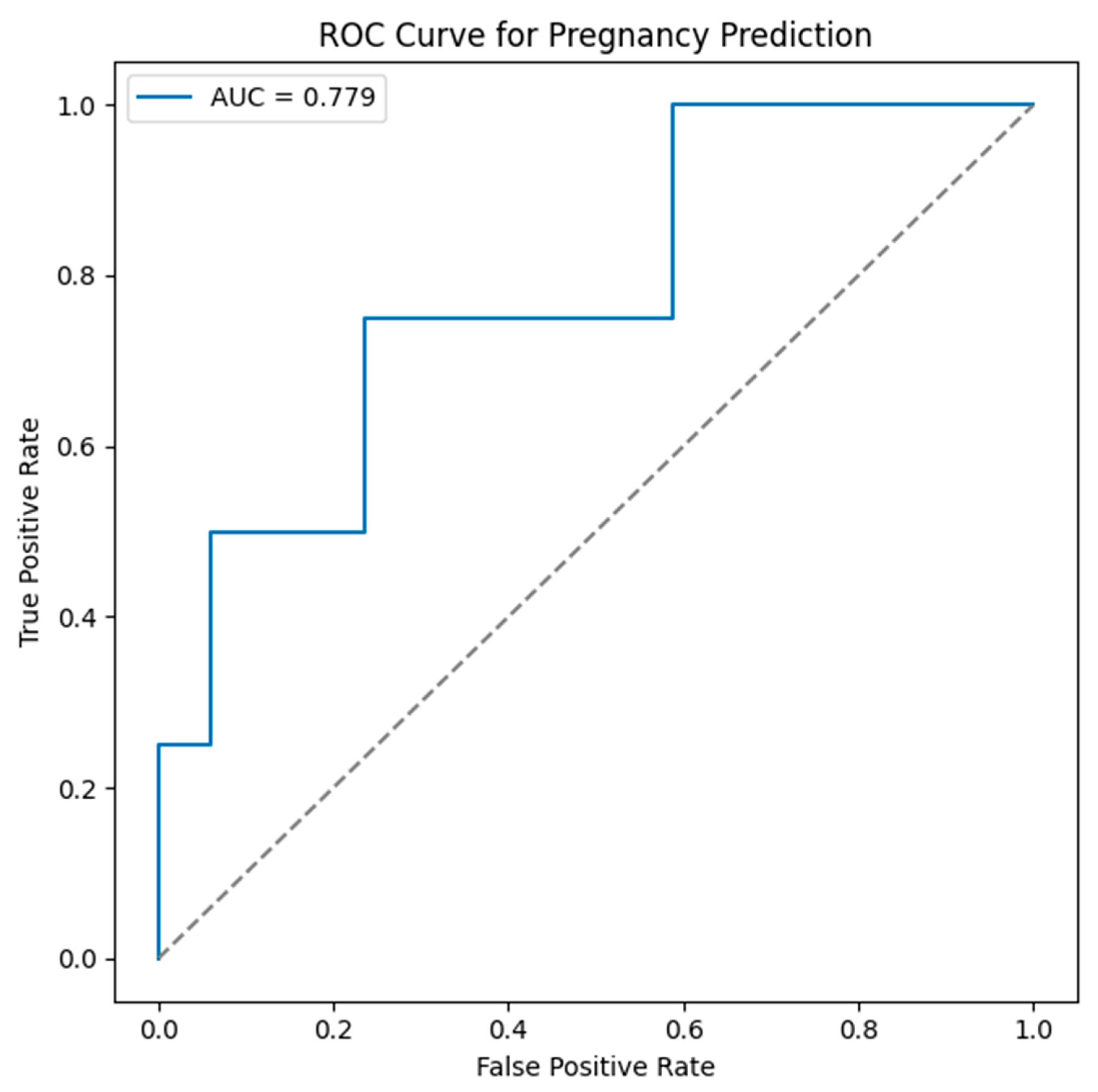
Multivariable Logistic Regression Analysis
- female age,
- stimulation protocol (letrozole plus late-onset rFSH vs conventional rFSH),
- AMH level,
- endometrial thickness on the day of hCG administration,
- number of follicles ≥17 mm on the trigger day.
Assessment of Interaction Effects
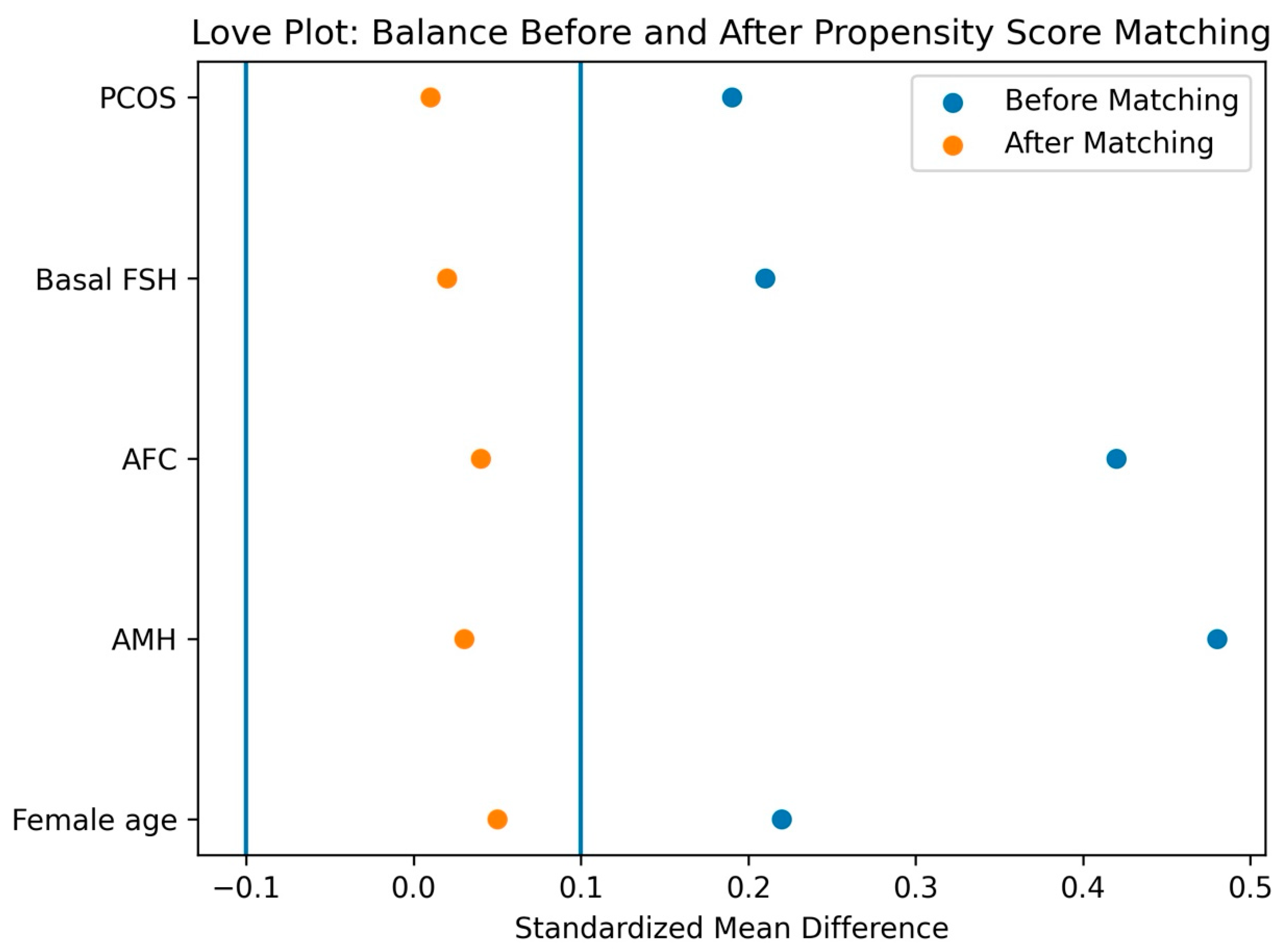
Propensity Score–Based Analyses
Outcome Analysis in the Matched Cohort
Inverse Probability of Treatment Weighting (IPTW)
Doubly Robust Estimation
Sensitivity Analyses
Results
Discussion
Strengths and Limitations
Clinical Implications and Individualized Treatment Strategies
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability
Acknowledgments
Conflicts of Interest
Abbreviations:
References
- Boivin, J.; Bunting, L.; Collins, J.A.; Nygren, K.G. International estimates of infertility prevalence and treatment-seeking: potential need and demand for infertility medical care. Hum Reprod 2007, 22, 1506–1512. [Google Scholar] [CrossRef]
- Mascarenhas, M.N.; Flaxman, S.R.; Boerma, T.; Vanderpoel, S.; Stevens, G.A. National, regional, and global trends in infertility prevalence since 1990: a systematic analysis of 277 health surveys. PLoS Med 2012, 9, e1001356. [Google Scholar] [CrossRef]
- Intrauterine insemination. Hum Reprod Update 2009, 15, 265–277. [CrossRef]
- Cohlen, B.J. Should we continue performing intrauterine inseminations in the year 2004? Gynecol Obstet Invest 2005, 59, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Cantineau, A.E.; Cohlen, B.J.; Heineman, M.J. Ovarian stimulation protocols (anti-oestrogens, gonadotrophins with and without GnRH agonists/antagonists) for intrauterine insemination (IUI) in women with subfertility. Cochrane Database Syst Rev 2007, CD005356. [Google Scholar] [CrossRef] [PubMed]
- Kamath, M.S.; Maheshwari, A.; Bhattacharya, S.; Lor, K.Y.; Gibreel, A. Oral medications including clomiphene citrate or aromatase inhibitors with gonadotropins for controlled ovarian stimulation in women undergoing in vitro fertilisation. Cochrane Database Syst Rev 2017, 11, Cd008528. [Google Scholar] [CrossRef]
- Guzick, D.S.; Carson, S.A.; Coutifaris, C.; Overstreet, J.W.; Factor-Litvak, P.; Steinkampf, M.P.; Hill, J.A.; Mastroianni, L., Jr.; Buster, J.E.; Nakajima, S.T. Efficacy of superovulation and intrauterine insemination in the treatment of infertility. New England Journal of Medicine 1999, 340, 177–183. [Google Scholar] [CrossRef]
- Wessel, J.A.; Danhof, N.A.; van Eekelen, R.; Diamond, M.P.; Legro, R.S.; Peeraer, K.; D’Hooghe, T.M.; Erdem, M.; Dankert, T.; Cohlen, B.J.; et al. Ovarian stimulation strategies for intrauterine insemination in couples with unexplained infertility: a systematic review and individual participant data meta-analysis. Hum Reprod Update 2022, 28, 733–746. [Google Scholar] [CrossRef]
- Mitwally, M.F.; Casper, R.F. Aromatase inhibition reduces the dose of gonadotropin required for controlled ovarian hyperstimulation. J Soc Gynecol Investig 2004, 11, 406–415. [Google Scholar] [CrossRef]
- Casper, R.F.; Mitwally, M.F. Aromatase inhibitors for ovulation induction. The Journal of Clinical Endocrinology & Metabolism 2006, 91, 760–771. [Google Scholar]
- Casper, R.F. Letrozole versus clomiphene citrate: which is better for ovulation induction? Fertil Steril 2009, 92, 858–859. [Google Scholar] [CrossRef] [PubMed]
- Legro, R.S.; Brzyski, R.G.; Diamond, M.P.; Coutifaris, C.; Schlaff, W.D.; Casson, P.; Christman, G.M.; Huang, H.; Yan, Q.; Alvero, R. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. New England Journal of Medicine 2014, 371, 119–129. [Google Scholar] [CrossRef]
- Alizzi, F.J. Letrozole with or without gonadotropin as a first-line ovulation induction in anovulatory infertile women due to polycystic ovary syndrome. Asian Journal of Pharmaceutical and Clinical Research 2018, 11, 129–133. [Google Scholar] [CrossRef]
- Arya, S.; Kupesic-Plavsic, S.; Mulla, Z.D.; Dwivedi, A.K.; Crisp, Z.; Jose, J.; Noble, L.S. Ovulation induction and controlled ovarian stimulation using letrozole gonadotropin combination: A single center retrospective cohort study. Eur J Obstet Gynecol Reprod Biol 2017, 218, 123–128. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Forman, R.; Mousa, N.A.; Al Inany, H.G.; Casper, R.F. Cost-effectiveness of aromatase inhibitor co-treatment for controlled ovarian stimulation. Human Reproduction 2006, 21, 2838–2844. [Google Scholar] [CrossRef] [PubMed]
- Tal, R.; Seifer, D.B. Ovarian reserve testing: a user’s guide. Am J Obstet Gynecol 2017, 217, 129–140. [Google Scholar] [CrossRef]
- Broer, S.L.; Broekmans, F.J.; Laven, J.S.; Fauser, B.C. Anti-Müllerian hormone: ovarian reserve testing and its potential clinical implications. Hum Reprod Update 2014, 20, 688–701. [Google Scholar] [CrossRef]
- Austin, P.C. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivariate Behav Res 2011, 46, 399–424. [Google Scholar] [CrossRef]
- Stuart, E.A. Matching methods for causal inference: A review and a look forward. Stat Sci 2010, 25, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Goverde, A.J.; McDonnell, J.; Vermeiden, J.P.; Schats, R.; Rutten, F.F.; Schoemaker, J. Intrauterine insemination or in-vitro fertilisation in idiopathic subfertility and male subfertility: a randomised trial and cost-effectiveness analysis. Lancet 2000, 355, 13–18. [Google Scholar] [CrossRef]
- Zeyneloglu, H.B.; Arici, A.; Olive, D.L.; Duleba, A.J. Comparison of intrauterine insemination with timed intercourse in superovulated cycles with gonadotropins: a meta-analysis. Fertil Steril 1998, 69, 486–491. [Google Scholar] [CrossRef] [PubMed]
- den Hartog, J.E.; Morre, S.A.; Land, J.A. Chlamydia trachomatis-associated tubal factor subfertility: Immunogenetic aspects and serological screening. Hum Reprod Update 2006, 12, 719–730. [Google Scholar] [CrossRef]
- Fuchs, C.S.; Giovannucci, E.L.; Colditz, G.A.; Hunter, D.J.; Stampfer, M.J.; Rosner, B.; Speizer, F.E.; Willett, W.C. Dietary fiber and the risk of colorectal cancer and adenoma in women. N Engl J Med 1999, 340, 169–176. [Google Scholar] [CrossRef]
- Chason, R.J.; Levens, E.D.; Yauger, B.J.; Payson, M.D.; Cho, K.; Larsen, F.W. Balloon fluoroscopy as treatment for intrauterine adhesions: a novel approach. Fertil Steril 2008, 90, 2005 e2015–2007. [Google Scholar] [CrossRef]
- Dessolle, L.; Biau, D. Proficiency in oocyte retrieval: how many procedures are necessary for training? Fertil Steril 2011, 96, e143; author reply e144. [Google Scholar] [CrossRef]
- Legro, R.S.; Brzyski, R.G.; Diamond, M.P.; Coutifaris, C.; Schlaff, W.D.; Casson, P.; Christman, G.M.; Huang, H.; Yan, Q.; Alvero, R.; et al. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med 2014, 371, 119–129. [Google Scholar] [CrossRef]
- Smitz, J.; Dolmans, M.M.; Donnez, J.; Fortune, J.E.; Hovatta, O.; Jewgenow, K.; Picton, H.M.; Plancha, C.; Shea, L.D.; Stouffer, R.L.; et al. Current achievements and future research directions in ovarian tissue culture, in vitro follicle development and transplantation: implications for fertility preservation. Hum Reprod Update 2010, 16, 395–414. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.J.; Zhang, X.M.; Chiriboga, L.; Yee, H.; Perle, M.A.; Mittal, K. Spatial differences in biologic activity of large uterine leiomyomata. Fertil Steril 2006, 85, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Kasius, A.; Smit, J.G.; Torrance, H.L.; Eijkemans, M.J.; Mol, B.W.; Opmeer, B.C.; Broekmans, F.J. Endometrial thickness and pregnancy rates after IVF: a systematic review and meta-analysis. Hum Reprod Update 2014, 20, 530–541. [Google Scholar] [CrossRef]
- Kim, H.H.; Speedy, S.E. The promised land of individualized ovarian stimulation: Are we there yet? Fertil Steril 2021, 115, 893–894. [Google Scholar] [CrossRef]
- Fauser, B.; Diedrich, K.; Devroey, P. Predictors of ovarian response: progress towards individualized treatment in ovulation induction and ovarian stimulation. Human reproduction update 2008, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
| Standard rFSH (n=372) | Letrozole + Late- rFSH (n=392) | P value | |
|---|---|---|---|
| Pregnancy n (%) | 37 (9.9%) | 58 (14.8%) | 0.042 |
|
Polycystic ovary syndrome (PCOS) n (%) |
61 (16.3%) | 127 (32.4%) | 0.001* |
| Cycle cancellation n (%) | 8 (2.1%) | 2(0.5%) | 0.12 |
| Miscarriage (n) | 8 | 14 | 0.28 |
| Ectopic pregnancy (n) | 2 | 1 | 0.61 |
| Ongoing pregnancy (n) | 5 | 24 | 0.001* |
| Single term delivery (n) | 16 | 17 | 1.0 |
| Twin term delivery (n) | 4 | 1 | 0.20 |
| Preterm delivery (n) | 2 | 1 | 0.61 |
| Variable | Standard rFSH (n=372) | Letrozole + Late-rFSH (n=392) | p-value |
|---|---|---|---|
| Female Age (years) | 30.2 ± 5.0 | 29.0 ± 5.0 | 0.002* |
| BMI (kg/m²) | 25.72 ± 4.39 | 26.37 ± 4.46 | 0.037* |
| Duration of Infertility (months) | 49.2 ± 31.7 | 47.8 ± 34.0 | 0.209 |
| Endometrial Thickness (mm) | 9.36 ± 2.12 | 8.65 ± 2.15 | 0.001* |
| Sperm Total Motility (%) | 48.06 ± 18.6 | 47.12 ± 17.1 | 0.28 |
| Antral Follicle Count | 12 (8–18) | 17 (12–24) | 0.001* |
| Basal LH (mIU/mL) | 4.7 (3.1–7.4) | 5.6 (3.6–8.8) | 0.005* |
| Basal FSH (mIU/mL) | 6.2 (5.1–7.1) | 5.9 (4.8–6.9) | 0.001* |
| AMH (ng/mL) | 2.6 (1.1–4.5) | 4.3 (2.0–6.2) | <0.001* |
| E2 on day 2–3 (pg/mL) | 35 (22–54) | 36 (24–51) | 0.374 |
| Initial Dose of OI (IU) | 75 (50–112.5) | 50 (37.5–75) | <0.001* |
| Total Gonadotrophin Dose (IU) | 750 (450–1050) | 375 (225–600) | <0.001* |
| HCG trigger day | 10.72 ± 2.21 | 10.80 ± 2.14 | 0.60 |
| Follicles ≥17mm on hCG day | 1 (1–2) | 1 (1–2) | 0.021* |
| Follicles 14–17mm on hCG day | 0 (0–1) | 1 (0–2) | 0.107 |
| E2 on hCG day (pg/mL) | 172 (32-2810) | 280 (23-4577) | 0.001* |
| Variable | β coefficient | Adjusted OR | 95% CI | p-value |
|---|---|---|---|---|
| Female age (years) | -0.36 | 0.70 | 0.64–0.77 | <0.001 |
| Stimulation protocol (rFSH vs Letrozole + Late-rFSH) | 0.09 | 1.09 | 0.68–1.74 | 0.657 |
| AMH (ng/mL) | 0.06 | 1.06 | 0.96–1.17 | 0.252 |
| Endometrial thickness on hCG day (mm) | -0.11 | 0.90 | 0.78–1.02 | 0.095 |
| Number of follicles ≥17 mm | -0.09 | 0.91 | 0.61–1.35 | 0.626 |
| Analysis method | Sample size | Pregnancy rate – conventional rFSH | Pregnancy rate – letrozole + late rFSH | Risk difference (Let − rFSH) | Odds ratio (Let vs rFSH) | p-value |
|---|---|---|---|---|---|---|
| Unadjusted (crude) | 764 | 37/372 (9.9%) | 58/392 (14.8%) | +4.9 pp | 1.57 (1.01–2.44) | 0.042 |
| Propensity score matching (PSM) | 590 (295 matched pairs) | 33/295 (11.2%) | 45/295 (15.3%) | +4.1 pp (95% CI −1.6 to +9.7 pp) | 1.40 (0.88–2.24) | 0.195 |
| Inverse probability of treatment weighting (IPTW) | 764 (weighted) | 11.4% | 14.5% | +3.2 pp | 1.33 (0.87–2.03) | 0.194 |
| Doubly robust (IPTW + outcome regression) | 764 (weighted) | — | — | — | 1.42 (0.92–2.18) | 0.113 |
| PSM + GEE logistic regression | 590 (295 matched pairs) | — | — | — | 1.43 (0.87–2.35) | 0.158 |
| Analysis method | Sample size | Pregnancy rate – Conventional rFSH | Pregnancy rate – Letrozole + late rFSH | Risk difference (Let − rFSH) | Odds ratio (Let vs rFSH) | p-value |
|---|---|---|---|---|---|---|
| Unadjusted (crude) | 764 | 9.9 (37/372) | 14.8 (58/392) | +4.8 pp | 1.57 (1.01–2.44) | 0.044 |
| Propensity score matching (PSM) | 574 (287 pairs) | 11.1 (32/287) | 13.6 (39/287) | +2.4 pp | 1.24 (0.76–2.02) | 0.457 |
| Inverse probability of treatment weighting (IPTW) | 764 (weighted) | 11.4 | 14.5 | +3.2 pp | 1.33 (0.87–2.03) | 0.194 |
| IPTW + outcome regression (doubly robust) | 764 (weighted) | — | — | — | 1.42 (0.92–2.18) | 0.113 |
| PSM + GEE logistic regression | 574 (287 pairs) | — | — | — | 1.25 (0.74–2.12) | 0.386 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




