Submitted:
21 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
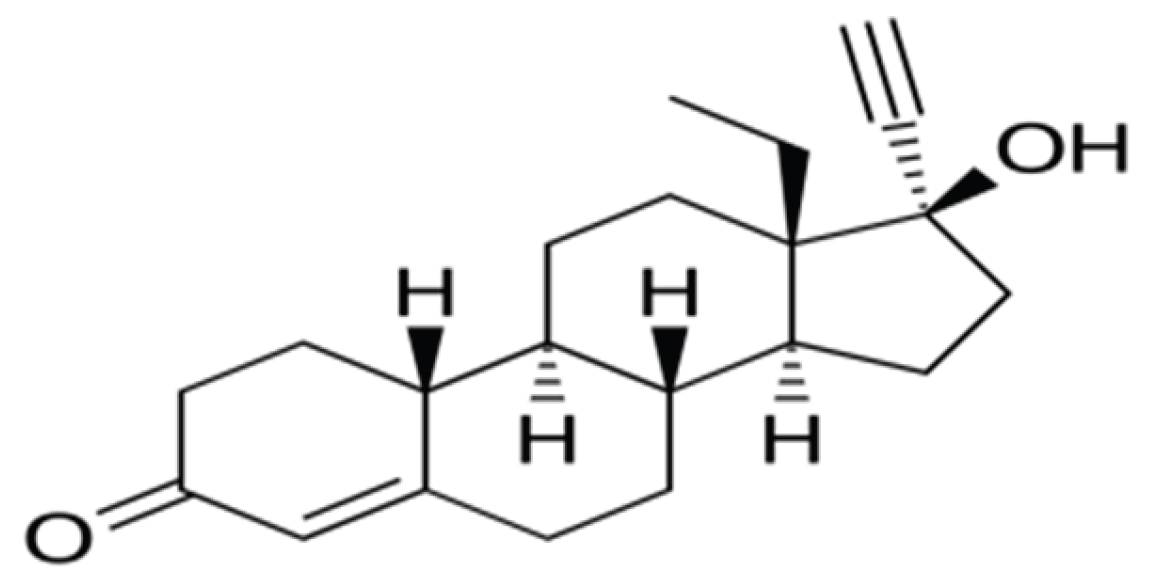
2.1. Chemicals and Reagents
2.2. LC–MS/MS Instrumentation
2.3. Calibration Standard and Quality Control Solution Preparation
2.4. Sample Preparation
2.5. Chromatographic Conditions
2.6. Mass Spectrometry Parameters
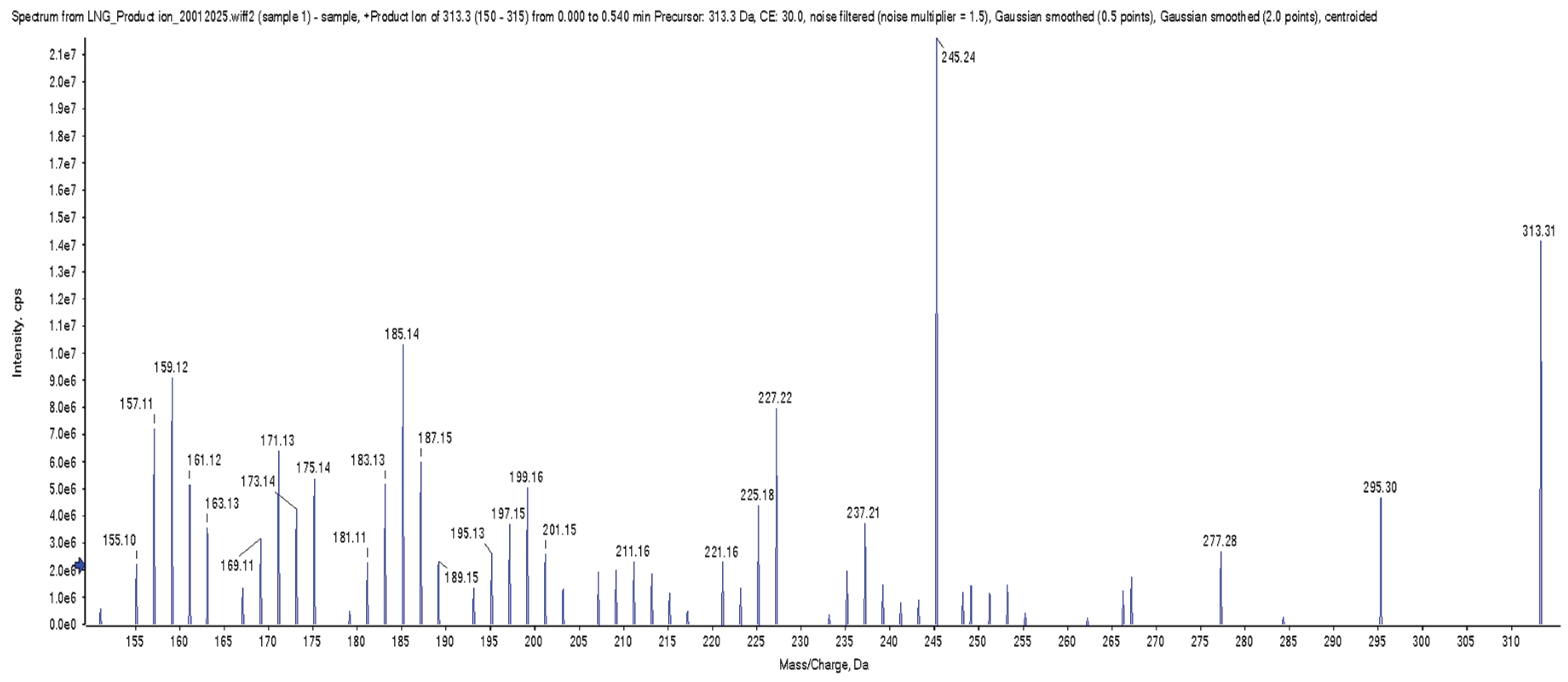
2.7. Collision-Induced Dissociation (CID)
2.8. Method Validation
2.8.1. Selectivity and Specificity
2.8.2. Calibration Curve and Linearity
2.8.3. Accuracy and Precision
2.9. Recovery and Matrix Effect
2.10. Stability Study
2.11. Clinical Application of the Validated Method
3. Results
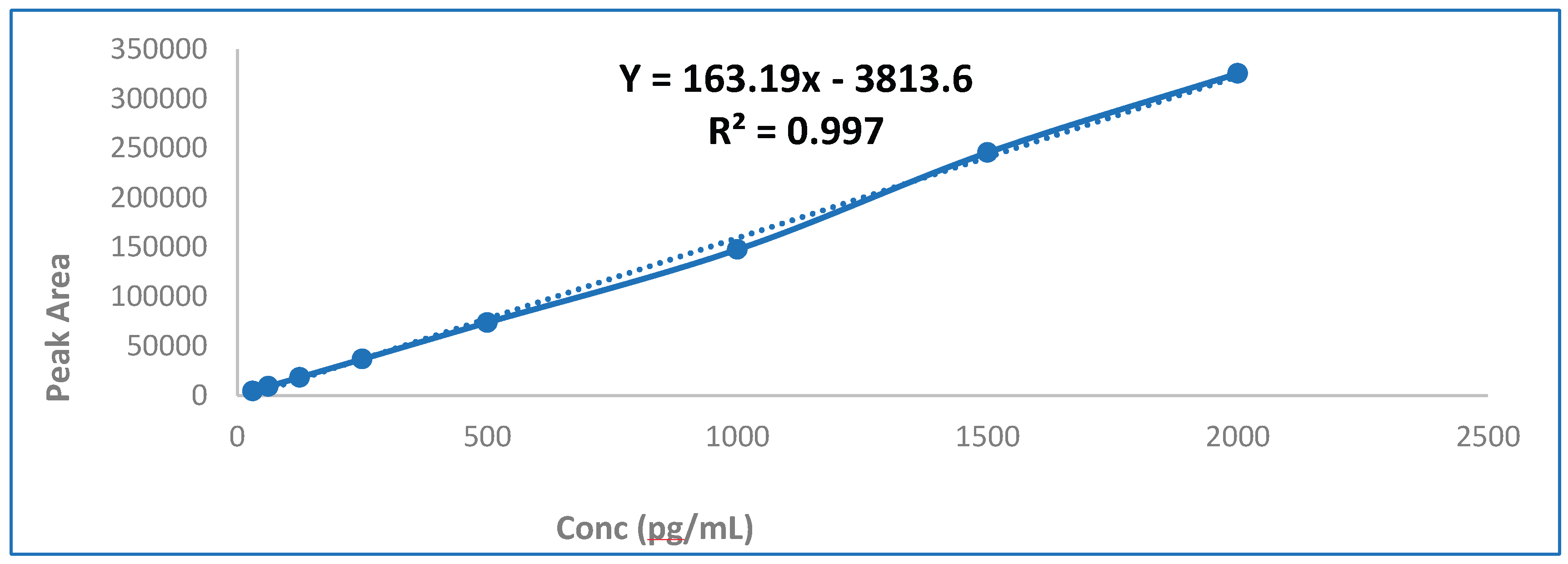
3.1. Linearity and Calibration Curve
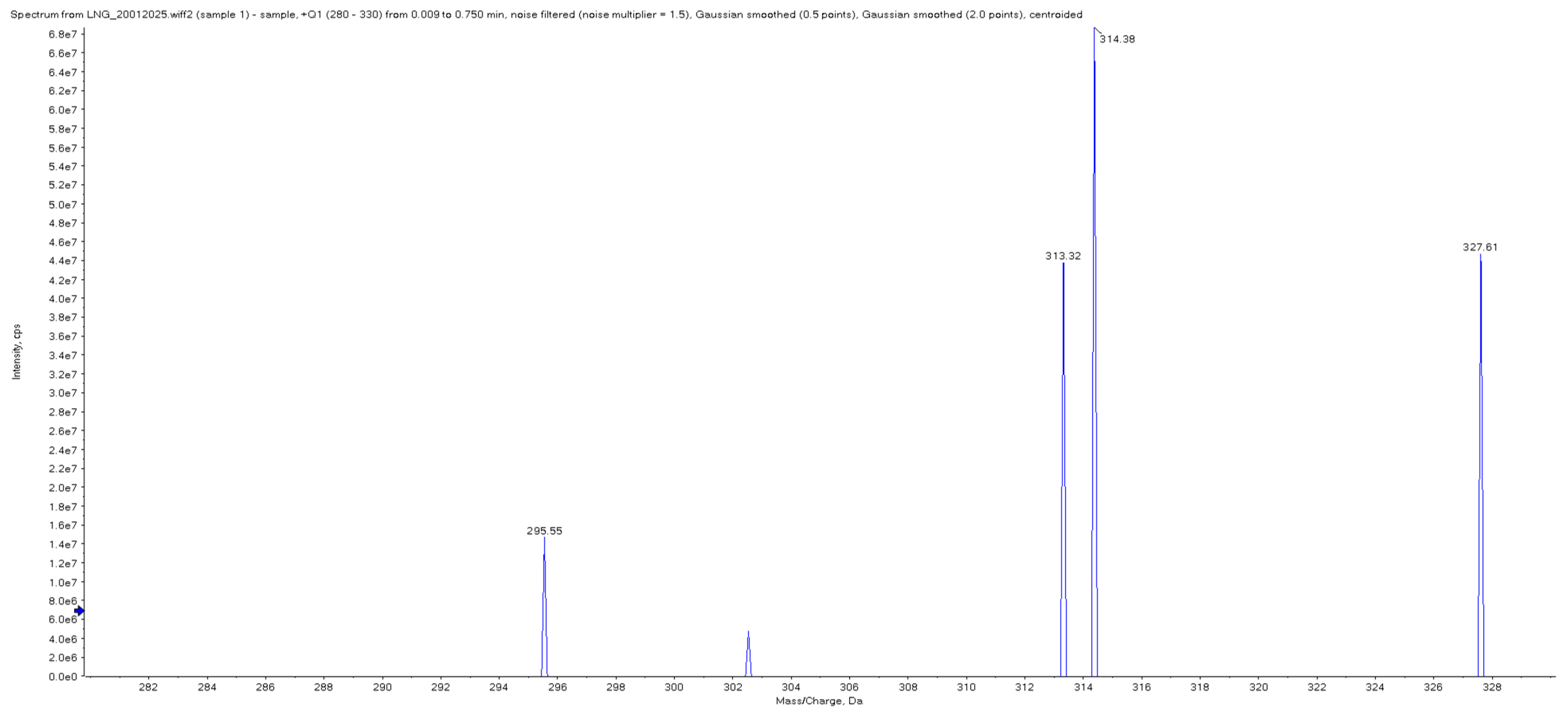
3.2. Selectivity and Specificity
3.3. Accuracy and Precision
3.4. Recovery and Matrix Effect
3.5. Stability Study
3.6. Clinical Application of the Validated Method
4. Discussion
5. Conclusions
Author Contributions
Funding
Ethics Statement
Acknowledgments
Conflicts of Interest
References
- Stefanović, O. D., 2017, 'Synergistic Activity of Antibiotics and Bioactive Plant Extracts: A Study Against Gram-Positive and Gram-Negative Bacteria', in S. Sahra (ed.), Bacterial Pathogenesis and Antibacterial Control, Intech Open, London. 10.5772/intechopen. 72026.
- Al Dalaty, A.; Gualeni, B.; Coulman, S.A.; Birchall, J.C. Models and methods to characterise levonorgestrel release from intradermally administered contraceptives. Drug Deliv. Transl. Res. 2021, 12, 335–349. [CrossRef]
- Sivin, I.; Nash, H.; Waldman, S. Jadelle® levonorgestrel rod implants: A summary of scientific data and lessons learned from programmatic experience; Population Council: New York, NY, United States, 2002; ISBN: .
- Rocca, M.L.; Palumbo, A.R.; Visconti, F.; Di Carlo, C. Safety and Benefits of Contraceptives Implants: A Systematic Review. Pharmaceuticals 2021, 14, 548. [CrossRef]
- Manoukian, O.S.; Arul, M.R.; Sardashti, N.; Stedman, T.; James, R.; Rudraiah, S.; Kumbar, S.G. Biodegradable polymeric injectable implants for long-term delivery of contraceptive drugs. J. Appl. Polym. Sci. 2017, 135. [CrossRef]
- Scarsi, K.K.; Darin, K.M.; Chappell, C.A.; Nitz, S.M.; Lamorde, M. Drug–Drug Interactions, Effectiveness, and Safety of Hormonal Contraceptives in Women Living with HIV. Drug Saf. 2016, 39, 1053–1072. [CrossRef]
- Steinauer, J.E.; Upadhyay, U.D.; Sokoloff, A.; Harper, C.C.; Diedrich, J.T.; Drey, E.A. Choice of the levonorgestrel intrauterine device, etonogestrel implant or depot medroxyprogesterone acetate for contraception after aspiration abortion. Contraception 2015, 92, 553–559. [CrossRef]
- Groskaufmanis, L.; Masho, S.W. Source of care and variation in long-acting reversible contraception use. Fertil. Steril. 2016, 105, 401–409. [CrossRef]
- Conde-Agudelo, A.; Belizán, J.M.; Lammers, C. Maternal-perinatal morbidity and mortality associated with adolescent pregnancy in Latin America: Cross-sectional study. Am. J. Obstet. Gynecol. 2005, 192, 342–349. [CrossRef]
- Gunardi. Monoplant the Indonesian implant: the overview of implant and its development. Indones J Obstet Gynecol. 2011; 35:35–45.
- Patton, G.C.; Coffey, C.; Sawyer, S.M.; Viner, R.M.; Haller, D.; Bose, K.; Vos, T.; Ferguson, J.; Mathers, C.D. Global patterns of mortality in young people: a systematic analysis of population health data. Lancet 2009, 374, 881–892. [CrossRef]
- Stover, J.; Ross, J. How Increased Contraceptive Use has Reduced Maternal Mortality. Matern. Child Heal. J. 2009, 14, 687–695. [CrossRef]
- Weisberg, E.; Bateson, D.; McGeechan, K.; Mohapatra, L. A three-year comparative study of continuation rates, bleeding patterns and satisfaction in Australian women using a subdermal contraceptive implant or progestogen releasing-intrauterine system. Eur. J. Contracept. Reprod. Heal. Care 2013, 19, 5–14. [CrossRef]
- Modesto, W.; Bahamondes, M.V.; Bahamondes, L. A randomized clinical trial of the effect of intensive versus non-intensive counselling on discontinuation rates due to bleeding disturbances of three long-acting reversible contraceptives. Hum. Reprod. 2014, 29, 1393–1399. [CrossRef]
- Phillips, S.J.; Tepper, N.K.; Kapp, N.; Nanda, K.; Temmerman, M.; Curtis, K.M. Progestogen-only contraceptive use among breastfeeding women: a systematic review. Contraception 2016, 94, 226–252. [CrossRef]
- Callahan, R.L.; Taylor, D.; Jenkins, D.W.; Owen, D.H.; Cheng, L.; Cancel, A.M.; Dorflinger, L.J.; Steiner, M.J. In vivo release of levonorgestrel from Sino-implant (II) — an innovative comparison of explant data. Contraception 2015, 92, 350–355. [CrossRef]
- Wathoni, N.; Alfauziah, T.Q.; Rantika, N. EVOLUTION OF CONTRACEPTIVE IMPLANTS: A REVIEW. Int. J. Appl. Pharm. 2018, 10, 16–22. [CrossRef]
- Fuchs, R.; Taylor, D.; Jenkins, D.W.; Brache, V.; Luo, D.; Dorflinger, L.J.; Steiner, M.J. Levonorgestrel release rates measured through analysis of two-rod contraceptive explants. Contraception: X 2020, 2. [CrossRef]
- Berzas, J.J.; Rodríguez, J.; Castañeda, G. Simultaneous Determination of Ethinylestradiol and Levonorgestrel in Oral Contraceptives by Derivative Spectrophotometry. Anal. 1997, 122, 41–44. [CrossRef]
- Theron, H.; Coetzee, C.; Sutherland, F.; Wiesner, J.; Swart, K. Selective and sensitive liquid chromatography–tandem mass spectrometry method for the determination of levonorgestrel in human plasma. J. Chromatogr. B 2004, 813, 331–336. [CrossRef]
- Wang, R.; Tian, Y.; Zhang, L.; Zhang, Z. Simultaneous Determination of Levonorgestrel and Two Endogenous Sex Hormones in Human Plasma Based on LC–MS/MS. Bioanalysis 2016, 8, 1133–1144. [CrossRef]
- Zhong, G.; Bi, H.; Zhou, S.; Chen, X.; Huang, M. Simultaneous determination of metformin and gliclazide in human plasma by liquid chromatography–tandem mass spectrometry: application to a bioequivalence study of two formulations in healthy volunteers. J. Mass Spectrom. 2005, 40, 1462–1471. [CrossRef]
- Cirrincione, L.R.; Penchala, S.D.; Scarsi, K.K.; Podany, A.T.; Winchester, L.C.; Back, D.J.; Khoo, S.H.; Fletcher, C.V.; Siccardi, M.; Else, L.J. Development, validation and utilization of a highly sensitive LC-MS/MS method for quantification of levonorgestrel released from a subdermal implant in human plasma. J. Chromatogr. B 2018, 1084, 106–112. [CrossRef]
- Veeran, M.G.; C., K.; B., B.; Painuly, D.; Aprem, A.S. RP-HPLC method validation for fast extraction and quantification of Levonorgestrel drug from silicone based intrauterine device intended for in-process and finished formulation. DARU J. Pharm. Sci. 2021, 29, 185–193. [CrossRef]
- Midhu GV, Karthikeyan C, Bakkiyaraja B, Painuly D, Abi SA. RP-HPLC method validation for fast extraction and quantification of Levonorgestrel drug from silicone based intrauterine device intended for in-process and finished formulation. DARU Journal of Pharmaceutical Sciences. 2021.185-193.
- Pal, V.K.; Pal, Y. ANALYTICAL METHOD DEVELOPMENT AND METHOD VALIDATION FOR DETERMINATION ASSAY AND CONTENT UNIFORMITY OF LEVONORGESTREL BY REVERSED-PHASE HIGHPERFORMANCE LIQUID CHROMATOGRAPHY. Asian J. Pharm. Clin. Res. 2020, 101–107. [CrossRef]
- Patel PU, Patal BM. Development and Validation of Stability Indicating Reverse Phase High Performance Liquid Chromatographic Method for Estimation of Levonorgestrel in Bulk Dosage Form. International Journal of Pharmaceutics and Drug Analysis. 2014; 2(4): 311-318.
- Chang, H.-F.; Wang, J.-Q.; Wang, B.; Deng, A.-P. An immunochromatographic assay for rapid and simultaneous detection of levonorgestrel and methylprednisolone in water samples. Chin. Chem. Lett. 2013, 24, 937–940. [CrossRef]
- Lee, J.H.; Na Park, H.; Kim, N.S.; Park, H.-J.; Park, S.; Shin, D.; Kang, H. Detection of Illegal Abortion-Induced Drugs Using Rapid and Simultaneous Method for the Determination of Abortion-Induced Compounds by LC–MS/MS. Chromatographia 2019, 82, 1365–1371. [CrossRef]
- Ezebialu, I.; Okafo, O.; Oringanje, C.; Ogbonna, U.; Udoh, E.; Odey, F.; Meremikwu, M.M. Surgical and nonsurgical interventions for vulvar and clitoral pain in girls and women living with female genital mutilation: A systematic review. Int. J. Gynecol. Obstet. 2017, 136, 34–37. [CrossRef]
- Bioanalytical Method Validation. Available online: https://www.ema.europa.eu/en/bioanalytical-method-validation#current-effective-version-section (accessed on 24 February 2021).
- US Food and Drug Administration. Bioanalytical Method Validation, Guidance for Industry. US Department of Health and Human Services, Center for Drug Evaluation and Research. 2018. Available online: https://www.fda.gov/media/70858/download (accessed on).
- Smith R, Taylor PJ. Electrospray ionization in bioanalytical mass spectrometry: advances and applications. J Chromatogr B. 2014;960:1–15.
- Xu Y, Wu W, Williams TD. Atmospheric pressure ionization for steroid analysis: comparison of APCI and ESI. Rapid Commun Mass Spectrom. 2017;31(10):843–852.
- Shah, V.P.; Midha, K.K.; Findlay, J.W.A.; Hill, H.M.; Hulse, J.D.; McGilveray, I.J.; McKay, G.; Miller, K.J.; Patnaik, R.N.; Powell, M.L.; et al. Bioanalytical Method Validation—A Revisit with a Decade of Progress. Pharm. Res. 2000, 17, 1551–1557. [CrossRef]
- US FDA. Bioanalytical Method Validation Guidance for Industry. Food and Drug Administration, 2018.
- European Medicines Agency (EMA). Guideline on Bioanalytical Method Validation. EMA, 2011.
- Peters FT, Drummer OH, Musshoff F. Validation of new methods. Forensic Sci Int. 2007;165(2–3):216–224.
- Taylor, P.J. Matrix effects: the Achilles heel of quantitative high-performance liquid chromatography–electrospray–tandem mass spectrometry. Clin. Biochem. 2005, 38, 328–334. [CrossRef]
- Viswanathan CT, Bansal S, Booth B, et al. Workshop/conference report: quantitative bioanalytical methods validation and implementation. AAPS J. 2007;9(1):E30–E42.
- Diaz S, Pavez M, Moo-Young AJ, et al. Clinical performance of levonorgestrel-releasing contraceptive implants: a pharmacokinetic and pharmacodynamic perspective. Contraception. 2006;74(1):59–67.
- Fraser IS, Weisberg E. A comprehensive review of levonorgestrel contraceptive implants. Contraception. 2011;84(5):402–417.
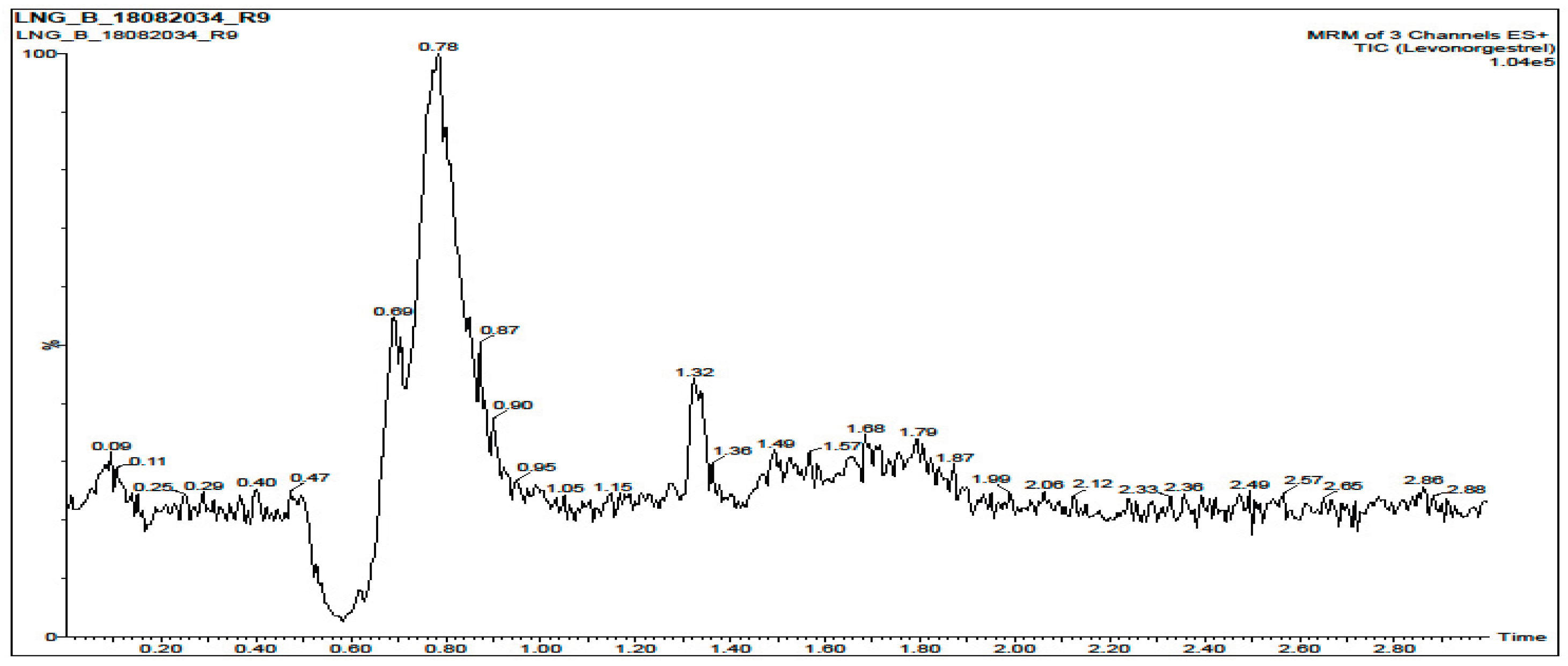
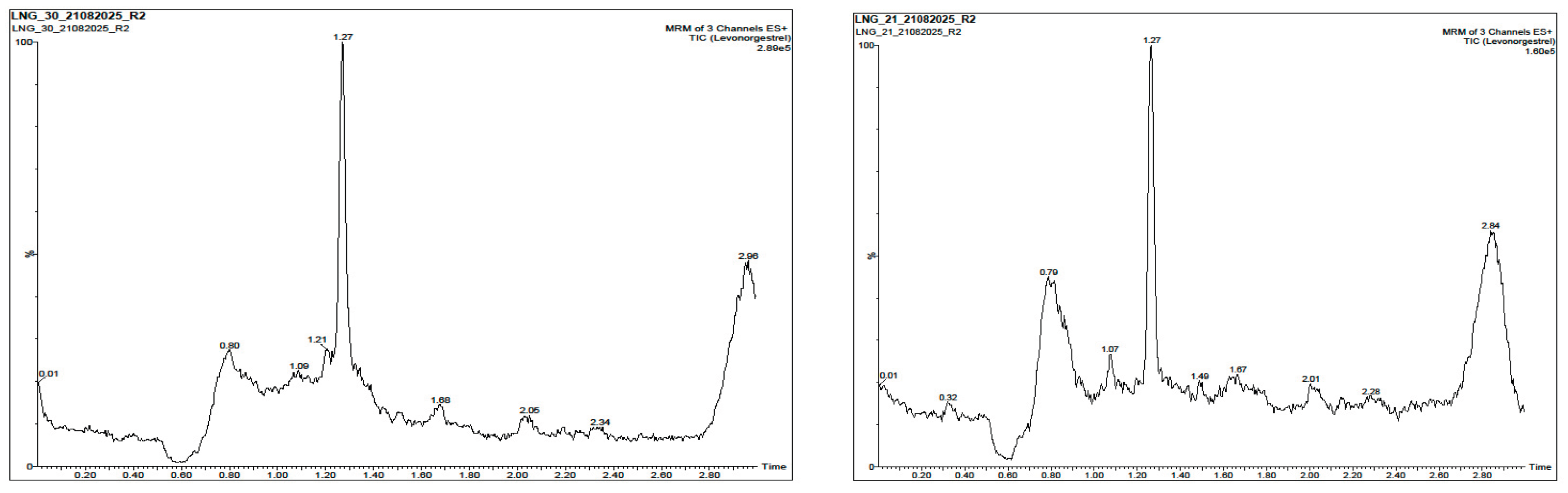
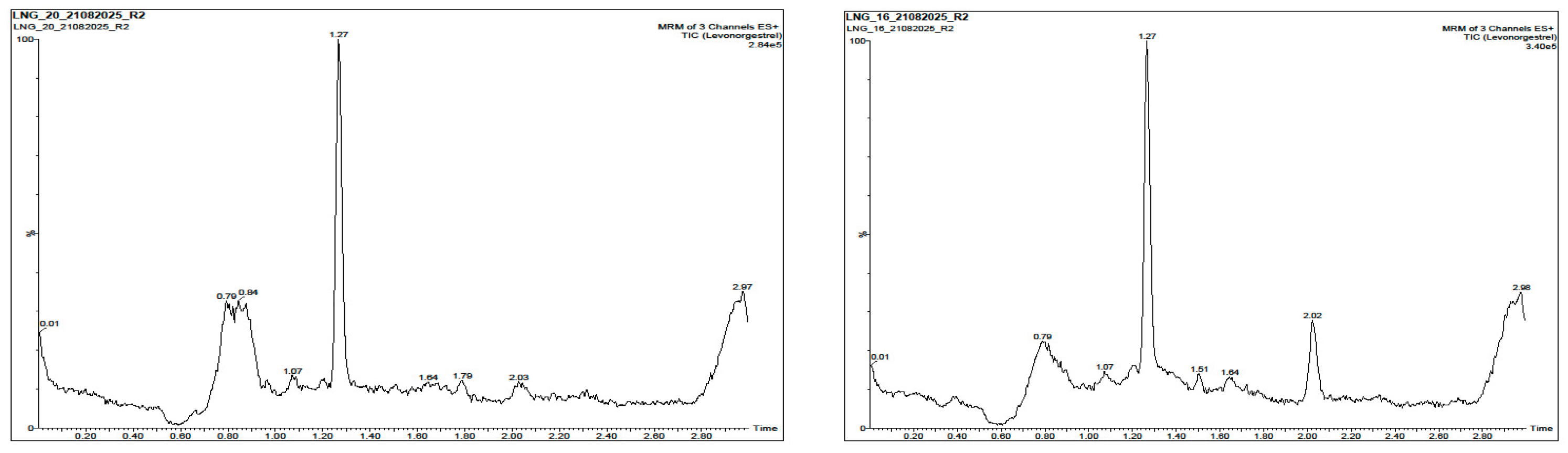
| Sample |
Parent ion (m/z) |
Product ion (m/z) |
Cone voltage (V) |
Collision Energy (V) |
| LNG | 312.985 | 81.468 | 72 | 40 |
| LNG | 312.985 | 90.921 | 72 | 42 |
| LNG | 312.985 | 108.963 | 72 | 24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




