Submitted:
15 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The CAR and Its Evolution, “Hijacking” Cytotoxic T-Cell Capacities to Eliminate Cellular Targets
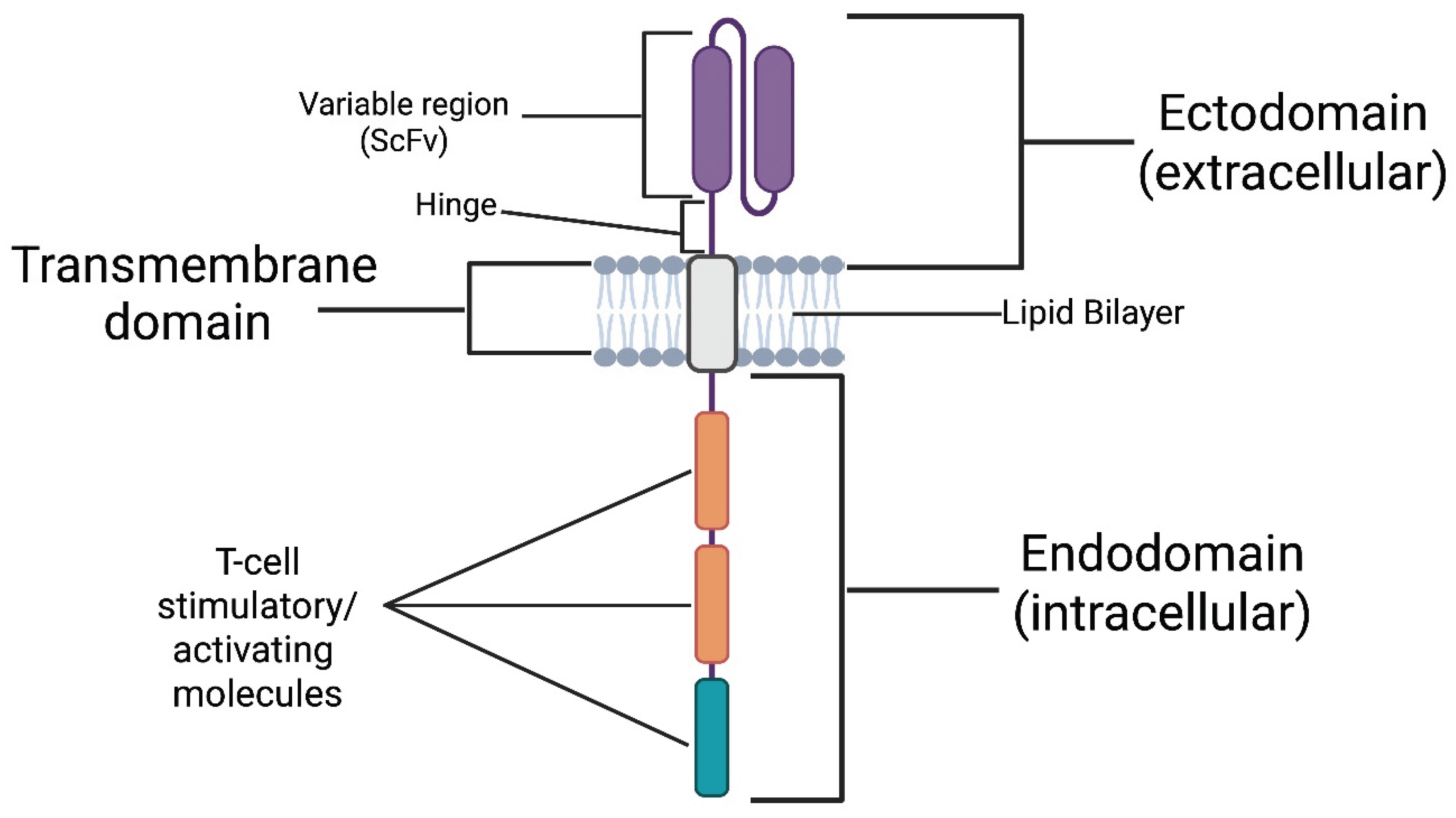
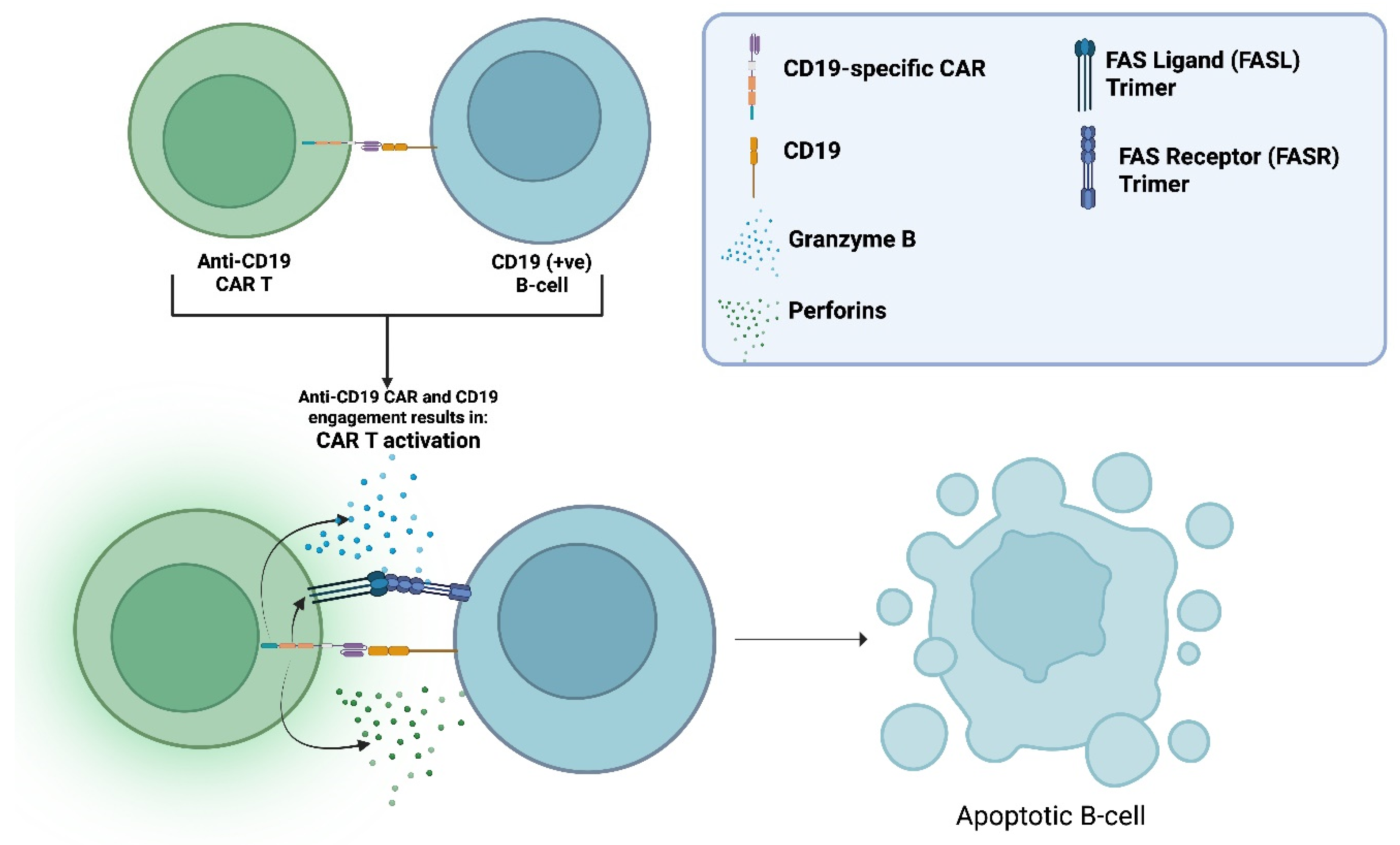
2.1. The Chimeric Antigen Receptor (CAR)
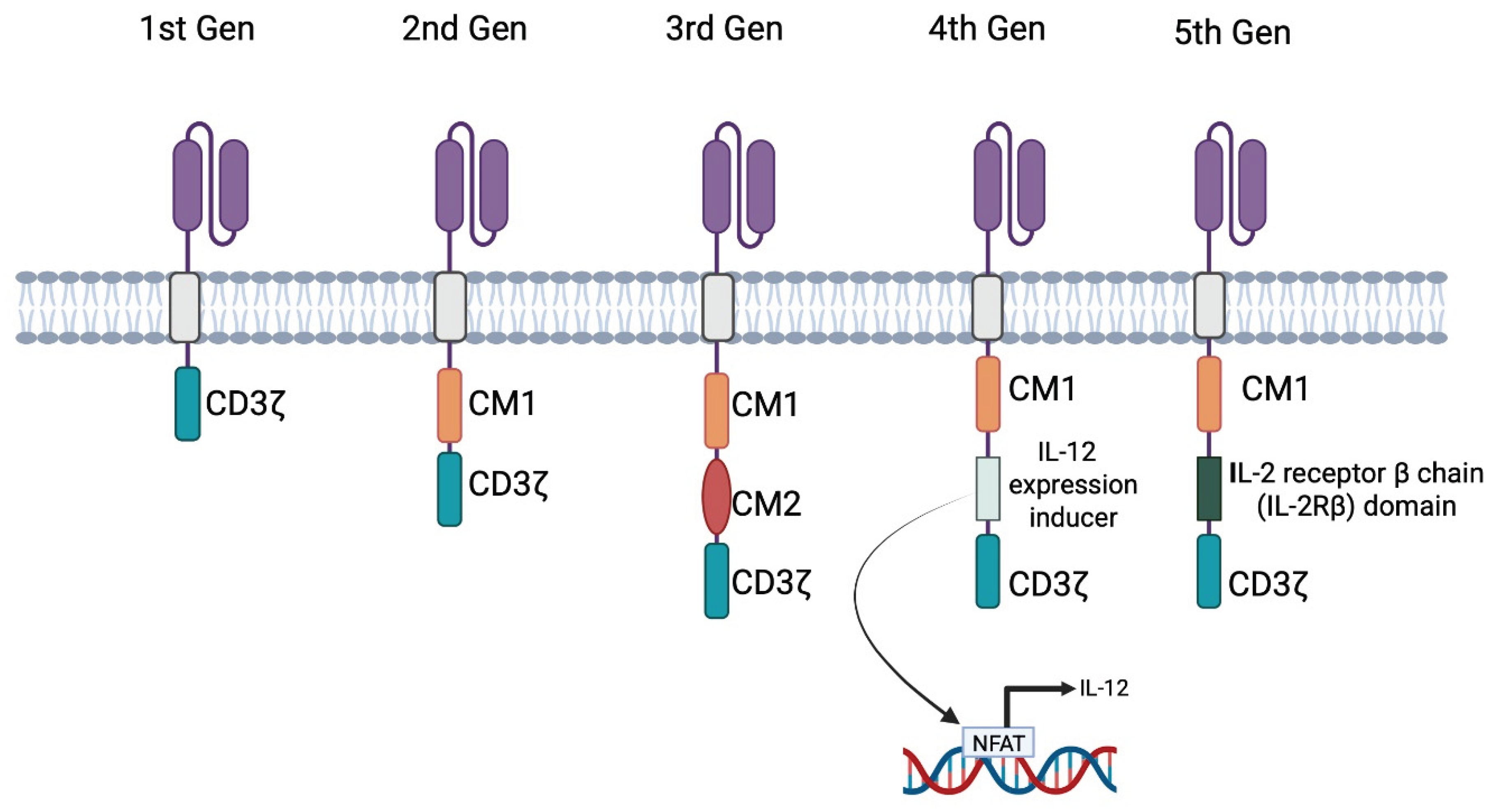
2.2. The History and the Different Generations of CARs
2.2.1. 1st Generation
2.2.2. 2nd Generation
2.2.3. 3rd to 5th Generation
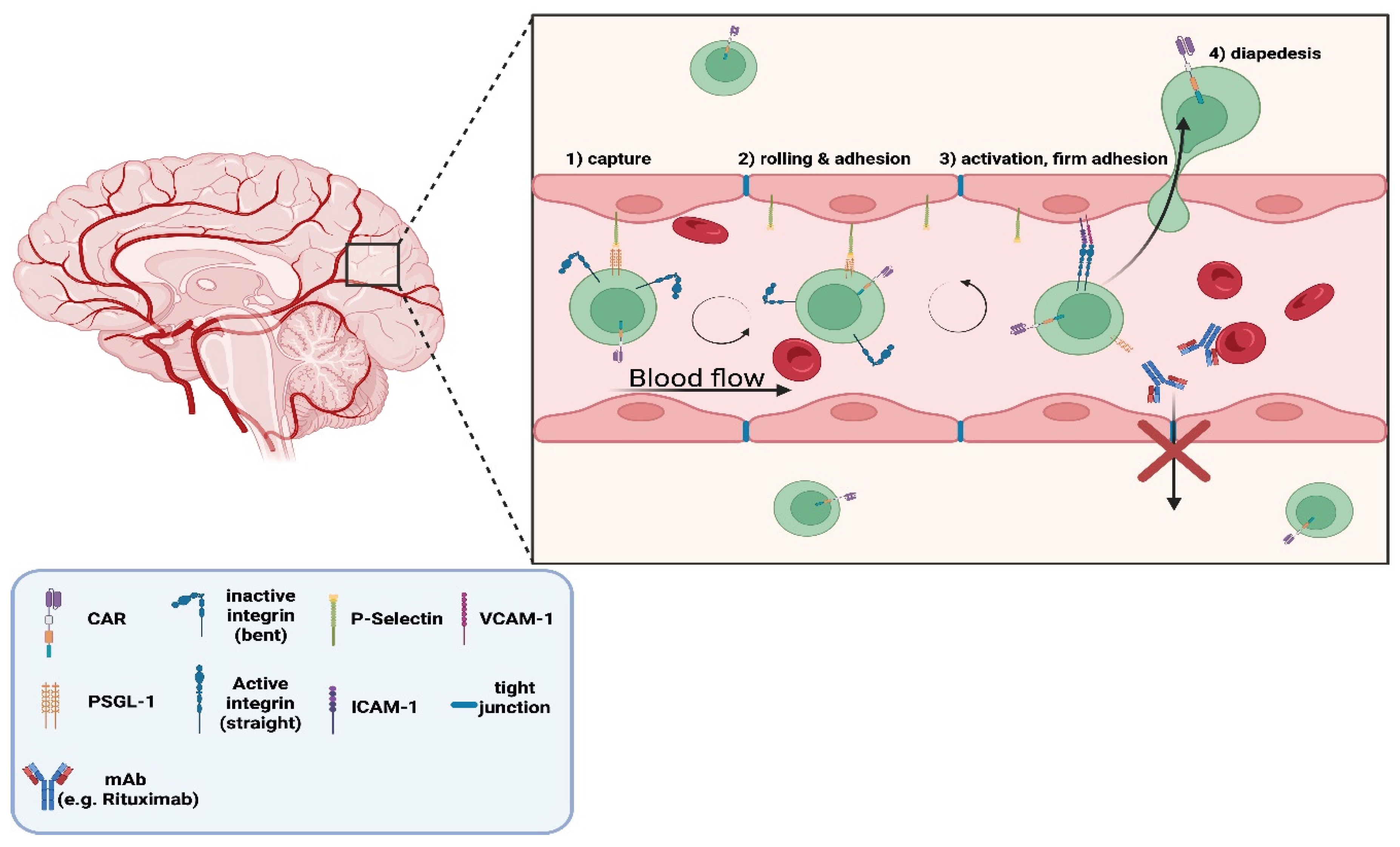
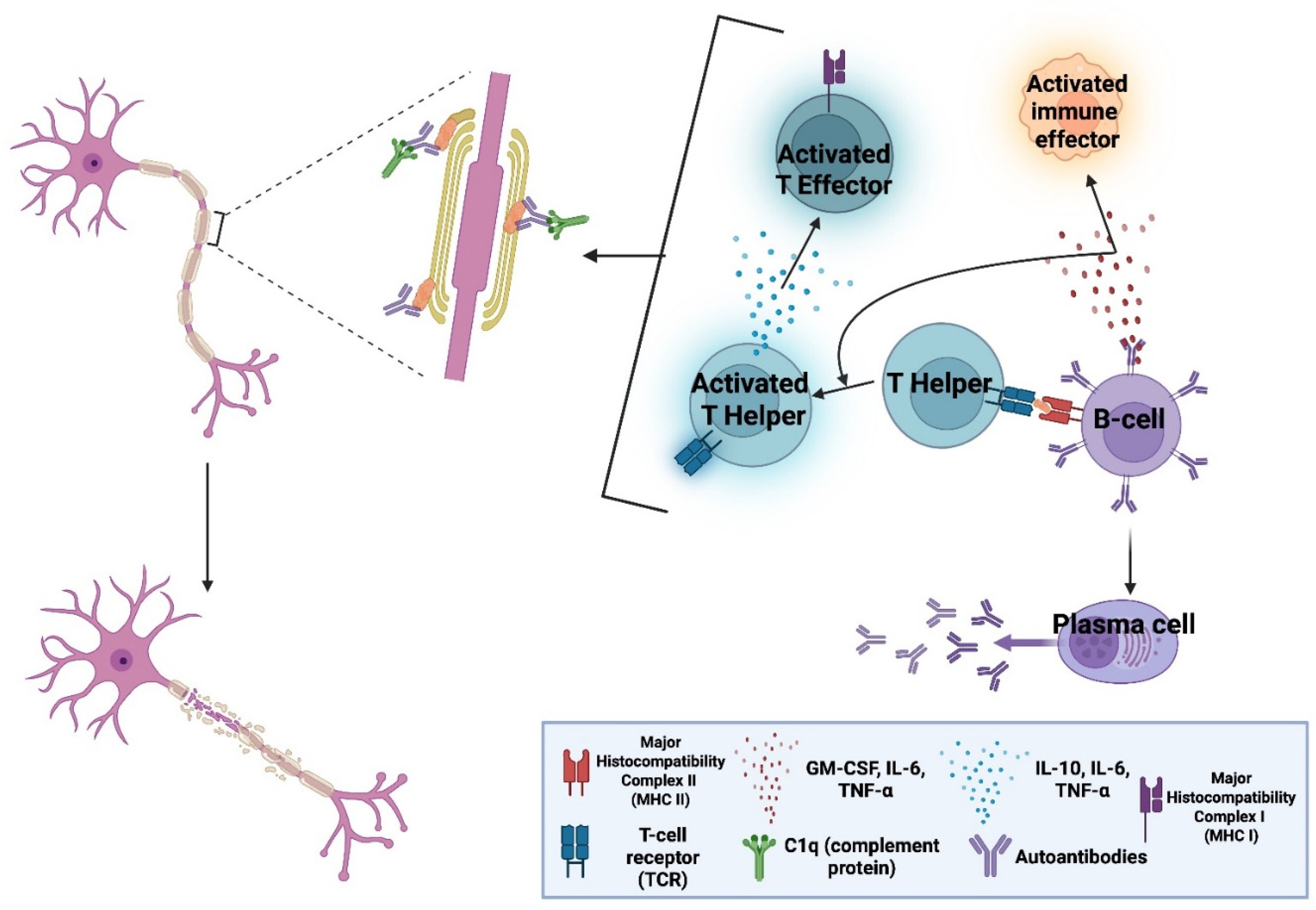
3. From mAbs to CAR T-Cells: Overcoming Immune-Access Limitations of mAbs and Compartmentalisation Barriers in CNS Autoimmunity
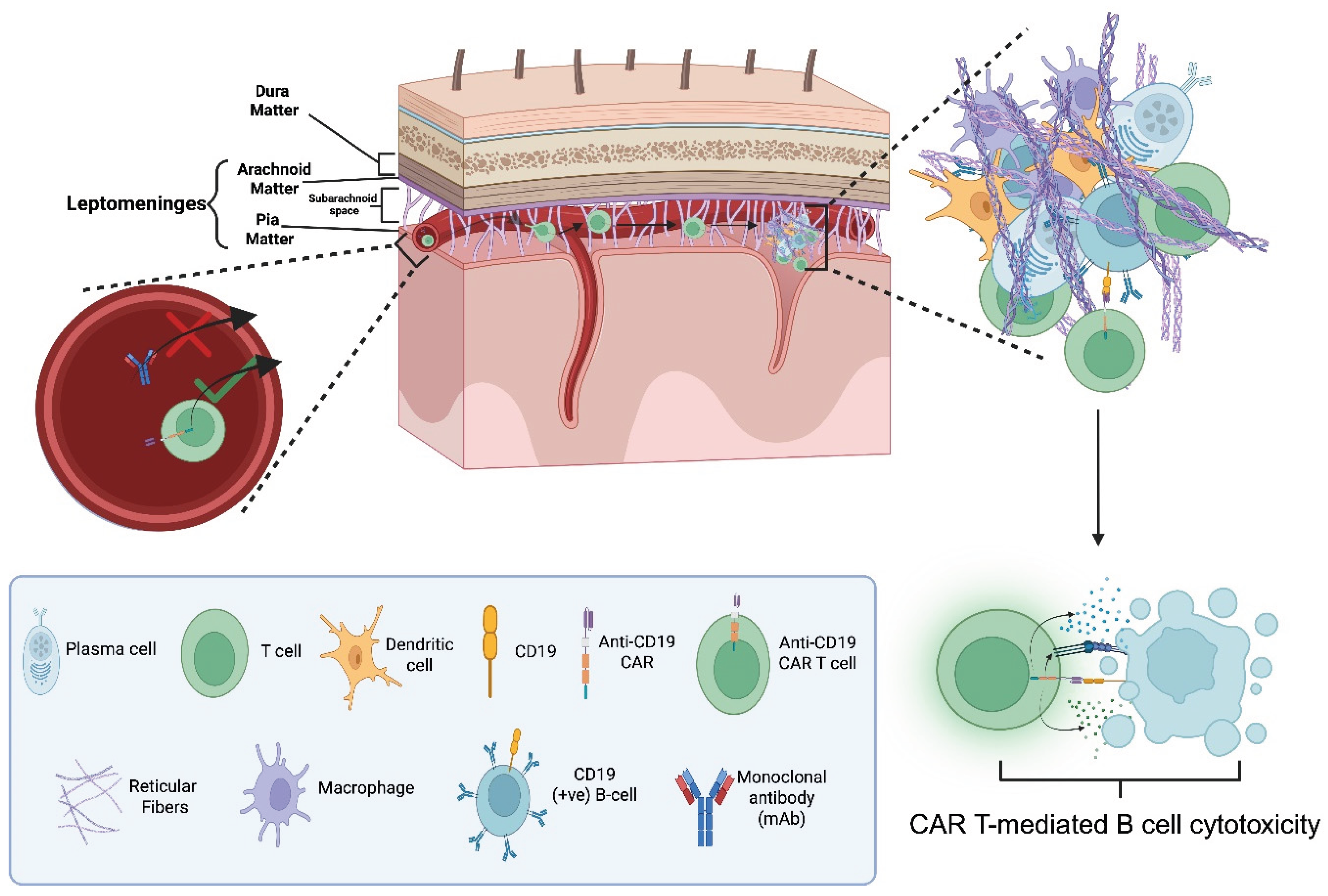
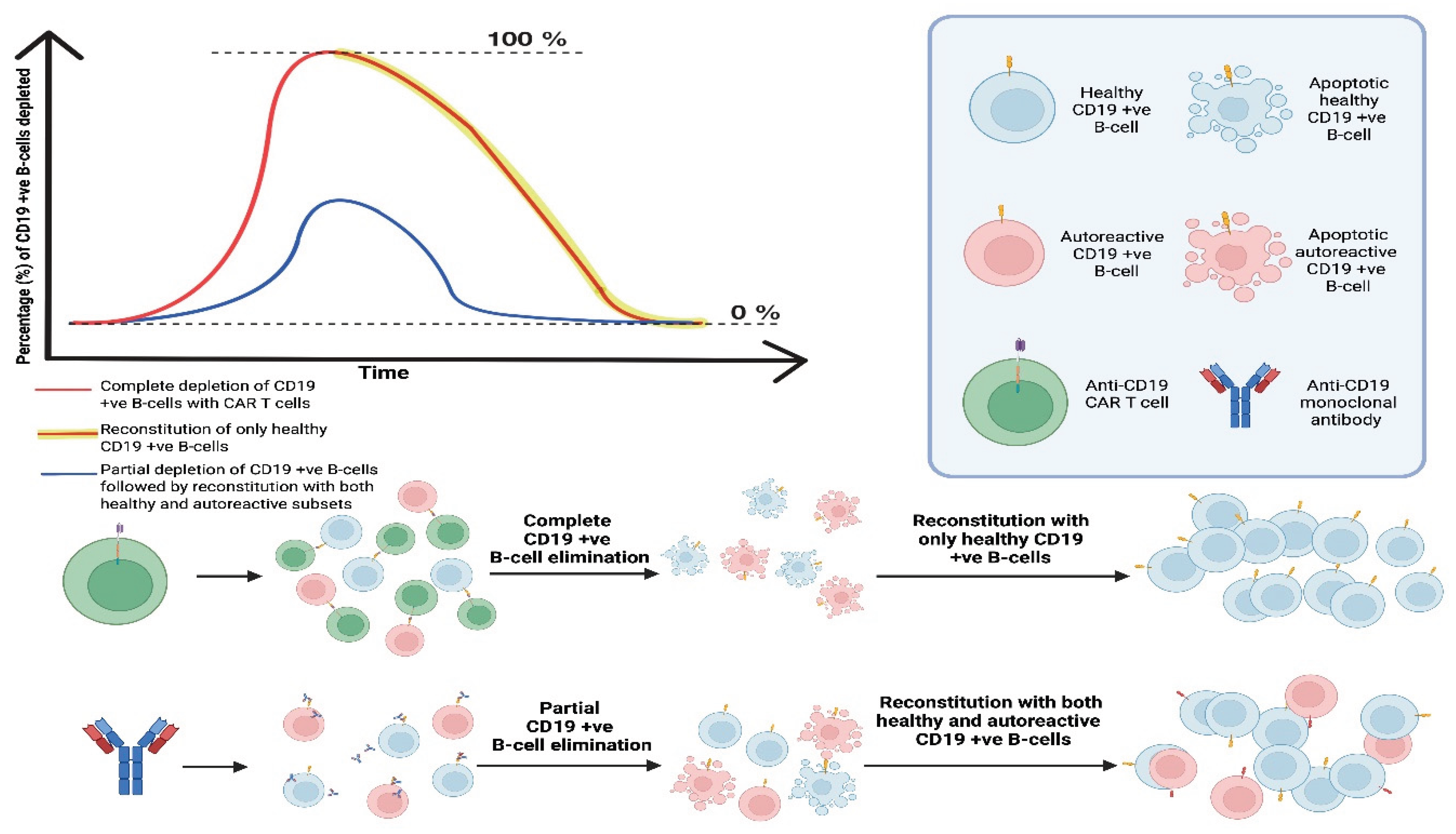
4. Could CAR T-Cell Therapies Achieve an “Immune Reset” to Cure CNS Autoimmunity?
4. Adverse Side-Effects Associated with CAR T-Cell Therapies and a Way to “Switch Off” Their Activity as a Safety Measure
4.1. Adverse Side-Effects
4.2. iCasp9 Suicide System [42]
5. The Plasticity in Different CAR T-Cell Constructs
5.1. Allogeneic CAR T-Cells
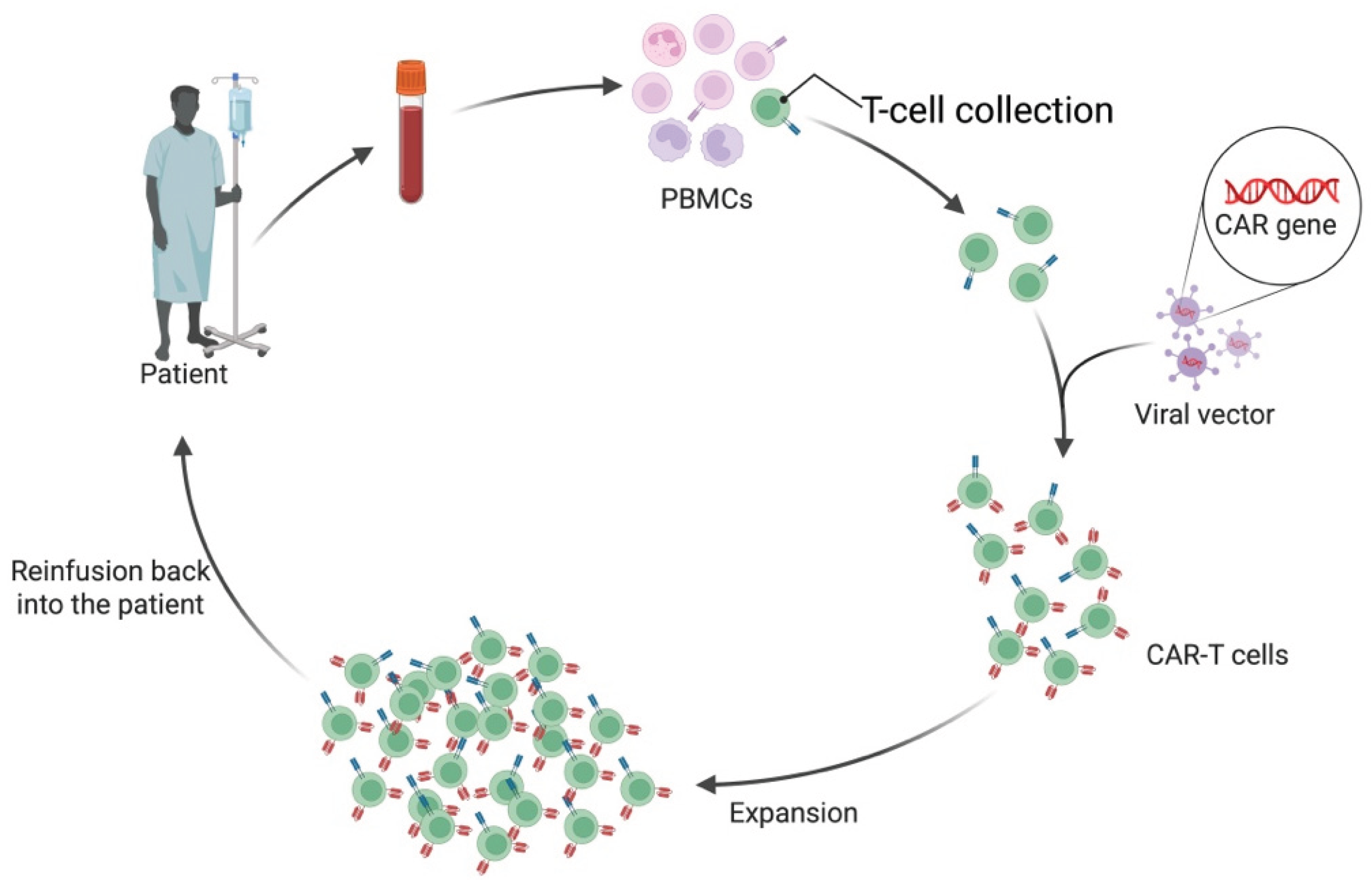
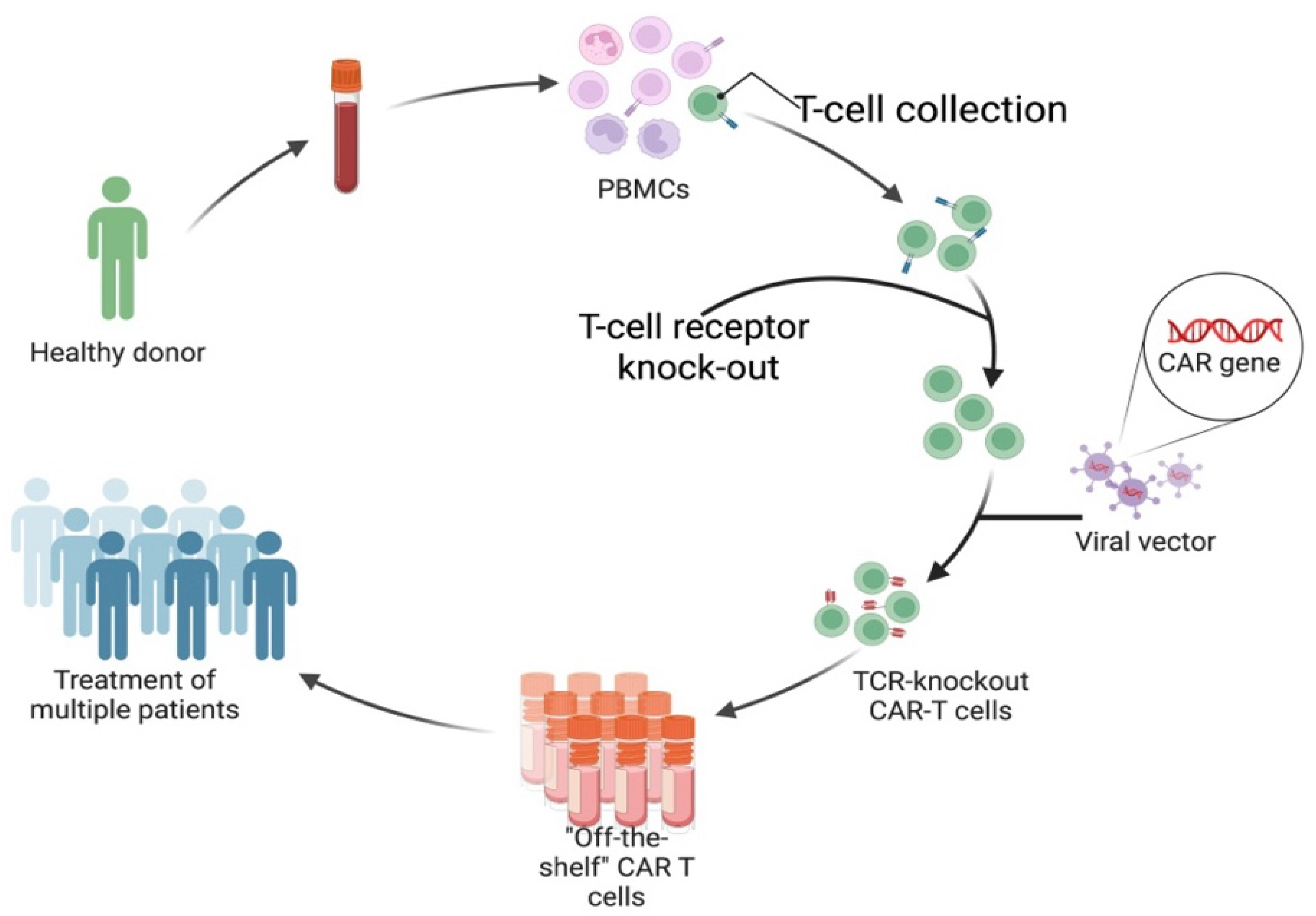
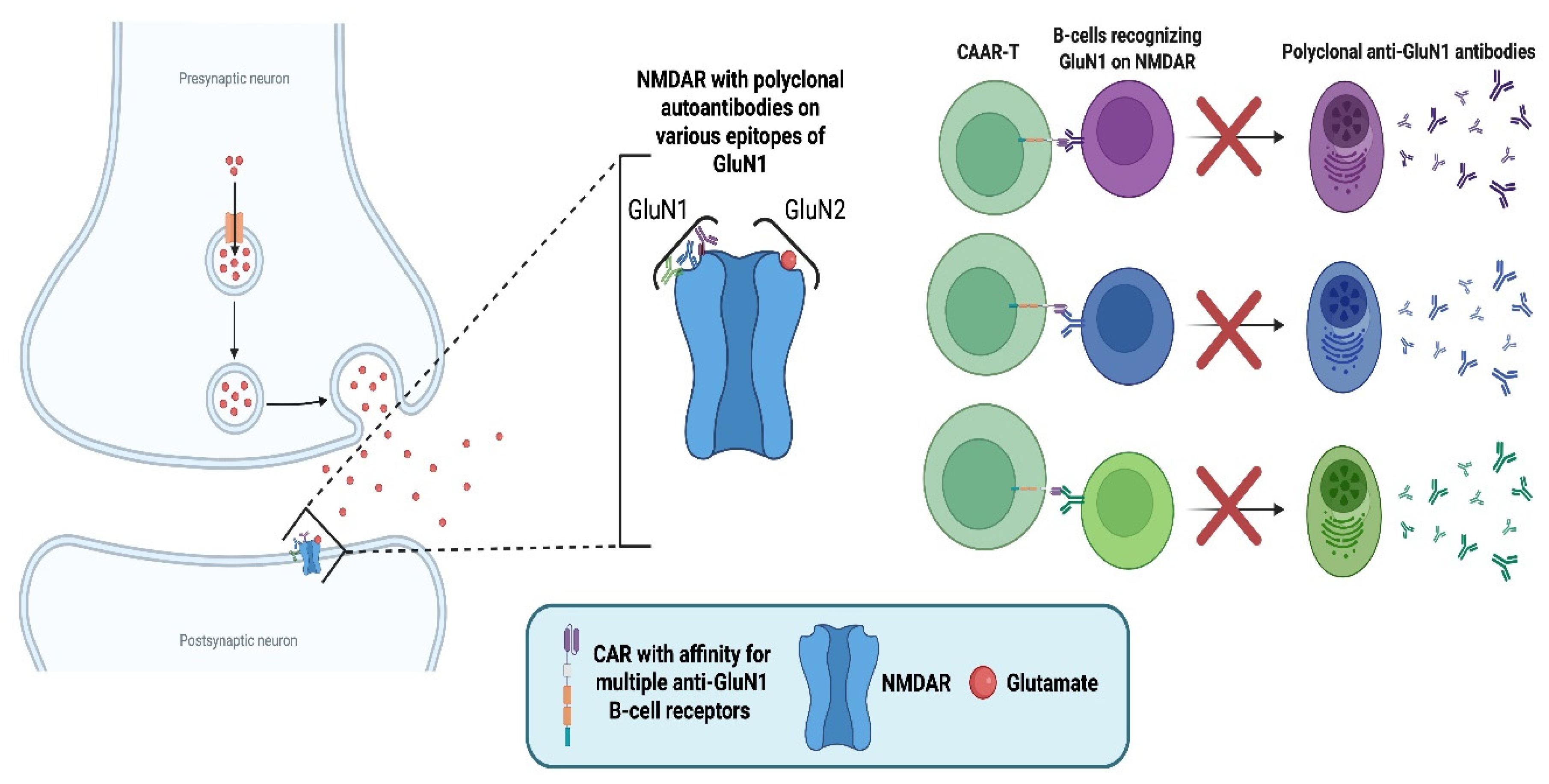
5.2. Anti-N-methyl-D-aspartate Receptor (NMDAR)-Specific Chimeric Autoantibody Receptor (CAAR) T-Cells for NMDAR Autoimmune Encephalitis (AE)
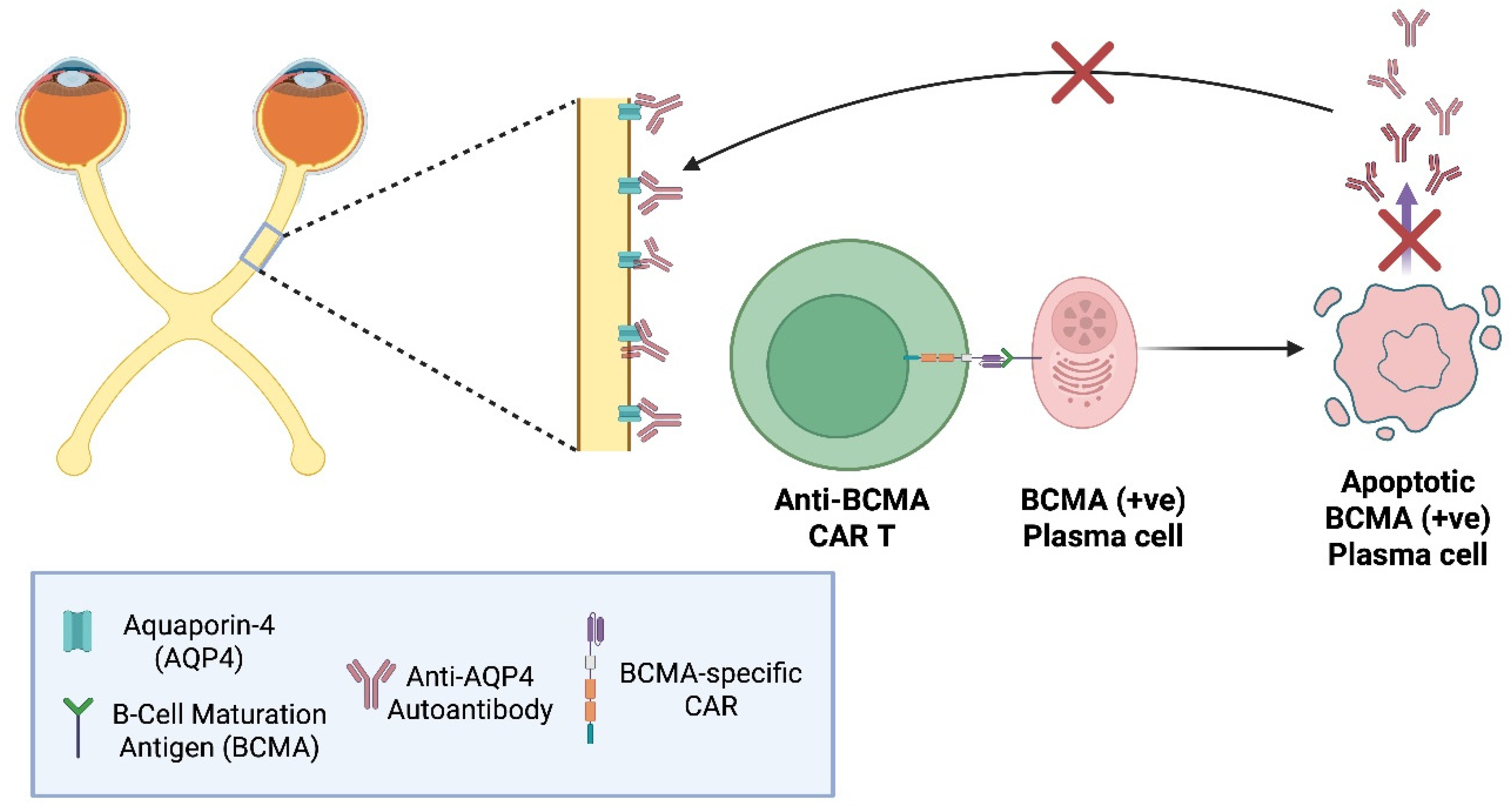
5.3. Anti-BCMA CAR T-Cells in NMOSD
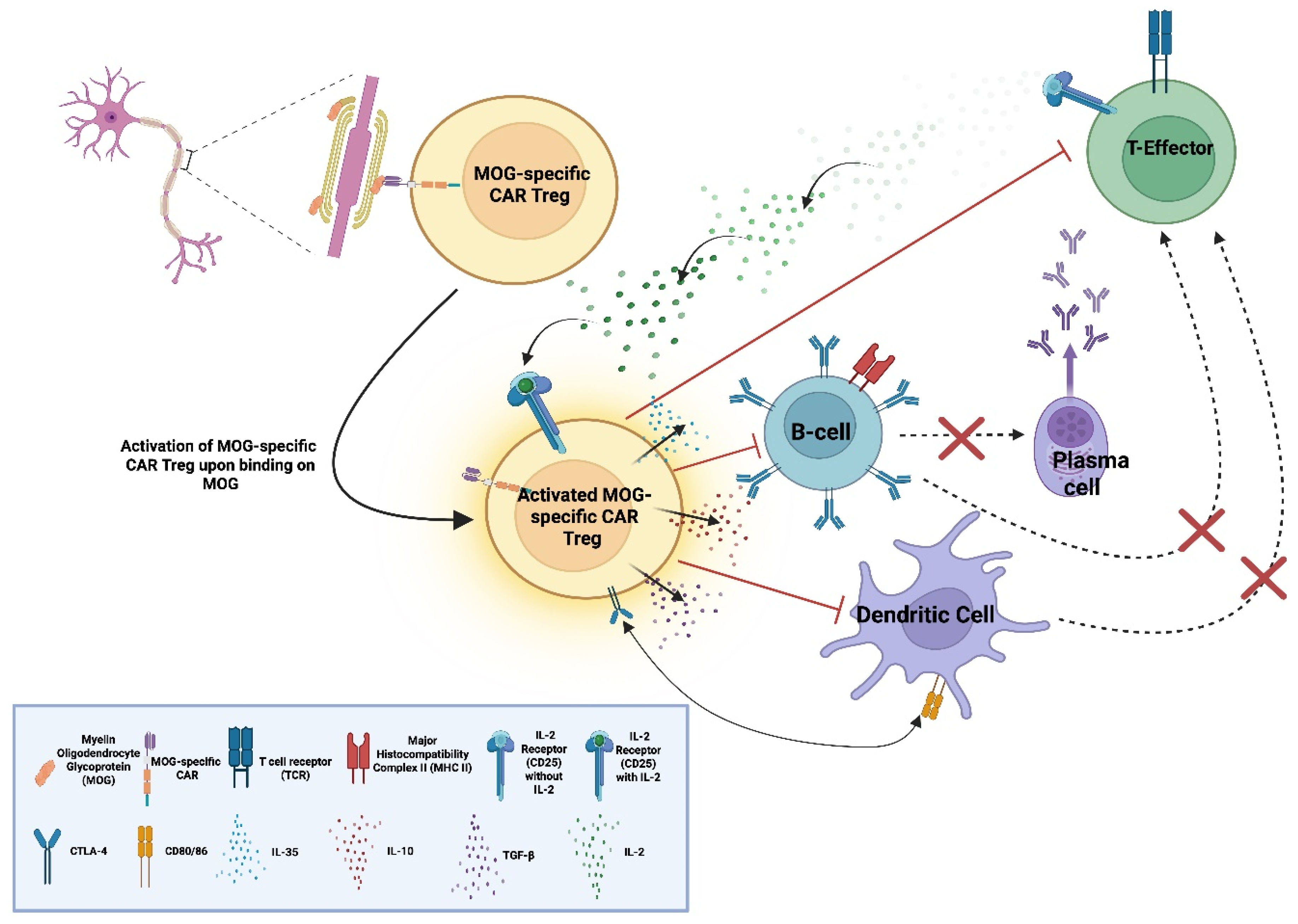
5.4. MOG-Specific CAR T-Regulatory Cells (CAR Tregs) in MS
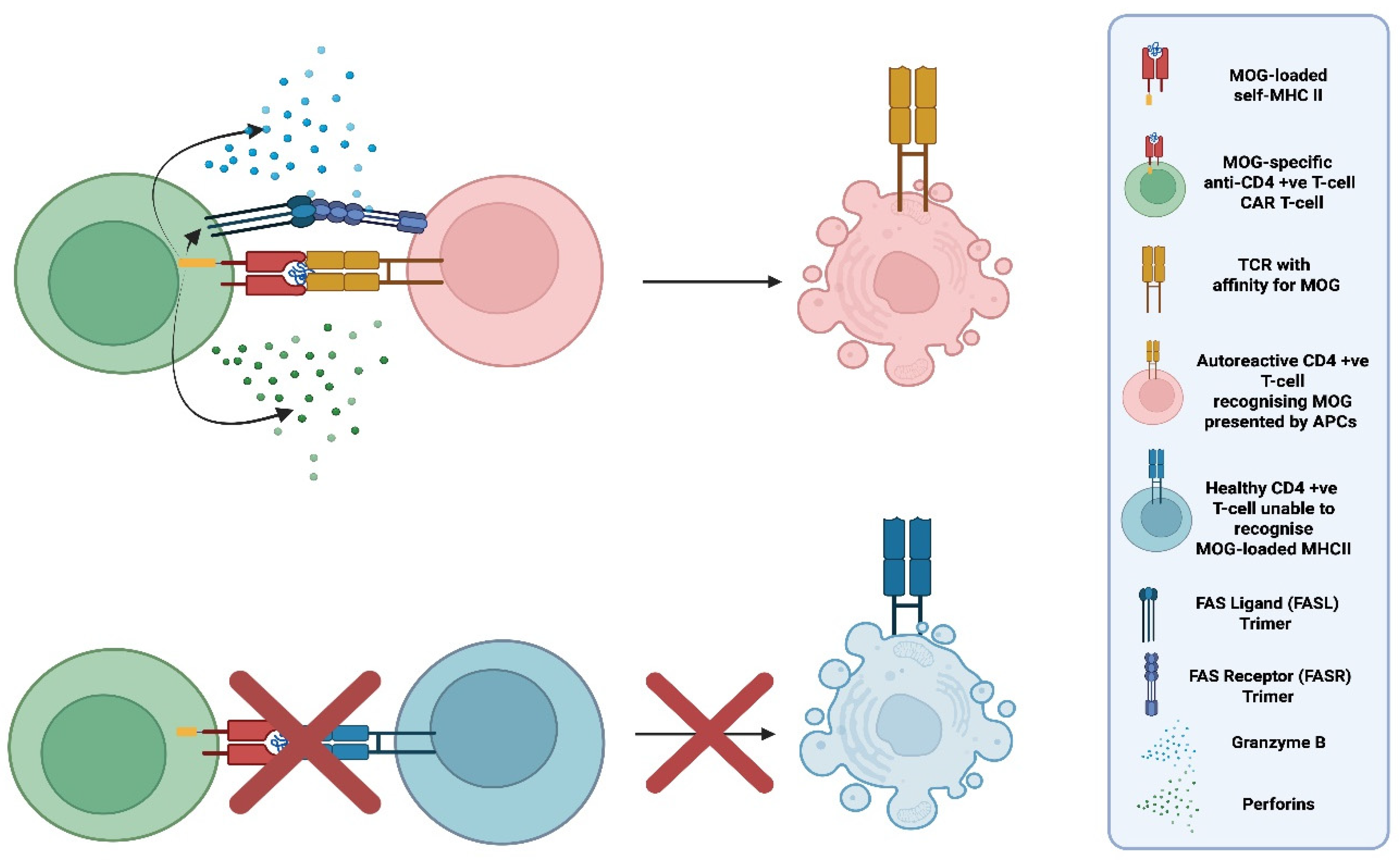
5.5. Peptide-Specific CAR T-Cells (pMHCII CAR T-Cells ) for the Selective Depletion of Autorective T-Cells [64]
5.6. mRNA-Induced CAR T-Cells
6. Case Reports of CAR T-Cell Use in CNS Autoimmunity
6.1. Anti-CD19 CAR T-Cellsin 2 MS Patients [69]
6.2. Anti-CD19 CAR T-Cells in a 25-year-old Male Patient with Refractory Myelin Oligodendrocyte Antibody-Associated Disease (MOGAD)
6.3. Anti-BCMA CAR T-Cells in Twelve NMOSD Patients [72]
7. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CNS | Central nervous system |
| DMT | Disease modifying therapy |
| BCMA | B-cell maturation antigen |
| CD | Cluster of differentiation |
| MS | Multiple sclerosis |
| mAbs | Monoclonal antibodies |
| NMOSD | Neuromyelitis optica spectrum disorder |
| MOGAD | Myelin oligodendrocyte antibody-associated disease |
| AE | Autoimmune encephalitis |
| AEm | Autoimmune encephalomyelitis |
| CAR | Chimeric antigen receptor |
| MHC | Major histocompatibility complex |
| TCR | T-cell receptor |
| IL | Interleukin |
| NFAT | Nuclear factor of activated T-cells |
| TNF | Tumour necrosis factor |
| TIL | Tumour infiltrating lymphocyte |
| CM | Co-stimulatory molecule |
| PSMA | Prostate-specific membrane antigen |
| TRUCKS | T-cells redirected for universal cytokine-mediated killing |
| ADCC | Antibody-dependent cellular cytotoxicity |
| ADCP | Antibody-dependent cellular phagocytosis |
| CDC | Complement-depended cytotoxicity |
| NK Cell | Natural killer cell |
| Neu | Neutrophil |
| MΦ | Macrophage |
| MAC | Membrane-attack complex |
| CSF | Cerebrospinal fluid |
| OCBs | Oligoclonal bands |
| SPMS | Secondary progressive MS |
| BBB | Blood-brain-barrier |
| TLS | Tertiary lymphoid structure |
| VCAM-1 | Vascular cell adhesion molecule-1 |
| ICAM-1 | Intercellular adhesion molecule-1 |
| aHSCT | Autologous hematopoietic stem-cell transplantation |
| SLE | Systemic lupous erythematosus |
| CRS | Cytokine release syndrome |
| ICANS | Immune effector cell-associated neurotoxicity syndrome |
| CID | Chemical Inducer of dimerization |
| Treg | T-regulatory cell |
| CTLA-4 | Cytotoxic T-lymphocyte–associated protein 4 |
| PD-1 | Programmed cell-death-protein 1 |
| GVHD | Graft-versus-host disease |
| HVGD | Host-versus-graft disease |
| HLA | Human leukocyte antigen |
| PBMCs | Peripheral blood mononuclear cells |
| NMDAR | N-methyl-D-aspartate receptor |
| CAAR | Chimeric autoantibody receptor |
| BCR | B-cell receptor |
| LGI1 | Leucine-rich glioma-inactivated 1 |
| AQP4 | Aquaporin-4 |
| MOG | Myelin oligodendrocyte glycoprotein |
| TGF | Transforming growth factor |
| APCs | Antigen-presenting cells |
| PK | Pharmacokinetics |
| PD | Pharmacodynamics |
| MG | Myasthenia gravis |
| rCAR T | RNA CAR T-cell |
| RRMS | Relapse-refractory multiple sclerosis |
| PPMS | Primary-progressive multiple sclerosis |
| ON | Optic neuritis |
| IVMTP | Intravenous methylprednisolone |
| OCT | Optic coherence tomography |
| EDSS | Expanded disability status scale |
References
- Ramanathan, S.; Brilot, F.; Irani, S. R.; Dale, R. C. Origins and immunopathogenesis of autoimmune central nervous system disorders. Nature Reviews Neurology 2023, 19, 172–190. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, L. D.; Cookfair, D. L.; Rudick, R. A.; Herndon, R. M.; Richert, J. R.; Salazar, A. M.; Fischer, J. S.; Goodkin, D. E.; Granger, C. v.; Simon, J. H.; Alam, J. J.; Bartoszak, D. M.; Bourdette, D. N.; Braiman, J.; Brownscheidle, C. M.; Coats, M. E.; Cohan, S. L.; Dougherty, D. S.; Kinkel, R. P.; Whitham, R. H. Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. Annals of Neurology 1996, 39, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K. P.; Brooks, B. R.; Cohen, J. A.; Ford, C. C.; Goldstein, J.; Lisak, R. P.; Myers, L. W.; Panitch, H. S.; Rose, J. W.; Schiffer, R. B.; Vollmer, T.; Weiner, L. P.; Wolinsky, J. S. Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis. Neurology 1995, 45, 1268–1276. [Google Scholar] [CrossRef]
- Skarlis, C.; Angelopoulou, E.; Rentzos, M.; Papageorgiou, S. G.; Anagnostouli, M. Monoclonal Antibodies as Therapeutic Agents in Autoimmune and Neurodegenerative Diseases of the Central Nervous System: Current Evidence on Molecular Mechanisms and Future Directions. International Journal of Molecular Sciences 2025, 26, 9398. [Google Scholar] [CrossRef]
- Skarlis, C.; Kotsari, M.; Anagnostouli, M. Advancing Treatment in Pediatric Multiple Sclerosis: The Promise of B-Cell-Targeting Therapies. International Journal of Molecular Sciences 2025, 26, 5989. [Google Scholar] [CrossRef] [PubMed]
- Cappell, K. M.; Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nature Reviews Clinical Oncology 2023, 20, 359–371. [Google Scholar] [CrossRef]
- Afzali, A. M.; Nirschl, L.; Sie, C.; Pfaller, M.; Ulianov, O.; Hassler, T.; Federle, C.; Petrozziello, E.; Kalluri, S. R.; Chen, H. H.; Tyystjärvi, S.; Muschaweckh, A.; Lammens, K.; Delbridge, C.; Büttner, A.; Steiger, K.; Seyhan, G.; Ottersen, O. P.; Öllinger, R.; Korn, T. B-cells orchestrate tolerance to the neuromyelitis optica autoantigen AQP4. Nature 2024, 627, 407–415. [Google Scholar] [CrossRef]
- Corbali, O.; Chitnis, T. Pathophysiology of myelin oligodendrocyte glycoprotein antibody disease. Frontiers in Neurology 2023, 14, 1137998. [Google Scholar] [CrossRef]
- Lanz, T. v.; Brewer, R. C.; Ho, P. P.; Moon, J.-S.; Jude, K. M.; Fernandez, D.; Fernandes, R. A.; Gomez, A. M.; Nadj, G.-S.; Bartley, C. M.; Schubert, R. D.; Hawes, I. A.; Vazquez, S. E.; Iyer, M.; Zuchero, J. B.; Teegen, B.; Dunn, J. E.; Lock, C. B.; Kipp, L. B.; Robinson, W. H. Clonally expanded B-cells in multiple sclerosis bind EBV EBNA1 and GlialCAM. Nature 2022, 603, 321–327. [Google Scholar] [CrossRef]
- Shang, H.; Shen, X.; Yu, X.; Zhang, J.; Jia, Y.; Gao, F. B-cell targeted therapies in autoimmune encephalitis: mechanisms, clinical applications, and therapeutic potential. Frontiers in Immunology 2024, 15, 1368275. [Google Scholar] [CrossRef]
- Comi, G.; Bar-Or, A.; Lassmann, H.; Uccelli, A.; Hartung, H.; Montalban, X.; Sørensen, P. S.; Hohlfeld, R.; Hauser, S. L. Role of B-cells in Multiple Sclerosis and Related Disorders. Annals of Neurology 2021, 89, 13–23. [Google Scholar] [CrossRef]
- Sadelain, M.; Brentjens, R.; Rivière, I. The Basic Principles of Chimeric Antigen Receptor Design. Cancer Discovery 2013, 3, 388–398. [Google Scholar] [CrossRef]
- Fujiwara, K.; Tsunei, A.; Kusabuka, H.; Ogaki, E.; Tachibana, M.; Okada, N. Hinge and Transmembrane Domains of Chimeric Antigen Receptor Regulate Receptor Expression and Signaling Threshold. Cells 2020, 9, 1182. [Google Scholar] [CrossRef] [PubMed]
- Abate-Daga, D.; Davila, M. L. CAR models: next-generation CAR modifications for enhanced T-cell function. Molecular Therapy - Oncolytics 2016, 3, 16014. [Google Scholar] [CrossRef]
- Davenport, A. J.; Jenkins, M. R. Programming a serial killer: CAR T cells form non-classical immune synapses. Oncoscience 2018, 5, 69–70. [Google Scholar] [CrossRef] [PubMed]
- Benmebarek, M.-R.; Karches, C. H.; Cadilha, B. L.; Lesch, S.; Endres, S.; Kobold, S. Killing Mechanisms of Chimeric Antigen Receptor (CAR) T Cells. International Journal of Molecular Sciences 2019, 20, 1283. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S. A.; Packard, B. S.; Aebersold, P. M.; Solomon, D.; Topalian, S. L.; Toy, S. T.; Simon, P.; Lotze, M. T.; Yang, J. C.; Seipp, C. A.; Simpson, C.; Carter, C.; Bock, S.; Schwartzentruber, D.; Wei, J. P.; White, D. E. Use of Tumor-Infiltrating Lymphocytes and Interleukin-2 in the Immunotherapy of Patients with Metastatic Melanoma. New England Journal of Medicine 1988, 319, 1676–1680. [Google Scholar] [CrossRef]
- Eshhar, Z.; Waks, T.; Gross, G.; Schindler, D. G. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proceedings of the National Academy of Sciences 1993, 90, 720–724. [Google Scholar] [CrossRef]
- Finney, H. M.; Lawson, A. D.; Bebbington, C. R.; Weir, A. N. Chimeric receptors providing both primary and costimulatory signalling in T cells from a single gene product. Journal of Immunology (Baltimore, Md.: 1950) 1998, 161, 2791–2797. [Google Scholar] [CrossRef]
- Milone, M. C.; Fish, J. D.; Carpenito, C.; Carroll, R. G.; Binder, G. K.; Teachey, D.; Samanta, M.; Lakhal, M.; Gloss, B.; Danet-Desnoyers, G.; Campana, D.; Riley, J. L.; Grupp, S. A.; June, C. H. Chimeric Receptors Containing CD137 Signal Transduction Domains Mediate Enhanced Survival of T Cells and Increased Antileukemic Efficacy In Vivo. Molecular Therapy 2009, 17, 1453–1464. [Google Scholar] [CrossRef]
- Maher, J.; Brentjens, R. J.; Gunset, G.; Rivière, I.; Sadelain, M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRζ /CD28 receptor. Nature Biotechnology 2002, 20, 70–75. [Google Scholar] [CrossRef]
- Ramos, C. A.; Rouce, R.; Robertson, C. S.; Reyna, A.; Narala, N.; Vyas, G.; Mehta, B.; Zhang, H.; Dakhova, O.; Carrum, G.; Kamble, R. T.; Gee, A. P.; Mei, Z.; Wu, M.-F.; Liu, H.; Grilley, B.; Rooney, C. M.; Heslop, H. E.; Brenner, M. K.; Dotti, G. In Vivo Fate and Activity of Second- versus Third-Generation CD19-Specific CAR-T Cells in B Cell Non-Hodgkin’s Lymphomas. Molecular Therapy 2018, 26, 2727–2737. [Google Scholar] [CrossRef]
- Hombach, A. A.; Rappl, G.; Abken, H. Arming Cytokine-induced Killer Cells With Chimeric Antigen Receptors: CD28 Outperforms Combined CD28–OX40 Super-stimulation. Molecular Therapy 2013, 21, 2268–2277. [Google Scholar] [CrossRef]
- Chmielewski, M.; Abken, H. TRUCKs: the fourth generation of CARs. Expert Opinion on Biological Therapy 2015, 15, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Chmielewski, M.; Abken, H. TRUCKS, the fourth-generation CAR T cells: Current developments and clinical translation. ADVANCES IN CELL AND GENE THERAPY 2020, 3. [Google Scholar] [CrossRef]
- Kagoya, Y.; Tanaka, S.; Guo, T.; Anczurowski, M.; Wang, C.-H.; Saso, K.; Butler, M. O.; Minden, M. D.; Hirano, N. A novel chimeric antigen receptor containing a JAK–STAT signalling domain mediates superior antitumor effects. Nature Medicine 2018, 24, 352–359. [Google Scholar] [CrossRef]
- Gogesch, P.; Dudek, S.; van Zandbergen, G.; Waibler, Z.; Anzaghe, M. The Role of Fc Receptors on the Effectiveness of Therapeutic Monoclonal Antibodies. International Journal of Molecular Sciences 2021, 22, 8947. [Google Scholar] [CrossRef]
- Rubenstein, J. L.; Combs, D.; Rosenberg, J.; Levy, A.; McDermott, M.; Damon, L.; Ignoffo, R.; Aldape, K.; Shen, A.; Lee, D.; Grillo-Lopez, A.; Shuman, M. A. Rituximab therapy for CNS lymphomas: targeting the leptomeningeal compartment. Blood 2003, 101, 466–468. [Google Scholar] [CrossRef]
- Cabrera, C. M. Oligoclonal bands: An immunological and clinical approach. Advances in Clinical Chemistry 2022, 109, 129–163. [Google Scholar] [PubMed]
- Magliozzi, R.; Howell, O.; Vora, A.; Serafini, B.; Nicholas, R.; Puopolo, M.; Reynolds, R.; Aloisi, F. Meningeal B-cell follicles in secondary progressive multiple sclerosis associate with early onset of disease and severe cortical pathology. Brain 2006, 130, 1089–1104. [Google Scholar] [CrossRef]
- Engelhardt, B.; Ransohoff, R. M. Capture, crawl, cross: the T cell code to breach the blood–brain barriers. Trends in Immunology 2012, 33, 579–589. [Google Scholar] [CrossRef]
- Choquet, S.; Soussain, C.; Azar, N.; Morel, V.; Metz, C.; Ursu, R.; Waultier-Rascalou, A.; di Blasi, R.; Houot, R.; Souchet, L.; Roos-Weil, D.; Uzunov, M.; Quoc, S. N.; Jacque, N.; Boussen, I.; Gauthier, N.; Ouzegdouh, M.; Blonski, M.; Campidelli, A.; Houillier, C. CAR T-cell therapy induces a high rate of prolonged remission in relapsed primary CNS lymphoma: Real-life results of the LOC network. American Journal of Hematology 2024, 99, 1240–1249. [Google Scholar] [CrossRef]
- Zhan, J.; Kipp, M.; Han, W.; Kaddatz, H. Ectopic lymphoid follicles in progressive multiple sclerosis: From patients to animal models. Immunology 2021, 164, 450–466. [Google Scholar] [CrossRef]
- Sai Santhosha Mrudula, A.; Avula, N. L. P.; Ahmed, S. K.; Salian, R. B.; Alla, D.; Jagannath, P.; Polasu, S. S. S. P.; Rudra, P.; Issaka, Y.; Khetan, M. S.; Gupta, T. Immunological outcomes of autologous hematopoietic stem cell transplantation for multiple sclerosis: a systematic review. Annals of Medicine & Surgery 2024, 86, 421–432. [Google Scholar] [CrossRef]
- Mackensen, A.; Müller, F.; Mougiakakos, D.; Böltz, S.; Wilhelm, A.; Aigner, M.; Völkl, S.; Simon, D.; Kleyer, A.; Munoz, L.; Kretschmann, S.; Kharboutli, S.; Gary, R.; Reimann, H.; Rösler, W.; Uderhardt, S.; Bang, H.; Herrmann, M.; Ekici, A. B.; Schett, G. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nature Medicine 2022, 28, 2124–2132. [Google Scholar] [CrossRef]
- Siegler, E. L.; Kenderian, S. S. Neurotoxicity and Cytokine Release Syndrome After Chimeric Antigen Receptor T Cell Therapy: Insights Into Mechanisms and Novel Therapies. Frontiers in Immunology 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Morris, E. C.; Neelapu, S. S.; Giavridis, T.; Sadelain, M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nature Reviews Immunology 2022, 22, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Gavriilaki, E.; Tzannou, I.; Vardi, A.; Tsonis, I.; Liga, M.; Gkirkas, K.; Ximeri, M.; Bousiou, Z.; Bouzani, M.; Sagiadinou, E.; Dolgyras, P.; Kotsiou, N.; Bampali, V.; Mallouri, D.; Tzenou, T.; Batsis, I.; Sotiropoulos, D.; Gigantes, S.; Papadaki, H. A.; Sakellari, I. Management strategies for CAR-T cell therapy-related toxicities: results from a survey in Greece. Frontiers in Medicine 2025, 12. [Google Scholar] [CrossRef]
- Lemoine, J.; Bachy, E.; Cartron, G.; Beauvais, D.; Gastinne, T.; di Blasi, R.; Rubio, M.-T.; Guidez, S.; Mohty, M.; Casasnovas, R.-O.; Joris, M.; Castilla-Llorente, C.; Haioun, C.; Hermine, O.; Loschi, M.; Carras, S.; Bories, P.; Fradon, T.; Herbaux, C.; Houot, R. Non relapse mortality after CAR T-cell therapy for large B-cell lymphoma: a LYSA study from the DESCAR-T registry. Blood Advances 2023, 7, 6589–6598. [Google Scholar] [CrossRef] [PubMed]
- Müller, F.; Taubmann, J.; Bucci, L.; Wilhelm, A.; Bergmann, C.; Völkl, S.; Aigner, M.; Rothe, T.; Minopoulou, I.; Tur, C.; Knitza, J.; Kharboutli, S.; Kretschmann, S.; Vasova, I.; Spoerl, S.; Reimann, H.; Munoz, L.; Gerlach, R. G.; Schäfer, S.; Schett, G. CD19 CAR T-Cell Therapy in Autoimmune Disease — A Case Series with Follow-up. New England Journal of Medicine 2024, 390, 687–700. [Google Scholar] [CrossRef]
- Morgan, R. A.; Yang, J. C.; Kitano, M.; Dudley, M. E.; Laurencot, C. M.; Rosenberg, S. A. Case Report of a Serious Adverse Event Following the Administration of T Cells Transduced With a Chimeric Antigen Receptor Recognizing ERBB2. Molecular Therapy 2010, 18, 843–851. [Google Scholar] [CrossRef]
- Hoyos, V.; Savoldo, B.; Quintarelli, C.; Mahendravada, A.; Zhang, M.; Vera, J.; Heslop, H. E.; Rooney, C. M.; Brenner, M. K.; Dotti, G. Engineering CD19-specific T lymphocytes with interleukin-15 and a suicide gene to enhance their anti-lymphoma/leukemia effects and safety. Leukemia 2010, 24, 1160–1170. [Google Scholar] [CrossRef]
- Ayala Ceja, M.; Khericha, M.; Harris, C. M.; Puig-Saus, C.; Chen, Y. Y. CAR-T cell manufacturing: Major process parameters and next-generation strategies. Journal of Experimental Medicine 2024, 221. [Google Scholar] [CrossRef]
- Papathanasiou, M. M.; Stamatis, C.; Lakelin, M.; Farid, S.; Titchener-Hooker, N.; Shah, N. Autologous CAR T-cell therapies supply chain: challenges and opportunities? Cancer Gene Therapy 2020, 27, 799–809. [Google Scholar] [CrossRef]
- Webster, J. A.; Luznik, L. This CAR won’t start: predicting nonresponse in ALL. Blood Advances 2023, 7, 4215–4217. [Google Scholar] [CrossRef]
- Tao, Z.; Chyra, Z.; Kotulová, J.; Celichowski, P.; Mihályová, J.; Charvátová, S.; Hájek, R. Impact of T cell characteristics on CAR-T cell therapy in hematological malignancies. Blood Cancer Journal 2024, 14, 213. [Google Scholar] [CrossRef]
- Mehta, P. H.; Fiorenza, S.; Koldej, R. M.; Jaworowski, A.; Ritchie, D. S.; Quinn, K. M. T Cell Fitness and Autologous CAR T Cell Therapy in Haematologic Malignancy. Frontiers in Immunology 2021, 12, 780442. [Google Scholar] [CrossRef] [PubMed]
- Graham, C.; Jozwik, A.; Pepper, A.; Benjamin, R. Allogeneic CAR-T Cells: More than Ease of Access? Cells 2018, 7, 155. [Google Scholar] [CrossRef]
- Liu, P.; Liu, M.; Lyu, C.; Lu, W.; Cui, R.; Wang, J.; Li, Q.; Mou, N.; Deng, Q.; Yang, D. Acute Graft-Versus-Host Disease After Humanized Anti-CD19-CAR T Therapy in Relapsed B-ALL Patients After Allogeneic Hematopoietic Stem Cell Transplant. Frontiers in Oncology 2020, 10, 573822. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, J. P.; Nicole, O.; Groc, L. NMDA receptor functions in health and disease: Old actor, new dimensions. Neuron 2023, 111, 2312–2328. [Google Scholar] [CrossRef] [PubMed]
- Hughes, E. G.; Peng, X.; Gleichman, A. J.; Lai, M.; Zhou, L.; Tsou, R.; Parsons, T. D.; Lynch, D. R.; Dalmau, J.; Balice-Gordon, R. J. Cellular and Synaptic Mechanisms of Anti-NMDA Receptor Encephalitis. The Journal of Neuroscience 2010, 30, 5866–5875. [Google Scholar] [CrossRef]
- Dalmau, J.; Lancaster, E.; Martinez-Hernandez, E.; Rosenfeld, M. R.; Balice-Gordon, R. Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. The Lancet Neurology 2011, 10, 63–74. [Google Scholar] [CrossRef]
- Titulaer, M. J.; McCracken, L.; Gabilondo, I.; Armangué, T.; Glaser, C.; Iizuka, T.; Honig, L. S.; Benseler, S. M.; Kawachi, I.; Martinez-Hernandez, E.; Aguilar, E.; Gresa-Arribas, N.; Ryan-Florance, N.; Torrents, A.; Saiz, A.; Rosenfeld, M. R.; Balice-Gordon, R.; Graus, F.; Dalmau, J. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. The Lancet Neurology 2013, 12, 157–165. [Google Scholar] [CrossRef]
- Dong, B.; Yue, Y.; Dong, H.; Wang, Y. N-methyl-D-aspartate receptor hypofunction as a potential contributor to the progression and manifestation of many neurological disorders. Frontiers in Molecular Neuroscience 2023, 16, 1174738. [Google Scholar] [CrossRef]
- Martinez-Hernandez, E.; Horvath, J.; Shiloh-Malawsky, Y.; Sangha, N.; Martinez-Lage, M.; Dalmau, J. Analysis of complement and plasma cells in the brain of patients with anti-NMDAR encephalitis. Neurology 2011, 77, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Reincke, S. M.; von Wardenburg, N.; Homeyer, M. A.; Kornau, H.-C.; Spagni, G.; Li, L. Y.; Kreye, J.; Sánchez-Sendín, E.; Blumenau, S.; Stappert, D.; Radbruch, H.; Hauser, A. E.; Künkele, A.; Edes, I.; Schmitz, D.; Prüss, H. Chimeric autoantibody receptor T cells deplete NMDA receptor-specific B cells. Cell 2023, 186, 5084–5097.e18. [Google Scholar] [CrossRef] [PubMed]
- Lennon, V. A.; Kryzer, T. J.; Pittock, S. J.; Verkman, A. S.; Hinson, S. R. IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. The Journal of Experimental Medicine 2005, 202, 473–477. [Google Scholar] [CrossRef]
- Pittock, S. J.; Berthele, A.; Fujihara, K.; Kim, H. J.; Levy, M.; Palace, J.; Nakashima, I.; Terzi, M.; Totolyan, N.; Viswanathan, S.; Wang, K.-C.; Pace, A.; Fujita, K. P.; Armstrong, R.; Wingerchuk, D. M. Eculizumab in Aquaporin-4–Positive Neuromyelitis Optica Spectrum Disorder. New England Journal of Medicine 2019, 381, 614–625. [Google Scholar] [CrossRef] [PubMed]
- Cree, B. A. C.; Gourraud, P.; Oksenberg, J. R.; Bevan, C.; Crabtree-Hartman, E.; Gelfand, J. M.; Goodin, D. S.; Graves, J.; Green, A. J.; Mowry, E.; Okuda, D. T.; Pelletier, D.; von Büdingen, H. -Christian; Zamvil, S. S.; Agrawal, A.; Caillier, S.; Ciocca, C.; Gomez, R.; Kanner, R.; Hauser, S. L. Long-term evolution of multiple sclerosis disability in the treatment era. Annals of Neurology 2016, 80, 499–510. [Google Scholar] [CrossRef]
- Pittock, S. J.; Barnett, M.; Bennett, J. L.; Berthele, A.; de Sèze, J.; Levy, M.; Nakashima, I.; Oreja-Guevara, C.; Palace, J.; Paul, F.; Pozzilli, C.; Yountz, M.; Allen, K.; Mashhoon, Y.; Kim, H. J. Ravulizumab in Aquaporin-4–Positive Neuromyelitis Optica Spectrum Disorder. Annals of Neurology 2023, 93, 1053–1068. [Google Scholar] [CrossRef]
- Shah, N.; Chari, A.; Scott, E.; Mezzi, K.; Usmani, S. Z. B-cell maturation antigen (BCMA) in multiple myeloma: rationale for targeting and current therapeutic approaches. Leukemia 2020, 34, 985–1005. [Google Scholar] [CrossRef] [PubMed]
- Vignali, D. A. A.; Collison, L. W.; Workman, C. J. How regulatory T cells work. Nature Reviews Immunology 2008, 8, 523–532. [Google Scholar] [CrossRef]
- Frikeche, J.; David, M.; Mouska, X.; Treguer, D.; Cui, Y.; Rouquier, S.; Lecorgne, E.; Proics, E.; Fall, P. B.; Lafon, A.; Lara, G.; Menardi, A.; Fenard, D.; Abel, T.; Gertner-Dardenne, J.; de la Rosa, M.; Dumont, C. MOG-specific CAR Tregs: a novel approach to treat multiple sclerosis. Journal of Neuroinflammation 2024, 21, 268. [Google Scholar] [CrossRef]
- Vh Yi, J.; Miller, A. T.; Archambault, A. S.; Jones, A. J.; Bradstreet, T. R.; Bandla, S.; Hsu, Y.-S.; Edelson, B. T.; Zhou, Y. W.; Fremont, D. H.; Egawa, T.; Singh, N.; Wu, G. F.; Hsieh, C.-S. Antigen-specific depletion of CD4 + T cells by CAR T cells reveals distinct roles of higher- and lower-affinity TCRs during autoimmunity. Science Immunology 2022, 7. [Google Scholar] [CrossRef]
- Tegenge, M. A.; Wang, X.; Liu, J.; Zhu, H.; Fashoyin-Aje, L. A. FDA Experience on CAR T Cell Pharmacokinetics/Pharmacodynamics and Model-Based Assessments. Clinical Pharmacology & Therapeutics 2025, 118, 324–330. [Google Scholar] [CrossRef]
- Yoon, S. H.; Lee, J. M.; Cho, H. I.; Kim, E. K.; Kim, H. S.; Park, M. Y.; Kim, T. G. Adoptive immunotherapy using human peripheral blood lymphocytes transferred with RNA encoding Her-2/neu-specific chimeric immune receptor in ovarian cancer xenograft model. Cancer Gene Therapy 2009, 16, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zheng, Z.; Cohen, C. J.; Gattinoni, L.; Palmer, D. C.; Restifo, N. P.; Rosenberg, S. A.; Morgan, R. A. High-Efficiency Transfection of Primary Human and Mouse T Lymphocytes Using RNA Electroporation. Molecular Therapy 2006, 13, 151–159. [Google Scholar] [CrossRef]
- Granit, V.; Benatar, M.; Kurtoglu, M.; Miljković, M. D.; Chahin, N.; Sahagian, G.; Feinberg, M. H.; Slansky, A.; Vu, T.; Jewell, C. M.; Singer, M. S.; Kalayoglu, M. v; Howard, J. F.; Mozaffar, T.; Granit, V.; Benatar, M.; Mozaffar, T.; Chahin, N.; Howard, J. F.; Kalayoglu, M. v. Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomised phase 1b/2a study. The Lancet Neurology 2023, 22, 578–590. [Google Scholar] [CrossRef]
- Fischbach, F.; Richter, J.; Pfeffer, L. K.; Fehse, B.; Berger, S. C.; Reinhardt, S.; Kuhle, J.; Badbaran, A.; Rathje, K.; Gagelmann, N.; Borie, D.; Seibel, J.; Ayuk, F.; Friese, M. A.; Heesen, C.; Kröger, N. CD19-targeted chimeric antigen receptor T cell therapy in two patients with multiple sclerosis. Med 2024, 5, 550–558.e2. [Google Scholar] [CrossRef]
- Shao, W.; Liu, X.; Li, J.; Sheng, T.; Li, Y.; Gu, Y.; Deng, B.; Wang, J.; Yang, W.; Yu, H.; Zhang, X.; Chen, X. Characteristics of cerebrospinal fluid oligoclonal band in anti-myelin oligodendrocyte glycoprotein (MOG) antibody associated disease. Heliyon 2024, 10, e24742. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Maqueda, J. M.; Sepulveda, M.; García, R. R.; Muñoz-Sánchez, G.; Martínez-Cibrian, N.; Ortíz-Maldonado, V.; Lorca-Arce, D.; Guasp, M.; Llufriu, S.; Martinez-Hernandez, E.; Armangue, T.; Fonseca, E. G.; Alba-Isasi, M. T.; Delgado, J.; Dalmau, J.; Juan, M.; Saiz, A.; Blanco, Y. CD19-Directed CAR T-Cells in a Patient With Refractory MOGAD. Neurology Neuroimmunology & Neuroinflammation 2024, 11. [Google Scholar] [CrossRef]
- Qin, C.; Tian, D.-S.; Zhou, L.-Q.; Shang, K.; Huang, L.; Dong, M.-H.; You, Y.-F.; Xiao, J.; Xiong, Y.; Wang, W.; Pang, H.; Guo, J.-J.; Cai, S.-B.; Wang, D.; Li, C.-R.; Zhang, M.; Bu, B.-T.; Wang, W. Anti-BCMA CAR T-cell therapy CT103A in relapsed or refractory AQP4-IgG seropositive neuromyelitis optica spectrum disorders: phase 1 trial interim results. Signal Transduction and Targeted Therapy 2023, 8, 5. [Google Scholar] [CrossRef] [PubMed]
| Condition | NCT no. | Antigen Target |
Phase | Patient enrolment |
Study Start |
Study Completion |
|
|---|---|---|---|---|---|---|---|
|
1 |
Relapsed /Refractory NMOSD |
NCT03605238 |
CD19/CD20 |
I *withdrawn due to recruitment issues |
N/A |
N/A |
N/A |
| 2 | (AQP4 +ve) NMOSD Recurrent & Refractory |
NCT05828212 | CD19 | I | 9 (actual) |
05-2023 | 30-7-2025 (actual) |
| 3 | Refractory Primary / Secondary Progressive MS |
NCT06384976 |
CD19 |
II |
120 (target) |
09-2024 |
01-2029 (estimated) |
| 4 | Relapsing / Progressive MS Refractory MG |
NCT06220201 | CD19 | I | 120 (estimated) |
03-2024 | 07-2027 (estimated) |
| 5 | Non-relapsing / Progressive MS | NCT06138132 | CD19 | I | 12 (estimated) |
04-2024 | 2027-06 (estimated) |
| 6 | Refractory MS / not responding to standard therapy | NCT06451159 | CD19 | I | 10 (estimated) |
06-2024 | 2027-06 (estimated) |
|
7 * |
Relapsing / Refractory NMOSD, MG, MS, MOGAD, CIDP, IIM, AE, POEMS syndrome |
NCT04561557 |
(Allogeneic) BCMA |
I |
36 (estimated) |
09-2020 |
05-2031 (estimated) |
|
8 ** |
Refractory to Standard Therapy SLE, IMNM, NMOSD, MS, MG, SSc |
NCT06249438 | Bispecific CD20&BCMA |
I |
30 (estimated) |
03-2024 |
03-2040 (estimated) |
|
9 * |
Refractory, failed standard treatment or lack effective treatment NMOSD, gMG, CIDP, MS |
NCT06485232 |
Allogeneic CD19 & BCMA |
I |
25 (estimated) |
02-2025 |
12-2027 (estimated) |
|
10 *** |
NMOSD, MOGAD, MS, MG |
NCT06869278 |
CD19 & CD20 & CD22 (Trispecific) | I | 37 (estimated) |
06-2025 (estimated) |
12-2029 (estimated) |
| 11 | MS, NMOSD, IIT, SLE, SSc, AAV, IIM, Sjögren's Syndrome (SS), MG |
NCT06548620 |
CD19 |
I |
18 (estimated) |
08-2024 | 08-2027 (estimated) |
| 12 | Refractory Neuroimmune Diseases |
NCT07022197 | BAFFR | I and II | 27 (estimated) |
04-2025 | 12-2027 (estimated) |
| 13 | Relapsing Multiple Sclerosis (MS) |
NCT06617793 | CD19 | I and II | 28 (estimated) |
02-2025 | 10-2030 (estimated) |
| 14 | Non-active Progressive Multiple Sclerosis (PMS) | NCT06675864 | CD19 | I and II | 28 (estimated) |
12-2024 | 06-2030 (estimated) |
| 15 | Relapsing / Progressive MS | NCT07006805 | CD19 | I and II | 12 (estimated) |
06-2026 | 10-2029 (estimated) |
|
16 * |
Relapse / Refractory Neurological Autoimmune Diseases |
NCT06939166 | CD19 / BCMA Allogeneic |
I | 12 (estimated) |
06-2025 (estimated) |
10-2027 (estimated) |
|
17 * |
B-cell Mediated Autoimmune Disorders |
NCT06680037 | CD19 Allogeneic |
I | 32 (estimated) |
2025-05-06 (actual) |
2029-01-01 (estimated) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




