Submitted:
12 December 2025
Posted:
16 December 2025
You are already at the latest version
Abstract
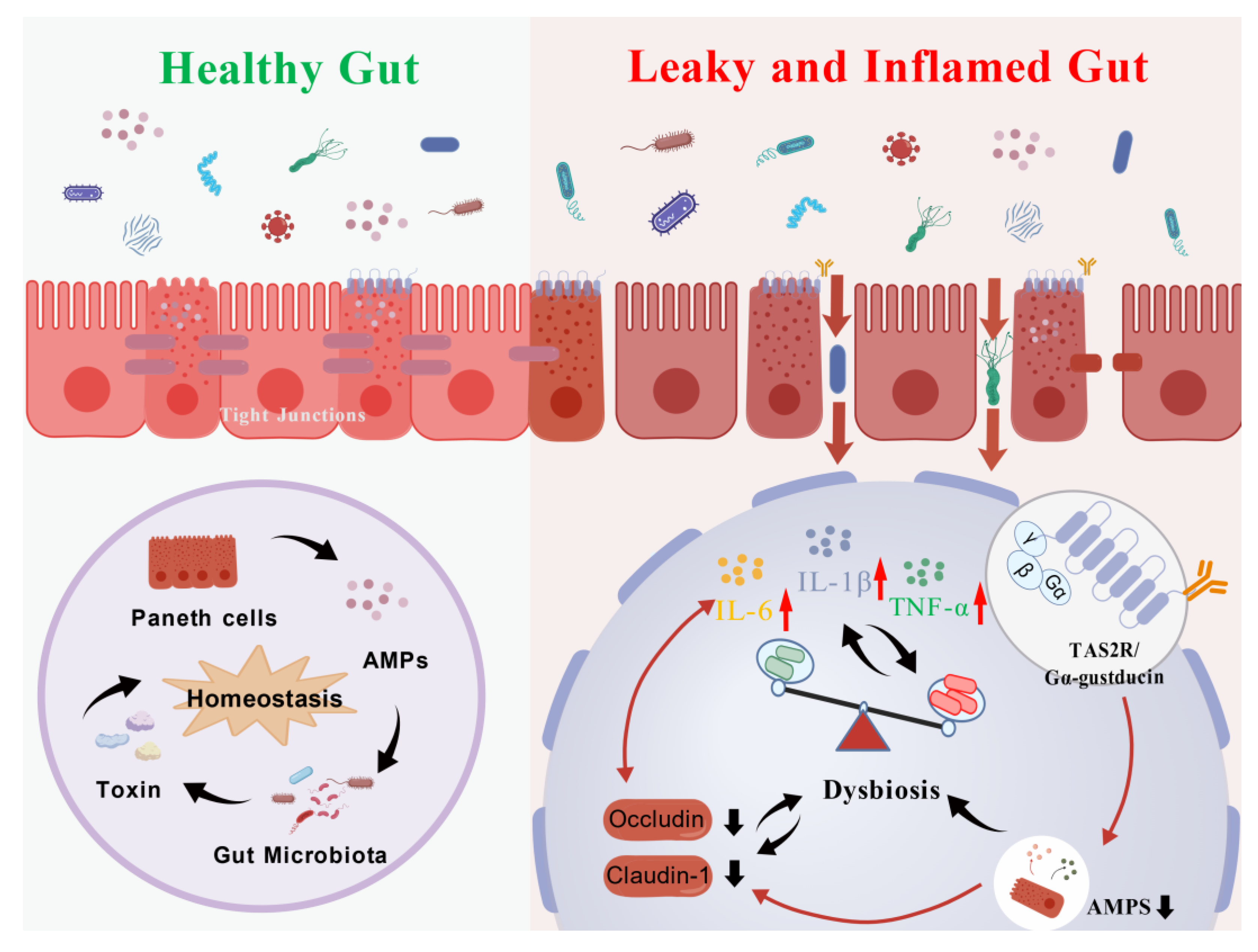
Clinical reports have shown that administration of Nauclea officinalis (Danmu in Chinese, DM) preparations may cause significant gastrointestinal discomfort. This study aimed to systematically evaluate the adverse effects of DM and its primary active constituent, strictosamide, on gastrointestinal motility, intestinal barrier integrity, and gut microbiota homeostasis. Furthermore, we sought to investigate the potential role of the bitter taste receptor (T2R) signaling pathway in mediating these effects. In vitro cell cultures and ex vivo intestinal tissues were employed to assess cell viability and molecular alterations. In vivo studies involved short-term (2 weeks) gavage of DM (0.54 and 1.08 g/kg) and long-term (16 weeks) intervention (0.4, 0.8, and 1.2 g/kg) in rodents. Evaluations included histopathological examination, serum levels of cytokines and oxidative stress markers (ELISA), expression of tight junction proteins (Western blot and qPCR), and 16S rDNA sequencing of cecal microbiota. Mechanistic analyses focused on α-defensin secretion and T2R-associated gene and protein expression. Administration of DM resulted in significant gastrointestinal dysfunction, characterized by delayed intestinal propulsion and increased gastric retention. Dose-dependent histopathological damage, disruption of the intestinal barrier (reduced occludin and claudin-1 expression), and elevated levels of pro-inflammatory cytokines (IL-6, TNF-α, IL-1β), oxidative stress markers (MDA, SOD, GSH-Px), and immune mediators (IFN-γ) were observed. Gut microbiota analysis revealed dysbiosis, marked by a decline in beneficial genera (e.g., Mucispirillum, Butyricicoccus, Roseburia) and an increase in potentially pathogenic bacteria (e.g., Citrobacter, Helicobacter). Mechanistically, DM suppressed α-defensin secretion and downregulated the expression of TAS2R108, TAS2R138, and Gα-gustducin both in vitro and in vivo. DM and strictosamide disrupt gut microbiota composition and compromise intestinal barrier function, likely through inhibition of the T2R/α-defensin pathway. These findings provide important mechanistic insights into drug-induced gastrointestinal toxicity and underscore the potential risks associated with prolonged use of DM-containing preparations.
Keywords:
1. Introduction
2. Results
2.1. Effects of Short-Term DM Intervention on Intestinal Function in Rats
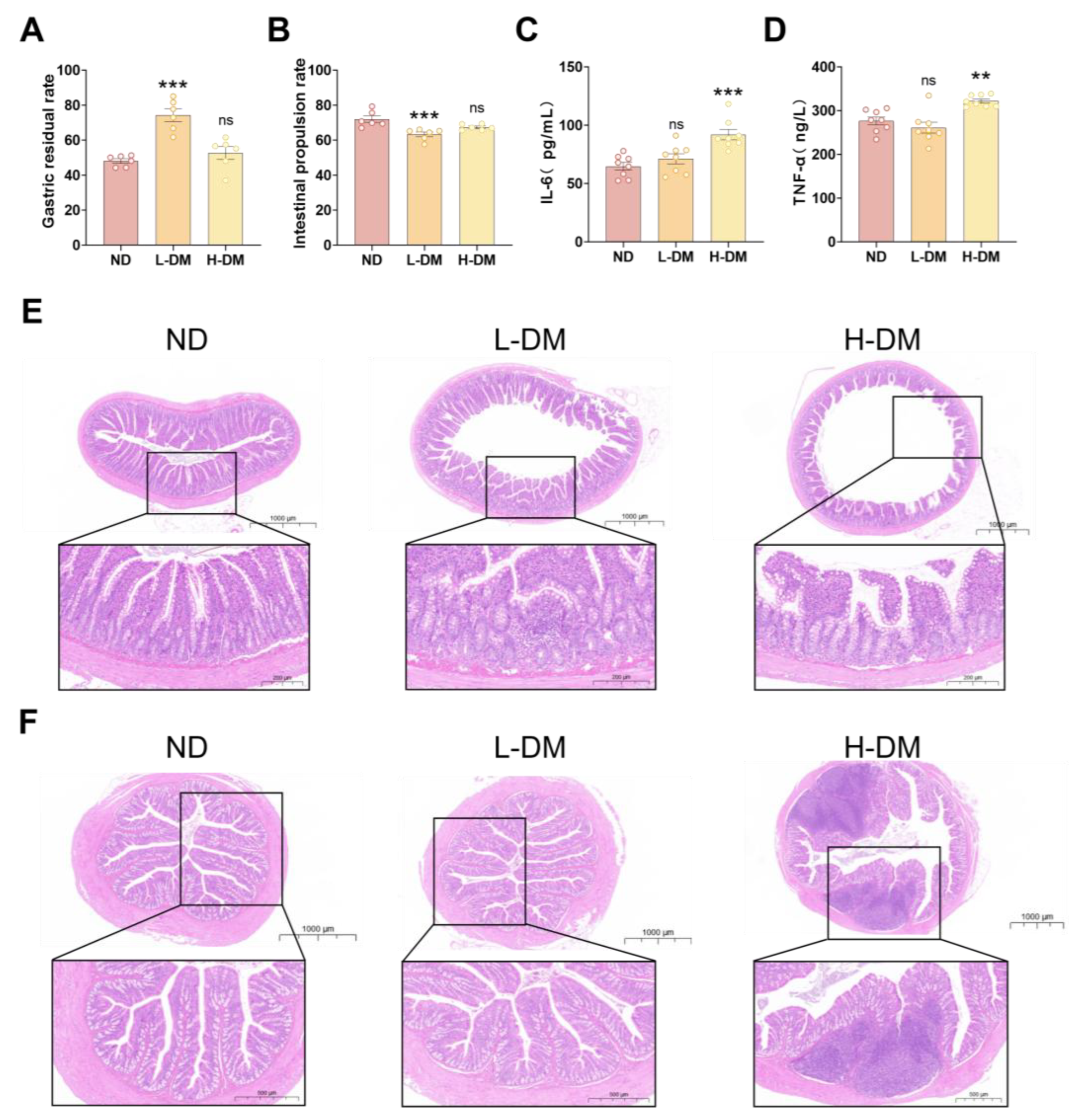
2.1.1. Effects of Short-Term DM Intervention on Gastrointestinal Motility in Rats
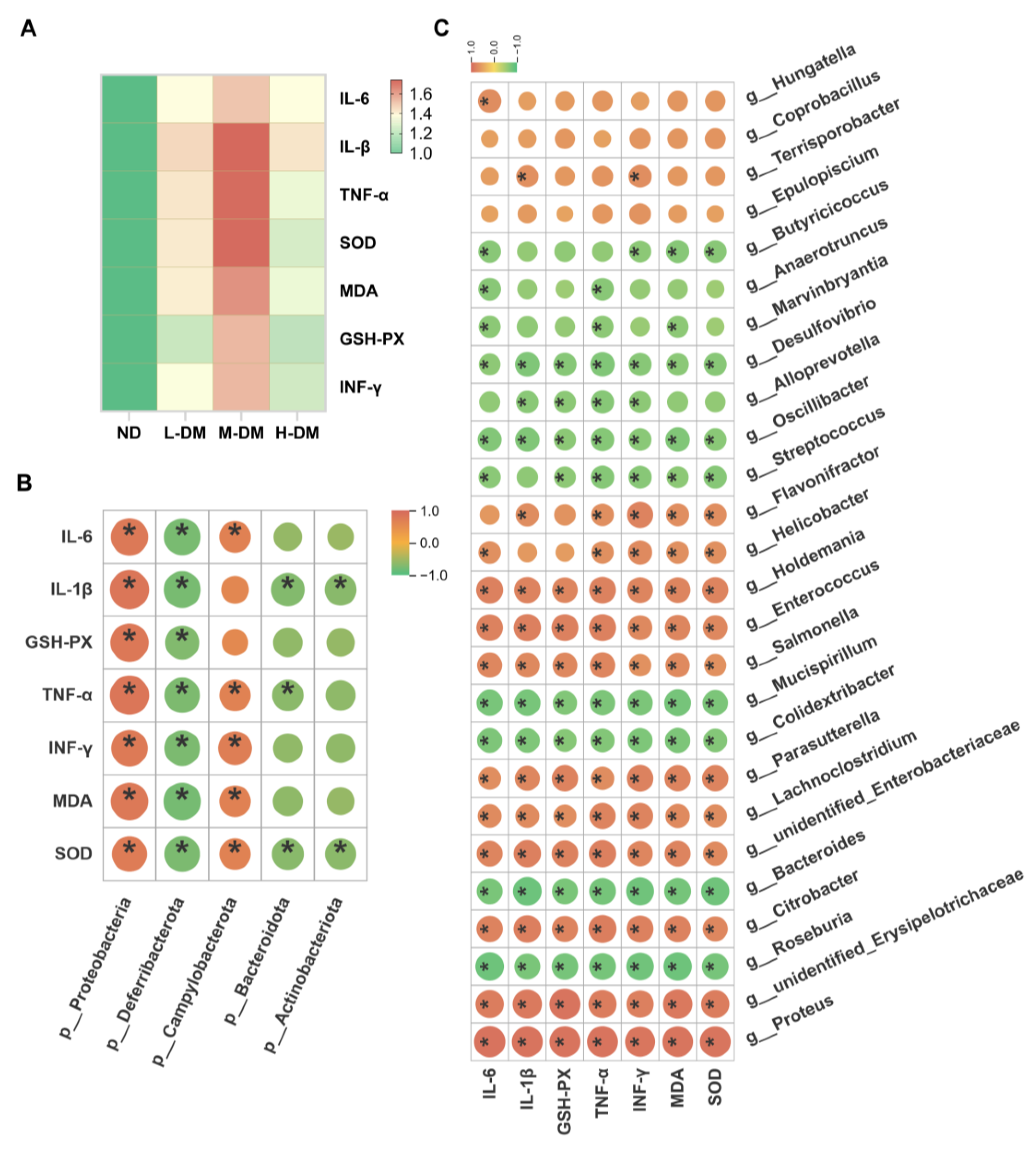
2.1.2. Effects of Short-Term DM Intervention on Inflammatory Levels in Rats
2.1.3. Effects of Short-Term DM Intervention on the Morphology of Ileum and Colon in Rats
2.2. Effects of Long-Term DM Intervention on the Intestine in Mice
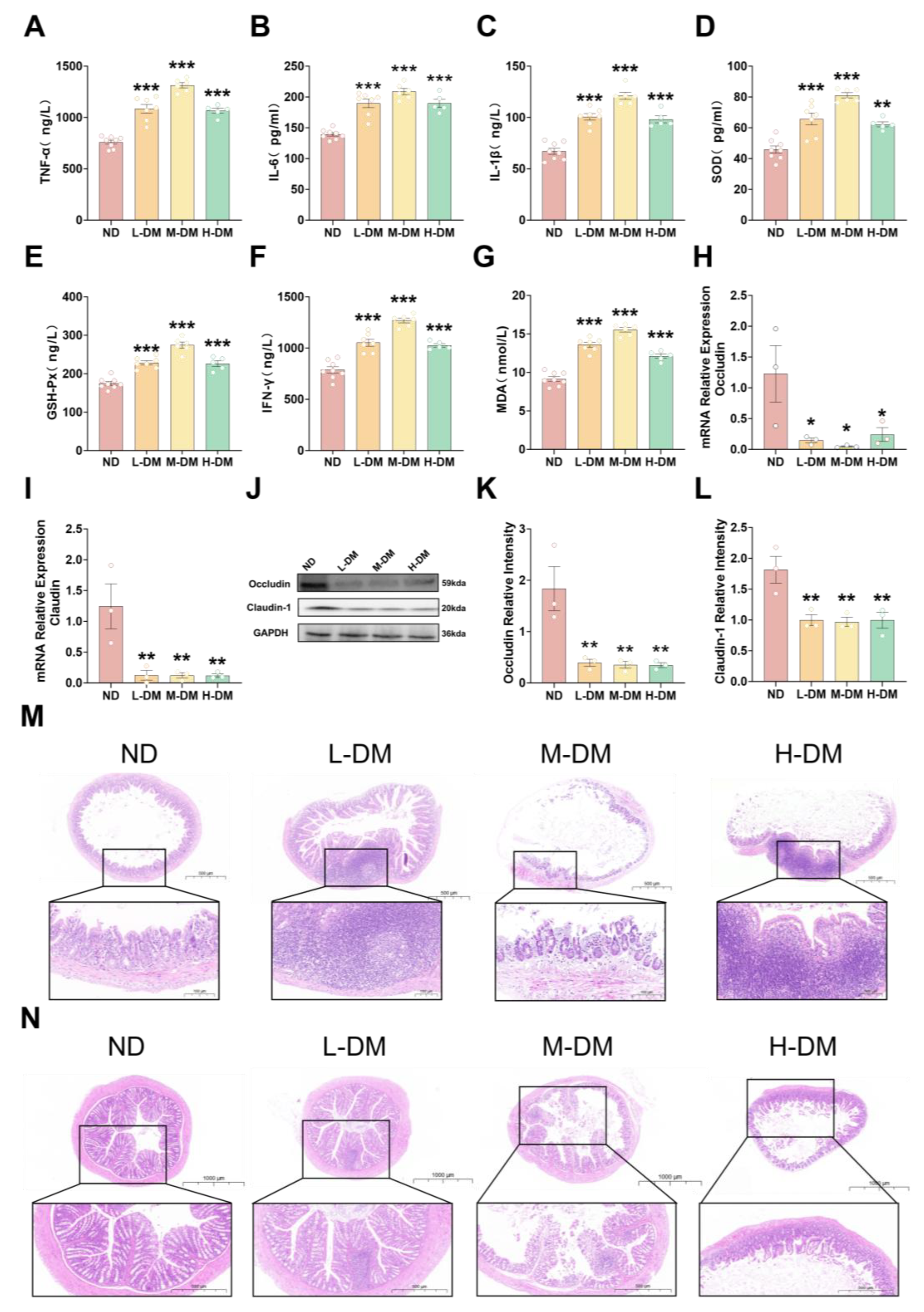
2.2.1. Effects of Long-Term DM Intervention on Intestinal Inflammatory Levels in Mice
2.2.2. Effects of Long-Term DM Intervention on Intestinal Wall Integrity in Mice
2.2.3. Effects of Long-Term DM Intervention on the Morphology of Ileum and Colon in Mice
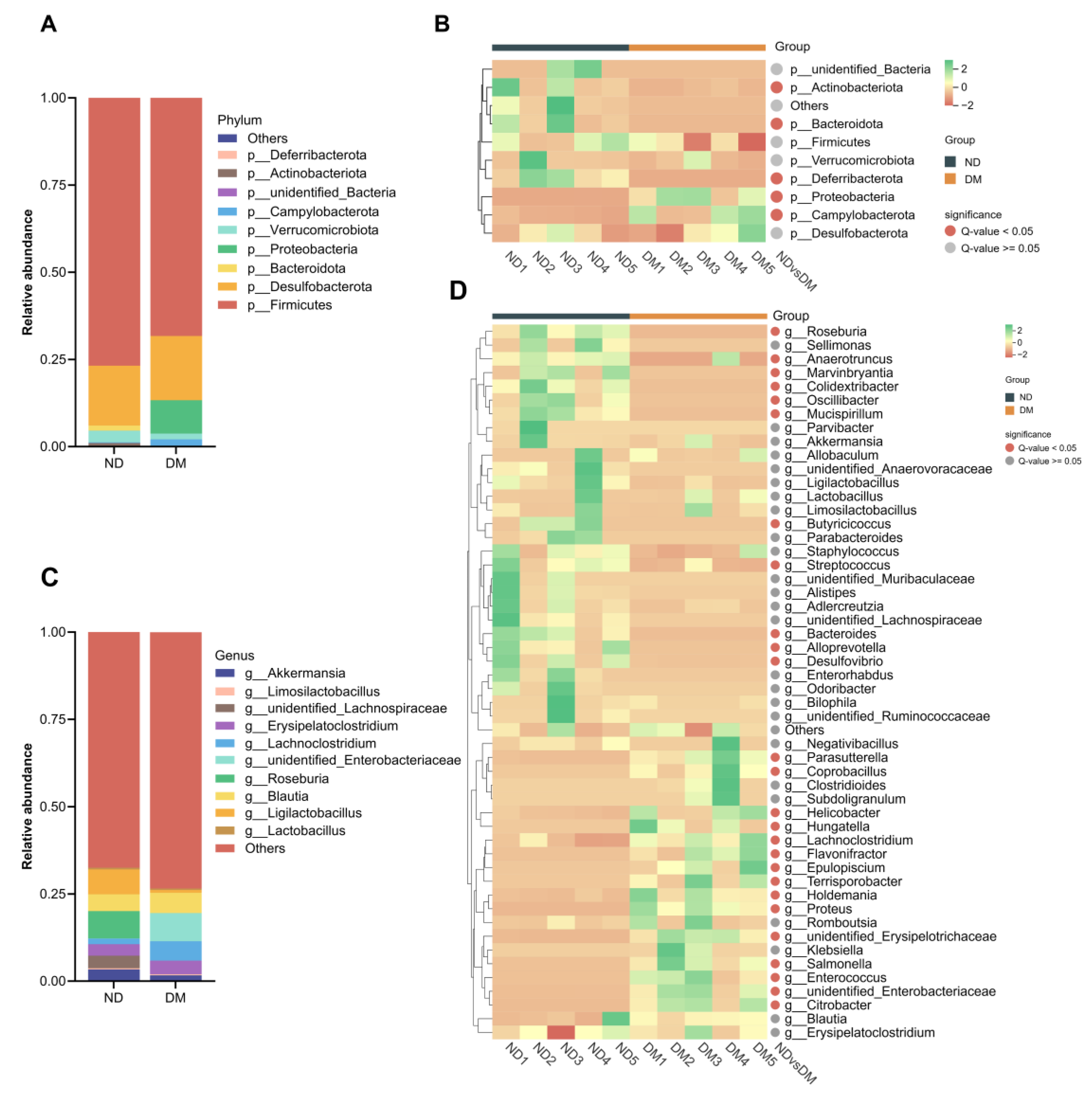
2.3. Effects of Long-Term DM Intervention on Intestinal Flora
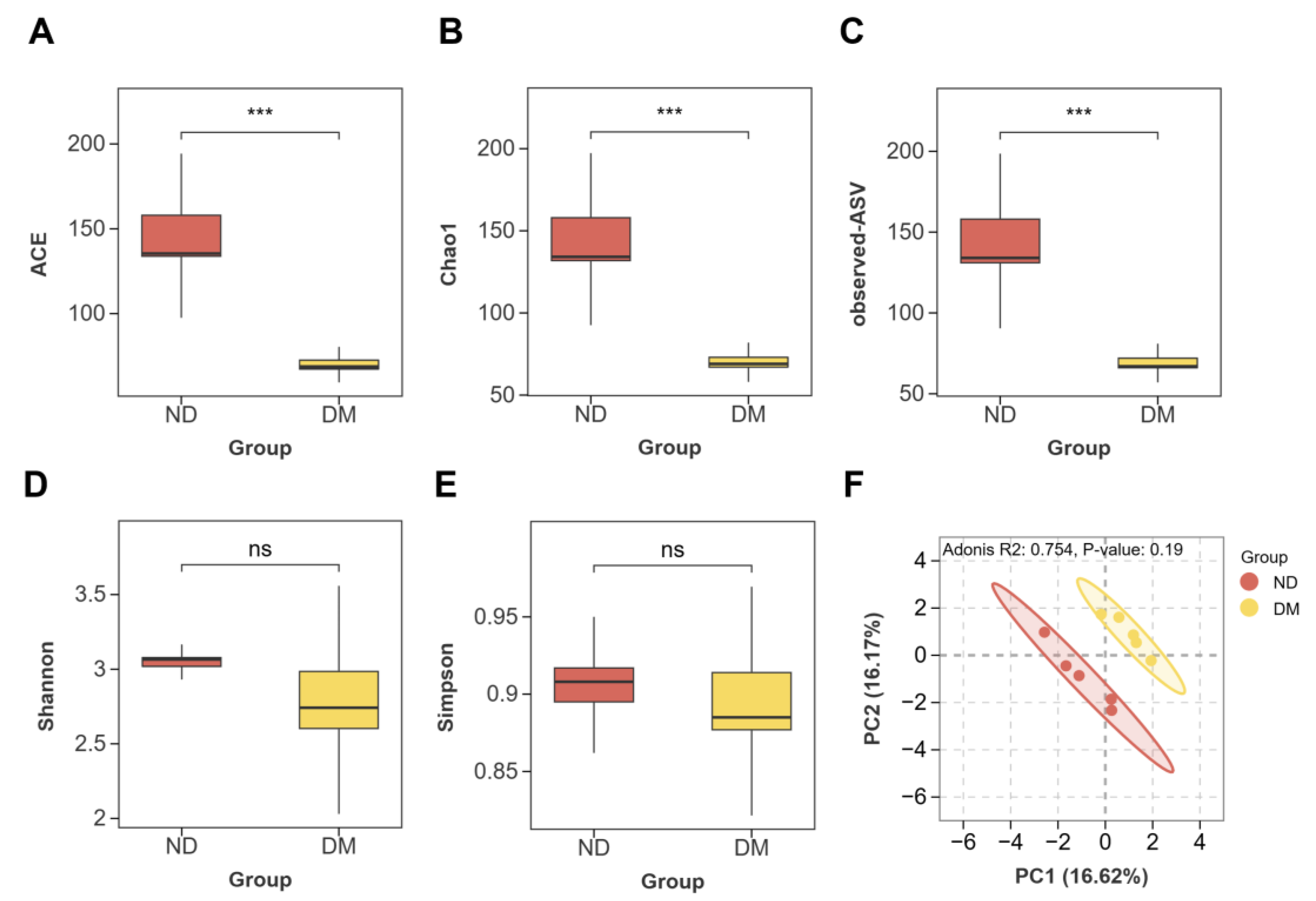
2.3.1. Effects of DM on Alpha and Beta Diversity
3.2. Effects of DM on the Composition of Intestinal Microorganisms
3.3. Correlation Analysis Between Gut Microbiota and Inflammatory/Oxidative Stress Biomarkers
2.4. Effects of DM on Cell/Tissue Viability and T2R/α-Defensin Expression
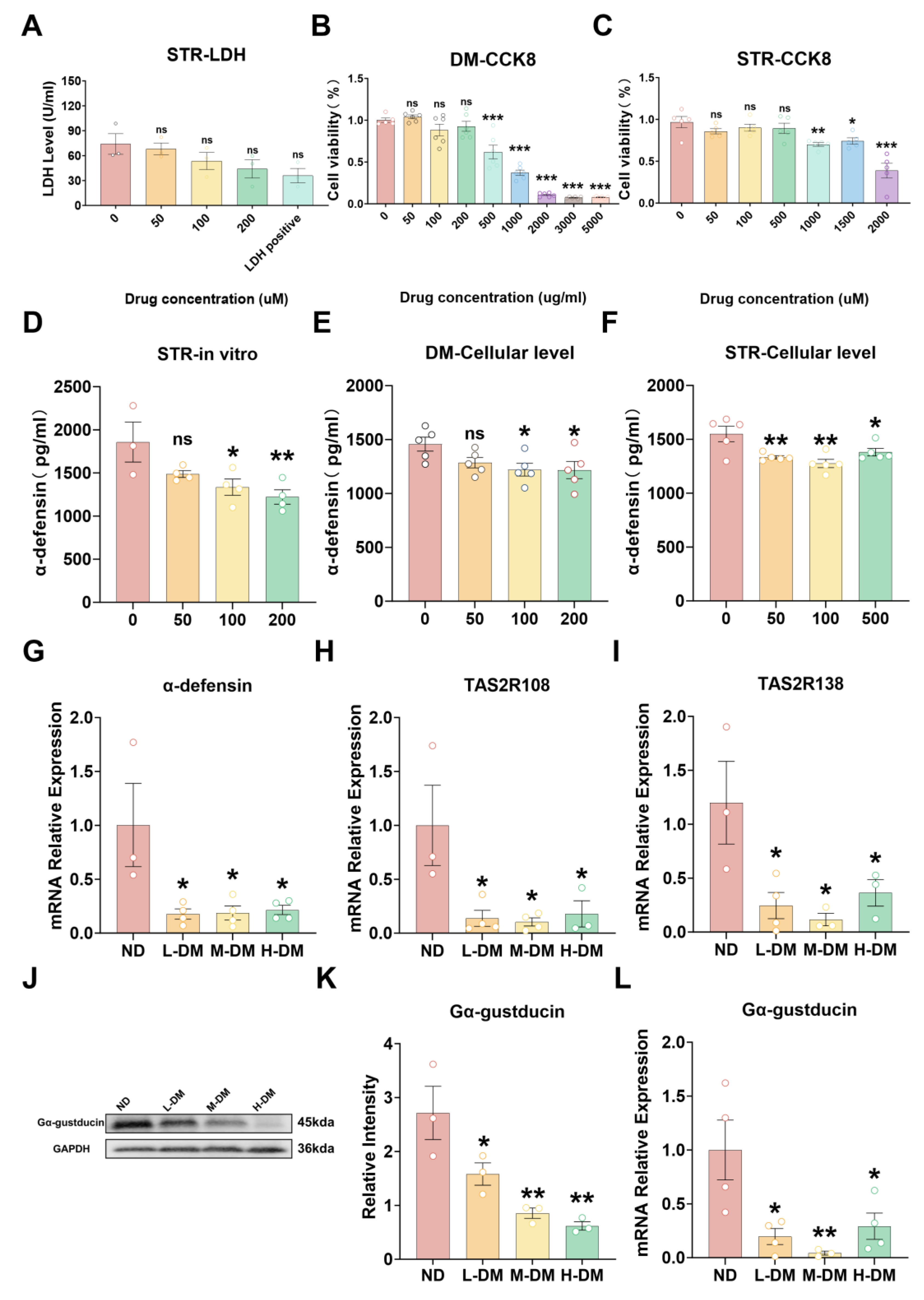
2.4.1. Effects of DM on Viability in STC-1 Cells and Ileal Ex Vivo Tissue
2.4.2. Effects of DM on the Expression of Antimicrobial Peptides
2.4.3. Effects of DM on the Expression of Bitter Taste Receptors
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials
5.2. Animals and Ethics Statement
5.3. Grouping and DM Treatment
5.4. Enzyme-Linked Immunosorbent Assay (ELISA)
5.5. Intestinal Flora Diversity
5.6. Gastrointestinal Motility Assessment: Small Intestinal Propulsion and Gastric Retention Rate
5.7. RNA Isolation and Real-Time PCR
| Primer Name | Primer Name |
|---|---|
| Actb-1R | GACCCATTCCCACCATC |
| Actb-1F | TCTTTGCAGCTCCTTCGT |
| Defa-5-R | GCAGCCTCTTATTCTACAATAGCA |
| Defa-5-F | CTAATACTGAGGAGCAGCCAGG |
| Ocln-1F | CTGCCTGCACGATGTCT |
| Ocln-1R | GAGTGTTCAGCCCAGTCAA |
| Cldn11-2F | CAGGTGGTGGGTTTCGT |
| Cldn11-2R | CAGGTGGGGATGGTGTAG |
| Tas2r108-2F | AACAGGACCAGCTTTTGGAATC |
| Tas2r108-2R | GAGGAAACAGATCATCAGCCTCAT |
| Tas2r138-1F | CACAACTACCAAGCCATCC |
| Tas2r138-1R | TGTGAGAGAAGCGGACAA |
| Gnat3-1F | CCCAGCCACTAACATCAAA |
| Gnat3-1R | TTCACAGTTCTTGCATCCCT |
5.8. Western Blot Assay
| Antibody | Product code | Manufacturer | Dilution ratio |
|---|---|---|---|
| GAPDH | AG8015 | Beyotime | 1:2000 |
| Occludin | AF7644 | Beyotime | 1:500 |
| Claudin-1 | AF6504 | Beyotime | 1:500 |
| Gα-gustducin | sc-518163 | SANTA | 1:100 |
5.9. Hematoxylin and Eosin Staining
5.10. Cells and Treatment
5.11. Cell Counting Kit-8 (CCK-8)
5.12. Primary Intestinal Cell Culture
5.13. Lactate Dehydrogenase (LDH) Release Assay
5.14. Statistic Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DM | Nauclea officinalis |
| T2R | bitter taste receptor |
| TNF-α | Tumor Necrosis Factor-alpha |
| IL-6 | Interleukin-6 |
| IL-1β | Interleukin-1 beta |
| IFN-γ | Interferon-gamma |
| SOD | Superoxide Dismutase |
| MDA | Malondialdehyde |
| GSH-PX | Glutathione Peroxidase |
References
- Wu, MH; Li, NX; Zhang, Y.; et al. Textual research on the origin of Nauclea officinalis used as medicine. Journal of Chinese Medicinal Materials 2019, 42, 2709–2714. [Google Scholar]
- Wang, Y.; Liao, J.H.; Sun, L.X. Research progress on Nauclea officinalis and its preparations. Asia-Pacific Traditional Medicine 2018, 14, 80–84. [Google Scholar]
- Huang, L; Miao, HQ; Su, X.J.; et al. Construction of a signal mining model for serious adverse drug reactions based on real-world data: an empirical study using Nauclea officinalis preparations as an example. China Food & Drug Administration Magazine 2024, 10, 102–111. [Google Scholar]
- Okumura, R.; Takeda, K. The role of the mucosal barrier system in maintaining gut symbiosis to prevent intestinal inflammation. Seminars in Immunopathology 2024, 47, 2. [Google Scholar] [CrossRef]
- Neurath, M.F.; Artis, D.; Becker, C. The intestinal barrier: a pivotal role in health, inflammation, and cancer. The Lancet Gastroenterology & Hepatology 2025, 10, 573–92. [Google Scholar] [CrossRef]
- Gieryńska, M.; Szulc-Dąbrowska, L.; Struzik, J.; et al. Integrity of the Intestinal Barrier: The Involvement of Epithelial Cells and Microbiota—A Mutual Relationship. Animals 2022, 12, 145. [Google Scholar] [CrossRef]
- Wang, F.; Graham, W.V.; Wang, Y.; et al. Interferon-gamma and tumor necrosis factor-alpha synergize to induce intestinal epithelial barrier dysfunction by up-regulating myosin light chain kinase expression. Am J Pathol 2005, 166, 409–419. [Google Scholar] [CrossRef]
- Qiu, P; Ishimoto, T; Fu, L.; et al. The Gut Microbiota in Inflammatory Bowel Disease. Front Cell Infect Microbiol 2022, 12, 733992. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, W. Probiotics in reducing obesity by reconfiguring the gut microbiota. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2024, 49, 1042–1051. [Google Scholar] [PubMed]
- Sternini, C.; Rozengurt, E. Bitter taste receptors as sensors of gut luminal contents. Nature Reviews Gastroenterology & Hepatology 2024, 22, 39–53. [Google Scholar] [CrossRef]
- Salzman, NH; Hung, K; Haribhai, D.; et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nature Immunology 2009, 11, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Howitt, MR; Lavoie, S; Michaud, M.; et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 2016, 351, 1329–1333. [Google Scholar] [CrossRef]
- Bevins, C.L.; Salzman, N.H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nat Rev Microbiol 2011, 9, 356–368. [Google Scholar] [CrossRef]
- Vaishnava, S; Behrendt, CL; Ismail, A.S.; et al. Paneth cells directly sense gut commensals and maintain homeostasis at the intestinal host-microbial interface. Proc Natl Acad Sci U S A 2008, 105, 20858–63. [Google Scholar] [CrossRef]
- Salzman, NH; Hung, K; Haribhai, D.; et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat Immunol 2010, 11, 76–83. [Google Scholar] [CrossRef]
- Yang, Y; Li, W; Sun, K.; et al. Berberine ameliorates dextran sulfate sodium -induced colitis through tuft cells and bitter taste signalling. BMC Biology 2024, 22, 280. [Google Scholar] [CrossRef]
- Feng, P; Chai, J; Yi, H.; et al. Aggravated gut inflammation in mice lacking the taste signaling protein α-gustducin. Brain, Behavior, and Immunity 2018, 71, 23–27. [Google Scholar] [CrossRef]
- Kim, RH; Lee, GE; Lee, K.; et al. Anti-inflammatory activities of black raspberry seed ellagitannins and their structural effects on the stimulation of glucagon-like peptide-1 secretion and intestinal bitter taste receptors. Food Funct 2023, 14, 4049–4064. [Google Scholar] [PubMed]
- Wu, Z; Yang, W; Wu, T.; et al. Long term Coptidis Rhizoma intake induce gastrointestinal emptying inhibition and colon barrier weaken via bitter taste receptors activation in mice. Phytomedicine 2025, 136, 156292. [Google Scholar] [CrossRef] [PubMed]
- Lu, YY; Xi, RH; Zheng, X.; et al. Signal transduction mechanisms of taste receptors and their regulation on microorganisms. West China Journal of Stomatology 2017, 35, 549–554. [Google Scholar]
- Thangaiyan, R; Sakwe, AM; Hawkins, A.T.; et al. Functional characterization of novel anti-DEFA5 monoclonal antibody clones 1A8 and 4F5 in inflammatory bowel disease colitis tissues. Inflammation Research 2025, 74, 30. [Google Scholar] [CrossRef] [PubMed]
- Salzman, N.H. Paneth cell defensins and the regulation of the microbiome. Gut Microbes 2014, 1, 401–406. [Google Scholar] [CrossRef]
- Muniz, L.R.; Knosp, C.; Yeretssian, G. Intestinal antimicrobial peptides during homeostasis, infection, and disease. Frontiers in Immunology 2012, 3310. [Google Scholar] [CrossRef]
- Xiao, Y; Lian, H; Zhong, X.S.; et al. Matrix metalloproteinase 7 contributes to intestinal barrier dysfunction by degrading tight junction protein Claudin-7. Frontiers in Immunology 2022, 131020902–1020902. [Google Scholar] [CrossRef]
- Singh, S; Singh, PP; Singh, A.G.; et al. Anti-diabetic medications and risk of pancreatic cancer in patients with diabetes mellitus: a systematic review and meta-analysis. Am J Gastroenterol 2013, 108, 510–9; quiz 520. [Google Scholar] [CrossRef]
- Dixit, R; Lacour, S; Gautier, J.-C.; et al. Cytokines as potential biomarkers of liver toxicity. Cancer Biomarkers 2005, 1, 29–39. [Google Scholar] [CrossRef]
- Zou, YY; Lu, H; Cui, X.Y.; et al. Interpretation and Discussion on Guiding Principles for Real-World Study Design and Statistical Analysis of Medical Devices. China Food & Drug Administration Magazine 2024, 10, 42–49. [Google Scholar]
- Ford, AC; Sperber, AD; Corsetti, M.; et al. Irritable bowel syndrome. Lancet 2020, 396, 1675–1688. [Google Scholar] [CrossRef]
- Barros, L.L.; Farias, A.Q.; Rezaie, A. Gastrointestinal motility and absorptive disorders in patients with inflammatory bowel diseases: Prevalence, diagnosis and treatment. World J Gastroenterol 2019, 25, 4414–4426. [Google Scholar] [CrossRef] [PubMed]
- Pal, S; Laskar, JA; Bhowmick, B.; et al. Helicobacter pylori in peptic ulcer disease: pathogenesis, gastric microbiome, and innovative therapies. Bulletin of the National Research Centre 2025, 49, 50. [Google Scholar] [CrossRef]
- Zhang, Y; Song, F; Yang, M.; et al. Gastrointestinal Dysmotility Predisposes to Colitis through Regulation of Gut Microbial Composition and Linoleic Acid Metabolism. Adv Sci (Weinh) 2024, 11, e2306297. [Google Scholar] [PubMed]
- Britza, SM; Musgrave, IF; Farrington, R.; et al. Intestinal epithelial damage due to herbal compounds - an in vitro study. Drug Chem Toxicol 2023, 46, 247–255. [Google Scholar]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med 2018, 50, 1–9. [Google Scholar]
- Suzuki, T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol Life Sci 2013, 70, 631–659. [Google Scholar]
- Feldman, G.J.; Mullin, J.M.; Ryan, M.P. Occludin: structure, function and regulation. Adv Drug Deliv Rev 2005, 57, 883–917. [Google Scholar]
- Tsukita, S.; Furuse, M. Occludin and claudins in tight-junction strands: leading or supporting players? Trends Cell Biol 1999, 9, 268–273. [Google Scholar] [CrossRef]
- Vivinus-Nébot, M; Frin-Mathy, G; Bzioueche, H.; et al. Functional bowel symptoms in quiescent inflammatory bowel diseases: role of epithelial barrier disruption and low-grade inflammation. Gut 2014, 63, 744–752. [Google Scholar]
- Michielan, A.; D'incà, R. Intestinal Permeability in Inflammatory Bowel Disease: Pathogenesis, Clinical Evaluation, and Therapy of Leaky Gut. Mediators Inflamm 2015, 2015, 628157. [Google Scholar] [PubMed]
- Huang, G; Sun, K; Yin, S.; et al. Burn Injury Leads to Increase in Relative Abundance of Opportunistic Pathogens in the Rat Gastrointestinal Microbiome. Front Microbiol 2017, 8, 1237. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, SN; Momeni, N; Chiti, H.; et al. Higher gut Bacteroidetes and Actinobacteria population in early pregnancy is associated with lower risk of gestational diabetes in the second trimester. BMC Pregnancy Childbirth 2025, 25, 106. [Google Scholar] [CrossRef]
- Palkovicsné Pézsa, N; Kovács, D; Gálfi, P.; et al. Effect of Enterococcus faecium NCIMB 10415 on Gut Barrier Function, Internal Redox State, Proinflammatory Response and Pathogen Inhibition Properties in Porcine Intestinal Epithelial Cells. Nutrients 2022, 14, 1486. [Google Scholar] [CrossRef]
- Hernandez-Patlan, D; Solis-Cruz, B; Pontin, K.P.; et al. Impact of a Bacillus Direct-Fed Microbial on Growth Performance, Intestinal Barrier Integrity, Necrotic Enteritis Lesions, and Ileal Microbiota in Broiler Chickens Using a Laboratory Challenge Model. Front Vet Sci 2019, 6108. [Google Scholar]
- Chavoya-Guardado, MA; Vasquez-Garibay, EM; Ruiz-Quezada, S.L.; et al. Firmicutes, Bacteroidetes and Actinobacteria in Human Milk and Maternal Adiposity. Nutrients 2022, 14, 2887. [Google Scholar] [CrossRef]
- Wang, L; Chai, M; Wang, J.; et al. Bifidobacterium longum relieves constipation by regulating the intestinal barrier of mice. Food Funct 2022, 13, 5037–5049. [Google Scholar] [CrossRef]
- Nie, K; Ma, K; Luo, W.; et al. Roseburia intestinalis: A Beneficial Gut Organism From the Discoveries in Genus and Species. Front Cell Infect Microbiol 2021, 11, 757718. [Google Scholar] [CrossRef]
- He, XQ; Liu, D; Liu, H.Y.; et al. Prevention of Ulcerative Colitis in Mice by Sweet Tea (Lithocarpus litseifolius) via the Regulation of Gut Microbiota and Butyric-Acid-Mediated Anti-Inflammatory Signaling. Nutrients 2022, 14, 2208. [Google Scholar] [CrossRef]
- Bourgonje, AR; Feelisch, M; Faber, K.N.; et al. Oxidative Stress and Redox-Modulating Therapeutics in Inflammatory Bowel Disease. Trends in Molecular Medicine 2020, 26, 1034–1046. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z; He, Z; Emara, A.M.; et al. Effects of malondialdehyde as a byproduct of lipid oxidation on protein oxidation in rabbit meat. Food Chem 2019, 288, 405–412. [Google Scholar] [PubMed]
- Mandal, M; Mamun, MAA; Rakib, A.; et al. Modulation of occludin, NF-κB, p-STAT3, and Th17 response by DJ-X-025 decreases inflammation and ameliorates experimental colitis. Biomed Pharmacother 2025, 185, 117939. [Google Scholar] [PubMed]
- Mostafavi Abdolmaleky, H.; Zhou, J.R. Gut Microbiota Dysbiosis, Oxidative Stress, Inflammation, and Epigenetic Alterations in Metabolic Diseases. Antioxidants (Basel) 2024, 13, 985. [Google Scholar]
- Salzman, N.H.; Underwood, M.A.; Bevins, C.L. Paneth cells, defensins, and the commensal microbiota: a hypothesis on intimate interplay at the intestinal mucosa. Semin Immunol 2007, 19, 70–83. [Google Scholar] [CrossRef]
- Liszt, KI; Wang, Q; Farhadipour, M.; et al. Human intestinal bitter taste receptors regulate innate immune responses and metabolic regulators in obesity. J Clin Invest 2022, 132, 1–16. [Google Scholar] [CrossRef]
- Yang, Y; Li, W; Sun, K.; et al. Berberine ameliorates dextran sulfate sodium -induced colitis through tuft cells and bitter taste signalling. BMC Biol 2024, 22, 280. [Google Scholar]
- Lee, RJ; Xiong, G; Kofonow, J.M.; et al. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J Clin Invest 2012, 122, 4145–4159. [Google Scholar] [CrossRef]
- Pei, XT; Peng, L; Zeng, J.K.; et al. Research progress on chemical constituents and pharmacological effects of Nauclea officinalis and predictive analysis of its quality markers. China Journal of Chinese Materia Medica 2024, 49, 2047–2063. [Google Scholar] [PubMed]
- Liu, R; Hao, YT; Liu, X.R.; et al. Effect of walnut oligopeptides on digestive function. Chinese Journal of Public Health 2024, 40, 50–56. [Google Scholar]
- Müller, M; Van Liefferinge, E; Navarro, M.; et al. CCK and GLP-1 release in response to proteinogenic amino acids using a small intestine ex vivo model in pigs. J Anim Sci 2022, 100. [Google Scholar]
- Müller, M; Xu, C; Navarro, M.; et al. Leucine (and lysine) increased plasma levels of the satiety hormone cholecystokinin (CCK), and phenylalanine of the incretin glucagon-like peptide 1 (GLP-1) after oral gavages in pigs. J Anim Sci 2023, 101. [Google Scholar] [CrossRef]
- Lewis, MA; Patil, K; Ettayebi, K.; et al. Divergent responses of human intestinal organoid monolayers using commercial in vitro cytotoxicity assays. PLoS ONE 2024, 19, e0304526. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




