Submitted:
07 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
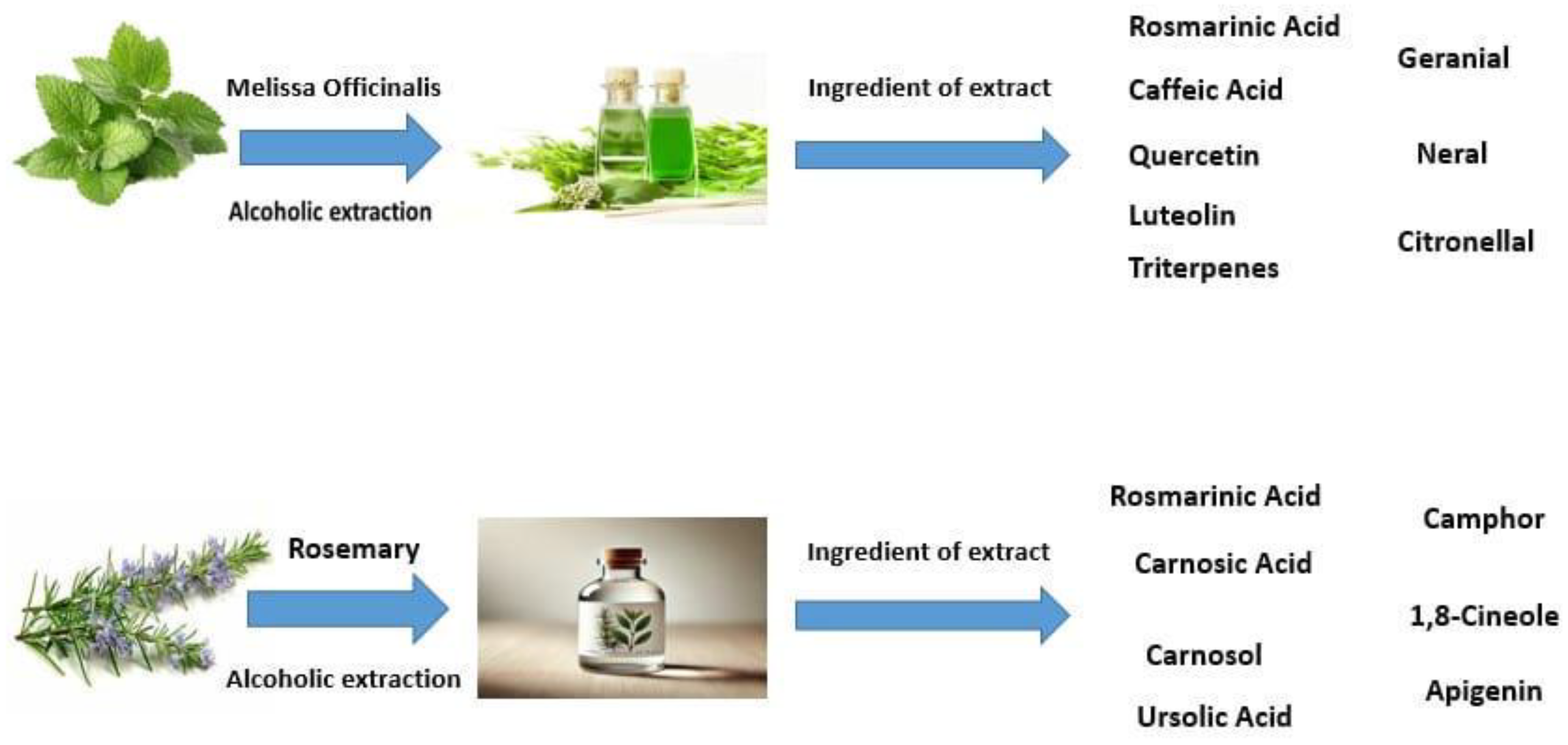
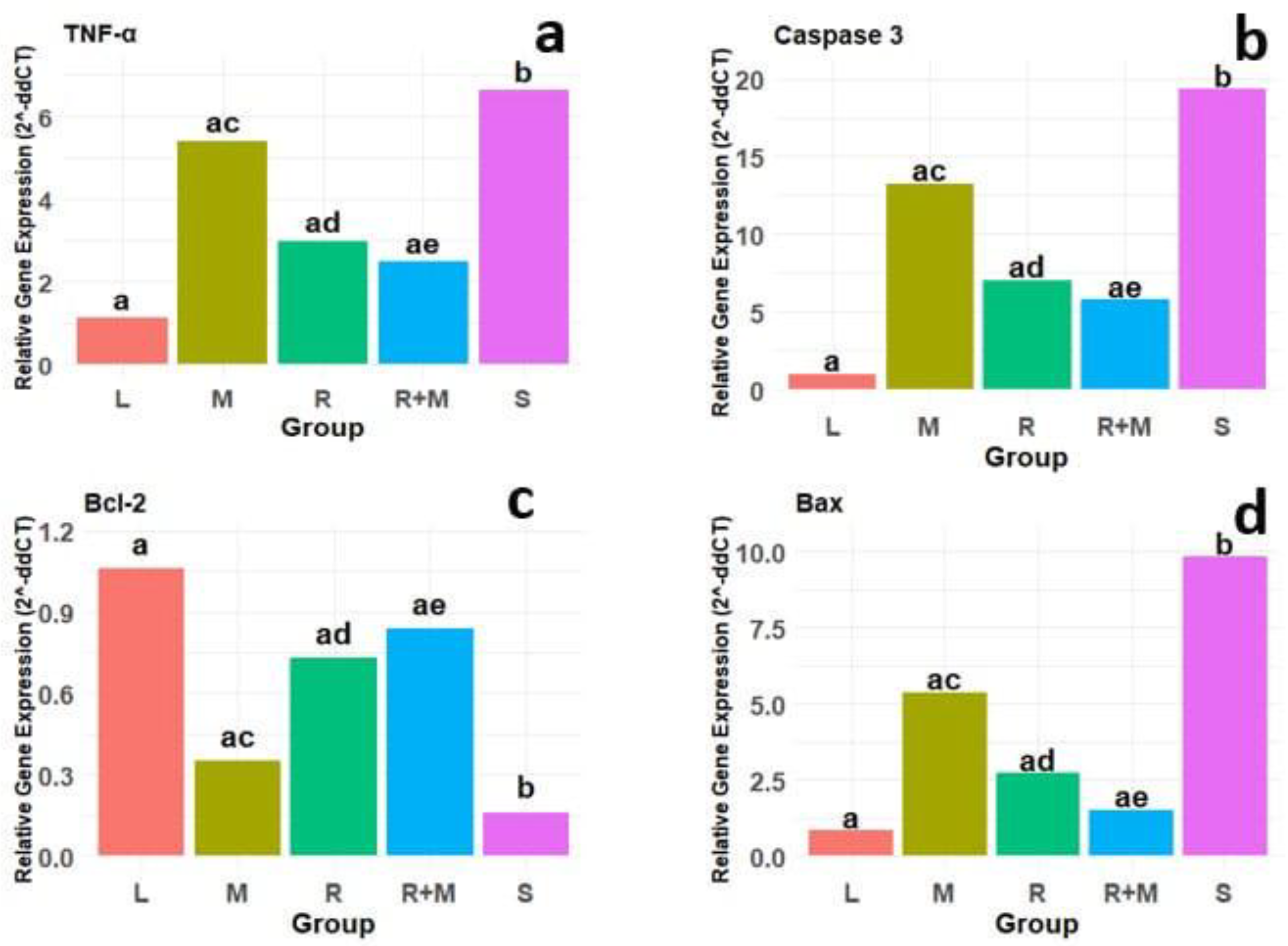
Spinal cord injury (SCI) is linked to a variety of negative outcomes and prognoses that can profoundly affect the lives of individuals, resulting in significant disruptions to multiple facets of their daily activities. A prominent secondary consequence of SCI is the onset of systemic infections, which may disseminate to other organs, including the kidneys, thereby impairing their functionality. Previous studies have demonstrated that the alcoholic extracts of Rosmarinus officinalis and Melissa officinalis possess antioxidant and neuroprotective properties, indicating their potential utility in the treatment and management of SCI and its associated secondary complications. Therefore, this study aimed to examine the combined effects of these extracts on sensory and motor functions, alterations in kidney tissue, and the expression of genes related to inflammation and apoptosis in a rat model of SCI. In this investigation, thirty-five adult male rats were divided into five experimental groups: a control group, a group subjected to spinal cord injury (SCI), a group treated with an alcoholic extract of Melissa officinalis, a group treated with an alcoholic extract of Rosmarinus officinalis, and a group receiving both extracts. The extracts were administered via intraperitoneal injection starting one-day post-SCI and continued for 28 days. Evaluations of sensory and motor functions were performed weekly, while changes in kidney tissue and the expression of genes associated with inflammation and apoptosis were assessed using histomorphometric techniques and quantitative real-time polymerase chain reaction (PCR). The results indicate that the alcoholic Melissa officinalis and Rosmarinus officinalis extracts significantly enhanced sensory and motor functions while reducing the expression levels of genes associated with inflammation (TNF-α) and apoptosis (caspase-3, Bax, and Bcl-2). These findings underscore the potential of these plant extracts in improving the management and treatment of spinal cord injury (SCI) and its secondary effects.
Keywords:
Introduction:
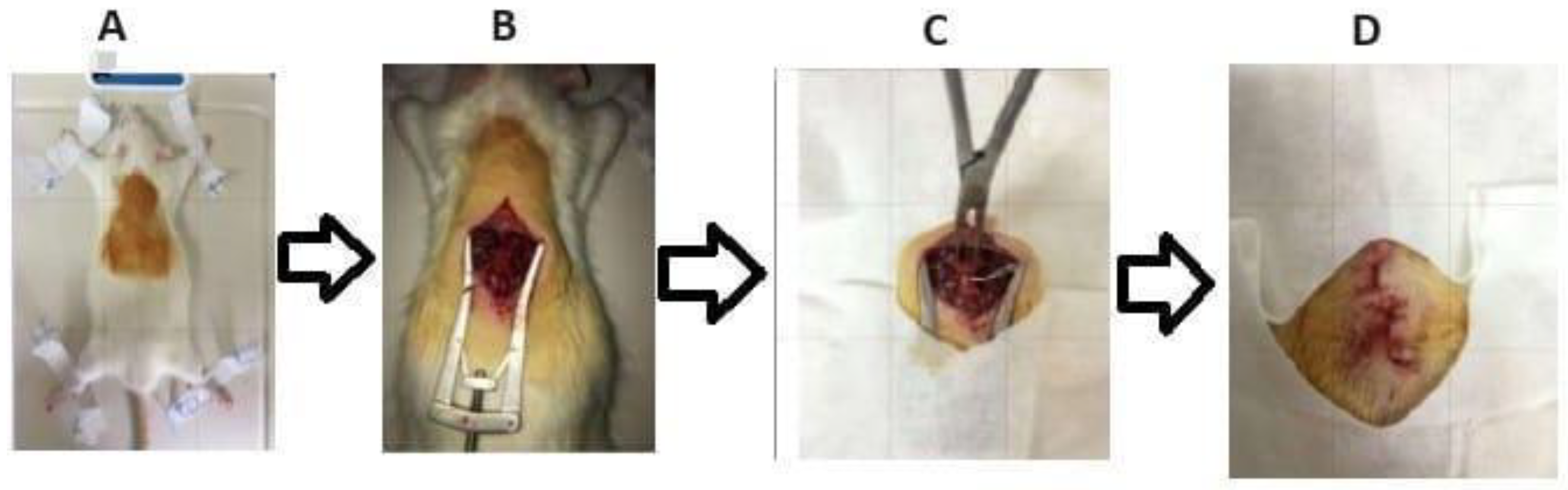
Materials and Methods:
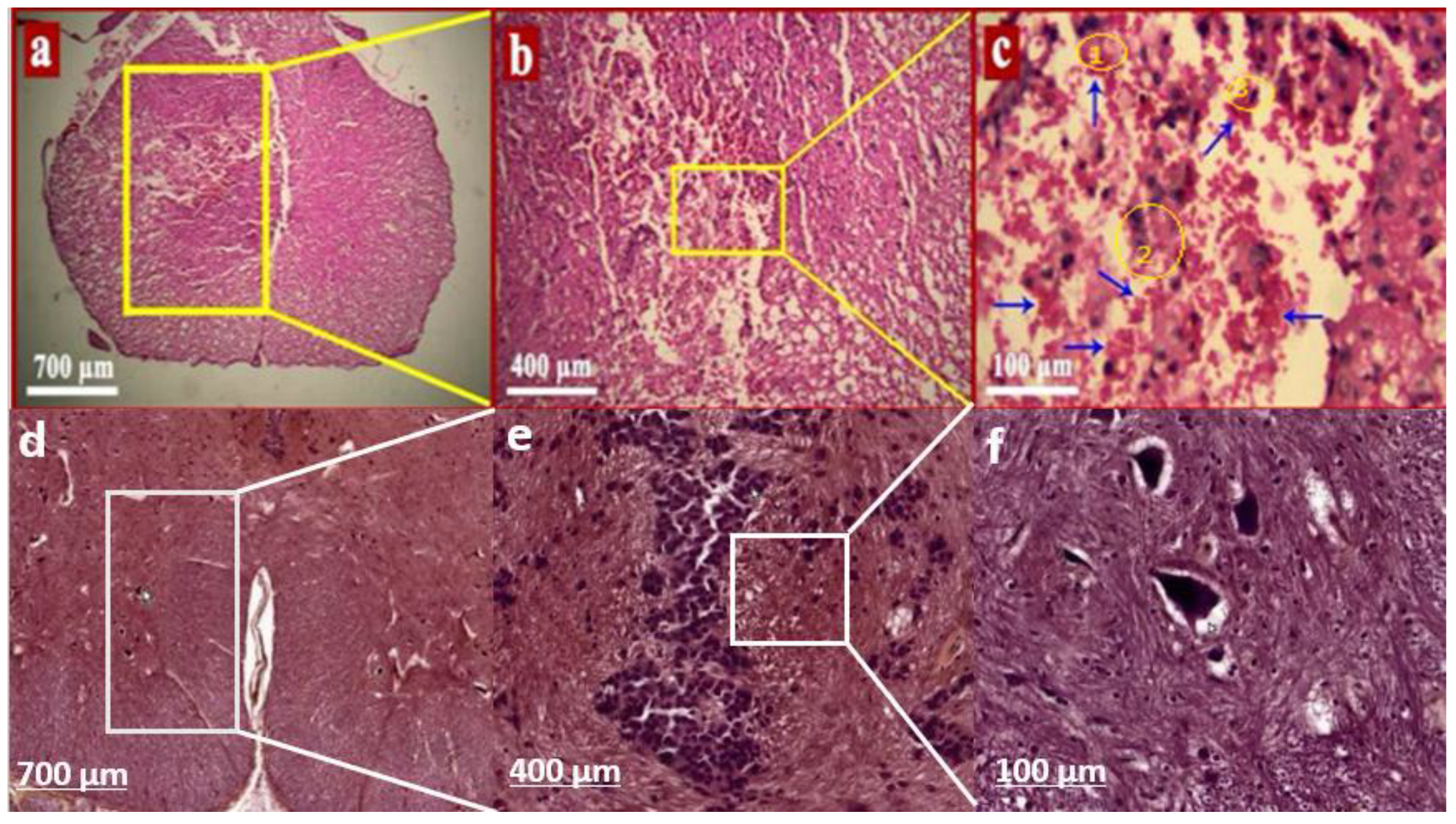
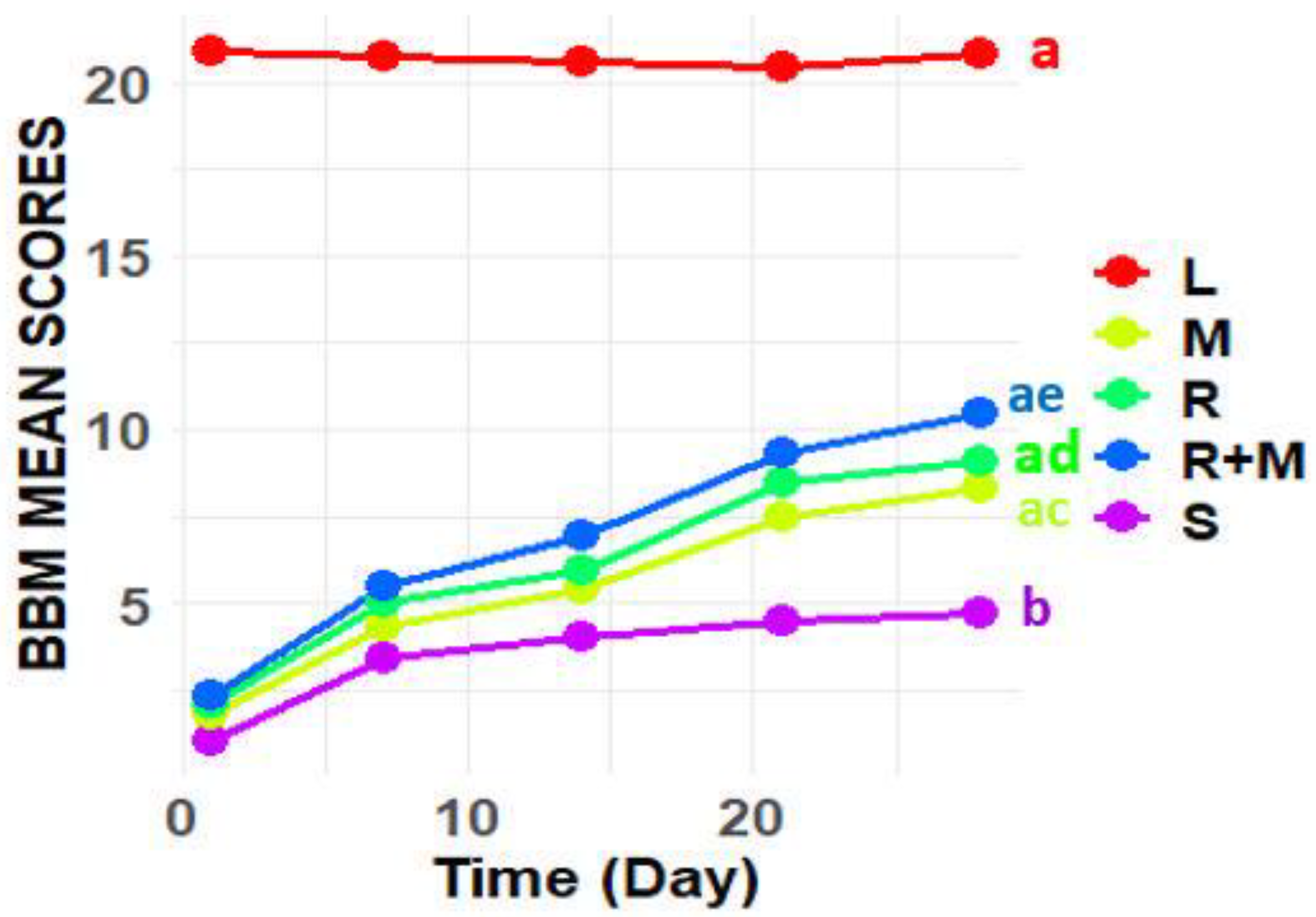
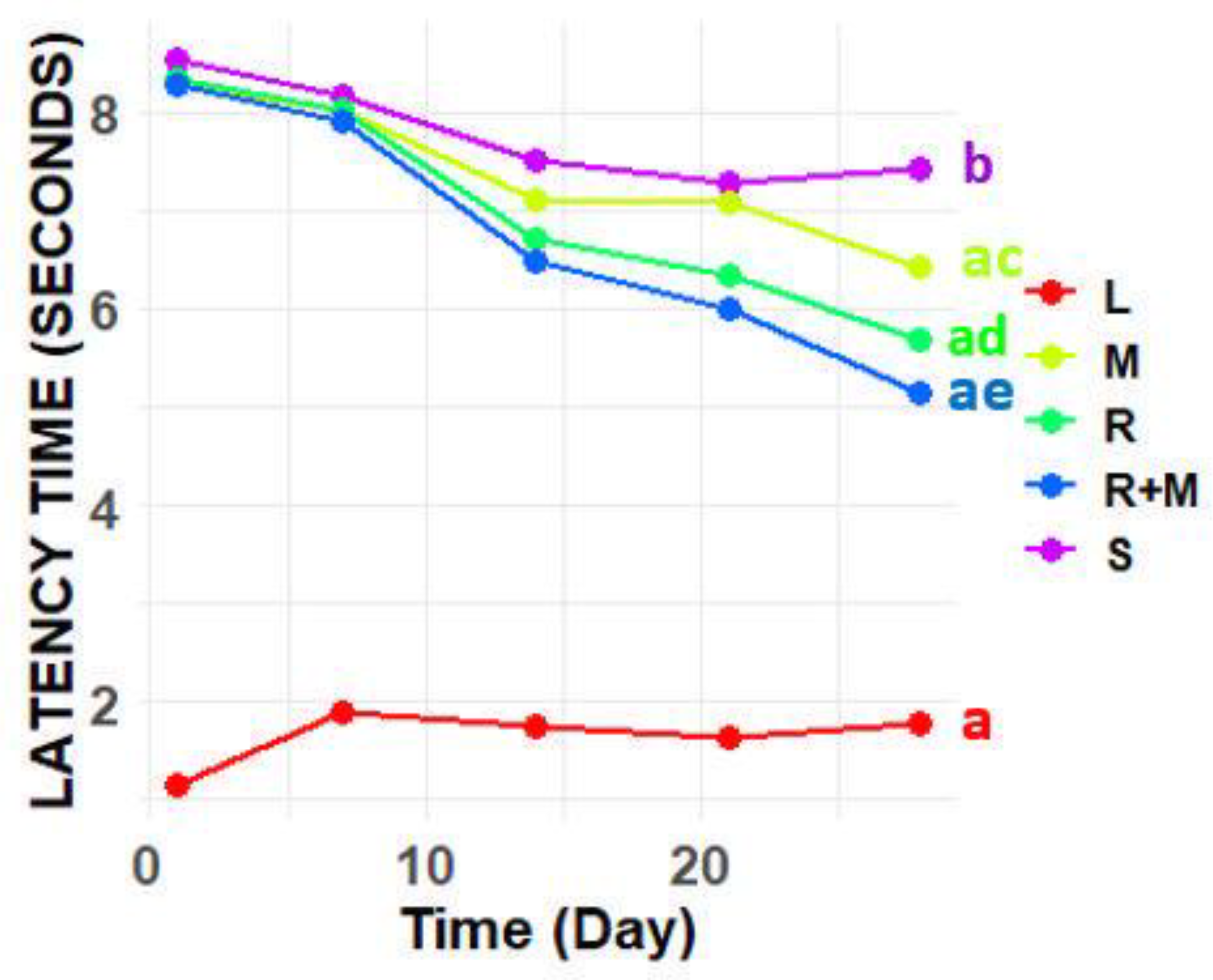
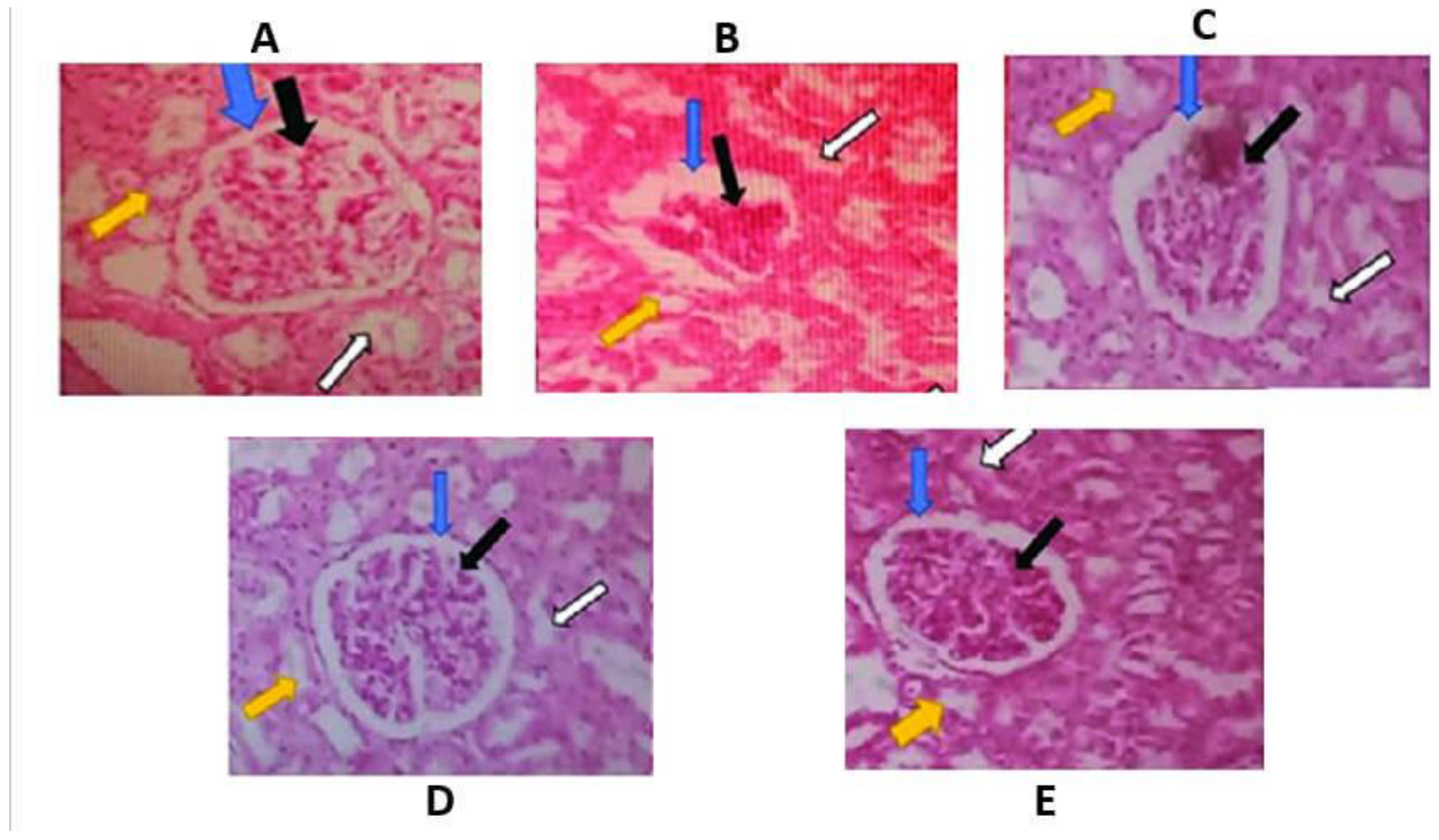
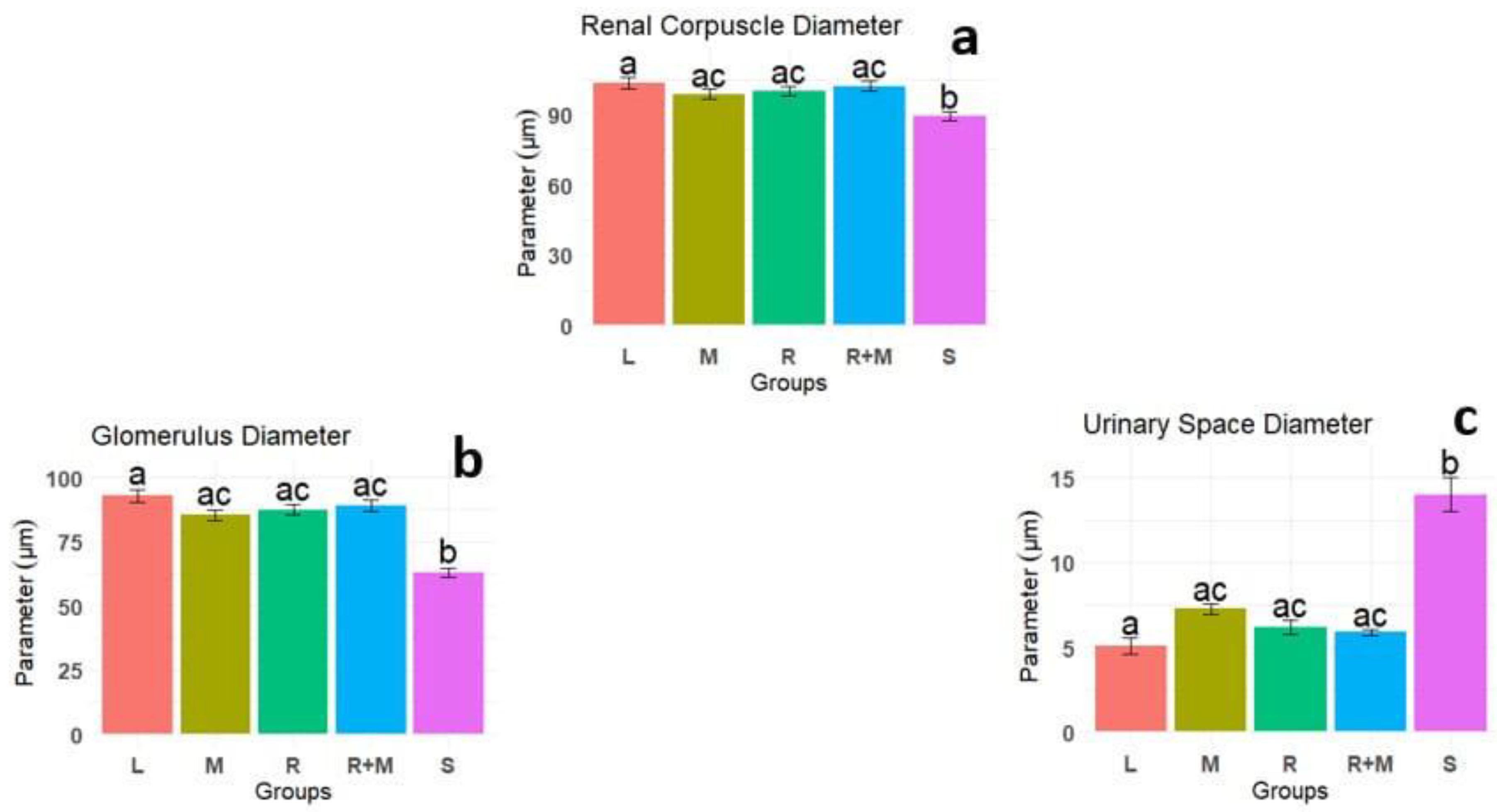
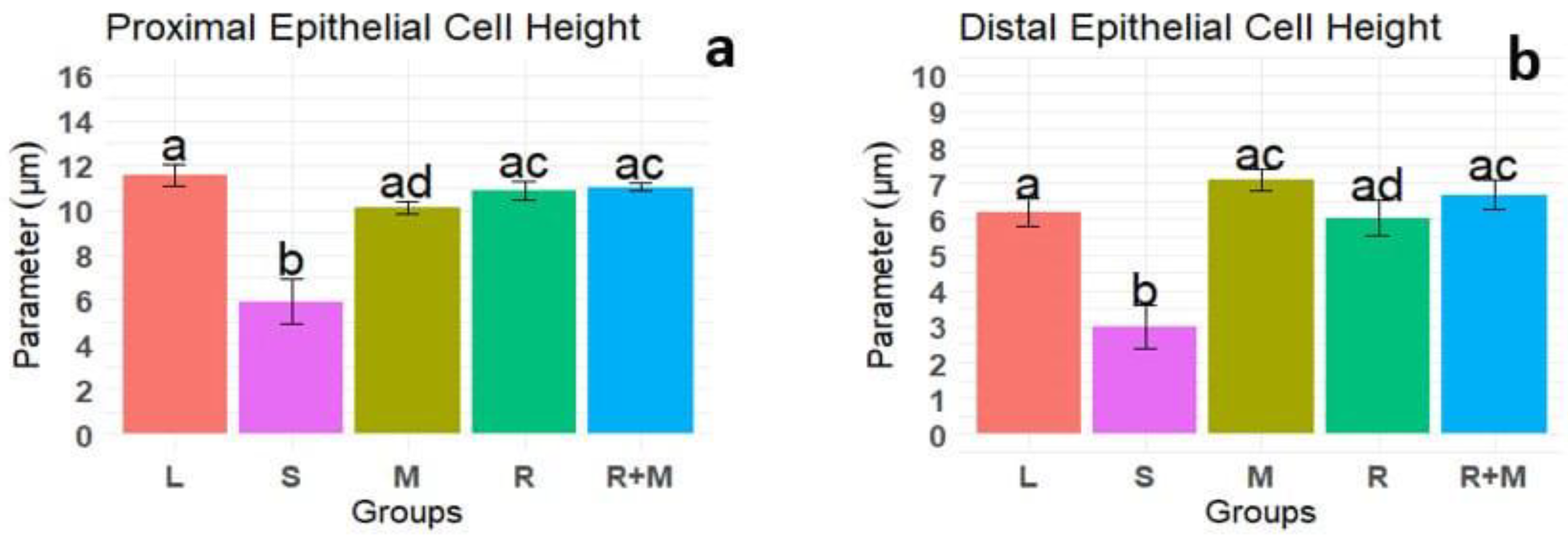
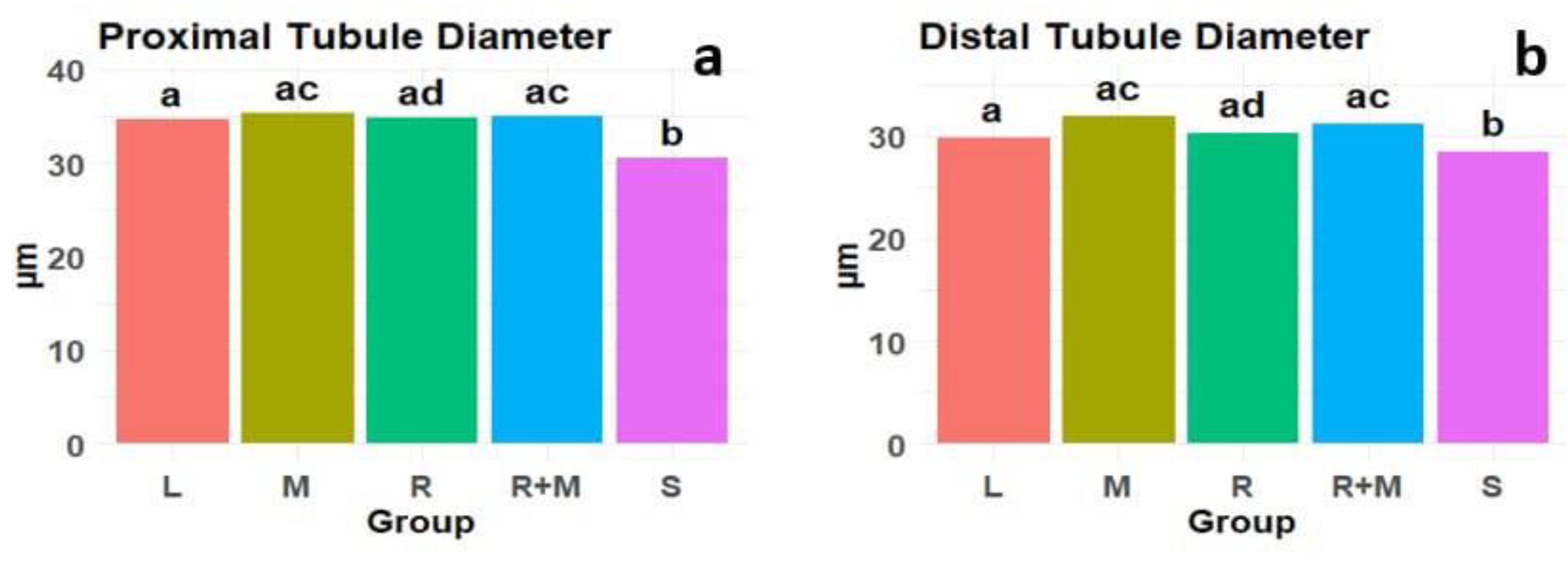
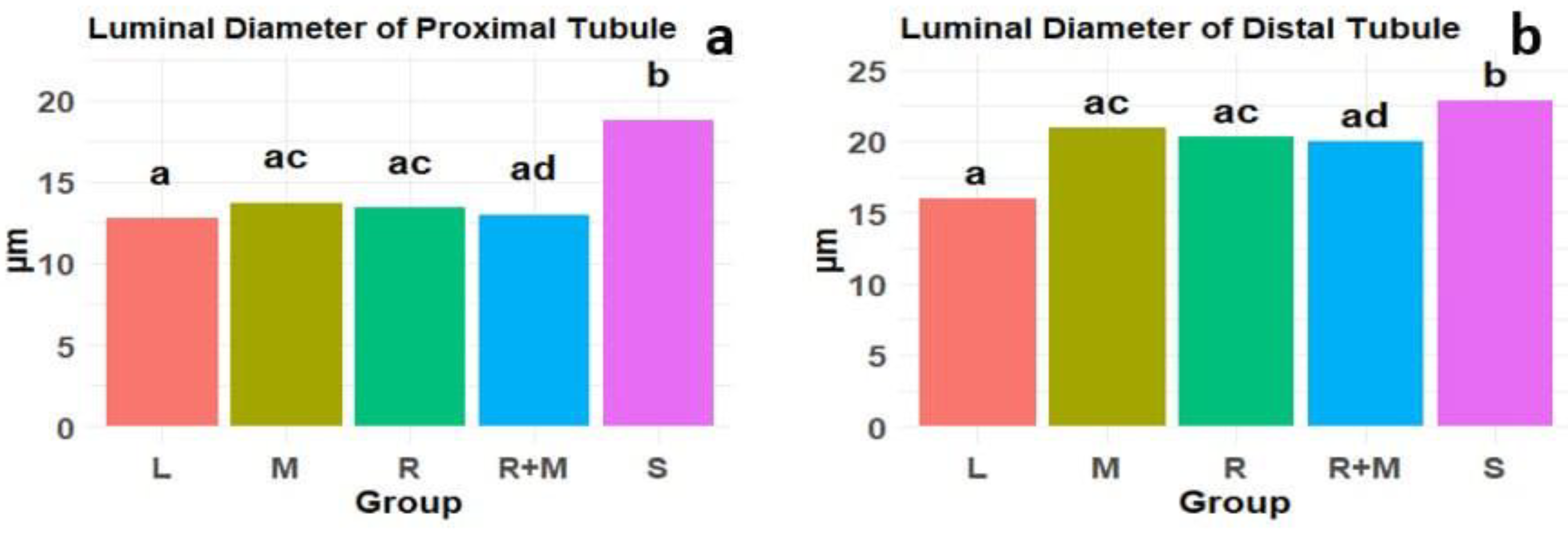
Results
Discussion:
Conclusion:
Abbreviation:
Author contributions
Funding
Data availability
Competing interests
Ethical approval
Consent to participate
Acknowledgements
Consent to publish
References
- Chen K, Yu W, Zheng G, Xu Z, Yang C, Wang Y, Yue Z, Yuan W, Hu B, Chen H: Biomaterial-based regenerative therapeutic strategies for spinal cord injury. NPG Asia Materials 2024, 16(1):5. [CrossRef]
- Xu J, Ding Y, Shi C, Yuan F, Sheng X, Liu Y, Xie Y, Lu H, Duan C, Hu J et al: Identification of Cathepsin B as a Therapeutic Target for Ferroptosis of Macrophage after Spinal Cord Injury. Aging Dis 2023, 15(1):421-443. [CrossRef]
- Mutepfa AR, Hardy JG, Adams CF: Electroactive Scaffolds to Improve Neural Stem Cell Therapy for Spinal Cord Injury. Frontiers in Medical Technology 2022, 4. [CrossRef]
- Ahuja CS, Nori S, Tetreault L, Wilson J, Kwon B, Harrop J, Choi D, Fehlings MG: Traumatic Spinal Cord Injury-Repair and Regeneration. Neurosurgery 2017, 80(3s):S9-s22. [CrossRef]
- Krause JS, Saunders LL: Health, secondary conditions, and life expectancy after spinal cord injury. Arch Phys Med Rehabil 2011, 92(11):1770-1775. [CrossRef]
- Anjum A, Yazid MDi, Fauzi Daud M, Idris J, Ng AMH, Selvi Naicker A, Ismail OHR, Athi Kumar RK, Lokanathan Y: Spinal Cord Injury: Pathophysiology, Multimolecular Interactions, and Underlying Recovery Mechanisms. International Journal of Molecular Sciences 2020, 21(20):7533. [CrossRef]
- Giverso C, Loy N, Lucci G, Preziosi L: Cell orientation under stretch: A review of experimental findings and mathematical modelling. J Theor Biol 2023, 572:111564. [CrossRef]
- Venkatesh K, Ghosh SK, Mullick M, Manivasagam G, Sen D: Spinal cord injury: pathophysiology, treatment strategies, associated challenges, and future implications. Cell Tissue Res 2019, 377(2):125-151. [CrossRef]
- Schaefer SD, Davies BM, Newcombe VFJ, Sutcliffe MPF: Could spinal cord oscillation contribute to spinal cord injury in degenerative cervical myelopathy? Brain Spine 2023, 3:101743. [CrossRef]
- Tran AP, Warren PM, Silver J: The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev 2018, 98(2):881-917. [CrossRef]
- Anwar MA, Al Shehabi TS, Eid AH: Inflammogenesis of Secondary Spinal Cord Injury. Front Cell Neurosci 2016, 10:98. [CrossRef]
- He X, Li Y, Deng B, Lin A, Zhang G, Ma M, Wang Y, Yang Y, Kang X: The PI3K/AKT signalling pathway in inflammation, cell death and glial scar formation after traumatic spinal cord injury: Mechanisms and therapeutic opportunities. Cell Prolif 2022, 55(9):e13275. [CrossRef]
- Clifford T, Finkel Z, Rodriguez B, Jose ph A, Cai L: Current Advancements in Spinal Cord Injury Research-Glial Scar Formation and Neural Regeneration. Cells 2023, 12(6). [CrossRef]
- Gong M, Qi S, Wu Z, Huang Y, Wu L, Wang X, He L, Lin L, Lin D: A novel therapeutic approach to modulate the inflammatory cascade: A timely exogenous local inflammatory response attenuates the sepsis-induced cytokine storm. Cytokine 2024, 176:156533. [CrossRef]
- Rönnbäck C, Hansson E: The Importance and Control of Low-Grade Inflammation Due to Damage of Cellular Barrier Systems That May Lead to Systemic Inflammation. Front Neurol 2019, 10:533. [CrossRef]
- Liu L, Pei FX, Tang KL, Xu JZ, Li QH: Expression and effect of Caspase-3 in neurons after tractive spinal cord injury in rats. Chin J Traumatol 2005, 8(4):220-224.
- Kwiecien JM, Dabrowski W, Dąbrowska-Bouta B, Sulkowski G, Oakden W, Kwiecien-Delaney CJ, Yaron JR, Zhang L, Schutz L, Marzec-Kotarska B et al: Prolonged inflammation leads to ongoing damage after spinal cord injury. PLoS One 2020, 15(3):e0226584. [CrossRef]
- Li R, Shang J, Zhou W, Jiang L, Xie D, Tu G: Overexpression of HIPK2 attenuates spinal cord injury in rats by modulating apoptosis, oxidative stress, and inflammation. Biomed Pharmacother 2018, 103:127-134. [CrossRef]
- Takagi T, Takayasu M, Mizuno M, Yoshimoto M, Yoshida J: Caspase activation in neuronal and glial apoptosis following spinal cord injury in mice. Neurol Med Chir (Tokyo) 2003, 43(1):20-29; discussion 29-30. [CrossRef]
- Brewer KL, Nolan TA: Spinal and supraspinal changes in tumor necrosis factor-alpha expression following excitotoxic spinal cord injury. J Mol Neurosci 2007, 31(1):13-21. [CrossRef]
- Wolf P, Schoeniger A, Edlich F: Pro-apoptotic complexes of BAX and BAK on the outer mitochondrial membrane. Biochim Biophys Acta Mol Cell Res 2022, 1869(10):119317. [CrossRef]
- Parvin S, Williams CR, Jarrett SA, Garraway SM: Spinal Cord Injury Increases Pro-inflammatory Cytokine Expression in Kidney at Acute and Sub-chronic Stages. Inflammation 2021, 44(6):2346-2361. [CrossRef]
- Rodríguez-Romero V, Cruz-Antonio L, Franco-Bourland RE, Guízar-Sahagún G, Castañeda-Hernández G: Changes in renal function during acute spinal cord injury: implications for pharmacotherapy. Spinal Cord 2013, 51(7):528-531. [CrossRef]
- Welk B, Fuller A, Razvi H, Denstedt J: Renal stone disease in spinal-cord-injured patients. J Endourol 2012, 26(8):954-959. [CrossRef]
- Gomez RA, Sequeira-Lopez MLS: Renin cells in homeostasis, regeneration and immune defence mechanisms. Nat Rev Nephrol 2018, 14(4):231-245. [CrossRef]
- Ramanauskiene K, Raudonis R, Majiene D: Rosmarinic Acid and Melissa officinalis Extracts Differently Affect Glioblastoma Cells. Oxid Med Cell Longev 2016, 2016:1564257. [CrossRef]
- Atanasova A, Petrova A, Teneva D, Ognyanov M, Georgiev Y, Nenov N, Denev P: Subcritical Water Extraction of Rosmarinic Acid from Lemon Balm (Melissa officinalis L.) and Its Effect on Plant Cell Wall Constituents. Antioxidants 2023, 12(4):888. [CrossRef]
- Ulbricht C, Abrams TR, Brigham A, Ceurvels J, Clubb J, Curtiss W, Kirkwood CD, Giese N, Hoehn K, Iovin R et al: An evidence-based systematic review of rosemary (Rosmarinus officinalis) by the Natural Standard Research Collaboration. J Diet Suppl 2010, 7(4):351-413. [CrossRef]
- Petrisor G, Motelica L, Craciun LN, Oprea OC, Ficai D, Ficai A: Melissa officinalis: Composition, Pharmacological Effects and Derived Release Systems-A Review. Int J Mol Sci 2022, 23(7). [CrossRef]
- Bayat M, Azami Tameh A, Hossein Ghahremani M, Akbari M, Mehr SE, Khanavi M, Hassanzadeh G: Neuroprotective properties of Melissa officinalis after hypoxic-ischemic injury both in vitro and in vivo. Daru 2012, 20(1):42. [CrossRef]
- Lešnik S, Bren U: Mechanistic Insights into Biological Activities of Polyphenolic Compounds from Rosemary Obtained by Inverse Molecular Docking. Foods 2022, 11(1):67. [CrossRef]
- Shang AJ, Yang Y, Wang HY, Tao BZ, Wang J, Wang ZF, Zhou DB: Spinal cord injury effectively ameliorated by neuroprotective effects of rosmarinic acid. Nutr Neurosci 2017, 20(3):172-179. [CrossRef]
- Ghasemzadeh MR, Amin B, Mehri S, Mirnajafi-Zadeh SJ, Hosseinzadeh H: Effect of alcoholic extract of aerial parts of Rosmarinus officinalis L. on pain, inflammation and apoptosis induced by chronic constriction injury (CCI) model of neuropathic pain in rats. J Ethnopharmacol 2016, 194:117-130. [CrossRef]
- Ghazizadeh J, Hamedeyazdan S, Torbati M, Farajdokht F, Fakhari A, Mahmoudi J, Araj-Khodaei M, Sadigh-Eteghad S: Melissa officinalis L. hydro-alcoholic extract inhibits anxiety and depression through prevention of central oxidative stress and apoptosis. Exp Physiol 2020, 105(4):707-720. [CrossRef]
- Mohamed WAM, Abd-Elhakim YM, Farouk SM: Protective effects of ethanolic extract of rosemary against lead-induced hepato-renal damage in rabbits. Experimental and Toxicologic Pathology 2016, 68(8):451-461. [CrossRef]
- Sheikhian L, Jamalifard Y: Multi-walled carbon nanotube-based dispersive solid phase extraction with following back-extraction for HPLC/UV determination of Rosmarinic acid in lemon balm and Rosemary plant samples. Journal of the Indian Chemical Society 2022, 99(8):100595. [CrossRef]
- Manolescu D, Uță G-a, Șuțan A, Ducu C, Din A, Moga S, Negrea D, Biță A, Bejena-Ru L, Bejenaru C-n: Biogenic synthesis of noble metal nanoparticles using Melissa officinalis L. and Salvia officinalis L. extracts and evaluation of their biosafety potential. Caryologia 2022, 75(3):65-83. [CrossRef]
- Appiah KS, Mardani HK, Omari RA, Eziah VY, Ofosu-Anim J, Onwona-Agyeman S, Amoatey CA, Kawada K, Katsura K, Oikawa Y et al: Involvement of Carnosic Acid in the Phytotoxicity of Rosmarinus officinalis Leaves. Toxins 2018, 10(12):498. [CrossRef]
- Hosseini SR, Kaka G, Joghataei MT, Hooshmandi M, Sadraie SH, Yaghoobi K, Mohammadi A: Assessment of Neuroprotective Properties of Melissa officinalis in Combination With Human Umbilical Cord Blood Stem Cells After Spinal Cord Injury. ASN Neuro 2016, 8(6). [CrossRef]
- Santos DRD, Teixeira RKC, Araújo NP, Calvo FC, Duarte TB, Ataíde LAP, Chaves RHF, Barros RSM: A new anesthetic protocol to medullary nerve roots access in rats. Acta Cir Bras 2021, 36(9):e360908. [CrossRef]
- Dasari VR, Spomar DG, Gondi CS, Sloffer CA, Saving KL, Gujrati M, Rao JS, Dinh DH: Axonal remyelination by cord blood stem cells after spinal cord injury. J Neurotrauma 2007, 24(2):391-410. [CrossRef]
- Byrnes KR, Fricke ST, Faden AI: Neuropathological differences between rats and mice after spinal cord injury. J Magn Reson Imaging 2010, 32(4):836-846. [CrossRef]
- Lin CY, Androjna C, Rozic R, Nguyen B, Parsons B, Midura RJ, Lee YS: Differential Adaptations of the Musculoskeletal System after Spinal Cord Contusion and Transection in Rats. J Neurotrauma 2018, 35(15):1737-1744. [CrossRef]
- Dinh P, Hazel A, Palispis W, Suryadevara S, Gupta R: Functional assessment after sciatic nerve injury in a rat model. Microsurgery: Official Journal of the International Microsurgical Society and the European Federation of Societies for Microsurgery 2009, 29(8):644-649. [CrossRef]
- Hayashibe M, Homma T, Fujimoto K, Oi T, Yagi N, Kashihara M, Nishikawa N, Ishizumi Y, Abe S, Hashimoto H et al: Locomotor improvement of spinal cord-injured rats through treadmill training by forced plantar placement of hind paws. Spinal Cord 2016, 54(7):521-529. [CrossRef]
- Huot-Lavoie M, Ting WK-C, Demers M, Mercier C, Ethier C: Impaired Motor Learning Following a Pain Episode in Intact Rats. Frontiers in Neurology 2019, 10. [CrossRef]
- Hosseini SR, Kaka G, Joghataei MT, Hooshmandi M, Sadraie SH, Yaghoobi K, Mansoori K, Mohammadi A: Coadministration of Dexamethasone and Melissa officinalis Has Neuroprotective Effects in Rat Animal Model with Spinal Cord Injury. Cell J 2017, 19(1):102-116. [CrossRef]
- Studzińska-Sroka E, Majchrzak-Celińska A, Bańdurska M, Rosiak N, Szwajgier D, Baranowska-Wójcik E, Szymański M, Gruszka W, Cielecka-Piontek J: Is Caperatic Acid the Only Compound Responsible for Activity of Lichen Platismatia glauca within the Nervous System? Antioxidants (Basel) 2022, 11(10). [CrossRef]
- Su X, Jing X, Jiang W, Li M, Liu K, Teng M, Wang D, Meng L, Zhang Y, Ji W: Curcumin-Containing polyphosphazene nanodrug for Anti-Inflammation and nerve regeneration to improve functional recovery after spinal cord injury. Int J Pharm 2023, 642:123197. [CrossRef]
- Wang J, Li H, Ren Y, Yao Y, Hu J, Zheng M, Ding Y, Chen YY, Shen Y, Wang LL et al: Local Delivery of β-Elemene Improves Locomotor Functional Recovery by Alleviating Endoplasmic Reticulum Stress and Reducing Neuronal Apoptosis in Rats with Spinal Cord Injury. Cell Physiol Biochem 2018, 49(2):595-609. [CrossRef]
- Caleja C, Barros L, Barreira JCM, Ciric A, Sokovic M, Calhelha RC, Beatriz M, Oliveira PP, Ferreira I: Suitability of lemon balm (Melissa officinalis L.) extract rich in rosmarinic acid as a potential enhancer of functional properties in cupcakes. Food Chem 2018, 250:67-74. [CrossRef]
- Moore J, Yousef M, Tsiani E: Anticancer Effects of Rosemary (Rosmarinus officinalis L.) Extract and Rosemary Extract Polyphenols. Nutrients 2016, 8(11). [CrossRef]
- Jacotet-Navarro M, Laguerre M, Fabiano-Tixier AS, Tenon M, Feuillère N, Bily A, Chemat F: What is the best ethanol-water ratio for the extraction of antioxidants from rosemary? Impact of the solvent on yield, composition, and activity of the extracts. Electrophoresis 2018. [CrossRef]
- Sabogal-Guáqueta AM, Hobbie F, Keerthi A, Oun A, Kortholt A, Boddeke E, Dolga A: Linalool attenuates oxidative stress and mitochondrial dysfunction mediated by glutamate and NMDA toxicity. Biomedicine & Pharmacotherapy 2019, 118:109295. [CrossRef]
- Bahtiyarca R: THE ESSENTIAL OIL OF LEMON BALM (Melissa officinalis L.), ITS COMPONENTS AND USING FIELDS. Jornal of Faculty of Agriculture, Omu 2006, 21:116-121.
- Shimojo Y, Kosaka K, Noda Y, Shimizu T, Shirasawa T: Effect of rosmarinic acid in motor dysfunction and life span in a mouse model of familial amyotrophic lateral sclerosis. J Neurosci Res 2010, 88(4):896-904. [CrossRef]
- Luo Y, Fu C, Wang Z, Zhang Z, Wang H, Liu Y: Mangiferin attenuates contusive spinal cord injury in rats through the regulation of oxidative stress, inflammation and the Bcl-2 and Bax pathway. Mol Med Rep 2015, 12(5):7132-7138. [CrossRef]
- Cheng YY, Zhao HK, Chen LW, Yao XY, Wang YL, Huang ZW, Li GP, Wang Z, Chen BY: Reactive astrocytes increase expression of proNGF in the mouse model of contused spinal cord injury. Neurosci Res 2020, 157:34-43. [CrossRef]
- Sun F, Zhang H, Huang T, Shi J, Wei T, Wang Y: miRNA-221 Regulates Spinal Cord Injury-Induced Inflammatory Response through Targeting TNF-α Expression. Biomed Res Int 2021, 2021:6687963. [CrossRef]
- Peng XM, Zhou ZG, Glorioso JC, Fink DJ, Mata M: Tumor necrosis factor-alpha contributes to below-level neuropathic pain after spinal cord injury. Ann Neurol 2006, 59(5):843-851. [CrossRef]
- Liu J, Peng L, Li J: The Lipoxin A4 Receptor Agonist BML-111 Alleviates Inflammatory Injury and Oxidative Stress in Spinal Cord Injury. Med Sci Monit 2020, 26:e919883. [CrossRef]
- Serapio-Palacios A, Navarro-Garcia F: EspC, an Autotransporter Protein Secreted by Enteropathogenic Escherichia coli, Causes Apoptosis and Necrosis through Caspase and Calpain Activation, Including Direct Procaspase-3 Cleavage. mBio 2016, 7(3). [CrossRef]
- Verma S, Singh A, Mishra A: Complex disruption effect of natural polyphenols on Bcl-2-Bax: molecular dynamics simulation and essential dynamics study. J Biomol Struct Dyn 2015, 33(5):1094-1106. [CrossRef]
- Kotipatruni RR, Dasari VR, Veeravalli KK, Dinh DH, Fassett D, Rao JS: p53- and Bax-mediated apoptosis in injured rat spinal cord. Neurochem Res 2011, 36(11):2063-2074. [CrossRef]
- Wu C, Zhang H, Hong H, Chen C, Chen J, Zhang J, Xue P, Jiang J, Cui Z: E3 ubiquitin ligase Triad1 promotes neuronal apoptosis by regulating the p53-caspase3 pathway after spinal cord injury. Somatosens Mot Res 2022, 39(1):21-28. [CrossRef]
- Akbari-Gharalari N, Ghahremani-Nasab M, Naderi R, Aliyari-Serej Z, Karimipour M, Shahabi P, Ebrahimi-Kalan A: Improvement of spinal cord injury symptoms by targeting the Bax/Bcl2 pathway and modulating TNF-α/IL-10 using Platelet-Rich Plasma exosomes loaded with dexamethasone. AIMS Neurosci 2023, 10(4):332-353. [CrossRef]
- Coelho VR, Viau CM, Staub RB, De Souza MS, Pflüger P, Regner GG, Pereira P, Saffi J: Rosmarinic Acid Attenuates the Activation of Murine Microglial N9 Cells through the Downregulation of Inflammatory Cytokines and Cleaved Caspase-3. Neuroimmunomodulation 2017, 24(3):171-181. [CrossRef]
- Bounihi A, Hajjaj G, Alnamer R, Cherrah Y, Zellou A: In Vivo Potential Anti-Inflammatory Activity of Melissa officinalis L. Essential Oil. Adv Pharmacol Sci 2013, 2013:101759. [CrossRef]
- Christopoulou SD, Androutsopoulou C, Hahalis P, Kotsalou C, Vantarakis A, Lamari FN: Rosemary Extract and Essential Oil as Drink Ingredients: An Evaluation of Their Chemical Composition, Genotoxicity, Antimicrobial, Antiviral, and Antioxidant Properties. Foods 2021, 10(12). [CrossRef]
- Miraj S, Rafieian K, Kiani S: Melissa officinalis L: A Review Study With an Antioxidant Prospective. J Evid Based Complementary Altern Med 2017, 22(3):385-394. [CrossRef]
- Ferreira A, Proença C, Serralheiro ML, Araújo ME: The in vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from Portugal. J Ethnopharmacol 2006, 108(1):31-37. [CrossRef]
- Eslami Farsani M, Razavi S, Rasoolijazi H, Esfandiari E, Seyedebrahimi R, Ababzadeh S: Neuroprotective effects of rosemary extract on white matter of prefrontal cortex in old rats. Iran J Basic Med Sci 2024, 27(4):518-523. [CrossRef]
- Zhou T, Wang J, Lin Z, Zhu H, Hu W, Zhang R, Chen X: Abietane diterpenoids with anti-neuroinflammation activity from Rosmarinus officinalis. Fitoterapia 2024, 174:105866. [CrossRef]
- Kara M, Sahin S, Rabbani F, Oztas E, Hasbal-Celikok G, Kanımdan E, Kocyigit A, Kanwal A, Wade U, Yakunina A et al: An in vitro analysis of an innovative standardized phospholipid carrier-based Melissa officinalis L. extract as a potential neuromodulator for emotional distress and related conditions. Front Mol Biosci 2024, 11:1359177. [CrossRef]
- Fareed SA, Yousef EM, Abd El-Moneam SM: Assessment of Effects of Rosemary Essential Oil on the Kidney Pathology of Diabetic Adult Male Albino Rats. Cureus 2023, 15(3):e35736. [CrossRef]
- salehi A: Effects of Melissa officinalis Alcoholic Extract on Kidney Tissue and Apoptosis Gene Expression in a Wistar Rat Model With Spinal Cord Injury. 1403.
- El-Desouky MA, Mahmoud MH, Riad BY, Taha YM: Nephroprotective effect of green tea, rosmarinic acid and rosemary on N-diethylnitrosamine initiated and ferric nitrilotriacetate promoted acute renal toxicity in Wistar rats. Interdiscip Toxicol 2019, 12(2):98-110. [CrossRef]
- Şengel N, Köksal Z, Dursun AD, Kurtipek Ö, Sezen Ş C, Arslan M, Kavutçu M: Effects of Dexmedetomidine Administered Through Different Routes on Kidney Tissue in Rats with Spinal Cord Ischaemia-Reperfusion Injury. Drug Des Devel Ther 2022, 16:2229-2239. [CrossRef]
- Choi JY, Jang TW, Song PH, Choi SH, Ku SK, Song CH: Combination Effects of Metformin and a Mixture of Lemon Balm and Dandelion on High-Fat Diet-Induced Metabolic Alterations in Mice. Antioxidants (Basel) 2022, 11(3). [CrossRef]
- Elsayed H, komy A, El-Shewy E, Elsayed F: Ameliorative Effect of Cinnamon and Rosemary Oils in Acrylamide–Induced Hepatic Injury in Rats. Bionatura 2024, 9:1-12. [CrossRef]
| Genes | Primer Sequence Forward | Primer Sequence Reverse | product size(bp) | Tm melt Forward (°C) | Tm melt Reverse (°C) |
| B2m (reference) | CTTTCTACATCCTGGCTCACA | GTCCAGATGATTCAGAGCTC |
91 |
52.40 |
51.78 |
| TNFa | CCCTCACACTCAGATCATCTTCT |
TCAGCCACTCCAGCTGCTCCTC |
94 |
55.27 |
60.43 |
| bcl2 |
GAGTGGGATACTGGAGATGAA |
TGGTAGCGACGAGAGAAGT |
90 |
52.40 |
51.09 |
| casp3 |
AAGTGATGGAGATGAAGGAGT |
CAGGCGTGAATGATGAAGAG |
90 |
51.00 |
51.78 |
| Bax |
GGAGACACCTGAGCTGAC |
CAGCAATCATCCTCTGCAGCT |
89 |
52.60 |
54.36 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




