1. Introduction
Adults diagnosed with attention-deficit/hyperactivity disorder often describe difficulties that go well beyond the core triad of inattention, hyperactivity and impulsivity. Problems with planning, affect regulation, anxiety and an intense drive toward perfectionism frequently persist even after guideline-directed treatment with atomoxetine or standard stimulant doses has been optimised [
1]. Clinicians therefore continue to look for add-on strategies that address this residual executive and emotional load without an undue side-effect burden.
Work in the fast-acting antidepressant field has shifted attention to glutamatergic signalling—specifically, to the idea that therapeutic benefit follows a transient tilt away from N-methyl-D-aspartate (NMDA) receptor traffic toward α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) throughput. Such a shift appears to set off a cascade involving brain-derived neurotrophic factor release, mTOR activation and rapid synaptogenesis, changes that unfold within hours of dose administration [
2,
3]. In animal models the behavioural effects of ketamine disappear if AMPA receptors are blocked, and they can be reproduced or amplified by drugs that act as positive allosteric modulators at the same receptors [
4,
5].
Two readily available agents touch this pathway from different angles. Methylphenidate, widely used for ADHD, produces a brief, dopamine-dependent rise in prefrontal glutamate [
6]. Piracetam, on the market for decades as a “nootropic,” enhances AMPA-mediated currents and promotes long-term potentiation while carrying little toxic baggage even at gram-level doses [
7,
8,
9]. Early controlled work and a large pool of off-label experience hint that the two drugs, taken together, may yield more than the sum of their parts in persons with attentional disorders [10).
The case that follows concerns a 28-year-old woman whose symptoms levelled off on atomoxetine yet improved strikingly when low-dose extended-release methylphenidate (18 mg) was combined with piracetam (1 200 mg daily). The speed and breadth of the response echo reports emerging from oral “ketamine-class” glutamatergic combinations [
11,
12] but were achieved with medications that are inexpensive, familiar and legally obtainable in many jurisdictions. Her clinical course offers a window onto a potential shortcut to AMPA-dominant neuroplasticity for selected adults with ADHD.
2. Methods
This account draws on routine clinical care delivered at Cheung Ngo Medical, a private outpatient psychiatry practice located in Tsim Sha Tsui, Hong Kong. The patient, a 28-year-old woman, reviewed and signed a written consent form permitting the anonymised use of her clinical information, rating-scale scores and self-kept logs for teaching and publication. The form meets the clinic’s standard for single-case reporting and conforms to local ethical guidance; no additional institutional approval was required.
2.1. Index visit and baseline testing
The index consultation took place on 15 November 2025. A structured clinical interview was conducted to confirm attention-deficit/hyperactivity disorder according to DSM-5 criteria and to screen for mood and anxiety comorbidity. Symptom severity was quantified with the Patient Health Questionnaire-9 (PHQ-9) and the Generalised Anxiety Disorder-7 (GAD-7). Objective attention and response-control were assessed with a computerised continuous-performance test that recorded omission and commission errors, mean reaction time and intra-individual variability. In addition, the patient completed a timed serial-search task in which she located target numbers within a 10 × 10 grid; total hits and completion time were recorded.
During the same visit she received a detailed explanation of the theoretical basis for augmenting Atomoxetine (only) with an AMPA-receptor positive allosteric modulator such as piracetam. Options for stimulant titration were reviewed, and she was encouraged to document any cognitive, motivational or emotional changes in a daily log.
2.2. Naturalistic trial and remote follow-up
After the clinic visit the patient returned to mainland China. Atomoxetine was stopped and lisdexamfetamine was not initiated. She tried extended-release methylphenidate with and without piracetam 1 200 mg/day, and kept a handwritten diary for all her feelings and observations.
A follow-up consultation was carried out on 5 December 2025 via encrypted WhatsApp messaging. At that point she forwarded scanned images of her diary and a structured medication log. Treatment recommendations were adjusted on the basis of these materials and her contemporaneous symptom report.
2.3. Data sources and handling
The present report is confined to information obtained from:
1) electronic clinic notes and rating-scale results from the index visit;
2) the computer-generated output of the continuous-performance and grid-search tasks;
3) pharmacy records confirming prescription details;
4) the patient’s original diary entries; and
5) verbatim excerpts from the encrypted WhatsApp correspondence.
No laboratory studies or neuroimaging were undertaken. All potentially identifying particulars—geographic, occupational or personal—have been removed or modified to preserve confidentiality.
2.4. Case Presentation
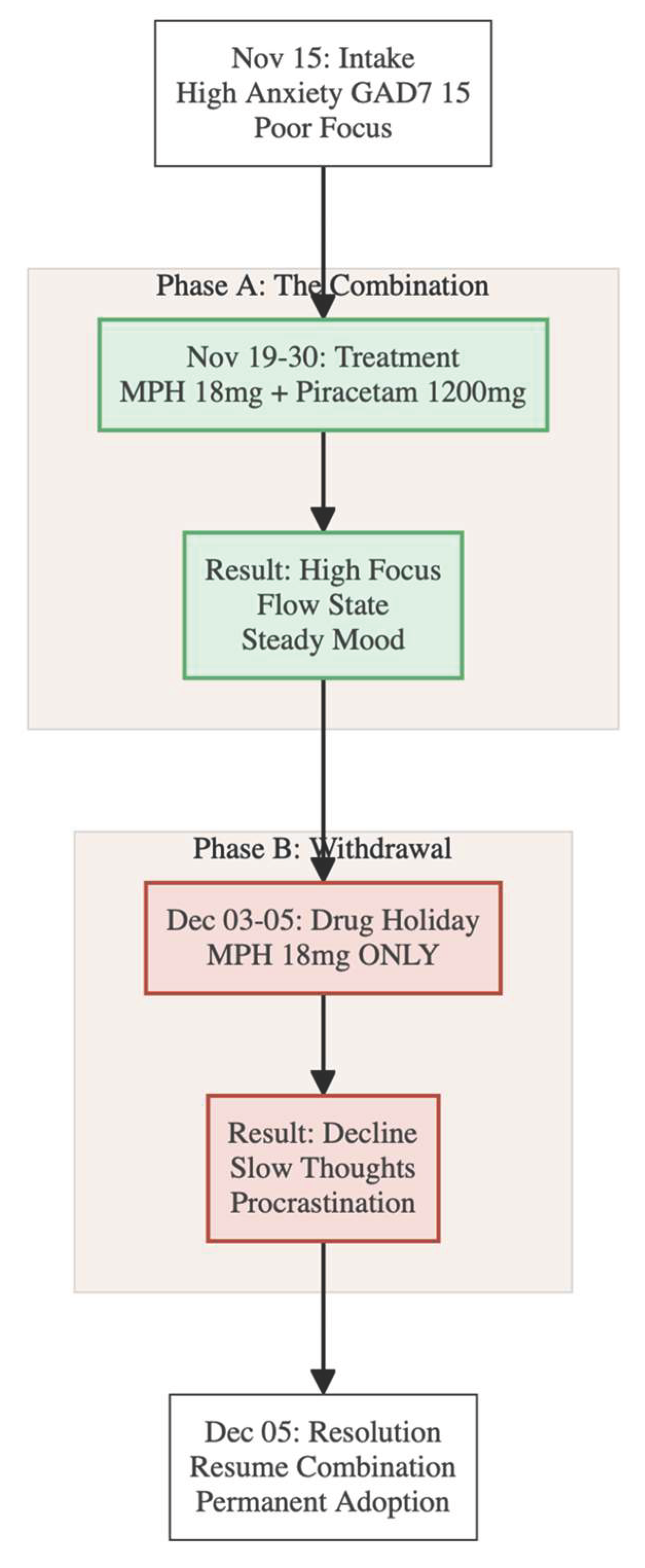
A 28-year-old woman first attended our clinic on 15 November 2025 seeking help for lifelong concentration problems that had recently begun to jeopardise her performance in a demanding postgraduate position (
Figure 1). She carried a previous diagnosis of attention-deficit/hyperactivity disorder, predominantly inattentive presentation, and described associated generalised anxiety of mild-to-moderate severity intertwined with perfectionistic and obsessive-compulsive personality traits. During the preceding four months she had taken atomoxetine, gradually titrated to 60 mg daily, but reported that the initial benefit had faded despite good adherence.
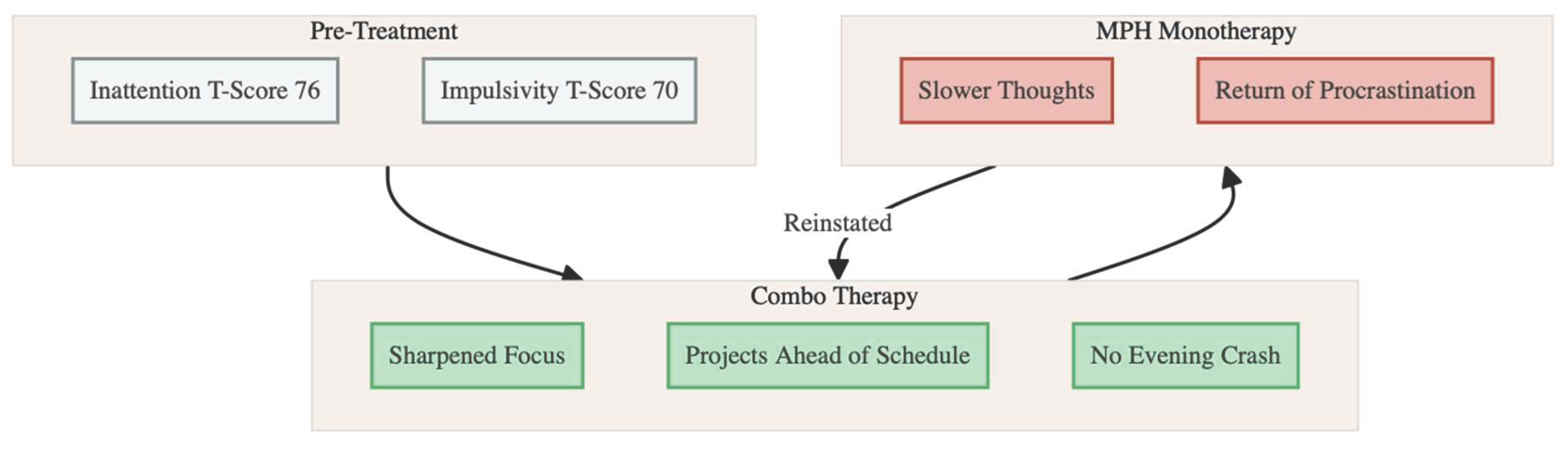
At the intake visit she appeared calm, made good eye contact and spoke fluently, though she frequently lost her train of thought when recounting recent events. Standard rating scales confirmed clinically significant symptoms (PHQ-9 = 11; GAD-7 = 15). A computerised continuous-performance test demonstrated markedly elevated omission and commission errors (T scores 76 and 70, respectively) with an otherwise average reaction time, supporting the impression of prominent inattention combined with impulsivity.
Because she wished to try a stimulant, we provided prescriptions for extended-release methylphenidate 18 mg once each morning and, as an alternative should methylphenidate prove ineffective, lisdexamfetamine 20 mg. She was also given piracetam in case she wanted to stay with atomoxetine, as an AMPA-positive allosteric modulator adjunct.
Four days after the consultation she left Hong Kong for professional travel. During that period she stopped atomoxetine and elected not to start lisdexamfetamine (
Figure 2). Instead, from 19 to 30 November she took methylphenidate 18 mg together with piracetam 1200 mg each morning and kept a detailed diary. Within the first hours of co-administration she noted a striking sharpening of focus, faster information processing and an ability to organise complex tasks that she had never experienced on atomoxetine. The heightened clarity and drive lasted most of the working day—approximately eight to ten hours—without the evening “crash” she had feared. She completed projects ahead of schedule, felt more creative and described her mood as “steady and resilient.” Adverse effects were mild: a subtle sense of muscular tension on some afternoons, brief awareness of an accelerated pulse and occasional difficulty falling asleep if dosing was delayed.
At the start of December she scheduled a two-day drug holiday. On 3, 4 and 5 December she resumed methylphenidate alone while withholding piracetam. Almost immediately she perceived a decline: thoughts felt slower, she reverted to procrastinating on routine e-mails and the effortless flow of ideas disappeared. On the evening of 5 December she contacted us through an encrypted messaging platform to report the contrast and asked whether she might reinstate the combination permanently.
Given the clear temporal association and absence of significant side-effects, we advised continuing piracetam at a dose between 600 and 1200 mg daily, alongside methylphenidate six days per week with a weekly pause to minimise tolerance. We also reiterated the importance of regular cardiovascular monitoring and sleep hygiene.
This case illustrates how low-dose methylphenidate, a standard first-line therapy for adult ADHD, can be augmented by piracetam, a putative positive allosteric modulator at AMPA receptors. In this patient the combination produced rapid, reproducible improvements in executive functioning, sustained motivation and emotional regulation that were not achieved with either atomoxetine monotherapy or methylphenidate alone.
3. Discussion
The present report describes a 28-year-old woman with predominantly inattentive ADHD who experienced an unexpectedly large improvement in focus, processing speed, creativity, motivation, and affect regulation after adding piracetam 1 200 mg to a low dose of extended-release methylphenidate (18 mg). Her own day-by-day notes created a quasi-experimental contrast: twelve consecutive days on the combination were followed by three days on methylphenidate alone, during which most of the gains promptly faded. Although n-of-1 in design, the temporal relationship is difficult to ignore and points to a genuine piracetam-dependent augmentation.
3.1. Rationale for the Observed Synergy
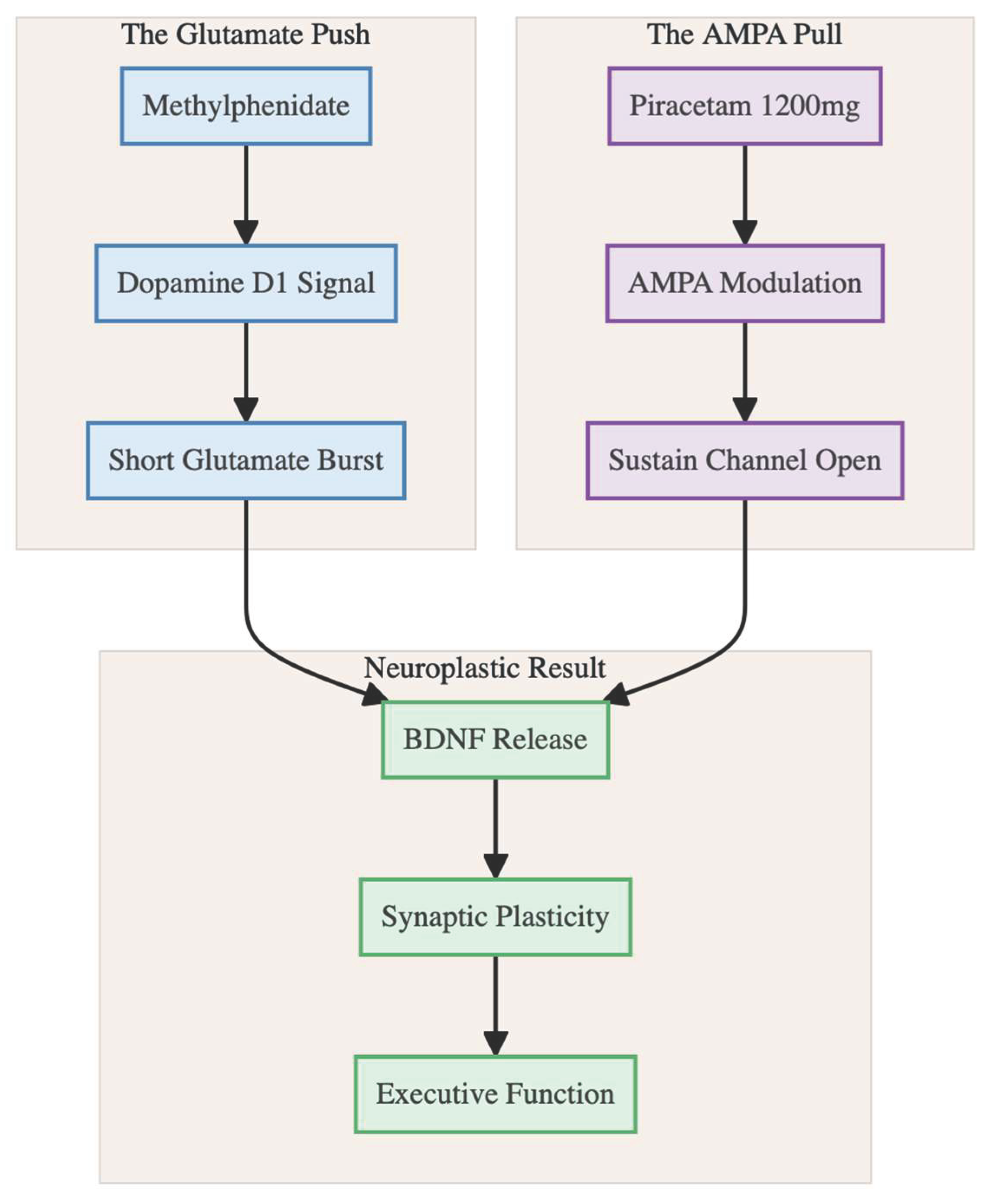
At therapeutic levels methylphenidate raises cortical dopamine (and, secondarily, noradrenaline) by blocking the respective transporters [
1]. Dopaminergic D1-receptor signalling disinhibits pyramidal cells, producing a short-lived glutamate rise of roughly 20–40% that is largely restricted to prefrontal cortex [
6]. Within minutes the same D1/ERK pathway shuttles GluA1-containing AMPA receptors to the synaptic membrane [
6]. Piracetam, an established positive allosteric modulator at AMPA receptors, slows desensitisation and boosts current flow through those channels [
7,
8]. The pairing therefore supplies both a modest glutamate “push” (methylphenidate) and an AMPA “pull” (piracetam), a constellation strikingly similar to the sequence believed to underlie ketamine’s rapid neuroplastic effects [
2,
4]. A comparable logic animates the four-drug Cheung Glutamatergic Regimen, where NMDA antagonism with dextromethorphan produces the glutamate burst that piracetam then amplifies [
11,
13]. In the present case methylphenidate simply substitutes for the NMDA-block step, yet the downstream AMPA-dominant state—and the sharp clinical benefit—appears the same (
Figure 3).
3.2. Safety Profile and Excitotoxicity Risk
Glutamate-driven toxicity requires a sustained calcium load, almost always via NMDA rather than GluA2-containing AMPA receptors [
14]. Methylphenidate’s glutamate rise is brief and self-limited, falling back to baseline within a few hours [
6,
15]. Higher doses of the drug may even dampen excitatory transmission [
6]. Piracetam, used in gram quantities for decades, has never been linked to excitotoxic injury or clinically significant neuro-adverse events [
9]. Taken together, the pharmacology offers a wide safety margin.
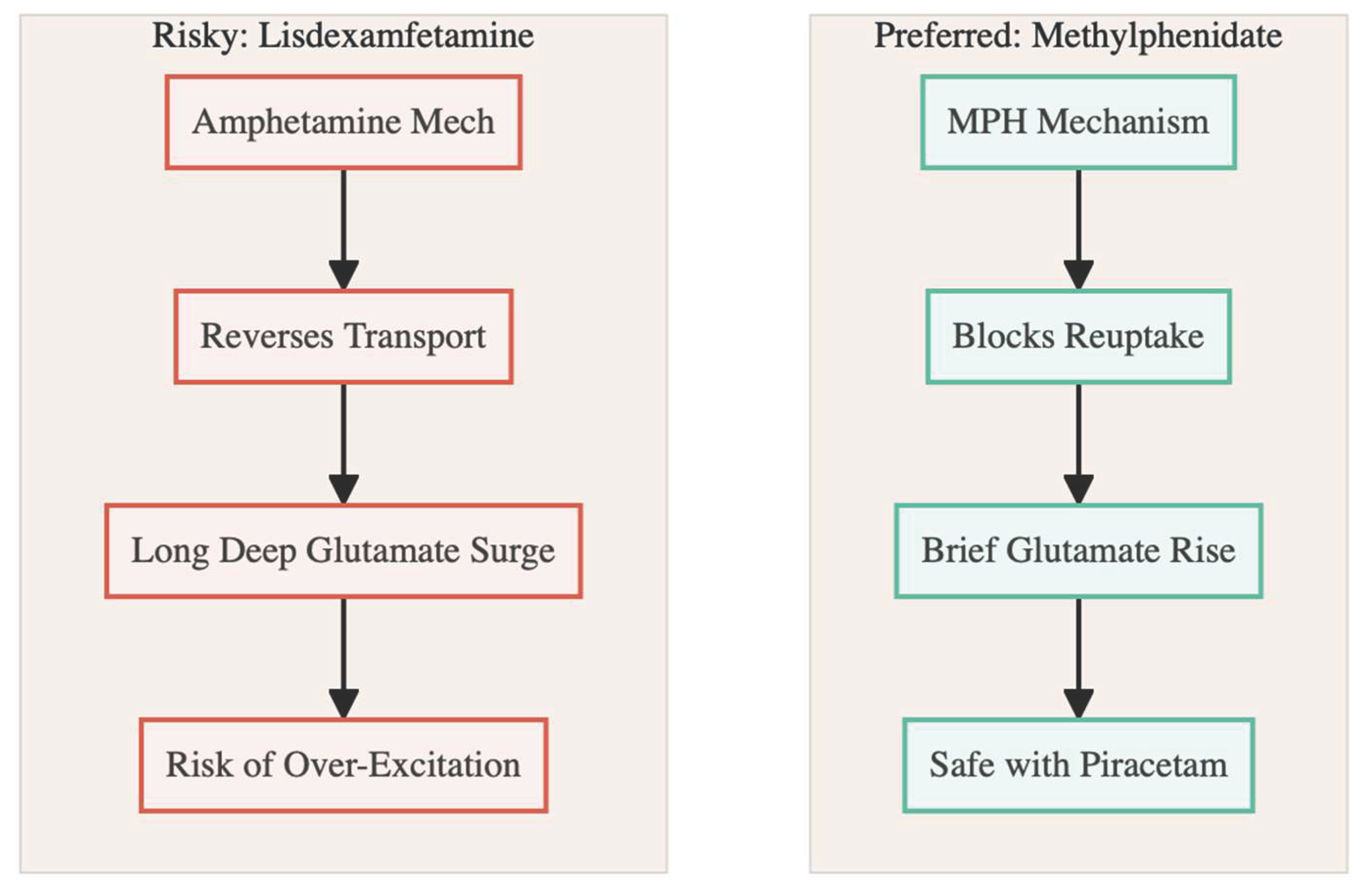
Amphetamine-class stimulants tell a different story: lisdexamfetamine and its congeners double extracellular glutamate for six to twelve hours, particularly in striatum, partly by reversing vesicular transport and astrocytic EAAT2 [
16,
17]. That longer, deeper surge erodes the safety cushion, especially at recreational doses [
18,
19]. Informal user reports echo these mechanistic warnings—piracetam plus amphetamine is often described as “jittery” or “overcaffeinated,” whereas the methylphenidate pairing feels “clean” and “smooth” [
10]. The contrast reinforces the biological case that methylphenidate is the safer glutamate partner for an AMPA modulator (
Figure 4).
3.3. Possible Place in the Cheung Glutamatergic Framework
The magnitude of benefit witnessed here rivals that claimed for the full four-drug Cheung stack in refractory mood and anxiety conditions [
11,
20,
21]. Because methylphenidate already carries regulatory approval for ADHD, a two-component variant—methylphenidate plus piracetam, optionally with oral glutamine—might offer a simpler, more acceptable route to the same neuroplastic target, especially in patients who need both stimulant therapy and help with executive dysfunction, anhedonia, or cognitive “fog.” For individuals with comorbid treatment-resistant depression or OCD, clinicians could still consider transitioning to, or layering in, the full Cheung protocol, as has been piloted elsewhere [
22].
3.4. Can Lisdexamfetamine Replace Methylphenidate in This Model?
Probably not without extra safeguards. The larger, longer glutamate wave induced by lisdexamfetamine, if combined with piracetam’s AMPA potentiation, risks tipping excitatory balance toward agitation, dysphoria, or theoretical excitotoxicity [
18]. Case reports suggest that adding an NMDA antagonist (memantine or low-dose dextromethorphan) can tame the amphetamine surge before AMPA amplification is attempted [
23,
24]. Thus, while an amphetamine-based variant is conceivable, methylphenidate appears the more forgiving partner in day-to-day practice.
In sum, this single case indicates that low-dose methylphenidate augmented with piracetam can harness the same AMPA-dominant plasticity cascade credited for ketamine-class antidepressants—yet with a safety profile familiar to everyday outpatient care. Controlled trials are now needed to evaluate durability, optimal dosing, and generalisability.
Conflict of Interest and Source of Funding Statement
None declared.
Funding Declaration
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics Declaration
Not applicable.
References
- Faraone SV. The pharmacology of amphetamine and methylphenidate: Relevance to the neurobiology of attention-deficit/hyperactivity disorder and other psychiatric comorbidities. Neuroscience & Biobehavioral Reviews. 2018;87:255–270.
- Li N, Lee B, Liu RJ, et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010;329(5994):959–964.
- Duman RS, Aghajanian GK. Synaptic dysfunction in depression: Potential therapeutic targets. Science. 2016;338(6103):68–72.
- Maeng S, Zarate CA Jr, Du J, et al. Cellular mechanisms underlying the antidepressant effects of ketamine: Role of AMPA receptors. Biological Psychiatry. 2008;63(4):349–352.
- Koike H, Iijima M, Chaki S. Involvement of AMPA receptor in both the rapid and sustained antidepressant-like effects of ketamine in animal models. Behavioural Brain Research. 2011;224(1):107–111.
- Cheng J, Xiong Z, Duffney LJ, et al. Methylphenidate exerts dose-dependent effects on glutamate receptors and behaviors. Biological Psychiatry. 2014;76(12):953–962.
- Ahmed AH, Oswald RE. Piracetam defines a new binding site for allosteric modulators of alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) receptors. Journal of Medicinal Chemistry. 2010;53(5):2197–2203.
- Gualtieri F, Manetti D, Romanelli MN, et al. Design and study of piracetam-like nootropics, controversial members of the problematic class of cognition-enhancing drugs. Current Pharmaceutical Design. 2002;8(2):125–138.
- Winblad B. Piracetam: A review of pharmacological properties and clinical uses. CNS Drug Reviews. 2005;11(2):169–182.
- Alavi K, Shirazi E, Akbari M, et al. Effects of piracetam as an adjuvant therapy on Attention-Deficit/Hyperactivity Disorder: A randomized, double-blind, placebo-controlled trial. Iranian Journal of Psychiatry and Behavioral Sciences. 2021;15(2).
- Cheung N. DXM, CYP2D6-inhibiting antidepressants, piracetam, and glutamine: Proposing a ketamine-class antidepressant regimen with existing drugs. Preprints. 2025. https://doi.org/10.20944/preprints202511.1815.v1.
- Cheung N. Clinical experience and optimisation of the Cheung glutamatergic regimen for refractory psychiatric diseases. Preprints. 2025. https://doi.org/10.20944/preprints202511.2246.v1.
- Cheung N. Clearing the fog with the Cheung’s regimen: A case of OTC glutamatergic augmentation in a student with depression, somatic symptoms and cognitive dysfunction. Preprints. 2025. https://doi.org/10.20944/preprints202512.0122.v1.
- Guo C, Ma YY. Calcium-permeable AMPA receptors and excitotoxicity in neurological disorders. Frontiers in Neural Circuits. 2021;15:711564.
- Koda K, Ago Y, Cong Y, et al. Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of monoamines in the mouse prefrontal cortex and striatum. Journal of Neurochemistry. 2010;114(1):259–270.
- Rowley HL, Kulkarni RS, Gosden J, et al. Differences in the neurochemical and behavioural profiles of lisdexamfetamine, methylphenidate and modafinil in freely moving rats. Journal of Psychopharmacology. 2014;28(3):254–269.
- Underhill SM, Colt MS, Amara SG. Amphetamine stimulates endocytosis of the norepinephrine and neuronal glutamate transporters in cultured locus coeruleus neurons. Neurochemical Research. 2020;45(6):1410–1419.
- Fleckenstein AE, Volz TJ, Riddle EL, et al. New insights into the mechanism of action of amphetamines. Annual Review of Pharmacology and Toxicology. 2007;47:681–698.
- Yamamoto BK, Moszczynska A, Gudelsky GA. Amphetamine toxicities: Classical and emerging mechanisms. Annals of the New York Academy of Sciences. 2010;1187:101–121.
- Cheung N. Case series: Marked improvement in treatment-resistant obsessive-compulsive symptoms with over-the-counter glutamatergic augmentation in routine clinical practice. Preprints. 2025. https://doi.org/10.20944/preprints202511.2223.v1.
- Cheung N. Rapid remission of adolescent binge-eating disorder after over-the-counter glutamatergic augmentations to bupropion. Preprints. 2025. https://doi.org/10.20944/preprints202511.2115.v1.
- Cheung N. Cheung’s regimen series: Successful conversion from one dose of esketamine to a low-cost oral ketamine-class glutamatergic regimen in treatment-resistant depression and OCD. Preprints. 2025. https://doi.org/10.20944/preprints202512.0185.v1.
- Biederman J, Fried R, Tarko L, et al. Memantine in the Treatment of Executive Function Deficits in Adults With ADHD. Journal of attention disorders. 2017;21(4):343–352.
- Cheung N. Cheung’s regimen series: Ultra-low-dose paroxetine as CYP2D6 inhibitor enables safe OTC glutamatergic augmentation in a depressive patient on lisdexamfetamine. Preprints. 2025. https://doi.org/10.20944/preprints202512.0057.v1.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).