Submitted:
01 December 2025
Posted:
02 December 2025
You are already at the latest version
Abstract
Activation of microglia and resulting neuroinflammation are central processes that significantly contribute to neurodegenerative disease progression. Treatments capable of attenuating neuroinflammation are therefore an urgent medical need. Vitis vinifera L., cultivated since ancient times for its fruits, is known for its antioxidant and anti-inflammatory activities. However, polyphenols, the main bioactive molecules in V. vinifera extracts, vary considerably due to numerous hard-to-control factors, making it difficult to obtain standardized extracts with consistent biological activity. To address this issue, plant cell culture biotechnology was used to produce a highly standardized V. vinifera phytocomplex (VP), and its anti-neuroinflammatory profile was investigated in LPS-stimulated microglial cells, an in vitro model of neuroinflammation. VP reduced the LPS-induced pro-inflammatory phenotype, improved cell viability and cell number, attenuated NF-κB activation and ERK1/2 phosphorylation, and increased SIRT1 levels. To overcome VP’s poor water solubility, water-soluble, nanocellulose-based formulations containing cellulose nanocrystal (CNC) were developed and tested. VP-CNC formulations markedly reduced the BV2 pro-inflammatory phenotype and increased cell viability under both basal and LPS-stimulated conditions. The nanoformulations also decreased pERK1/2 levels and increased SIRT1 expression, exhibiting biological activities comparable to VP alone. V. vinifera phytocomplex derived from plant cell cultures represents an innovative and standardized product with promising antineuroinflammatory properties.
Keywords:
1. Introduction
2. Results
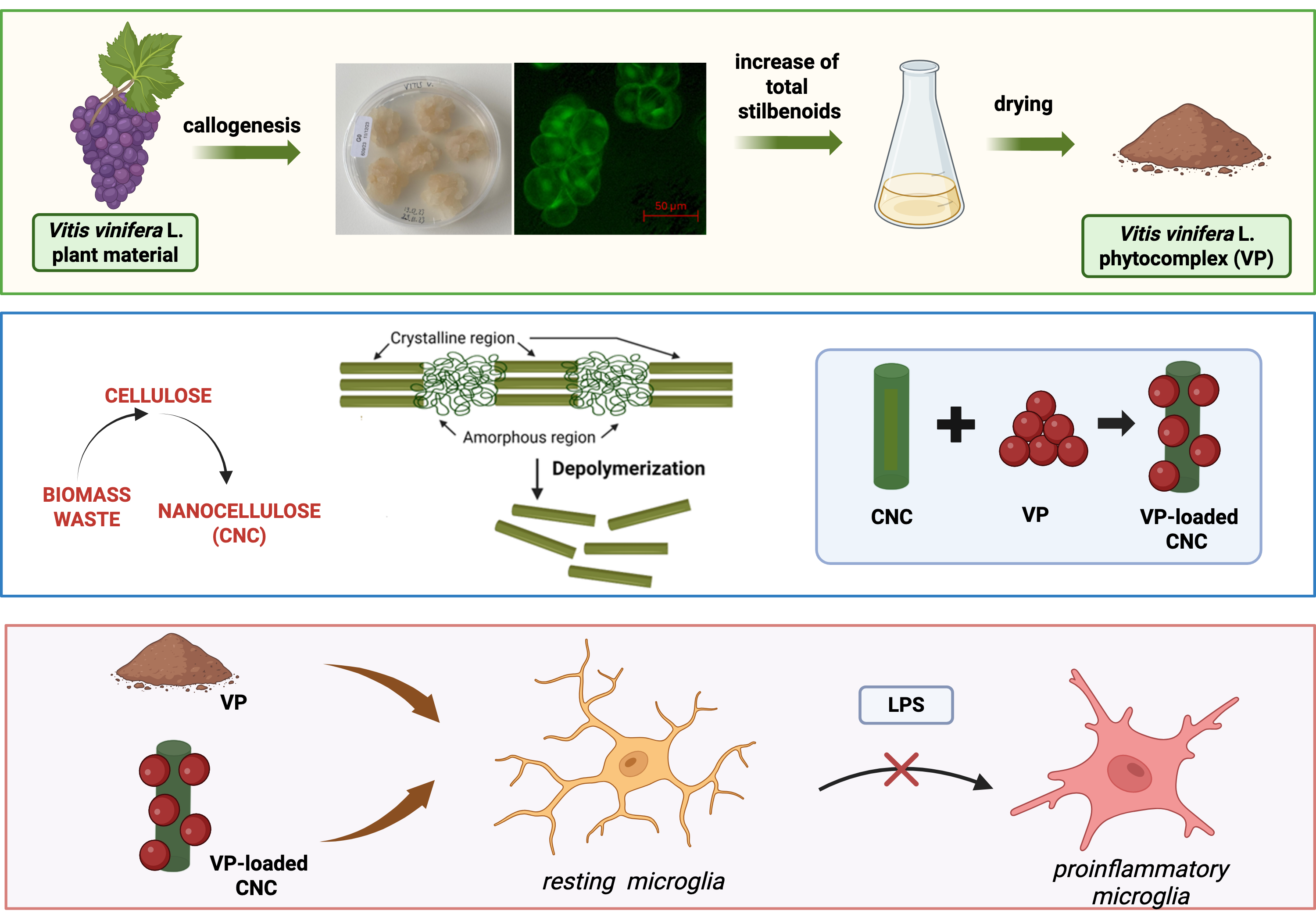
2.1. Characterization of Vitis vinifera L. Standardized Phytocomplex Obtained from Cell Culture Suspensions
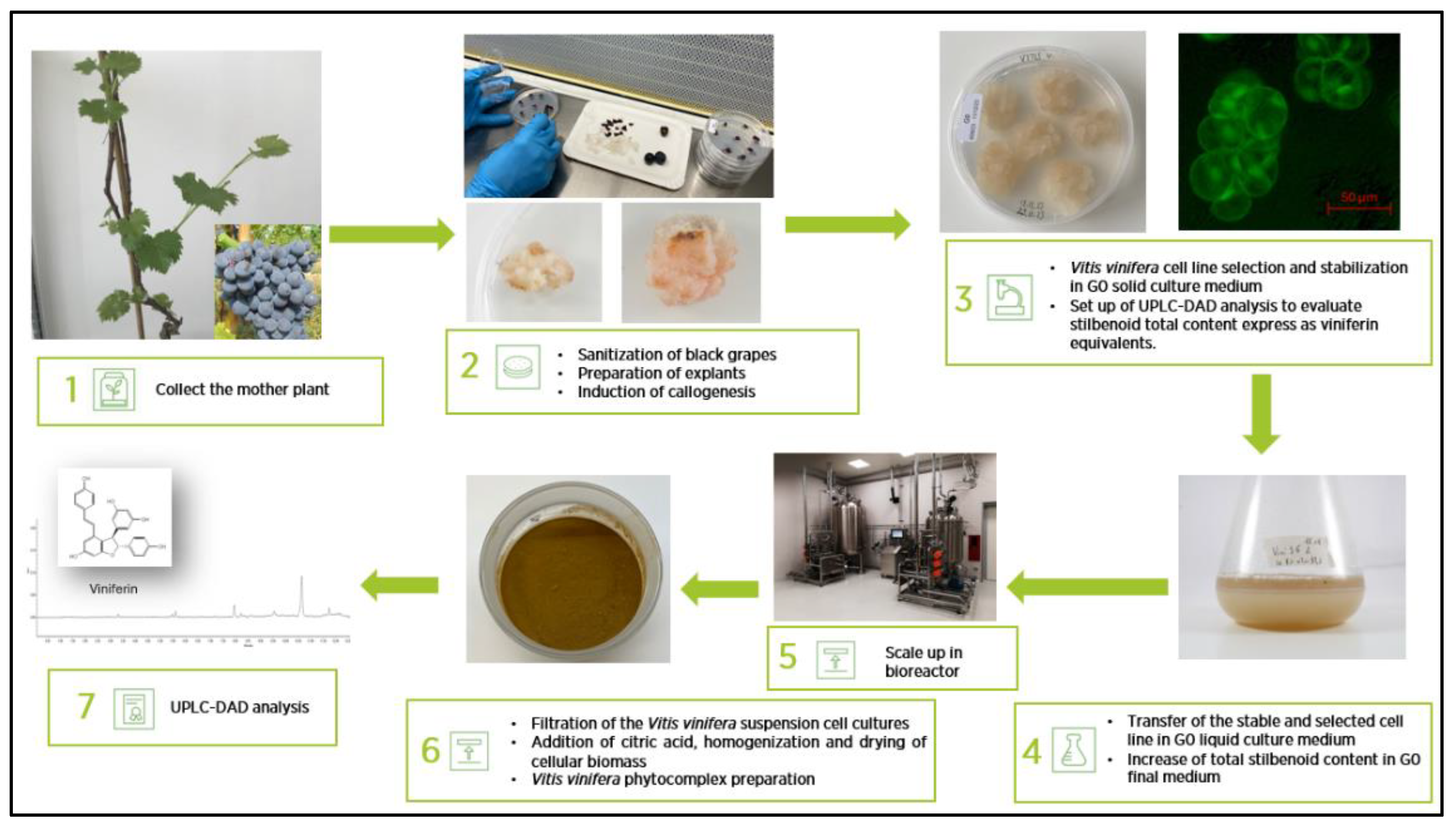
2.1.1. Development and Preparation of V. vinifera L. Standardized Phytocomplex (VP)
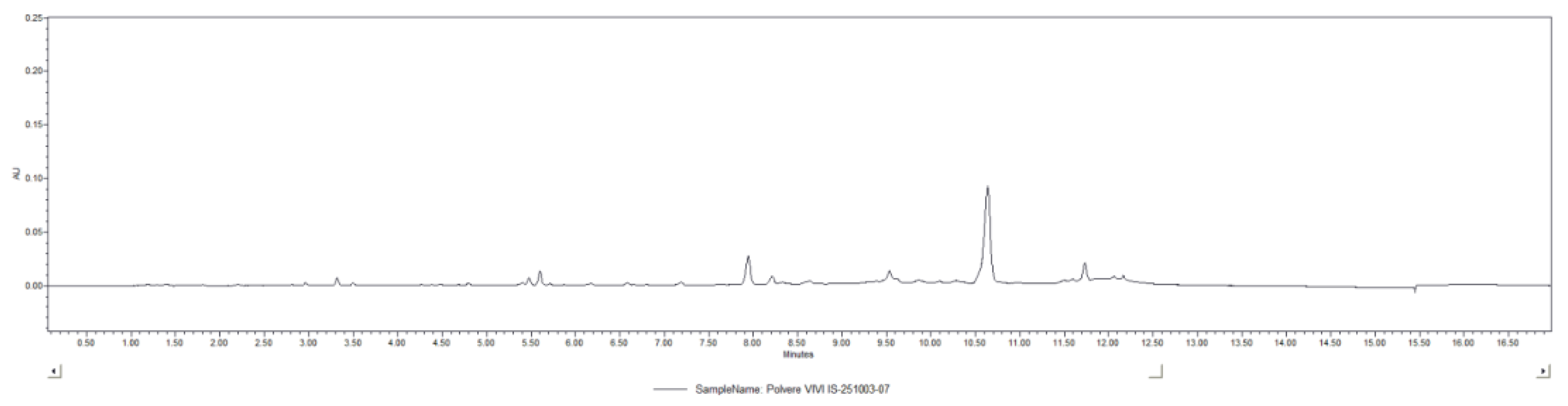
2.1.2. UPLC-DAD Analysis
2.2. Effect of VP on an In Vitro Model of Neuroinflammation
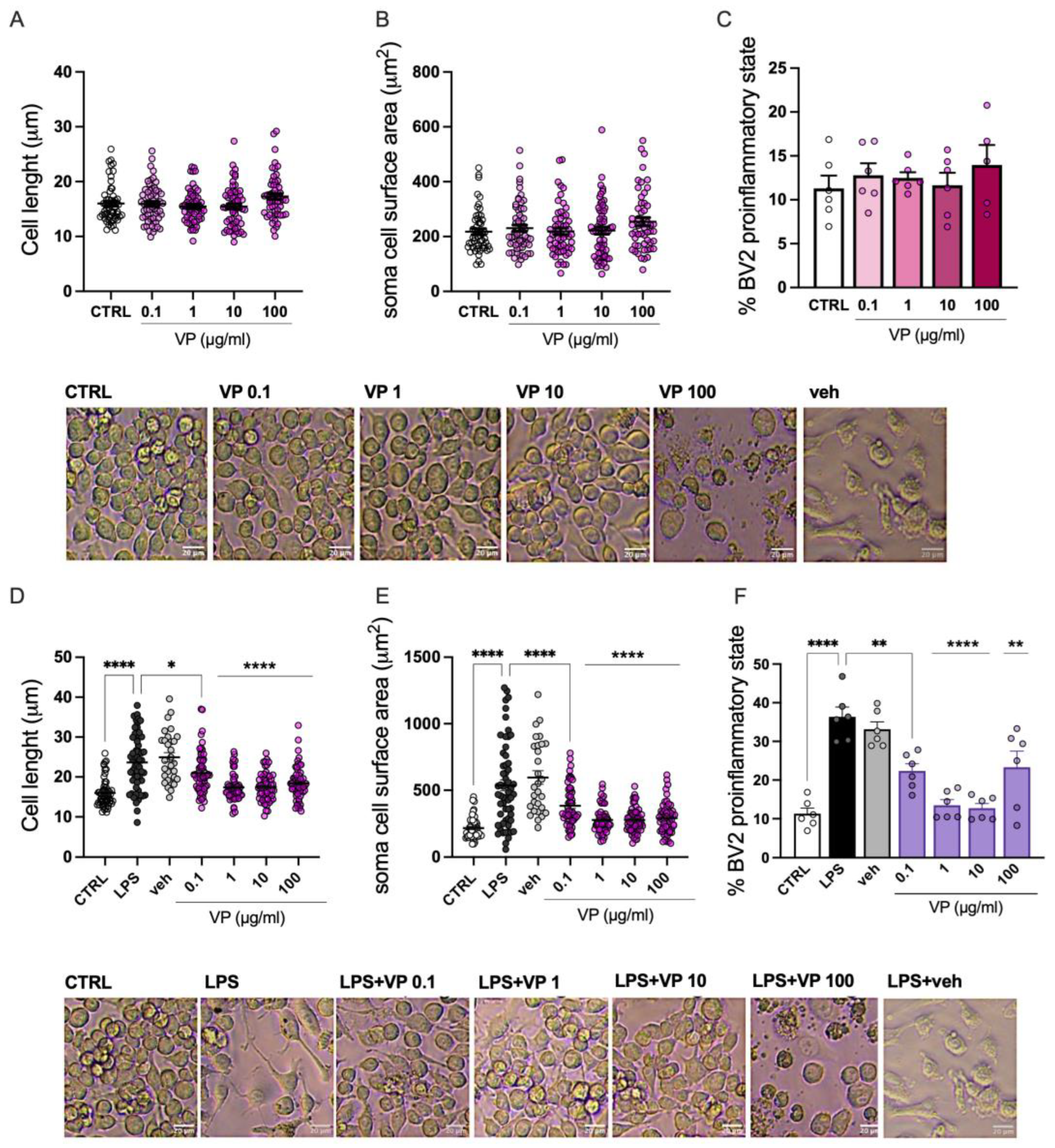
2.2.1. Suppression of Microglia Proinflammatory Phenotype by VP
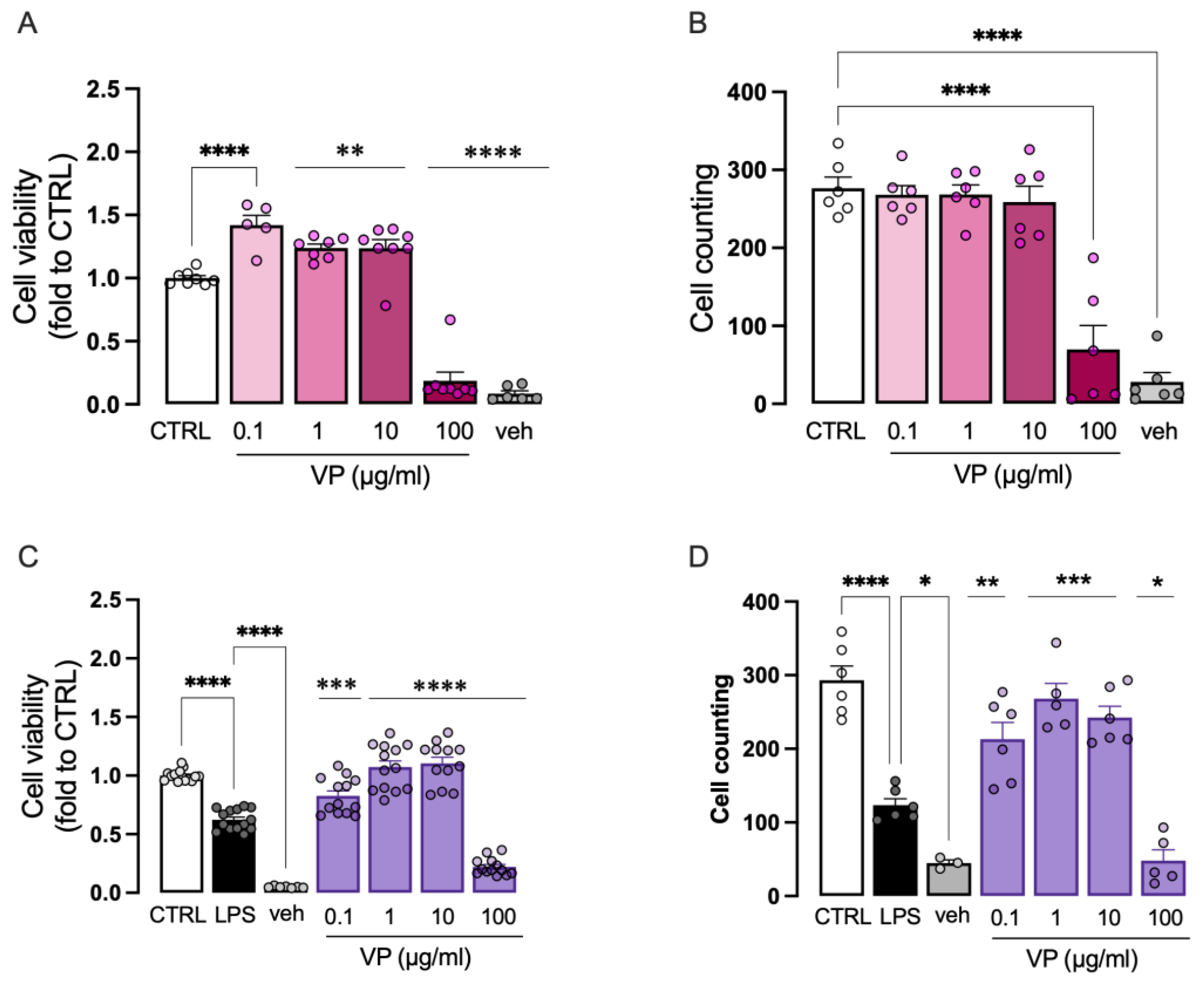
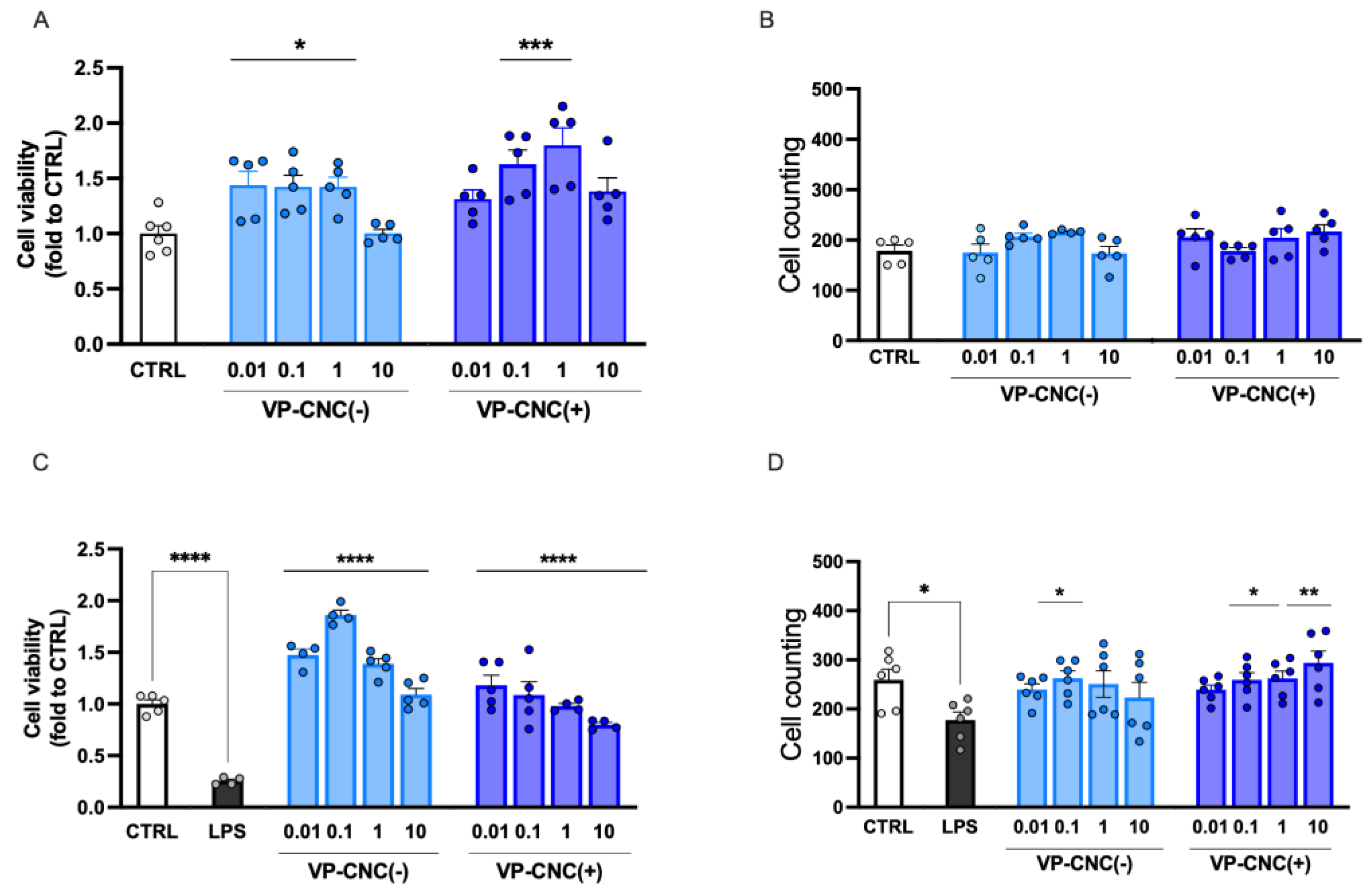
2.2.2. Effect of VP on Microglia Cell Viability
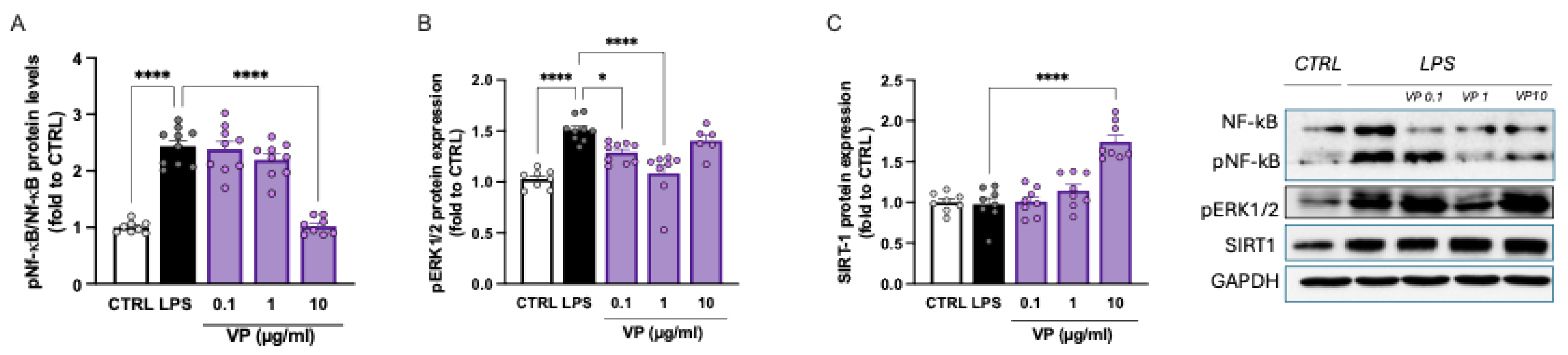
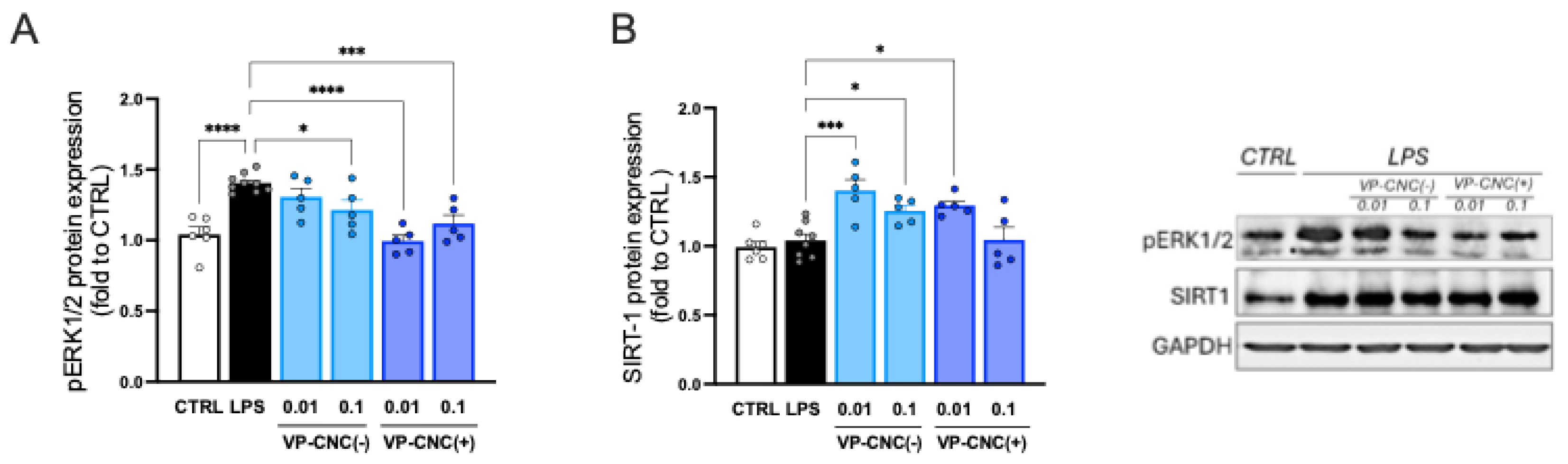
2.2.3. Modulation of Neuroinflammation Markers by VP
2.3. Pharmacological Profile of VP Nanocellulose Formulations
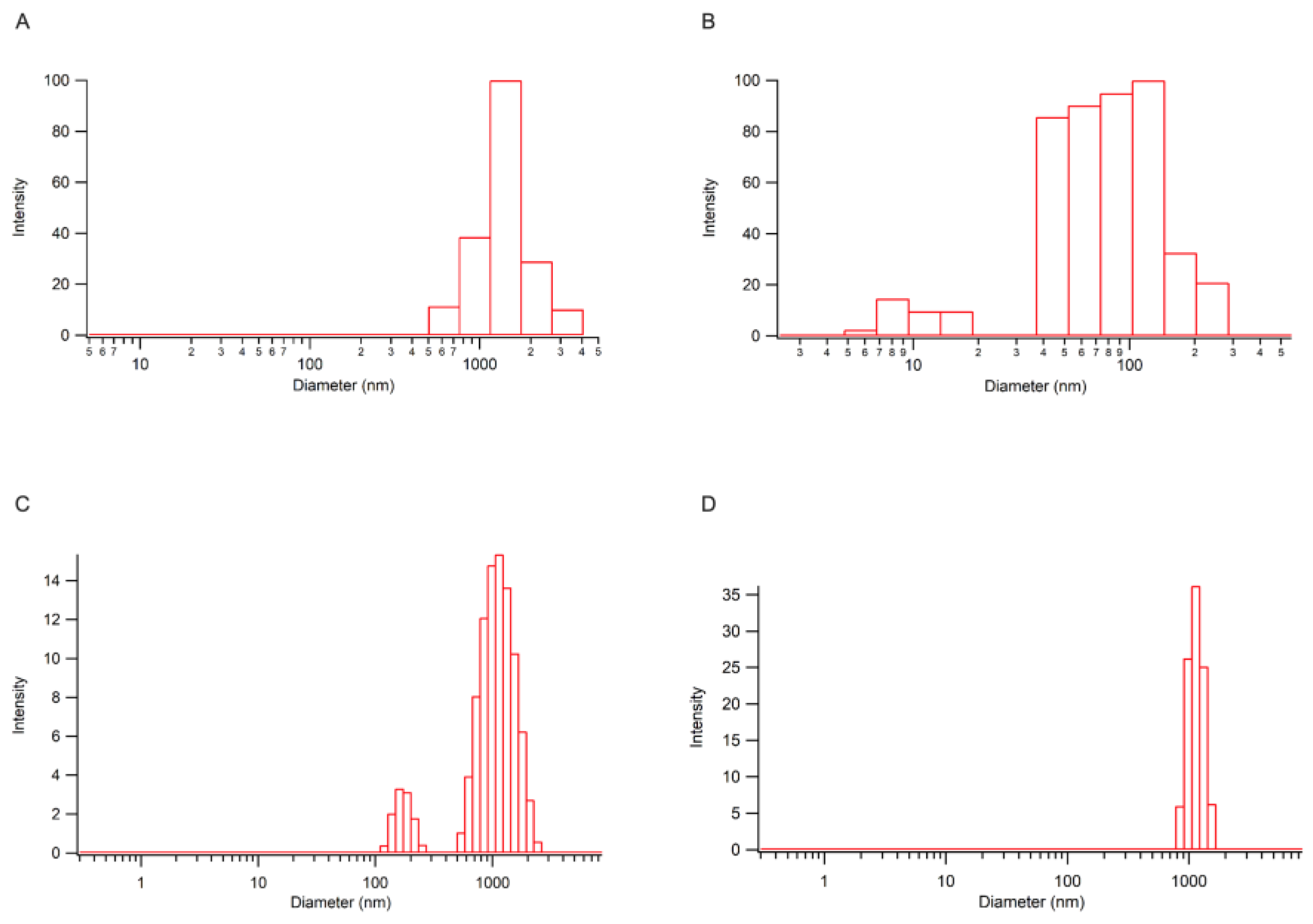
2.3.1. Analysis of VP Nano Cellulose Formulations
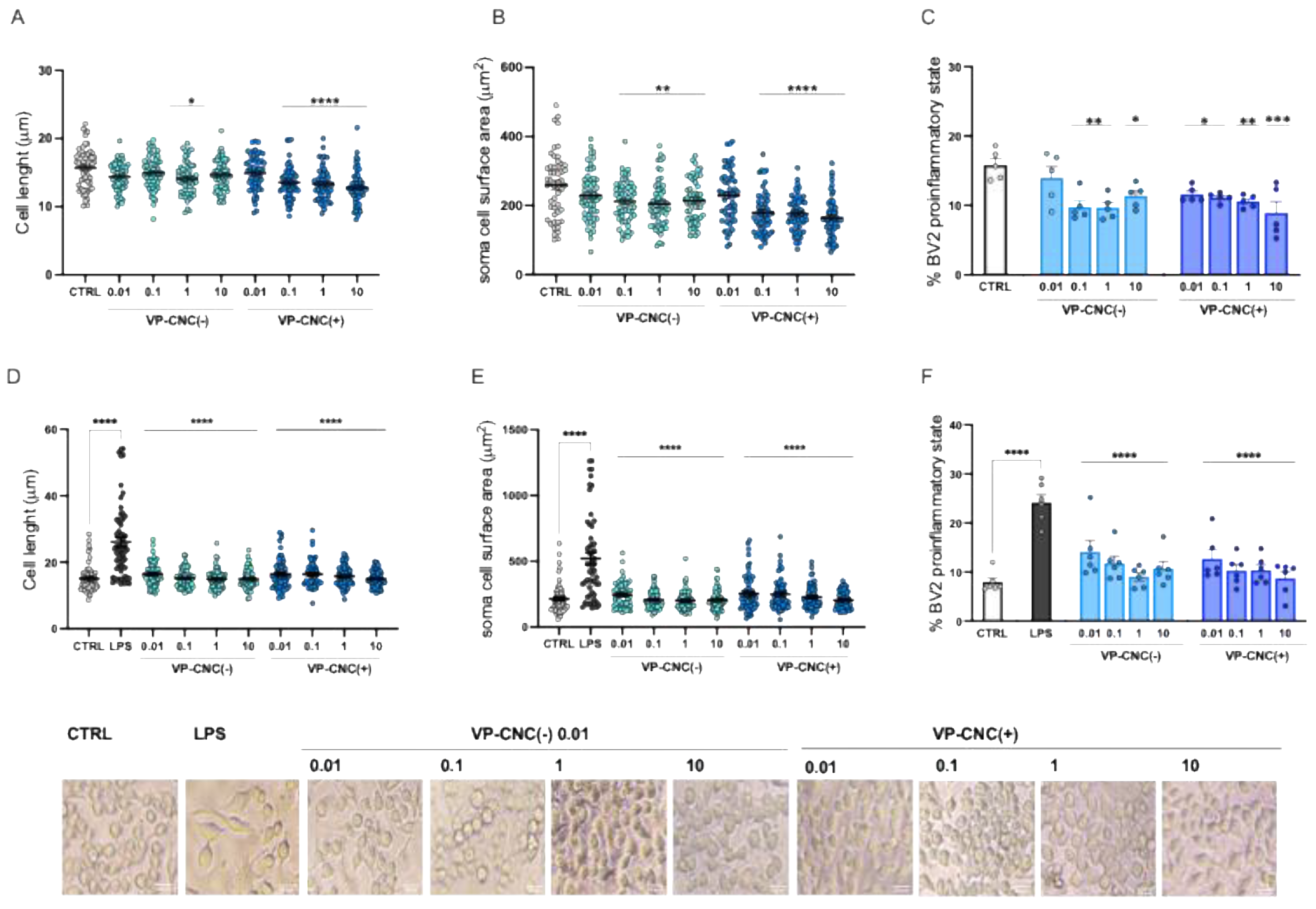
2.3.2. Attenuation of Proinflammatory Morphology by VP-CNC Nanoformulations
2.3.3. Improvement of Microglia Cell Viability by VP Nanoformulations
2.3.4. VP-CNC Nanoformulation Effect on pERK1/2 and SIRT1 Levels
3. Discussion
4. Materials and Methods
4.1. Vitis vinifera L. Phytocomplex (VP) from Cell Culture Suspensions
4.2. UPLC-DAD Analysis of Vitis vinifera L. Standardized Phytocomplex (VP)
4.3. Synthesis CNC (+)
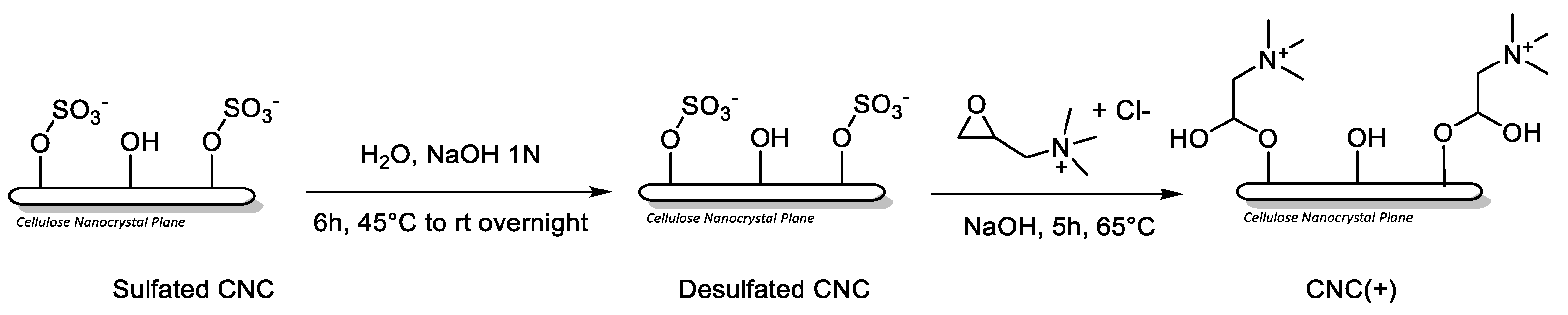
4.4. Preparation of VP-CNC Nanoformulations
4.5. BV2 Cell Culture
4.6. Treatments
4.7. Sulforhodamine B (SRB) Assay
4.8. Cell Counting and Morphology
4.9. Western Blot Analysis
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKT CNC CNS |
activation of protein kinase B cellulose nanocrystal central nervous system |
| CSF1R ERK LPS MAPK NF-κB RNS ROS |
colony-stimulating factor 1 receptor extracellular signal-regulated kinases bacterial lipopolysaccharide from Gram- mitogen activated protein kinase nuclear factor κB reactive nitrogen species reactive oxygen species |
| SIRT1 | Silent information regulator sirtuin 1 |
| TLR4 VP |
toll-like receptor 4 Vitis vinifera L. phytocomplex |
References
- Waisman, A.; Liblau, R.S.; Becher, B. Innate and Adaptive Immune Responses in the CNS. Lancet Neurol 2015, 14, 945–955. [Google Scholar] [CrossRef]
- Adamu, A.; Li, S.; Gao, F.; Xue, G. The Role of Neuroinflammation in Neurodegenerative Diseases: Current Understanding and Future Therapeutic Targets. Front Aging Neurosci 2024, 16, 1347987. [Google Scholar] [CrossRef]
- DiSabato, D.J.; Quan, N.; Godbout, J.P. Neuroinflammation: The Devil Is in the Details. J Neurochem 2016, 139 (Suppl. 2), 136–153. [Google Scholar] [CrossRef]
- Kölliker-Frers, R.; Udovin, L.; Otero-Losada, M.; Kobiec, T.; Herrera, M.I.; Palacios, J.; Razzitte, G.; Capani, F. Neuroinflammation: An Integrating Overview of Reactive-Neuroimmune Cell Interactions in Health and Disease. Mediators Inflamm 2021, 2021. [Google Scholar] [CrossRef] [PubMed]
- Ajoolabady, A.; Kim, B.; Abdulkhaliq, A.A.; Ren, J.; Bahijri, S.; Tuomilehto, J.; Borai, A.; Khan, J.; Pratico, D. Dual Role of Microglia in Neuroinflammation and Neurodegenerative Diseases. Neurobiol Dis 2025, 216, 107133. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Barres, B.A. Microglia and Macrophages in Brain Homeostasis and Disease. Nat Rev Immunol 2018, 18, 225–242. [Google Scholar] [CrossRef]
- Qin, J.; Ma, Z.; Chen, X.; Shu, S. Microglia Activation in Central Nervous System Disorders: A Review of Recent Mechanistic Investigations and Development Efforts. Front Neurol 2023, 14, 1103416. [Google Scholar] [CrossRef]
- Kwon, H.S.; Koh, S.H. Neuroinflammation in Neurodegenerative Disorders: The Roles of Microglia and Astrocytes. Transl Neurodegener 2020, 9. [Google Scholar] [CrossRef]
- Gao, C.; Jiang, J.; Tan, Y.; Chen, S. Microglia in Neurodegenerative Diseases: Mechanism and Potential Therapeutic Targets. Signal Transduct Target Ther 2023, 8. [Google Scholar] [CrossRef]
- Shi, F.D.; Yong, V.W. Neuroinflammation across Neurological Diseases. Science 2025, 388. [Google Scholar] [CrossRef] [PubMed]
- Goufo, P.; Singh, R.K.; Cortez, I. A Reference List of Phenolic Compounds (Including Stilbenes) in Grapevine (Vitis Vinifera L.) Roots, Woods, Canes, Stems, and Leaves. Antioxidants 2020, 9, 398. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Li, J.; Xiong, R.G.; Saimaiti, A.; Huang, S.Y.; Wu, S.X.; Yang, Z.J.; Shang, A.; Zhao, C.N.; Gan, R.Y.; et al. Bioactive Compounds, Health Benefits and Food Applications of Grape. Foods 2022, 11, 2755. [Google Scholar] [CrossRef]
- Chen, H.; Yang, J.; Deng, X.; Lei, Y.; Xie, S.; Guo, S.; Ren, R.; Li, J.; Zhang, Z.; Xu, T. Foliar-Sprayed Manganese Sulfate Improves Flavonoid Content in Grape Berry Skin of Cabernet Sauvignon (Vitis Vinifera L.) Growing on Alkaline Soil and Wine Chromatic Characteristics. Food Chem 2020, 314, 126182. [Google Scholar] [CrossRef]
- Tetik, F.; Civelek, S.; Cakilcioglu, U. Traditional Uses of Some Medicinal Plants in Malatya (Turkey). J Ethnopharmacol 2013, 146, 331–346. [Google Scholar] [CrossRef]
- Ishtiaq, M.; Mahmood, A.; Maqbool, M. Indigenous Knowledge of Medicinal Plants from Sudhanoti District (AJK), Pakistan. J Ethnopharmacol 2015, 168, 201–207. [Google Scholar] [CrossRef]
- Nassiri-Asl, M.; Hosseinzadeh, H. Review of the Pharmacological Effects of Vitis Vinifera (Grape) and Its Bioactive Constituents: An Update. Phytother Res 2016, 30, 1392–1403. [Google Scholar] [CrossRef]
- Lakshmi, B.V.S.; Sudhakar, M.; Anisha, M. Neuroprotective Role of Hydroalcoholic Extract of Vitis Vinifera against Aluminium-Induced Oxidative Stress in Rat Brain. Neurotoxicology 2014, 41, 73–79. [Google Scholar] [CrossRef]
- Pazos-Tomas, C.C.; Cruz-Venegas, A.; Pérez-Santiago, A.D.; Sánchez-Medina, M.A.; Matías-Pérez, D.; García-Montalvo, I.A. Vitis Vinifera: An Alternative for the Prevention of Neurodegenerative Diseases. J Oleo Sci 2020, 69, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Krasteva, G.; Georgiev, V.; Pavlov, A. Recent Applications of Plant Cell Culture Technology in Cosmetics and Foods. Eng Life Sci 2020, 21, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, V.; Slavov, A.; Vasileva, I.; Pavlov, A. Plant Cell Culture as Emerging Technology for Production of Active Cosmetic Ingredients. Eng Life Sci 2018, 18, 779–798. [Google Scholar] [CrossRef]
- Biermann, O.; Hädicke, E.; Koltzenburg, S.; Müller-Plathe, F.; Müller-Plathe, F.; Biermann, D.-C.O.; Hädicke, E.; Koltzenburg, S. Hydrophilicity and Lipophilicity of Cellulose Crystal Surfaces. Angew. Chem. Int. Ed 2001, 40. [Google Scholar] [CrossRef]
- Lindman, B.; Medronho, B.; Alves, L.; Norgren, M.; Nordenskiöld, L. Hydrophobic Interactions Control the Self-Assembly of DNA and Cellulose. Q Rev Biophys 2021, 54. [Google Scholar] [CrossRef]
- Kumar, R.; Chauhan, S. Cellulose Nanocrystals Based Delivery Vehicles for Anticancer Agent Curcumin. Int J Biol Macromol 2022, 221, 842–864. [Google Scholar] [CrossRef]
- Mazeau, K.; Wyszomirski, M. Modelling of Congo Red Adsorption on the Hydrophobic Surface of Cellulose Using Molecular Dynamics. Cellulose 2012 19:5 2012, 19, 1495–1506. [Google Scholar] [CrossRef]
- Piras, C.C.; Fernández-Prieto, S.; De Borggraeve, W.M. Ball Milling: A Green Technology for the Preparation and Functionalisation of Nanocellulose Derivatives. Nanoscale Adv 2019, 1, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Van Hove, H.; De Feo, D.; Greter, M.; Becher, B. Central Nervous System Macrophages in Health and Disease. Annu Rev Immunol 2025, 43, 589–613. [Google Scholar] [CrossRef] [PubMed]
- Colonna, M.; Butovsky, O. Microglia Function in the Central Nervous System During Health and Neurodegeneration. Annu Rev Immunol 2017, 35, 441–468. [Google Scholar] [CrossRef]
- Borgonetti, V.; Anceschi, L.; Brighenti, V.; Corsi, L.; Governa, P.; Manetti, F.; Pellati, F.; Galeotti, N. Cannabidiol-Rich Non-Psychotropic Cannabis Sativa L. Oils Attenuate Peripheral Neuropathy Symptoms by Regulation of CB2-Mediated Microglial Neuroinflammation. Phytother Res 2022. [Google Scholar] [CrossRef]
- Sangiovanni, E.; Di Lorenzo, C.; Piazza, S.; Manzoni, Y.; Brunelli, C.; Fumagalli, M.; Magnavacca, A.; Martinelli, G.; Colombo, F.; Casiraghi, A.; et al. Vitis Vinifera L. Leaf Extract Inhibits In Vitro Mediators of Inflammation and Oxidative Stress Involved in Inflammatory-Based Skin Diseases. Antioxidants 2019, 8, 134. [Google Scholar] [CrossRef]
- Acero, N.; Manrique, J.; Muñoz-Mingarro, D.; Martínez Solís, I.; Bosch, F. Vitis Vinifera L. Leaves as a Source of Phenolic Compounds with Anti-Inflammatory and Antioxidant Potential. Antioxidants 2025, 14, 279. [Google Scholar] [CrossRef]
- Dai, X.M.; Ryan, G.R.; Hapel, A.J.; Dominguez, M.G.; Russell, R.G.; Kapp, S.; Sylvestre, V.; Stanley, E.R. Targeted Disruption of the Mouse Colony-Stimulating Factor 1 Receptor Gene Results in Osteopetrosis, Mononuclear Phagocyte Deficiency, Increased Primitive Progenitor Cell Frequencies, and Reproductive Defects. Blood 2002, 99, 111–120. [Google Scholar] [CrossRef]
- Arthur, J.S.C.; Ley, S.C. Mitogen-Activated Protein Kinases in Innate Immunity. Nat Rev Immunol 2013, 13, 679–692. [Google Scholar] [CrossRef]
- Liu, P.; Li, Y.; Wang, W.; Bai, Y.; Jia, H.; Yuan, Z.; Yang, Z. Role and Mechanisms of the NF-ĸB Signaling Pathway in Various Developmental Processes. Biomedicine & Pharmacotherapy 2022, 153, 113513. [Google Scholar] [CrossRef]
- Gandhi, D.; Bhandari, S.; Maity, S.; Mahapatra, S.K.; Rajasekaran, S. Activation of ERK/NF-KB Pathways Contributes to the Inflammatory Response in Epithelial Cells and Macrophages Following Manganese Exposure. Biol Trace Elem Res 2025, 203, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, Y.; Wang, Y.; Chao, Y.; Zhang, J.; Jia, Y.; Tie, J.; Hu, D. Regulation of SIRT1 and Its Roles in Inflammation. Front Immunol 2022, 13, 831168. [Google Scholar] [CrossRef]
- Yeung, F.; Hoberg, J.E.; Ramsey, C.S.; Keller, M.D.; Jones, D.R.; Frye, R.A.; Mayo, M.W. Modulation of NF-KappaB-Dependent Transcription and Cell Survival by the SIRT1 Deacetylase. EMBO J 2004, 23, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Wang, J.G.; Xiao, D.M.; Fan, M.; Wang, D.P.; Xiong, J.Y.; Chen, Y.; Ding, Y.; Liu, S.L. Resveratrol Inhibits Interleukin 1β-Mediated Inducible Nitric Oxide Synthase Expression in Articular Chondrocytes by Activating SIRT1 and Thereby Suppressing Nuclear Factor-ΚB Activity. Eur J Pharmacol 2012, 674, 73–79. [Google Scholar] [CrossRef]
- Hu, T.; Fan, X.; Ma, L.; Liu, J.; Chang, Y.; Yang, P.; Qiu, S.; Chen, T.; Yang, L.; Liu, Z. TIM4-TIM1 Interaction Modulates Th2 Pattern Inflammation through Enhancing SIRT1 Expression. Int J Mol Med 2017, 40, 1504–1510. [Google Scholar] [CrossRef]
- Li, Y.; Liu, T.; Li, Y.; Han, D.; Hong, J.; Yang, N.; He, J.; Peng, R.; Mi, X.; Kuang, C.; et al. Baicalin Ameliorates Cognitive Impairment and Protects Microglia from LPS-Induced Neuroinflammation via the SIRT1/HMGB1 Pathway. Oxid Med Cell Longev 2020, 2020. [Google Scholar] [CrossRef]
- Sun, H.; Li, D.; Wei, C.; Liu, L.; Xin, Z.; Gao, H.; Gao, R. The Relationship between SIRT1 and Inflammation: A Systematic Review and Meta-Analysis. Front Immunol 2024, 15, 1465849. [Google Scholar] [CrossRef]
- Al-Khayri, J.M.; Mascarenhas, R.; Harish, H.M.; Gowda, Y.; Lakshmaiah, V.V.; Nagella, P.; Al-Mssallem, M.Q.; Alessa, F.M.; Almaghasla, M.I.; Rezk, A.A.S. Stilbenes, a Versatile Class of Natural Metabolites for Inflammation-An Overview. Molecules 2023, 28, 3786. [Google Scholar] [CrossRef] [PubMed]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small Molecule Activators of Sirtuins Extend Saccharomyces Cerevisiae Lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef]
- Rogina, B.; Tissenbaum, H.A. SIRT1, Resveratrol and Aging. Front Genet 2024, 15, 1393181. [Google Scholar] [CrossRef]
- Wu, C.W.; Nakamoto, Y.; Hisatome, T.; Yoshida, S.; Miyazaki, H. Resveratrol and Its Dimers ε-Viniferin and δ-Viniferin in Red Wine Protect Vascular Endothelial Cells by a Similar Mechanism with Different Potency and Efficacy. Kaohsiung J Med Sci 2020, 36, 535–542. [Google Scholar] [CrossRef]
- Hung, M.W.; Wu, C.W.; Kokubu, D.; Yoshida, S.; Miyazaki, H. ε-Viniferin Is More Effective than Resveratrol in Promoting Favorable Adipocyte Differentiation with Enhanced Adiponectin Expression and Decreased Lipid Accumulation. Food Sci Technol Res 2019, 25, 817–826. [Google Scholar] [CrossRef]
- Hasan, N.; Rahman, L.; Kim, S.H.; Cao, J.; Arjuna, A.; Lallo, S.; Jhun, B.H.; Yoo, J.W. Recent Advances of Nanocellulose in Drug Delivery Systems. Journal of Pharmaceutical Investigation 2020, 50, 553–572. [Google Scholar] [CrossRef]
- Karimian, A.; Parsian, H.; Majidinia, M.; Rahimi, M.; Mir, S.M.; Samadi Kafil, H.; Shafiei-Irannejad, V.; Kheyrollah, M.; Ostadi, H.; Yousefi, B. Nanocrystalline Cellulose: Preparation, Physicochemical Properties, and Applications in Drug Delivery Systems. Int J Biol Macromol 2019, 133, 850–859. [Google Scholar] [CrossRef]
- Huo, Y.; Liu, Y.; Xia, M.; Du, H.; Lin, Z.; Li, B.; Liu, H. Nanocellulose-Based Composite Materials Used in Drug Delivery Systems. Polymers 2022, Vol. 14, Page 2648 2022, 14, 2648. [Google Scholar] [CrossRef] [PubMed]
- Gamborg, O.L.; Miller, R.A.; Ojima, K. Nutrient Requirements of Suspension Cultures of Soybean Root Cells. Exp Cell Res 1968, 50, 151–158. [Google Scholar] [CrossRef]
- Liu, Y.; Li, M.; Qiao, M.; Ren, X.; Huang, T.S.; Buschle-Diller, G. Antibacterial Membranes Based on Chitosan and Quaternary Ammonium Salts Modified Nanocrystalline Cellulose. Polym Adv Technol 2017, 28, 1629–1635. [Google Scholar] [CrossRef]
- Borgonetti, V.; Pressi, G.; Bertaiola, O.; Guarnerio, C.; Mandrone, M.; Chiocchio, I.; Galeotti, N. Attenuation of Neuroinflammation in Microglia Cells by Extracts with High Content of Rosmarinic Acid from in Vitro Cultured Melissa Officinalis L. Cells. J Pharm Biomed Anal 2022, 220. [Google Scholar] [CrossRef] [PubMed]
- Videtta, G.; Sasia, C.; Galeotti, N. High Rosmarinic Acid Content Melissa Officinalis L. Phytocomplex Modulates Microglia Neuroinflammation Induced by High Glucose. Antioxidants 2025, 14, 161. [Google Scholar] [CrossRef] [PubMed]
- Motulsky, H.J.; Brown, R.E. Detecting Outliers When Fitting Data with Nonlinear Regression—a New Method Based on Robust Nonlinear Regression and the False Discovery Rate. BMC Bioinformatics 2006, 7. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



