Submitted:
27 November 2025
Posted:
28 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Generation and Validation of hiPSCs Carrying Homozygous Nonsense Mutations in USH2A Exon-13 by CRISPR/Cas9
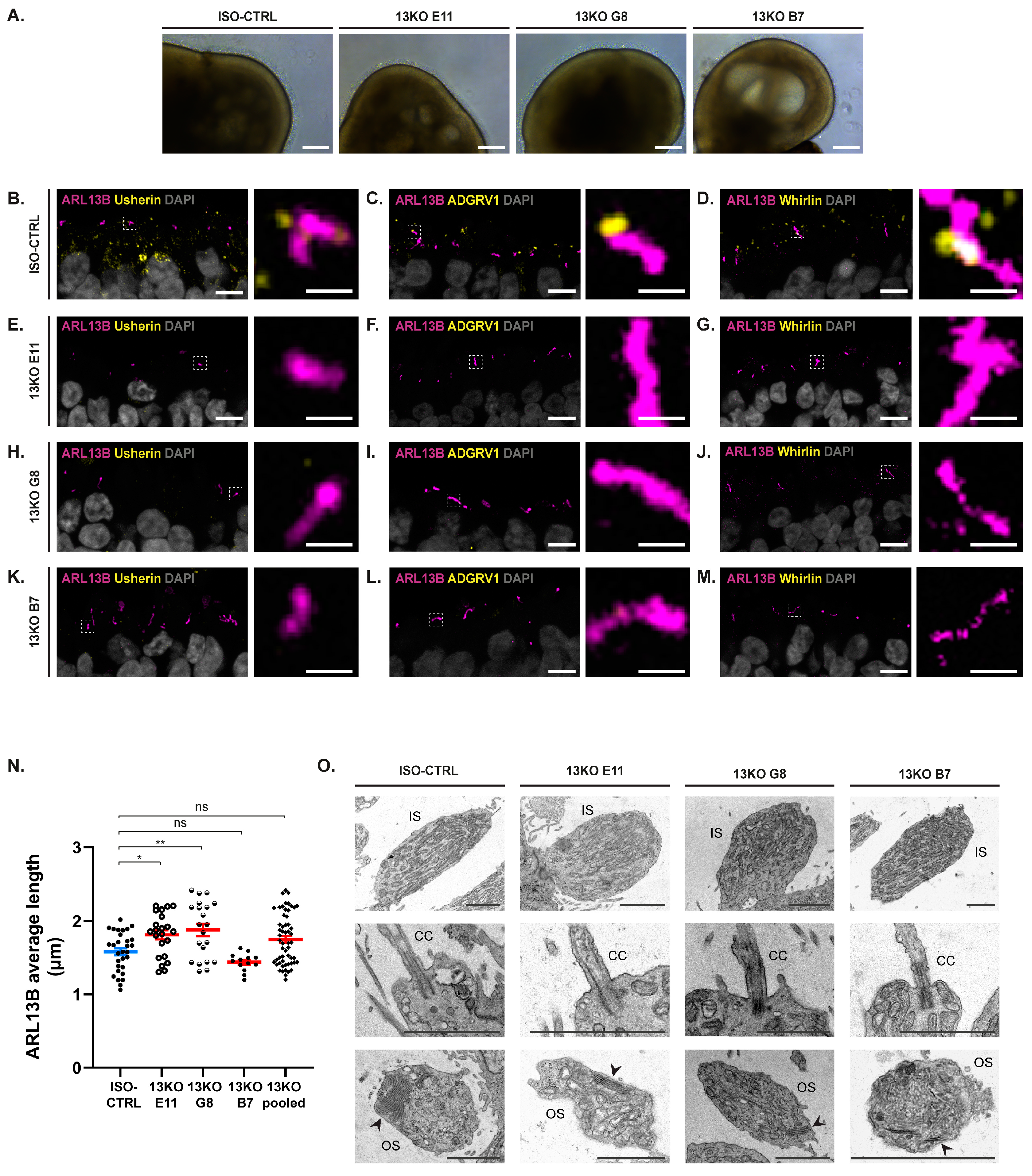
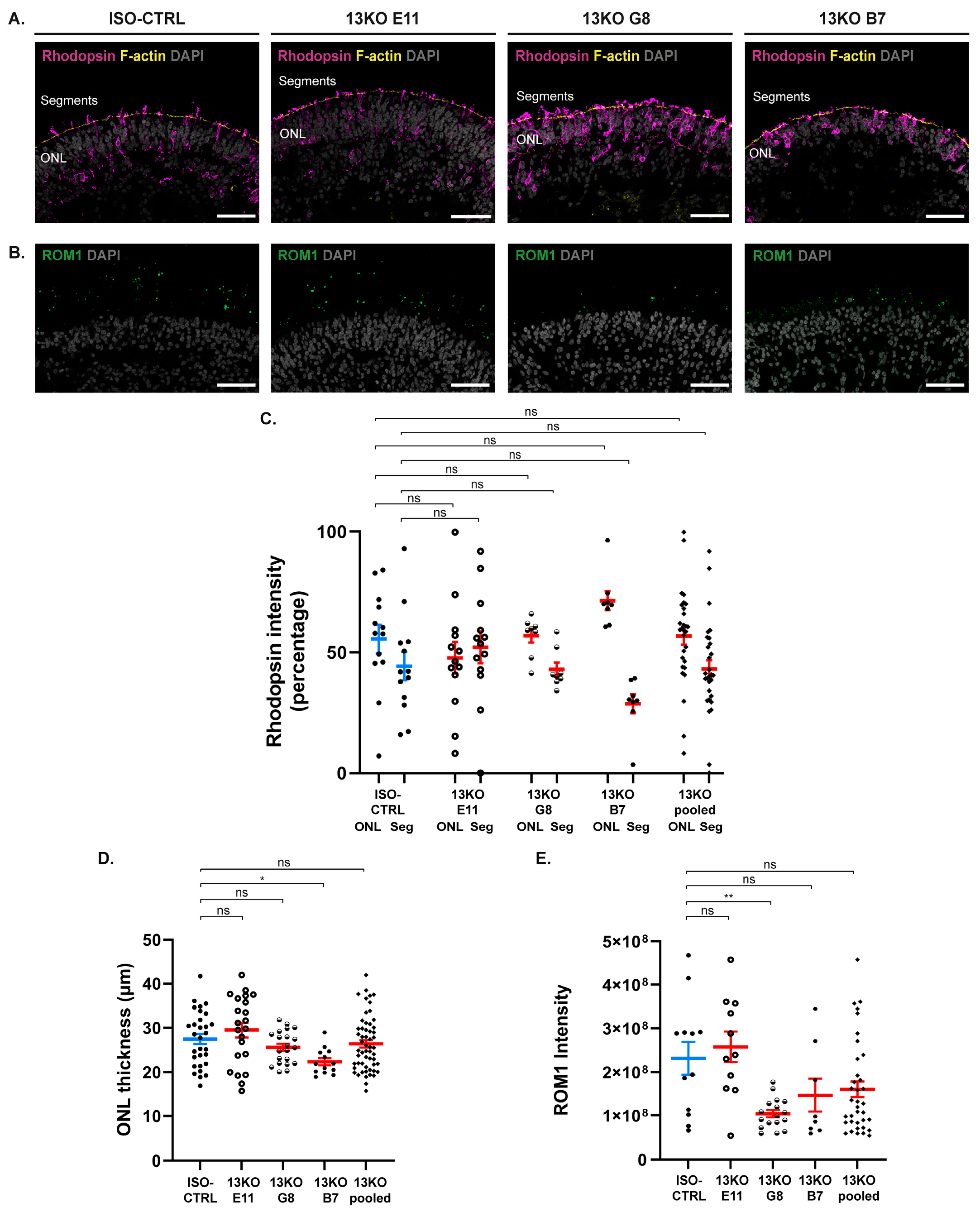
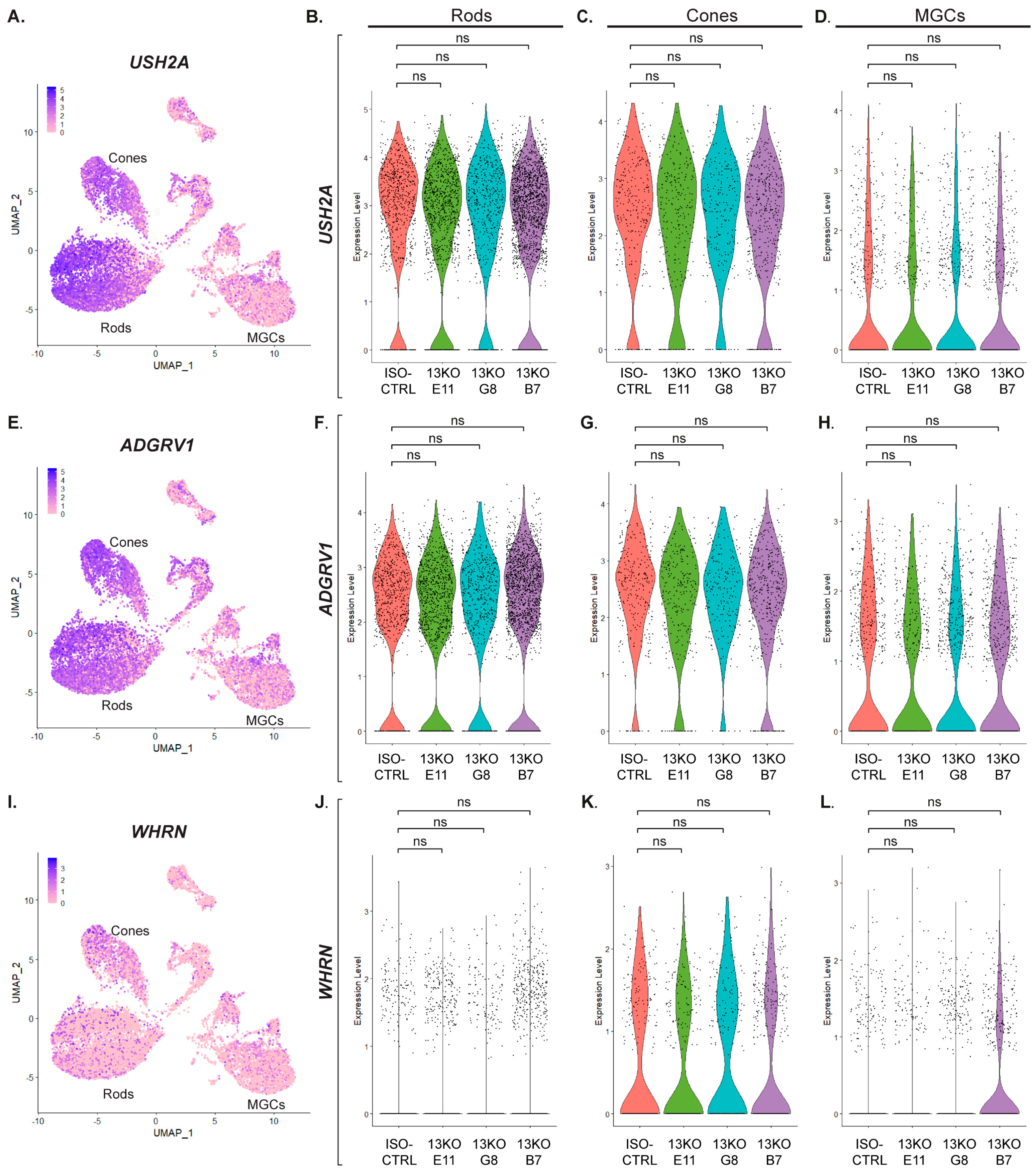
2.2. USH2A 13KO hiPSC-Derived Retinal Organoids Show Loss of Ciliary Localization of Usherin and Its Interacting Proteins
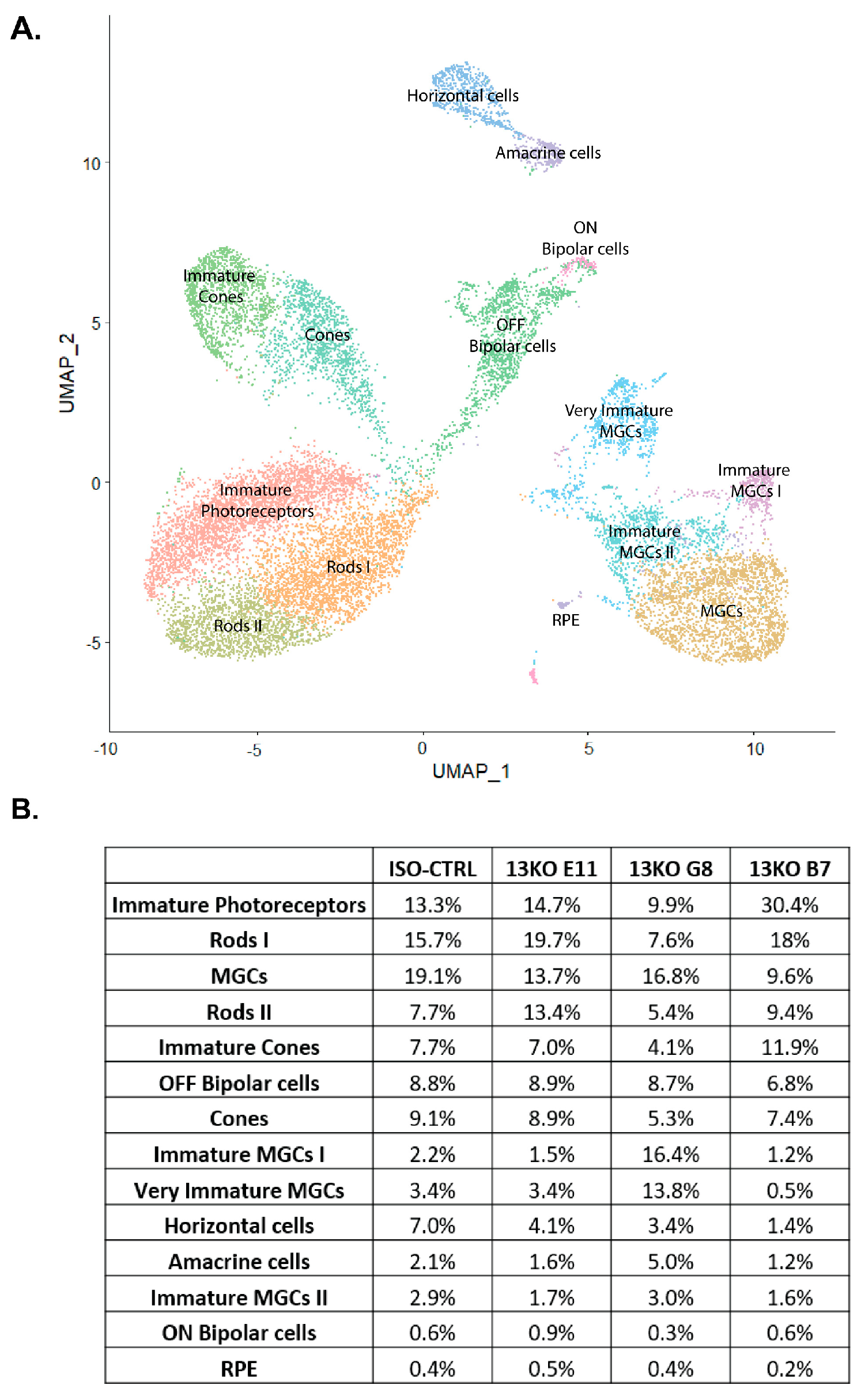
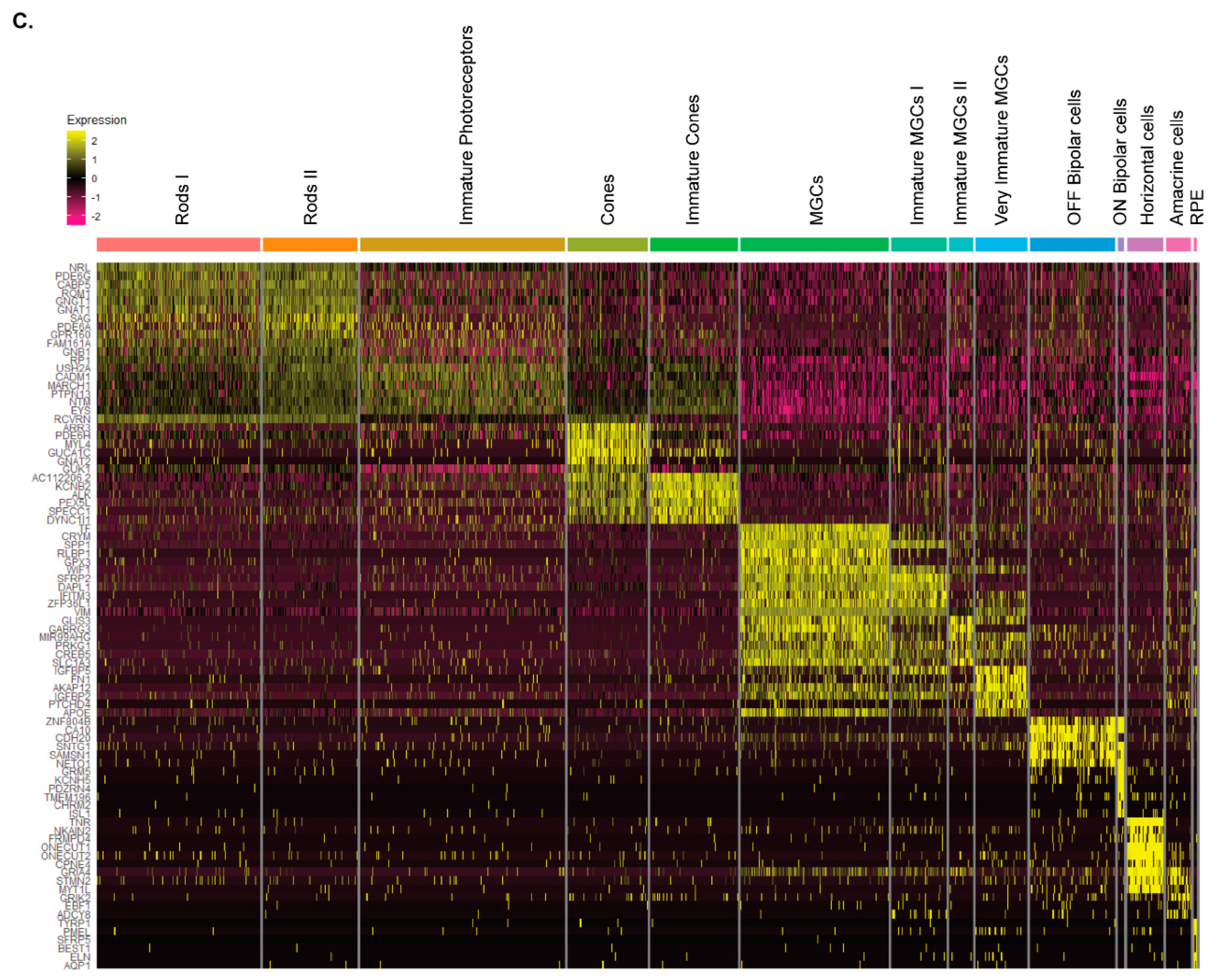
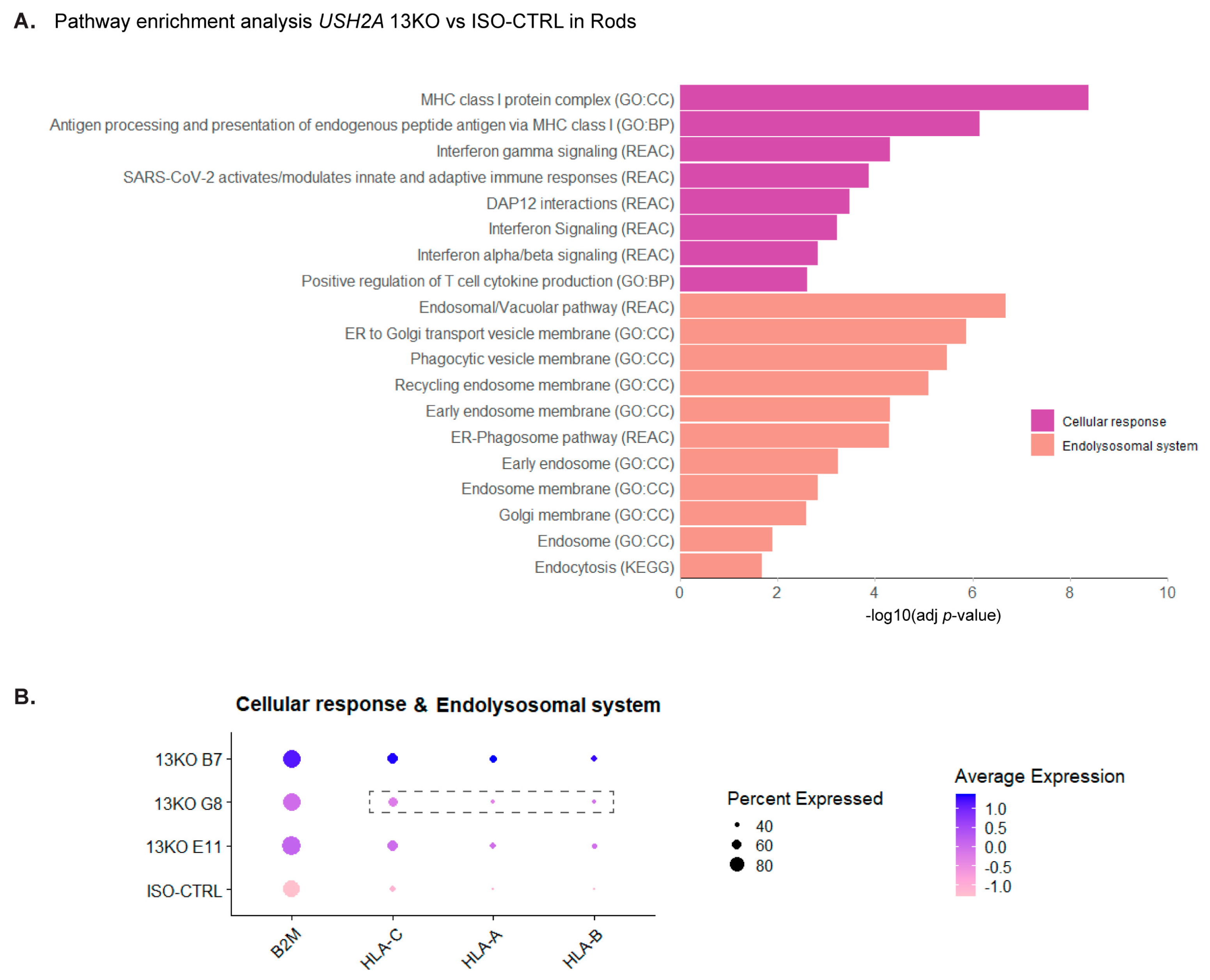
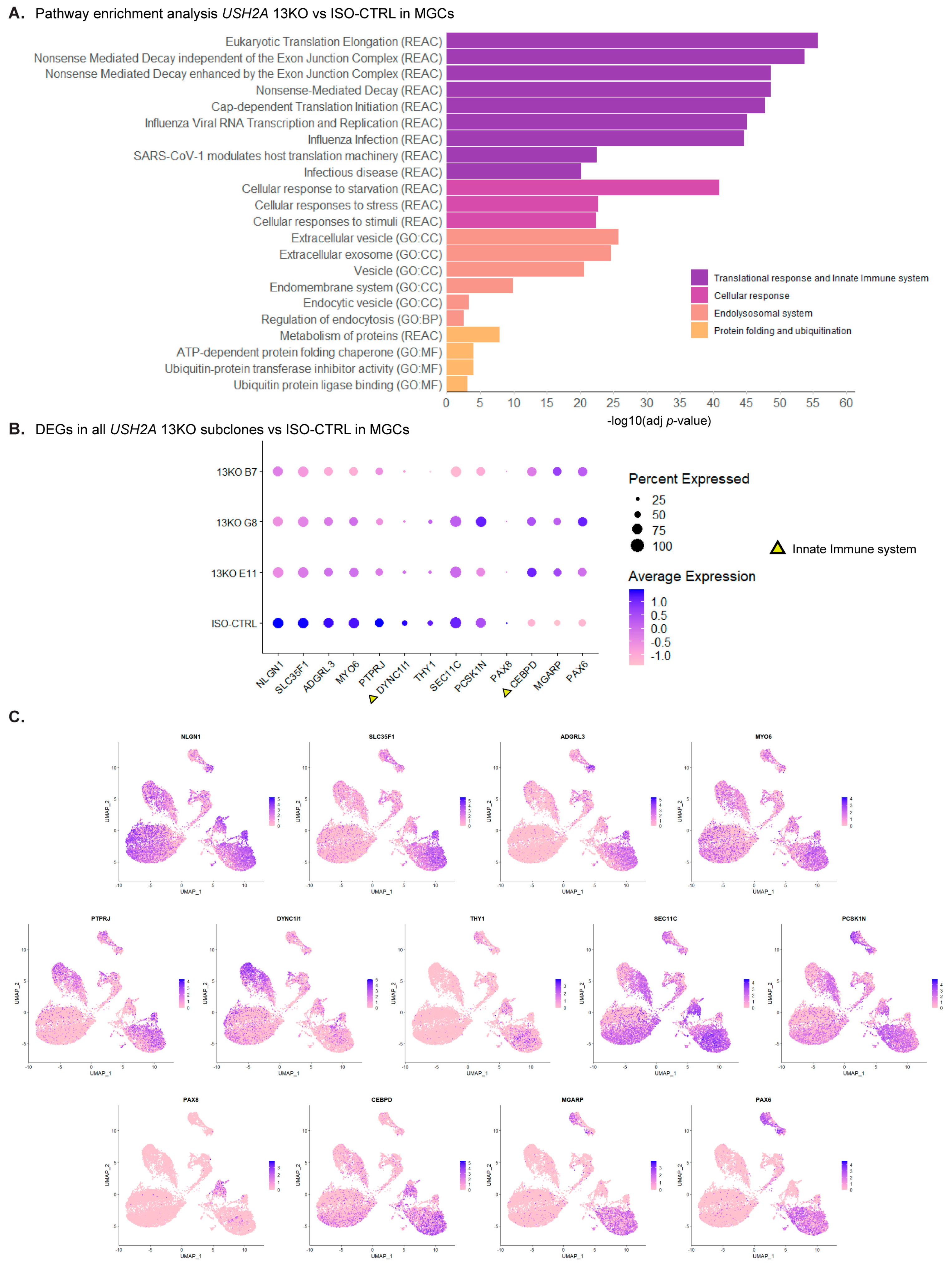
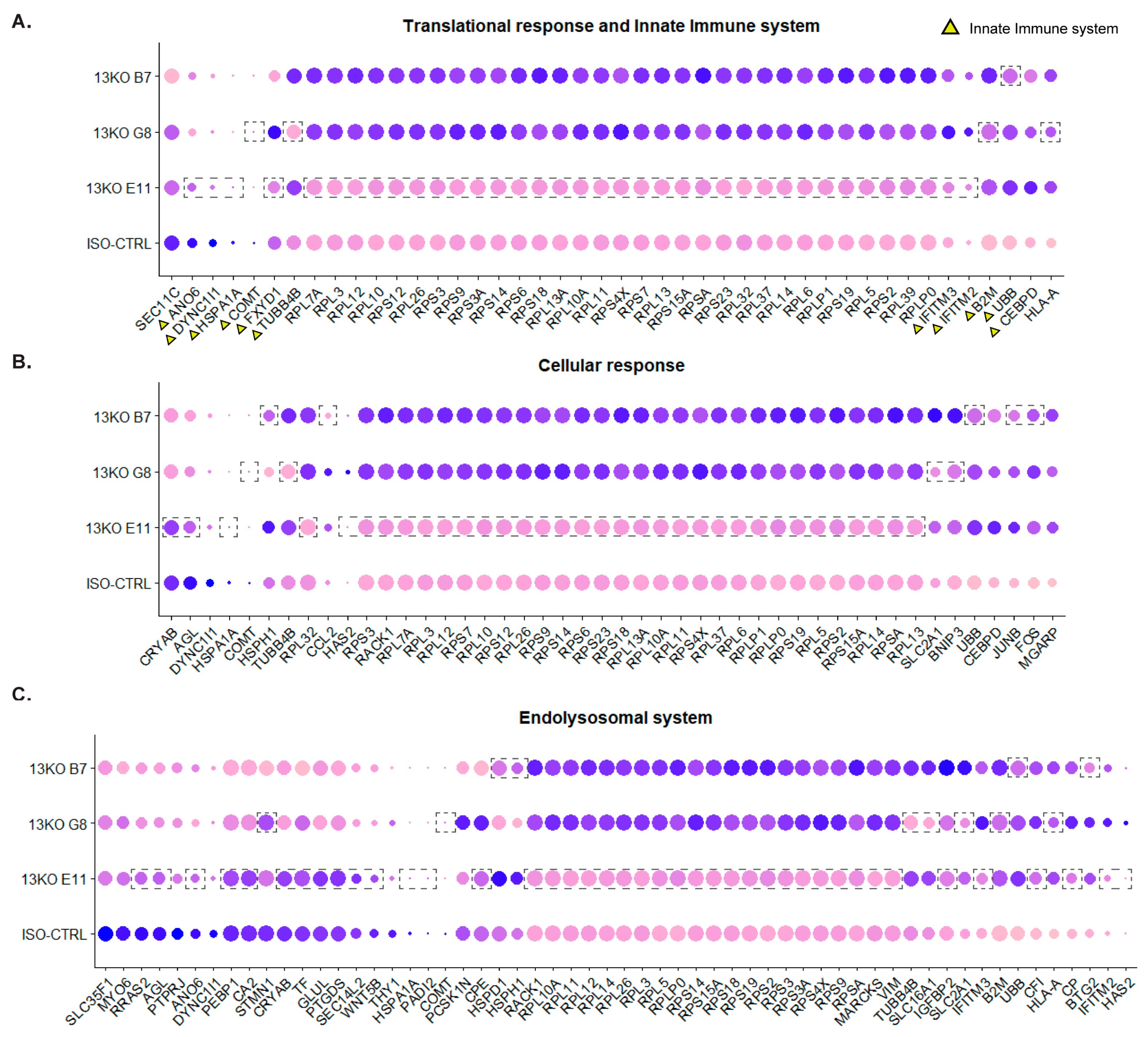
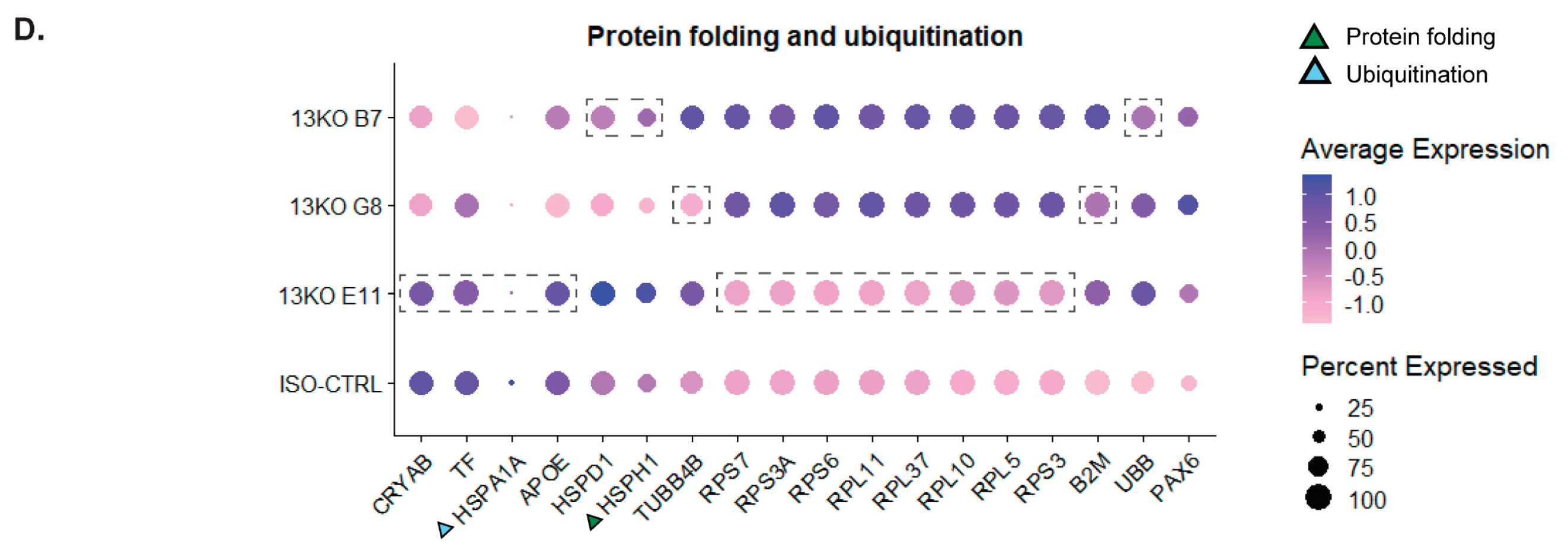
2.2. scRNA-Seq Analysis Reveals Activation of the Innate Immune Response and Disrupted Homeostasis in the USH2A 13KO Müller Glial Cells


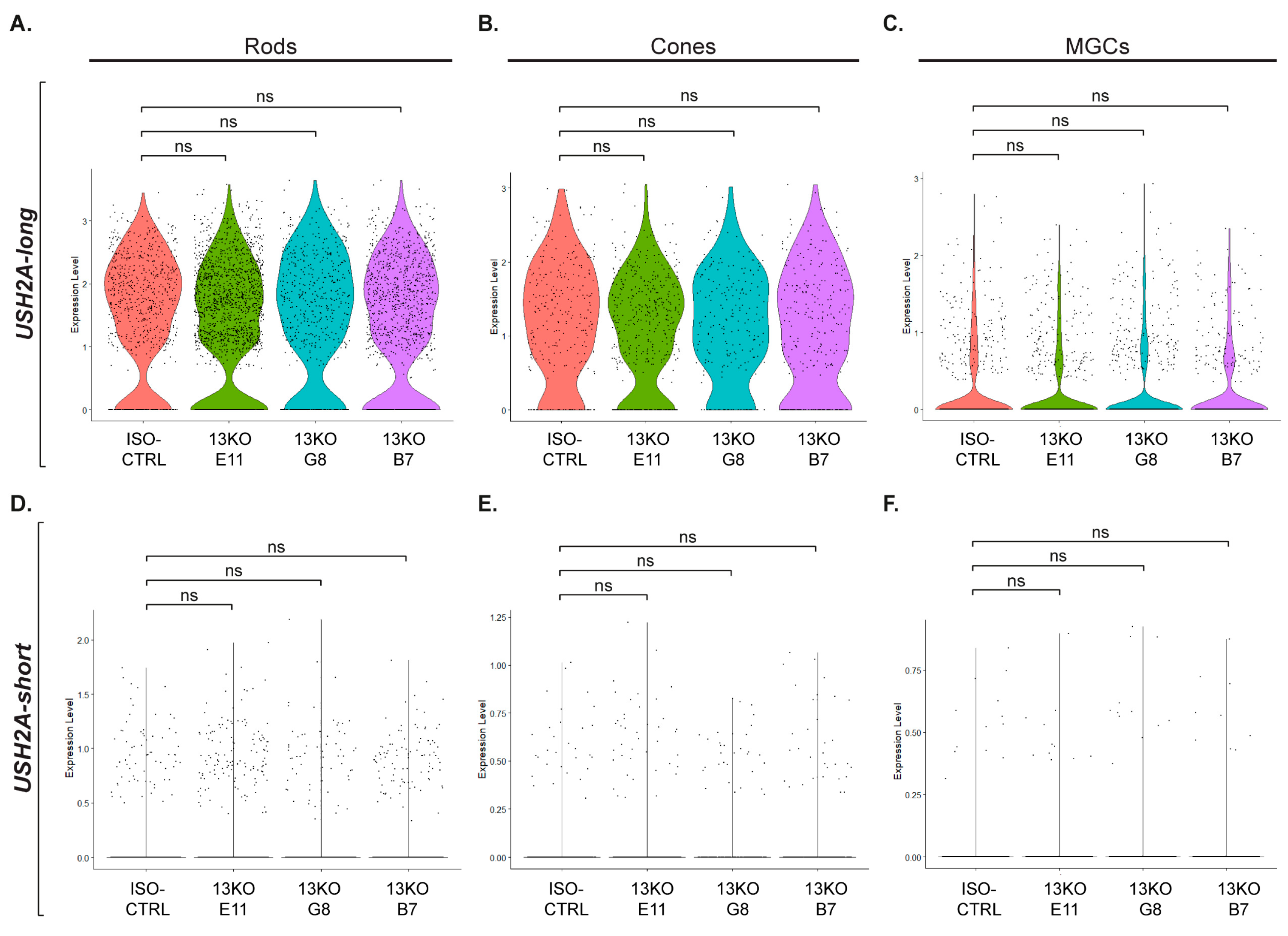
2.3. Expression of USH2A Transcripts Encoding Usherin Isoform A and Usherin Isoform B in the Retinal Organoids
2.4. The USH2A 13KO Müller Glial Cells Do Not Exhibit Morphological Changes
3. Discussion
4. Materials and Methods
4.1. CRISPR/Cas9-Mediated Genome Editing in hiPSCs
4.2. Cell Culture and Retinal Organoids Differentiation
4.3. Immunohistochemical Analysis
4.4. Quantification and Statistical Analysis
4.5. Single-Cell RNA Sequencing
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | Adeno-associated viral vectors |
| DD | Differentiation day |
| DEG | Differentially expressed gene |
| MGCs | Müller glial cells |
| ONL | Outer nuclear layer |
| TEM | Transmission electron microscopy |
| UMAP | Uniform Manifold Approximation and Projection |
References
- Ordonez-Labastida, V.; Chacon-Camacho, O.F.; Lopez-Rodriguez, V.R.; Zenteno, J.C. USH2A mutational spectrum causing syndromic and non-syndromic retinal dystrophies in a large cohort of Mexican patients. Mol Vis 2023, 29, 31–38. [Google Scholar]
- Eudy, J.D.; Weston, M.D.; Yao, S.; Hoover, D.M.; Rehm, H.L.; Ma-Edmonds, M.; Yan, D.; Ahmad, I.; Cheng, J.J.; Ayuso, C.; et al. Mutation of a gene encoding a protein with extracellular matrix motifs in Usher syndrome type IIa. Science 1998, 280, 1753–1757. [Google Scholar] [CrossRef]
- Liu, X.Z.; Hope, C.; Liang, C.Y.; Zou, J.M.; Xu, L.R.; Cole, T.; Mueller, R.F.; Bundey, S.; Nance, W.; Steel, K.P.; et al. A mutation (2314delG) in the Usher syndrome type IIA gene: high prevalence and phenotypic variation. Am J Hum Genet 1999, 64, 1221–1225. [Google Scholar] [CrossRef]
- Weston, M.D.; Eudy, J.D.; Fujita, S.; Yao, S.; Usami, S.; Cremers, C.; Greenberg, J.; Ramesar, R.; Martini, A.; Moller, C.; et al. Genomic structure and identification of novel mutations in usherin, the gene responsible for Usher syndrome type IIa. Am J Hum Genet 2000, 66, 1199–1210. [Google Scholar] [CrossRef]
- Baux, D.; Blanchet, C.; Hamel, C.; Meunier, I.; Larrieu, L.; Faugere, V.; Vache, C.; Castorina, P.; Puech, B.; Bonneau, D.; et al. Enrichment of LOVD-USHbases with 152 USH2A genotypes defines an extensive mutational spectrum and highlights missense hotspots. Hum Mutat 2014, 35, 1179–1186. [Google Scholar] [CrossRef]
- Dreyer, B.; Brox, V.; Tranebjaerg, L.; Rosenberg, T.; Sadeghi, A.M.; Moller, C.; Nilssen, O. Spectrum of USH2A mutations in Scandinavian patients with Usher syndrome type II. Hum Mutat 2008, 29, 451. [Google Scholar] [CrossRef] [PubMed]
- Aller, E.; Larrieu, L.; Jaijo, T.; Baux, D.; Espinos, C.; Gonzalez-Candelas, F.; Najera, C.; Palau, F.; Claustres, M.; Roux, A.F.; et al. The USH2A c.2299delG mutation: dating its common origin in a Southern European population. Eur J Hum Genet 2010, 18, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, B.; Tranebjaerg, L.; Brox, V.; Rosenberg, T.; Moller, C.; Beneyto, M.; Weston, M.D.; Kimberling, W.J.; Cremers, C.W.; Liu, X.Z.; et al. A common ancestral origin of the frequent and widespread 2299delG USH2A mutation. Am J Hum Genet 2001, 69, 228–234. [Google Scholar] [CrossRef]
- Perez-Carro, R.; Blanco-Kelly, F.; Galbis-Martinez, L.; Garcia-Garcia, G.; Aller, E.; Garcia-Sandoval, B.; Minguez, P.; Corton, M.; Mahillo-Fernandez, I.; Martin-Merida, I.; et al. Unravelling the pathogenic role and genotype-phenotype correlation of the USH2A p.(Cys759Phe) variant among Spanish families. PLoS One 2018, 13, e0199048. [Google Scholar] [CrossRef] [PubMed]
- Rivolta, C.; Berson, E.L.; Dryja, T.P. Paternal uniparental heterodisomy with partial isodisomy of chromosome 1 in a patient with retinitis pigmentosa without hearing loss and a missense mutation in the Usher syndrome type II gene USH2A. Arch Ophthalmol 2002, 120, 1566–1571. [Google Scholar] [CrossRef]
- Han, S.; Wang, Q.; Cheng, M.; Hu, Y.; Liu, P.; Hou, W.; Liang, L. The effects of ush2a gene knockout on vesicle transport in photoreceptors. Gene 2024, 892, 147885. [Google Scholar] [CrossRef]
- Liu, X.; Bulgakov, O.V.; Darrow, K.N.; Pawlyk, B.; Adamian, M.; Liberman, M.C.; Li, T. Usherin is required for maintenance of retinal photoreceptors and normal development of cochlear hair cells. Proc Natl Acad Sci U S A 2007, 104, 4413–4418. [Google Scholar] [CrossRef] [PubMed]
- Sahly, I.; Dufour, E.; Schietroma, C.; Michel, V.; Bahloul, A.; Perfettini, I.; Pepermans, E.; Estivalet, A.; Carette, D.; Aghaie, A.; et al. Localization of Usher 1 proteins to the photoreceptor calyceal processes, which are absent from mice. J Cell Biol 2012, 199, 381–399. [Google Scholar] [CrossRef]
- Dona, M.; Slijkerman, R.; Lerner, K.; Broekman, S.; Wegner, J.; Howat, T.; Peters, T.; Hetterschijt, L.; Boon, N.; de Vrieze, E.; et al. Usherin defects lead to early-onset retinal dysfunction in zebrafish. Exp Eye Res 2018, 173, 148–159. [Google Scholar] [CrossRef]
- Zou, J.; Li, R.; Wang, Z.; Yang, J. Studies of the Periciliary Membrane Complex in the Syrian Hamster Photoreceptor. Adv Exp Med Biol 2019, 1185, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Luo, L.; Shen, Z.; Chiodo, V.A.; Ambati, B.K.; Hauswirth, W.W.; Yang, J. Whirlin replacement restores the formation of the USH2 protein complex in whirlin knockout photoreceptors. Invest Ophthalmol Vis Sci 2011, 52, 2343–2351. [Google Scholar] [CrossRef]
- Reiners, J.; van Wijk, E.; Marker, T.; Zimmermann, U.; Jurgens, K.; te Brinke, H.; Overlack, N.; Roepman, R.; Knipper, M.; Kremer, H.; et al. Scaffold protein harmonin (USH1C) provides molecular links between Usher syndrome type 1 and type 2. Hum Mol Genet 2005, 14, 3933–3943. [Google Scholar] [CrossRef] [PubMed]
- Sorusch, N.; Bauss, K.; Plutniok, J.; Samanta, A.; Knapp, B.; Nagel-Wolfrum, K.; Wolfrum, U. Characterization of the ternary Usher syndrome SANS/ush2a/whirlin protein complex. Hum Mol Genet 2017, 26, 1157–1172. [Google Scholar] [CrossRef]
- Knapp, B.; Roedig, J.; Roedig, H.; Krzysko, J.; Horn, N.; Guler, B.E.; Kusuluri, D.K.; Yildirim, A.; Boldt, K.; Ueffing, M.; et al. Affinity Proteomics Identifies Interaction Partners and Defines Novel Insights into the Function of the Adhesion GPCR VLGR1/ADGRV1. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Linnert, J.; Knapp, B.; Guler, B.E.; Boldt, K.; Ueffing, M.; Wolfrum, U. Usher syndrome proteins ADGRV1 (USH2C) and CIB2 (USH1J) interact and share a common interactome containing TRiC/CCT-BBS chaperonins. Front Cell Dev Biol 2023, 11, 1199069. [Google Scholar] [CrossRef]
- Krzysko, J.; Maciag, F.; Mertens, A.; Guler, B.E.; Linnert, J.; Boldt, K.; Ueffing, M.; Nagel-Wolfrum, K.; Heine, M.; Wolfrum, U. The Adhesion GPCR VLGR1/ADGRV1 Regulates the Ca(2+) Homeostasis at Mitochondria-Associated ER Membranes. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Linnert, J.; Guler, B.E.; Krzysko, J.; Wolfrum, U. The adhesion G protein-coupled receptor VLGR1/ADGRV1 controls autophagy. Basic Clin Pharmacol Toxicol 2023, 133, 313–330. [Google Scholar] [CrossRef] [PubMed]
- Maddalena, A.; Tornabene, P.; Tiberi, P.; Minopoli, R.; Manfredi, A.; Mutarelli, M.; Rossi, S.; Simonelli, F.; Naggert, J.K.; Cacchiarelli, D.; et al. Triple Vectors Expand AAV Transfer Capacity in the Retina. Mol Ther 2018, 26, 524–541. [Google Scholar] [CrossRef] [PubMed]
- Sanjurjo-Soriano, C.; Erkilic, N.; Baux, D.; Mamaeva, D.; Hamel, C.P.; Meunier, I.; Roux, A.F.; Kalatzis, V. Genome Editing in Patient iPSCs Corrects the Most Prevalent USH2A Mutations and Reveals Intriguing Mutant mRNA Expression Profiles. Mol Ther Methods Clin Dev 2020, 17, 156–173. [Google Scholar] [CrossRef]
- Tachida, Y.; Manian, K.V.; Butcher, R.; Levy, J.M.; Pendse, N.; Hennessey, E.; Liu, D.R.; Pierce, E.A.; Liu, Q.; Comander, J. Systematic empirical evaluation of individual base editing targets: validating therapeutic targets in USH2A and comparison of methods. Mol Ther 2025. [Google Scholar] [CrossRef]
- Samanta, A.; Stingl, K.; Kohl, S.; Ries, J.; Linnert, J.; Nagel-Wolfrum, K. Ataluren for the Treatment of Usher Syndrome 2A Caused by Nonsense Mutations. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Dulla, K.; Slijkerman, R.; van Diepen, H.C.; Albert, S.; Dona, M.; Beumer, W.; Turunen, J.J.; Chan, H.L.; Schulkens, I.A.; Vorthoren, L.; et al. Antisense oligonucleotide-based treatment of retinitis pigmentosa caused by USH2A exon 13 mutations. Mol Ther 2021, 29, 2441–2455. [Google Scholar] [CrossRef]
- Liu, X.; Lillywhite, J.; Zhu, W.; Huang, Z.; Clark, A.M.; Gosstola, N.; Maguire, C.T.; Dykxhoorn, D.; Chen, Z.Y.; Yang, J. Generation and Genetic Correction of USH2A c.2299delG Mutation in Patient-Derived Induced Pluripotent Stem Cells. Genes (Basel) 2021, 12. [Google Scholar] [CrossRef]
- Boon, N.; Lu, X.; Andriessen, C.A.; Moustakas, I.; Buck, T.M.; Freund, C.; Arendzen, C.H.; Bohringer, S.; Mei, H.; Wijnholds, J. AAV-mediated gene augmentation therapy of CRB1 patient-derived retinal organoids restores the histological and transcriptional retinal phenotype. Stem Cell Reports 2023, 18, 1123–1137. [Google Scholar] [CrossRef]
- Buck, T.M.; Quinn, P.M.J.; Pellissier, L.P.; Mulder, A.A.; Jongejan, A.; Lu, X.; Boon, N.; Koot, D.; Almushattat, H.; Arendzen, C.H.; et al. CRB1 is required for recycling by RAB11A+ vesicles in human retinal organoids. Stem Cell Reports 2023, 18, 1793–1810. [Google Scholar] [CrossRef]
- Sanjurjo-Soriano, C.; Jimenez-Medina, C.; Erkilic, N.; Cappellino, L.; Lefevre, A.; Nagel-Wolfrum, K.; Wolfrum, U.; Van Wijk, E.; Roux, A.F.; Meunier, I.; et al. USH2A variants causing retinitis pigmentosa or Usher syndrome provoke differential retinal phenotypes in disease-specific organoids. HGG Adv 2023, 4, 100229. [Google Scholar] [CrossRef]
- Suzuki, K.; Tsunekawa, Y.; Hernandez-Benitez, R.; Wu, J.; Zhu, J.; Kim, E.J.; Hatanaka, F.; Yamamoto, M.; Araoka, T.; Li, Z.; et al. In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 2016, 540, 144–149. [Google Scholar] [CrossRef]
- Quinn, P.M.; Buck, T.M.; Mulder, A.A.; Ohonin, C.; Alves, C.H.; Vos, R.M.; Bialecka, M.; van Herwaarden, T.; van Dijk, E.H.C.; Talib, M.; et al. Human iPSC-Derived Retinas Recapitulate the Fetal CRB1 CRB2 Complex Formation and Demonstrate that Photoreceptors and Muller Glia Are Targets of AAV5. Stem Cell Reports 2019, 12, 906–919. [Google Scholar] [CrossRef]
- Guo, C.; Ma, X.; Gao, F.; Guo, Y. Off-target effects in CRISPR/Cas9 gene editing. Front Bioeng Biotechnol 2023, 11, 1143157. [Google Scholar] [CrossRef]
- McDonald, A.; Gallego, C.; Andriessen, C.; Orlova, M.; Goncalves, M.; Wijnholds, J. Conventional and Tropism-Modified High-Capacity Adenoviral Vectors Exhibit Similar Transduction Profiles in Human iPSC-Derived Retinal Organoids. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Mathur, P.D.; Zheng, T.; Wang, Y.; Almishaal, A.; Park, A.H.; Yang, J. Individual USH2 proteins make distinct contributions to the ankle link complex during development of the mouse cochlear stereociliary bundle. Hum Mol Genet 2015, 24, 6944–6957. [Google Scholar] [CrossRef] [PubMed]
- Tebbe, L.; Mwoyosvi, M.L.; Crane, R.; Makia, M.S.; Kakakhel, M.; Cosgrove, D.; Al-Ubaidi, M.R.; Naash, M.I. The usherin mutation c.2299delG leads to its mislocalization and disrupts interactions with whirlin and VLGR1. Nat Commun 2023, 14, 972. [Google Scholar] [CrossRef]
- Gomez-Herranz, M.; Taylor, J.; Sloan, R.D. IFITM proteins: Understanding their diverse roles in viral infection, cancer, and immunity. J Biol Chem 2023, 299, 102741. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Wang, L.; Liao, X.; Huang, B.; Luo, C.; Liao, G.; Yuan, L.; Liu, X.; Luo, H.; Shu, Y. Research Progress into the Biological Functions of IFITM3. Viruses 2024, 16. [Google Scholar] [CrossRef]
- Wang, J.; Luo, Y.; Katiyar, H.; Liang, C.; Liu, Q. The Antiviral Activity of Interferon-Induced Transmembrane Proteins and Virus Evasion Strategies. Viruses 2024, 16. [Google Scholar] [CrossRef]
- Navneet, S.; Wilson, K.; Rohrer, B. Muller Glial Cells in the Macula: Their Activation and Cell-Cell Interactions in Age-Related Macular Degeneration. Invest Ophthalmol Vis Sci 2024, 65, 42. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Larbi, D.; Andrade, M.; Reardon, S.; Reh, T.A.; Wohl, S.G. A Comparative Analysis of Reactive Muller Glia Gene Expression After Light Damage and microRNA-Depleted Muller Glia-Focus on microRNAs. Front Cell Dev Biol 2020, 8, 620459. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.S.; Farzan, M. The broad-spectrum antiviral functions of IFIT and IFITM proteins. Nat Rev Immunol 2013, 13, 46–57. [Google Scholar] [CrossRef] [PubMed]
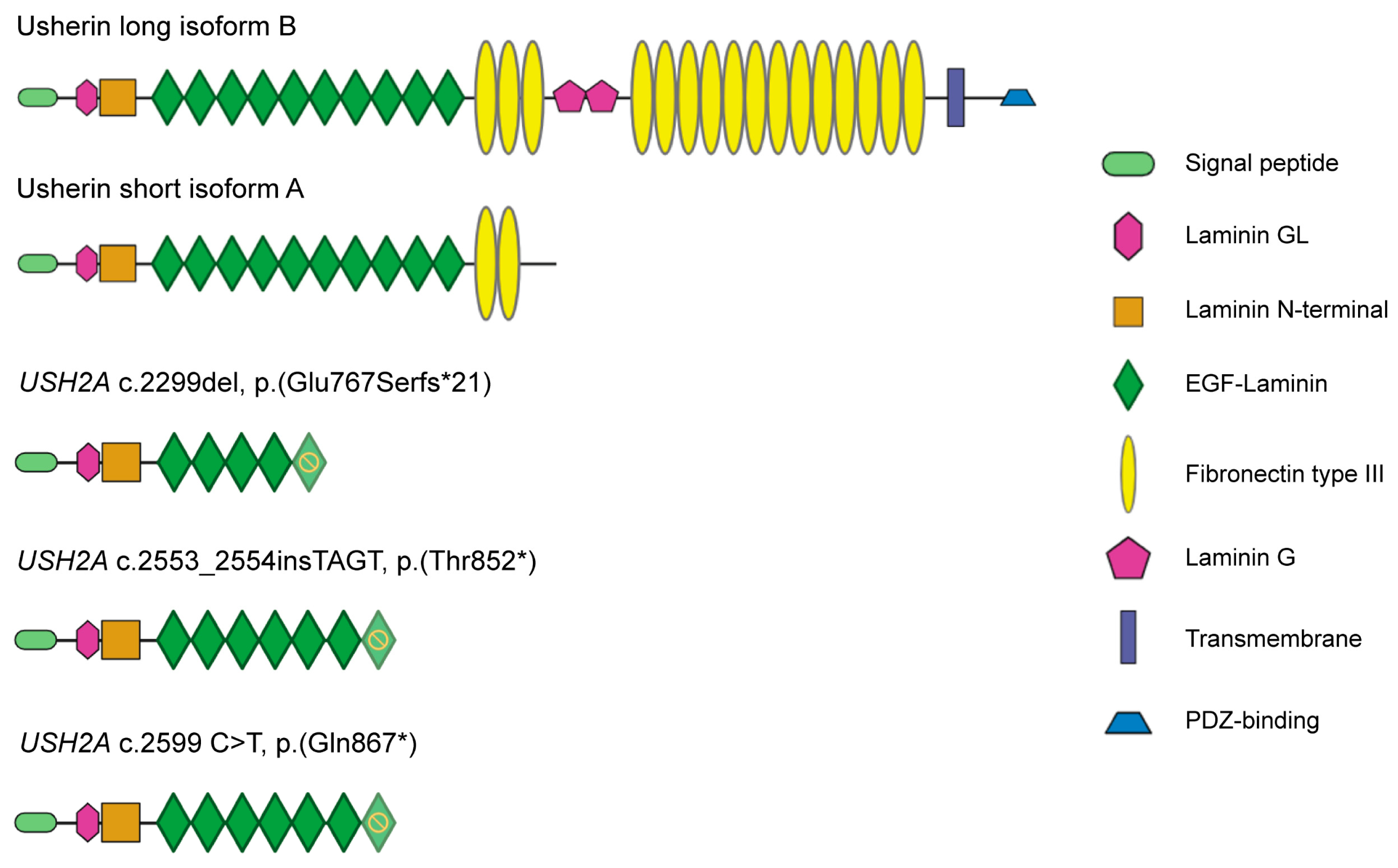
| USH2A mutation | Usherin change | Usherin size (aa) | Protein domain affected |
|---|---|---|---|
| c.2311 G>T | p.(Glu771Ter) | 770 | EGF-Lam like 5 |
| c.2299del | p.(Glu767Serfs*21) | 786 (766+20) | EGF-Lam like 5 |
| c.2310del | p.(Glu771Lysfs*17) | 786 (770+16) | EGF-Lam like 5 |
| c.2314delG | p.(Ala772Profs*16) | 786(771+15) | EGF-Lam like 5 |
| c.2344del | p.Glu782Lysfs*6) | 786 (781+5) | EGF-Lam like 5 |
| c.2391_2392del | p.(Cys797*) | 796 | EGF-Lam like 6 |
| c.2431 A>T | p.(Lys811*) | 810 | EGF-Lam like 6 |
| c.2431_2432_del | p.(Lys811Aspfs*11) | 820 (810+10) | EGF-Lam like 6 |
| c.2440 C>T | p.(Gln814*) | 813 | EGF-Lam like 6 |
| c.2512 C>T | p.(Glu838*) | 837 | EGF-Lam like 6 |
| c.2541 C>A | p.(Cys847*) | 846 | EGF-Lam like 7 |
| c.2553_2554insTAGT | p.(Thr852*) | 851 | EGF-Lam like 7 |
| c.2599 C>T | p.(Gln867*) | 866 | EGF-Lam like 7 |
| c.2610 C>A | p.(Cys870*) | 869 | EGF-Lam like 7 |
| C.2661 C>A | p.(Tyr887) | 886 | EGF-Lam like 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




