Submitted:
26 November 2025
Posted:
27 November 2025
You are already at the latest version
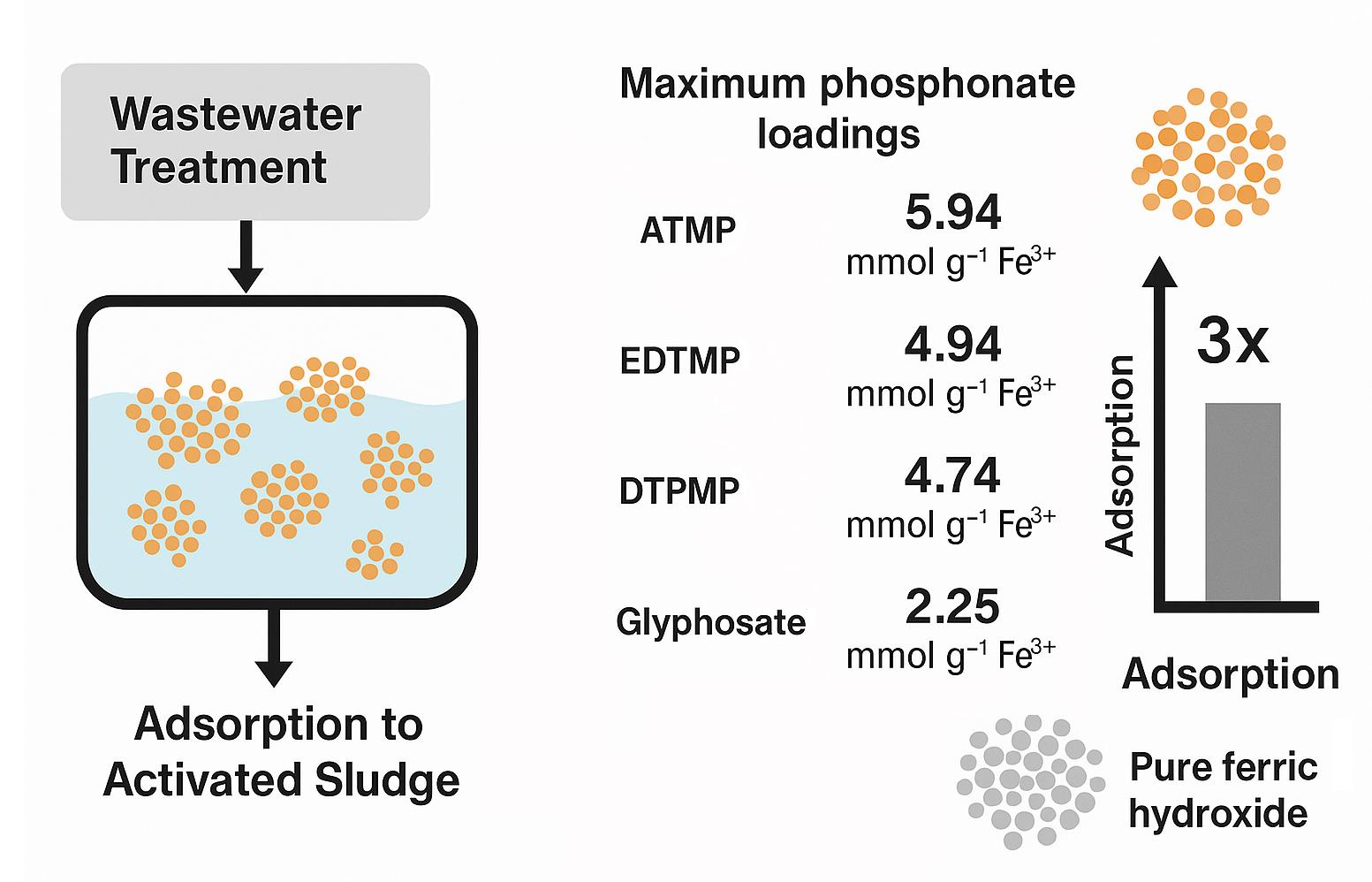
Abstract
Keywords:
1. Introduction
2. Materials and Methods
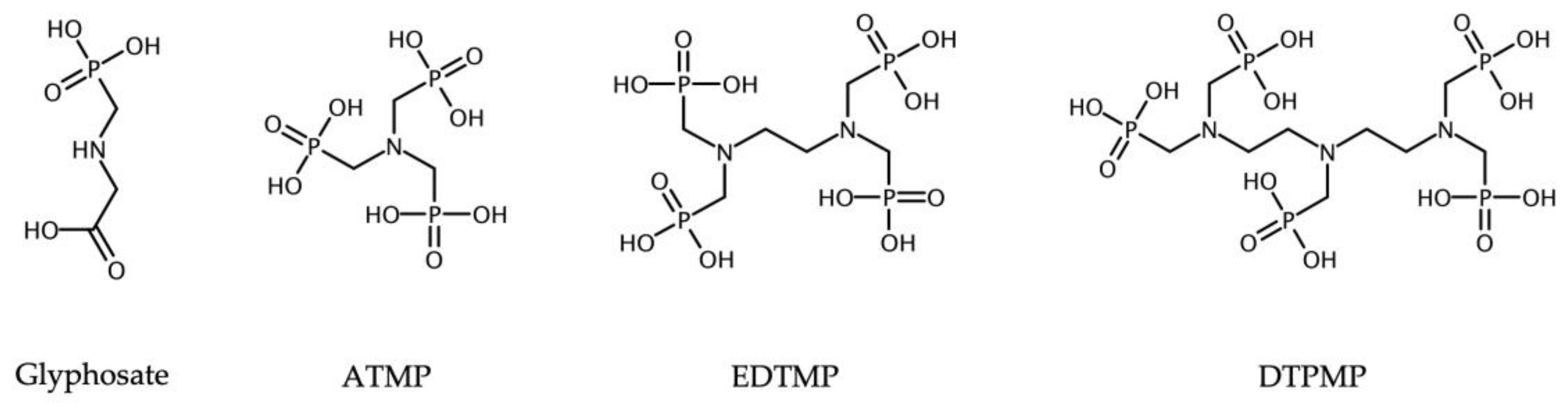
2.1. Chemicals
2.2. Experimental Procedure
2.3. Analytical Methods
2.4. Adsorption Isotherm Calculations
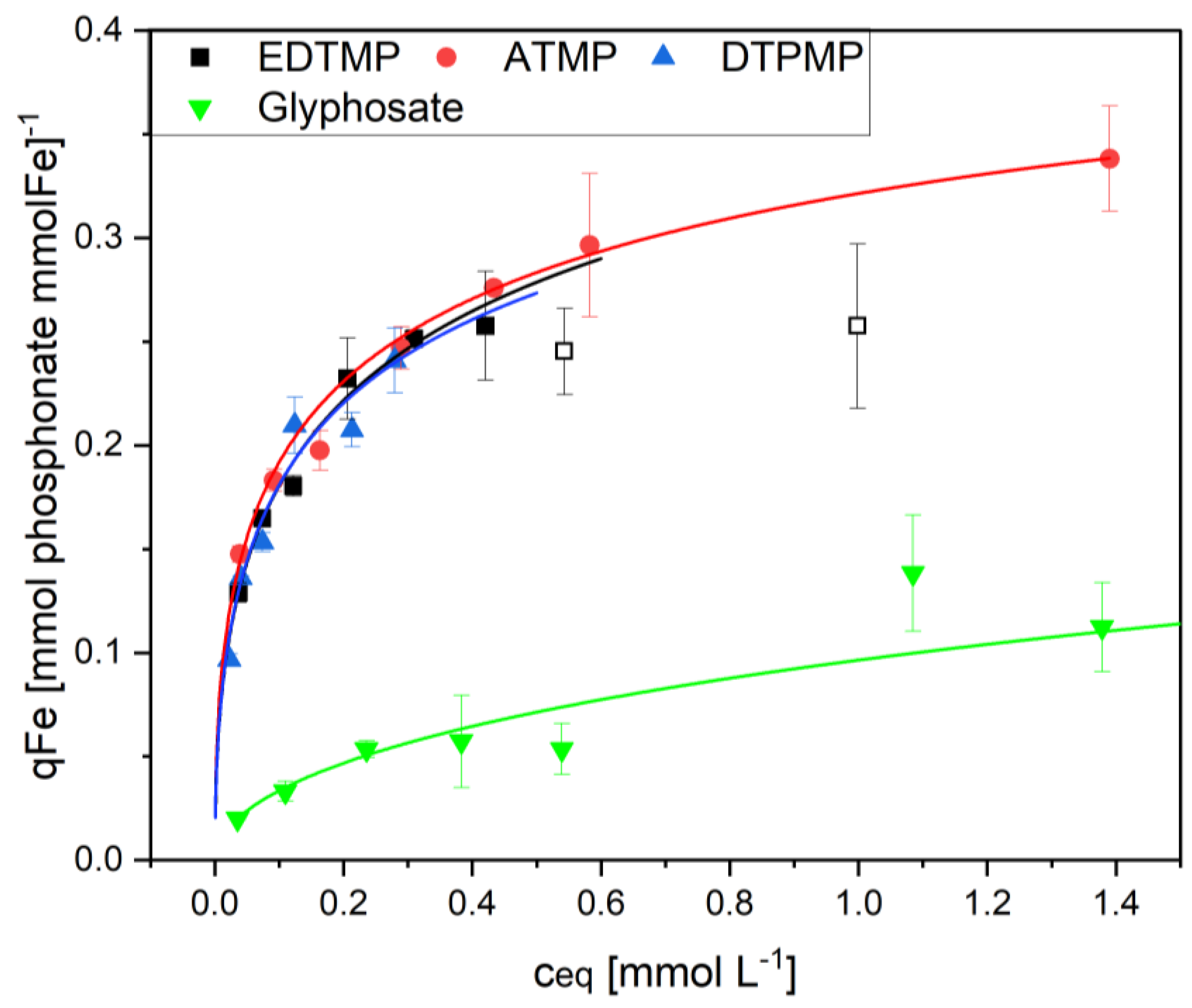
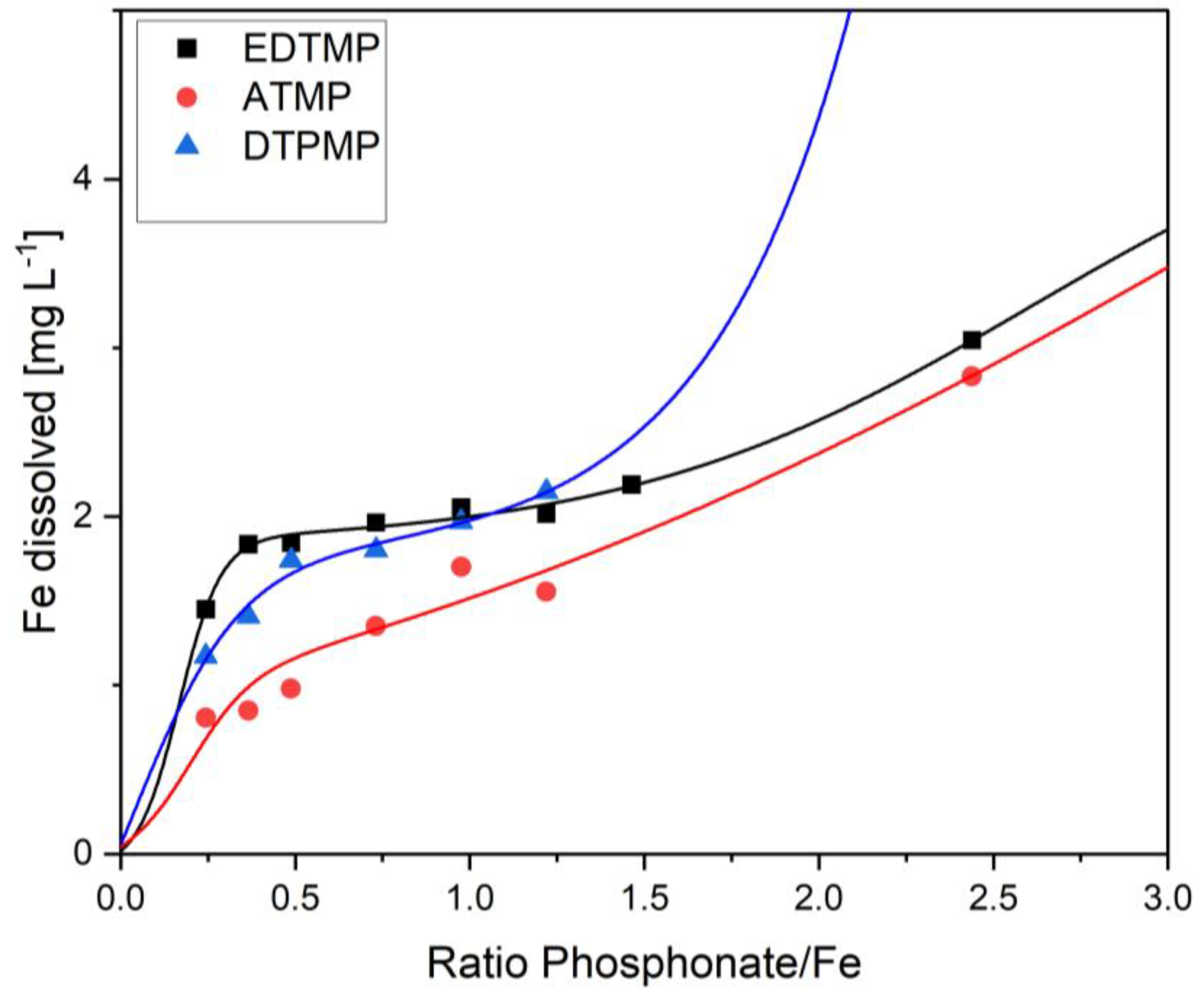
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATMP | Aminotrimethylene Phosphonic Acid |
| EDTMP | Ethylenediaminetetra(methylene Phosphonic Acid) |
| DTPMP | Diethylenetriaminepenta(methylene Phosphonic Acid) |
| HEDP | Hydroxyethylidene Diphosphonic Acid |
| IDMP | Iminodi(methylene Phosphonic Acid) |
| DUP | Dissolved unreactive phosphorus |
| PP | Particulate phosphorus |
| WWTP | Wastewater treatment plant |
References
- Rott, E.; Steinmetz, H.; Metzger, J. W. Organophosphonates: A review on environmental relevance, biodegradability and removal in wastewater treatment plants. Sci.Total Environ. 2018, 615, 1176–1191. [Google Scholar] [CrossRef]
- Rott, E.; Happel, O.; Armbruster, D.; Minke, R. Behavior of PBTC, HEDP, and Aminophosphonates in the Process of Wastewater Treatment. Water 2020, 12, 53. [Google Scholar] [CrossRef]
- Li, C.; Yang, Q.; Lu, S.; Liu, Y. Adsorption and mechanism study for phosphonate antiscalant HEDP removal from reverse osmosis concentrates by magnetic La/Zn/Fe3O4@ PAC composite. Colloid. Surf. A 2021, 613, 126056. [Google Scholar] [CrossRef]
- Rott, E.; Nouri, M.; Meyer, C.; Minke, R.; Schneider, M.; Mandel, K.; Drenkova-Tuhtan, A. Removal of phosphonates from synthetic and industrial wastewater with reusable magnetic adsorbent particles. Water Res. 2018, 145, 608–617. [Google Scholar] [CrossRef]
- He, G.; Lao, Q.; Jin, G.; Zhu, Q.; Chen, F. Increasing eutrophication driven by the increase of phosphate discharge in a subtropical bay in the past 30 years. Front. Mar. Sci., Sec. Marine Biogeochem 2023, 10. [Google Scholar] [CrossRef]
- Poiger, T.; Keller, M.; Buerge, I.J.; Balmer, M.E. Behavior of glyphosate in wastewater treatment plants. Chimia 2020, 74. [Google Scholar] [CrossRef] [PubMed]
- Venditti, S.; Kiesch, A.; Hansen, J. Fate of glyphosate and its metabolite AminoMethylPhosponic acid (AMPA) from point source through wastewater sludge and advanced treatment. Chemosphere 2023, 340, 139843. [Google Scholar] [CrossRef] [PubMed]
- Manav, M. C.; Sofos, N.; Hove-Jensen, B.; Brodersen, D. E. The Abc of Phosphonate Breakdown: A Mechanism for Bacterial Survival. BioEssays 2018, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ruffolo, F.; Dinhof, T.; Murray, L.; Zangelmi, E.; Chin, J. P.; Pallitsch, K.; Peracchi, A. The Microbial Degradation of Natural and Anthropogenic Phosphonates. Molecules 2023, 28, 6863. [Google Scholar] [CrossRef]
- Riedel, R.; Krahl, K.; Buder, K.; Böllmann, J.; Braun, B.; Martienssen, M. Novel standard biodegradation test for synthetic phosphonates, J. Microbiol. Methods 2023, 212, 106793. [Google Scholar] [CrossRef]
- Nowack, B. Aminopolyphosphonate removal during wastewater treatment. Water Res. 2002, 36, 4636–4642. [Google Scholar] [CrossRef] [PubMed]
- Altaf, R.; Sun, B.; Lu, H.; Zhao, H.; Liu, D. Removal and recovery of phosphonates from wastewater via adsorption. Crit. Rev. Environ. Sci. Technol. 2022, 53(9), 1032–1058. [Google Scholar] [CrossRef]
- Armbruster, D.; Rott, E.; Minke, R.; Happel, O. Trace-level determination of phosphonates in liquid and solid phase of wastewater and environmental samples by IC-ESI-MS/MS. Bioanal. Chem. 2020, 412, 4807–4825. [Google Scholar] [CrossRef]
- Michalska, J.; Turek-Szytow, J.; Dudło, A.; Surmacz-Górska, J. Characterization of humic substances recovered from the sewage sludge and validity of their removal from this waste. EFB Bioeconomy J. 2022, 2, 100026. [Google Scholar] [CrossRef]
- Tang, S.; Zheng, C.; Zhang, Z. Effect of inherent minerals on sewage sludge pyrolysis: Product characteristics, kinetics and thermodynamics. Waste Manag. 2018, 80, 175–185. [Google Scholar] [CrossRef]
- Nowack, B.; Stone, A. T. Adsorption of Phosphonates onto the Goethite–Water Interface. J. Colloid Interface Sci. 1999, 214, 20–30. [Google Scholar] [CrossRef]
- Reinhardt, T.; Gómez Elordi, M.; Minke, R.; Schönberger, H.; Rott, E. Batch studies of phosphonate adsorption on granular ferric hydroxides. Water Sci. Technol. 2020, 81, 10–20. [Google Scholar] [CrossRef]
- Rott, E.; Minke, R.; Steinmetz, H. Removal of phosphorus from phosphonate-loaded industrial wastewaters via precipitation/flocculation. J. Water Process Eng. 2017, 17, 188–196. [Google Scholar] [CrossRef]
- Liu, Y.; Yao, M.; Jin, Z. Simultaneous adsorption of organic phosphonate and orthophosphate from municipal sewage on nanoparticle Fe3O4 with molecular simulation. npj Clean Water 2025, 8, 40. [Google Scholar] [CrossRef]
- Chen, Y.; Baygents, J.C.; Farrell, J. Removing phosphonate antiscalants from membrane concentrate solutions using granular ferric hydroxide. J. Water Process. Eng. 2017, 19, 18–25. [Google Scholar] [CrossRef]
- Lu, J.; Liu, H.; Zhao, X.; Jefferson, W.; Cheng, F.; Qu, J. Phosphate removal from water using freshly formed Fe–Mn binary oxide: adsorption behaviors and mechanisms. Colloid. Surf. A 2014, 455, 11–18. [Google Scholar] [CrossRef]
- Irfan, I.; Inam, M. A.; Iftikhar, R. Comparative phosphate sorption and recovery potential of mono and bimetallic iron-lanthanum impregnated biochar derived via co-pyrolysis of sewage sludge and wheat straw: Highly effective phosphatic fertilizer. J. Water Process. Eng. 2024, 66, 106110. [Google Scholar] [CrossRef]
- Hinz, C. Description of sorption data with isotherm equations. Geoderma 2001, 99, 225–243. [Google Scholar] [CrossRef]
- Nowack, B. , Stone, A. T. Competitive adsorption of phosphate and phosphonates onto goethite. Water Res. 2006, 40, 2201–2209. [Google Scholar] [CrossRef]
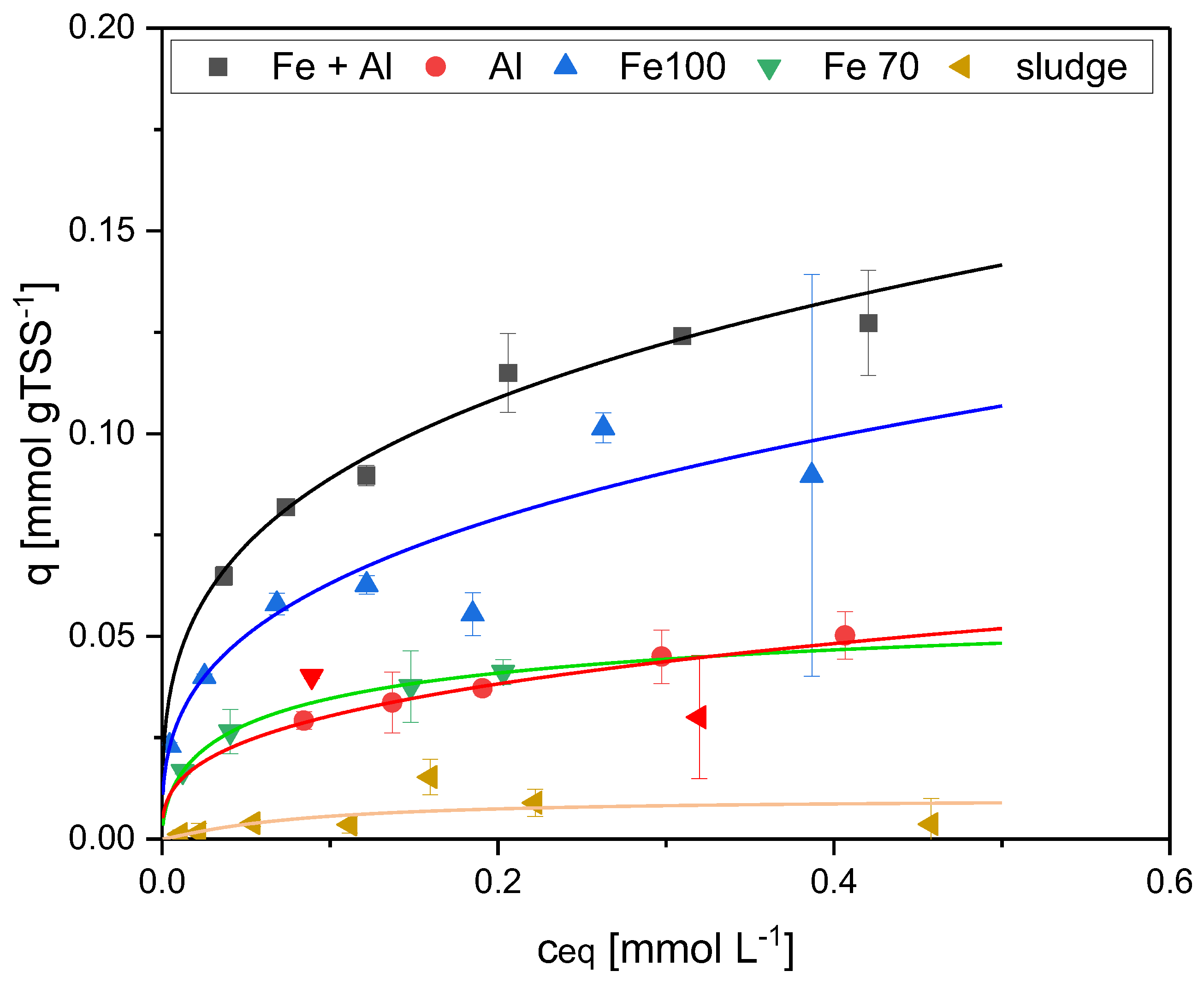
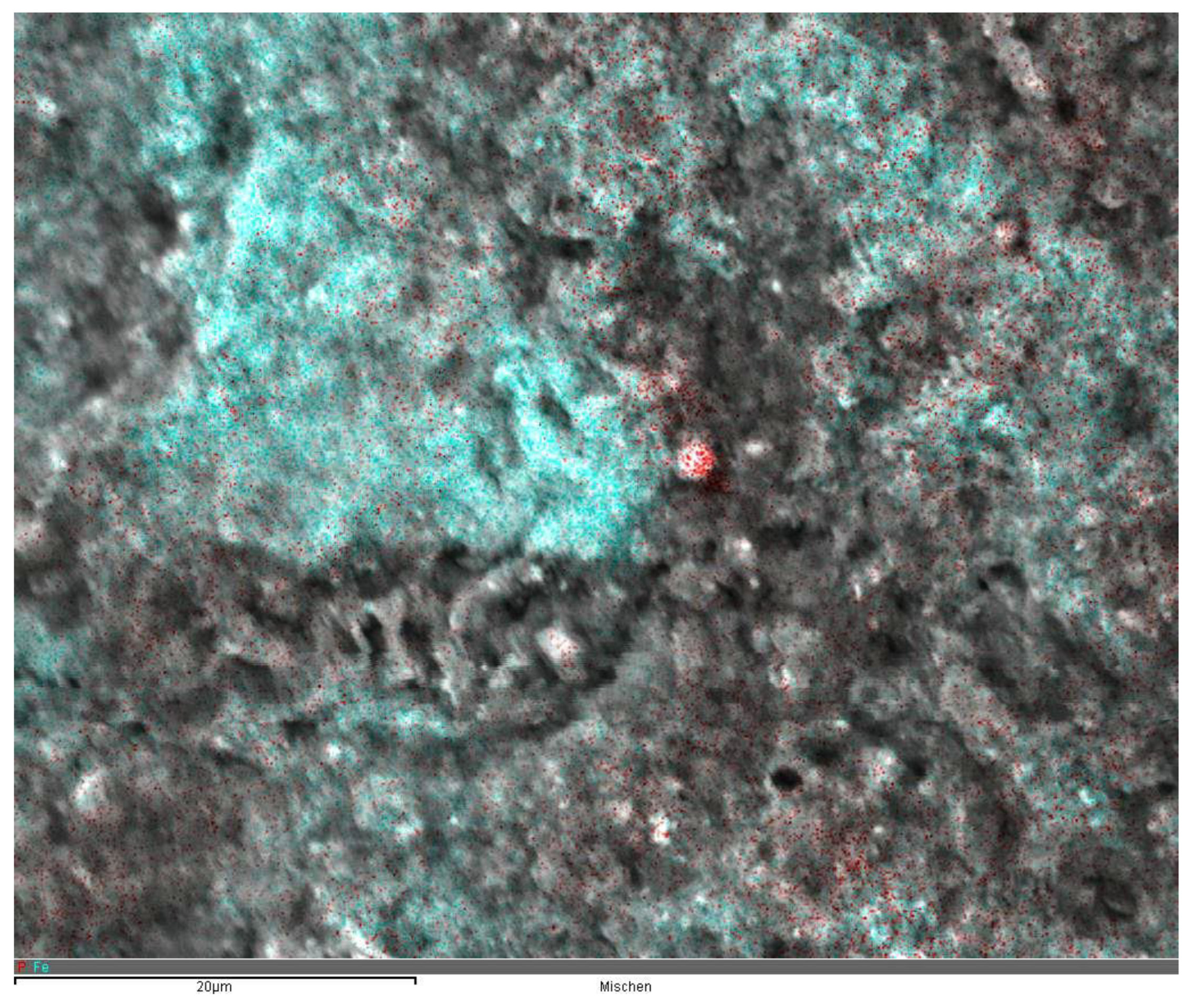
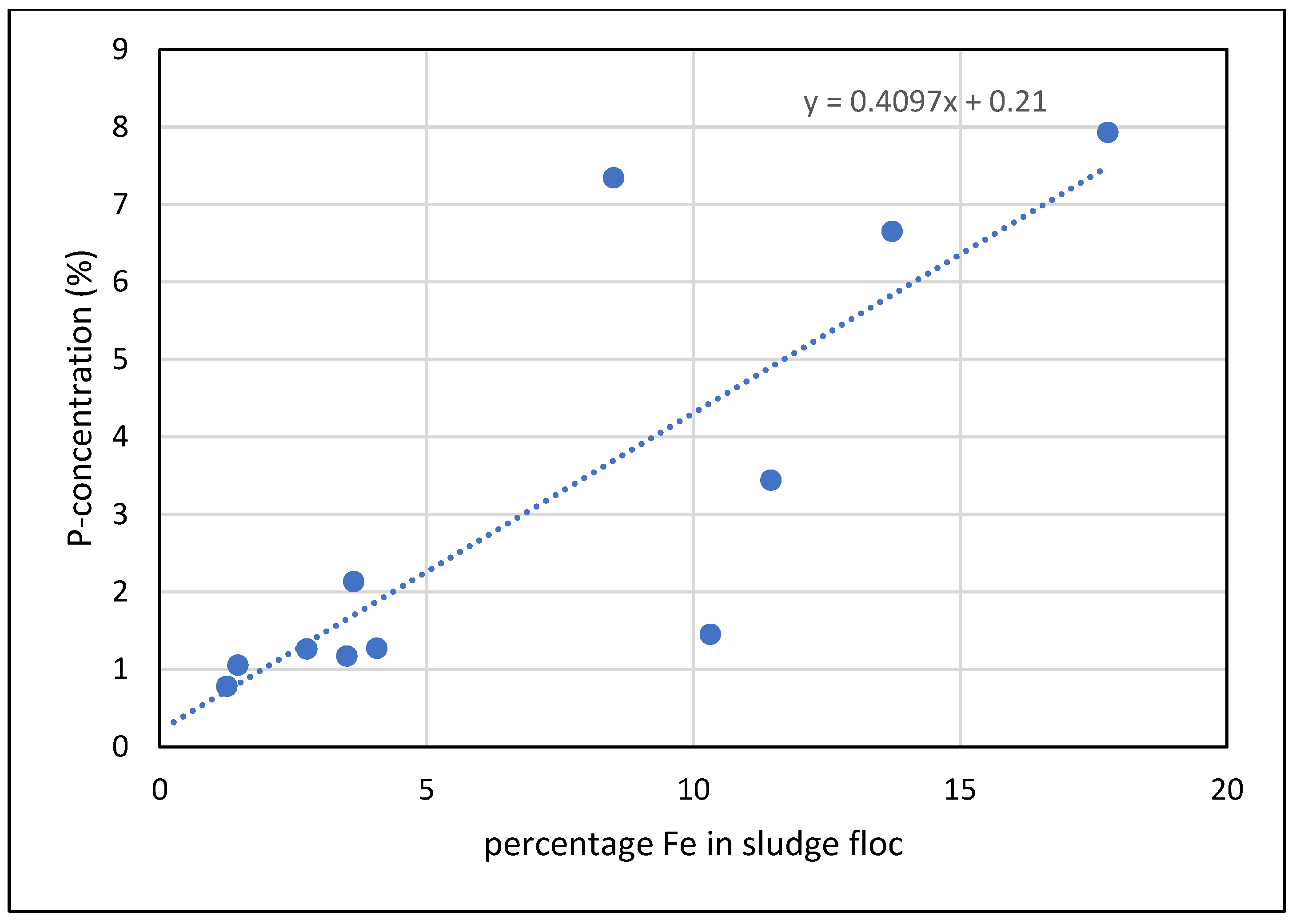
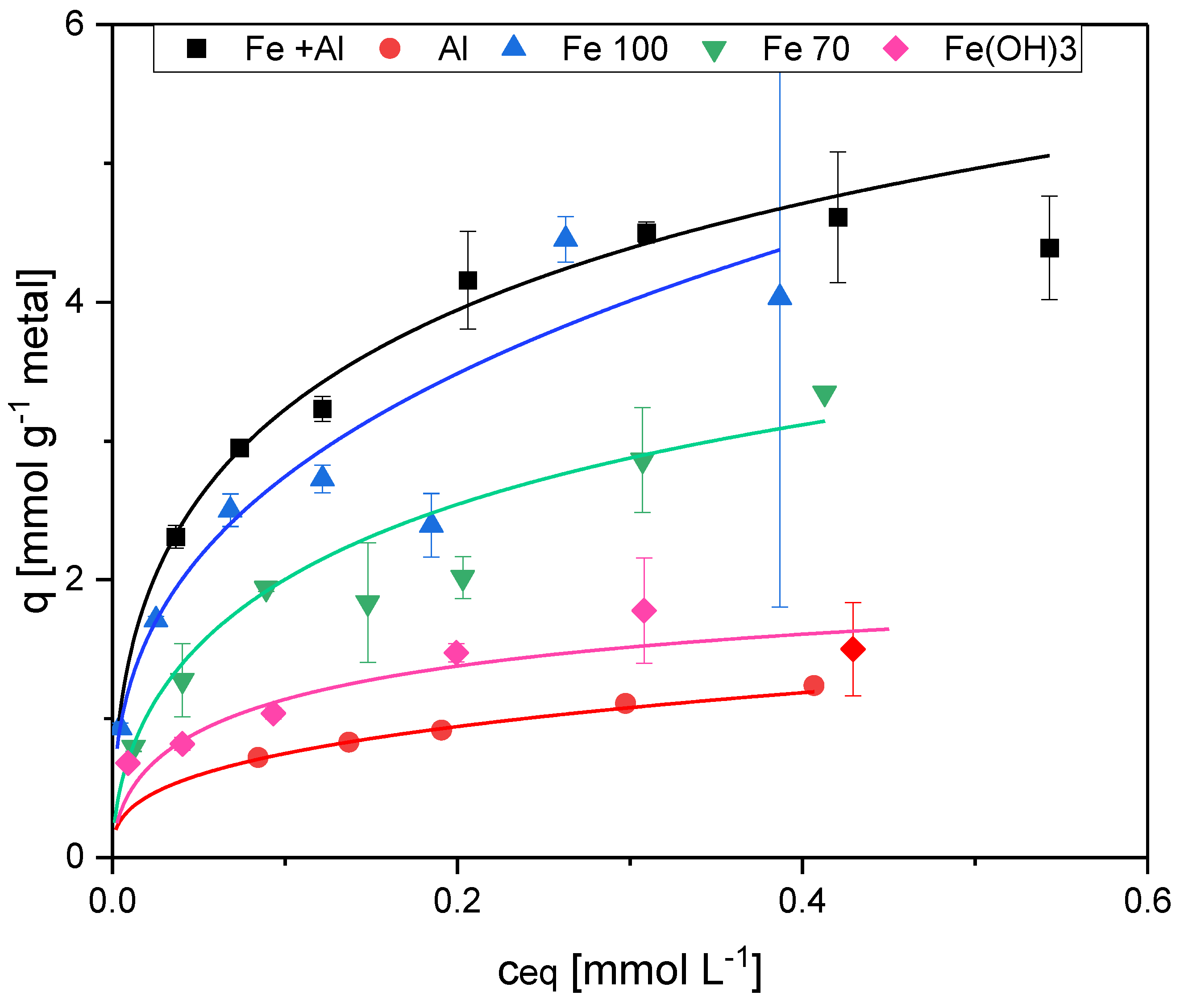
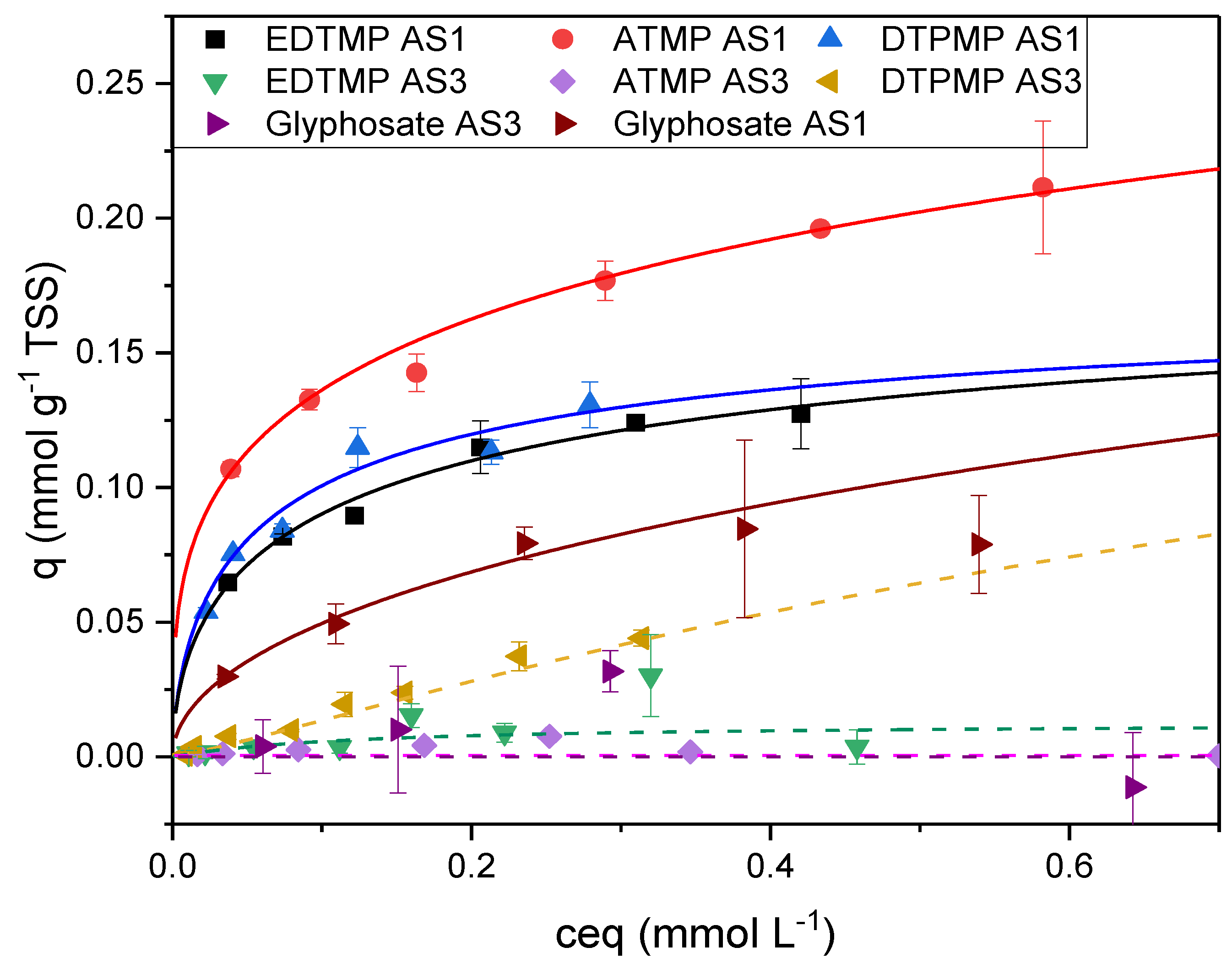
| Sorbent No. | Condition | TSS [g L-1] | Fe [mg L-1] | [Al mg L-1] |
| 1 | Fe+Al | 6.0 | 205.0 | 130.8 |
| 2 | Al | 7.0 | - | 284.4 |
| 3 | w/o | 3.9 | - | - |
| 4 | Fe100 | 4.0 | 103.3 | - |
| 5 | Fe70 | 3.2 | 69.9 | - |
| 6 | Fe pure | - | 104.7 | - |
| Langmuir | Freundlich | ||||||||||
| # | qmax | K | RSS | R2 | K | n | RSS | R2 | |||
| qmax | 1 | ATMP | 0.23 | 14.70 | 1.3E-03 | 0.908 | 0.23 | 0.22 | 3.7E-04 | 0.971 | |
| [mmol | EDTMP | 0.14 | 19.05 | 1.4E-04 | 0.957 | 0.28 | 0.28 | 9.5E-05 | 0.971 | ||
| gTSS-1] | DTPMP | 0.14 | 25.36 | 2.2E-04 | 0.948 | 0.31 | 0.31 | 2.8E-04 | 0.934 | ||
| 2 | ATMP | 0.13 | 736.53 | 7.4E-04 | 0.688 | 0.10 | 0.10 | 5.3E-04 | 0.777 | ||
| EDTMP | 0.06 | 9.22 | 1.2E-05 | 0.959 | 0.36 | 0.36 | 1.5E-06 | 0.995 | |||
| DTPMP | 0.10 | 15.04 | 1.6E-04 | 0.896 | 0.25 | 0.25 | 8.0E-06 | 0.0995 | |||
| 4 | EDTMP | 0.10 | 20.77 | 1.2E-03 | 0.813 | 0.26 | 0.26 | 9.2E-04 | 0.831 | ||
| 5 | EDTMP | 0.05 | 42.48 | 2.6E-05 | 0.942 | 0.29 | 0.29 | 5.1E-05 | 0.886 | ||
| 6 | EDTMP | 0.05 | 25.53 | 1.3E-04 | 0.808 | 0.32 | 0.32 | 2.6E-05 | 0.952 | ||
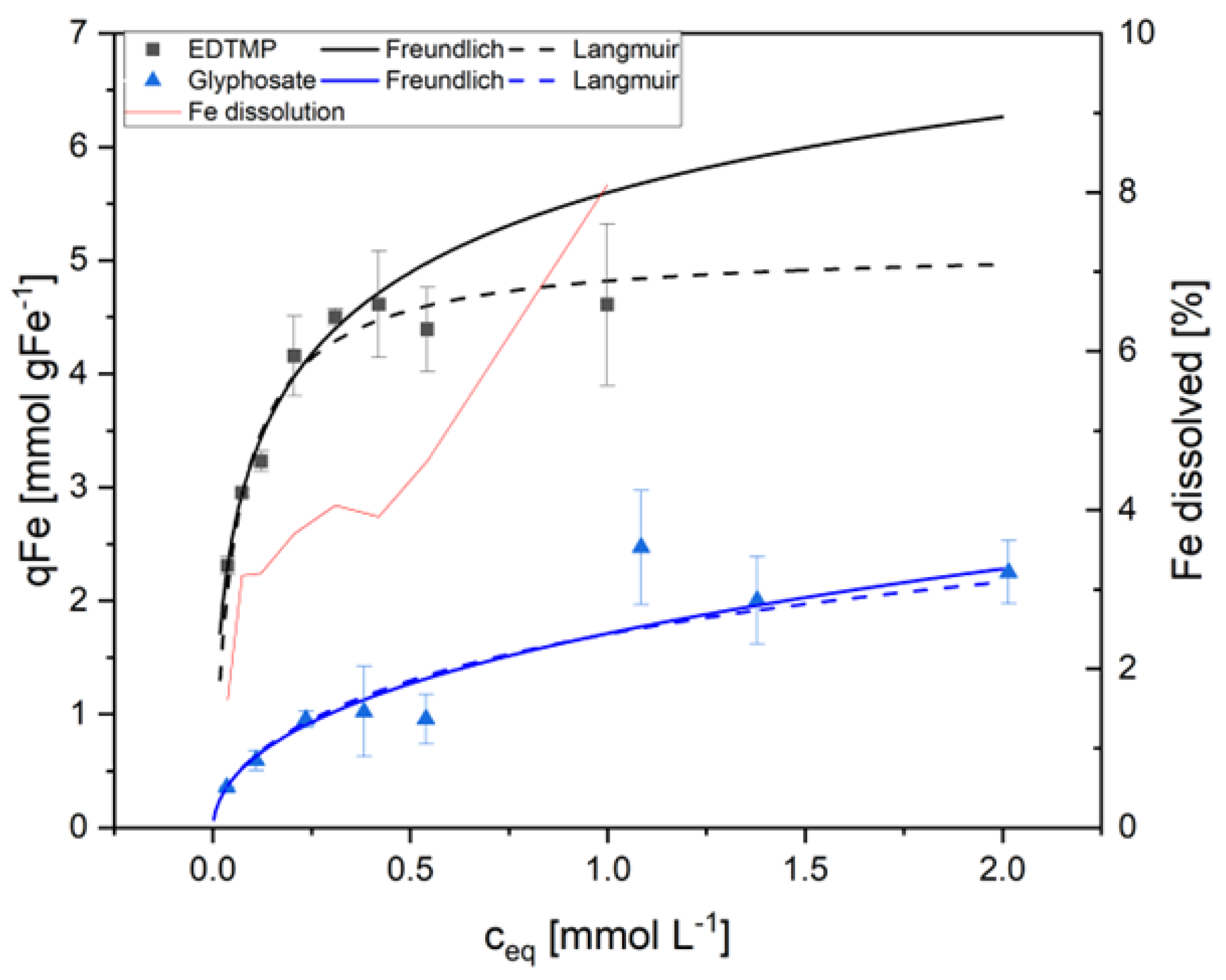
| qmax | 1 | ATMP | 5.94 | 13.18 | 9.4E-01 | 0.905 | 0.24 | 0.24 | 1.9E-01 | 0.979 | |
| [mmol | EDTMP | 4.94 | 21.23 | 2.6E-01 | 0.953 | 0.19 | 0.19 | 8.8E-01 | 0.841 | ||
| gFe-1] | DTPMP | 4.74 | 24.08 | 2.4E-01 | 0.950 | 0.32 | 0.32 | 3.0E-01 | 0.937 | ||
| 4 | EDTMP | 5.13 | 11.56 | 2.6E-01 | 0.847 | 0.31 | 0.31 | 1.5E-01 | 0.893 | ||
| 5 | EDTMP | 4.08 | 8.04 | 9.4E-01 | 0.853 | 0.45 | 0.45 | 4.0E-01 | 0.926 | ||
| 6 | EDTMP | 1.70 | 30.04 | 2.3E-01 | 0.798 | 0.26 | 0.26 | 1.2E-01 | 0.874 | ||
| qmax | 1 | ATMP | 0.33 | 13.18 | 2.9E-03 | 0.905 | 0.24 | 0.24 | 5.9E-04 | 0.979 | |
| [mmolFe-1] | EDTMP | 0.28 | 21.23 | 8.2E-04 | 0.953 | 0.19 | 0.19 | 2.8E-03 | 0.841 | ||
| DTPMP | 0.26 | 24.08 | 7.3E-04 | 0.950 | 0.32 | 0.32 | 9.3E-04 | 0.937 | |||
| 4 | EDTMP | 0.29 | 11.56 | 8.2E-03 | 0.847 | 0.31 | 0.31 | 4.6E-03 | 0.893 | ||
| 5 | EDTMP | 0.23 | 8.04 | 2.9E-03 | 0.853 | 0.45 | 0.45 | 1.2E-03 | 0.926 | ||
| 6 | EDTMP | 0.10 | 30.04 | 7.1E-04 | 0.798 | 0.26 | 0.26 | 3.7E-04 | 0.874 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




