Submitted:
25 November 2025
Posted:
27 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
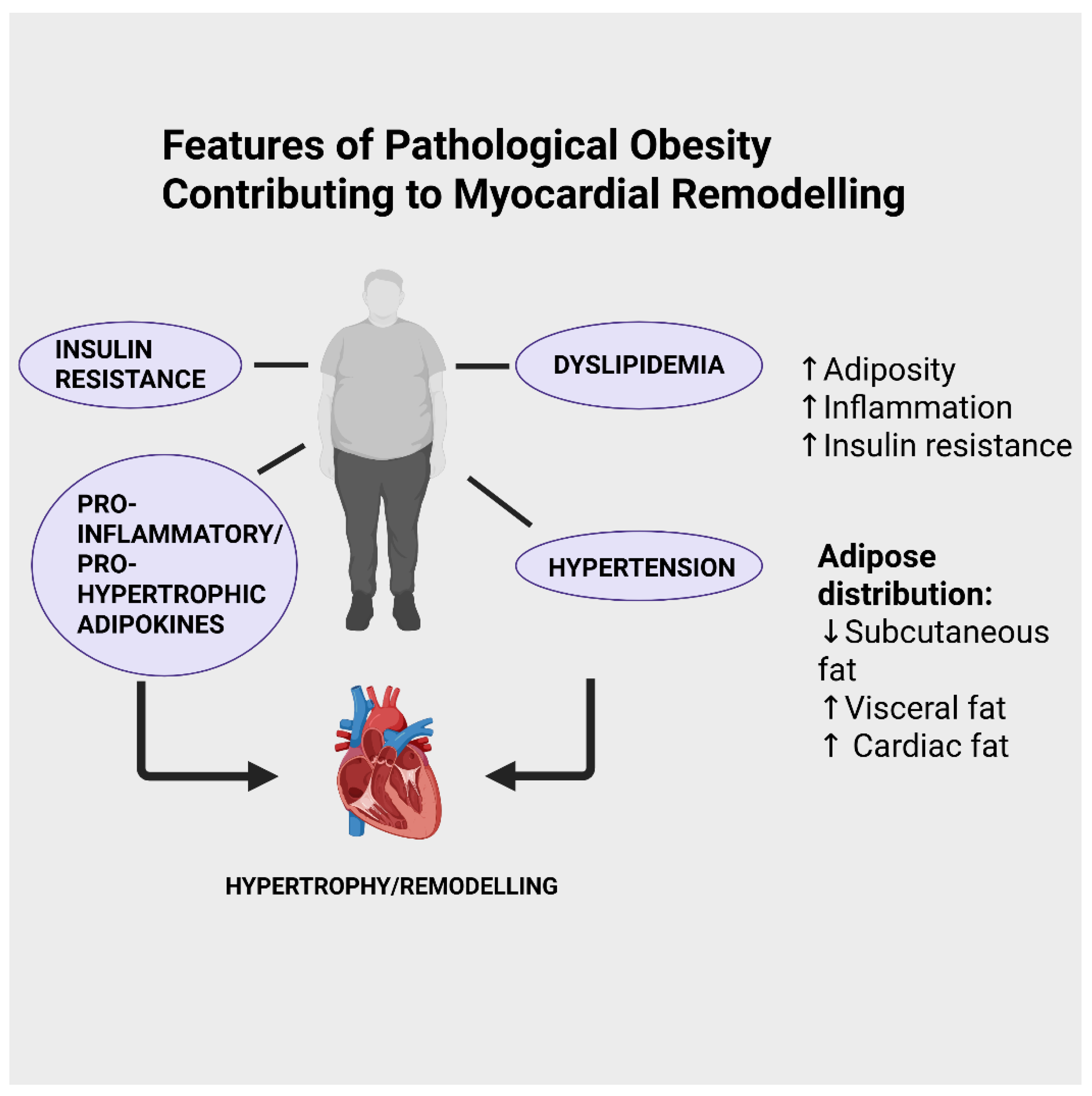
2. Obesity and Heart Disease with Particular Emphasis on Myocardial Remodelling and Heart Failure
3. The “Obesity Paradox”: A Role for Adipokines?
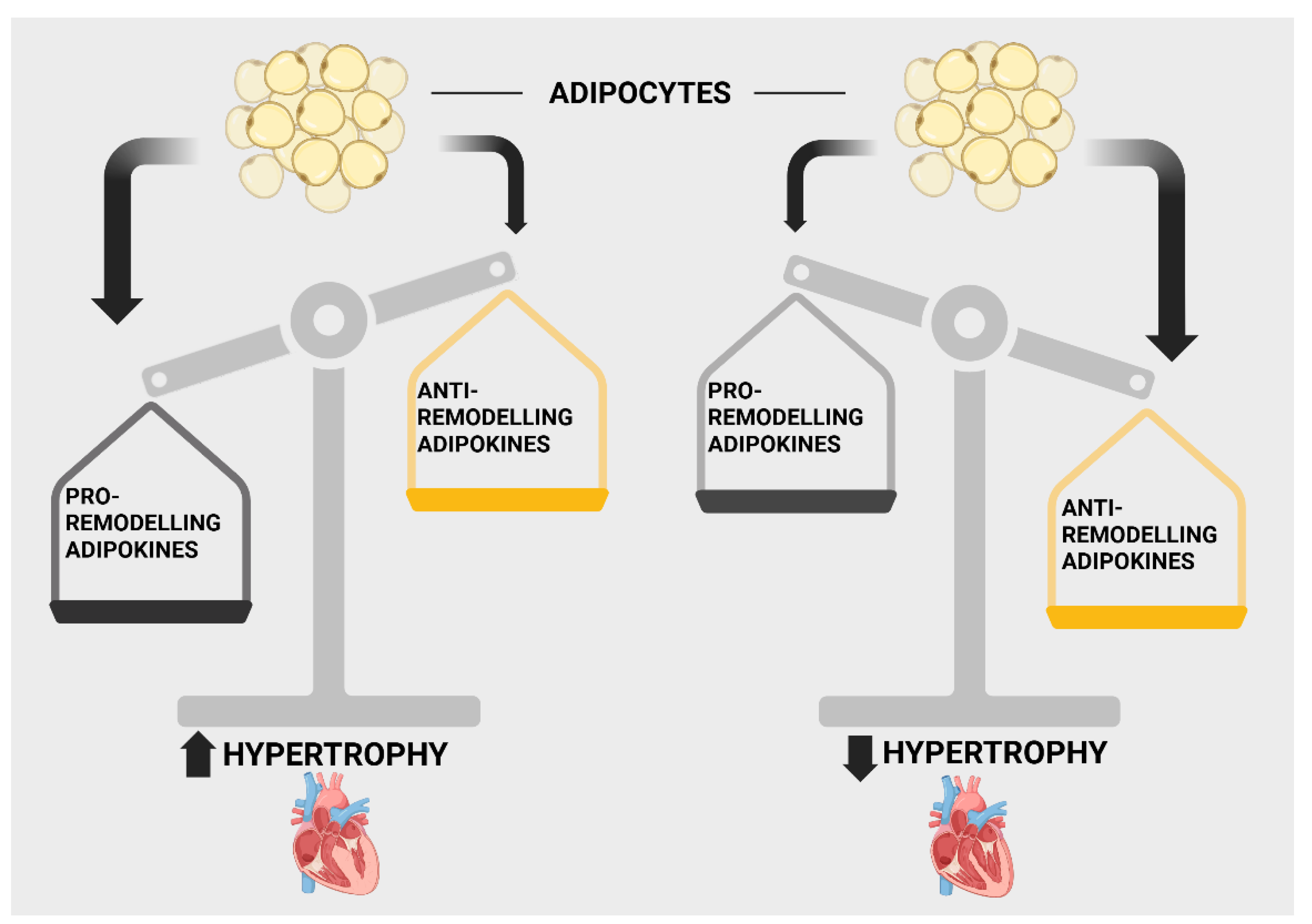
4. Adipocytes as Endocrine Organs
5. Obesity, Myocardial Remodelling and Heart Failure
6. Leptin and Adiponectin and the Critical Importance of the Leptin/Adiponectin Balance
7. Omentin
8. Resistin
9. Visfatin
10. Apelin
11. Vaspin
12. Adipsin
13. Asprosin
14. Chemerin
15. Meteorin-like Protein
16. Progranulin
17. Neuroregulin 4
18. Retinol Protein Binding 4
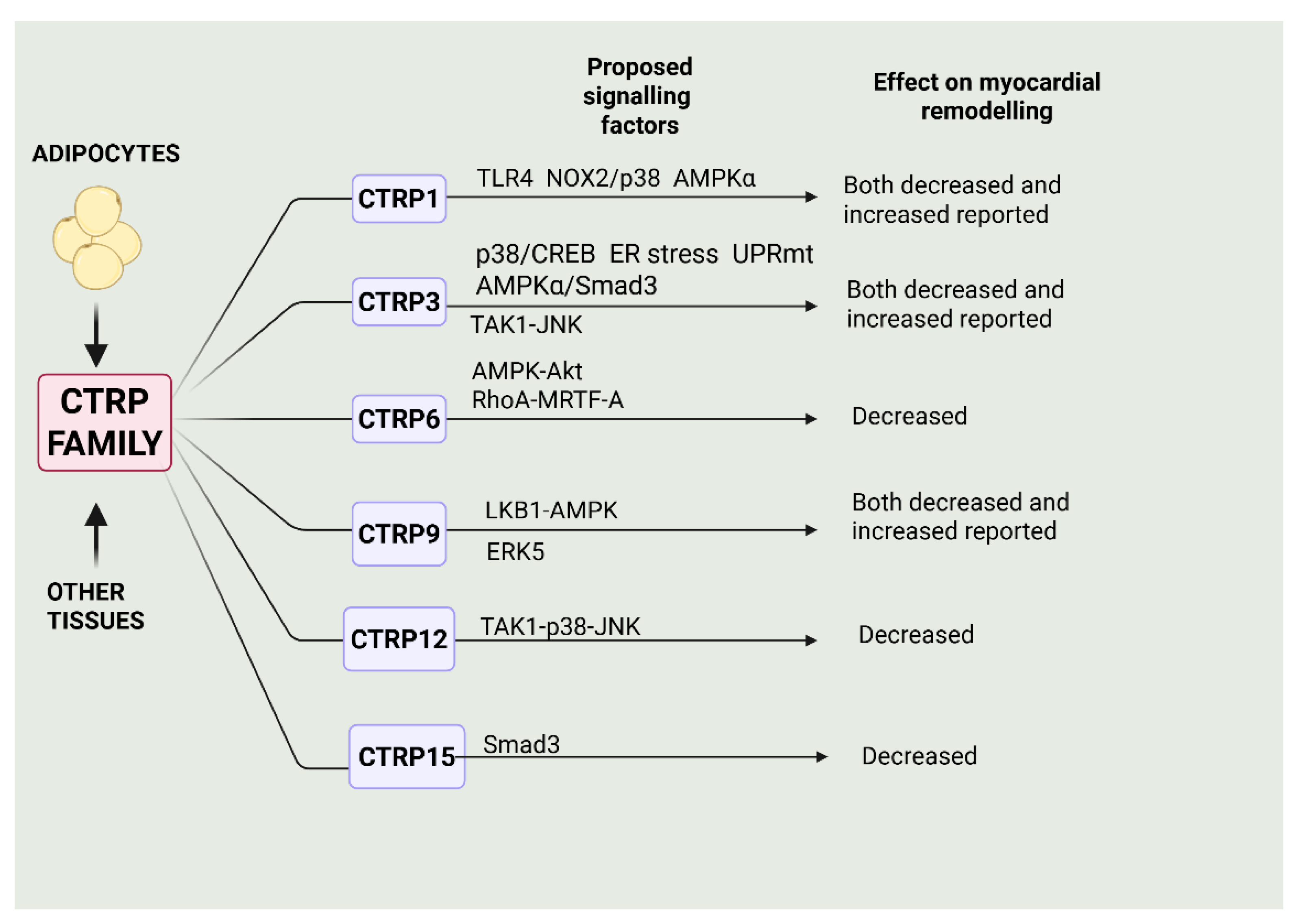
19. CTRP Family
19.1. CTRP1
19.2. CTRP3
19.3. CTRP6
19.4. CTRP9
19.5. CTRP12
19.6. CTRP15
20. Summary, Overall Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| WHO | World health organization |
| HFpEF | Heart failure with preserved ejection fraction |
| BMI | Body mass index |
| WATs | White adipose tissues |
| BATs | Brown adipose tissues |
| ObR (or LepR) | Leptin receptor |
| JAK2-STAT3 | Janus Kinase 3-Signal Transducer and Activator of Transcription 3 |
| NRCM | neonatal rat cardiac myocytes |
| p38 MAPK | p38 mitogen activated protein kinase |
| PHVM | pediatric human ventricular myocytes |
| ET-1 | endothelin 1 |
| ROS | reactive oxygen species |
| mTOR | mammalian target of rapamycin |
| RhoA/ROCK | Ras homolog gene family, member A/Rho-associated protein kinase |
| PPARα | peroxisome proliferator-activated receptor α |
| NFAT | nuclear factor of activated T cells |
| CUX1 | cut-like homeobox 1 |
| FTO | fat mass and obesity-associated protein |
| TGFβ | transforming growth factor β |
| AdipoR | adiponectin receptor |
| ERK | extracellular signal-regulated kinase |
| AMPK | AMP activated protein kinase |
| TAC | thoracic aorta constriction |
| STZ | streptozotocin |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| Brg1 | Brahma related gene 1 |
| DMD | Duchenne muscular dystrophy |
| HO-1 | heme oxygenase 1 |
| CAMKK2 | Calcium/calmodulin-dependent protein kinase 2 |
| PGC-1α | Peroxisome proliferator-activated receptor-gamma coactivator 1-alpha |
| Cx43 | connexin 43 |
| Ang II | angiotensin 2 |
| MiR-133a | microRNA 133a |
| UNX | uni-nephrectomized |
| NRAM | neonatal rat atrial myocytes |
| Mef2 | myocyte enhancer factor-2 |
| TLR4 | toll-like receptor 4 |
| MyD88 | myeloid differentiation factor 88 |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| miRNA | micro RNA |
| LKB1 | liver kinase B1 (also referred to as serine/threonine-protein kinase 11) |
| P70S6K | Ribosomal protein S6 kinase beta |
| NAMPT | nicotinamide phosphoribosyltransferase |
| REV-ERB | nuclear receptor subfamily 1 group D |
| PI3K | phosphatidylinositol 3-kinase |
| JNK | c-Jun N-terminal kinase |
| TRPC1 | transient receptor potential canonical channel 1 |
| APJ | apelin receptor |
| AT1 | angiotensin 2 type 1 receptor |
| ACE | angiotensin converting enzyme |
| RAS | renin angiotensin system |
| SGLT2 | sodium-glucose cotransporter-2 |
| PEG | polyethylene glycol |
| AKT | protein kinase B |
| cAMP | cyclic adenosine monophosphate |
| PKA | protein kinase A |
| CCRL2 | C-C chemokine receptor-like 2 |
| Metrnl | Meteorin-like protein |
| KIT | KIT Proto-Oncogene, Receptor Tyrosine Kinase |
| SIRT1 | sirtuin type 1 |
| ULK1 | Unc-51 Like Autophagy Activating Kinase 1 |
| BRCA2 | BRCA2 DNA repair associated |
| TGF-β | transforming growth factor beta |
| IL | interleukin |
| Wnt | Wingless-related integration site |
| Nrg4 | Neuregulin 4 |
| Rbp4 | Retinol protein binding 4 |
| CTRP | C1q/TNF-related proteins |
| UPRmt | mitochondrial unfolded protein |
| TAK1 | transforming growth factor-β activated kinase 1 |
| MRTF-A | myocardin-related transcription factor-A |
| IR | insulin receptor |
| IRS1 | insulin receptor substrate 1 |
References
- Kyle, T.K.; Dhurandhar, E.J.; Allison, D.B. Regarding obesity as a disease: Evolving policies and their implications. Endocrinol. Metab. Clin. North Am. 2016, 45, 511-520. [CrossRef]
- Lingvay, I.; Cohen, R.V.; Roux, C.W.L.; Sumithran, P. Obesity in adults. Lancet 2024, 404, 972-987. [CrossRef]
- Kenchaiah, S.; Evans, J.C.; Levy, D.; Wilson, P.W.; Benjamin, E,J.; Larson, M.G.; Kannel, W.B.; Vasan, R.S. Obesity and the risk of heart failure. N Engl J Med. 2002, 347, 305-13. [CrossRef]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; St-Onge, M.P.; American Heart Association Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Epidemiology and Prevention; and Stroke Council. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation 2021, 143, e984-e1010. [CrossRef]
- Koskinas, K.C.; Van Craenenbroeck, E.M.; Antoniades, C.; Blüher, M.; Gorter, T.M.; Hanssen, H.; Marx, N.; McDonagh, T.A.; Mingrone, G.; Rosengren, A.; Prescott, E.B. Obesity and cardiovascular disease: an ESC clinical consensus statement. Eur. J. Prev. Cardiol. 2025, 32,:184-220. [CrossRef]
- Mahabadi, A.A.; Massaro, J.M.; Rosito, G.A.; Levy, D.; Murabito, J.M.; Wolf, P.A.; O’Donnell, C.J.; Fox C,S.; Hoffmann, U. Association of pericardial fat, intrathoracic fat, and visceral abdominal fat with cardiovascular disease burden: the Framingham Heart Study. Eur. Heart J. 2009, 30, 850-856. [CrossRef]
- Upadhaya, S.; Le Jemtel, T.H. Epicardial Adipose Tissue and Heart Failure. Trends Cardiovasc. Med. 2025,35,339-340. [CrossRef]
- Janssen-Telders, C.; Eringa, E.C.; de Groot, J.R.; de Man, F.S.; Handoko, M. The role of epicardial adipose tissue remodelling in heart failure with preserved ejection fraction. Cardiovasc. Res. 2025, 121, 860-870. [CrossRef]
- Whitman, J.; Kozaily, E.; Michos, E.D.; Silverman, D.N.; Fudim, M.; Mentz, R.J.; Tedford, R.J.; Rao V.N. Epicardial fat in heart failure and preserved ejection fraction: Novel insights and future perspectives. Curr. Heart Fail. Rep. 2025, 22,13. [CrossRef]
- Ebong, I.A.; Goff, D.C. Jr.; Rodriguez, C.J.; Chen, H.; Bertoni, A.G. Mechanisms of heart failure in obesity. Obes. Res. Clin. Pract. 2014, 8, e540-8. [CrossRef]
- Alansari, H.; Lazzara, G.; Taha, M.B.; Gorthi, J.R. The impact of obesity on cardiovascular diseases: Heart failure. Methodist Debakey Cardiovasc. J. 2025, 21, 44-52. [CrossRef]
- Piché, M.E.; Tchernof, A.; Després, J.P. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ. Res. 2020, 126, 1477-1500. [CrossRef]
- Cesaro, A.; De Michele, G.; Fimiani, F.; Acerbo, V.; Scherillo, G.; Signore, G.; Rotolo, F.P.; Scialla, F.; Raucci, G.; Panico, D.; Gragnano, F.; Moscarella, E.; Scudiero, O.; Mennitti, C.; Calabrò, P. Visceral adipose tissue and residual cardiovascular risk: a pathological link and new therapeutic options. Front. Cardiovasc. Med. 2023, 10, 1187735. [CrossRef]
- Amundson, D.E.; Djurkovic, S.; Matwiyoff, G.N. The obesity paradox. Crit. Care Clin. 2010, 26, 583-596. [CrossRef]
- Dramé, M.; Godaert, L. The obesity paradox and mortality in older adults: A systematic review. Nutrients 2023, 15,1780. [CrossRef]
- Horwich, T.B.; Fonarow, G.C.; Clark, A.L. Obesity and the obesity paradox in heart failure. Prog. Cardiovasc. Dis. 2018, 61,151-156. [CrossRef]
- Antonopoulos, A.S.; Tousoulis, D. The molecular mechanisms of obesity paradox. Cardiovasc. Res. 2017, 113, 1074-1086. [CrossRef]
- Pan, J.; Yin, J.; Gan, L.; Xue, J. Two-sided roles of adipose tissue: Rethinking the obesity paradox in various human diseases from a new perspective. Obes. Rev. 2023, 24, e13521. [CrossRef]
- Butt, J.H.; Thune, J.J.; Nielsen, J.C.; Haarbo, J.; Videbæk, L.; Gustafsson, F.; Kristensen, S.L.; Bruun, N.E.; Eiskjær, H.; Brandes, A.; Hassager, C.; Svendsen, J.H.; Høfsten, D.E.; Torp-Pedersen, C.; Schou, M.; Pehrson, S.; Packer, M.; McMurray, J.J.V.; Køber, L. Anthropometric measures and long-term mortality in non-ischaemic heart failure with reduced ejection fraction: Questioning the obesity paradox. Eur. J. Heart Fail. 2025, 27, 527-536. [CrossRef]
- Wertheimer, E.; Shapiro, B. The physiology of adipose tissue. Physiol. Rev. 1948, 28, 451-464. [CrossRef]
- Kershaw, E.E.; Flier, J,S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548-2556. [CrossRef]
- Coelho, M.; Oliveira, T.; Fernandes, R. Biochemistry of adipose tissue: an endocrine organ. Arch. Med. Sci. 2013, 9, 191-200. [CrossRef]
- Scheja, L,; Heeren J. The endocrine function of adipose tissues in health and cardiometabolic disease. Nat. Rev. Endocrinol. 2019, 15, 507-524. [CrossRef]
- Chaldakov, G.N.; Stankulov, I.S.; Hristova, M.; Ghenev, P. Adipobiology of disease: adipokines and adipokine-targeted pharmacology. Curr. Pharm. Des. 2003, 9. [CrossRef]
- Yang, F.T.; Stanford, K.I. Batokines: Mediators of Inter-Tissue Communication (a Mini-Review). Curr. Obes. Rep. 2022, 11,1-9. [CrossRef]
- Bays, H.E.; Kirkpatrick, C.F.; Maki, K.C.; Toth, P.P.; Morgan, R.T.; Tondt, J.; Christensen, S.M.; Dixon, D.L.; Jacobson, T.A. Obesity, dyslipidemia, and cardiovascular disease: A joint expert review from the Obesity Medicine Association and the National Lipid Association 2024. J. Clin. Lipidol. 2024, 18, e320-e350. [CrossRef]
- Sharma, K.; Kass, D.A. Heart failure with preserved ejection fraction: mechanisms, clinical features, and therapies. Circ. Res. 2014, 115, 79-96. [CrossRef]
- Rao, V.N.; Zhao, D.; Allison, M.A.; Guallar, E.; Sharma, K.; Criqui, M.H.; Cushman, M.; Blumenthal, R.S.; Michos, E.D. Adiposity and Incident Heart Failure and its Subtypes: MESA (Multi-Ethnic Study of Atherosclerosis). JACC Heart Fail. 2018, 6, 999-1007. [CrossRef]
- Neeland, I.J.; Gupta, S.; Ayers, C.R.; Turer, A.T.; Rame, J.E.; Das, S.R.; Berry, JD.; Khera, A.; McGuire, D.K.; Vega, G.L.; Grundy, S.M.; de Lemos, J.A.; Drazner, M.H. Relation of regional fat distribution to left ventricular structure and function. Circ. Cardiovasc. Imaging. 2013, 6, 800-807. [CrossRef]
- Raggi, P.; Stillman, A.E. Clinical Role of Epicardial Adipose Tissue. Can. J. Cardiol. 2025, Feb 17, S0828-282X(25)00131-X. [CrossRef]
- Vincenzi, M.; Nebigil, C.G. Uncovering the role of prokineticin pathway on Epicardial Adipose Tissue (EAT) development and EAT-associated cardiomyopathy. Trends Cardiovasc. Med. 2025, 35, 328-338. [CrossRef]
- Ketema, E.; Lopaschuk, G.D. The Impact of obesity on cardiac energy metabolism and efficiency in heart failure with preserved ejection fraction. Can. J. Cardiol. 2025, Jan 30: S0828-282X(25)00099-6. [CrossRef]
- Borlaug, B.A.; Jensen, M.D.; Kitzman, D.W.; Lam, C.S.P.; Obokata, M.; Rider. O.J. Obesity and heart failure with preserved ejection fraction: new insights and pathophysiological targets. Cardiovasc. Res. 2023, 118, 3434-3450. [CrossRef]
- Rider, O.J.; Lewis, A.J.; Neubauer, S. Structural and metabolic effects of obesity on the myocardium and the aorta. Obes. Facts. 2014, 7, 329-338. [CrossRef]
- Zhou, R.; Zhang, Z.; Li, X.; Duan, Q.; Miao, Y.; Zhang, T.; Wang, M.; Li, J.; Zhang, W.; Wang, L.; Jones, O.D.; Xu, M.; Liu, Y.; Xu, X. Autophagy in high-fat diet and streptozotocin-induced metabolic cardiomyopathy: mechanisms and therapeutic implications. Int. J. Mol. Sci. 2025, 26, 1668. [CrossRef]
- Ren, J.; Wu, N.N.; Wang, S.; Sowers, J,R.; Zhang, Y. Obesity cardiomyopathy: evidence, mechanisms, and therapeutic implications. Physiol. Rev. 2021, 101, 1745-1807. [CrossRef]
- Alpert, M.A.; Karthikeyan, K.; Abdullah, O.; Ghadban, R. Obesity and cardiac remodeling in adults: Mechanisms and clinical implications. Prog. Cardiovasc. Dis. 2018, 61, 114-123. [CrossRef]
- Theodorakis, N.; Kreouzi, M.; Hitas, C.; Anagnostou, D.; Nikolaou, M. Adipokines and cardiometabolic heart Failure with preserved ejection fraction: A State-of-the-Art review. Diagnostics (Basel) 2024, 14, 2677. [CrossRef]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 372, 1994, 425–432, 1994. [CrossRef]
- Karmazyn, M.; Gan, X.T. Molecular and cellular mechanisms underlying the cardiac hypertrophic and pro-remodelling effects of leptin. Int. J. Mol. Sci. 2024, 25, 1137. [CrossRef]
- Theodorakis, N.; Nikolaou, M. Leptin and heart failure: the chicken or the egg? Heart Fail. Rev. 2025, 30, 749-757. [CrossRef]
- Leifheit-Nestler, M.; Wagner, N.M.; Gogiraju, R.; Didié, M, Konstantinides, S.; Hasenfuss, G.; Schäfer, K. Importance of leptin signaling and signal transducer and activator of transcription-3 activation in mediating the cardiac hypertrophy associated with obesity. J. Transl. Med. 2013, 11, 170. [CrossRef]
- Kang, K.W.; Ok, M.; Lee, S,K. Leptin as a key between obesity and cardiovascular disease. J. Obes. Metab. Syndr. 2020, 29, 248-259. [CrossRef]
- Allison, M.B.; Myers, M.G. Jr. 20 years of leptin: connecting leptin signaling to biological function. J. Endocrinol. 2014, 223, T25-35. [CrossRef]
- Liu, Z.; Xiao, T.; Liu H. Leptin signaling and its central role in energy homeostasis. Front. Neurosci. 2023,17, 1238528. [CrossRef]
- Jiang, L.; Li, Z.; Rui, L. Leptin stimulates both JAK2-dependent and JAK2-independent signaling pathways. J. Biol. Chem. 2008, 283, 28066-28073. [CrossRef]
- Rajapurohitam, V.; Gan, X.; Kirshenbaum, L.A.; Karmazyn, M. The obesity-associated peptide leptin induces hypertrophy in neonatal rat ventricular myocytes. Circ. Res. 2003, 93, 277-279. [CrossRef]
- Xu, F.P.; Chen, M.S.; Wang, Y,Z.; Yi, Q.; Lin, S.B.; Chen, A.F.; Luo, JD. Leptin induces hypertrophy via endothelin-1-reactive oxygen species pathway in cultured neonatal rat cardiomyocytes. Circulation 2004, 110, 1269-1275. [CrossRef]
- Madani, S.; De Girolamo, S.; Muñoz, D.M.; Li, R.K.; Sweeney, G. Direct effects of leptin on size and extracellular matrix components of human pediatric ventricular myocytes. Cardiovasc. Res. 2006, 69, 716-725. [CrossRef]
- Zeidan, A.; Javadov, S.; Karmazyn, M. Essential role of Rho/ROCK-dependent processes and actin dynamics in mediating leptin-induced hypertrophy in rat neonatal ventricular myocytes. Cardiovasc. Res. 2006, 72, 101-111. [CrossRef]
- Moey, M.; Rajapurohitam, V.; Zeidan, A.; Karmazyn, M. Ginseng (Panax quinquefolius) attenuates leptin-induced cardiac hypertrophy through inhibition of p115Rho guanine nucleotide exchange factor-RhoA/Rho-associated, coiled-coil containing protein kinase-dependent mitogen-activated protein kinase pathway activation. J. Pharmacol. Exp. Ther. 2011, 339, 746-756. [CrossRef]
- Zeidan, A.; Hunter, J.C.; Javadov, S.; Karmazyn, M. mTOR mediates RhoA-dependent leptin-induced cardiomyocyte hypertrophy. Mol. Cell. Biochem. 2011, 352, 99-108. [CrossRef]
- Abe, Y.; Ono, K.; Kawamura, T.; Wada, H.; Kita, T.; Shimatsu, A.; Hasegawa, K. Leptin induces elongation of cardiac myocytes and causes eccentric left ventricular dilatation with compensation. Am. J. Physiol. Heart Circ. Physiol. 2007, 292, H2387-H2396. [CrossRef]
- Zeidan, A.; Javadov, S.; Chakrabarti, S.; Karmazyn, M. Leptin-induced cardiomyocyte hypertrophy involves selective caveolae and RhoA/ROCK-dependent p38 MAPK translocation to nuclei. Cardiovasc. Res. 2008, 77,64-72. [CrossRef]
- Hou, N.; Luo, M.S.; Liu, S.M.; Zhang, H.N.; Xiao, Q.; Sun, P.; Zhang, G.S.; Luo, J.D.; Chen, M.S. Leptin induces hypertrophy through activating the peroxisome proliferator-activated receptor α pathway in cultured neonatal rat cardiomyocytes. Clin. Exp. Pharmacol. Physiol. 2010, 37, 1087-1095. [CrossRef]
- Rajapurohitam, V.; Izaddoustdar, F.; Martinez-Abundis, E.; Karmazyn. M. Leptin-induced cardiomyocyte hypertrophy reveals both calcium-dependent and calcium-independent/RhoA-dependent calcineurin activation and NFAT nuclear translocation. Cell. Signal. 2012, 24, 2283-2290. [CrossRef]
- Gan, X,T.; Zhao, G.; Huang, C.X.; Rowe, A.C.; Purdham, D.M.; Karmazyn, M. Identification of fat mass and obesity associated (FTO) protein expression in cardiomyocytes: regulation by leptin and its contribution to leptin-induced hypertrophy. PLoS One 2013, 8, e74235. [CrossRef]
- Kain, D.; Simon, A.J.; Greenberg, A.; Ben Zvi, D.; Gilburd, B.; Schneiderman, J. Cardiac leptin overexpression in the context of acute MI and reperfusion potentiates myocardial remodeling and left ventricular dysfunction. PLoS One 2018, 13, e0203902. [CrossRef]
- Scherer, P.E.; Williams, S.; Fogliano, M,; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q, produced exclusively in adipocytes. J. Biol. Chem. 1995, 270, 26746-26749. [CrossRef]
- Arita, Y.; Kihara, S.; Ouchi, N.; Takahashi, M.; Maeda K.; Miyagawa, J.; Hotta, K.;, Shimomura, I.; Nakamura, T.; Miyaoka, K.; Kuriyama, H.; Nishida, M.; Yamashita, S.; Okubo, K.; Matsubara, K.; Muraguchi, M.; Ohmoto, Y.; Funahashi, T.; Matsuzawa, Y. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem. Biophys. Res. Commun. 1999, 257, 79-83. [CrossRef]
- Maeda, N.; Funahashi, T.; Matsuzawa, Y.; Shimomura, I. Adiponectin, a unique adipocyte-derived factor beyond hormones. Atherosclerosis 292, 2020, 292, 1-9. [CrossRef]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [CrossRef]
- Ding, G.; Qin, Q.; He, N.; Francis-David, S.C.; Hou, J.; Liu, J.; Ricks, E.; Yang, Q. Adiponectin and its receptors are expressed in adult ventricular cardiomyocytes and upregulated by activation of peroxisome proliferator-activated receptor gamma. J. Mol. Cell. Cardiol. 2007, 43, 73-84. [CrossRef]
- Shibata, R.; Ouchi, N.; Ito, M.; Kihara, S.; Shiojima, I.; Pimentel, D.R.; Kumada, M.; Sato, K.; Schiekofer, S.; Ohashi, K.; Funahashi, T.; Colucci, W.S.; Walsh, K. Adiponectin-mediated modulation of hypertrophic signals in the heart. Nat. Med. 2004, 10, 1384-1389. [CrossRef]
- Fujioka, D.; Kawabata, K.; Saito, Y.; Kobayashi, T.; Nakamura, T.; Kodama, Y.; Takano, H.; Obata, J.E.; Kitta, Y.; Umetani, K.; Kugiyama, K. Role of adiponectin receptors in endothelin-induced cellular hypertrophy in cultured cardiomyocytes and their expression in infarcted heart. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H2409-H2416. [CrossRef]
- Dhandapany, P.S.; Kang, S.; Kashyap, D.; Rajagopal, R.; Sundaresan, N.;, Singh, R.; Thangaraj, K.; Jayaprakash, S.; Manjunath, C.N.; Shenthar, J.; Lebeche, D. Adiponectin receptor 1 variants contribute to hypertrophic cardiomyopathy that can be reversed by rapamycin. Sci. Adv. 2021, 7, eabb3991. [CrossRef]
- Li, H.; Yao, W.; Irwin, M.G.; Wang, T.; Wang, S.; Zhang, L.; Xia, Z. Adiponectin ameliorates hyperglycemia-induced cardiac hypertrophy and dysfunction by concomitantly activating Nrf2 and Brg1. Free Radic. Biol. Med. 2015, 84, 311-321. [CrossRef]
- Abou-Samra, M.; Dubuisson, N.; Marino, A.; Selvais, C.M.; Romain, V.; Davis-López de Carrizosa, M.A.; Noel, L.; Beauloye, C.; Brichard, S.M.; Horman, S. Striking cardioprotective effects of an adiponectin receptor agonist in an aged mouse model of Duchenne muscular dystrophy. Antioxidants (Basel) 2024, 13, 1551. [CrossRef]
- Cho, S.; Dadson, K.; Sung, H.K.; Ayansola, O.; Mirzaesmaeili, A.; Noskovicova, N.; Zhao, Y.; Cheung, K.; Radisic, M.; Hinz, B.; Sater, A.A.A.; Hsu, H.H.; Lopaschuk, G.D.; Sweeney, G. Cardioprotection by the adiponectin receptor agonist ALY688 in a preclinical mouse model of heart failure with reduced ejection fraction (HFrEF). Biomed. Pharmacother. 2024, 171, 16119. [CrossRef]
- Leffler, K.E.; Abdel-Rahman, A.A. Restoration of adiponectin-connexin43 signaling mitigates myocardial inflammation and dysfunction in diabetic female rats. J. Cardiovasc. Pharmacol. 2020, 75, 259-267. [CrossRef]
- Han, X.; Wang, Y.; Fu, M.; Song, Y.; Wang, J.; Cui, X.; Fan, Y.; Cao, J.; Luo, J.; Sun, A.; Zou, Y.; Hu, K.; Zhou, J.; Ge, J. Effects of adiponectin on diastolic function in mice underwent transverse aorta constriction. J. Cardiovasc. Transl. Res. 2020, 13, 225-237. [CrossRef]
- Zhang, N.; Wei, W.Y.; Liao, H.H.; Yang, Z.; Hu, C.; Wang, S.S.; Deng, W.; Tang, Q.Z. AdipoRon, an adiponectin receptor agonist, attenuates cardiac remodeling induced by pressure overload. J. Mol. Med. (Berl). 2018, 96, 1345-1357. [CrossRef]
- Li, Y.; Cai. X.;, Guan, Y.; Wang, L.; Wang, S.; Li, Y.;, Fu, Y.; Gao, X.; Su, G. Adiponectin upregulates MiR-133a in cardiac hypertrophy through AMPK activation and reduced ERK1/2 phosphorylation. PLoS One 2016, 11,e0148482. [CrossRef]
- Tanaka, K.; Wilson, R.M.; Essick, E.E.; Duffen, J.L.; Scherer, P.E.; Ouchi, N.; Sam, F. Effects of adiponectin on calcium-handling proteins in heart failure with preserved ejection fraction. Circ. Heart Fail. 2014, 7, 976-985. [CrossRef]
- Cao, T.; Gao, Z.; Gu, L.; Chen, M.; Yang, B.; Cao, K.; Huang, H.; Li M. AdipoR1/APPL1 potentiates the protective effects of globular adiponectin on angiotensin II-induced cardiac hypertrophy and fibrosis in neonatal rat atrial myocytes and fibroblasts. PLoS One 2014, 9, e103793. [CrossRef]
- Fujishima, Y.; Maeda, N.; Matsuda, K.; Komura, N.; Hirata, A.; Mori, T.; Sekimoto, R.; Tsushima, Y.; Nishizawa, H.; Funahashi, T.; Shimomura, I. Effect of adiponectin on cardiac beta-catenin signaling pathway under angiotensin II infusion. Biochem. Biophys. Res. Commun. 2014, 444, 224-229. [CrossRef]
- Dadson, K.; Turdi, S.; Hashemi. S.; Zhao, J.; Polidovitch, N.; Beca, S.; Backx, P.H.; McDermott, J.C.; Sweeney, G. Adiponectin is required for cardiac MEF2 activation during pressure overload induced hypertrophy. J. Mol. Cell. Cardiol. 2015, 86, 10102-9. [CrossRef]
- Liao, Y.; Takashima, S.; Maeda, N.; Ouchi, N.; Komamura, K.; Shimomura, I.; Hori, M.; Matsuzawa, Y.; Funahashi, T.; Kitakaze, M. Exacerbation of heart failure in adiponectin-deficient mice due to impaired regulation of AMPK and glucose metabolism. Cardiovasc. Res. 2005, 67, 705-713. [CrossRef]
- Yang, R.Z.; Lee, M.J.; Hu, H.; Pray, J.; Wu, H.B.; Hansen, B.C.; Shuldiner, A.R.; Fried, S.K.; McLenithan, J.C.; Gong, D.W. Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E1253-E1261. [CrossRef]
- Biegański, H.M.; Dąbrowski, K.;, Różańska-Walędziak, A. Omentin-general overview of Its role in obesity, metabolic syndrome and other diseases; Problem of current research state. Biomedicines 2025, 13, 632. [CrossRef]
- Sena, C.M. Omentin: A key player in glucose homeostasis, atheroprotection, and anti-inflammatory potential for cardiovascular health in obesity and diabetes. Biomedicines 2024, 12, 284. [CrossRef]
- Brunetti, L.; Leone, S.; Orlando, G.; Ferrante, C.; Recinella, L.; Chiavaroli, A.; Di Nisio, C.; Shohreh, R.; Manippa, F.; Ricciuti.;A, Vacca, M. Hypotensive effects of omentin-1 related to increased adiponectin and decreased interleukin-6 in intra-thoracic pericardial adipose tissue. Pharmacol. Rep. 2014, 66, 991-995. [CrossRef]
- Bilovol, O.M.;, Knyazkova, I.I.; Al-Travneh, O.V.; Bogun, M.V.; Berezin, A.E. Altered adipocytokine profile predicts early stage of left ventricular remodeling in hypertensive patients with type 2 diabetes mellitus. Diabetes Metab. Syndr. 2020, 14, 109-116. [CrossRef]
- Matsuo, K.; Shibata, R.; Ohashi, K.; Kambara, T.; Uemura, Y.; Hiramatsu-Ito, M.; Enomoto, T.; Yuasa, D.; Joki, Y.; Ito, M.; Hayakawa, S.; Ogawa, H.; Kihara, S.; Murohara, T.; Ouchi, N. Omentin functions to attenuate cardiac hypertrophic response. J. Mol. Cell. Cardiol. 2015, 79, 195-202. [CrossRef]
- Ito, M.; Shibata, R.; Ohashi, K.; Otaka, N.; Yamaguchi, S.; Ogawa, H.; Enomoto, T.; Masutomi, T.; Murohara, T.; Ouchi, N. Omentin modulates chronic cardiac remodeling after myocardial infarction. Circ. Rep. 2023, 5, 46-54. [CrossRef]
- Yan, X.; Wu, L.; Gao, M.; Yang, P.; Yang, J.; Deng, Y. Omentin inhibits the resistin-induced hypertrophy of H9c2 cardiomyoblasts by inhibiting the TLR4/MyD88/NF-kappaB signaling pathway. Exp. Ther. Med. 2022, 23, 292. [CrossRef]
- Su, Z.; Tian, S.; Liang, W.; Wu, L. Association between omentin-1 and heart failure with preserved ejection fraction in Chinese elderly patients. Clin. Cardiol. 2023, 47, e24181. [CrossRef]
- Huang, Y.;, Lin, Y.; Zhang, S.; Wang, Z.; Zhang, J.; Chang, C.; Liu, L.; Ji, Q.; Liu, X. Circulating omentin-1 levels are decreased in dilated cardiomyopathy patients with overt heart failure. Dis. Markers. 2016, 2016:6762825. [CrossRef]
- Jiang, H.; Zhao, G.; Li, X.; Jin, H.; Yang, G.; Jin, K.; Piao, L.; Zhu, E.; Lei, Y.; Fang, E.; Han, X.; Nan, Y.; Jin, Q.; Cheng, X. Association between omentin and echo parameters in patients with chronic heart failure. Minerva Cardioangiol. 2017, 65, 8-15. [CrossRef]
- Steppan, C.M.; Brown, E.J.; Wright, C.M.; Bhat, S.; Banerjee, R.R.; Dai, C.Y.; Enders, G.H.; Silberg, D.G.; Wen, X.; Wu, G.D.; Lazar, M.A. A family of tissue-specific resistin-like molecules. Proc. Natl. Acad. Sci. USA 2001, 98, 502-506. [CrossRef]
- Nagaev, I.; Bokarewa, M.; Tarkowski, A.; Smith, U. Human resistin is a systemic immune-derived proinflammatory cytokine targeting both leukocytes and adipocytes. PLoS One 2006, 1, e31. [CrossRef]
- Dhurandhar, Y.; Tomar, S.; Das, A.; Prajapati, J.L.; Singh, A.P.; Bodake, S.H.; Namdeo, K.P. Chronic inflammation in obesity and neurodegenerative diseases: exploring the link in disease onset and progression. Mol. Biol. Rep. 2025, 52, 424. [CrossRef]
- Kim, M.; Oh, J.K.; Sakata, S.; Liang, I.; Park, W.; Hajjar, R.J.; Lebeche, D. Role of resistin in cardiac contractility and hypertrophy. J. Mol. Cell, Cardiol. 2008, 45, 270-280. [CrossRef]
- Chemaly, E.R.; Hadri, L.; Zhang, S.; Kim, M.; Kohlbrenner, E.; Sheng, J.; Liang, L.; Chen, J.; K-Raman, P.; Hajjar, R.J.; Lebeche, D. Long-term in vivo resistin overexpression induces myocardial dysfunction and remodeling in rats. J. Mol. Cell. Cardiol. 2011, 51, 144-155. [CrossRef]
- Zhao, B.; Bouchareb, R.; Lebeche, D. Resistin deletion protects against heart failure injury by targeting DNA damage response. Cardiovasc. Res. 2022, 118, 1947-1963. [CrossRef]
- Lebeche, D. Diabetic cardiomyopathy: is resistin a culprit? Cardiovasc. Diagn. Ther. 2015, 5, 387-393. [CrossRef]
- Liu, P.; Cheng, G.C.; Ye, Q.H.; Deng, Y.Z.; Wu, L. LKB1/AMPK pathway mediates resistin-induced cardiomyocyte hypertrophy in H9c2 embryonic rat cardiomyocytes. Biomed. Rep. 2016, 4, 387-391. [CrossRef]
- Yan, X.; Wu, L.; Gao, M.; Yang, P.; Yang, J.; Deng, Y. Omentin inhibits the resistin-induced hypertrophy of H9c2 cardiomyoblasts by inhibiting the TLR4/MyD88/NF-kappaB signaling pathway. Exp. Ther. Med. 2022, 23, 292. [CrossRef]
- Luo, J.W.; Zheng, X.; Cheng, G.C.; Ye, Q.H.; Deng, Y.Z.; Wu, L. Resistin-induced cardiomyocyte hypertrophy is inhibited by apelin through the inactivation of extracellular signal-regulated kinase signaling pathway in H9c2 embryonic rat cardiomyocytes. Biomed. Rep. 2016, 5, 473-478. [CrossRef]
- Kang, S.; Chemaly, E.R.; Hajjar, R.J.; Lebeche, D. Resistin promotes cardiac hypertrophy via the AMP-activated protein kinase/mammalian target of rapamycin (AMPK/mTOR) and c-Jun N-terminal kinase/insulin receptor substrate 1 (JNK/IRS1) pathways. J. Biol. Chem. 2011, 286, 18465-18473. [CrossRef]
- Lin, Q.; Kumar, S.; Kariyawasam, U.; Yang, X.; Yang, W.; Skinner, J.T.; Gao, W.D.; Johns, R.A. Human resistin induces cardiac dysfunction in pulmonary hypertension. J. Am. Heart. Assoc. 2023, 12, e027621. [CrossRef]
- Takeishi, Y.; Niizeki, T.; Arimoto, T.; Nozaki, N.; Hirono, O.; Nitobe, J.; Watanabe, T.; Takabatake. N.; Kubota, I. Serum resistin is associated with high risk in patients with congestive heart failure--a novel link between metabolic signals and heart failure. Circ. J. 2007, 71, 460-464. [CrossRef]
- Butler, J.; Kalogeropoulos, A.; Georgiopoulou, V.; de Rekeneire, N.; Rodondi, N.; Smith, A.L.; Hoffmann, U.; Kanaya, A.; Newman, A.B.; Kritchevsky, S.B.; Vasan, R.S.; Wilson, P.W.; Harris, T.B.; Health ABC Study. Serum resistin concentrations and risk of new onset heart failure in older persons: the health, aging, and body composition (Health ABC) study. Arterioscler. Thromb, Vasc. Biol. 2009, 29, 1144-1149. [CrossRef]
- Bhalla, V.; Kalogeropoulos, A.; Georgiopoulou, V.; Butler, J. Serum resistin: physiology, pathophysiology and implications for heart failure. Biomark. Med. 2010, 4, 445-452. [CrossRef]
- Frankel, D.S.; Vasan, R.S.; D’Agostino, R.B.Sr.; Benjamin, E.J.; Levy, D.; Wang, T.J.; Meigs, J.B. Resistin, adiponectin, and risk of heart failure the Framingham offspring study. J. Am. Coll. Cardiol. 2009, 53, 754-762. [CrossRef]
- Khan, R.S.; Kato, T.S.; Chokshi, A.; Chew, M.; Yu, S.; Wu, C.; Singh, P.; Cheema, F.;H.; Takayama, H.; Harris, C.; Reyes-Soffer, G.; Knöll, R.; Milting, H.; Naka, Y.; Mancini, D.; Schulze, P.C. Adipose tissue inflammation and adiponectin resistance in patients with advanced heart failure: correction after ventricular assist device implantation. Circ. Heart Fail. 2012 , 5, 340-348. [CrossRef]
- Cai, X.; Allison, M.A.; Ambale-Venkatesh, B.; Jorgensen, N.W.; Lima, J.A.C.; Muse, E.D.; McClelland, R.L.; Shea, S.; Lebeche, D. Resistin and risks of incident heart failure subtypes and cardiac fibrosis: the Multi-Ethnic Study of Atherosclerosis. ESC Heart Fail. 2022, 9, 3452-3460. [CrossRef]
- Samal, B.; Sun, Y.; Stearns, G.; Xie, C.; Suggs, S.; McNiece, I. Cloning and characterization of the cDNA encoding a novel human pre-B-cell colony-enhancing factor. Mol. Cell. Biol. 1994, 14, 1431-1437. [CrossRef]
- Wang, T.; Zhang, X.; Bheda, P.; Revollo, J.R.; Imai, S.; Wolberger, C. Structure of Nampt/PBEF/visfatin, a mammalian NAD+ biosynthetic enzyme. Nat. Struct. Mol. Biol. 2006, 13, 661-662. [CrossRef]
- Adeghate, E. Visfatin: structure, function and relation to diabetes mellitus and other dysfunctions. Curr. Med. Chem. 2008, 15, 1851-1862. [CrossRef]
- Stastny, J.; Bienertova-Vasku, J.; Vasku, A. Visfatin and its role in obesity development. Diabetes Metab. Syndr. 2012, 6, 120-124. [CrossRef]
- Chang, L.; Yang, R.; Wang, M.; Liu, J.; Wang, Y.; Zhang, H.; Li, Y. Angiotensin II type-1 receptor-JAK/STAT pathway mediates the induction of visfatin in angiotensin II-induced cardiomyocyte hypertrophy. Am. J. Med. Sci. 2012, 343, 220-226. [CrossRef]
- Yang, R.; Chang, L.; Wang, M.; Zhang, H.; Liu, J.; Wang, Y.; Jin, X.; Xu, L.; Li, Y. MAPK pathway mediates the induction of visfatin in neonatal SD rat cardiomyocytes pretreated with glucose. Biomed. Rep. 2014, 2, 282-286. [CrossRef]
- Dierickx, P.; Zhu, K.; Carpenter, B.J.; Jiang, C.; Vermunt, M.W.; Xiao, Y.; Luongo, T.S.; Yamamoto, T.; Martí-Pàmies, Í.; Mia, S.; Latimer, M.; Diwan, A.; Zhao. J.; Hauck, A.K.; Krusen, B.; Nguyen, H.C.B.; Blobel, G.A.; Kelly, D.P.; Pei, L.; Baur, J.A.; Young, M.E.; Lazar, MA. Circadian REV-ERBs repress E4bp4 to activate NAMPT-dependent NAD+ biosynthesis and sustain cardiac function. Nat. Cardiovasc. Res. 2022, 1, 45-58. [CrossRef]
- Yang, R.; Chang, L.; Liu, S.; Jin, X.; Li, Y. High glucose induces Rho/ROCK-dependent visfatin and type I procollagen expression in rat primary cardiac fibroblasts. Mol. Med. Rep. 2014,10, 1992-1998. [CrossRef]
- Zheng, M.; Lu, N.; Ren, M.; Chen, H. Visfatin associated with major adverse cardiovascular events in patients with acute myocardial infarction. BMC Cardiovasc. Disord. 2020, 20, 271. [CrossRef]
- Bełtowski J. Apelin and visfatin: unique “beneficial” adipokines upregulated in obesity? Med. Sci. Monit. 2006, 12, RA112-9.
- Lim, S.Y.; Davidson, S.M.; Paramanathan, A.J.; Smith, C.C.; Yellon, D.M.; Hausenloy D.J. The novel adipocytokine visfatin exerts direct cardioprotective effects. J. Cell. Mol. Med. 2008, 12, 1395-1403. [CrossRef]
- Hsu, C.P.; Hariharan, N.; Alcendor, R.R.; Oka, S.; Sadoshima, J. Nicotinamide phosphoribosyltransferase regulates cell survival through autophagy in cardiomyocytes. Autophagy 2009, 5, 1229-1231. [CrossRef]
- Yano, M.; Akazawa, H.; Oka, T.; Yabumoto, C.; Kudo-Sakamoto, Y.; Kamo, T.; Shimizu, Y.; Yagi, H.; Naito, A.T.; Lee, J.K.; Suzuki, J.; Sakata, Y.; Komuro, I. Monocyte-derived extracellular Nampt-dependent biosynthesis of NAD(+) protects the heart against pressure overload. Sci. Rep. 2015, 5, 15857. [CrossRef]
- Yu, X.Y.; Qiao, S.B.; Guan, H.S.; Liu, S.W.; Meng, X.M. Effects of visfatin on proliferation and collagen synthesis in rat cardiac fibroblasts. Horm. Metab. Res. 2010, 42, 507-513. [CrossRef]
- Pillai, V.B.; Sundaresan, N.R.; Kim, G.; Samant, S.; Moreno-Vinasco, L.; Garcia, J.G.; Gupta, M.P. Nampt secreted from cardiomyocytes promotes development of cardiac hypertrophy and adverse ventricular remodeling. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H415-H426. [CrossRef]
- Li, J.; Wu, W.; Zhao, M.; Liu, X. Involvement of TRPC1 in Nampt-induced cardiomyocyte hypertrophy through the activation of ER stress. Cell. Mol. Biol. (Noisy-le-grand) 2017, 63, 33-37. [CrossRef]
- Shen, C,; Fang, R.; Wang, J.; Wu, N.; Wang, S.; Shu, T.; Dai, J.; Feng, M.; Chen, X. Visfatin aggravates transverse aortic constriction-induced cardiac remodelling by enhancing macrophage-mediated oxidative stress in mice. J. Cell. Mol. Med. 2023, 27, 2562-2571. [CrossRef]
- Liu, Z.; Sammani, S.; Barber, C.J.; Kempf, C.L.; Li, F.; Yang, Z.; Bermudez, R.T.; Camp, S.M.; Herndon, V.R.; Furenlid, L.R.; Martin, D.R.; Garcia, J.G.N. An eNAMPT-neutralizing mAb reduces post-infarct myocardial fibrosis and left ventricular dysfunction. Biomed. Pharmacother. 2024, 170, 116103. [CrossRef]
- Majak, P.; Lunde, I.G.; Hasic, A.K.; Husebye, T.; Christensen, G.; Tønnessen, T.; Bjørnstad, J.L. Reduced visfatin levels in aortic stenosis increase after aortic valve replacement and may contribute to reverse left ventricular remodelling. J. Cardiovasc. Surg. (Torino), 2015, 56, 483-492.
- Oka, S.I.; Byun, J.; Huang, C.Y.; Imai, N.; Ralda, G.; Zhai, P.; Xu, X.; Kashyap, S.; Warren, J.; Alan Maschek, J.; Tippetts, T.S.; Tong, M.; Venkatesh, S.; Ikeda, Y.; Mizushima, W.; Kashihara, T.; Sadoshima, J. Nampt potentiates antioxidant defense in diabetic dardiomyopathy. Circ. Res. 2021, 129, 114-130. [CrossRef]
- Byun, J.; Oka, S.I.; Imai, N.; Huang, C.Y.; Ralda, G.; Zhai, P.; Ikeda, Y.; Ikeda, S.; Sadoshima, J. Both gain and loss of Nampt function promote pressure overload-induced heart failure. Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H711-H725. [CrossRef]
- Doan, K.V.; Luongo, T.S.; Ts’olo, T.T.; Lee, W.D.; Frederick, D.W.; Mukherjee, S.; Adzika, G.K.; Perry, C.E.; Gaspar, R.B.; Walker, N.; Blair, M.C.; Bye, N.; Davis, J.G.; Holman, C.D.; Chu, Q.; Wang, L.; Rabinowitz, J.D.; Kelly, D.P.; Cappola, T.P.; Margulies, K.B.; Baur, J.A. Cardiac NAD+ depletion in mice promotes hypertrophic cardiomyopathy and arrhythmias prior to impaired bioenergetics. Nat. Cardiovasc. Res. 2024, 3, 1236-1248. [CrossRef]
- Tatemoto, K.; Hosoya, M.; Habata, Y.; Fujii, R.; Kakegawa, T.; Zou, M.X.; Kawamata, Y.; Fukusumi, S.; Hinuma, S.; Kitada, C.; Kurokawa, T.; Onda, H.; Fujino, M. Isolation and characterization of a novel endogenous peptide ligand for the human APJ receptor. Biochem. Biophys. Res. Commun. 1998, 251, 471-476. [CrossRef]
- O’Dowd, B.F.; Heiber, M.; Chan, A.; Heng, H.H.; Tsui, L.C.; Kennedy, J.L.; Shi, X.; Petronis, A.; George, S.R.; Nguyen, T. A human gene that shows identity with the gene encoding the angiotensin receptor is located on chromosome 11. Gene 1993, 136, 355-360. [CrossRef]
- Siddiquee, K.; Hampton, J.; McAnally, D.; May, L.; Smith, L. The apelin receptor inhibits the angiotensin II type 1 receptor via allosteric trans-inhibition. Br. J. Pharmacol. 2013, 168, 1104-1117. [CrossRef]
- Siddiquee, K.; Hampton, J.; Khan, S.; Zadory, D.; Gleaves, L.; Vaughan, D.E.; Smith, L.H. Apelin protects against angiotensin II-induced cardiovascular fibrosis and decreases plasminogen activator inhibitor type-1 production. J. Hypertens. 2011, 29, 724-731. [CrossRef]
- Scimia, M.C.; Hurtado, C.; Ray, S.; Metzler, S.; Wei, K.; Wang, J.; Woods, C.E.; Purcell, N.H.; Catalucci, D.; Akasaka, T.; Bueno, O.F.; Vlasuk, G.P.; Kaliman, P.; Bodmer, R.; Smith, L.H.; Ashley, E.; Mercola, M.; Brown, J.H.; Ruiz-Lozano, P. APJ acts as a dual receptor in cardiac hypertrophy. Nature 2012, 488, 394-398. [CrossRef]
- Parikh, V.N.; Liu, J.; Shang, C.; Woods, C.; Chang, A.C.; Zhao, M.; Charo, D.N.; Grunwald, Z.; Huang, Y.; Seo, K.; Tsao, P.S.; Bernstein, D.; Ruiz-Lozano, P.; Quertermous, T.; Ashley, E.A. Apelin and APJ orchestrate complex tissue-specific control of cardiomyocyte hypertrophy and contractility in the hypertrophy-heart failure transition. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H348-H356. [CrossRef]
- Zhen, E.Y.; Higgs, R.E.; Gutierrez, J.A. Pyroglutamyl apelin-13 identified as the major apelin isoform in human plasma. Anal. Biochem. 2013, 442, 1-9. [CrossRef]
- Maguire, J.J.; Kleinz, M.J.; Pitkin, S.L.; Davenport, A.P. [Pyr1]apelin-13 identified as the predominant apelin isoform in the human heart: vasoactive mechanisms and inotropic action in disease. Hypertension 2009, 54, 598-604. [CrossRef]
- Chng, S.C.; Ho, L.; Tian, J.; Reversade, B. ELABELA: a hormone essential for heart development signals via the apelin receptor. Dev. Cell. 2013, 27, 672-680. [CrossRef]
- Wang, X.; Zhang, L.; Feng, M.; Xu, Z.; Cheng, Z.; Qian, L. ELA-11 protects the heart against oxidative stress injury induced apoptosis through ERK/MAPK and PI3K/AKT signaling pathways. Front. Pharmacol. 2022, 13, 873614. [CrossRef]
- Sato, T.; Sato, C.; Kadowaki, A.; Watanabe, H.; Ho, L.; Ishida, J.; Yamaguchi, T.; Kimura, A.; Fukamizu, A.; Penninger, J.M.; Reversade, B.; Ito, H.; Imai, Y.; Kuba, K. ELABELA-APJ axis protects from pressure overload heart failure and angiotensin II-induced cardiac damage. Cardiovasc. Res. 2017, 113, 760-769. [CrossRef]
- Zhou, H.; Gong, D.W.; Chen, L.; Chen, H.; Wang, Q.;, Ullah, M.; Ahmad, S.; Jeelani, I.; Zhao, Q. Fc-Elabela mitigates heart failure without liver and renal toxicity in mice. Front. Pharmacol. 2025, 16, 1555728. [CrossRef]
- Santos, R.A.S.; Sampaio, W.O.; Alzamora, A.C.; Motta-Santos, D.; Alenina, N.; Bader, M.; Campagnole-Santo, M.J. The ACE2/Angiotensin-(1-7)/Mas axis of the renin-angiotensin system: focus on angiotensin-(1-7). Physiol. Rev. 2018, 98, 505–553. [CrossRef]
- Sato, T.; Suzuki, T.; Watanabe, H.; Kadowaki, A.; Fukamizu, A.; Liu, P.P.; Kimura, A.; Ito, H.; Penninger, J.M.; Imai, Y.; Kuba, K. Apelin is a positive regulator of ACE2 in failing hearts. J. Clin. Invest. 2013, 123, 5203-5211. [CrossRef]
- Zhang, T.; Wang, X.; Wang, Z.; Zhai, J.; He, L.; Wang, Y.; Zuo, Q.; Ma, S.; Zhang, G.; Guo, Y. Canagliflozin ameliorates ventricular remodeling through Apelin/Angiotensin-converting enzyme 2 signaling in heart failure with preserved ejection fraction rats. Pharmacology 2023, 108, 478-491. [CrossRef]
- Chen, Z.; Wu, D.; Li, L.; Chen, L. Apelin/APJ System: A novel therapeutic target for myocardial ischemia/reperfusion Injury. DNA Cell Biol. 2016, 35, 766-775. [CrossRef]
- Tatemoto, K.; Takayama, K.; Zou, M.X.; Kumaki, I.; Zhang, W.; Kumano, K.; Fujimiya, M. The novel peptide apelin lowers blood pressure via a nitric oxide-dependent mechanism. Regul. Pept. 2001, 99, 87-92. [CrossRef]
- Li, L.; Zeng, H.; Chen, J.X. Apelin-13 increases myocardial progenitor cells and improves repair postmyocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H605-H618. [CrossRef]
- Li, L.; Zeng, H.; Hou, X.; He, X.; Chen, J.X. Myocardial injection of apelin-overexpressing bone marrow cells improves cardiac repair via upregulation of Sirt3 after myocardial infarction. PLoS One 2013, 8, e71041. [CrossRef]
- Zeng, H.; He, X.; Hou, X.; Li, L.; Chen, J.X. Apelin gene therapy increases myocardial vascular density and ameliorates diabetic cardiomyopathy via upregulation of sirtuin 3. Am. J. Physiol. Heart Circ. Physiol. 2014, 306, H585-H597. [CrossRef]
- Lv, W.; Zhang, L.; Cheng, X.; Wang, H.; Qin, W.; Zhou, X.; Tang, B. Apelin inhibits angiotensin II-induced atrial fibrosis and atrial fibrillation via TGF-β1/Smad2/α-SMA pathway. Front. Physiol. 2020, 11, 583570. [CrossRef]
- Chapman, F.A.; Maguire, J.J.; Newby, D.E.; Davenport, A.P.; Dhaun, N. Targeting the apelin system for the treatment of cardiovascular diseases. Cardiovasc. Res. 2023, 119, 2683-2696. [CrossRef]
- Pang, B.; Jiang, Y.R.; Xu, J.Y.; Shao, D.X.; Hao, L.Y. Apelin/ELABELA-APJ system in cardiac hypertrophy: Regulatory mechanisms and therapeutic potential. Eur. J. Pharmacol. 2023, 949, 175727. [CrossRef]
- Iwanaga, Y.; Kihara, Y.; Takenaka, H.; Kita, T. Down-regulation of cardiac apelin system in hypertrophied and failing hearts: Possible role of angiotensin II-angiotensin type 1 receptor system. J. Mol. Cell. Cardiol. 2006, 41, 798-806. [CrossRef]
- Kuba, K.; Zhang, L.; Imai, Y.; Arab, S.; Chen, M.; Maekawa, Y.; Leschnik, M.; Leibbrandt, A.; Markovic, M.; Schwaighofer, J.; Beetz, N.; Musialek, R.; Neely, G.G.; Komnenovic, V.; Kolm, U.; Metzler, B.; Ricci, R.; Hara, H.; Meixner, A.; Nghiem, M.; Chen, X.; Dawood, F.; Wong, K.M.; Sarao, R.; Cukerman, E.; Kimura, A.; Hein, L.; Thalhammer, J.; Liu, P.P.; Penninger, J.M. Impaired heart contractility in Apelin gene-deficient mice associated with aging and pressure overload. Circ. Res. 2007, 101, e32-42. [CrossRef]
- Falcão-Pires, I.; Gonçalves, N.; Gavina, C.; Pinho, S.; Teixeira, T.; Moura, C.; Amorim, M.J.; Pinho, P.; Areias, J.C.; Leite-Moreira, A. Correlation between plasma levels of apelin and myocardial hypertrophy in rats and humans: possible target for treatment? Expert Opin. Ther. Targets. 2010, 14, 231-241. [CrossRef]
- Ye, L.; Ding, F.; Zhang, L.; Shen, A.; Yao, H.; Deng, L.; Ding, Y. Serum apelin is associated with left ventricular hypertrophy in untreated hypertension patients. J. Transl. Med. 2015, 13, 290. [CrossRef]
- Helske, S.; Kovanen, P.T.; Lommi, J.; Turto, H.; Kupari, M. Transcardiac gradients of circulating apelin: extraction by normal hearts vs. release by hearts failing due to pressure overload. J. Appl. Physiol. 2010, 109, 1744-1748. [CrossRef]
- Szokodi, I.; Tavi, P.; Földes, G.; Voutilainen-Myllylä, S.; Ilves, M.; Tokola, H.; Pikkarainen, S.; Piuhola, J.; Rysä, J.; Tóth, M.; Ruskoaho, H. Apelin, the novel endogenous ligand of the orphan receptor APJ, regulates cardiac contractility. Circ. Res. 2002, 91, 434-440. [CrossRef]
- Ashley, E.A.; Powers, J.; Chen, M.; Kundu, R.; Finsterbach, T.; Caffarelli, A.; Deng, A.; Eichhorn, J.; Mahajan, R.; Agrawal, R.; Greve, J.; Robbins, R.; Patterson, A.J.; Bernstein, D.; Quertermous, T. The endogenous peptide apelin potently improves cardiac contractility and reduces cardiac loading in vivo. Cardiovasc. Res. 2005, 65, 73-82. [CrossRef]
- Barnes, G.D.; Alam, S.; Carter, G.; Pedersen, C.M.;, Lee, K.M.; Hubbard, T.J.; Veitch, S.; Jeong, H.; White, A.; Cruden, N.L.; Huson, L.; Japp, A.G.; Newby, D.E. Sustained cardiovascular actions of APJ agonism during renin-angiotensin system activation and in patients with heart failure. Circ. Heart Fail. 2013, 6, 482-491. [CrossRef]
- Japp, A.G.; Cruden, N.L.; Barnes, G.; van Gemeren, N.; Mathews, J.; Adamson, J.; Johnston, N.R.; Denvir, M.A.; Megson, I.L.; Flapan, A.D.; Newby, D.E. Acute cardiovascular effects of apelin in humans: potential role in patients with chronic heart failure. Circulation 2010, 121, 1818-1827. [CrossRef]
- Li, Y.; Lu, H.; Xu, W.; Shang, Y.; Zhao, C.; Wang, Y.; Yang, R.; Jin, S.; Wu, Y.; Wang, X.; Teng, X. Apelin ameliorated acute heart failure via inhibiting endoplasmic reticulum stress in rabbits. Amino Acids 2021, 53, 417-427. [CrossRef]
- Gargalovic, P.; Wong, P.; Onorato, J.; Finlay, H.; Wang, T.; Yan, M.; Crain, E.; St-Onge, S.; Héroux, M.; Bouvier, M.; Xu, C.; Chen, X.Q.; Generaux, C.; Lawrence, M.; Wexler, R.; Gordon, D. In vitro and In vivo evaluation of a small-molecule APJ (apelin receptor) agonist, BMS-986224, as a potential treatment for heart failure. Circ. Heart Fail. 2021, 14, e007351. [CrossRef]
- Girault-Sotias, P.E.; Deloux, R.; De Mota, N.; Riché, S.; Daubeuf, F.; Iturrioz, X.; Parlakian, A.; Berdeaux, A.; Agbulut, O.; Bonnet, D.; Boitard, S.E.; Llorens-Cortes, C. The metabolically resistant apelin-17 analogue LIT01-196 reduces cardiac dysfunction and remodelling in heart failure after myocardial infarction. Can. J. Cardiol. 2025, 41, 911-924. [CrossRef]
- Ceylan-Isik, A.F.; Kandadi, M.R.; Xu, X.; Hua, Y.; Chicco, A.J.; Ren, J.; Nair, S. Apelin administration ameliorates high fat diet-induced cardiac hypertrophy and contractile dysfunction. J. Mol. Cell. Cardiol. 2013, 63, 4-13. [CrossRef]
- Alfarano, C.; Foussal, C.; Lairez, O.; Calise, D.; Attané, C.; Anesia, R.; Daviaud, D.; Wanecq, E.; Parini, A.; Valet, P.; Kunduzova, O. Transition from metabolic adaptation to maladaptation of the heart in obesity: role of apelin. Int. J. Obes. (Lond) 2015, 39, 312-320. [CrossRef]
- Pchejetski, D.; Foussal, C.; Alfarano, C.; Lairez, O.; Calise, D.; Guilbeau-Frugier, C.; Schaak, S.; Seguelas, M.H.; Wanecq, E.; Valet, P.; Parini, A.; Kunduzova, O. Apelin prevents cardiac fibroblast activation and collagen production through inhibition of sphingosine kinase 1. Eur. Heart J. 2012, 33, 2360-2369. [CrossRef]
- Foussal, C.; Lairez, O.; Calise, D.; Pathak, A.; Guilbeau-Frugier, C.; Valet, P.; Parini, A.; Kunduzova O. Activation of catalase by apelin prevents oxidative stress-linked cardiac hypertrophy. FEBS Lett. 2010, 584, 2363-2370. [CrossRef]
- Wei, X.; Luo, L.; Lu, H.; Li, S.; Deng, X.; Li, Z.; Gong, D.; Chen, B. Apelin-13’s Actions in controlling hypertension-related cardiac hypertrophy and the expressions of inflammatory cytokines. Chem. Biol. Drug Des. 2024, 104, e14628. [CrossRef]
- Japp, A.G.; Newby, D.E. Unlocking the therapeutic potential of apelin. Hypertension 2016, 68, 307-309. [CrossRef]
- Serpooshan, V.; Sivanesan, S.; Huang, X.; Mahmoudi, M.; Malkovskiy, A.V.; Zhao, M.; Inayathullah, M.; Wagh, D.; Zhang, X.J.; Metzler, S.; Bernstein, D.; Wu, J.C.; Ruiz-Lozano, P.; Rajadas, J. [Pyr1]-Apelin-13 delivery via nano-liposomal encapsulation attenuates pressure overload-induced cardiac dysfunction. Biomaterials 2015, 37, 289-98. [CrossRef]
- Xie, F.; Liu, W.; Feng, F.; Li, X.; Yang, L.; Lv, D.; Qin, X.; Li, L.; Chen, L. A static pressure sensitive receptor APJ promote H9c2 cardiomyocyte hypertrophy via PI3K-autophagy pathway. Acta Biochim. Biophys. Sin. (Shanghai) 2014, 46, 699-708. [CrossRef]
- Xie, F.; Liu, W.; Feng, F.; Li, X.; He, L.; Lv, D.; Qin, X.; Li, L.; Li, L.; Chen, L. Apelin-13 promotes cardiomyocyte hypertrophy via PI3K-Akt-ERK1/2-p70S6K and PI3K-induced autophagy. Acta Biochim. Biophys. Sin. (Shanghai) 2015, 47, 969-980. [CrossRef]
- Wu, D.; Xie, F.; Xiao, L.; Feng, F.; Huang, S.; He, L.; Liu, M.; Zhou, Q.; Li, L.; Chen, L. Caveolin-1-autophagy pathway mediated cardiomyocyte hypertrophy induced by apelin-13. DNA Cell. Biol. 2017, 36, 611-618. [CrossRef]
- Xie, F.; Wu, D.; Huang, S.F.; Cao, J.G.; Li, H.N.; He, L.; Liu, M.Q.; Li, L.F.; Chen, L.X. The endoplasmic reticulum stress-autophagy pathway is involved in apelin-13-induced cardiomyocyte hypertrophy in vitro. Acta Pharmacol. Sin. 2017, 38, 1589-1600. [CrossRef]
- Hida, K.; Wada, J.; Eguchi, J.; Zhang, H.; Baba, M.; Seida, A.; Hashimoto, I.; Okada, T.; Yasuhara, A.; Nakatsuka, A.; Shikata, K.; Hourai, S.; Futami, J.; Watanabe, E.; Matsuki, Y.; Hiramatsu, R.; Akagi, S.; Makino, H.; Kanwar, Y.S. Visceral adipose tissue-derived serine protease inhibitor: a unique insulin-sensitizing adipocytokine in obesity. Proc. Natl. Acad. Sci. USA 2005, 102, 10610-10615. [CrossRef]
- Zhou, X.; Chen, Y.; Tao, Y.; Zhang, W.; Xu, W.; Lu, X. Serum Vaspin as a Predictor of Adverse Cardiac Events in Acute Myocardial Infarction. J. Am. Heart Assoc. 2019, 8, e010934. [CrossRef]
- Ji, M.; Li, Y.; Liu, Y.; Ma, G. Vaspin ameliorates cardiac remodeling by suppressing phosphoinositide 3-kinase/protein kinase B pathway to improve oxidative stress in heart failure rats. J. Cardiovasc. Pharmacol. 2022, 80, 442-452. [CrossRef]
- Rui, H.; Yu, H.; Zou, D.; Chi, K.; Xu, P.; Song, X.; Liu, L.; Wu, X.; Wang, J.; Xue, L. Vaspin alleviates pathological cardiac hypertrophy by regulating autophagy-dependent myocardial senescence. Emerg. Crit. Care Med. 2024, 4, 4-15. [CrossRef]
- Zhang, D.; Zhu, H.; Zhan, E.; Wang, F.; Liu, Y.; Xu, W.; Liu, X.;Liu, J.; Li, S.; Pan, Y.; Wang, Y.; Cao, W. Vaspin mediates the intraorgan crosstalk between heart and adipose tissue in lipoatrophic mice. Front. Cell Dev. Biol. 2021, 9, 647131. [CrossRef]
- Cook, K.S.; Min, H.Y.; Johnson, D.; Chaplinsky, R.J.; Flier, J.S.; Hunt, C.R.; Spiegelman, B.M. Adipsin: a circulating serine protease homolog secreted by adipose tissue and sciatic nerve. Science 1987, 237, 402-405. [CrossRef]
- Rosen, B.S.; Cook, K.S.; Yaglom, J.; Groves, D.L.; Volanakis, J.E.; Damm, D.; White, T.; Spiegelman, B.M. Adipsin and complement factor D activity: an immune-related defect in obesity. Science 1989,244, 1483-1487. [CrossRef]
- Lo, J.C.; Ljubicic, S.; Leibiger, B.; Kern, M.; Leibiger, I.B.; Moede, T.; Kelly, M.E.; Chatterjee Bhowmick, D.; Murano, I.; Cohen, P.; Banks, A.S.; Khandekar, M.J.; Dietrich, A.; Flier, J.S.; Cinti, S.; Blüher, M.; Danial, N.N.; Berggren, P.O.; Spiegelman, B.M. Adipsin is an adipokine that improves beta cell function in diabetes. Cell 2014, 158, 41-53. [CrossRef]
- Gómez-Banoy, N.; Lo, J.C. Adipokines as key players in β cell function and failure. Clin. Sci. (Lond) 2019, 133, 2317-2327. [CrossRef]
- Dare, A.;, Chen, S.Y. Adipsin in the pathogenesis of cardiovascular diseases. Vascul. Pharmacol. 2024, 154, 107270. [CrossRef]
- Romere, C.; Duerrschmid, C.; Bournat, J.; Constable, P.; Jain, M.; Xia, F.; Saha, P.K.; Del Solar, M.; Zhu, B.; York, B.; Sarkar, P.; Rendon, D.A.; Gaber, M.W.; LeMaire, S.A.; Coselli, J.S.; Milewicz, D.M.; Sutton, V.R.; Butte, N.F.; Moore, D.D.; Chopra, A.R. Asprosin, a fasting-induced glucogenic protein hormone. Cell 2016, 165, 566-579. [CrossRef]
- Kocaman, N.; Kuloğlu, T. Expression of asprosin in rat hepatic, renal, heart, gastric, testicular and brain tissues and its changes in a streptozotocin-induced diabetes mellitus model. Tissue Cell 2020, 66, 101397. [CrossRef]
- Wen, M.S.; Wang, C.Y.; Yeh, J.K.; Chen, C.C.; Tsai, M.L.; Ho, M.Y.; Hung, K.C.; Hsieh, I.C. The role of asprosin in patients with dilated cardiomyopathy. BMC Cardiovasc. Disord. 2020, 20, 402. [CrossRef]
- Savcılıoglu, M.D.; Duzen, I.V.; Tuluce, S.Y.; Savcılıoglu, N.; Vuruskan, E.; Altunbas, G.; Kaplan, M.; Baloglu, M.; Tabur, S.; Sucu, M.; Taysı. S. The effects of SGLT-2 inhibitors on echocardiographic indices and antioxidative properties in patients with heart failure with reduced ejection fraction and diabetes mellitus. Eur. Rev. Med. Pharmacol. Sci. 2024, 28, 4121-4135. [CrossRef]
- Wang, G.; Fan, C.; Chai, Y.; Yu, X.; Xing, M.; Lv, Z.; Yuan, S.; Dai, H. Association of serum asprosin concentrations with heart failure. BMC Cardiovasc. Disord. 2023, 23, 617. [CrossRef]
- Liang, D.; Shi, G.; Xu, M.; Yin, J.; Liu, Y.; Yang, J.; Xu, L. The correlation between serum asprosin and left ventricular diastolic dysfunction in elderly patients with type 2 diabetes mellitus in the community. J. Diabetes Investig. 2024, 15, 608-613. [CrossRef]
- Nagpal, S.; Patel, S.; Jacobe, H.; DiSepio, D.; Ghosn, C.; Malhotra, M.; Teng, M.; Duvic, M.; Chandraratna, R.A. Tazarotene-induced gene 2 (TIG2), a novel retinoid-responsive gene in skin. J. Invest. Dermatol. 1997, 109, 91-95. [CrossRef]
- Rourke, J.L.; Dranse, H.J.; Sinal, C.J. Towards an integrative approach to understanding the role of chemerin in human health and disease. Obes. Rev. 2013, 14, 245-262. [CrossRef]
- Helfer, G.; Wu, Q.F. Chemerin: a multifaceted adipokine involved in metabolic disorders. J. Endocrinol. 2018, 238, R79-R94. [CrossRef]
- Goralski, K.B.; McCarthy, T.C.; Hanniman, E.A.; Zabel, B.A.; Butcher, E.C.; Parlee, S.D.; Muruganandan, S.; Sinal, C.J. Chemerin, a novel adipokine that regulates adipogenesis and adipocyte metabolism. J. Biol. Chem. 2007, 282, 28175-28188. [CrossRef]
- Inci, S.; Aksan, G.; Doğan, P. Chemerin as an independent predictor of cardiovascular event risk. Ther. Adv. Endocrinol. Metab. 2016, 7, 57-68. [CrossRef]
- Zhou, X.; Tao, Y.; Chen, Y.; Xu, W.; Qian, Z.; Lu, X. Serum chemerin as a novel prognostic indicator in chronic heart failure. J. Am. Heart Assoc. 2019, 8, e012091. [CrossRef]
- Chen, L.; Meng, J.; Zheng, J.; Zhao, T.; Li, Q.; Lu, C. Role of CCRL2 in the pathogenesis of experimental autoimmune myocarditis via P21-activated kinase 1/NOD-like receptor protein 3 pathway. Int. Heart J. 2024, 65, 339-348. [CrossRef]
- Imiela, A.M.; Stępnicki, J.; Zawadzka, P.S.; Bursa, A.; Pruszczyk, P. Chemerin as a driver of cardiovascular diseases: New perspectives and future directions. Biomedicines 2025, 13, 1481. [CrossRef]
- Li, Z.; Gao, Z.; Sun, T.; Zhang, S.; Yang, S.; Zheng, M.; Shen, H. Meteorin-like/Metrnl, a novel secreted protein implicated in inflammation, immunology, and metabolism: A comprehensive review of preclinical and clinical studies. Front. Immunol. 2023, 14, 1098570. [CrossRef]
- Lee, D.E.; McKay, L.K.; Bareja, A.; Li, Y.; Khodabukus, A.; Bursac, N.; Taylor, G.A.; Baht, G.S.; White, J.P. Meteorin-like is an injectable peptide that can enhance regeneration in aged muscle through immune-driven fibro/adipogenic progenitor signaling. Nat. Commun. 2022, 13, 7613. [CrossRef]
- Li, Z.Y.; Zheng, S.L.; Wang, P.; Xu, T.Y.; Guan, Y.F.; Zhang, Y.J.; Miao, C.Y. Subfatin is a novel adipokine and unlike Meteorin in adipose and brain expression. CNS Neurosci. Ther. 2014, 20, 344-354. [CrossRef]
- Rupérez, C.; Ferrer-Curriu, G.; Cervera-Barea, A.; Florit, L.; Guitart-Mampel, M.; Garrabou, G.; Zamora, M.; Crispi, F.; Fernandez-Solà, J.; Lupón, J.; Bayes-Genis, A.; Villarroya, F.; Planavila, A. Meteorin-like/Meteorin-β protects heart against cardiac dysfunction. J. Exp, Med. 2021, 218, e20201206. [CrossRef]
- Reboll, M.R.; Klede, S.; Taft, M.H.; Cai, C.L.; Field, L.J.; Lavine, K.J.;, Koenig, A.L.; Fleischauer, J.; Meyer, J.; Schambach, A.; Niessen, H.W.; Kosanke, M.; van den Heuvel, J.; Pich, A.; Bauersachs, J.; Wu, X.; Zheng, L.; Wang, Y.; Korf-Klingebiel, M.; Polten, F.; Wollert, K.C. Meteorin-like promotes heart repair through endothelial KIT receptor tyrosine kinase. Science 2022, 376, 1343-1347. [CrossRef]
- Chen, D.X.; Feng, Y.Y.; Wang, H.Y.; Lu, C.H.; Liu, D.Z.; Gong, C.; Xue, Y.; Na, N.; Huang, F. Metrnl ameliorates myocardial ischemia-reperfusion injury by activating AMPK-mediated M2 macrophage polarization. Mol. Med. 2025, 31, 98. [CrossRef]
- Hu, C.; Zhang, X.; Song, P.; Yuan, Y.P.; Kong, C.Y.; Wu, H.M.; Xu, S.C.; Ma, Z.G.; Tang, Q.Z. Meteorin-like protein attenuates doxorubicin-induced cardiotoxicity via activating cAMP/PKA/SIRT1 pathway. Redox Biol. 2020, 37, 101747. [CrossRef]
- Cao, H.; Liao, Y.; Hong, J. Protective effects of METRNL overexpression against pathological cardiac remodeling. Gene 2024, 901, 148171. [CrossRef]
- Lu, Q.B.; Ding, Y.; Liu, Y.; Wang, Z.C.; Wu, Y.J.; Niu, K.M.; Li, K.X.; Zhang, J.R.; Sun, H.J. Metrnl ameliorates diabetic cardiomyopathy via inactivation of cGAS/STING signaling dependent on LKB1/AMPK/ULK1-mediated autophagy. J. Adv, Res. 2023, 51, 161-179. [CrossRef]
- Mizushima, N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr. Opin. Cell. Biol. 2010, 22, 132-139. [CrossRef]
- Li, J.; Hong, Y.; Zhong, Y.; Yang, S.; Pei, L.; Huang, Z.; Long, H.; Chen, X.; Zhou, C.; Zheng, G.; Zeng, C.; Wu, H.; Wang, T. Meteorin-like (METRNL) attenuates hypertensive induced cardiac hypertrophy by inhibiting autophagy via activating BRCA2. Biochim. Biophys. Acta Mol. Basis Dis. 2024, 1870, 167113. [CrossRef]
- Shangguan, J.; Liu, G.; Xiao, L.; Zhang, W.; Zhu, X.; Li, L. Meteorin-like/meteorin-β protects against cardiac dysfunction after myocardial infarction in mice by inhibiting autophagy. Exp. Ther. Med. 2024, 28, 293. [CrossRef]
- Cai, J.; Wang, Q.M.; Li, J.W.; Xu, F.; Bu, Y.L.; Wang, M.; Lu, X.; Gao, W. Serum Meteorin-like is associated with weight loss in the elderly patients with chronic heart failure. J. Cachexia Sarcopenia Muscle. 2022, 13, 409-417. [CrossRef]
- Anido-Varela, L.; Aragón-Herrera, A.; González-Maestro, A.; Bellas, CT.; Tarazón, E.; Solé-González, E.; Martínez-Sellés, M.; Guerra-Ramos, J.M.; Carrasquer, A.; Morán-Fernández, L.; García-Vega, D.; Seoane-Blanco, A.; Moure-González, M.; Seijas-Amigo, J.; Rodríguez-Penas, D.; García-Seara, J.; Moraña-Fernández, S.; Vázquez-Abuín, X.; Roselló-Lletí, E.; Portolés, M.; Eiras, S.; Agra, R.M.; Álvarez, E.; González-Juanatey, J.R.; Feijóo-Bandín, S.; Lago, F.; REDINSCOR III registry. Meteorin-like protein plasma levels are associated with worse outcomes in de novo heart failure. Eur. J. Clin. Invest. 2025, 55, e14380. [CrossRef]
- Zhou, J.; Gao, G.; Crabb, J.W.; Serrero, G. Purification of an autocrine growth factor homologous with mouse epithelin precursor from a highly tumorigenic cell line. J. Biol. Chem. 1993, 268, 10863-10869. [CrossRef]
- Bateman, A.; Bennett, H.P. Granulins: the structure and function of an emerging family of growth factors. J. Endocrinol. 1998, 158, 145-151. [CrossRef]
- Wang, X.; Liang, Y.; Yang, F.; Shi, Y.; Shao, R.; Jing, R.; Yang, T.; Chu, Q.; An, D.; Zhou, Q.; Song, J.; Chen, H.; Liu, C. Molecular mechanisms and targeted therapy of progranulin in metabolic diseases. Front. Endocrinol. (Lausanne). 2025, 16, 1553794. [CrossRef]
- Alyahya. A.M. The role of progranulin in ischemic heart disease and its related risk factors. Eur. J. Pharm. Sci. 2022, 175, 106215. [CrossRef]
- Alyahya, A.M.; Al-Masri, A.; Hersi, A.; El Eter, E.; Husain, S.; Lateef, R.; Mawlana, O.H. The effects of progranulin in a rat model of acute myocardial ischemia/reperfusion are mediated by activation of the P13K/Akt Signaling pathway. Med. Sci. Monit. Basic Res. 2019, 25, 229-237. [CrossRef]
- Saeedi-Boroujeni, A.; Purrahman, D.; Shojaeian, A.; Poniatowski, Ł.A.; Rafiee, F.; Mahmoudian-Sani, M.R. Progranulin (PGRN) as a regulator of inflammation and a critical factor in the immunopathogenesis of cardiovascular diseases. J. Inflamm. (Lond) 2023, 20, 1. [CrossRef]
- Sasaki, T.; Shimazawa, M.; Kanamori, H.; Yamada, Y.; Nishinaka, A.; Kuse, Y.; Suzuki, G.; Masuda, T.; Nakamura, S.; Hosokawa, M.; Minatoguchi, S.; Hara, H. Effects of progranulin on the pathological conditions in experimental myocardial infarction model. Sci. Rep. 2020, 10, 11842. [CrossRef]
- Sasaki, T.; Kuse, Y.; Nakamura, S.; Shimazawa, M.; Hara, H. Progranulin deficiency exacerbates cardiac remodeling after myocardial infarction. FASEB Bioadv 2023, 5, 395-411. [CrossRef]
- Zhu, Y.; Ohama, T.; Kawase, R.; Chang, J.; Inui, H.; Kanno, K.; Okada, T.; Masuda, D.; Koseki, M.; Nishida, M.; Sakata, Y.; Yamashita, S. Progranulin deficiency leads to enhanced age-related cardiac hypertrophy through complement C1q-induced β-catenin activation. J. Mol. Cell. Cardiol. 2020, 138, 197-211. [CrossRef]
- Bergmann, M.W. WNT signaling in adult cardiac hypertrophy and remodeling: lessons learned from cardiac development. Circ. Res. 2010, 107, 1198-1208. [CrossRef]
- Minatoguchi, S.; Satake, A.; Murase, H.; Yoshizumi, R.; Komaki, H.; Baba, S.; Yasuda, S.; Ojio, S.; Tanaka, T.; Okura, H.; Minatoguchi, S. Elevated plasma progranulin levels in the acute phase are correlated with recovery of left ventricular function in the chronic phase in patients with acute myocardial infarction. PLoS One 2024, 19, e0313014. [CrossRef]
- Harari, D.; Tzahar, E.; Romano, J.; Shelly, M.; Pierce, J.H.; Andrews, G.C.; Yarden, Y. Neuregulin-4: a novel growth factor that acts through the ErbB-4 receptor tyrosine kinase. Oncogene 1999, 18, 2681-2689. [CrossRef]
- Chen, M.; Zhu, J.; Luo, H.; Mu, W.; Guo, L. The journey towards physiology and pathology: Tracing the path of neuregulin 4. Genes Dis. 2023, 11, 687-700. [CrossRef]
- Liu, Y.; Chen, M. Neuregulin 4 as a novel adipokine in energy metabolism. Front. Physiol. 2023, 13, 1106380. [CrossRef]
- Wang, H.; Wang, L.; Hu, F.; Wang, P.; Xie, Y.; Li, F.; Guo, B. Neuregulin-4 attenuates diabetic cardiomyopathy by regulating autophagy via the AMPK/mTOR signalling pathway. Cardiovasc. Diabetol. 2022, 21, 205. [CrossRef]
- Wang, P.; Guo, X.; Wang, H.; Wang, L.; Ma, M.; Guo, B. Neuregulin-4 protects cardiomyocytes against high-glucose-induced ferroptosis via the AMPK/NRF2 signalling pathway. Biol. Direct 2024, 19, 62. [CrossRef]
- Wei, H.; Guo, X.; Yan, J.; Tian, X.; Yang, W.; Cui, K.; Wang, L.; Guo, B. Neuregulin-4 alleviates isoproterenol (ISO)-induced cardial remodeling by inhibiting inflammation and apoptosis via AMPK/NF-κB pathway. Int. Immunopharmacol. 2024,143, 113301. [CrossRef]
- Yang, Z.; Yaling, W.; Tao, L.; Mingyue, R.; Yongjun, L. Cardioprotective effect of NRG-4 gene expression on spontaneous hypertension rats and its mechanism through mediating the activation of ErbB signaling pathway. Cell. Mol. Biol. (Noisy-le-grand) 2022, 68, 89-101. [CrossRef]
- Zhang, Y.; Liu, Y.; Wang, Y. PCBP2 Promotes NRG4 mRNA Stability to Diminish Angiotensin II-Induced Hypertrophy, NLRP3 Inflammasome Activation, and Oxidative Stress of AC16 Cardiomyocytes. Cell Biochem. Biophys. 2025, Jul 18. [CrossRef]
- Zheng, L.; Zhang, C.; Bu, S.; Guo, W.; Li, T.; Xu, Y.; Liu, Y.; Yuan, C.; Feng, C.; Zong, G.; Zhu, J.; Xing, M.; Geng, X. The Causal Effect of Serum Lipid Levels Mediated by Neuregulin 4 on the Risk of Four Atherosclerosis Subtypes: Evidence from Mendelian Randomization Analysis. Vasc Health Risk Manag. 2024, 20, 351-357. [CrossRef]
- Tian, Q., Liu, M.L.; Tang, C.S.; Xue, L.; Pang, Y.Z.; Qi, Y.F. Association of Circulating Neuregulin-4 with Presence and Severity of Coronary Artery Disease. Int. Heart. J. 2019, 60, 45-49. [CrossRef]
- Craig, R.L.; Chu, W.S.; Elbein, S.C. Retinol binding protein 4 as a candidate gene for type 2 diabetes and prediabetic intermediate traits. Mol. Genet. Metab. 2007, 90, 338–344. [CrossRef]
- Zhang, K.; Wang, L.; Gao, W.; Guo, R. Retinol-binding protein 4 in skeletal and cardiac muscle: molecular mechanisms, clinical implications, and future perspectives. Front. Cell Dev. Biol. 2025, 13, 1587165. [CrossRef]
- Gao, W.; Wang, H.; Zhang, L.; Cao, Y.; Bao, J.Z.; Liu, Z.X.; Wang, L.S.; Yang, Q.; Lu, X. Retinol-Binding Protein 4 Induces Cardiomyocyte Hypertrophy by Activating TLR4/MyD88 Pathway. Endocrinology 2016, 157, 2282-2293. [CrossRef]
- Shan, H.; Ji, Y.; Gu, H.; Li, H.; Zhu, J.; Feng, Y.; Peng, H.; You, T.; Gu, X. Elevated Serum Retinol Binding Protein 4 is Associated with the Risk of Diabetic Cardiomyopathy. Rev. Cardiovasc. Med. 2022, 23, 115. [CrossRef]
- Li, X.Z.; Zhang, K.Z.; Yan, J.J.; Wang, L.; Wang, Y.; Shen, X.; Sun, H.X.; Liu, L.; Zhao, C.; He, H.W.; Wang, L.S.; Gao, W.; Lu, X. Serum retinol-binding protein 4 as a predictor of cardiovascular events in elderly patients with chronic heart failure. ESC Heart Fail. 2020, 7, 542-550. [CrossRef]
- Si, Y.; Fan, W.; Sun, L. A. Review of the relationship between CTRP family and coronary artery disease. Curr. Atheroscler. Rep. 2020, 22, 22. [CrossRef]
- Guo, S.; Mao, X.; Liu, J. Multi-faceted roles of C1q/TNF-related proteins family in atherosclerosis. Front. Immunol. 2023, 14, 1253433. [CrossRef]
- Gu, Y.; Hu, X.; Ge, P.B.; Chen, Y.; Wu, S.; Zhang, X.W. CTRP1 aggravates cardiac dysfunction post myocardial infarction by modulating TLR4 in macrophages. Front. Immunol. 2021, 12, 635267. [CrossRef]
- Wu, L.; Gao, L.; Zhang, D.; Yao, R.; Huang, Z.; Du, B.; Wang, Z.; Xiao, L.; Li, P.; Li, Y.; Liang, C.; Zhang, Y. C1QTNF1 attenuates angiotensin II-induced cardiac hypertrophy via activation of the AMPKa pathway. Free Radic. Biol. Med. 2018, 121, 215-230. [CrossRef]
- Li, C.; Ying, S.; Wu, X.; Zhu, T.; Zhou, Q.; Zhang, Y.; Liu, Y.; Zhu, R.; Hu, H. CTRP1 Aggravates Cardiac Fibrosis by Regulating The NOX2/P38 Pathway in Macrophages. Cell J. 2022, 24, 732-740. [CrossRef]
- Yi, W.; Sun, Y.; Yuan, Y.; Lau, W.B.; Zheng, Q.; Wang, X.; Wang, Y.; Shang, X.; Gao, E.; Koch, W.J.; Ma, X.L. C1q/tumor necrosis factor-related protein-3, a newly identified adipokine, is a novel antiapoptotic, proangiogenic, and cardioprotective molecule in the ischemic mouse heart. Circulation 2012, 125, 3159-3169. [CrossRef]
- Zhang, B.; Zhang, P.; Tan, Y.; Feng, P.; Zhang, Z.; Liang, H.; Duan, W.; Jin, Z.; Wang, X.; Liu, J.; Gao, E.; Yu, S.; Yi, D.; Sun, Y.; Yi, W. C1q-TNF-related protein-3 attenuates pressure overload-induced cardiac hypertrophy by suppressing the p38/CREB pathway and p38-induced ER stress. Cell Death Discov. 2019, 10, 520. [CrossRef]
- Shi, L.; Tan, Y.; Zheng, W.; Cao, G.; Zhou, H.; Li, P.; Cui, J.; Song, Y.; Feng, L.; Li, H.; Shan, W.; Zhang, B.; Yi, W. CTRP3 alleviates mitochondrial dysfunction and oxidative stress injury in pathological cardiac hypertrophy by activating UPRmt via the SIRT1/ATF5 axis. Cell Death Discov. 2024, 10, 53. [CrossRef]
- Ma, Z.G.; Yuan, Y,P.; Xu, S.C.; Wei, W,Y.; Xu, C.R.; Zhang, X.; Wu, Q.Q.; Liao, H.H.; Ni, J.; Tang, Q.Z. CTRP3 attenuates cardiac dysfunction, inflammation, oxidative stress and cell death in diabetic cardiomyopathy in rats. Diabetologia 2017,60, 1126-1137. [CrossRef]
- Wei, W.Y.; Ma, Z.G.; Zhang, N.; Xu, S.C.; Yuan, Y.P.; Zeng, X.F.; Tang, Q.Z. Overexpression of CTRP3 protects against sepsis-induced myocardial dysfunction in mice. Mol. Cell. Endocrinol. 2018, 476, 27-36. [CrossRef]
- Wu, D.; Lei, H.; Wang, J.Y.; Zhang, C.L.; Feng, H.; Fu, F.Y.; Li, L.; Wu, L.L. CTRP3 attenuates post-infarct cardiac fibrosis by targeting Smad3 activation and inhibiting myofibroblast differentiation. J. Mol. Med. (Berl). 2015, 93, 1311-1325. [CrossRef]
- Ma, Z.G.; Yuan, Y.P.; Zhang, X.; Xu, S.C.; Kong, C.Y.; Song, P.; Li, N.; Tang, Q.Z. C1q-tumour necrosis factor-related protein-3 exacerbates cardiac hypertrophy in mice. Cardiovasc. Res. 2019, 115, 1067-1077. [CrossRef]
- Fan, T.; Zhu, N.; Li, M.; Wang, Z.; Lin, X. CTRP6-mediated cardiac protection in heart failure via the AMPK/SIRT1/PGC-1α signalling pathway. Exp. Physiol. 2024, 109, 2031-2045. [CrossRef]
- Lei, H.; Wu, D.; Wang, J.Y.; Li, L.; Zhang, C.L.; Feng, H.; Fu, F.Y.; Wu, L.L. C1q/tumor necrosis factor-related protein-6 attenuates post-infarct cardiac fibrosis by targeting RhoA/MRTF-A pathway and inhibiting myofibroblast differentiation. Basic Res. Cardiol. 2015, 110, 35. [CrossRef]
- Niemann, B.; Li, L.; Siegler, D.; Siegler, B.H.; Knapp, F.; Hanna, J.; Aslam, M.; Kracht, M.; Schulz, R.; Rohrbach, S. CTRP9 mediates protective effects in cardiomyocytes via AMPK- and adiponectin receptor-mediated induction of anti-oxidant response. Cells 2020, 9, 1229. [CrossRef]
- Zuo, A.; Zhao, X.; Li, T.; Li, J.; Lei, S.; Chen, J.; Xu, D.; Song, C.; Liu, T.; Li, C.; Guo, Y. CTRP9 knockout exaggerates lipotoxicity in cardiac myocytes and high-fat diet-induced cardiac hypertrophy through inhibiting the LKB1/AMPK pathway. J. Cell. Mol. Med. 2020, 24, 2635-2647. [CrossRef]
- Appari, M.; Breitbart, A.; Brandes, F.; Szaroszyk, M.; Froese, N.; Korf-Klingebiel, M.; Mohammadi, M.M.; Grund, A.; Scharf, G.M.; Wang, H.; Zwadlo, C.; Fraccarollo, D.; Schrameck, U.; Nemer, M.; Wong, G.W.; Katus, H.A.; Wollert, K.C.; Müller, O.J.; Bauersachs, J.; Heineke, J. C1q-TNF-related protein-9 promotes cardiac hypertrophy and failure. Circ. Res. 2017, 120, 66-77. [CrossRef]
- Bai, B.; Ji, Z.; Wang, F.; Qin, C.; Zhou, H.; Li, D.; Wu, Y. CTRP12 ameliorates post-myocardial infarction heart failure through down-regulation of cardiac apoptosis, oxidative stress and inflammation by influencing the TAK1-p38 MAPK/JNK pathway. Inflamm. Res. 2023, 72, 1375-1390. [CrossRef]
- Zhao, Q.; Zhang, C.L.; Xiang R,L.; Wu, L.L.; Li, L. CTRP15 derived from cardiac myocytes attenuates TGFβ1-induced fibrotic response in cardiac fibroblasts. Cardiovasc. Drugs Ther. 2020, 34, 591-604. [CrossRef]
- Sawicka, M.; Janowska, J.; Chudek, J. Potential beneficial effect of some adipokines positively correlated with the adipose tissue content on the cardiovascular system. Int. J. Cardiol. 2016, 222, 581-589. [CrossRef]
- Packer M. The adipokine hypothesis of heart failure with a preserved ejection fraction: A novel framework to explain pathogenesis and guide treatment. J. Am. Coll. Cardiol. 2025, 86, 1269-1373. [CrossRef]
- Packer, M.; Butler, J.; Lam, C.S.P.; Zannad, F.; Vaduganathan, M.; Borlaug, B.A. Central adiposity or hypertension: Which drives heart failure with a preserved ejection fraction? J. Am. Coll. Cardiol. 2025, 86, 1935-1949. [CrossRef]
| Experimental model | Proposed cellular mechanisms | Reference |
| NRCM | p38 MAPK | [47] |
| NRCM | ET-1/ROS pathway | [48] |
| PHVM | JAK and MAPK pathways | [49] |
| NRCM | RhoA/ROCK | [50,51] |
| NRCM | mTOR-dependent RhoA/ROCK | [52] |
| NRCM | JAK/STAT activation | [53] |
| NRCM | RhoA/ROCK-caveolae upregulation | [54] |
| NRCM | PPARα activation | [55] |
| NRCM | calcineurin/NFAT | [56] |
| NRCM | JAK/STAT/CUX1-dependent FTO upregulation | [57] |
| Myocardial leptin overexpressing mice | TGFβ upregulation | [58] |
| Experimental model | Proposed cellular mechanisms | Reference |
| Mouse TAC | AMPK activation | [64] |
| NRCM | AMPK activation | [65] |
| STZ diabetic rat | Nrf2/ Brg1 activation & HO-1 induction | [67] |
| Aged mouse DMD model | CAMKK2/pAMPK/PGC-1α activation | [68] |
| Mouse TAC | cytokine reduction, other metabolic effects | [69] |
| STZ diabetic female rats | increased Cx43 expression | [70] |
| Mouse TAC | AMPK activation | [71,72] |
| Ang II infusion [rat] | AMPK activation/MiR-133a upregulation | [73] |
| Aldosterone infusion in UNX mice | reduced cytokines and oxidative stress | [74] |
| NRAM | AMPK activation | [75] |
| Ang II infusion (mouse) | Inhibition of β-catenin pathway | [76] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




