Submitted:
25 November 2025
Posted:
26 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
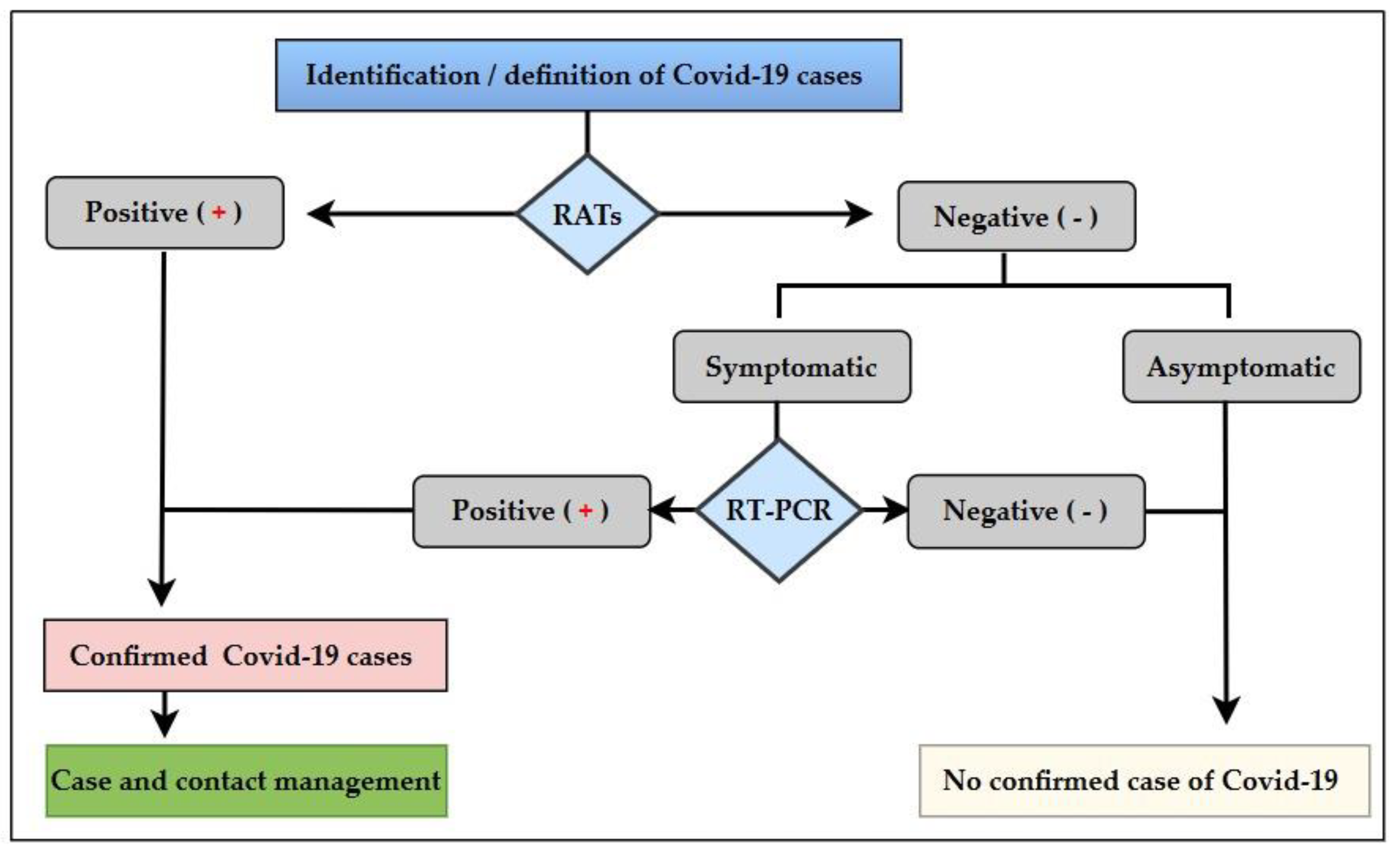
2.1. Settings and Design
2.2. Population and Selection Criteria
2.3. Data Collection and Analysis
2.4. Sample Collection and Laboratory Procedures
2.5. Ethical Considerations
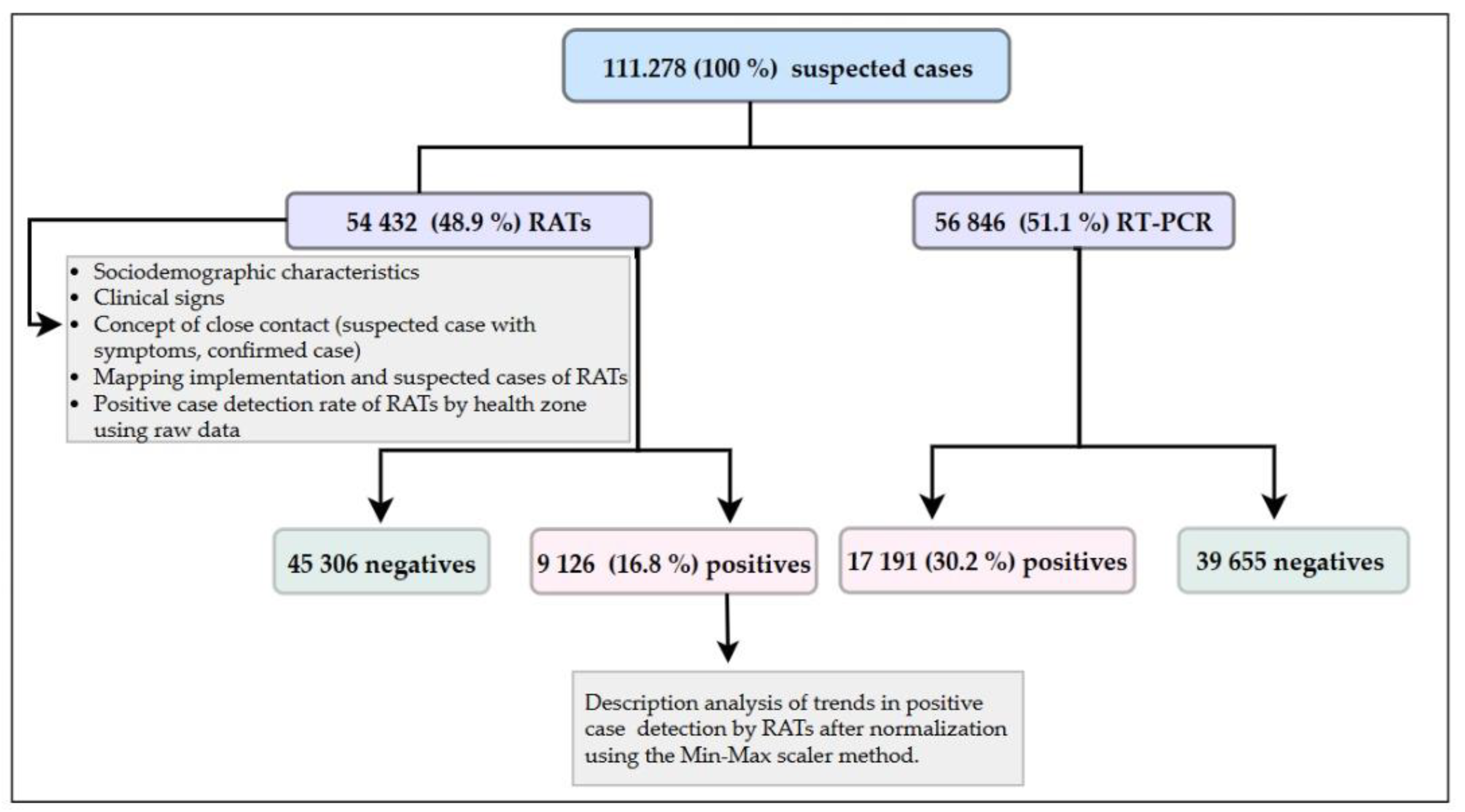
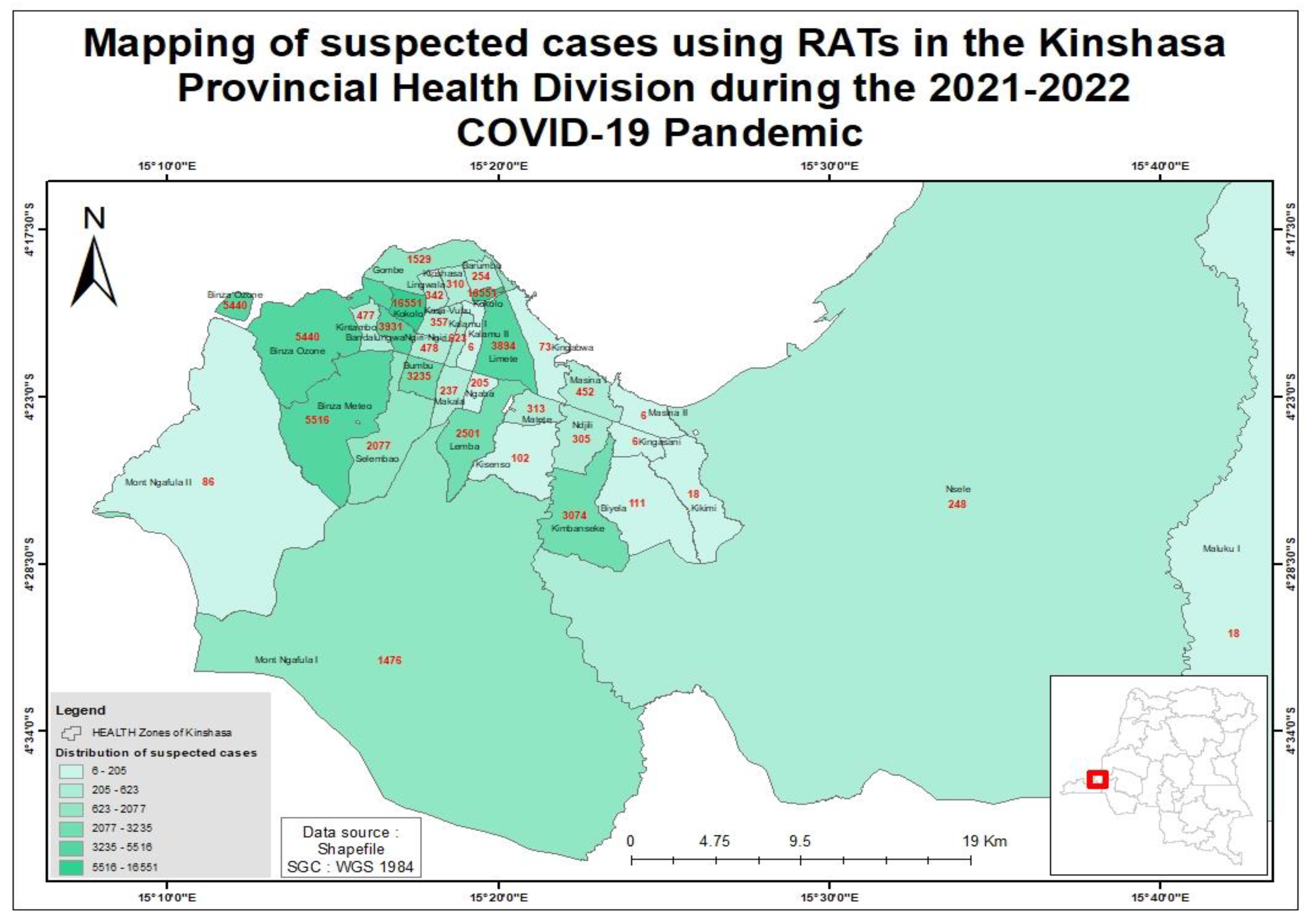
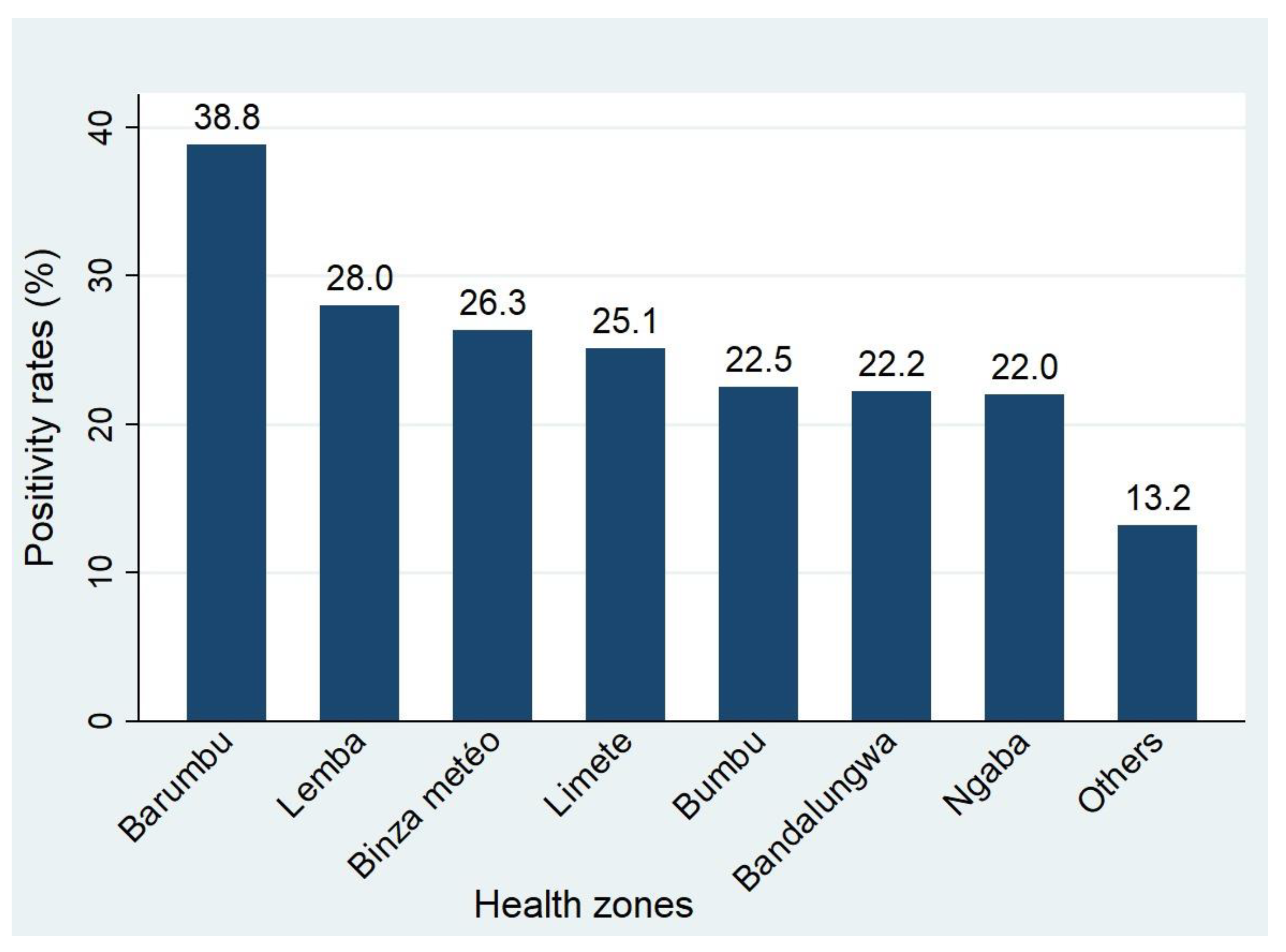
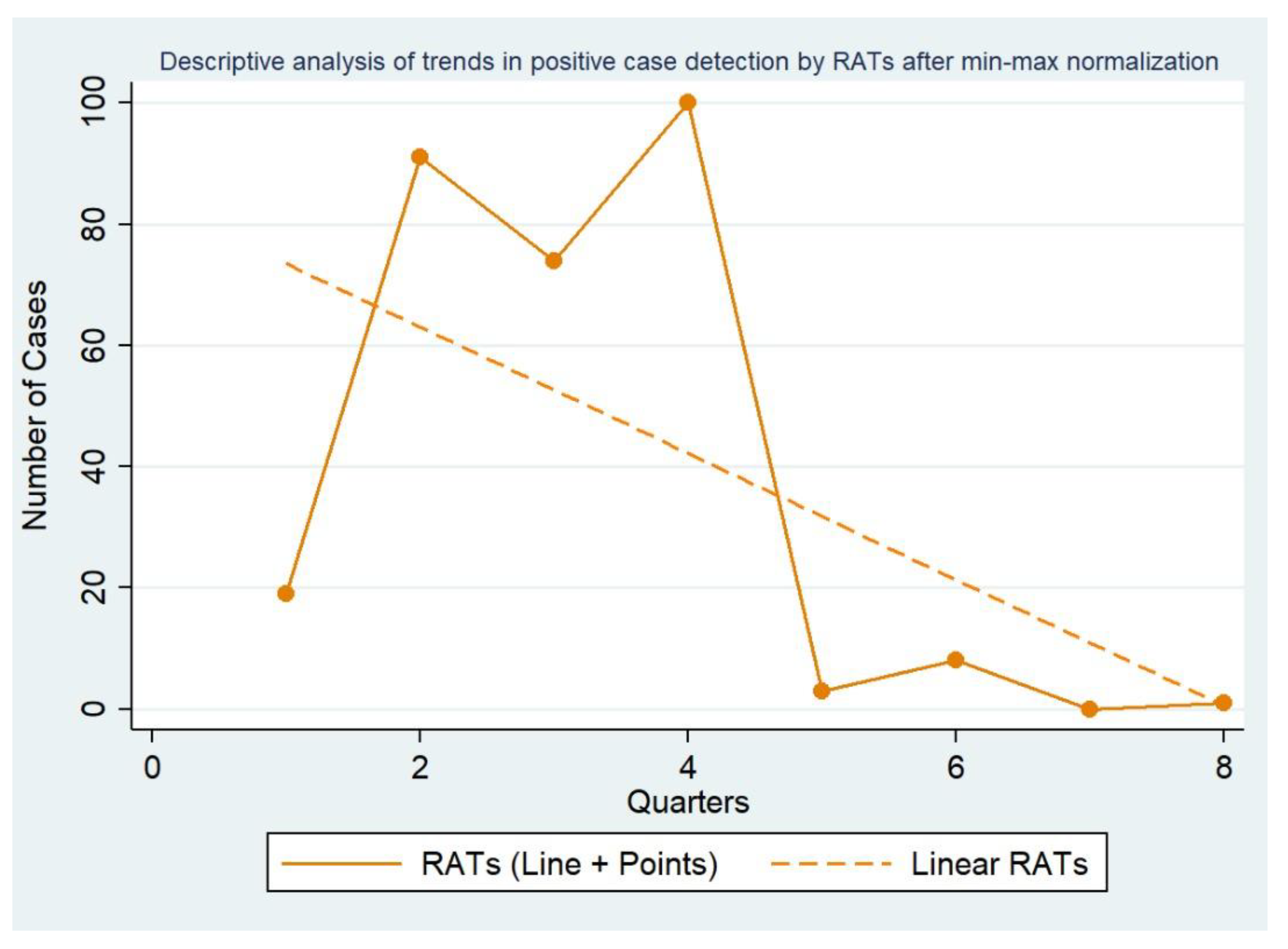
3. Results
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.-L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2022, 20, 141–154. [Google Scholar] [CrossRef]
- Otshudiema, J.O.; Folefack, G.L.T.; Nsio, J.M.; Mbala-Kingebeni, P.; Kakema, C.H.; Kosianza, J.B.; et al. Epidemiological Comparison of Four COVID-19 Waves in the Democratic Republic of the Congo, March 2020–January 2022. J. Epidemiol. Glob. Health 2022, 12, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Mansourabadi, A.H.; Sadeghalvad, M.; Mohammadi-Motlagh, H.R.; Amirzargar, A. Serological and Molecular Tests for COVID-19: a recent update. Iran. J. Immunol. 2021, 18, 13–33. [Google Scholar]
- Jayamohan, H.; Lambert, C.J.; Sant, H.J.; Jafek, A.; Patel, D.; Feng, H.; et al. SARS-CoV-2 pandemic: a review of molecular diagnostic tools including sample collection and commercial response with associated advantages and limitations. Anal. Bioanal. Chem. 2021, 413, 49–71. [Google Scholar] [CrossRef]
- Organisation Mondiale de la Santé. Tests diagnostiques pour le dépistage du SARS-CoV-2 : Orientations provisoires 11 septembre 2020; World Health Organization: Geneva, Switzerland, 2020; Available online: https://iris.who.int/handle/10665/335724 (accessed on 15 April 2025).
- Organisation Mondiale de la Santé. Détection des antigènes à l’aide de tests immunologiques rapides pour le diagnostic de l’infection à SARS-CoV-2, Orientations provisoires 11 septembre 2020; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/publications/i/item/antigen-detection-in-the-diagnosis-of-sars-cov-2infection-using-rapid-immunoassays (accessed on 29 August 2025).
- World Health Organization. Antigen Detection in the Diagnosis of SARS-CoV-2 Infection; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Clop, G. Impact du test antigénique de dépistage rapide du SARS-CoV2 sur le temps de passage des patients atteints de Covid-19 dans un service d’urgence. Ph.D. Thesis, Université de Bordeaux, Bordeaux, France, 2022; pp. 1–28. [Google Scholar]
- Mukadi-Bamuleka, D.; Bulabula-Penge, J.; Jacobs, B.K.M.; De Weggheleire, A.; Edidi-Atani, F.; Mambu-Mbika, F.; et al. Head-to-head comparison of diagnostic accuracy of four Ebola virus disease rapid diagnostic tests versus GeneXpert® in eastern Democratic Republic of the Congo outbreaks: a prospective observational study. EBioMedicine 2023, 91, 104568. [Google Scholar] [CrossRef]
- Mukadi-Bamuleka, D.; Bulabula-Penge, J.; De Weggheleire, A.; Jacobs, B.K.M.; Edidi-Atani, F.; Mambu-Mbika, F.; et al. Field performance of three Ebola rapid diagnostic tests used during the 2018–20 outbreak in the eastern Democratic Republic of the Congo: a retrospective, multicenter observational study. Lancet Infect. Dis. 2022, 22, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Organisation Mondiale de la Santé. Orientations pour la lutte anti-infectieuse dans les établissements de soins de longue durée dans le contexte de la COVID-19, Orientations provisoires 21 mars 2020; World Health Organization: Geneva, Switzerland, 2020; Available online: https://apps.who.int/iris/bitstream/handle/10665/331642/WHO-2019-nCoV-IPC_long_term_care-2020.1-fre.pdf (accessed on 29 August 2025).
- Organisation Mondiale de la Santé. Orientations pour la lutte anti-infectieuse dans les établissements de soins de longue durée dans le contexte de la Covid-19, orientations provisoires 8 janvier 2021; World Health Organization: Geneva, Switzerland, 2021; Available online: https://iris.who.int/handle/10665/338935 (accessed on 15 April 2025).
- World Health Organization. Prevention, Identification and Management of Health Worker Infection in the Context of COVID-19; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/publications/i/item/10665-336265 (accessed on 15 April 2025).
- République Démocratique du Congo, Ministère de la Santé, Hygiène et Prévoyance sociale, Comité Multisectoriel de la lutte contre la COVID-19 RDC. Procédures opérationnelles de la Surveillance Épidémiologique de la COVID-19 centrée sur la Recherche active des cas, de la détection précoce des alertes et des cas suspects, à l’isolement et prise en charge médicale rapide des cas confirmés et au suivi des contacts, Décembre 2020; Ministère de la Santé: Kinshasa, Democratic Republic of the Congo, 2020. [Google Scholar]
- World Health Organization. WHO COVID-19 Case Definition; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-Surveillance_Case_Definition-2020.2 (accessed on 28 May 2025).
- Agulló, V.; Fernandez-González, M.; Ortiz de la Tabla, V.; Gonzalo-Jiménez, N.; García, J.A.; Masiá, M.; Gutiérrez, F. Evaluation of the rapid antigen test Panbio COVID-19 in saliva and nasal swabs in a population-based point-of-care study. J. Infect. 2020, 82, 186–230. [Google Scholar] [CrossRef]
- Alemany, A.; Baró, B.; Ouchi, D.; Rodó, P.; Ubals, M.; Corbacho-Monné, M.; et al. Analytical and clinical performance of the panbio COVID-19 antigen-detecting rapid diagnostic test. J. Infect. 2021, 82, 186–230. [Google Scholar] [CrossRef]
- Haute Autorité de Santé. Revue rapide sur les tests de détection antigénique du virus SARS-CoV-2; Haute Autorité de Santé: Saint-Denis, France, 2020; Available online: https://www.has-sante.fr (accessed on 15 June 2025).
- Haute Autorité de Santé. Évaluation de l’intérêt des tests antigéniques rapides sur le prélèvement nasal pour la détection du virus SARS-CoV-2 (Méta-analyse); Haute Autorité de Santé: Saint-Denis, France, 2021; Available online: https://www.has-sante.fr (accessed on 15 June 2025).
- Hardy, É.J.L.; Flori, P. Spécificités épidémiologiques de la COVID-19 en Afrique : préoccupation de santé publique actuelle ou future ? Ann. Pharm. Fr. 2020, 79, 216–226. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune response. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Pison, G.; Meslé, F. La Covid-19 plus meurtrière pour les hommes que pour les femmes. Popul. Sociétés 2022, 598, 1–5. [Google Scholar] [CrossRef]
- Bornfleth, I.; Kutschka, A.; Gardemann, B.; Isermann, B.; Goette, A. Protective regulation of the ACE2/ACE gene expression by estrogen in human atrial tissue from elderly men. Exp. Biol. Med. 2017, 242, 1412–1423. [Google Scholar] [CrossRef]
- Gebhard, C.; Regitz-Zagrosek, V.; Neuhauser, H.K.; Morgan, R.; Klein, S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex Differ. 2020, 11, 29. [Google Scholar] [CrossRef]
- Plaçais, L.; Richier, Q. COVID-19 : caractéristiques cliniques, biologiques et radiologiques chez l’adulte, la femme enceinte et l’enfant. Une mise au point de la pandémie. Rev. Med. Interne 2020, 41, 308–318. [Google Scholar] [CrossRef]
- Chow, E.J.; Schwartz, N.G.; Tobolowsky, F.A.; et al. Symptom Screening at Illness Onset of Health Care Personnel With SARS-CoV-2 Infection in King County, Washington. JAMA 2020, 323, 2087–2089. [Google Scholar] [CrossRef]
- Yombi, J.C.; De Greef, J.; Marsin, A.S.; Simon, A.; Rodriguez-Villalobos, H.; Penaloza, A.; Belkhir, L. Symptom-based screening for COVID-19 in healthcare workers: the importance of fever. J. Hosp. Infect. 2020, 105, 428–429. [Google Scholar] [CrossRef]
- Smith, N.; Goncalves, P.; Charbit, B.; Grzelak, L.; Beretta, M.; Planchais, C.; et al. Distinct systemic and mucosal immune responses during acute SARS-CoV-2 infection. Nat. Immunol. 2021, 22, 1428–1439. [Google Scholar] [CrossRef]
- Nkwembe, E.N.; Ituneme, Y.N.; Mufwaya, G.M.; Lebo, L.N.; Matondo, M.K.; Yalungu, H.M.; et al. Impact of COVID-19 on the Circulation of Respiratory Viruses in Kinshasa: Analysis of the Prevalence and Viral Etiologies of Acute Respiratory Infections. Open J. Epidemiol. 2024, 14, 669–682. [Google Scholar] [CrossRef]
- Liu, Y.; Gayle, A.A.; Wilder-Smith, A.; Rocklöv, J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J. Travel Med. 2020, 27, taaa021. [Google Scholar] [CrossRef]
- Billah, M.A.; Miah, M.M.; Khan, M.N. Reproductive number of coronavirus: A systematic review and meta-analysis based on global level evidence. PLoS ONE 2020, 15, e0242128. [Google Scholar] [CrossRef]
- Mai, N.T.; Tran, G.T.H.; Dang, A.H.; Cao, P.T.B.; Nguyen, T.T.; Pham, H.T.L.; Vu, T.T.T.; Dong, H.V.; Huynh, L.T.M. Comparison of the Basic Reproduction Numbers for COVID-19 through Four Waves of the Pandemic in Vietnam. Int. J. Transl. Med. 2023, 3, 1–11. [Google Scholar] [CrossRef]
- Dozol, J.-M. Place des tests antigéniques dans le dépistage et la stratégie diagnostique de la COVID-19 : retour d’expérience de pharmaciens d’officine de la Région SUD. Ph.D. Thesis, Université de Bordeaux, Bordeaux, France, 2025. Available online: https://dumas.ccsd.cnrs.fr/dumas-04957403. Available online: https://dumas.ccsd.cnrs.fr/dumas-04957403 (accessed on 15 June 2025).
- Ganser, I.; Buckeridge, D.L.; Heffernan, J.; Prague, M.; Thiébaut, R. Estimating the population effectiveness of interventions against COVID-19 in France: A modelling study. Epidemics 2024, 46, 100740. [Google Scholar] [CrossRef]
- Wang, J.; Tang, K.; Feng, K.; Lin, X.; Lv, W.; Chen, K.; Wang, F. Impact of temperature and relative humidity on the transmission of COVID-19: a modelling study in China and the United States. BMJ Open 2021, 11, e043863. [Google Scholar] [CrossRef]
- Zheng, C.; Shao, W.; Chen, X.; Zhang, B.; Wang, G.; Zhang, W. Real-world effectiveness of COVID-19 vaccines: a literature review and meta-analysis. Int. J. Infect. Dis. 2022, 114, 252–260. [Google Scholar] [CrossRef]
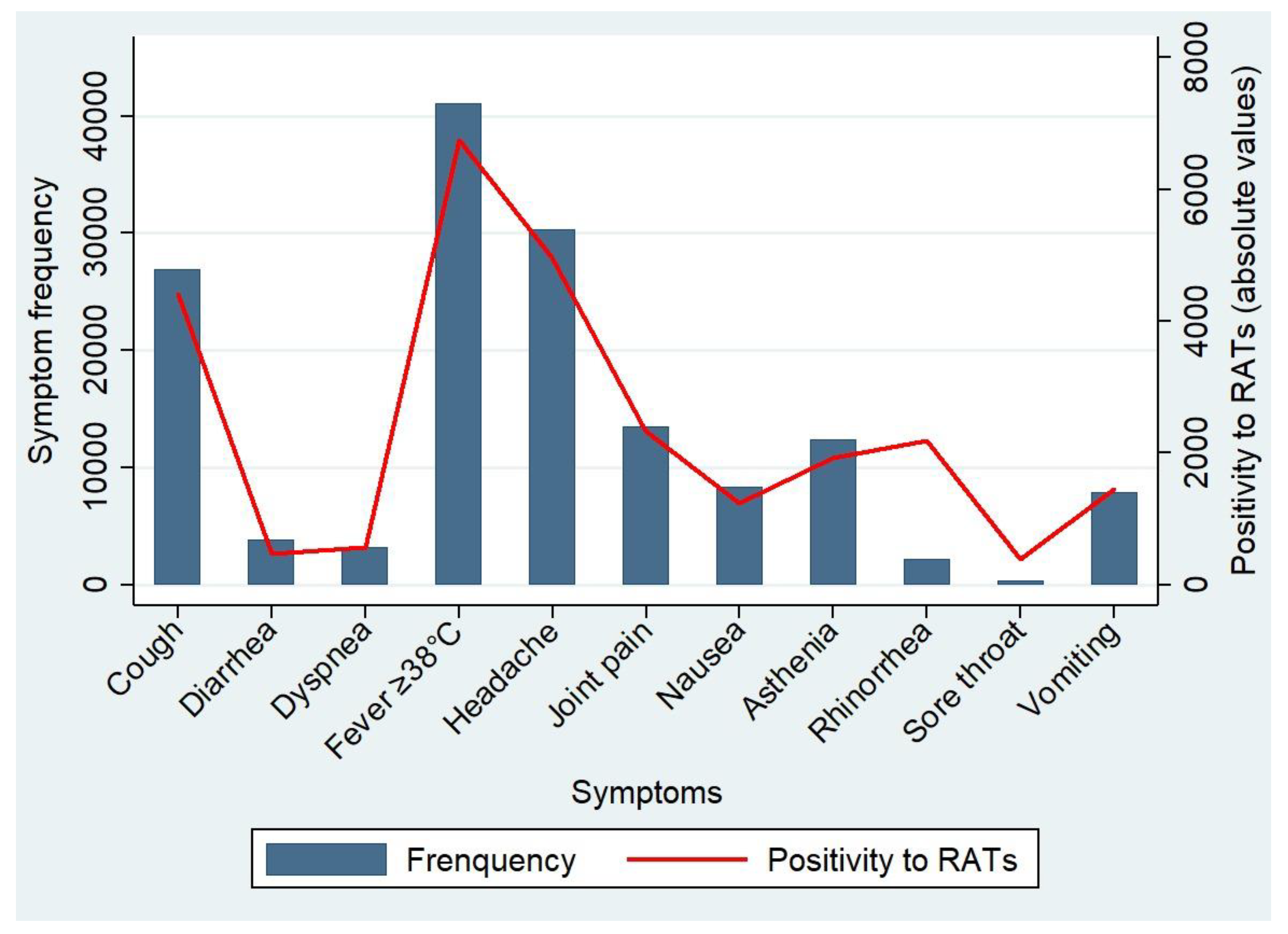
| Variables |
N=54432 | Negative (%) | Positive (%) |
|---|---|---|---|
| Age (years), mean age: 36.9±15.9 | |||
| ≤21 | 6143 | 4972 (80.9) | 1171 (19.1) |
| 21-40 | 27221 | 22435 (82.4) | 4786 (17.6) |
| 41-60 | 16056 | 13581 (84.6) | 2475 (15.4) |
| ≥60 | 5012 | 4318 (86.2) | 694 (13.8) |
| Sex | |||
| M | 29832 | 24923 (83.5) | 4909 (16.5) |
| F | 24600 | 20383 (82.9) | 4217 (17.1) |
| Variables | N = 54432 | Negative [n (%)] | Positive [n (%)] | Chi-square | p-value |
|---|---|---|---|---|---|
| Symptoms | |||||
| Sore throat | |||||
| No | 51453 | 42710 (83) | 8743 (17) | 34.51 | < 0.001 |
| Yes | 2979 | 2596 (87.1) | 383 (12.9) | ||
| Rhinorrhea | |||||
| No | 42097 | 35158 (83.5) | 6939 (16.5) | 10.63 | 0.001 |
| Yes | 12335 | 10148 (82.3) | 2187 (17.7) | ||
| Cough | |||||
| No | 27501 | 22787 (82.9) | 4714 (17.1) | 5.61 | 0.018 |
| Yes | 26931 | 22519 (83.6) | 4412 (16.4) | ||
| Dyspnea | |||||
| No | 51267 | 42708 (83.3) | 8559 (16.7) | 3.18 | 0.075 |
| Yes | 3165 | 2598 (821) | 567 (17.9) | ||
| Physical asthenia | |||||
| No | 42024 | 34817 (82.9) | 7207 (17.1) | 19.47 | < 0.001 |
| Yes | 12408 | 10489 (84.5) | 1919 (15.5) | ||
| Vomiting | |||||
| No | 46550 | 38866 (83.5) | 7684 (16.5) | 15.44 | < 0.001 |
| Yes | 7882 | 6440 (81.7) | 1442 (18.3) | ||
| Nausea | |||||
| No | 46059 | 38167 (82.9) | 7892 (17.1) | 29.16 | < 0.001 |
| Yes | 8373 | 7139 (85.3)) | 1234 (14.7) | ||
| Diarrhea | |||||
| No | 50516 | 41852 (82.8) | 8664 (17.2) | 74.63 | < 0.001 |
| Yes | 3916 | 3454 (88.2) | 462 (11.8) | ||
| Headache | |||||
| No | 24125 | 19958 (82.7) | 4167 (17.3) | 7.97 | 0.005 |
| Yes | 30307 | 25348 (83.6) | 4959 (16.4) | ||
| Joint pain | |||||
| No | 40925 | 33930 (82.9) | 6995 (17.1) | 12.59 | < 0.001 |
| Yes | 13507 | 11376 (84.2) | 2131 (15.8) | ||
| Fever ≥38°C | |||||
| No | 13353 | 10986 (82.3) | 2367 (17.7) | 11.70 | < 0.001 |
| Yes | 41079 | 34320 (83.5) | 6759 (16.5) | ||
| History of close contact with a symptomatic person | |||||
| Sex Female | |||||
| No | 18939 | 16488 (87.1) | 2451 (12.9) | 1022.46 | < 0.001 |
| Yes | 5661 | 3895 (68.8) | 1766 (31.2) | ||
| Sex Male | |||||
| No | 22488 | 19831 (88.2) | 2657 (11.8) | 1430.75 | < 0.001 |
| Yes | 7344 | 5092 (69.3) | 2252 (30.7) | ||
| History of contact with a confirmed COVID-19 case | |||||
| Sex Female | |||||
| No | 18309 | 16071 (87.8) | 2238 (12.2) | 1219.53 | 0.001 |
| Yes | 6291 | 4312 (68.5) | 1979 (31.5) | ||
| Sex Male | |||||
| No | 23526 | 20561 (87.4) | 2965 (12.6) | 1201.47 | 0.001 |
| Yes | 6306 | 4362 (69.2) | 1944 (30.8) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




