Submitted:
21 November 2025
Posted:
24 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Materials
Preparation of PDA Vesicle and PDA/L64 Suspensions
Preparation of PDA/L64/α-CD Mixtures
Isothermal Titration Calorimetry Experiments
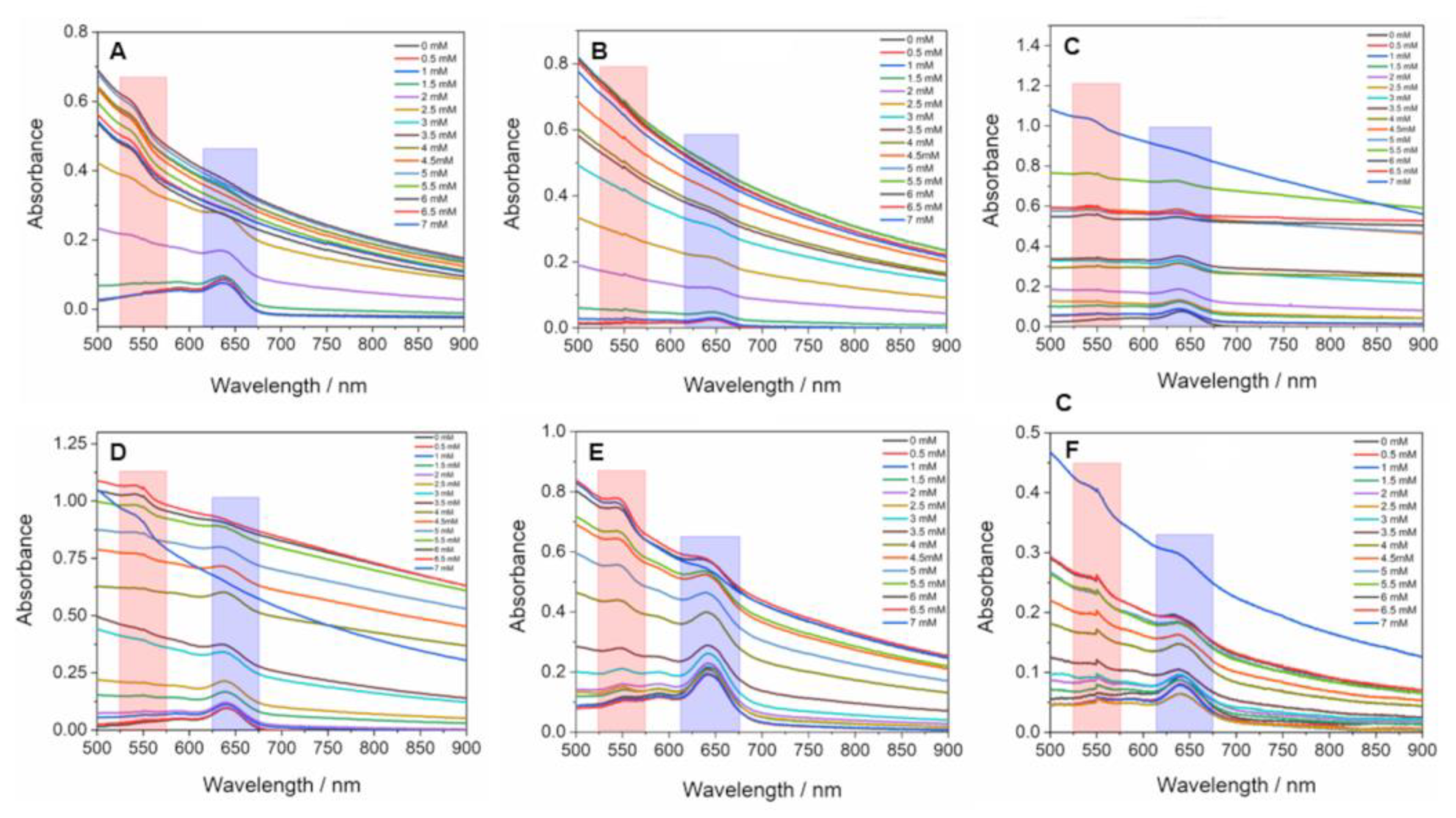
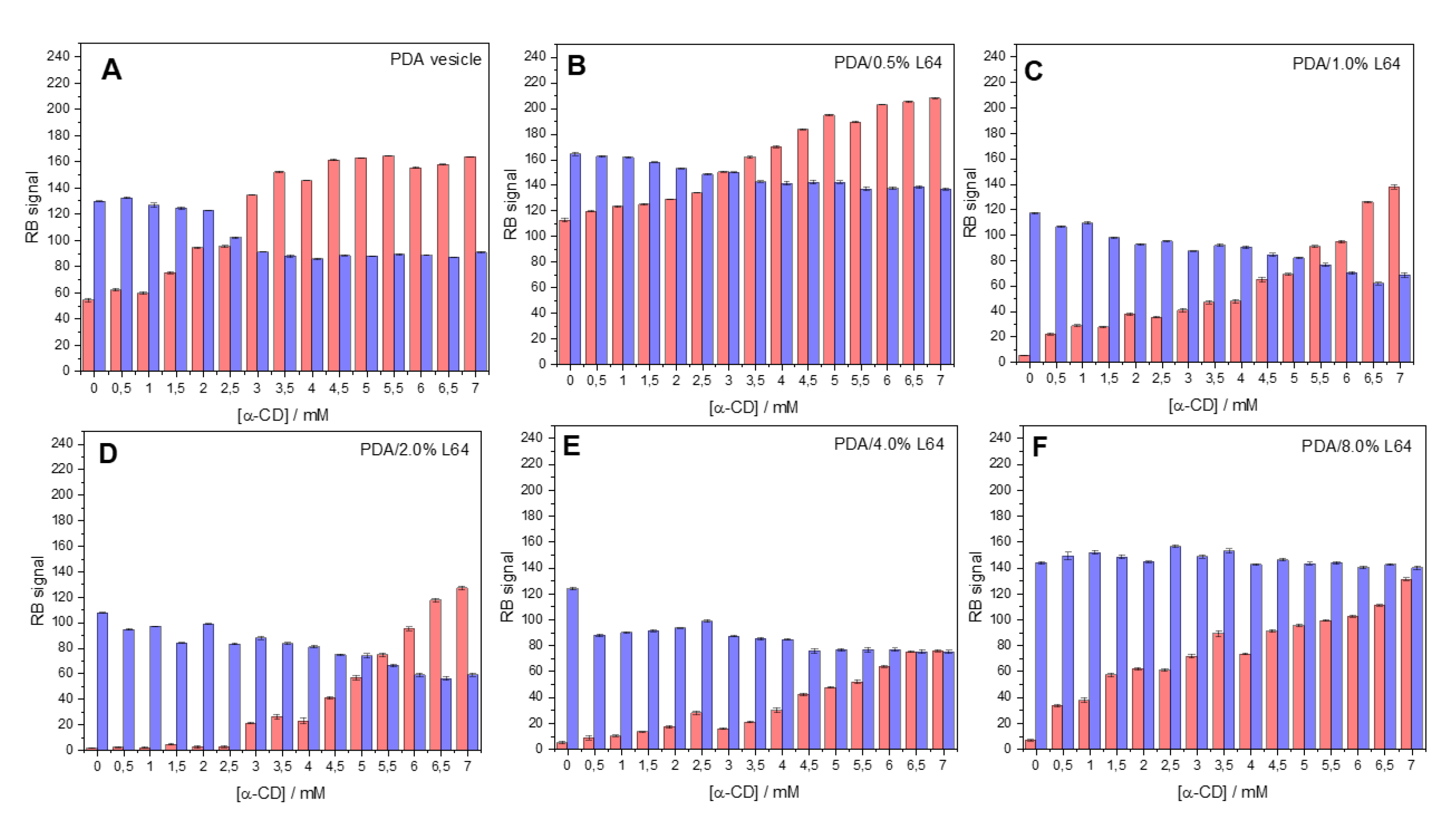
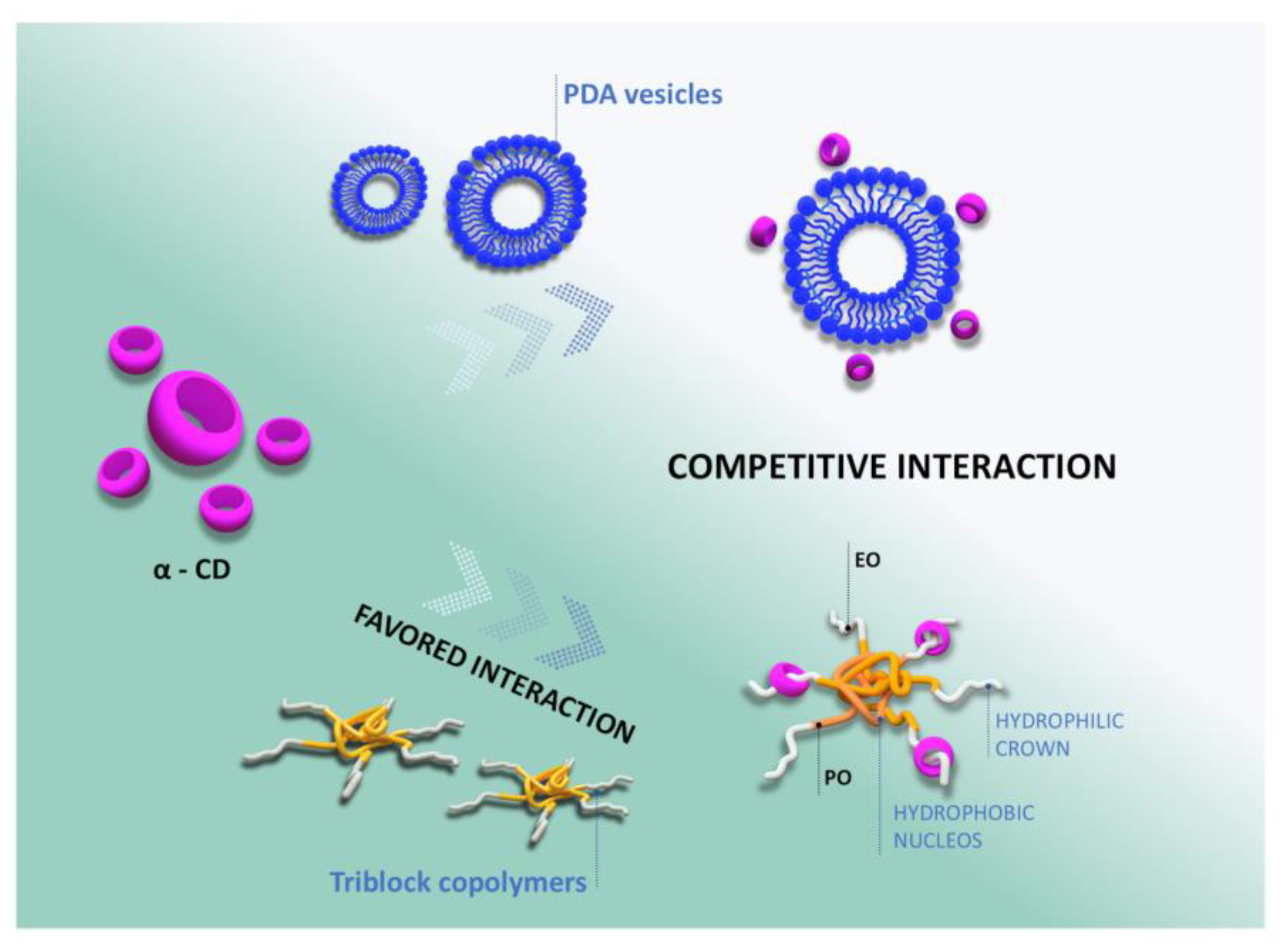
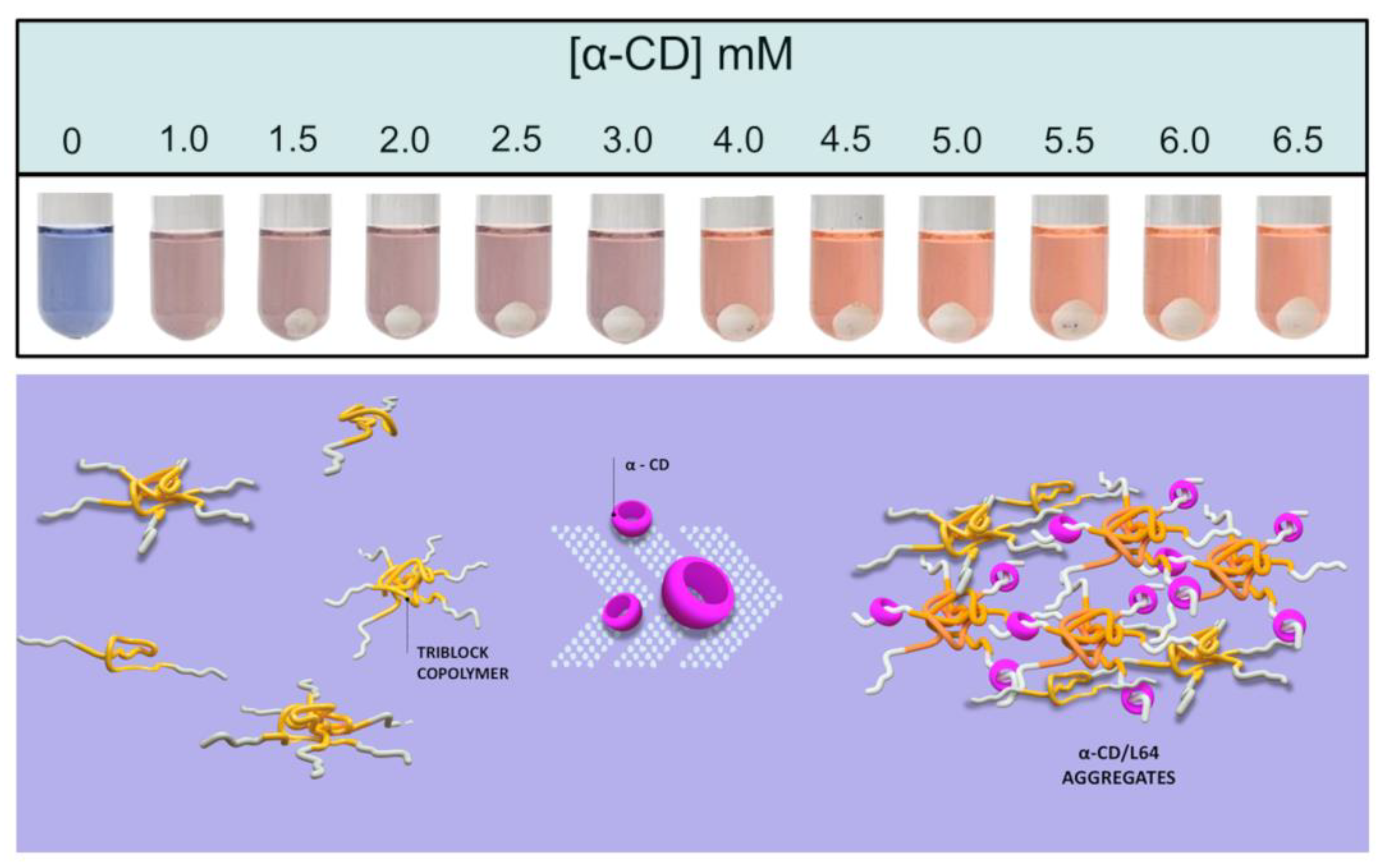
3. Results and Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PDAs | Polydiacetylenes |
| UV | Ultraviolet |
| EO | Ethylene oxide |
| PO | Propylene oxide |
| CD | Cyclodextrin |
| ICs | Inclusion complexes |
| L64 | Poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol) |
| MW | Molecular weight |
| PCDA | 10,12-pentacosadiynoic acid |
| RGB | Red–Green–Blue color model |
| ROIs | Regions of interest |
| UV–Vis | Ultraviolet–Visible spectroscopy |
| ITC | Isothermal titration calorimetry |
| n | Stoichiometric coefficients |
| K | Association constant |
| VP-ITC | Variable-Pressure Isothermal Titration Calorimeter |
| CR | Colorimetric response |
| CMC | Critical micelle concentration |
| PPO | Polypropylene oxide |
| PEO | Polyethylene oxide |
Appendix A
Appendix A.1
References
- Ortega, P.F.R.; Galvao, B.R.L.; de Oliveira, P.S.C.; Bastos, G.A.A.; Bernardes, M.R.F.; Lavall, R.L.; Trigueiro, J.P.C. Thermochromism in Polydiacetylene/Poly(vinyl alcohol) Hydrogels Obtained by the Freeze-Thaw Method: A Theoretical and Experimental Study. Ind. Eng. Chem. Res. 2021, 60, 13243–13252.
- Huo, J.; Hu, Z.; He, G.; Hong, X.; Yang, Z.; Luo, S.; Ye, X.; Li, Y.; Zhang, Y.; Zhang, M.; Chen, H.; Fan, T.; Zhang, Y.; Xiong, B.; Wang, Z.; Zhu, Z.; Chen, D. High temperature thermochromic polydiacetylenes: Design and colorimetric properties. Appl. Surf. Sci. 2017, 423, 951–956. [CrossRef]
- Wilk-Kozubek, M.; Potaniec, B.; Gazinska, P.; Cybinska, J. Exploring the Origins of Low-Temperature Thermochromism in Polydiacetylenes. Polymers 2024, 16. [CrossRef]
- Singh, Y.; Jayaraman, N. Visual Detection of pH and Biomolecular Interactions at Micromolar Concentrations Aided by a Trivalent Diacetylene-Based Vesicle. Macromol. Chem. Phys. 2017, 218. [CrossRef]
- Beliktay, G.; Shaikh, T.; Koca, E.; Cingil, H.E. Effect of UV Irradiation Time and Headgroup Interactions on the Reversible Colorimetric pH Response of Polydiacetylene Assemblies. ACS Omega 2023, 8, 37213–37224.
- Weston, M.; Tjandra, A.D.; Chandrawati, R. Tuning chromatic response, sensitivity, and specificity of polydiacetylene-based sensors. Polym. Chem. 2020, 11, 166–183. [CrossRef]
- Kim, T.; Moon, D.; Park, J.H.; Yang, H.; Cho, S.; Park, T.H.; Ahn, D.J. Visual detection of odorant geraniol enabled by integration of a human olfactory receptor into polydiacetylene/lipid nano-assembly. Nanoscale 2019, 11, 7582–7587. [CrossRef]
- Charych, D.; Nagy, J.; Spevak, W.; Bednarski, M. Direct colorimetric detection of a receptor-ligand interaction by a polymerized bilayer assembly. Science 1993, 261, 585–588. [CrossRef]
- Cho, J.; Woo, S.; Ahn, D.; Ahn, K.; Lee, H.; Kim, J. Cyclodextrin-induced color changes in polymerized diacetylene Langmuir-Schaefer films. Chem. Lett. 2003, 32, 282–283. [CrossRef]
- Seto, K.; Hosoi, Y.; Furukawa, Y. Raman spectra of Langmuir-Blodgett and Langmuir-Schaefer films of polydiacetylene prepared from 10,12-pentacosadiynoic acid. Chem. Phys. Lett. 2007, 444, 328–332. [CrossRef]
- Geiger, E.; Hug, P.; Keller, B. Chromatic transitions in polydiacetylene Langmuir-Blodgett films due to molecular recognition at the film surface studied by spectroscopic methods and surface analysis. Macromol. Chem. Phys. 2002, 203, 2422–2431. [CrossRef]
- Iimori, Y.; Onodera, T.; Kasai, H.; Mitsuishi, M.; Miyashita, T.; Oikawa, H. Fabrication of pseudo single crystalline thin films composed of polydiacetylene nanofibers and their optical properties. Opt. Mater. Express 2017, 7, 2218–2223.
- Velarde, M.G.; Chetverikov, A.P.; Ebeling, W.; Wilson, E.G.; Donovan, K.J. On the electron transport in polydiacetylene crystals and derivatives. EPL 2014, 106.
- Lebegue, E.; Farre, C.; Jose, C.; Saulnier, J.; Lagarde, F.; Chevalier, Y.; Chaix, C.; Jaffrezic-Renault, N. Responsive Polydiacetylene Vesicles for Biosensing Microorganisms. Sensors 2018, 18. [CrossRef]
- Miller, J.S.; Finney, T.J.; Ilagan, E.; Frank, S.; Chen-Izu, Y.; Suga, K.; Kuhl, T.L. Fluorogenic Biosensing with Tunable Polydiacetylene Vesicles. Biosensors 2025, 15.
- Yu, Z.; MuYu, C.; Xu, H.; Zhao, J.; Yang, G. Recent progress in the design of conjugated polydiacetylenes with reversible thermochromic performance: a review. Polym. Chem. 2023, 14, 2266–2290. [CrossRef]
- Jelinek, R.; Ritenberg, M. Polydiacetylenes – recent molecular advances and applications. RSC Adv. 2013, 3, 21192–21201. [CrossRef]
- Nguyen, L.H.; Oveissi, F.; Chandrawati, R.; Dehghani, F.; Naficy, S. Naked-Eye Detection of Ethylene Using Thiol-Functionalized Polydiacetylene-Based Flexible Sensors. ACS Sens. 2020, 5, 1921–1928. [CrossRef]
- Guo, J.; Yang, L.; Zhu, L.; Chen, D. Selective detection of metal ions based on nanocrystalline ionochromic polydiacetylene. Polymer 2013, 54, 743–749. [CrossRef]
- Tjandra, A.D.; Chandrawati, R. Polydiacetylene/copolymer sensors to detect lung cancer breath volatile organic compounds. RSC Appl. Polym. 2024, 2, 1043–1056.
- Jang, H.; Jeon, J.; Shin, M.; Kang, G.; Ryu, H.; Kim, S.M.; Jeon, T.-J. Polydiacetylene (PDA) Embedded Polymer-Based Network Structure for Biosensor Applications. Gels 2025, 11. [CrossRef]
- Dias Ferreira, G.M.; Dias Ferreira, G.M.; Hespanhol, M.C.; Rezende, J.P.; Pires, A.C.S.; Ortega, P.F.R.; Mendes da Silva, L.H. A simple and inexpensive thermal optic nanosensor formed by triblock copolymer and polydiacetylene mixture. Food Chem. 2018, 241, 358–363. [CrossRef]
- Morais, D.C.; Vieira, B.B.M.; Carvalho, M.C.; Miguez, F.B.; Lopes, J.F.; Sousa, F.B.D. Thermodynamic investigation of biperiden hydrochloride and cyclodextrins supramolecular systems. Chem. Phys. Lett. 2024, 851. [CrossRef]
- Passos, J.J.; De Sousa, F.B.; Mundim, I.M.; Bonfim, R.R.; Melo, R.; Viana, A.F.; Stolz, E.D.; Borsoi, M.; Rates, S.M.K.; Sinisterra, R.D. Double continuous injection preparation method of cyclodextrin inclusion compounds by spray drying. Chem. Eng. J. 2013, 228, 345–351. [CrossRef]
- Passos, J.J.; De Sousa, F.B.; Mundim, I.M.; Bonfim, R.R.; Melo, R.; Viana, A.F.; Stolz, E.D.; Borsoi, M.; Rates, S.M.K.; Sinisterra, R.D. In vivo evaluation of the highly soluble oral β-cyclodextrin–Sertraline supramolecular complexes. Int. J. Pharm. 2012, 436, 478–485. [CrossRef]
- Utzeri, G.; Matias, P.M.C.; Murtinho, D.; Valente, A.J.M. Cyclodextrin-Based Nanosponges: Overview and Opportunities. Front. Chem. 2022, 10. [CrossRef]
- Roy, I.; Stoddart, J.F. Cyclodextrin Metal-Organic Frameworks and Their Applications. Acc. Chem. Res. 2021, 54, 1440–1453.
- Valente, A.J.M.; Soderman, O. The formation of host–guest complexes between surfactants and cyclodextrins. Adv. Colloid Interface Sci. 2014, 205, 156–176.
- Champaiboon, T.; Tumcharern, G.; Potisatityuenyong, A.; Wacharasindhu, S.; Sukwattanasinitt, M. A polydiacetylene multilayer film for naked eye detection of aromatic compounds. Sens. Actuators B Chem. 2009, 139, 532–537. [CrossRef]
- Martínez, M.; Ballesteros, S.; Almarza, E.; de la Torre, C.; Búa, S. Acute nitrobenzene poisoning with severe associated methemoglobinemia: Identification in whole blood by GC-FID and GC-MS. J. Anal. Toxicol. 2003, 27, 221–225.
- Tchieno, F.M.M.; Tonle, I.K. p-Nitrophenol determination and remediation: an overview. Rev. Anal. Chem. 2018, 37. [CrossRef]
- de Castro, K.C.; Coco, J.C.; dos Santos, E.M.; Ataide, J.A.; Martinez, R.M.; Monteiro do Nascimento, M.H.; Prata, J.; Lopes da Fonte, P.R.M.; Severino, P.; Mazzola, P.G.; Baby, A.R.; Souto, E.B.; de Araujo, D.R.; Lopes, A.M. Pluronic® triblock copolymer-based nanoformulations for cancer therapy: A 10-year overview. J. Control. Release 2023, 353, 802–822.
- Liu, D.; Yang, M.; Wang, D.; Jing, X.; Lin, Y.; Feng, L.; Duan, X. DPD Study on the Interfacial Properties of PEO/PEO-PPO-PEO/PPO Ternary Blends: Effects of Pluronic Structure and Concentration. Polymers 2021, 13. [CrossRef]
- Mayer, B.; Klein, C.; Topchieva, I.; Köhler, G. Selective assembly of cyclodextrins on poly(ethylene oxide)-poly(propylene oxide) block copolymers. J. Comput.-Aided Mol. Des. 1999, 13, 373–383.
- Almgren, M.; Brown, W.; Hvidt, S. Self-aggregation and phase-behavior of poly(ethylene oxide) poly(propylene oxide) poly(ethylene oxide) block-copolymers in aqueous solution. Colloid Polym. Sci. 1995, 273, 2–15.
- Mata, J.; Majhi, P.; Guo, C.; Liu, H.; Bahadur, P. Concentration, temperature, and salt-induced micellization of a triblock copolymer Pluronic L64 in aqueous media. J. Colloid Interface Sci. 2005, 292, 548–556. [CrossRef]
- Marinov, G.; Michels, B.; Zana, R. Study of the state of the triblock copolymer poly(ethylene oxide) poly(propylene oxide) poly(ethylene oxide) L64 in aqueous solution. Langmuir 1998, 14, 2639–2644. [CrossRef]
- Pradal, C.; Jack, K.S.; Grondahl, L.; Cooper-White, J.J. Gelation kinetics and viscoelastic properties of Pluronic and α-cyclodextrin-based pseudopolyrotaxane hydrogels. Biomacromolecules 2013, 14, 3780–3792.
- Tsai, C.-C.; Zhang, W.-B.; Wang, C.-L.; Van Horn, R.M.; Graham, M.J.; Huang, J.; Chen, Y.; Guo, M.; Cheng, S.Z.D. Evidence of formation of site-selective inclusion complexation between β-cyclodextrin and poly(ethylene oxide)-block-poly(propylene oxide)-block-poly(ethylene oxide) copolymers. J. Chem. Phys. 2010, 132.
- Yang, C.; Ni, X.; Li, J. Synthesis of polyrotaxanes consisting of multiple α-cyclodextrin rings threaded on reverse Pluronic PPO-PEO-PPO triblock copolymers based on block-selected inclusion complexation. Eur. Polym. J. 2009, 45, 1570–1579. [CrossRef]
- Meira, L.H.R.; Soares, G.A.B.; Bonomini, H.I.M.; Lopes, J.F.; De Sousa, F.B. Thermodynamic compatibility between cyclodextrin supramolecular complexes and surfactant. Int. J. Pharm. 2018, 544, 203–212. [CrossRef]

| Supramolecular system | K | ΔHo / kJ mol-1 | TΔSo / kJ mol-1 | ΔGo / kJ mol-1 |
|---|---|---|---|---|
| α-CD with L64 | 11 300 ± 1.250 | -0.73 | 21.05 | -21.78 |
| α-CD with PDA | 4 000 ± 353 | -0.94 | 19.60 | -21.54 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




