Submitted:
15 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Rare Genetic Diseases
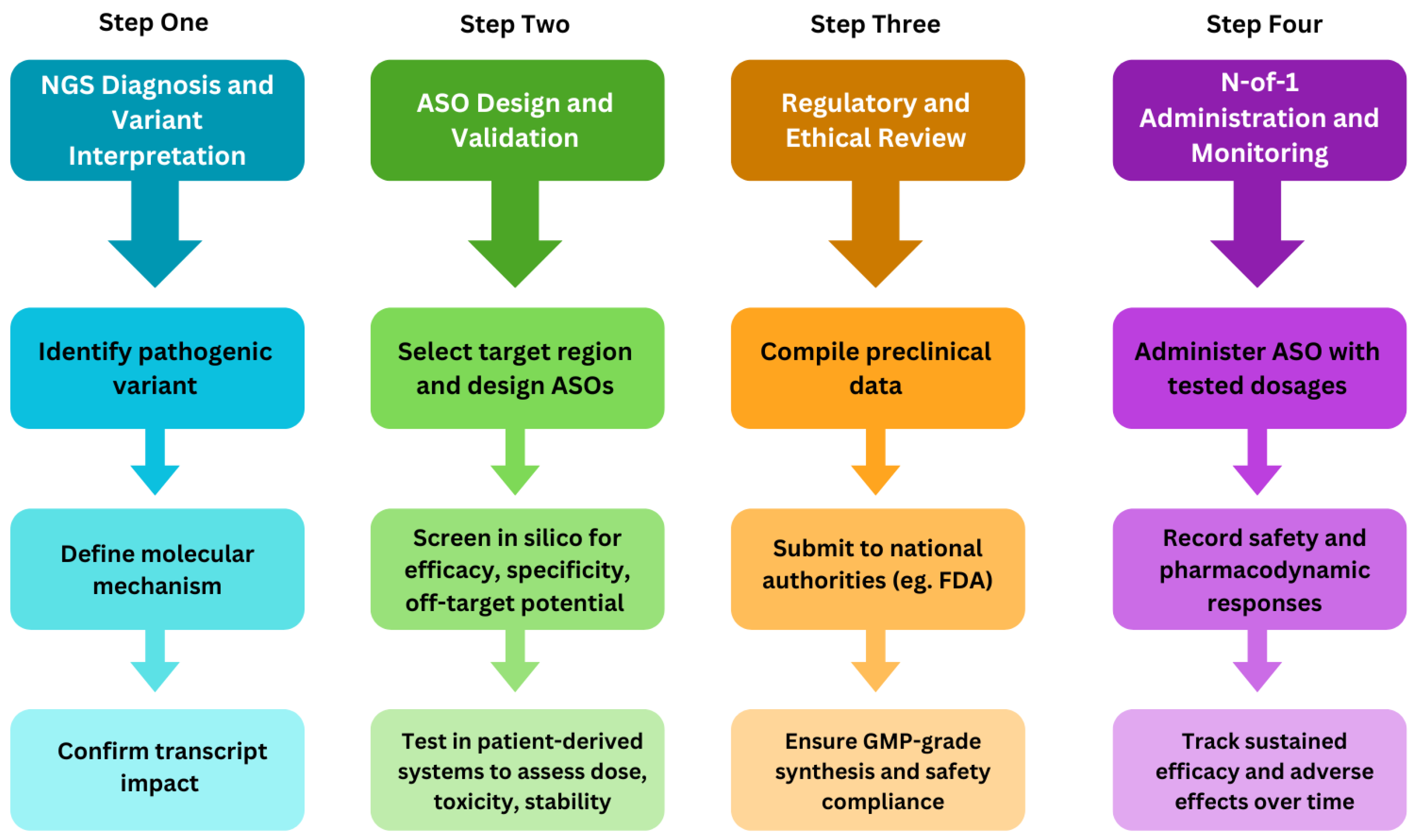
1.2. From Diagnosis to Design
2. NGS for Therapy Selection in Rare Diseases
2.1. NGS Assays and Detection
2.2. Mapping Variant Classes to ASO Strategies
2.3. Assay Limitations and Solutions
3. Antisense Oligonucleotide Therapies
3.1. Mechanisms of ASO Therapies
3.2. Delivery Strategies
3.3. Case Studies: N-of-1 and Platform-Based ASO Development
3.3.1. Milasen: Rapid Customization for Batten Disease
3.3.2. Valeriasen: Targeted Knockdown in KCNT1-Related Epileptic Encephalopathy
3.3.3. Atipeksen: Splice Correction for Ataxia-Telangiectasia
3.4. Toward Scalable Personalization
4. N-of-1 Therapies
4.1. Regulatory Evolution and Foundational Cases
4.2. Feasibility of N-of-1 Treatments
4.3. Ethical and Societal Considerations
5. Challenges and Outlooks
5.1. Delivery and Durability
5.2. Timing and Therapeutic Window
5.3. Economic and Regulatory Barriers
5.4. Data Integration and AI-Driven Design
5.5. Outlook
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Moliner, A.M., Waligora, J. (2017). The European Union Policy in the Field of Rare Diseases. In: Posada de la Paz, M., Taruscio, D., Groft, S. (eds) Rare Diseases Epidemiology: Update and Overview. Advances in Experimental Medicine and Biology, vol 1031. Springer, Cham. [CrossRef]
- Duan, D., Goemans, N., Takeda, S. et al. Duchenne muscular dystrophy. Nat Rev Dis Primers 7, 13 (2021). [CrossRef]
- Phillips, C., Parkinson, A., Namsrai, T., Chalmers, A., Dews, C., Gregory, D., Kelly, E., Lowe, C., & Desborough, J. (2024). Time to diagnosis for a rare disease: managing medical uncertainty. A qualitative study. Orphanet journal of rare diseases, 19(1), 297. [CrossRef]
- Rare Diseases International. Rare Diseases: Leaving no one Behind in Universal Health Coverage. (2019). Available online at:https://d254mlohi4u805.cloudfront.net/rdi/2019/RDI%20UHC%20Paper%20Final%20October%202019.pdf.
- Thompson, R., Spendiff, S., Roos, A., Bourque, P. R., Chardon, J. W., Kirschner, J., Horvath, R. & Lochmüller, H. (2020). Advances in the diagnosis of inherited neuromuscular diseases and implications for therapy development. The Lancet Neurology, 19(6), 522-532. [CrossRef]
- Cohen, E., Bonne, G., Rivier, F. & Hamroun, D. (2021). The 2022 version of the gene table of neuromuscular disorders (nuclear genome). Neuromuscular Disorders, 31(12), 1313-1357. [CrossRef]
- Wilton-Clark, H., Yan, E., & Yokota, T. (2024). Preparing for Patient-Customized N-of-1 Antisense Oligonucleotide Therapy to Treat Rare Diseases. Genes, 15(7), 821. [CrossRef]
- Aoki Y, Wood MJA. Emerging Oligonucleotide Therapeutics for Rare Neuromuscular Diseases. Journal of Neuromuscular Diseases. 2021;8(6):869-884. [CrossRef]
- Qin D. Next-generation sequencing and its clinical application. Cancer Biol Med. 2019 Feb;16(1):4-10. [CrossRef] [PubMed] [PubMed Central]
- Klein, Christopher J. et al. Neurology Individualized Medicine: When to Use Next-Generation Sequencing Panels. Mayo Clinic Proceedings, Volume 92, Issue 2, 292 - 305. [CrossRef]
- Radziwonik-Fraczyk, W., Elert-Dobkowska, E., Karpinski, M. et al. (2024). Next generation sequencing panel as an effective approach to genetic testing in patients with a highly variable phenotype of neuromuscular disorders. Neurogenetics 25, 233–247. [CrossRef]
- Yubero, D., Natera-de Benito, D., Pijuan, J., Armstrong, J., Martorell, L., Fernàndez, G., Maynou, J., Jou, C., Roldan, M., Ortez, C., Nascimento, A., Hoenicka, J., & Palau, F. (2021). The Increasing Impact of Translational Research in the Molecular Diagnostics of Neuromuscular Diseases. International Journal of Molecular Sciences, 22(8), 4274. [CrossRef]
- Pagnamenta, A.T., Camps, C., Giacopuzzi, E. et al.(2023). Structural and non-coding variants increase the diagnostic yield of clinical whole genome sequencing for rare diseases.Genome Med 15, 94. [CrossRef]
- Nurchis, M.C., Altamura, G., Riccardi, M.T. et al.(2023). Whole genome sequencing diagnostic yield for paediatric patients with suspected genetic disorders: systematic review, meta-analysis, and GRADE assessment. Arch Public Health 81, 93. [CrossRef]
- Bodle, E. E., Zhu, W., Velez-Bartolomei, F., Tesi-Rocha, A., Liu, P., & Bernstein, J. A. (2021). Combined Genome Sequencing and RNA Analysis Reveals and Characterizes a Deep Intronic Variant in IGHMBP2 in a Patient With Spinal Muscular Atrophy With Respiratory Distress Type 1. Pediatric neurology, 114, 16–20. [CrossRef]
- Tan, J.K., Awuah, W.A., Ahluwalia, A. et al. (2024).Genes to therapy: a comprehensive literature review of whole-exome sequencing in neurology and neurosurgery. Eur J Med Res 29, 538. [CrossRef]
- Luo, H., Liu, Y., Wei, C., & Xiong, H. (2025). RNA sequencing facilitates the identification of genetic causes of Duchenne muscular dystrophy and proposes a stepwise DMD diagnostic procedure. Gene, 936, 149089. [CrossRef]
- Lauffer, M.C., van Roon-Mom, W., Aartsma-Rus, A. et al. (2024). Possibilities and limitations of antisense oligonucleotide therapies for the treatment of monogenic disorders. Commun Med 4, 6. [CrossRef]
- Tsoumpra MK, Fukumoto S, Matsumoto T, Takeda S, Wood MJA, Aoki Y. (2019). Peptide-conjugate antisense based splice-correction for Duchenne muscular dystrophy and other neuromuscular diseases. EBioMedicine [Internet].;45:630–45. [CrossRef]
- Porensky, P. N., & Burghes, A. H. (2013). Antisense oligonucleotides for the treatment of spinal muscular atrophy. Human gene therapy, 24(5), 489–498. [CrossRef]
- Okubo, M., Noguchi, S., Awaya, T. et al. (2023). RNA-seq analysis, targeted long-read sequencing and in silico prediction to unravel pathogenic intronic events and complicated splicing abnormalities in dystrophinopathy. Hum Genet 142, 59–71. [CrossRef]
- Lim, K. H., Han, Z., Jeon, H. Y., Kach, J., Jing, E., Weyn-Vanhentenryck, S., Downs, M., Corrionero, A., Oh, R., Scharner, J., Venkatesh, A., Ji, S., Liau, G., Ticho, B., Nash, H., & Aznarez, I. (2020). Antisense oligonucleotide modulation of non-productive alternative splicing upregulates gene expression. Nature communications, 11(1), 3501. [CrossRef]
- McDowall, S., Aung-Htut, M., Wilton, S. & Li, D. (2024). Antisense oligonucleotides and their applications in rare neurological diseases. Frontiers in Neuroscience, 18. [CrossRef]
- Ek, M., Nilsson, D., Engvall, M., Malmgren, H., Thonberg, H., Pettersson, M., Anderlid, B. M., Hammarsjö, A., Helgadottir, H. T., Arnardottir, S., Naess, K., Nennesmo, I., Paucar, M., Hjartarson, H. T., Press, R., Solders, G., Sejersen, T., Lindstrand, A., & Kvarnung, M. (2023). Genome sequencing with comprehensive variant calling identifies structural variants and repeat expansions in a large fraction of individuals with ataxia and/or neuromuscular disorders. Frontiers in neurology, 14, 1170005. [CrossRef]
- Lin, X., Yang, Y., Melton, P. E., Singh, V., Simpson-Yap, S., Burdon, K. P., Taylor, B. V., & Zhou, Y. (2022). Integrating Genetic Structural Variations and Whole-Genome Sequencing Into Clinical Neurology. Neurology. Genetics, 8(4), e200005. [CrossRef]
- Davis, R. L., Kumar, K. R., Puttick, C., Liang, C., Ahmad, K. E., Edema-Hildebrand, F., Park, J., Minoche, A. E., Gayevskiy, V., Mallawaarachchi, A. C., Christodoulou, J., Schofield, D., Dinger, M. E., Cowley, M. J. & Sue, C. M. (2022). Use of whole-genome sequencing for mitochondrial disease diagnosis. Neurology, 99(7). [CrossRef]
- Bennett, C. F. & Swayze, E. E. (2010). Rna targeting therapeutics: Molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annual Review of Pharmacology and Toxicology, 50(1), 259-293. [CrossRef]
- van Deutekom, J. C., Bremmer-Bout, M., Janson, A. A., Ginjaar, I. B., Baas, F., den Dunnen, J. T., & van Ommen, G. J. (2001). Antisense-induced exon skipping restores dystrophin expression in DMD patient derived muscle cells. Human molecular genetics, 10(15), 1547–1554. [CrossRef]
- Mallory A. Havens, Michelle L. Hastings. (2019). Splice-switching antisense oligonucleotides as therapeutic drugs, Nucleic Acids Research, Volume 44, Issue 14, Pages 6549–6563. [CrossRef]
- Gushchina, L. V., Vetter, T. A., Frair, E. C., Bradley, A. J., Grounds, K. M., Lay, J. W., Huang, N., Suhaiba, A., Schnell, F. J., Hanson, G., Simmons, T. R., Wein, N., & Flanigan, K. M. (2022). Systemic PPMO-mediated dystrophin expression in the Dup2 mouse model of Duchenne muscular dystrophy. Molecular therapy. Nucleic acids, 30, 479–492. [CrossRef]
- Yau, W., Sullivan, R., O’Connor, E., Pellerin, D., Parkinson, M., Giunti, P., Dicaire, M., Danzi, M., Züchner, S., Brais, B., Wood, N., Houlden, H., Vandrovcova, J. (2025). Diagnostic yield and limitations of whole-genome sequencing for hereditary cerebellar ataxia, Brain Communications, Volume 7, Issue 3, fcaf188. [CrossRef]
- Kuijper EC, Bergsma AJ, Pijnappel WWMP, Aartsma-Rus A. Opportunities and challenges for antisense oligonucleotide therapies. J Inherit Metab Dis. (2021) ;44(1):72-87. [CrossRef] [PubMed] [PubMed Central]
- Chwalenia, K., Wood, M. J. A. & Roberts, T. C. (2025). Progress and prospects in antisense oligonucleotide-mediated exon skipping therapies for duchenne muscular dystrophy. Journal of Muscle Research and Cell Motility. [CrossRef]
- Geary, R. S., Norris, D., Yu, R. & Bennett, C. F. (2015). Pharmacokinetics, biodistribution and cell uptake of antisense oligonucleotides. Advanced Drug Delivery Reviews, 87, 46-51. [CrossRef]
- Gan, L., Wu, L. C. L., Wood, J. A., Yao, M., Treleaven, C. M., Estrella, N. L., Wentworth, B. M., Hanson, G. J., & Passini, M. A. (2022). A cell-penetrating peptide enhances delivery and efficacy of phosphorodiamidate morpholino oligomers in mdx mice. Molecular therapy. Nucleic acids, 30, 17–27. [CrossRef]
- Xiulong Shen, David R Corey, Chemistry, mechanism and clinical status of antisense oligonucleotides and duplex RNAs, Nucleic Acids Research, Volume 46, Issue 4, 28 February 2018, Pages 1584–1600. [CrossRef]
- Juliano, R., Alam, M. R., Dixit, V., & Kang, H. (2008). Mechanisms and strategies for effective delivery of antisense and siRNA oligonucleotides. Nucleic acids research, 36(12), 4158–4171. [CrossRef]
- Hsia, H., Zanini, C., Simonneau, C., Fraidling, J., Kraft, T., Mayer, K., Sommer, A., Indlekofer, A., Wirth, T., Benz, J., Georges, G., Langer, L., Gassner, C., Larraillet, V., Lohmann, S., Koller, E., Mansø, M., Ravn, J., Hofer, K., Emrich, T., Niewöhner, J., Schumacher, F., Brinkmann, U. (2025). Improved targeted delivery of antisense oligonucleotide with an antibody mask, Nucleic Acids Research, Volume 53, Issue 20, gkaf487. [CrossRef]
- Shah, M. N. A., Wilton-Clark, H., Haque, F., Powell, B., Sutanto, L. E., Maradiya, R., Zhabyeyev, P., Roshmi, R. R., Anwar, S., Aslesh, T., Lim, K. R. Q., Maruyama, R., Bigot, A., Young, C. S., Bittner, S., Spencer, M. J., Moulton, H. M., Oudit, G. Y., & Yokota, T. (2025). DG9 boosts PMO nuclear uptake and exon skipping to restore dystrophic muscle and cardiac function. Nature communications, 16(1), 4477. [CrossRef]
- Goto, A., Yamamoto, S., & Iwasaki, S. (2023). Biodistribution and delivery of oligonucleotide therapeutics to the central nervous system: Advances, challenges, and future perspectives. Biopharmaceutics and Drug Disposition, 44(1), 26–47. [CrossRef]
- Kennedy Z, Gilbert JW, Godinho BMDC.(2022 Feb 25). Intrathecal Delivery of Therapeutic Oligonucleotides for Potent Modulation of Gene Expression in the Central Nervous System. In: Arechavala-Gomeza V, Garanto A, editors. Antisense RNA Design, Delivery, and Analysis [Internet]. New York: Humana; 2022. Chapter 24. Available from: https://www.ncbi.nlm.nih.gov/books/NBK584238/doi: 10.1007/978-1-0716-2010-6_24. [CrossRef]
- Lindow, M., Vornlocher, HP., Riley, D. et al.(2012). Assessing unintended hybridization-induced biological effects of oligonucleotides. Nat Biotechnol 30, 920–923. [CrossRef]
- Kim, J., Hu, C., Moufawad El Achkar, C., Black, L. E., Douville, J., Larson, A., Pendergast, M. K., Goldkind, S. F., Lee, E. A., Kuniholm, A., Soucy, A., Vaze, J., Belur, N. R., Fredriksen, K., Stojkovska, I., Tsytsykova, A., Armant, M., DiDonato, R. L., Choi, J., … Yu, T. W. (2019). Patient-customized oligonucleotide therapy for a rare genetic disease. New England Journal of Medicine, 381(17), 1644–1652. [CrossRef]
- Wilton-Clark, H., Yan, E., Yokota, T. (2025). Milasen: The Emerging Era of Patient-Customized N-of-1 Antisense Oligonucleotides as Therapeutic Agents for Genetic Diseases. In: Echigoya, Y., Maruyama, R., Yokota, T. (eds) Exon Skipping and Inclusion Therapies. Methods in Molecular Biology, vol 2964. Humana, New York, NY. [CrossRef]
- Burbano, L. E., Li, M., Jancovski, N., Jafar-Nejad, P., Richards, K., Sedo, A., Soriano, A., Rollo, B., Jia, L., Gazina, E. V., Piltz, S., Adikusuma, F., Thomas, P. Q., Kopsidas, H., Rigo, F., Reid, C. A., Maljevic, S., & Petrou, S. (2022). Antisense oligonucleotide therapy for KCNT1 encephalopathy. JCI insight, 7(23), e146090. [CrossRef]
- Kim, J., Woo, S., de Gusmao, C.M. et al. (2023). A framework for individualized splice-switching oligonucleotide therapy. Nature 619, 828–836. [CrossRef]
- Roshmi, R.R., Yokota, T. (2023). Viltolarsen: From Preclinical Studies to FDA Approval. In: Maruyama, R., Yokota, T. (eds) Muscular Dystrophy Therapeutics. Methods in Molecular Biology, vol 2587. Humana, New York, NY. [CrossRef]
- Bou-Jaoudeh, M., Piaton-Breda, G., Pereme, F. et al. (2025). Toward an Extensible Regulatory Framework for N-of-1 to N-of-Few Personalized RNA Therapy Design. Ther Innov Regul Sci59, 505–518. [CrossRef]
- IND Submissions for Individualized Antisense Oligonucleotide Drug Products for Severely Debilitating or Life-Threatening Diseases: Clinical Recommendations. FDA-2021-D-1140. (2021). https://www.federalregister.gov/d/2021-26453.
- Aartsma-Rus, A., Garanto, A., van Roon-Mom, W., McConnell, E. M., Suslovitch, V., Yan, W. X., Watts, J. K., & Yu, T. W. (2023). Consensus Guidelines for the Design and In Vitro Preclinical Efficacy Testing N-of-1 Exon Skipping Antisense Oligonucleotides. Nucleic acid therapeutics, 33(1), 17–25. [CrossRef]
- Synofzik, M., van Roon-Mom, W. M. C., Marckmann, G., van Duyvenvoorde, H. A., Graessner, H., Schüle, R., & Aartsma-Rus, A. (2022). Preparing n-of-1 Antisense Oligonucleotide Treatments for Rare Neurological Diseases in Europe: Genetic, Regulatory, and Ethical Perspectives. Nucleic acid therapeutics, 32(2), 83–94. [CrossRef]
- Goyenvalle, A., Jimenez-Mallebrera, C., van Roon, W., Sewing, S., Krieg, A. M., Arechavala-Gomeza, V., & Andersson, P. (2023). Considerations in the Preclinical Assessment of the Safety of Antisense Oligonucleotides. Nucleic acid therapeutics, 33(1), 1–16. [CrossRef]
- Schüle, R., Graessner, H., Aartsma-Rus, A., Roon-Mom, W. M. v. & Synofzik, M. (2025). Tailored antisense oligonucleotides for ultrarare cns diseases: An experience-based best practice framework for individual patient evaluation. Molecular Therapy Nucleic Acids, 36(3), 102615. [CrossRef]
- Pacanowski, M., Vitarello, J., Hyun, I., Yu, T., & Zineh, I. (2023). A Multistakeholder Perspective on Advancing Individualized Therapeutics. In Clinical Pharmacology and Therapeutics (Vol. 114, Issue 5, pp. 994–1001). John Wiley and Sons Inc. [CrossRef]
- n-Lorem Foundation. (2025). About. https://www.nlorem.org/.
- Bladen, C. L., Salgado, D., Monges, S., Foncuberta, M. E., Kekou, K., Kosma, K., Dawkins, H., Lamont, L., Roy, A. J., Chamova, T., Guergueltcheva, V., Chan, S., Korngut, L., Campbell, C., Dai, Y., Wang, J., Barišić, N., Brabec, P., Lahdetie, J., Walter, M. C., … Lochmüller, H. (2015). The TREAT-NMD DMD Global Database: analysis of more than 7,000 Duchenne muscular dystrophy mutations. Human mutation, 36(4), 395–402. [CrossRef]
- Mohammad Shadid, Mohamed Badawi & Abedelnasser Abulrob, (2021) Antisense oligonucleotides: absorption, distribution, metabolism, and excretion, Expert Opinion on Drug Metabolism & Toxicology, 17:11, 1281-1292. [CrossRef]
- Nakmode, D.D., Singh, B., Abdella, S. et al. Long-acting parenteral formulations of hydrophilic drugs, proteins, and peptide therapeutics: mechanisms, challenges, and therapeutic benefits with a focus on technologies. Drug Deliv. and Transl. Res. 15, 1156–1180 (2025). [CrossRef]
- Chen, P., Wei, Y., Sun, T., Lin, J., & Zhang, K. (2024). Enabling safer, more potent oligonucleotide therapeutics with bottlebrush polymer conjugates. Journal of controlled release : official journal of the Controlled Release Society, 366, 44–51. [CrossRef]
- Brancato, D., Treccarichi, S., Bruno, F., Coniglio, E., Vinci, M., Saccone, S., Calì, F., & Federico, C. (2025). NGS Approaches in Clinical Diagnostics: From Workflow to Disease-Specific Applications. International Journal of Molecular Sciences, 26(19), 9597. [CrossRef]
- Pietro Delre, Carmen Cerchia, Antonio Lavecchia.(2025) Artificial intelligence in the development of small nucleic acid therapeutics: toward smarter and safer medicines. Drug Discovery Today. Volume 30, Issue 11, 104488. ISSN 1359-6446. [CrossRef]
- Bou-Jaoudeh, M., Piaton-Breda, G., Pereme, F., & Gilbert, S. (2025). Toward an Extensible Regulatory Framework for N-of-1 to N-of-Few Personalized RNA Therapy Design. Therapeutic innovation & regulatory science, 59(3), 505–518. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



