Submitted:
10 November 2025
Posted:
12 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design, Period, and Setting
2.2. Ethical Consideration
2.3. Collection of Clinical Samples and Data
2.4. Clinical Specimens Testing by Allplex Multiplex rRT-qPCR
2.4.1. RNA Extraction
2.4.2. Pooled RNA Samples Testing
2.5. Retesting of Samples with Invalid Allplex rRT-qPCR Results
2.6. Clinical Specimens Testing by the ML Ag Combo Test
2.7. Statistical Analysis
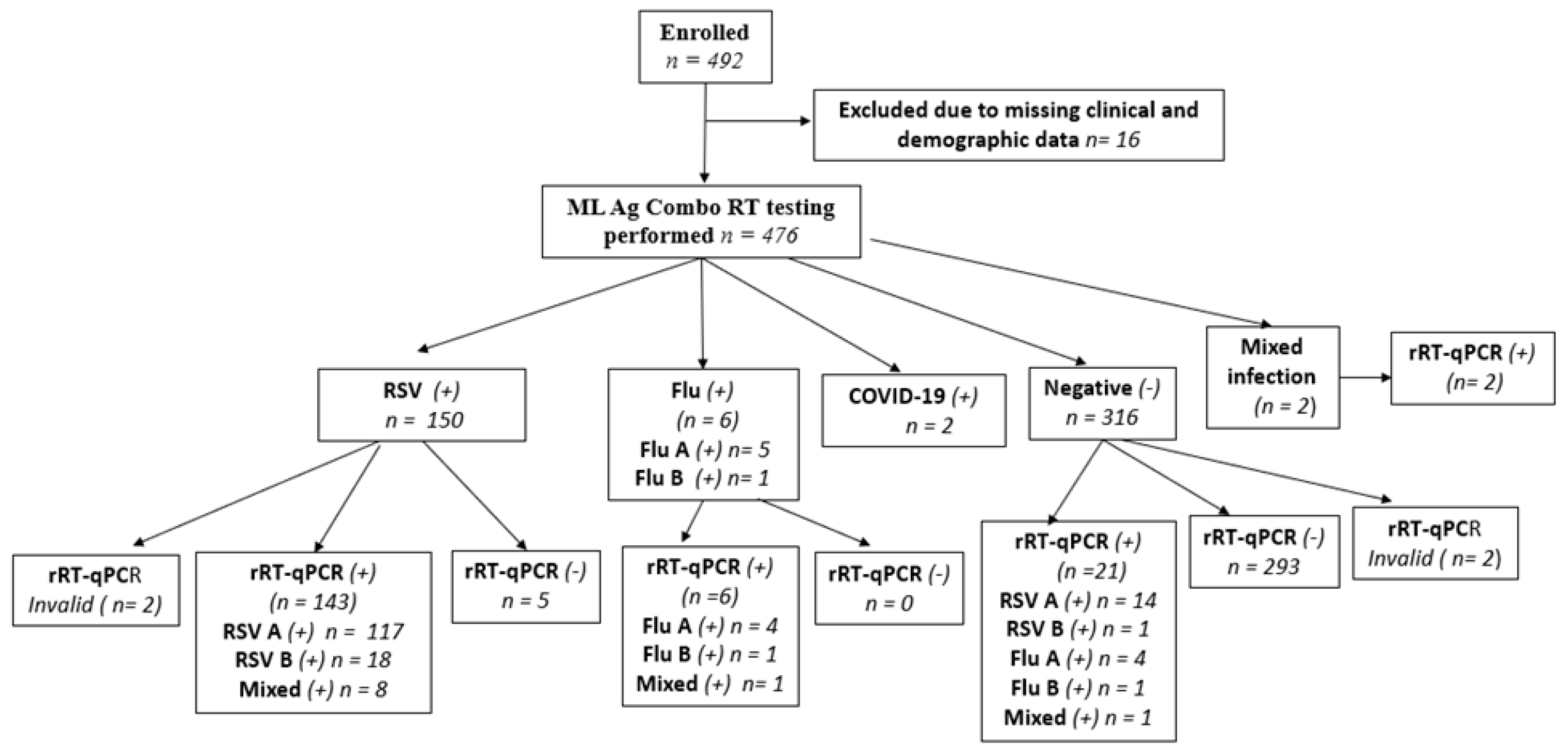
3. Results
3.1. Mixed Infections
3.2. Clinical Performance of the ML Ag Como Rapid Test Using Allplex rRT-qPCR as a Reference
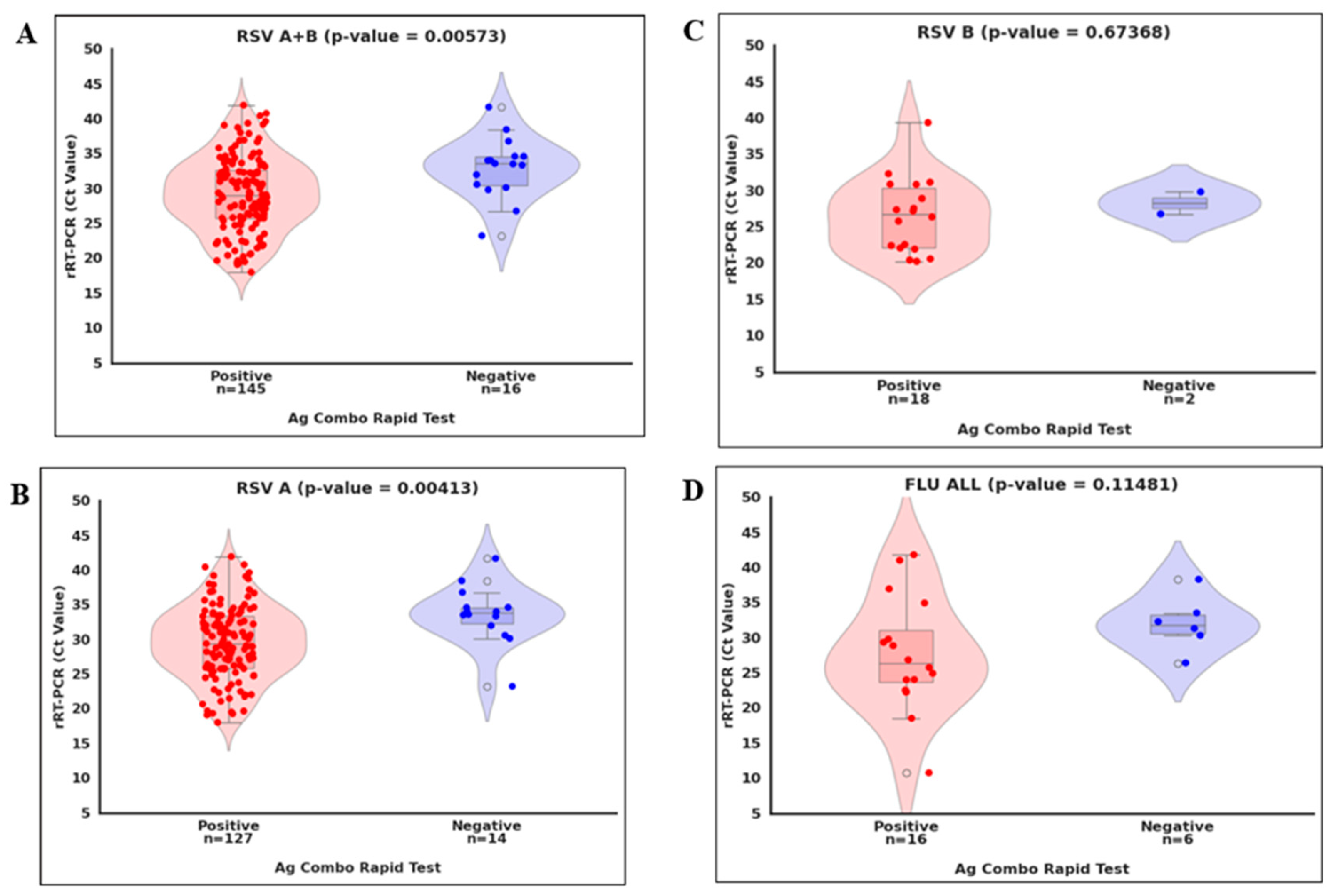
3.2.1. ML Ag Combo RDT Sensitivity Depending on the Ct Values
3.2.2. ML Ag Combo RDT Sensitivity Depending on the Ct Values
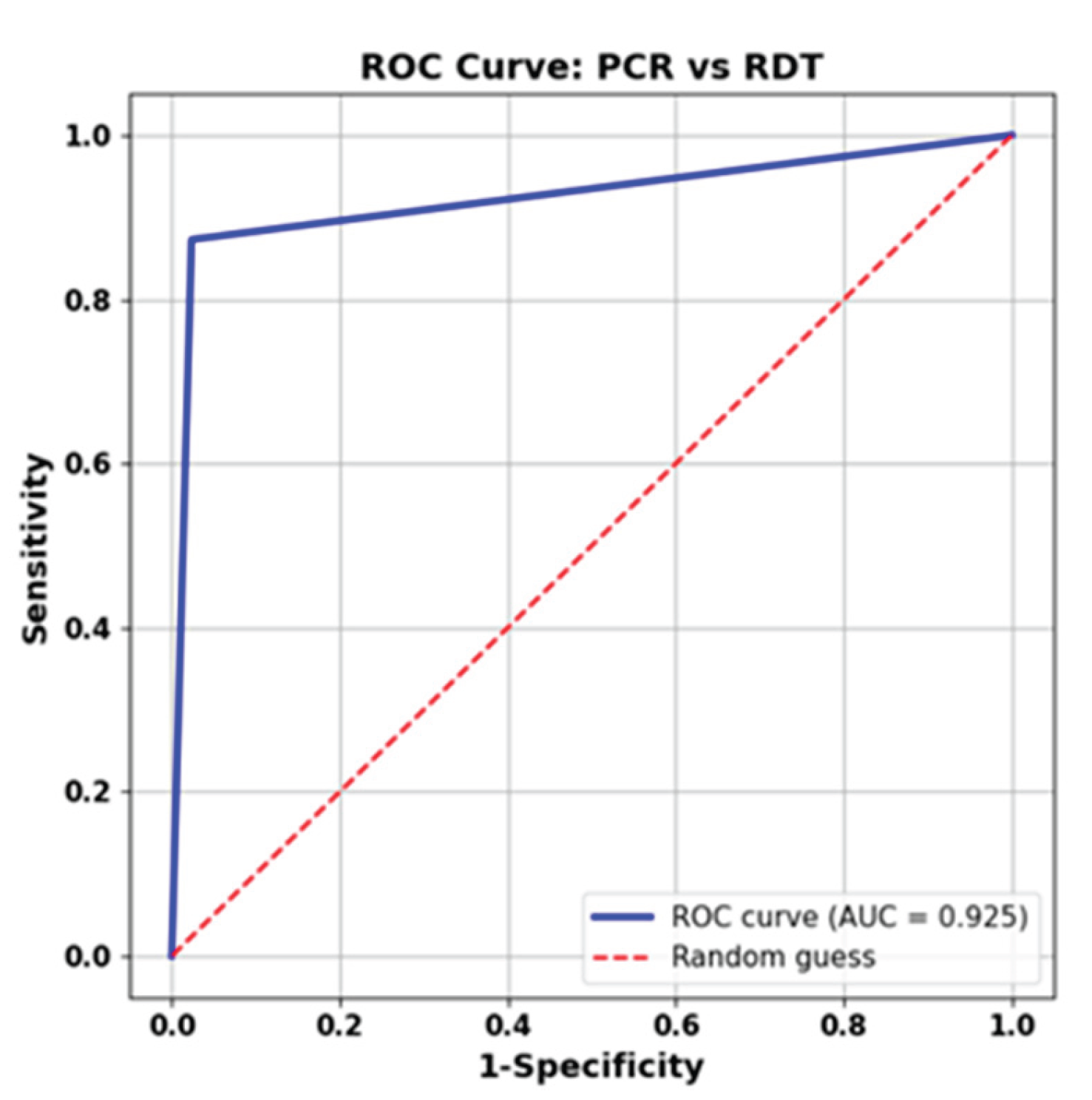
3.2.3. ML Ag Combo RDT Showed High Performance Across Different Metrics in Pediatric Hospital Settings
4. Discussion
5. Study Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AHRI | Armauer Hansen Research Institute |
| ARI | Acute Respiratory Infections |
| AUC | Arear Under Curve |
| CDC | Center for Disease Control |
| CI | Confidence Interval |
| COVID-19 | Coronavirus Disease 2019 |
| CRP | C-Reactive Protein |
| CT | Cycle Threshold |
| FIA | Fluorescence immunoassay |
| ILI | Influenza-like Illness |
| IQR | Inter Quartile Range |
| LFA | Lateral Flow Assay |
| LRTI | Lower Respiratory Tract Infection |
| ML | MobiLab |
| MxA | Myxovirus Resistance Protein A |
| NP | Nucleoprotein |
| NPS | Nasopharyngeal Swab |
| NPV | Negative Predictive Value |
| POCT | Point-of-care Test |
| PPV | Positive Predictive Value |
| RDT | Rapid Diagnostic Test |
| RNA | Ribo Nucleic Acid |
| RSV | Respiratory Syncytial Virus |
| Flu A | Influenza A virus |
| Flu B | Influenza B virus |
| RSV A | Respiratory syncytial virus A |
| RSV B | respiratory syncytial virus B. |
| ROC | Receiver Operating Characteristics |
| rRT-qPCR | Real-Time Reverse Transcription Quantitative Polymerase Chain Reaction |
| SARS-COV2 | Severe Acute Respiratory Syndrome- Corona Virus 2 |
| VTM | Viral Transport Medium |
References
- Wang X, Li Y, Deloria-Knoll M, Madhi SA, Cohen C, Arguelles VL, Basnet S, Bassat Q, Brooks WA, Echavarria M, Fasce RA, Gentile A, Goswami D, Homaira N, Howie SRC, Kotloff KL, Khuri-Bulos N, Krishnan A, Lucero MG, Lupisan S, Mathisen M, McLean KA, Mira-Iglesias A, Moraleda C, Okamoto M, Oshitani H, O’Brien KL, Owor BE, Rasmussen ZA, Rath BA, Salimi V, Sawatwong P, Scott JAG, Simões EAF, Sotomayor V, Thea DM, Treurnicht FK, Yoshida L-M, Zar HJ, Campbell H, Nair H. Global burden of acute lower respiratory infection associated with human parainfluenza virus in children younger than 5 years for 2018: a systematic review and meta-analysis. Lancet Glob Health 2021, 9, e1077–e1087. [Google Scholar] [CrossRef] [PubMed]
- Robertson SE, Roca A, Alonso P, Simoes EAF, Kartasasmita CB, Olaleye DO, Odaibo GN, Collinson M, Venter M, Zhu Y, Wright PF. Respiratory syncytial virus infection: denominator-based studies in Indonesia, Mozambique, Nigeria and South Africa. Bull World Health Organ 2004, 82, 914–922. [Google Scholar]
- 2023 Respiratory Virus Response - NSSP Emergency Department Visits - COVID-19, Flu, RSV, Combined | Data | Centers for Disease Control and Prevention. https://data.cdc.gov/Public-Health-Surveillance/2023-Respiratory-Virus-Response-NSSP-Emergency-Dep/vutn-jzwm/about_data. Retrieved 28 October 2025.
- Langley JM, Bianco V, Domachowske JB, Madhi SA, Stoszek SK, Zaman K, Bueso A, Ceballos A, Cousin L, D’Andrea U, Dieussaert I, Englund JA, Gandhi S, Gruselle O, Haars G, Jose L, Klein NP, Leach A, Maleux K, Nguyen TLA, Puthanakit T, Silas P, Tangsathapornpong A, Teeratakulpisarn J, Vesikari T, Cohen RA. Incidence of Respiratory Syncytial Virus Lower Respiratory Tract Infections During the First 2 Years of Life: A Prospective Study Across Diverse Global Settings. J Infect Dis 2022, 226, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Mazur NI, Caballero MT, Nunes MC. Severe respiratory syncytial virus infection in children: burden, management, and emerging therapies.
- Moyes J, Tempia S, Walaza S, McMorrow ML, Treurnicht F, Wolter N, von Gottberg A, Kahn K, Cohen AL, Dawood H, Variava E, Cohen C. The economic burden of RSV-associated illness in children aged < 5 years, South Africa 2011–2016. BMC Medicine 2023, 21, 146.
- Getzzg. 2025. Estimating the economic burden of respiratory syncytial virus infection among children <2 years old receiving care in Maputo, Mozambique. JOGH. https://jogh.org/2025/jogh-15-04076/. Retrieved 28 October 2025.
- Tayachew A, Teka G, Gebeyehu A, Shure W, Biru M, Chekol L, Berkessa T, Tigabu E, Gizachew L, Agune A, Gonta M, Hailemariam A, Gedefaw E, Woldeab A, Alemu A, Getaneh Y, Lisanwork L, Yibeltal K, Abate E, Abayneh A, Wossen M, Hailu M, Workineh F. Prevalence of respiratory syncytial virus infection and associated factors in children aged under five years with severe acute respiratory illness and influenza-like illness in Ethiopia. IJID Reg 2024, 10, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Wadilo F, Feleke A, Gebre M, Mihret W, Seyoum T, Melaku K, Howe R, Mulu A, Mihret A. Viral etiologies of lower respiratory tract infections in children < 5 years of age in Addis Ababa, Ethiopia: a prospective case–control study. Virol J 2023, 20, 163. [Google Scholar]
- Tayachew A, Mekuria Z, Shure W, Arimide DA, Gebeyehu A, Berkesa T, Gonta M, Teka G, Kebede M, Melese D, Wossen M, Abte M, Hailu M, Berhe N, Medstrand P, Kebede N. Epidemiology of respiratory syncytial virus and its subtypes among cases of influenza like illness and severe acute respiratory infection: findings from nationwide sentinel surveillance in Ethiopia. BMC Infect Dis 2025, 25, 941. [Google Scholar]
- Shure W, Tayachew A, Berkessa T, Teka G, Biru M, Gebeyehu A, Woldeab A, Tadesse M, Gonta M, Agune A, Hailemariam A, Haile B, Addis B, Moges M, Lisanwork L, Gizachew L, Tigabu E, Mekuria Z, Yimer G, Dereje N, Aliy J, Lulseged S, Melaku Z, Abate E, Gebreyes W, Wossen M, Abayneh A. SARS-CoV-2 co-detection with influenza and human respiratory syncytial virus in Ethiopia: Findings from the severe acute respiratory illness (SARI) and influenza-like illness (ILI) sentinel surveillance, January 01, 2021, to June 30, 2022. PLOS Glob Public Health 2024, 4, e0003093. [Google Scholar]
- Caldwell JM, Espinosa CM, Banerjee R, Domachowske JB. Rapid diagnosis of acute pediatric respiratory infections with Point-of-Care and multiplex molecular testing. Infection 2025, 53, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Jeong HW, Heo JY, Park JS, Kim WJ. Effect of the Influenza Virus Rapid Antigen Test on a Physician’s Decision to Prescribe Antibiotics and on Patient Length of Stay in the Emergency Department. [CrossRef]
- Diagnostic utility of rapid antigen testing as point-of-care test for influenza and other respiratory viruses in patients with acute respiratory illness. [CrossRef]
- Swets MC, Russell CD, Harrison EM, Docherty AB, Lone N, Girvan M, Hardwick HE, Visser LG, Openshaw PJM, Groeneveld GH, Semple MG, Baillie JK. SARS-CoV-2 co-infection with influenza viruses, respiratory syncytial virus, or adenoviruses. The Lancet 2022, 399, 1463–1464. [Google Scholar] [CrossRef] [PubMed]
- Rapid multiplex PCR for respiratory viruses reduces time to result and improves clinical care: Results of a systematic review and meta-analysis. [CrossRef]
- Nelson PP, Rath BA, Fragkou PC, Antalis E, Tsiodras S, Skevaki C. Current and Future Point-of-Care Tests for Emerging and New Respiratory Viruses and Future Perspectives. Front Cell Infect Microbiol 2020, 10, 181. [Google Scholar]
- Tai C-S, Jian M-J, Lin T-H, Chung H-Y, Chang C-K, Perng C-L, Hsieh P-S, Shang H-S. Analytical performance evaluation of a multiplex real-time RT-PCR kit for simultaneous detection of SARS-CoV-2, influenza A/B, and RSV. PeerJ 2025, 13, e19693. [Google Scholar] [CrossRef] [PubMed]
- Schober T, Wong K, DeLisle G, Caya C, Brendish NJ, Clark TW, Dendukuri N, Doan Q, Fontela PS, Gore GC, Li P, McGeer AJ, Noël KC, Robinson JL, Suarthana E, Papenburg J. Clinical Outcomes of Rapid Respiratory Virus Testing in Emergency Departments: A Systematic Review and Meta-Analysis. JAMA Intern Med 2024, 184, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Lim H-J, Lee J-Y, Baek Y-H, Park M-Y, Youm D-J, Kim I, Kim M-J, Choi J, Sohn Y-H, Park J-E, Yang Y-J. Evaluation of Multiplex Rapid Antigen Tests for the Simultaneous Detection of SARS-CoV-2 and Influenza A/B Viruses. Biomedicines 2023, 11, 3267. [Google Scholar]
- 2020. Biological evaluation of medical devices—Part 1: Evaluation and testing within a risk management processANSI/AAMI/ISO 10993-1:2018; Biological evaluation of medical devices—Part 1: Evaluation and testing within a risk management process. AAMI.
- Meier, K. Guidance for Industry and FDA Staff - Statistical Guidance on Reporting Results from Studies Evaluating Diagnostic Tests.
- McHugh, ML. 2012. Interrater reliability: the kappa statistic. Biochem Med 276–282.
- Lee S. Guide to ROC and AUC for Statistical Analysis. https://www.numberanalytics.com/blog/roc-auc-guide-statistical-analysis. Retrieved 2 November 2025.
- Chekol MT, Sugerman D, Tayachew A, Mekuria Z, Tesfay N, Alemu A, Gashu A, Shura W, Gonta M, Agune A, Hailemariam A, Assefa Y, Wossen M, Hassen A, Michele P, Silver R, Delelegn H, Briana L, Kasa T, Kebede N. Frontiers | Clinical and epidemiological characteristics of influenza and SARS-CoV-2 virus among patients with acute febrile illness in selected sites of Ethiopia 2021–2022. [CrossRef]
- Dulo B, Hinsene G, Mannekulih E. 2024. Viral etiology of respiratory infections among patients at Adama Hospital Medical College, a facility-based surveillance site in Oromia, Ethiopia. [CrossRef]
- CDC. 2025. Multiplex Assays Authorized for Simultaneous Detection of Influenza Viruses and SARS-CoV-2 by FDA. Influenza (Flu). https://www.cdc.gov/flu/hcp/testing-methods/flu-covid19-detection.html. Retrieved 28 October 2025.
- Basile K, Kok J, Dwyer DE. Point-of-care diagnostics for respiratory viral infections. Expert Rev Mol Diagn 2017, 18, 75–83. [Google Scholar]
- Batra R, Blandford E, Kulasegaran-Shylini R, Futschik ME, Bown A, Catton M, Conti-Frith H, Alexandridou A, Gill R, Milroy C, Harper S, Gettings H, Noronha M, Harrison H-L, Douthwaite S, Nebbia G, Klapper PE, Tunkel S, Vipond R, Hopkins S, Fowler T. 2025. Multiplex lateral flow test sensitivity and specificity in detecting influenza A, B and SARS-CoV-2 in adult patients in a UK emergency department. [CrossRef]
- Carlton HC, Savović J, Dawson S, Mitchelmore PJ, Elwenspoek MMC. Novel point-of-care biomarker combination tests to differentiate acute bacterial from viral respiratory tract infections to guide antibiotic prescribing: a systematic review.
- Rosenblatt KP, Romeu H, Romeu C, Granger E. Frontiers | Performance evaluation of a SARS-CoV-2 and influenza A/B combo rapid antigen test. [CrossRef]
- Bayart J-L, Gillot C, Dogné J-M, Roussel G, Verbelen V, Favresse J, Douxfils J. Clinical performance evaluation of the Fluorecare® SARS-CoV-2 & Influenza A/B & RSV rapid antigen combo test in symptomatic individuals. J Clin Virol 2023, 161, 105419. [Google Scholar]
- Aboagye FT, Annison L, Hackman HK, Acquah ME, Ashong Y, Owusu-Frimpong I, Egyam BC, Annison S, Osei-Adjei G, Antwi-Baffour S. Comparative evaluation of RT-PCR and antigen-based rapid diagnostic tests (Ag-RDTs) for SARS-CoV-2 detection: performance, variant specificity, and clinical implications. Microbiol Spectr 2024, 12, e00073–24. [Google Scholar]
- Widyasari K, Kim S, Kim S, Lim CS. Performance Evaluation of STANDARD Q COVID/FLU Ag Combo for Detection of SARS-CoV-2 and Influenza A/B. Diagnostics (Basel) 2022, 13, 32. [Google Scholar] [CrossRef] [PubMed]
- Franck KT, Schneider UV, Ma CMG, Knudsen D, Lisby G. Evaluation of immuview RSV antigen test (SSI siagnostica) and BinaxNOW RSV card (alere) for rapid detection of respiratory syncytial virus in retrospectively and prospectively collected respiratory samples. [CrossRef]
- Cantais A, Mory O, Plat A, Giraud A, Pozzetto B, Pillet S. Analytical performances of the BD VeritorTM System for the detection of respiratory syncytial virus and influenzaviruses A and B when used at bedside in the pediatric emergency department. Journal of Virological Methods 2019, 270, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Zhu M, Chen L, Cao J, Cai J, Huang S, Wang H, He H, Chen Z, Huang R, Ye H. Frontiers | Clinical application of Myxovirus resistance protein A as a diagnostic biomarker to differentiate viral and bacterial respiratory infections in pediatric patients. [CrossRef]
- Jung J-A, Wi P-H, Kim H. Comparisons of clinical characteristics between Respiratory Syncytial Virus A and B infection.
- van der Zalm MM, Sam-Agudu NA, Verhagen LM. Respiratory adenovirus infections in children: a focus on Africa. Curr Opin Pediatr 2024, 36, 342–348. [Google Scholar] [CrossRef] [PubMed]
| Cta (N gene) | RSV | Flu A+B |
|---|---|---|
| < 20 | (7/7) 100 % | (1/1)100 % |
| < 25 | (31/32) 96.9 % | (6/6) 100 % |
| < 30 | (78/81) 96.3 % | (11/12)91.6% |
| < 35 | (125/138) 90.6% | (12/17) 70.6% |
| < 42 | (145/161) 90.1 % | 15/2171.4% |
| 20 – 25 | (24/25) 96 % | (5/5) 100 % |
| 25 – 30 | (47/49) 95.9 % | (5/6)83.3 % |
| 30-35 | (46/56)82.1 % | (1/5)20.0 % |
| 35-42 | 86.9 % (20/23) | (3/4) 75.0% |
| Mean Ct (< 42) [95% CI] | 29.6 (28.7-30.4) | 29.7(26.8-32.6) |
| Neg. with RT- qPCR [95% CI] | 98.3 % (96.13 - 99.45) | 100 % (98.78 - 100) |
| RSV | 95 % CI1 | FLU A/B | 95 % CI |
|---|---|---|---|
| Sensitivity | 90.06 % | 84.36 % - 94.21 % | 73.68 % |
| Specificity | 98.33% | 96.13 % - 99.45 % | 100 % |
| PPV | 93.45 % | 85.66– 97.15 % | 100 % |
| NPV | 97.38 % | 95.90 % - 98.34 % | 93.46 % |
| Accuracy | 96.59% | 94.49 % - 98.05 % | 94.47 % |
| Cohen’s κ | 0.90 | 0.86–0.94 | 0.82 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




