Submitted:
08 November 2025
Posted:
10 November 2025
You are already at the latest version
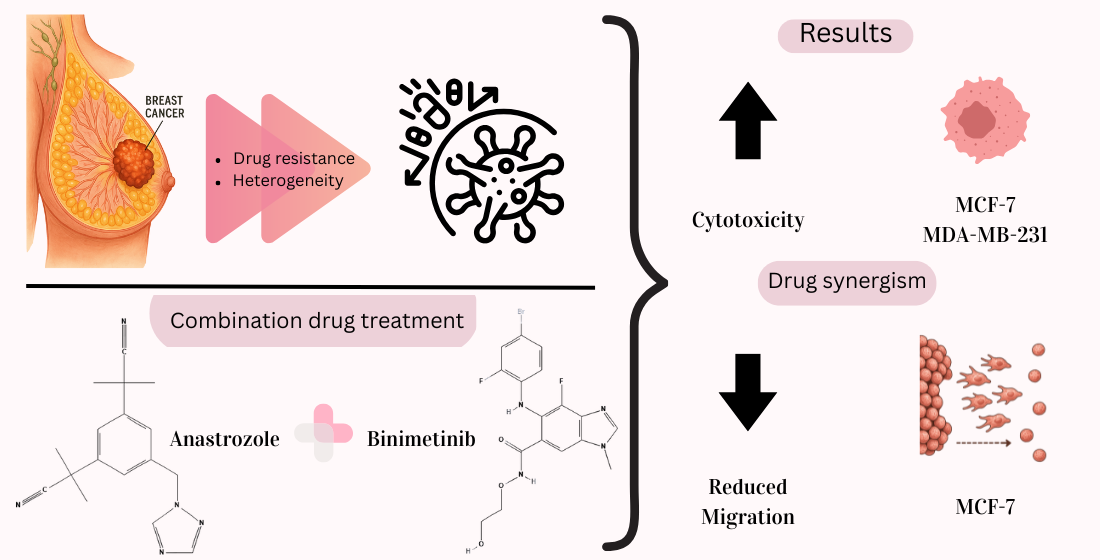
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Drugs
2.2. Cell Culture
2.3. Cytotoxicity Assay
2.4. Migration Assay
2.5. Statistical Analysis
3. Results
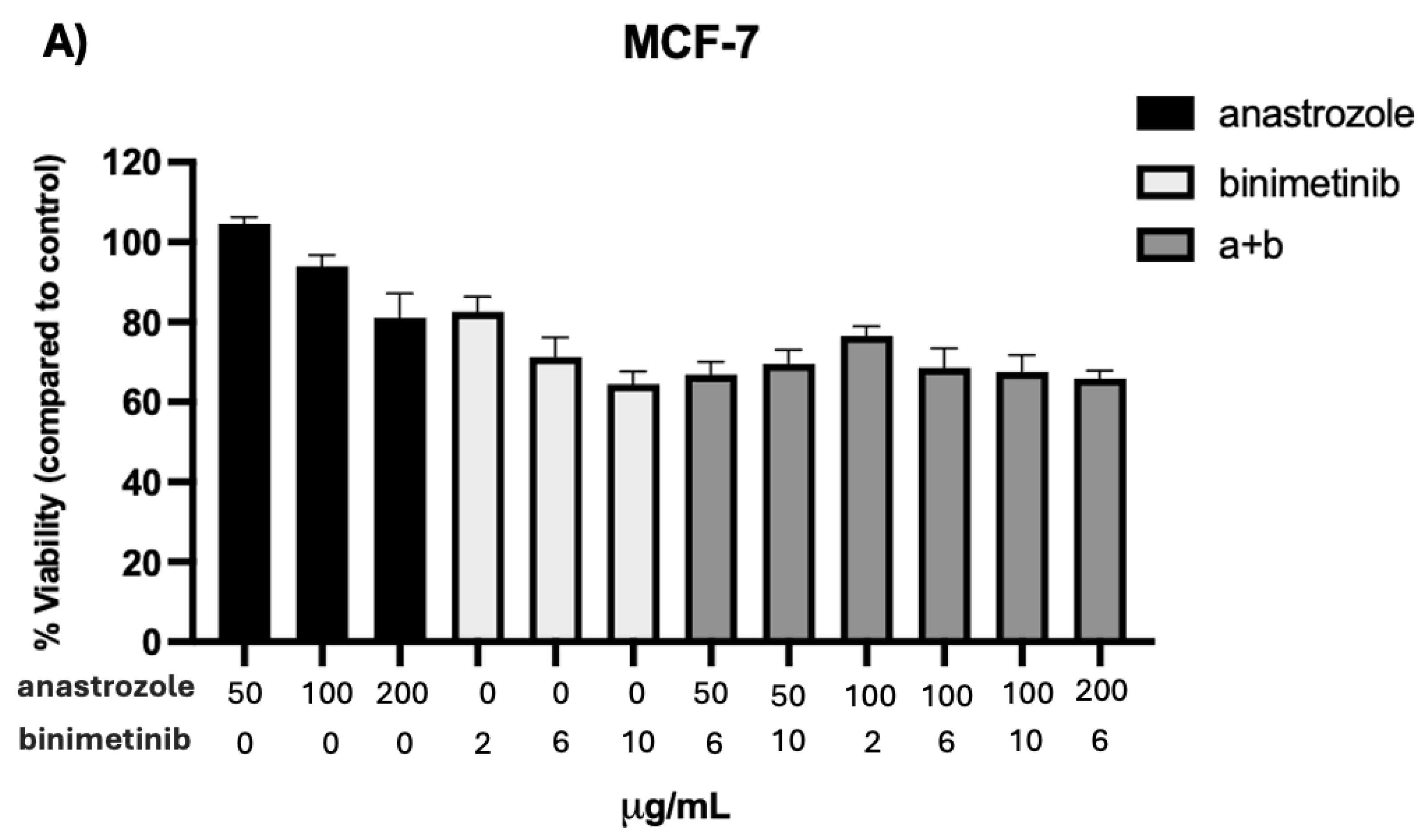
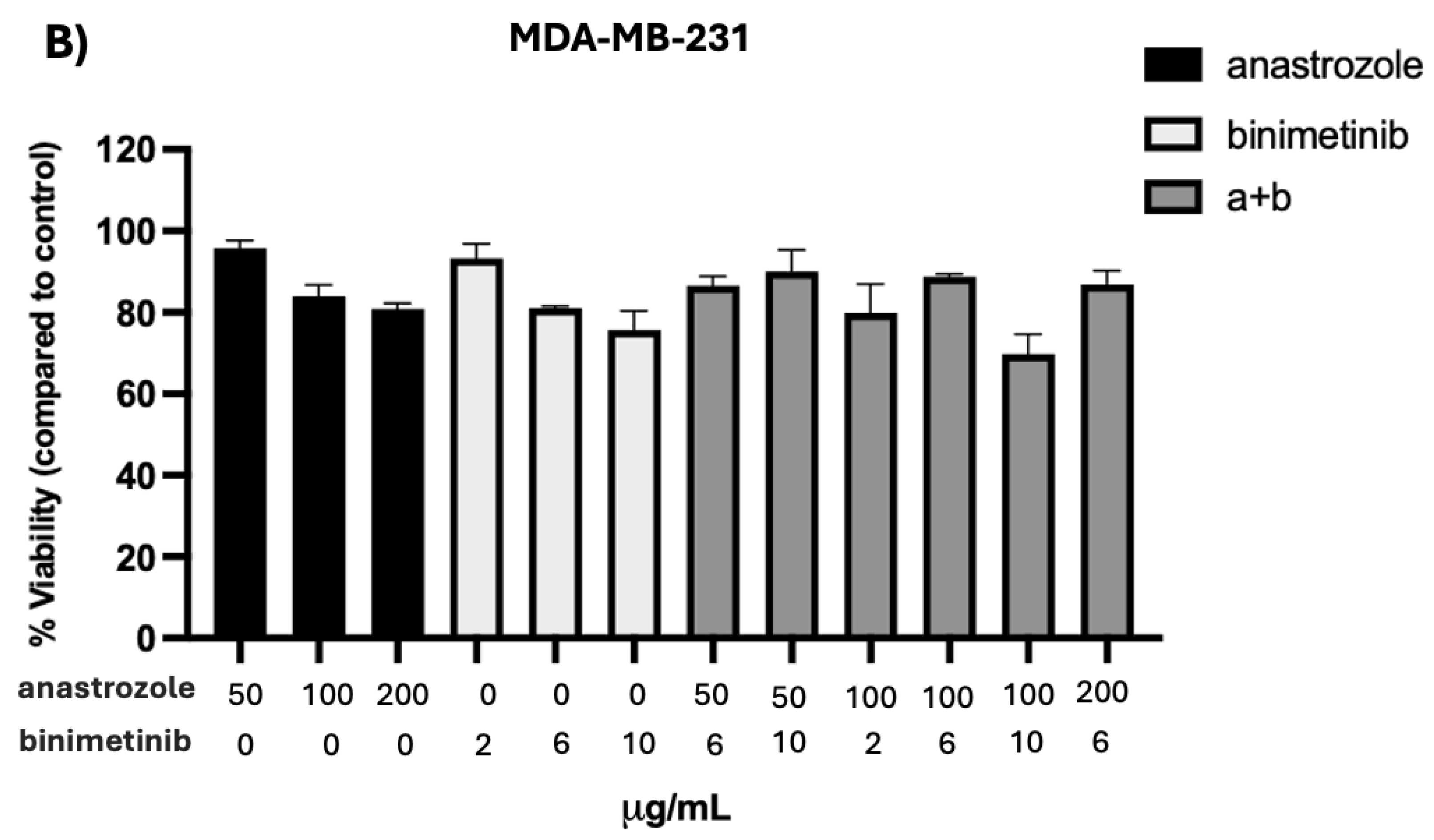
3.1. Cell Cytotoxicity (MTT)
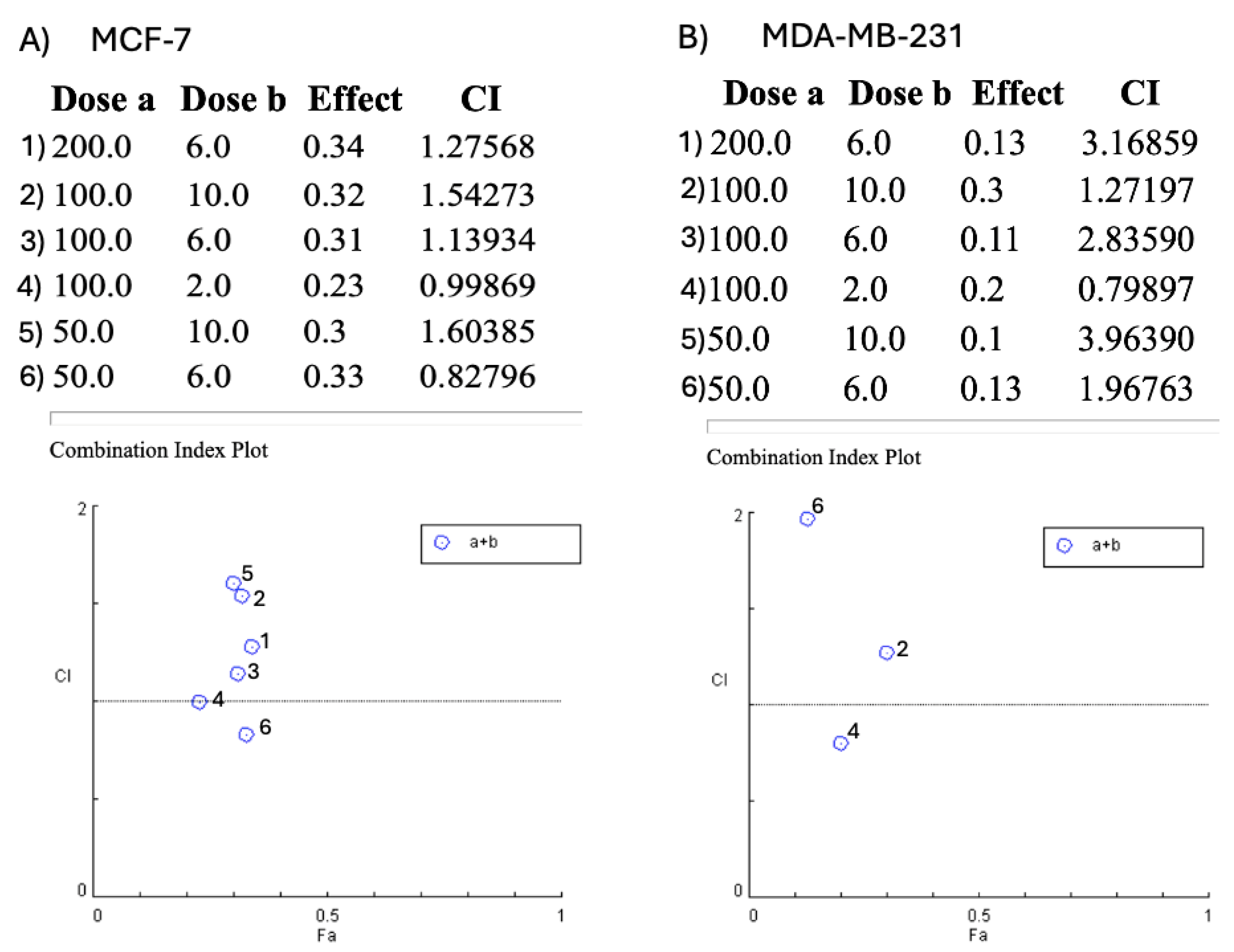
3.2. Drug–Drug Interaction (Combination Index)
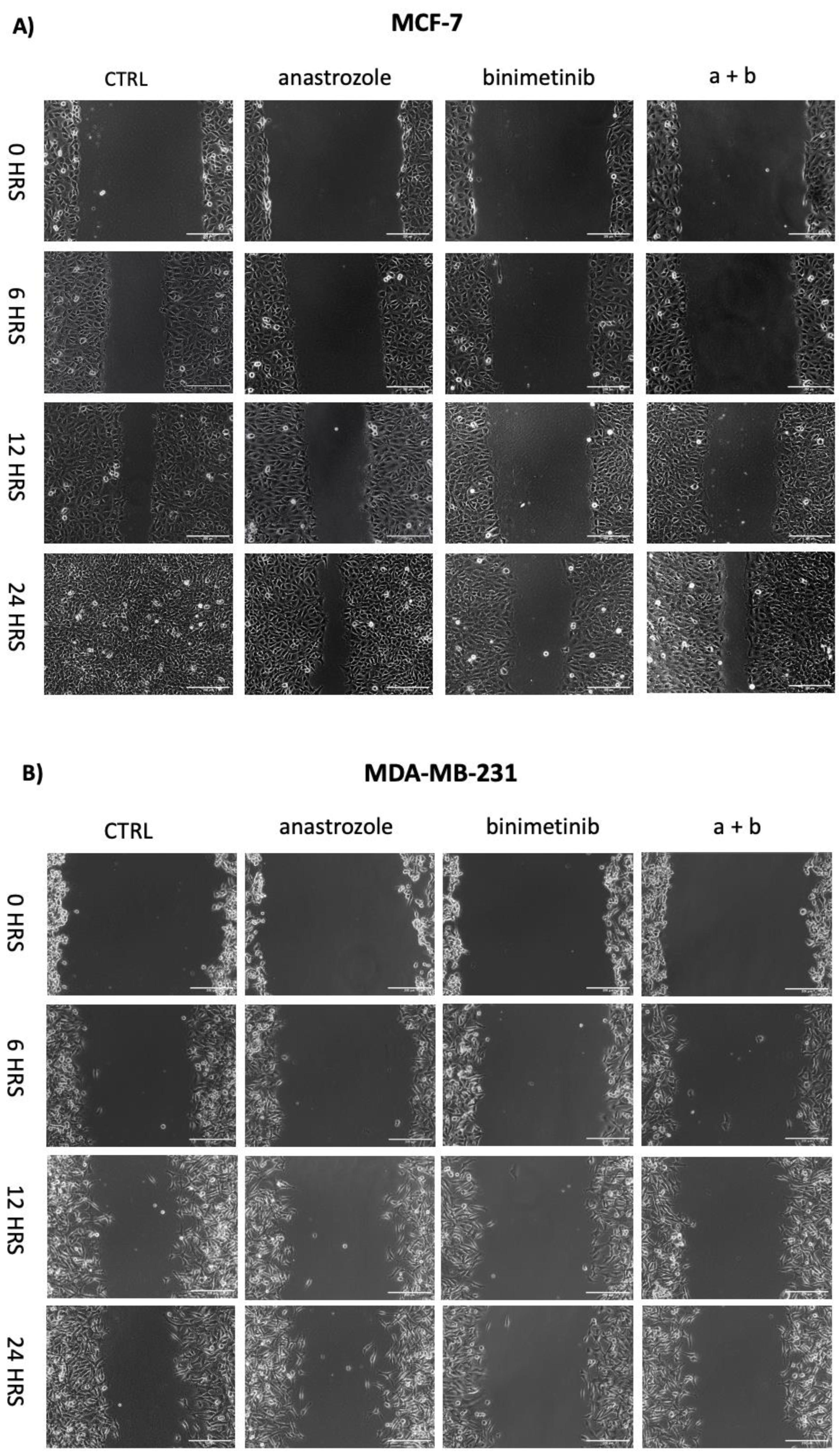
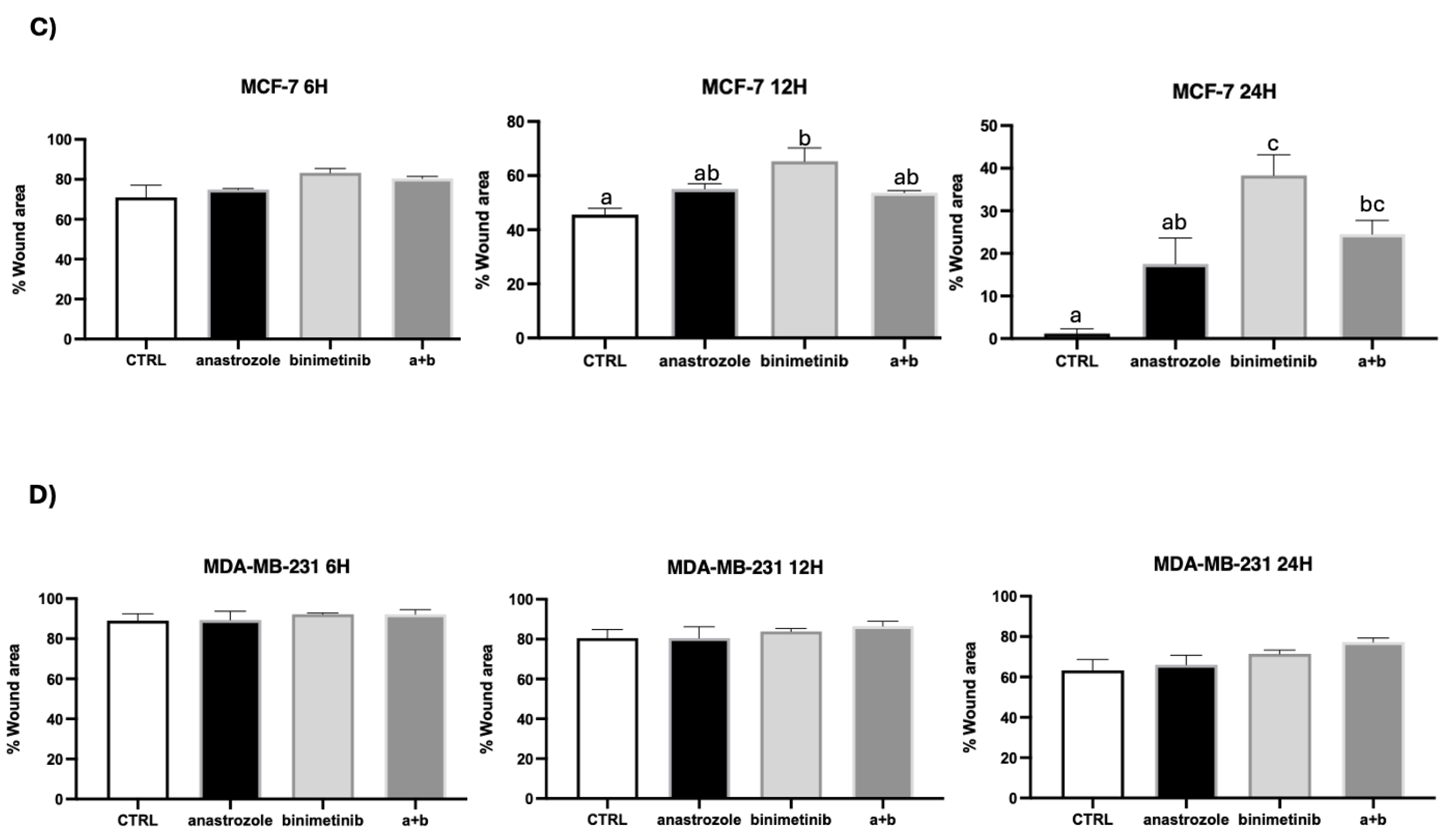
3.3. Cell Migration by Wound-Healing Assay
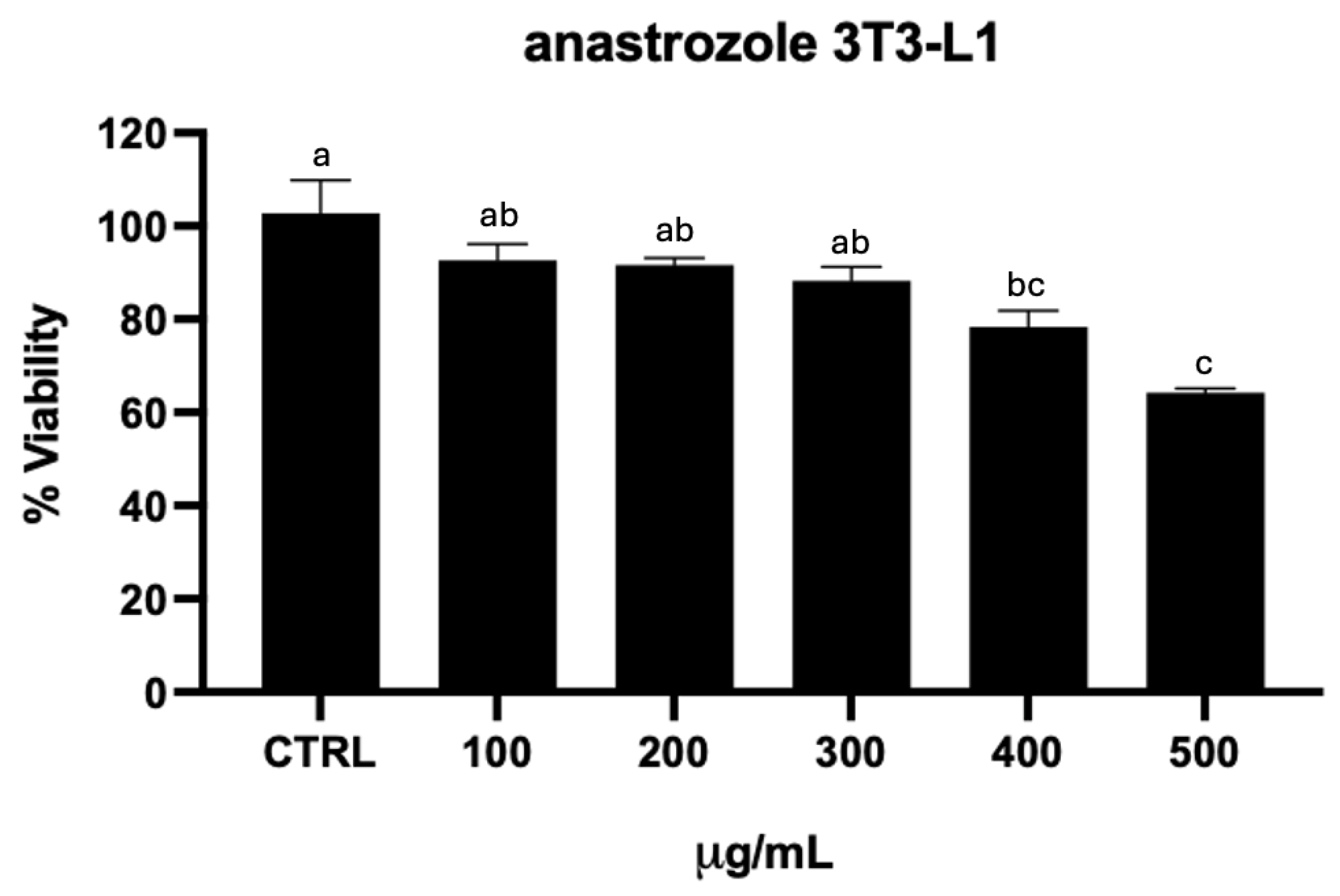
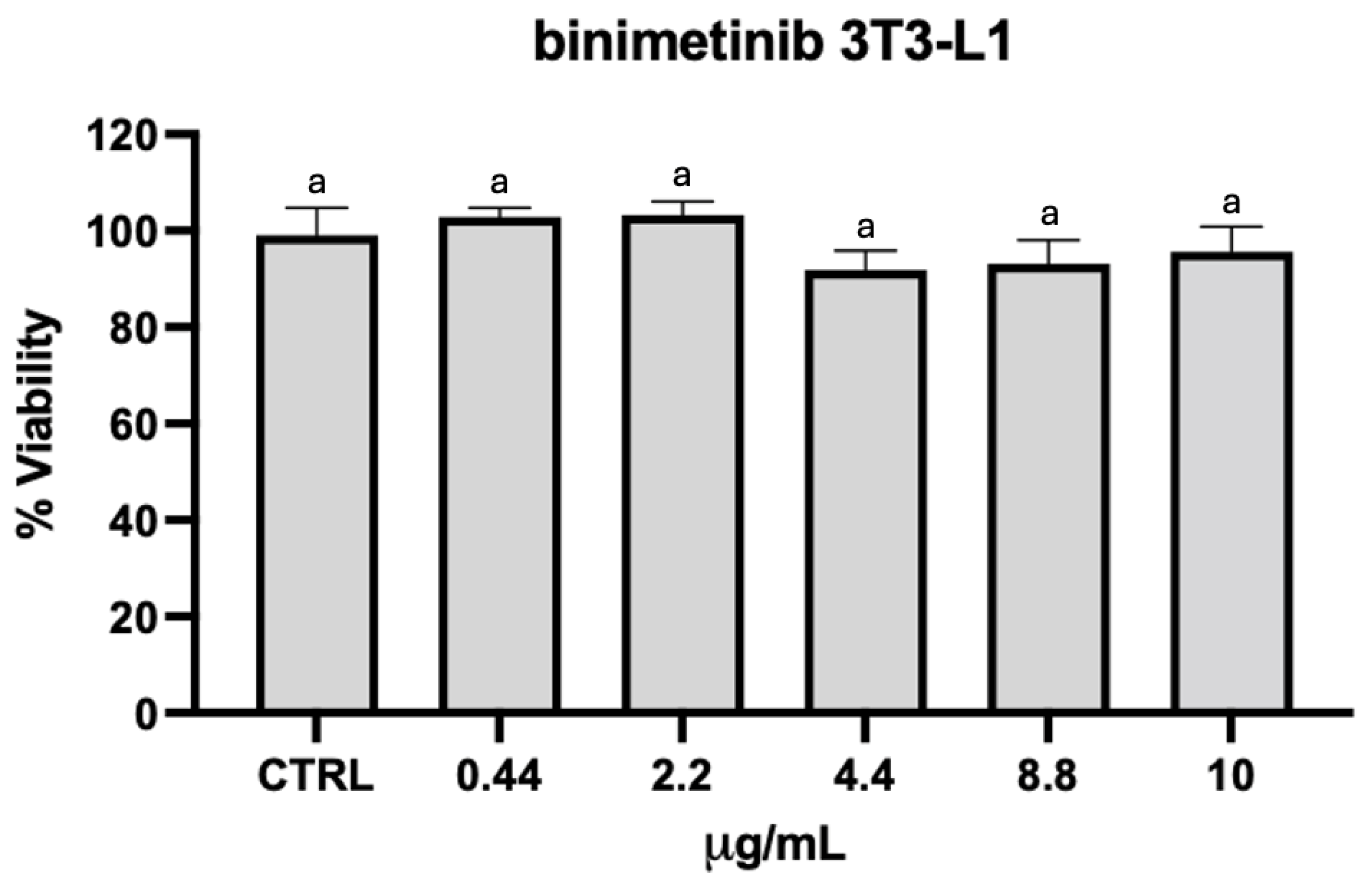
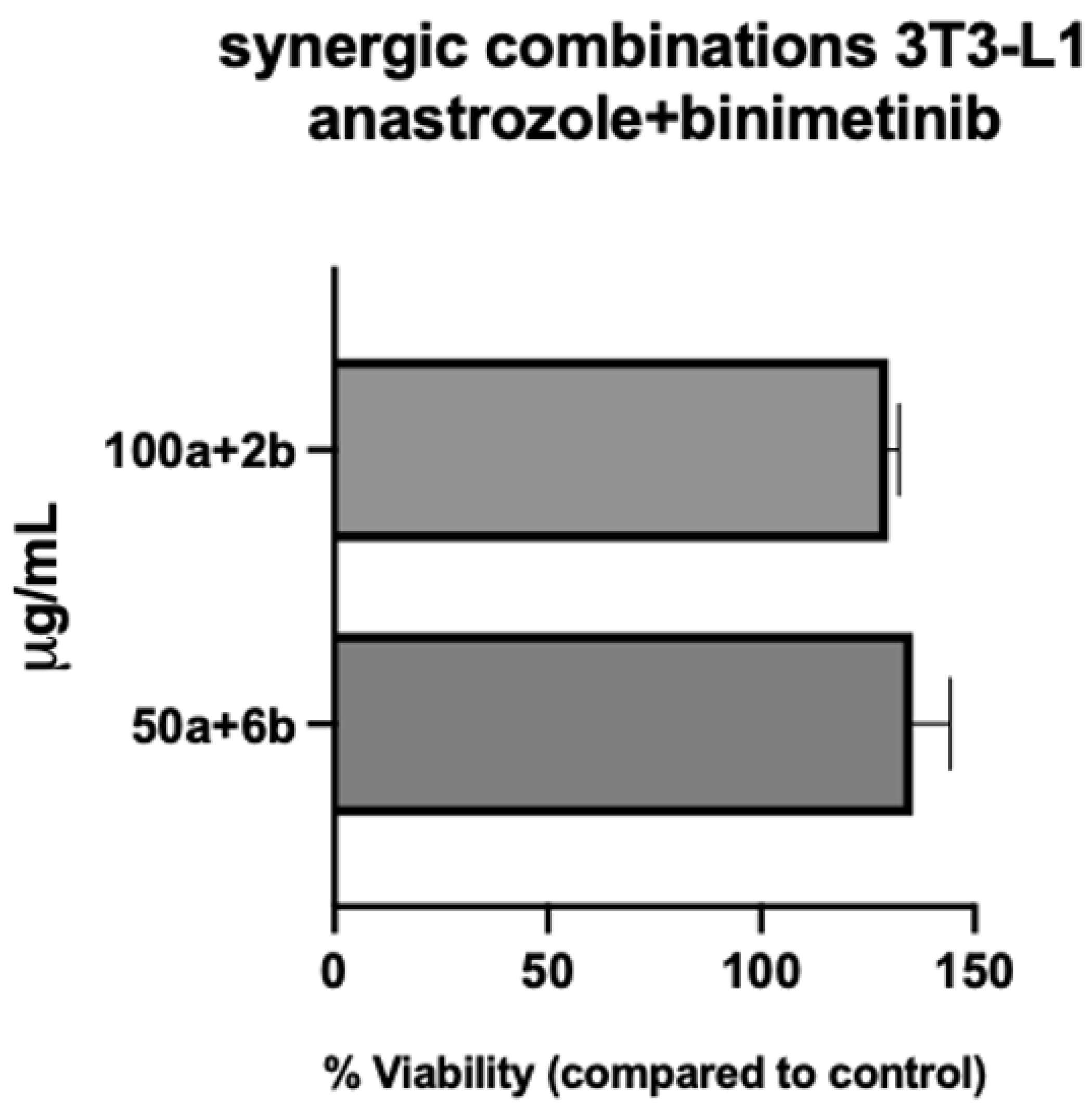
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABC | ATP-binding cassette |
| ABCB1 | ATP-binding cassette P-glycoprotein |
| DMSO | Dimethyl sulfoxide |
| ER | Estrogen receptor |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide |
| TNBC | triple-negative breast cancer |
References
- Oms, 2022.
- Asleh, K.; Riaz, N.; Nielsen, T.O. Heterogeneity of triple negative breast cancer: Current advances in subtyping and treatment implications. J. Exp. Clin. Cancer Res. 2022, 41, 265. [CrossRef]
- Kinnel, B.; Singh, S.K.; Oprea-Ilies, G.; Singh, R. Targeted Therapy and Mechanisms of Drug Resistance in Breast Cancer. Cancers 2023, 15, 1320. [CrossRef]
- Gervasini G, Jara C, Olier C, Romero N, Martínez R, Carrillo JA. Polymorphisms in ABCB1 and CYP19A1 genes affect anastrozole plasma concentrations and clinical outcomes in postmenopausal breast cancer patients. Br J Clin Pharmacol. 2017 Mar;83(3):562-571. doi: 10.1111/bcp.13130.
- Wang, Y.; Minden, A. Current Molecular Combination Therapies Used for the Treatment of Breast Cancer. Int. J. Mol. Sci. 2022, 23, 11046. [CrossRef]
- Herrera-Abreu, M.T.; Guan, J.; Khalid, U.; et al. Inhibition of GPX4 enhances CDK4/6 inhibitor and endocrine therapy activity in breast cancer. Nat. Commun. 2024, 15, 9550. [CrossRef]
- Swain, S.M.; Shastry, M.; Hamilton, E. Targeting HER2-positive breast cancer: advances and future directions. Nat. Rev. Drug Discov. 2023, 22, 101–126. [CrossRef]
- Ye, J.; Wu, J.; Liu, B. Therapeutic strategies of dual-target small molecules to overcome drug resistance in cancer therapy. Biochim. Biophys. Acta Rev. Cancer 2023, 1878, 188866. [CrossRef]
- Gautam, S.; Maurya, R.; Vikal, A.; Patel, P.; Thakur, S.; Singh, A.; Gupta, G.D.; Kurmi, B.D. Understanding drug resistance in breast cancer: Mechanisms and emerging therapeutic strategies. Med. Drug Discov. 2025, 26, 100210. [CrossRef]
- Zivadinovic, D.; Gametchu, B.; Watson, C.S. Membrane estrogen receptor-α levels in MCF-7 breast cancer cells predict cAMP and proliferation responses. Breast Cancer Res. 2004, 7, R101. [CrossRef]
- Telang, N.T. The Divergent Effects of Ovarian Steroid Hormones in the MCF-7 Model for Luminal A Breast Cancer: Mechanistic Leads for Therapy. Int. J. Mol. Sci. 2022, 23, 4800. [CrossRef]
- Cairns, J.; Ingle, J.N.; Dudenkov, T.M.; Kalari, K.R.; Carlson, E.E.; Na, J.; Buzdar, A.U.; Robson, M.E; Ellis, M.J.; Goss, P.E.; Shepherd, L.E.; Goodnature, B.; Goetz, M.P.; Weinshilboum, R.M.; Li, H.; Bari, M.G.; Wang, L. Pharmacogenomics of aromatase inhibitors in postmenopausal breast cancer and additional mechanisms of anastrozole action. JCI Insight 2020, 5, e137571. [CrossRef]
- Kun, E.; Tsang, Y.T.M.; Ng, C.W.; Gershenson, D.M.; Wong, K.K. MEK inhibitor resistance mechanisms and recent developments in combination trials. Cancer Treat. Rev. 2021, 92, 102137. [CrossRef]
- Correia, A.S.; Gärtner, F.; Vale, N. Drug combination and repurposing for cancer therapy: the example of breast cancer. Heliyon 2021, 7, e05948. [CrossRef]
- Chu, Y.H.; Huang, Y.C.; Chiu, P.Y.; Kuo, W.H.; Pan, Y.R.; Kuo, Y.T.; Wang, R.H.; Kao, Y.C.; Wang, Y.H.; Lin, Y.F.; Lin, K.T. Combating breast cancer progression through combination therapy with hypomethylating agent and glucocorticoid. iScience 2023, 26, 106597. [CrossRef]
- Thornton, D.; Heck, K.; Patrick, M.; Kromtit, R.; Benedict, C.; Pondugula, S.R.; Shen, J.; Calderón, A.I. Açaí extract and anticancer drug combination promotes synergistic toxicity and apoptosis in MCF-10A cells of breast cancer model. J. Ethnopharmacol. 2025, 353, 120361. [CrossRef]
- Rushing, B.R.; Molina, S.; Sumner, S. Metabolomics Analysis Reveals Altered Metabolic Pathways and Response to Doxorubicin in Drug-Resistant Triple-Negative Breast Cancer Cells. Metabolites 2023, 13, 865. [CrossRef]
- Obidiro, O.; Battogtokh, G.; Akala, E.O. Triple Negative Breast Cancer Treatment Options and Limitations: Future Outlook. Pharmaceutics 2023, 15, 1796. [CrossRef]
- Moon, A. Ras Signaling in Breast Cancer. In Translational Research in Breast Cancer; Noh, D.Y., Han, W., Toi, M., Eds.; Advances in Experimental Medicine and Biology; Springer: Singapore, 2021; Volume 1187. [CrossRef]
- Lei, B.; Sun, S.; Zhang, X.; Feng, C.; Xu, J.; Wen, Y.; Huang, Y.; Wu, M.; Yu, Y. Bisphenol AF exerts estrogenic activity in MCF-7 cells through activation of Erk and PI3K/Akt signals via GPER signaling pathway. Chemosphere 2019, 220, 122–131. [CrossRef]
- Zhang, H.P.; Jiang, R.Y.; Zhu, J.Y.; Sun, K.N.; Huang, Y.; Zhou, H.H.; Zheng, Y.B.; Wang, X.J. PI3K/AKT/mTOR signaling pathway: an important driver and therapeutic target in triple-negative breast cancer. Breast Cancer 2024, 31, 539–551. [CrossRef]
- Elwakeel, A.; Soudan, H.; Eldoksh, A.; Shalaby, M.; Eldemellawy, M.; Ghareeb, D.; Abouseif, M.; Fayad, A.; Hassan, M.; Saeed, H. Implementation of the Chou-Talalay method for studying the in vitro pharmacodynamic interactions of binary and ternary drug combinations on MDA-MB-231 triple negative breast cancer cells. Synergy 2019, 8, 100047. [CrossRef]
- Acosta-Casique, A.; Montes-Alvarado, J.B.; Barragán, M.; Larrauri-Rodríguez, K.A.; Perez-Gonzalez, A.; Delgado-Magallón, A.; Millán-Perez-Peña, L.; Rosas-Murrieta, N.H.; Maycotte, P. ERK activation modulates invasiveness and Reactive Oxygen Species (ROS) production in triple negative breast cancer cell lines. Cell. Signal. 2023, 101, 110487. [CrossRef]
- Yoshikawa, Y. RAS Inhibition Suppresses the Progression and Metastasis of Triple-Negative Breast Cancer. Ph.D. Thesis, Kobe University, Kobe, Japan, 2024.
- Wang, D.; Zhang, Y.; Li, Q.; Li, Y; Li, W.; Zhang, A.; Xu, J.; Meng, J.; Tang, L.; Lyu, S. Epigenetics: Mechanisms, potential roles, and therapeutic strategies in cancer progression. Genes Dis. 2023, 11, 101020. [CrossRef]
- Molehin, D.; Rasha, F.; Rahman, R.L.; et al. Regulation of aromatase in cancer. Mol. Cell. Biochem. 2021, 476, 2449–2464. [CrossRef]
- Bilir, B.; Kucuk, O.; Moreno, C.S. Wnt signaling blockage inhibits cell proliferation and migration, and induces apoptosis in triple-negative breast cancer cells. J. Transl. Med. 2013, 11, 280. [CrossRef]
- Xu, X.; Zhang, M.; Xu, F.; et al. Wnt signaling in breast cancer: biological mechanisms, challenges and opportunities. Mol. Cancer 2020, 19, 165. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



