Submitted:
28 November 2025
Posted:
01 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Blood Collection, Processing, and Storage
2.3. Biochemical and Hematological Analyses
2.4. Cardiovascular, Inflammatory, and Oxidative Biomarkers
2.5. Lp-PLA₂ (PLA2G7) mRNA Expression by RT-qPCR
| Title 1 | Title 2 | Title 3 |
| IL-6 | 5'- CAC CGG GAA CGA AAG AGA AG -3' 5'- GGG CGG CTA CAT CTT TGG AAT C -3 |
For Rev |
| TNF-α |
5'- AAG AAT TCA AAC TGG GGC CT -3' 5'- GAG GAA GGC CTA AGG TCC AC -3' |
For Rev |
| Lp-PLA₂ | 5'- CCA CCC AAA TTG CAT GTG C -3' 5'- GCC AGT CAA AAG GAT AAA CCA CA -3' |
For Rev |
| GAPDH | 5'- CAA GGT CAT CCA TGA CAA CTT TG -3' 5'- GTC CAC CAC CCT GTT GCT GTA G -3' |
For Rev |
2.6. Statistical Analysis
3. Results
3.1. Study population and analysis sets
| Healthy | CVD | P-value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Age | 53+14 | 55+10 | |||||||
| BMI | 26+1 | 29+5 | 0.01 | ||||||
| Hb (g/dL) | 14+1 | 14+3 | |||||||
| Glucose (mmol/L) | 5.8+1.5 | 6.8+2 | 0.05 | ||||||
| Cholesterol (mmol/L) | 3.6+0.5 | 4.2+0.6 | 0.001 | ||||||
| HDL (mmol/L) | 1.3+0.3 | 1.1+0.1 | 0.001 | ||||||
| LDL (mmol/L) | 1.8+0.6 | 3+0.6 | 0.00001 | ||||||
| Triglycerides(mmol/L) | 1.3+0.5 | 1.3+0.5 | |||||||
| ALT (IU/L) | 22+7 | 38+30 | 0.05 | ||||||
| Creatinine (umol/L) | 81+12 | 107+24 | 0.001 | ||||||
| Urea (mmol/L) | 4.8+1.2 | 10+5 | 0.01 | ||||||
| Healthy (mean±SD) | CVD (mean±SD) | P-vaule | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WBC (10e3/uL) | 7+1.6 | 10.6+5 | 0.001 | ||||||||||||
| RBC (10e6/uL) | 5+0.35 | 4.7+0.9 | |||||||||||||
| Hematocrit % | 43+3 | 43+8 | |||||||||||||
| MCV (fL) | 83+4 | 90+3.7 | 0.0001 | ||||||||||||
| MCH (pg) | 27+2 | 29+2 | 0.01 | ||||||||||||
| MCHC (g/dL) | 33+1.2 | 32+1.3 | 0.01 | ||||||||||||
| RDW-CV % | 13+1.5 | 12+2 | 0.05 | ||||||||||||
| MPV (fL) | 10+0.6 | 8+1.2 | 0.001 | ||||||||||||
| Platelets (10e3/uL) | 268+50 | 275+103 | |||||||||||||
| Neutrophils % | 47+11 | 65+11 | 0.0001 | ||||||||||||
| Lymphocytes % | 40+10 | 22+11 | 0.0001 | ||||||||||||
| Monocytes % | 8+1.8 | 8.6+2.3 | |||||||||||||
| Eosinophils % | 3+1.3 | 2.3+1.3 | 0.05 | ||||||||||||
| Basophils % | 0.7+0.2 | 0.8+0.3 | 0.05 | ||||||||||||
| Measure | Healthy (mean±SD) | CVD (mean±SD) | p-value |
|---|---|---|---|
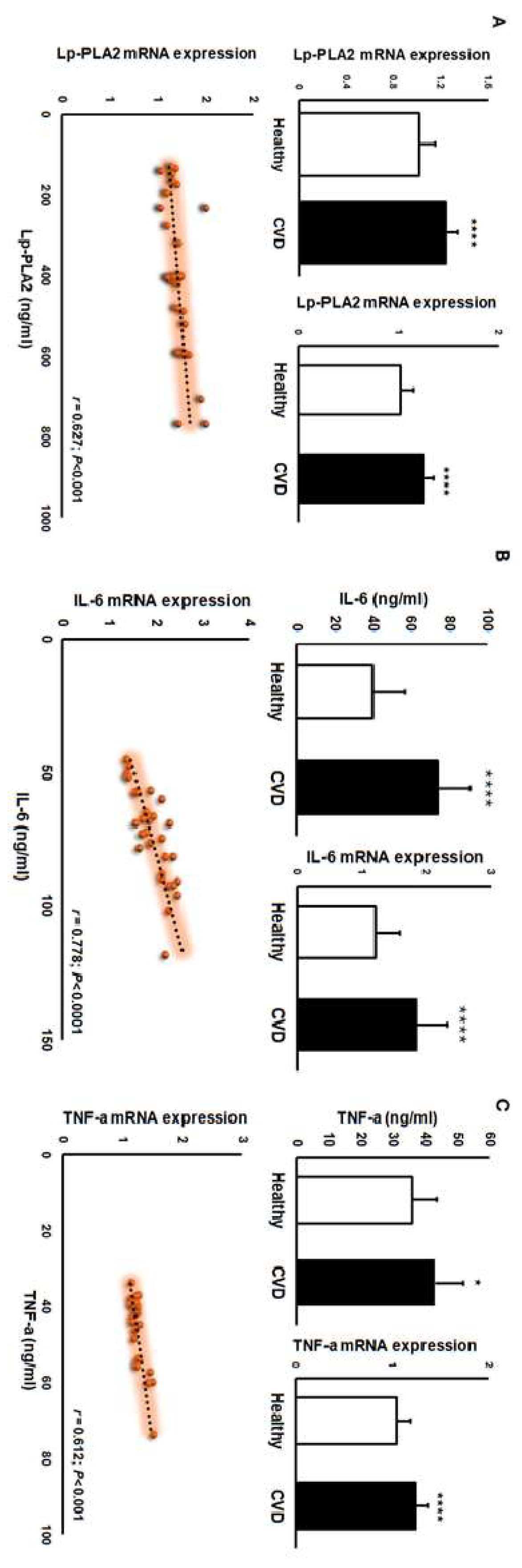
| Lp-PLA2 (ng/ml) | 101+31 | 419+185 | 0.0001 |
| IL-6 (ng/ml) | 40+17 | 74+17 | 0.0001 |
| TNF-a (ng/ml) | 3.6+0.8 | 4.3+0.9 | 0.0001 |
| ox-LDL (nmol/ml) | 242+66 | 430+147 | 0.0001 |
| LP-a (nmol/L) | 90+33 | 134+31 | 0.0001 |
| FABP3 (ng/ml) | 3.1+0.1 | 3.4+1 | ns |
3.2. Baseline clinical, biochemical and hematological characteristics
3.3. Inflammatory and oxidative biomarkers
3.4. Paired protein–mRNA analyses
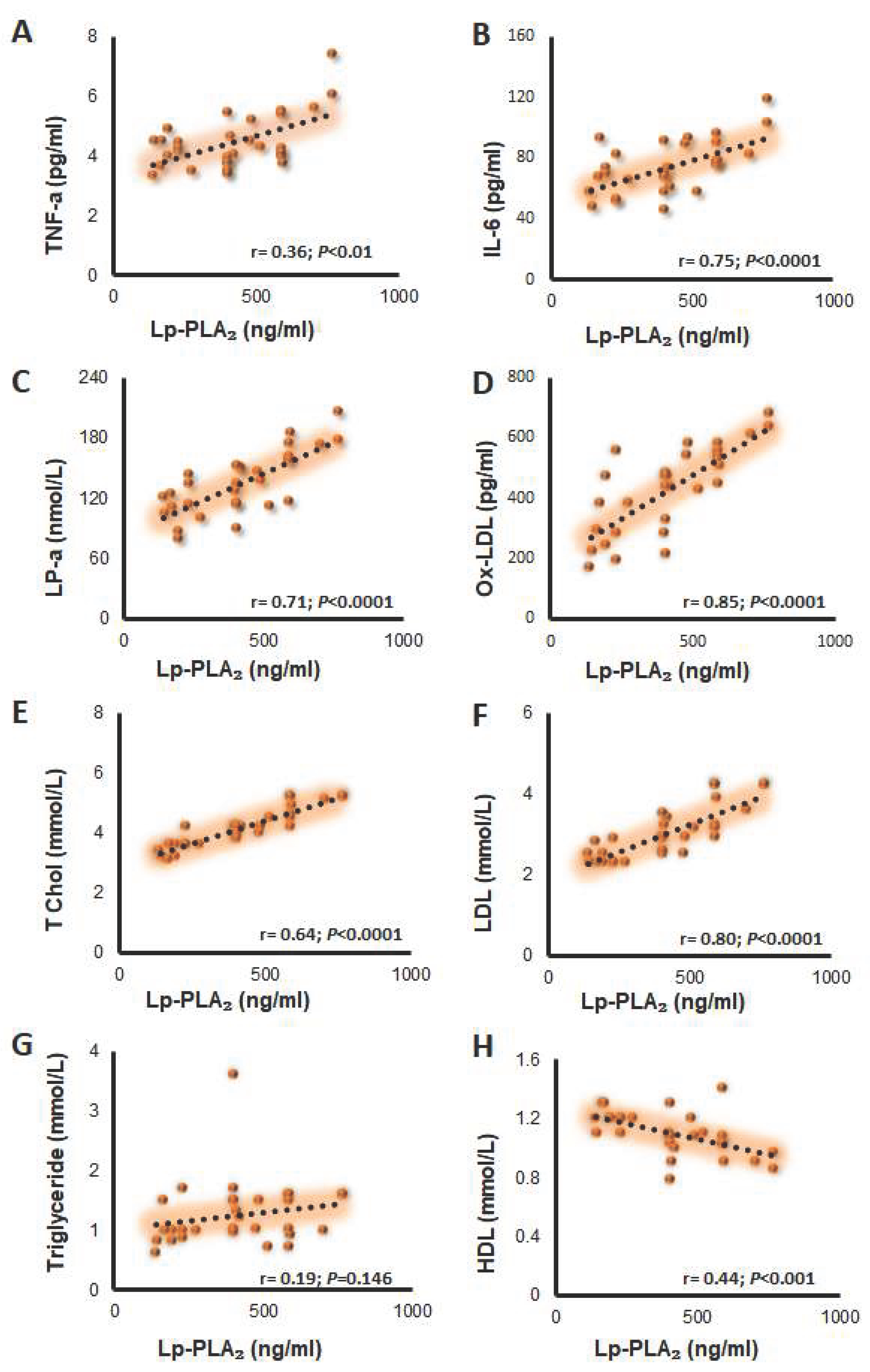
3.5. Associations between Lp-PLA2 concentration and circulating lipid profiles, inflammatory, and oxidative biomarkers
3.6. Discrimination of CVD from healthy
4. Discussion
5. Conclusions
Author Contributions
Disclosures
Sources of Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
References
- World Health Organization. Cardiovascular diseases (CVDs) Fact Sheet. 31 Jul 2025.
- Rosenson RS, Stafforini DM. Lipoprotein-Associated and Secreted Phospholipases A₂ in Cardiovascular Disease. Circulation. 2011;124:2749–2769. [CrossRef]
- Tselepis AD, et al. Pathophysiological Role and Clinical Significance of Lp-PLA₂ bound to LDL and HDL. Curr Med Chem. 2014;20(40):6256–6269. [CrossRef]
- O’Donoghue M, et al. Lp-PLA₂ in Acute Coronary Syndrome: distribution across lipoprotein classes. Can J Cardiol. 2013;29(10):1256–1262.
- The Lp-PLA₂ Studies Collaboration. Meta-analysis of observational studies of Lp-PLA₂ mass/activity and CVD risk. Eur J Cardiovasc Prev Rehabil. 2007;14(1):3–11.
- The Emerging Risk Factors Collaboration. Lp-PLA₂ and risk of coronary disease and stroke: collaborative analysis of 32 prospective studies. Lancet. 2010;375:1536–1544. [CrossRef]
- Temporal profile and prognostic value of Lp-PLA₂ mass and activity in acute stroke. Atherosclerosis. 2012;220(2):394–399. [CrossRef]
- Chaudhary R, et al. Biochemical differences in the mass vs activity tests of Lp-PLA₂. Clin Chim Acta. 2017;471:32–37. [CrossRef]
- Kolodgie FD, et al. Lp-PLA₂ protein expression in human coronary atheroma: association with high-risk lesions. Arterioscler Thromb Vasc Biol. 2006;26(7):e164–e171. [CrossRef]
- The STABILITY Investigators. Darapladib for preventing ischemic events in stable CHD. N Engl J Med. 2014;370:1702–1711. [CrossRef]
- O’Donoghue ML, et al. Effect of darapladib on major coronary events after ACS (SOLID-TIMI 52). JAMA. 2014;312(10):1006–1015. [CrossRef]
- Holmes MV, Talmud PJ. Deciphering the causal role of sPLA₂s and Lp-PLA₂ in CHD (Mendelian randomization). Arterioscler Thromb Vasc Biol. 2015;35:2281–2289. [CrossRef]
- Patel RS, et al. Carriage of the V279F null allele of PLA2G7 and coronary risk in East Asians. PLoS One. 2011;6(4):e18208. [CrossRef]
- Wang L, et al. Mechanisms of lysophosphatidylcholine in atherosclerosis. Life Sci. 2020;247:117443.
- Ismaeel S, Qadri A. ATP release drives inflammation with lysophosphatidylcholine. ImmunoHorizons. 2021;5(4):219–233. [CrossRef]
- Luigi M, et al. Role of free fatty acids in endothelial dysfunction. J Biomed Sci. 2017;24:50. [CrossRef]
- van der Valk FM, et al. Oxidized phospholipids on Lp(a) elicit arterial wall inflammation in humans. Circulation. 2016;134:611–624. [CrossRef]
- Tsimikas S, et al. Oxidized phospholipid modification of Lp(a): epidemiology, genetics, and pathophysiology. Atherosclerosis. 2022;349:76–86. [CrossRef]
- Packard CJ, Libby P. Inflammation in atherosclerosis: from vascular biology to biomarker discovery and risk prediction. Clin Chem. 2008;54(1):24–38. [CrossRef]
- Hansson GK, Hermansson A. The immune system in atherosclerosis. Nat Immunol. 2011;12(3):204–212. [CrossRef]
- Libby P, Ridker PM, Hansson GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473(7347):317–325. [CrossRef]
- Dong S, Zhou Y, Gao X, Xu F, Liu D, Lu X, et al. Increased Lp-PLA2 activity is associated with inflammatory markers and atherosclerotic burden in patients with coronary artery disease. BMC Cardiovasc Disord. 2023;23:45.
- Ferguson JF, Matthews GJ, Townsend RR, et al. Genetic variants in the Lp-PLA2/PLA2G7 gene are associated with plasma Lp-PLA2 levels and cardiovascular risk. J Lipid Res. 2012;53(11):2436–2443.
- Jackisch L, Hummel J, Schulze MB, et al. Circulating inflammatory cytokines and cardiovascular risk: associations of IL-6, IL-1β, TNF-α with subclinical atherosclerosis. Atherosclerosis. 2018;278:21–27.
- Ridker PM, Luscher TF. Anti-inflammatory therapies for cardiovascular disease. Eur Heart J. 2014;35(27):1782–1791. [CrossRef]
- Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med. 1999;340(2):115–126. [CrossRef]
- Ridker PM, et al. Interleukin-6 signaling and the pathogenesis of cardiovascular disease. Atherosclerosis. 2018;269:203–209. [CrossRef]
- Kishimoto T. IL-6: from its discovery to clinical applications. Int Immunol. 2010;22(5):347–352. [CrossRef]
- Tzoulaki I, Murray GD, Lee AJ, Rumley A, Lowe GD, Fowkes FG. Inflammatory markers and the risk of coronary heart disease: prospective study and meta-analysis. Eur Heart J. 2007;28(22):2743–2749. [CrossRef]
- Macphee CH, Moores KE, Boyd HF, et al. Lipoprotein-associated phospholipase A2, platelet-activating factor acetylhydrolase, generates two bioactive products during the oxidation of low-density lipoprotein. Biochem J. 1999;338:479–487.
- Wilensky RL, et al. Inhibition of lipoprotein-associated phospholipase A2 reduces complex coronary atherosclerotic plaque development. Nat Med. 2008;14(10):1059–1066. [CrossRef]
- Tellis CC, Tselepis AD. The role of lipoprotein-associated phospholipase A2 in atherosclerosis may depend on its lipoprotein carrier in plasma. Biochim Biophys Acta. 2009;1791(5):327–338. [CrossRef]
- Zalewski A, Macphee C. Role of lipoprotein-associated phospholipase A2 in atherosclerosis. J Clin Invest. 2005;115(12):3380–3387. [CrossRef]
- Tsimikas S, et al. Lipoprotein(a): novel target and emerging biomarker in cardiovascular disease. J Am Coll Cardiol. 2018;71(2):177–192.
- Mehta JL, Chen J, Hermonat PL, Romeo F, Novelli G. Lectin-like, oxidized low-density lipoprotein receptor-1 (LOX-1): a critical player in the development of atherosclerosis and related disorders. Cardiovasc Res. 2006;69(1):36–45. [CrossRef]
- Bergmark C, Dewan A, Orsoni A, et al. A novel function of lipoprotein(a) as a preferential carrier of oxidized phospholipids in human plasma. J Lipid Res. 2008;49(10):2230–2239. [CrossRef]
- Tabas I, Garcia-Cardena G, Owens GK. Recent insights into the cellular biology of atherosclerosis. J Cell Biol. 2015;209(1):13–22. [CrossRef]
- Libby P, Buring JE, Badimon L, et al. Inflammation and atherosclerosis: report from a scientific session. Atherosclerosis. 2019;283:91–100.
- Tedgui A, Mallat Z. Cytokines in atherosclerosis: pathogenic and regulatory pathways. Physiol Rev. 2006;86(2):515–581. [CrossRef]
- Monaco C, Andreakos E, Kiriakidis S, et al. Canonical pathway of nuclear factor kappaB activation selectively regulates proinflammatory and prothrombotic responses in human atherosclerosis. Proc Natl Acad Sci U S A. 2004;101(15):5634–5639. [CrossRef]
- White HD, Held C, Stewart R, et al. Darapladib for preventing ischemic events in stable coronary heart disease. N Engl J Med. 2014;370(18):1702–1711. 1702. [CrossRef]
- O’Donoghue ML, Braunwald E, White HD, et al. Effect of darapladib on major coronary events after an acute coronary syndrome: the SOLID-TIMI 52 randomized clinical trial. JAMA. 2014;312(10):1006–1015.
- Packard CJ, et al. Lipoprotein-associated phospholipase A2 as an independent predictor of coronary heart disease. N Engl J Med. 2000;343(16):1148–1155. [CrossRef]
- Mallat Z, Lambeau G, Tedgui A. Lipoprotein-associated and secreted phospholipases A2 in cardiovascular disease: roles as biological effectors and biomarkers. Circulation. 2010;122(21):2183–2200. [CrossRef]
- Steg PG, Bhatt DL, Hamm CW, et al. Clinical outcomes with Lp-PLA2 inhibition after acute coronary syndromes. Atherosclerosis. 2019;286:1–9.
- Sabatine MS, et al. Lipid, inflammatory, and metabolic biomarkers and the risk of recurrent cardiovascular events. Circulation. 2012;125(4):450–458.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





