Submitted:
05 November 2025
Posted:
06 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Systematize the biochemical and biophysical basis of biophoton emission.
- Present the experimental methods currently in use and their limitations.
- Evaluate quantum biological theories and their empirical basis.
- Review the possibilities of cell-level and intercellular light-based communication.
- Present scientific evidence for photon emission as a means of DNA communication.
- Identify currently open questions and suggest directions for further research.
2. Biological Mechanisms of Photon Emission
2.1. Biochemical Background
2.2. Photon Emission at the Cellular Level
- -
- -
2.3. Open Questions
- -
- Which specific molecular reactions are responsible for the highest photon emission? Is it possible to selectively inhibit or enhance these reactions?
- -
- To what extent is photon emission cell type-dependent? Are there cell lines that emit light particularly strongly or regularly?
- -
- How does the internal architecture of the cell (e. g., the spatial distribution of mitochondria) affect the emission pattern?
- -
- Is real-time, spectrally selective mapping of photon emission from cells in vitro and in vivo possible?
3. Experimental Approaches and Challenges
3.1. Measurement Techniques
3.1.1. Photon Detection Systems
- -
- Photomultiplier tubes (PMTs):
- -
- Cooled CCD cameras (Charge-Coupled Device):
- -
- SPAD (Single-Photon Avalanche Diodes):
- -
- CMOS (Complementary Metal-Oxide Semiconductor)
3.1.2. Environmental and Measurement Conditions
- -
- Darkroom: Complete exclusion of environmental photons is necessary – often with multi-layer shielding, vibration-free flooring, and thermostatic control.
- -
- Calibration: It is important to know the exact dark count and the spectral response of the detector.
- -
- Temperature and pH control: Cell metabolism and thus photon emission are extremely sensitive to environmental factors.
3.2. Samples and Experimental Protocols
- -
- In vitro cell cultures: provide a standardized, well-controlled environment. Common models: HeLa, fibroblasts, plant cells.
- -
- Isolated cell organelles: e. g., mitochondria or membrane fragments for targeted biochemical analysis.
- -
- Tissue samples or whole living organisms (e. g., zebrafish embryos, mouse embryos, Arabidopsis leaves): for studying physiological relevance.
- -
- Sample preparation is a critical step: mechanical stress, oxidative environment, or metabolic disturbances can falsely increase emissions.
- -
- Low signal-to-noise ratio: Even under ideal conditions, it is difficult to separate true photon emission from background noise.
- -
- Lack of standardization: Research groups use different measurement procedures, which makes comparison difficult.
- -
- Environmental effects: Temperature, humidity, and electromagnetic noise can affect the accuracy of measurements.
- -
- Biological variability: The natural heterogeneity and dynamic state of cells pose a challenge to reproducibility.
4. Quantum Biological Interpretations
4.1. The Coherent Biophoton Theory
- -
- A low-entropy photon field in which the temporal and spatial patterns of photon emission are not completely random.
- -
- The photon emission spectrum may also contain monochromatic components, especially during certain physiological processes.
- -
- Cells, as quantum oscillators, may be capable of generating light pulses and synchronizing with each other.
4.2. Quantum Coherence and Quantum Information in Living Systems
- -
- Photosynthesis: light energy can be transferred in a quantum coherent manner in photosynthetic complexes (e. g., FMO complexes), as confirmed by femtosecond spectroscopy [57].
- -
- -
- -
- Photons may function not only as emissions but also as internal communication signals.
- -
- Cells may behave as a quantum network, where electromagnetic fields synchronize activity.
- -
- Emission may follow nonlinear dynamics, which can also be observed in chaotic or fractal patterns [46].
4.3. Critical Remarks and Methodological Issues
- -
- Coherence time: biological systems operate in a thermal, noisy environment where the duration of quantum coherence is extremely short—typically on the order of picoseconds.
- -
- Lack of empirical evidence: few direct measurements confirm coherent biophoton emission. Measurements are sensitive to noise, and statistical processing requires great caution.
- -
- Alternative explanations: the observed patterns can often be explained by classical nonlinear systems (e. g., oscillation, stochastic resonance).
- -
- Nevertheless, quantum biological interpretations offer important new perspectives for the study of living systems. To move forward, multidisciplinary experimental protocols combining quantum optical, biophysical, and biochemical methods are needed.
5. Light-Based Communication at the Cellular and Intercellular Levels
5.1. Intracellular Light Signals
- -
- Mitochondria: Reactive oxygen species (ROS) generated during cellular respiration and their chemical reactions can lead to the emission of photons. These "internal light points" may also encode temporal and spatial patterns of intracellular activity [63].
- -
- -
5.2. Intercellular Photon Communication
- -
- Gurwitsch's mitogenetic radiation (1920s): long-distance light signaling between the apical regions of onion bulbs, which may have induced cell division.
- -
- Kobayashi et al. [46]: non-chemical interactions between different plant cell cultures were demonstrated, which were reduced by optical shielding.
- -
5.3. Possible Mechanisms
- -
- Emission: The emitter cell is capable of emitting photons in a controlled manner, either in response to environmental stimuli or according to internal rhythms.
- -
- Detection: The receiving cell detects the photons and responds functionally based on these signals. This can be activation via receptors or membrane proteins, or even modification of intracellular signaling pathways.
5.4. The Question of Photon Patterns and Information Content
- -
- The photons emitted by cells can carry information not only based on their quantity, but also on temporal fluctuations (pulsation, oscillation), spectral characteristics (wavelength dispersion, bands), polarization, and other quantum characteristics. Experimental results show that certain photon patterns may be unique to a cell line, developmental stage, or stress state [71]. This information encoding is similar to classical electrical activity patterns (e. g., EEG, action potentials), but occurs with a photonic signal.
6. Present Scientific Evidence for Photon Emission as a Means of DNA Communication
The Interaction Between Photon Emission and DNA
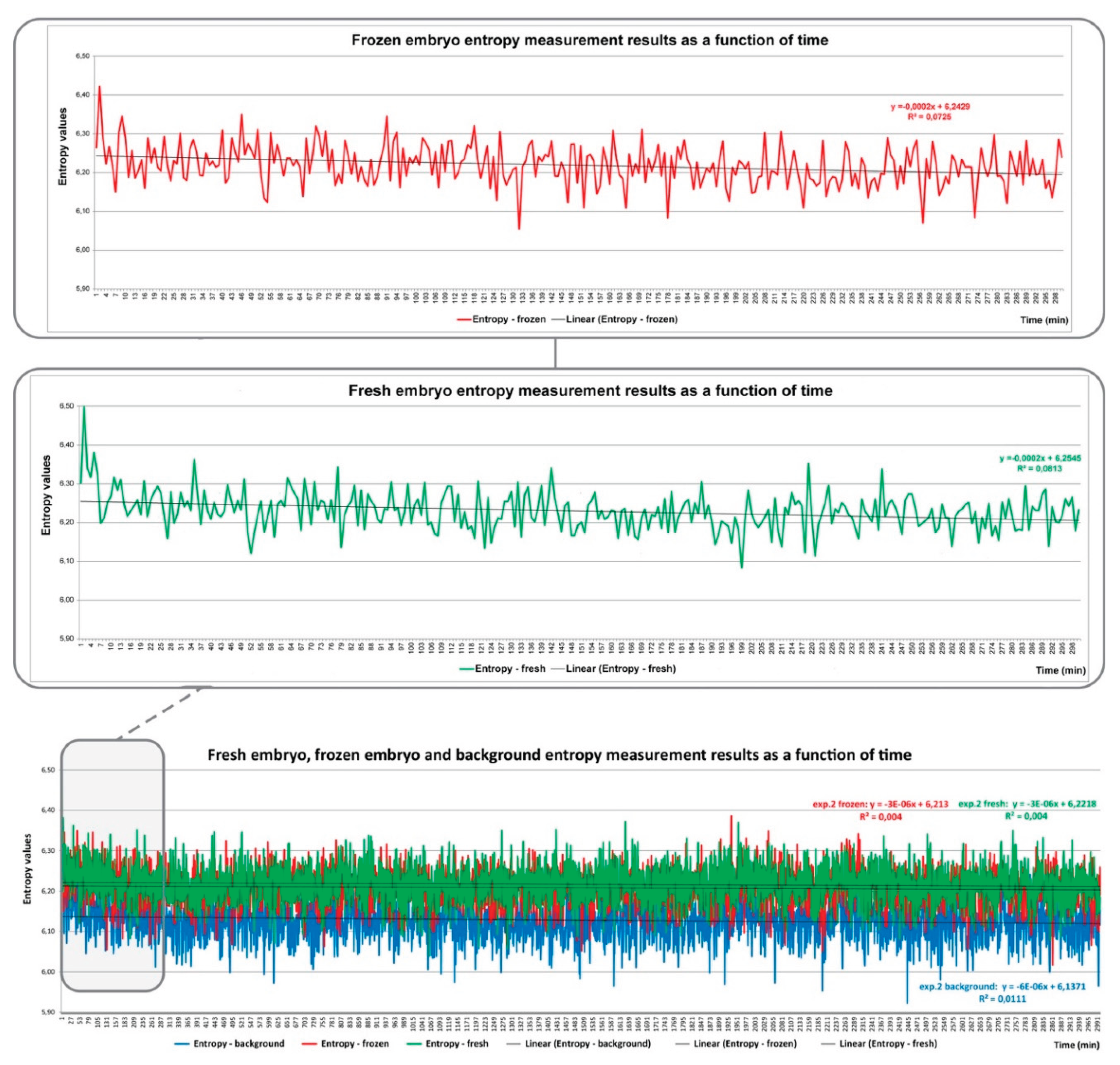
7. Summary of Our Own Research
8. Future Research Directions
9. Conclusions
- -
- understanding coherent processes at the cellular level,
- -
- broadening the toolkit of quantum biology,
- -
- and, in the long term, exploring the physical dimensions of biological information processing.
Acknowledgments
Conflicts of Interest
References
- Steven H D Haddock 1, Mark A Moline, James F Case. Bioluminescence in the sea. Ann Rev Mar Sci. 2010, 2, 443–93. [CrossRef]
- E. A. Widder, Bioluminescence in the Ocean: Origins of Biological, Chemical, and Ecological Diversity, Science. 2010.
- Marieh B Al-Handawi, Srujana Polavaram, Anastasiya Kurlevskaya, Patrick Commins, Stefan Schramm, César Carrasco-López, Nathan M Lui, Kyril M Solntsev, Sergey P Laptenok, Isabelle Navizet, Panče Naumov Spectrochemistry of Firefly Bioluminescence. Chem Rev. 2022, 122, 13207–13234. [CrossRef]
- Niggli, H.J. Ultraweak photons emitted by cells: biophotons. J Photochem Photobiol B. 1992, 14, 144–6. [Google Scholar] [CrossRef] [PubMed]
- Devaraj, M. , and P. Martosubroto (Eds). 1997. Small Pelagic Resources and their Fisheries in the Asia-Pacific region. Proceedings of the APFIC.
- R Vogel , R Süssmuth. Low level chemiluminescence from liquid culture media. J Appl Microbiol. 1999, 86, 999–1007. [CrossRef] [PubMed]
- Niggli, H.J. Temperature dependence of ultraweak photon emission in fibroblastic differentiation after irradiation with artificial sunligIndian J Exp Biol. 2003, 41, 419–23.
- Joseph Tafur, M.D. ,1 Eduard P.A. Van Wijk, Ph.D.,2,3 Roeland Van Wijk, Ph.D.,2,4 and Paul J. Mills, Ph.D. Biophoton Detection and Low-Intensity Light Therapy: A Potential Clinical Partnership. Photomedicine and Laser Surgery Volume 28, Number 1, 2010 ª Mary Ann Liebert, Inc. Pp. 23–30. [CrossRef]
- Ankush Prasad & Pavel Pospíšil. Towards the two-dimensional imaging of spontaneous ultra-weak photon emission from microbial, plant and animal cells. Scientific Reports 2013, 3, 1211.
- Yan, Z. Zhang X. Preliminary study of the human body surface photon emission. Prog. Biochem. Biophysics 1979, 2, 48–52. [Google Scholar]
- Popp et al., Popp FA, Nagi W, Li KH, Scholz W, Weingartner O, Wolf R. Biophoton emission. New evidence for coherence and DNA as source. Cell Biophys. 1984, 6, 33–51. [CrossRef]
- Enrique Cadenas, Helmut Siesi, Ana Campa, Giuseppe Cilento. Electronically Excited States in Microsomal Membranes: Use of Chlorophyll-a As An Indicator of Triplet Carbonyls. Photochemistry and Photobiology. [CrossRef]
- T. I. Quickenden, M. J. T. I. Quickenden, M. J. Comarmond, R. N. Tilbury. Ultra Weak Bioluminescence Spectra of Stationary Phase Saccharomyces cerevisiae And Schizosaccharomyces pombe. 1985. [Google Scholar] [CrossRef]
- Inaba, H. Super-High Sensitivity Systems for Detection and Spectral Analysis of Ultraweak Photon Emission from Biological Cell Cells and Tissues. Experientia 1988, 44, 550–559. [Google Scholar] [CrossRef]
- A.G. Gurwitsch. Die natur des spezifischen erregers der zellteilung. Dev Genes Evol 1923, 100, 11.
- Laager, F. Light based cellular interactions: hypotheses and perspectives Front. Phys., 2015, 3, 2015. [Google Scholar] [CrossRef]
- T I Quickenden 1, R N Tilbury Luminescence spectra of exponential and stationary phase cultures of respiratory deficient Saccharomyces cerevisiae. J Photochem Photobiol B. 1991, 8, 169–74. [CrossRef]
- Gallep, C.M. and dos Santos, S.R. Photon-Counts during Germination of Wheat (Triticum aestivum) in Wastewater Sediment Solutions Correlated with Seedling Growth. Seed Science and Technology 2007, 35, 607–614. [Google Scholar] [CrossRef]
- S Cohen , F A Popp. Biophoton emission of human body. Indian J Exp Biol. 2003, 41, 440–5.
- Schwabl H, Klima H. Spontaneous ultraweak photon emission from biological systems and the endogenous light field. Forsch Komplementarmed Klass Naturheilkd. 2005, 12, 84–9. [CrossRef] [PubMed]
- Pospíšil, P. Prasad A., Rác M. Mechanism of the formation of electronically excited species by oxidative metabolic processes: role of reactive oxygen species. Biomolecules 2019, 9, E258. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A. Mihačová, E., Manoharan, R.R. et al. Application of ultra-weak photon emission imaging in plant stress assessment. J Plant Res 2025, 138, 389–400. [Google Scholar] [CrossRef]
- Michal Cifra a, Pavel Pospíšil b Ultra-weak photon emission from biological samples: Definition, mechanisms, properties, detection and applications Journal of Photochemistry and Photobiology B: Biology 2014, 139, 2–10.
- Pospíšil P, Prasad A, Rac M. Role of reactive oxygen species in ultra-weak photon emission in biological systems. J Photochem Photobiol B 2014, 139, 11–23. [CrossRef]
- Pospíšil P, Yamamoto Y. Damage to photosystem II by lipid peroxidation products. Biochim Biophys Acta Gen Subj 2017, 1861, 457–466. [CrossRef] [PubMed]
- A Shanei 1, Z Alinasab 1*, A Kiani 1, MA Nematollahi Detection of Ultraweak Photon Emission (UPE) from Cells as a Tool for Pathological Studies J Biomed Phys Eng. 2017, 7, 389–396.
- Antonio Ayala 1, Mario F Muñoz 1, Sandro Argüelles. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev. 2014, 2014, 360438. [CrossRef]
- John, R. Bucher, Oxidative stress and radical-induced signalling IARC Scientific Publications, No. 165. Baan RA, Stewart BW, Straif K, editors. Lyon (FR): International Agency for Research on Cancer; 2019.
- Kaznacheev, A.V.P. Mikhailova, L.P. & Kartashov, N.B. Distant intercellular electromagnetic interaction between two tissue cultures. Bull Exp Biol Med 1980, 89, 345–348. [Google Scholar] [CrossRef]
- Michal Cifra 1, Jeremy Z Fields, Ashkan Farhadi. Electromagnetic cellular interactions Prog Biophys Mol Biol. 2011, 105, 223–46. [CrossRef]
- Michal Cifra, Christian Brouderb, Michaela Nerudov. Biophotons, coherence and photocount statistics: a critical review. 2015; arXiv:1502.07316v1.
- Bat’yanov A., P. Distant-optical interaction of mitochondria through quartz. Biull Eksp. Biol. Med. 1984, 97, 675–677. [Google Scholar] [CrossRef]
- Scholkmann, F. Fels D., Cifra M. Non-chemical and non-contact cell-to-cell communication: a short review. Am. J. Transl. Res. 2013, 5, 586–593. [Google Scholar] [PubMed]
- Rhys R Mould 1,†,#, Alasdair M Mackenzie 2,†,*,#, Ifigeneia Kalampouka 1, Alistair V W Nunn 1,3, E Louise Thomas 1, Jimmy D Bell 1, Stanley W Botchway 2Ultra weak photon emission—a brief review. Front Physiol. 2024, 15, 1348915. [CrossRef]
- Roland Thar 1, Michael Kühl, Propagation of electromagnetic radiation in mitochondria? J Theor Biol. 2004, 230, 261–70. [CrossRef]
- Craddock T. J., A. Friesen D., Mane J., Hameroff S., Tuszynski J. A. The feasibility of coherent energy transfer in microtubules. J. R. Soc. Interface 2014, 11, 20140677. [Google Scholar] [CrossRef]
- Prasad, A. Pospíšil, P. Linoleic Acid-Induced Ultra-Weak Photon Emission from Chlamydomonas reinhardtii as a Tool for Monitoring of Lipid Peroxidation in the Cell Membranes. PLoS ONE 2011, 6, e22345. [Google Scholar] [CrossRef]
- Nunn A. V., W. Guy G. W., Bell J. D. Bioelectric fields at the beginnings of life. Bioelectricity 2022, 4, 237–247. [Google Scholar] [CrossRef] [PubMed]
- N S Dhalla 1, R M Temsah, T Netticadan Role of oxidative stress in cardiovascular diseases. J Hypertens. 2000, 18, 655–73. [CrossRef]
- Rusty Rodriguez and Regina Redman (2005) Balancing the generation and elimination of reactive oxygen species PNAS. [CrossRef]
- Philip Newsholme 1, Vinicius Fernandes Cruzat 1 2, Kevin Noel Keane 1, Rodrigo Carlessi 1, Paulo Ivo Homem de Bittencourt Jr Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem J. 2016, 473, 4527–4550. [CrossRef]
- Prasad, S. , Gupta S. C., Tyagi A. K. (2017). Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 387, 95–105. [CrossRef]
- Phull A., R. Nasir B., Haq I. U., Kim S. J. Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chem. Biol. Interact. 2018, 281, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Sumien et al., 2021 Sumien N., Cunningham J. T., Davis D. L., Engelland R., Fadeyibi O., Farmer G. E., et al. (2021). Neurodegenerative disease: Roles for sex, hormones, and oxidative stress. Endocrinology 162 (11), bqab185. [CrossRef]
- Popp F., A. , Nagl W., Li K. H., Scholz W., Weingärtner O., Wolf R. (1984). Biophoton emission - new evidence for coherence and DNA as source. Cell Biophys. 6, 33–52. [CrossRef]
- Kobayashi, M. , Takeda M., Sato T., Yamazaki Y., Kaneko K., Ito K. I., Kato H., Inaba H. (1999): In vivo imaging of spontaneous ultraweak photon emission from a rat‘s brain correlated with cerebral energy metabolism and oxidative stress. Neurosci. Res. 34, 103–113. [CrossRef]
- Kobayashi, M. Sasaki K., Enomoto M., Ehara Y. Highly sensitive determination of transient generation of biophotons during hypersensitive response to cucumber mosaic virus in cowpea. J. Exp. Bot. 2006, 58, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Photonis (2002). Photomultiplier tube basics. Available online: https://psec.uchicago.edu/library/photomultipliers/Photonis_PMT_basics.pdf (accessed on 11 April 2023).
- Chen Y., S. Chen B. T. 2003 Measuring of a three-dimensional surface by use of a spatial distance computation, Applied Optics, 42 11 1958 1972.
- Lijian Zhang, Leonardo Neves, Jeff S Lundeen and Ian A Walmsley. A characterization of the single-photon sensitivity of an electron multiplying charge-coupled device Journal of Physics B: Atomic, Molecular and Optical Physics , Volume 42, Number 11.
- Saeidfirozeh, H. , Shafiekhani, A., Cifra, M. et al. Endogenous Chemiluminescence from Germinating Arabidopsis Thaliana Seeds. Sci Rep 8, 16231 (2018). [CrossRef]
- Khaoua, I. , Graciani, G., Kim, A. et al. Detectivity optimization to measure ultraweak light fluxes using an EM-CCD as binary photon counter array. Sci Rep 11, 3530 (2021). [CrossRef]
- Christian Kurtsiefer1, Markus Oberparleiter1, and Harald Weinfurter1,2 High-efficiency entangled photon pair collection in type-II parametric fluorescence. Phys. Rev. A 64, 023802 – Published 2 July, 2001.
- Rhys R Mould 1,†,#, Alasdair M Mackenzie 2,†,*,#, Ifigeneia Kalampouka 1, Alistair V W Nunn 1,3, E Louise Thomas 1, Jimmy D Bell 1, Stanley W Botchway Ultra weak photon emission—a brief review Front Physiol. 2024 Feb14;15:1348915. [CrossRef]
- Popp, F.-A. (1979) Photon Storage in Biological Systems. In: Popp, F.-A., Becker, G., Koenig, H.L. and Peschka, W., Eds., Electromagnetic Bio-Information, Urban & Schwarzenberg, Munich, 123-148.
- Fritz-Albert Popp Properties of biophotons and their theoretical implications Indian J Exp Biol. 2003, 41, 391–402.
- Engel, G. , Calhoun, T., Read, E. et al. Evidence for wavelike energy transfer through quantum coherence in photosynthetic systems. Nature 446, 782–786 (2007). [CrossRef]
- Thorsten Ritz Quantum effects in biology: Bird navigation. Procedia Chemistry Volume 3, Issue 1, 2011, Pages 262-275.
- R. A. Holland True navigation in birds: from quantum physics to global migration Journal of Zoology. 2014. [CrossRef]
- S M Sagar 1, R J Thomas, L T Loverock, M F Spittle. Olfactory sensations produced by high-energy photon irradiation of the olfactory receptor mucosa in humans. Int J Radiat Oncol Biol Phys. 1991, 20, 771–6. [CrossRef]
- Anashe Bandari Human smell perception is governed by quantum spin-residual information. Scilight News 2019 Volume 2019, Issue 30. [CrossRef]
- Willeford, K. The Luminescence Hypothesis of Olfaction. Sensors (Basel). 2023, 23, 1333. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Karu, T. Primary and secondary mechanisms of action of visible to near-IR radiation on cells. J Photochem Photobiol B. 1999, 49, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Mari Jibu, Kunio Yasue, Scott Hagan Evanescent (tunneling) photon and cellular ‘vision’ BioSystems 42 (1997) 65–73.
- Hameroff, S. and Penrose, R. (1996) Orchestrated Reduction of Quantum Coherence in Brain Microtubules: A Model for Consciousness. Mathematics and Computers in Simulation, 40, 453-480. [CrossRef]
- Kučera O, Cifra M. Cell-to-cell signaling through light: just a ghost of chance? Cell Commun Signal. 2013 Nov 12;11:87. [CrossRef] [PubMed] [PubMed Central]
- Mould RR, Kalampouka I, Thomas EL, Guy GW, Nunn AVW, Bell JD. Non-chemical signalling between mitochondria. Front Physiol. 2023 Sep 22;14:1268075. [CrossRef] [PubMed] [PubMed Central]
- Tang, R. and Dai, J. (2014) Biophoton Signal Transmission and Processing in the Brain. Journal of Photochemistry and Photobiology B 139, 71–75. [CrossRef]
- Kumar, S. , Boone, K., Tuszyński, J. et al. Possible existence of optical communication channels in the brain. Sci Rep, 2016; 6, 36508. [Google Scholar] [CrossRef]
- Esmaeilpour, T. , Fereydouni, E., Dehghani, F. et al. An Experimental Investigation of Ultraweak Photon Emission from Adult Murine Neural Stem Cells. Sci Rep, 2020; 10, 463. [Google Scholar] [CrossRef]
- Eduard, P.A. Van Wijk a b, Roeland Van Wijk a, Saskia Bosman a Using ultra-weak photon emission to determine the effect of oligomeric proanthocyanidins on oxidative stress of human skin. Journal of Photochemistry and Photobiology B: Biology Volume 98, Issue 3, 8 March 2010, Pages 199-206.
- Pietruszka, M. , Marzec, M. Ultra-weak photon emission from DNA. Sci Rep 14, 28915 (2024). [CrossRef]
- Knappe, G. A. , Wamhoff, E. C. & Bathe, M. Functionalizing DNA origami to investigate and interact with biological systems. Nat. Rev. Mater. 8, 123–138 2022.
- Organick, L.; et al. Probing the physical limits of reliable DNA data retrieval. Nat. Commun. 11, 616,2020.
- Kaznacheev, V.P. and Mihaylova, M.P. (1981) Ultra-Weak Radiations in Intercellular Interactions. Science, Novosibirsk.
- Bischof, M. (2003) Introduction to Integrative Biophysics. In: Popp, F.-A. and Beloussov, L., Eds., Integrative Biophysics, Springer, Dordrecht, 1-115. [CrossRef]
- F. A. Popp, K. H. Li, W. P. Mei, et al., (1988) Physical aspects of biophotons. Experientia, 44, 576-585.
- Tanabe, T. , Notomi, M., Kuramochi, E., Shinya, A., & Taniyama, H. (2007). Trapping and delaying photons for one nanosecond in an ultrasmall high-Q photonic-crystal nanocavity. Nature Photonics, 1(1), 49-52. [CrossRef]
- Feldmann A, Ivanek R, Murr R, Gaidatzis D, Burger L, Schübeler D (2013) Transcription Factor Occupancy Can Mediate Active Turnover of DNA Methylation at Regulatory Regions. PLoS Genet 9, e1003994. [CrossRef]
- Fröhlich, H. Long-range coherence and energy storage in biological systems. Int. J. Quantum Chem. 2, 641–649,1968.
- Vasconcellos, Á. R. , Vannucchi, F. S., Mascarenhas, S. & Luzzi, R. Fröhlich Condensate: emergence of Synergetic Dissipative structures in Information Processing Biological and Condensed Matter systems. Information 3, 601–620,2012.
- Nakamura, Y. Pashkin, Y. A. & Tsai, J. S. Coherent control of macroscopic quantum states in a single-Cooper-pair box. Nature 398, 786–788,1999.
- Zhang, Z. Agarwal, G. S. & Scully, M. O. Quantum fluctuations in the Fröhlich Condensate of Molecular vibrations Driven Far from Equilibrium. Phys. Rev. Lett. 122, 158101, 2019.
- Dong, B.; et al. Superresolution intrinsic fluorescence imaging of chromatin utilizing native, unmodified nucleic acids for contrast. Proc. Natl. Acad. Sci. U S A. 113, 9716–9721 (2016).
- Berke et al, 2024, Unique algorithm for the evaluation of embryo photon emission and viability Sci Rep. 2024, 14, 15066. [CrossRef]
- Bódis et al. 2024 Detection of ultra-weak photon emissions from mouse embryos with implications for Assisted reproduction Journal of Health Care Communications Volume 9, Issue 4.
- Shannon, C. E. A mathematical theory of communication. Bell Syst. Tech. J. 1948; 27, 623–656. [Google Scholar]
- Shannon, C. E. Prediction and entropy of printed English. Bell Syst. Tech. J. 1951; 30, 50–64. [Google Scholar]
- Riera Aroche, R. , Ortiz García, Y.M., Martínez Arellano, M.A. et al. DNA as a perfect quantum computer based on the quantum physics principles. Sci Rep 14, 11636 (2024). [CrossRef]
- Popp, F.A. , et al. (1992) Recent Advances in Biophoton Research and Its Aplication. World Scientific, Singapore City, London, New York. [CrossRef]
- Choi, K.H. Lee, J.H. Kim, M.Y. Kim, J.H. Lim, J. Lee Effect of 710 nm visible light irradiation on neurite outgrowth in primary rat cortical neurons following ischemic insult. Biochem. Biophys. Res. Commun., 422 (2012), pp. 274–279.
- Van Wijk, R. and Van Wijk, E.P.A. (2005) An Introduction to Human Biophoton Emission. Forsch Komplementärmed Klass Naturheilkd, 12, 77-83. [CrossRef]
- Szent-Györgyi, A. Towards a New Biochemistry? Science. 1941, 93, 609–11. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



