Submitted:
04 November 2025
Posted:
05 November 2025
You are already at the latest version
Abstract
Keywords:
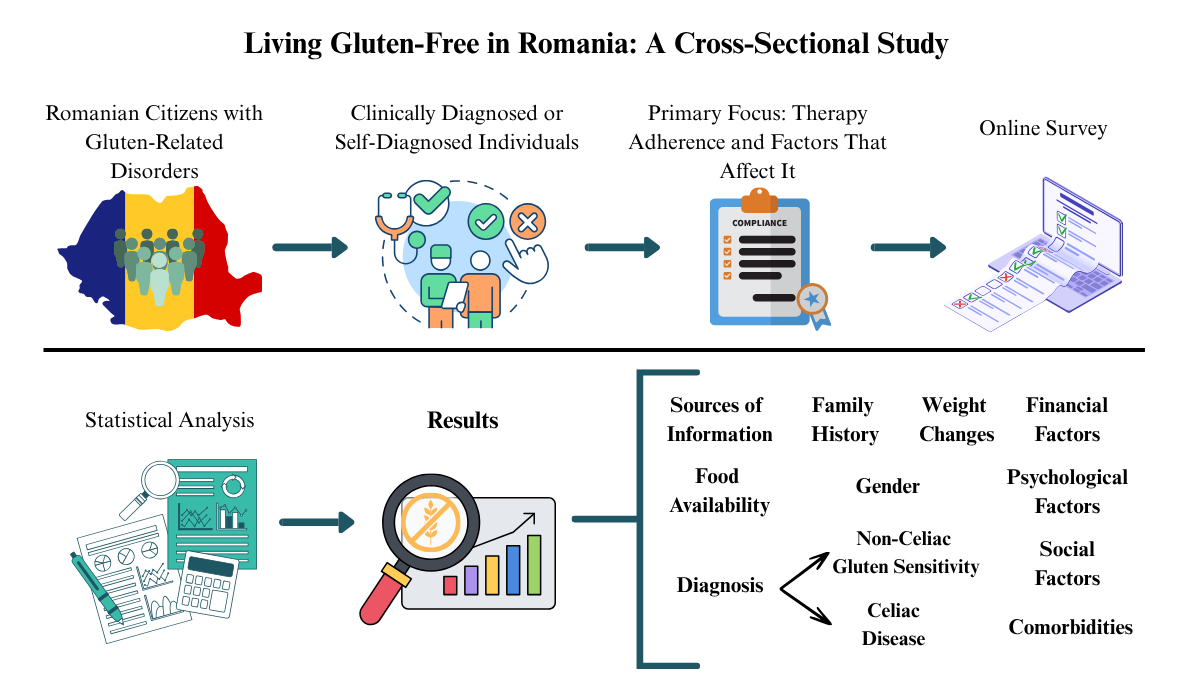
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Ethical Aspects
2.3. Questionnaire Components
- Residence and gender (items 2-3): to identify the Romanian citizens who continue to reside in Romania and to help us determine the male-to-female ratio for these disorders.
- Diagnostic background, familial incidence, and comorbidities (items 4–11): to explore how diagnoses are made and to identify self-diagnostic practices, the hereditary factor, emotional adaptation to the diagnosis, and coexistent chronic diseases.
- Dietary knowledge and attitudes (items 12–16): sources of information and shifting perceptions of importance and difficulty.
- Accessibility of GF foods (items 17–20): frequency of finding suitable products in stores and restaurants, both at time of diagnosis and at the time of completing the questionnaire.
- Physical and social consequences (items 21–23): weight changes, feelings of discrimination, and social absences due to dietary restrictions.
- Economic burden (item 24): perceived expense of GF alternatives.
- Adherence behaviors and beliefs (items 25–33): self-reported dietary adherence, label-reading habits, trust in certified products, and strategies in social contexts.
2.4. Adherence with the GFD
2.5. Study Hypotheses and Analyses
2.6. Statistical Analysis
3. Results
Descriptive Statistics and Individual Differences
3.1. Impact of Medical and Individual Factors on GFD Adherence
3.2. Impact of Psychosocial Factors on GFD Adherence
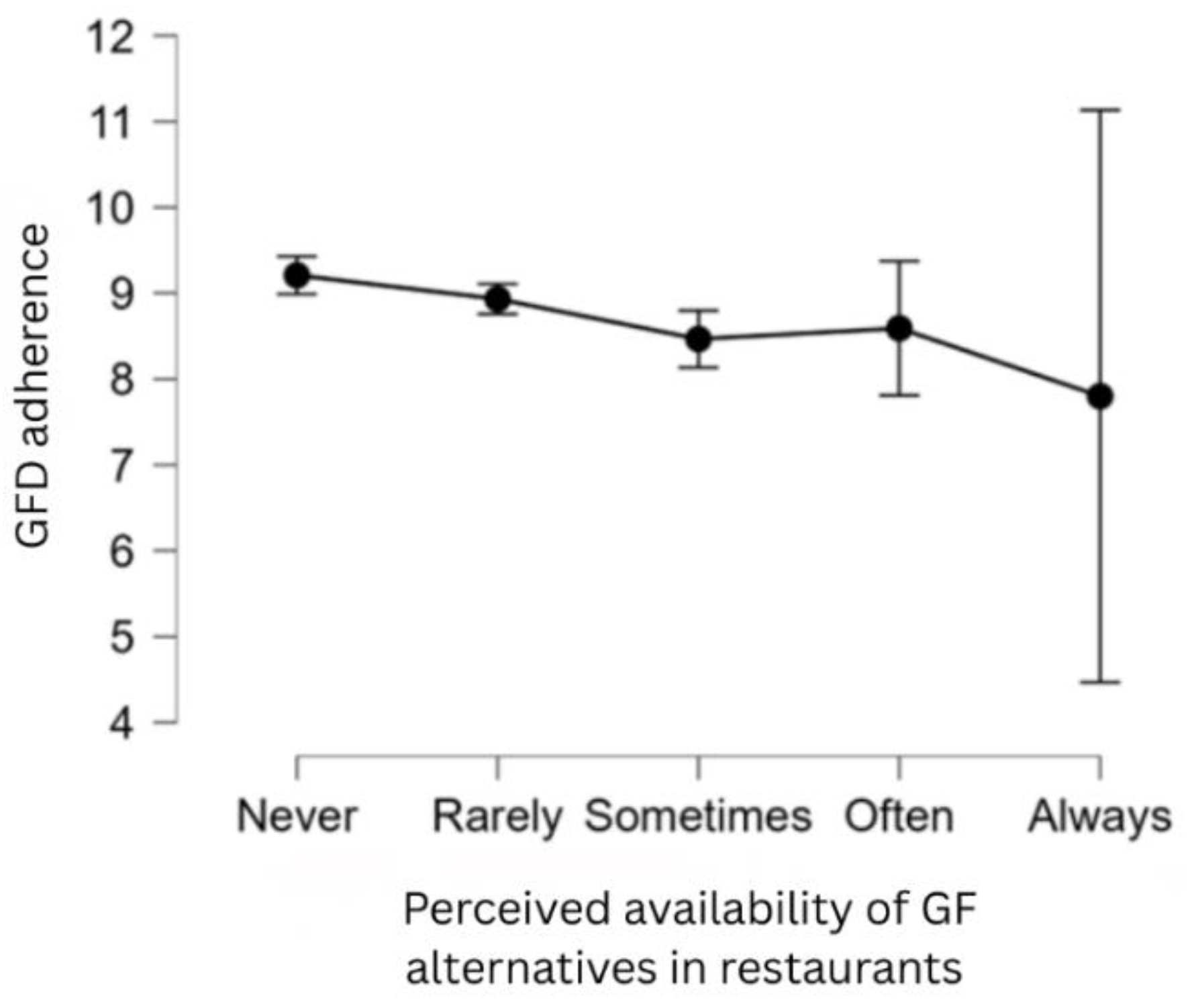
3.3. Impact of Economic Factors and Availability on GFD Adherence
4. Discussion
5. Conclusions
- Strengthening diagnostic pathways to diminish self-diagnosis and guarantee the proper initiation of restrictive diets.
- Improving the accuracy of food labeling and oversight of certification to reduce unintentional gluten exposure.
- Increasing access to specialized nutritional counseling as a fundamental aspect of GRDs care.
- Tackling economic obstacles through policy initiatives aimed at alleviating the financial burden associated with GF products.
- Incorporating psychosocial support to alleviate feelings of isolation, discrimination, and impairments in QoL.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ARIG | Romanian Association for Gluten Intolerance |
| CD | Celiac Disease |
| GC | Gluten-Containing |
| GERD | Gastroesophageal Reflux Disease |
| GF | Gluten-Free |
| GFD | Gluten-Free Diet |
| GRD | Gluten-Related Disorder |
| IBS | Irritable Bowel Syndrome |
| LI | Lactose Intolerance |
| NCGS | Non-Celiac Gluten Sensitivity |
| NCWS | Non-Celiac Wheat Sensitivity |
| QoL | Quality of Life |
| SRGS | Self-Reported Gluten Sensitivity |
References
- Jansson-Knodell, C.L.; Rubio-Tapia, A. Gluten-Related Disorders From Bench to Bedside. Clinical Gastroenterology and Hepatology 2024, 22, 693-704.e1. [Google Scholar] [CrossRef] [PubMed]
- Valenti, S.; Corica, D.; Ricciardi, L.; Romano, C. Gluten-Related Disorders: Certainties, Questions and Doubts. Ann Med 2017, 49, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Al-Toma, A.; Volta, U.; Auricchio, R.; Castillejo, G.; Sanders, D.S.; Cellier, C.; Mulder, C.J.; Lundin, K.E.A. European Society for the Study of Coeliac Disease (ESsCD) Guideline for Coeliac Disease and Other Gluten-Related Disorders. United European Gastroenterol J 2019, 7, 583–613. [Google Scholar] [CrossRef]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology 2018, 16, 823-836.e2. [Google Scholar] [CrossRef]
- Pinto-Sanchez, M.I.; Silvester, J.A.; Lebwohl, B.; Leffler, D.A.; Anderson, R.P.; Therrien, A.; Kelly, C.P.; Verdu, E.F. Society for the Study of Celiac Disease Position Statement on Gaps and Opportunities in Coeliac Disease. Nat Rev Gastroenterol Hepatol 2021, 18, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Bonner, E.R.; Tschollar, W.; Anderson, R. Review Article : Novel Enzyme Therapy Design for Gluten Peptide Digestion Through Exopeptidase Supplementation. Aliment Pharmacol Ther 2025, 1–17. [Google Scholar] [CrossRef]
- Gnodi, E.; Meneveri, R.; Barisani, D. Celiac Disease: From Genetics to Epigenetics. World J Gastroenterol 2022, 28, 449–463. [Google Scholar] [CrossRef]
- Durazzo, M.; Ferro, A.; Brascugli, I.; Mattivi, S.; Fagoonee, S.; Pellicano, R. Extra-Intestinal Manifestations of Celiac Disease. What Should We Know in 2022? J Clin Med 2022, 11. [Google Scholar] [CrossRef]
- Tomer, R.; Patiyal, S.; Dhall, A.; Raghava, G.P.S. Prediction of Celiac Disease Associated Epitopes and Motifs in a Protein. Front Immunol 2023, 14, 1–10. [Google Scholar] [CrossRef]
- Alkalay, M.J. Nutrition in Patients with Lactose Malabsorption, Celiac Disease, and Related Disorders. Nutrients 2022, 14, 2. [Google Scholar] [CrossRef]
- Verdelli, A.; Corrà, A.; Mariotti, E.B.; Aimo, C.; Quintarelli, L.; Ruffo di Calabria, V.; Donati, M.E.; Bonciolini, V.; Antiga, E.; Caproni, M. Skin Gluten-Related Disorders: New and Old Cutaneous Manifestations to Be Considered. Front Med (Lausanne) 2023, 10, 1155288. [Google Scholar] [CrossRef]
- Yu, X.B.; Uhde, M.; Green, P.H.; Alaedini, A. Autoantibodies in the Extraintestinal Manifestations of Celiac Disease. Nutrients 2018, 10, 1–16. [Google Scholar] [CrossRef]
- King, J.A.; Jeong, J.; Underwood, F.E.; Quan, J.; Panaccione, N.; Windsor, J.W.; Coward, S.; Debruyn, J.; Ronksley, P.E.; Shaheen, A.A.; et al. Incidence of Celiac Disease Is Increasing over Time: A Systematic Review and Meta-Analysis. American Journal of Gastroenterology 2020, 115, 507–525. [Google Scholar] [CrossRef] [PubMed]
- Rouvroye, M.D.; Oldenburg, L.; Slottje, P.; Joosten, J.H.K.; de Menezes, R.X.; Reinders, M.E.; Bouma, G. Testing for Coeliac Disease Rarely Leads to a Diagnosis: A Population-Based Study. Scand J Prim Health Care 2021, 39, 315–321. [Google Scholar] [CrossRef]
- Wagh, S.K.; Lammers, K.M.; Padul, M.V.; Rodriguez-Herrera, A.; Dodero, V.I. Celiac Disease and Possible Dietary Interventions: From Enzymes and Probiotics to Postbiotics and Viruses. Int J Mol Sci 2022, 23, 11748. [Google Scholar] [CrossRef]
- Stanciu, D.; Staykov, H.; Dragomanova, S.; Tancheva, L.; Pop, R.S.; Ielciu, I.; Crișan, G. Gluten Unraveled: Latest Insights on Terminology, Diagnosis, Pathophysiology, Dietary Strategies, and Intestinal Microbiota Modulations—A Decade in Review. Nutrients 2024, 16. [Google Scholar] [CrossRef]
- Goel, G.; Tye-Din, J.A.; Qiao, S.W.; Russell, A.K.; Mayassi, T.; Ciszewski, C.; Sarna, V.K.; Wang, S.; Goldstein, K.E.; Dzuris, J.L.; et al. Cytokine Release and Gastrointestinal Symptoms after Gluten Challenge in Celiac Disease. Sci Adv 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Caio, G.; Volta, U.; Sapone, A.; Leffler, D.A.; De Giorgio, R.; Catassi, C.; Fasano, A. Celiac Disease: A Comprehensive Current Review. BMC Med 2019, 17, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Green, P.H.R.; Fasano, A. Extraintestinal Manifestations of Coeliac Disease. Nat Rev Gastroenterol Hepatol 2015, 12, 561–571. [Google Scholar] [CrossRef]
- Therrien, A.; Kelly, C.P.; Silvester, J.A. Celiac Disease: Extraintestinal Manifestations and Associated Conditions; 2020; Vol. 54, ISBN 0000000000. [Google Scholar]
- Nardecchia, S.; Auricchio, R.; Discepolo, V.; Troncone, R. Extra-Intestinal Manifestations of Coeliac Disease in Children: Clinical Features and Mechanisms. Front Pediatr 2019, 7, 56. [Google Scholar] [CrossRef]
- Singh, A.D.; Ellias, S.; Singh, P.; Ahuja, V.; Makharia, G.K. The Prevalence of the Celiac Disease in Patients with Dyspepsia: A Systematic Review and Meta-Analysis. Dig Dis Sci 2022, 67, 3067–3079. [Google Scholar] [CrossRef]
- Lamjadli, S.; Oujamaa, I.; Souli, I.; Eddehbi, F.; Lakhouaja, N.; Bouchra, M.; Salami, A.; Guennouni, M. Micronutrient Deficiencies in Patients with Celiac Disease : A Systematic Review and Meta-Analysis. Int J Immunopathol Pharmacol 2025, 39, 1–24. [Google Scholar] [CrossRef]
- Lauret, E.; Rodrigo, L. Celiac Disease and Autoimmune-Associated Conditions. Biomed Res Int 2013, 2013, 127589. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Mangge, H.; Schenk, M.; Enko, D. Non-Responsive Celiac Disease May Coincide with Additional Food Intolerance/Malabsorption, Including Histamine Intolerance. Med Hypotheses 2021, 146, 110404. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, W.; Zopf, Y. Gluten and FODMAPS — Sense of a Restriction / When. Nutrients 2019, 11, 1–12. [Google Scholar] [CrossRef]
- An, C.; Yang, J.; Pinto-Sanchez, M.I.; Verdu, E.F.; Lebwohl, B.; Green, P.H.; Alaedini, A. Molecular Triggers of Non-Celiac Wheat Sensitivity: A Scoping Review and Analysis. American Journal of Gastroenterology 2025, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Elli, L.; Branchi, F.; Tomba, C.; Villalta, D.; Norsa, L.; Ferretti, F.; Roncoroni, L.; Bardella, M.T. Diagnosis of Gluten Related Disorders: Celiac Disease, Wheat Allergy and Non-Celiac Gluten Sensitivity. World J Gastroenterol 2015, 21, 7110–7119. [Google Scholar] [CrossRef] [PubMed]
- Shahbazkhani, B.; Fanaeian, M.M.; Farahvash, M.J.; Aletaha, N.; Alborzi, F.; Elli, L.; Shahbazkhani, A.; Zebardast, J.; Rostami-Nejad, M. Prevalence of Non-Celiac Gluten Sensitivity in Patients with Refractory Functional Dyspepsia: A Randomized Double-Blind Placebo Controlled Trial. Sci Rep 2020, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Catassi, G.; Naspi, L. Nonceliac Gluten Sensitivity. Curr Opin Clin Nutr Metab Care 2023, 26, 490–494. [Google Scholar] [CrossRef]
- Expósito Miranda, M.; García-Valdés, L.; Espigares-Rodríguez, E.; Leno-Durán, E.; Requena, P. Non-Celiac Gluten Sensitivity: Clinical Presentation, Etiology and Differential Diagnosis. Gastroenterología y Hepatología (English Edition) 2023, 46, 562–571. [Google Scholar] [CrossRef]
- Roszkowska, A.; Pawlicka, M.; Mroczek, A.; Bałabuszek, K.; Nieradko-Iwanicka, B. Non-Celiac Gluten Sensitivity: A Review. Medicina (B Aires) 2019, 55, 222. [Google Scholar] [CrossRef]
- Graziano, M.; Rossi, M. An Update on the Cutaneous Manifestations of Coeliac Disease and Non-Coeliac Gluten Sensitivity. Int Rev Immunol 2018, 37, 291–300. [Google Scholar] [CrossRef]
- Skodje, G.I.; Minelle, I.H.; Rolfsen, K.L.; Iacovou, M.; Lundin, K.E.A.; Veierød, M.B.; Henriksen, C. Dietary and Symptom Assessment in Adults with Self-Reported Non-Coeliac Gluten Sensitivity. Clin Nutr ESPEN 2019, 31, 88–94. [Google Scholar] [CrossRef]
- Molina-Infante, J.; Santolaria, S.; Sanders, D.S.; Fernández-Bañares, F. Systematic Review: Noncoeliac Gluten Sensitivity. Aliment Pharmacol Ther 2015, 41, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small Amounts of Gluten in Subjects With Suspected Nonceliac Gluten Sensitivity: A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Clinical Gastroenterology and Hepatology 2015, 13, 1604–1612.e3. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, M.R.; Cremon, C.; Stanghellini, V.; Barbara, G. Recent Advances in Understanding Non-Celiac Gluten Sensitivity. F1000Res 2018, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yoosuf, S.; Makharia, G.K. Treatment of Gluten-Related Disorders. Gluten-Related Disorders: Diagnostic Approaches, Treatment Pathways, and Future Perspectives 2022, 149–182. [Google Scholar] [CrossRef]
- Buriánek, F.; Gege, C.; Marinković, P. New Developments in Celiac Disease Treatments. Drug Discov Today 2024, 29. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: New York, 1988; ISBN 978-1-134-74270-7. [Google Scholar]
- Al-sunaid, F.F.; Al-homidi, M.M.; Al-qahtani, R.M.; Al-ashwal, R.A.; Mudhish, G.A.; Hanbazaza, M.A.; Al-zaben, A.S. The Influence of a Gluten-Free Diet on Health-Related Quality of Life in Individuals with Celiac Disease. BMC Gastroenterol 2021, 21, 1–9. [Google Scholar] [CrossRef]
- Demirkesen, I.; Ozkaya, B. Recent Strategies for Tackling the Problems in Gluten-Free Diet and Products. Crit Rev Food Sci Nutr 2022, 62, 571–597. [Google Scholar] [CrossRef]
- Bascuñán, K.A.; Vespa, M.C.; Araya, M. Celiac Disease: Understanding the Gluten-Free Diet. Eur J Nutr 2017, 56, 449–459. [Google Scholar] [CrossRef]
- Green, P.H.R.; Lebwohl, B.; Greywoode, R. Celiac Disease. Journal of Allergy and Clinical Immunology 2015, 135, 1099–1106. [Google Scholar] [CrossRef]
- Cheng, F.W.; Handu, D. Nutrition Assessment, Interventions, and Monitoring for Patients with Celiac Disease: An Evidence Analysis Center Scoping Review. J Acad Nutr Diet 2020, 120, 1381–1406. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, D.; Staykov, H.; Dragomanova, S.; Tancheva, L.; Pop, R.S.; Ielciu, I.; Crisan, G. Gluten Unraveled: Latest Insights on Terminology, Diagnosis, Pathophysiology, Dietary Strategies, and Intestinal Microbiota Modulations—A Decade in Review. Nutrients 2024, 16, 3636. [Google Scholar] [CrossRef] [PubMed]
- Manza, F.; Lungaro, L.; Costanzini, A.; Caputo, F.; Carroccio, A.; Mansueto, P.; Seidita, A.; Raju, S.A.; Volta, U.; De Giorgio, R.; et al. Non-Celiac Gluten/Wheat Sensitivity—State of the Art: A Five-Year Narrative Review. Nutrients 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Chávez, F.; Dezar, G.V.A.; Islas-Zamorano, A.P.; Espinoza-Alderete, J.G.; Vergara-Jiménez, M.J.; Magaña-Ordorica, D.; Ontiveros, N. Prevalence of Self-Reported Gluten Sensitivity and Adherence to a Gluten-Free Diet in Argentinian Adult Population. Nutrients 2017, 9, 1–11. [Google Scholar] [CrossRef]
- Niland, B.; Cash, B.D. Health Benefits and Adverse Effects of a Gluten-Free Diet in Non-Celiac Disease Patients. Gastroenterol Hepatol (N Y) 2018, 14, 82–91. [Google Scholar]
- Volta, U.; Bardella, M.T.; Calabrò, A.; Troncone, R.; Corazza, G.R.; Bagnato, C.; Belcari, C.; Bellantoni, A.; Caio, G.; Calella, F.; et al. An Italian Prospective Multicenter Survey on Patients Suspected of Having Non-Celiac Gluten Sensitivity. BMC Med 2014, 12, 4–11. [Google Scholar] [CrossRef]
- Manza, F.; Lungaro, L.; Costanzini, A.; Caputo, F.; Carroccio, A.; Mansueto, P.; Seidita, A.; Raju, S.A.; Volta, U.; De Giorgio, R.; et al. Non-Celiac Gluten/Wheat Sensitivity—State of the Art: A Five-Year Narrative Review. Nutrients 2025, 17. [Google Scholar] [CrossRef]
- Cabrera-Chávez, F.; Dezar, G.V.A.; Islas-Zamorano, A.P.; Espinoza-Alderete, J.G.; Vergara-Jiménez, M.J.; Magaña-Ordorica, D.; Ontiveros, N. Prevalence of Self-Reported Gluten Sensitivity and Adherence to a Gluten-Free Diet in Argentinian Adult Population. Nutrients 2017, 9, 1–11. [Google Scholar] [CrossRef]
- van Gils, T.; Nijeboer, P.; Ijssennagger, C.E.; Sanders, D.S.; Mulder, C.J.J.; Bouma, G. Prevalence and Characterization of Self-Reported Gluten Sensitivity in The Netherlands. Nutrients 2016, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Giambalvo, O.; La Blasca, F.; Iacobucci, R.; D’Alcamo, A.; Mansueto, P. Self-Reported Non-Celiac Wheat Sensitivity in High School Students: Demographic and Clinical Characteristics. Nutrients 2017, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zanini, B.; Baschè, R.; Ferraresi, A.; Ricci, C.; Lanzarotto, F.; Marullo, M.; Villanacci, V.; Hidalgo, A.; Lanzini, A. Randomised Clinical Study: Gluten Challenge Induces Symptom Recurrence in Only a Minority of Patients Who Meet Clinical Criteria for Non-Coeliac Gluten Sensitivity. Aliment Pharmacol Ther 2015, 42, 968–976. [Google Scholar] [CrossRef]
- Silvester, J.A.; Weiten, D.; Graff, L.A.; Walker, J.R.; Duerksen, D.R. Living Gluten-Free: Adherence, Knowledge, Lifestyle Adaptations and Feelings towards a Gluten-Free Diet. Journal of Human Nutrition and Dietetics 2016, 29, 374–382. [Google Scholar] [CrossRef]
- Jeanes, Y.; Orlandi, L.; Muhammad, H.; Reeves, S. Telemedicine in Coeliac Disease: In-Person Appointments Are Favoured by Patients With a Lower Education Attainment and Lower Household Income. Journal of Human Nutrition and Dietetics 2025, 38, e70014. [Google Scholar] [CrossRef]
- Centrul Regional Pentru Managementul Bolii Celiace. Available online: https://www.insmc.ro/compartimente/centrul-regional-pentru-managementul-bolii-celiace/ (accessed on 19 March 2025).
- Siminiuc, R.; Ṭurcanu, D. Food Security of People with Celiac Disease in the Republic of Moldova through Prism of Public Policies. Front Public Health 2022, 10. [Google Scholar] [CrossRef]
- Members of Association of European Coeliac Societies | AOECS. Available online: https://www.aoecs.org/about-us/members/ (accessed on 19 March 2025).
- Asociația Română Pentru Intoleranță La Gluten (ARIG). Available online: https://arig.ro/organizare/ (accessed on 19 March 2025).
- Avena-Woods, C.; Mangione, R.A.; Wu, W.K. Exploring the Community Pharmacist’s Knowledge of Celiac Disease. Am J Pharm Educ 2018, 82, 100–105. [Google Scholar] [CrossRef]
- Poslt Königová, M.; Sebalo Vňuková, M.; Řehořková, P.; Anders, M.; Ptáček, R. The Effectiveness of Gluten-Free Dietary Interventions: A Systematic Review. Front Psychol 2023, 14. [Google Scholar] [CrossRef]
- Isaac, D.M.; Wu, J.; Mager, D.R.; Turner, J.M. Managing the Pediatric Patient with Celiac Disease: A Multidisciplinary Approach. J Multidiscip Healthc 2016, 9, 529–536. [Google Scholar] [CrossRef]
- Kaul, R.; Jansson-Knodell, C.; Simons, M.L.; Weekley, K.; Gardinier, D.; Rubio-Tapia, A. Avoidant/Restrictive Food Intake Disorder in Celiac Disease. Nutrients 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Zarkadas, M.; Dubois, S.; Macisaac, K.; Cantin, I.; Rashid, M.; Roberts, K.C.; La Vieille, S.; Godefroy, S.; Pulido, O.M. Living with Coeliac Disease and a Gluten-Free Diet: A Canadian Perspective. Journal of Human Nutrition and Dietetics 2013, 26, 10–23. [Google Scholar] [CrossRef]
- Niland, B.; Cash, B.D. Health Benefits and Adverse Effects of a Gluten-Free Diet in Non-Celiac Disease Patients. Gastroenterol Hepatol (N Y) 2018, 14, 82–91. [Google Scholar]
- Leffler, D.A.; Edwards-George, J.; Dennis, M.; Schuppan, D.; Cook, F.; Franko, D.L.; Blom-Hoffman, J.; Kelly, C.P. Factors That Influence Adherence to a Gluten-Free Diet in Adults with Celiac Disease. Dig Dis Sci 2008, 53, 1573–1581. [Google Scholar] [CrossRef]
- Lebwohl, B.; Sanders, D.S.; Green, P.H.R. Coeliac Disease. The Lancet 2018, 391, 70–81. [Google Scholar] [CrossRef]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition Guidelines for the Diagnosis of Coeliac Disease. J Pediatr Gastroenterol Nutr 2012, 54, 136–160. [Google Scholar] [CrossRef] [PubMed]
- Burden, M.; Mooney, P.D.; Blanshard, R.J.; White, W.L.; Cambray-Deakin, D.R.; Sanders, D.S. Cost and Availability of Gluten-Free Food in the UK: In Store and Online. Postgrad Med J 2015, 91, 622–626. [Google Scholar] [CrossRef]
- Ribeiro, C. dos S.; Pratesi, C.B.; Zandonadi, R.P. Celiac Disease and Gluten-Free Diets: A Path or Barrier to Food (In)Security? Nutrients 2025, 17. [Google Scholar]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology 2018, 16, 823-836.e2. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. ACG Clinical Guidelines: Diagnosis and Management of Celiac Disease. American Journal of Gastroenterology 2013, 108, 656–676. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Murray, J.A. Epidemiology of Celiac Disease. Gastroenterol Clin North Am 2019, 48, 1–18. [Google Scholar] [CrossRef]
- Bardella, M.T.; Fredella, C.; Saladino, V.; Trovato, C.; Cesana, B.M.; Quatrini, M.; Prampolini, L. Gluten Intolerance: Gender- and Age-Related Differences in Symptoms. Scand J Gastroenterol 2005, 40, 15–19. [Google Scholar] [CrossRef]
- Jansson-Knodell, C.L.; Hujoel, I.A.; West, C.P.; Taneja, V.; Prokop, L.J.; Rubio-Tapia, A.; Murray, J.A. Sex Difference in Celiac Disease in Undiagnosed Populations: A Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology 2019, 17, 1954–1968.e13. [Google Scholar] [CrossRef]
- Ciccocioppo, R.; Kruzliak, P.; Cangemi, G.C.; Pohanka, M.; Betti, E.; Lauret, E.; Rodrigo, L. The Spectrum of Differences between Childhood and Adulthood Celiac Disease. Nutrients 2015, 7, 8733–8751. [Google Scholar] [CrossRef]
- Megiorni, F.; Mora, B.; Bonamico, M.; Barbato, M.; Montuori, M.; Viola, F.; Trabace, S.; Mazzilli, M.C. HLA-DQ and Susceptibility to Celiac Disease: Evidence for Gender Differences and Parent-of-Origin Effects. American Journal of Gastroenterology 2008, 103, 997–1003. [Google Scholar] [CrossRef]
- Hernangomez-Laderas, A.; Cilleros-Portet, A.; Martínez Velasco, S.; Marí, S.; Legarda, M.; González-García, B.P.; Tutau, C.; García-Santisteban, I.; Irastorza, I.; Fernandez-Jimenez, N.; et al. Sex Bias in Celiac Disease: XWAS and Monocyte EQTLs in Women Identify TMEM187 as a Functional Candidate Gene. Biol Sex Differ 2023, 14. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex Differences in Immune Responses. Nat Rev Immunol 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Salazar, C.; García-Cárdenas, J.M.; Paz-y-miño, C. Understanding Celiac Disease from Genetics to the Future Diagnostic Strategies. Clin Med Insights Gastroenterol 2017, 10. [Google Scholar] [CrossRef]
- Dochat, C.; Afari, N.; Satherley, R.-M.; Coburn, S.; McBeth, J.F. Celiac Disease Symptom Profiles and Their Relationship to Gluten-Free Diet Adherence, Mental Health, and Quality of Life. BMC Gastroenterol 2024, 24, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Bardella, M.T.; Calabrò, A.; Troncone, R.; Corazza, G.R.; Bagnato, C.; Belcari, C.; Bellantoni, A.; Caio, G.; Calella, F.; et al. An Italian Prospective Multicenter Survey on Patients Suspected of Having Non-Celiac Gluten Sensitivity. BMC Med 2014, 12, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Molina-Infante, J.; Santolaria, S.; Sanders, D.S.; Fernández-Bañares, F. Systematic Review: Noncoeliac Gluten Sensitivity. Aliment Pharmacol Ther 2015, 41, 807–820. [Google Scholar] [CrossRef]
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of Gluten-Related Disorders: Consensus on New Nomenclature and Classification. BMC Med 2012, 10. [Google Scholar] [CrossRef] [PubMed]

| Category | Signs and Symptoms |
|---|---|
| Gastrointestinal Manifestations | Diarrhea, constipation, vomiting, bloating, abdominal distension, abdominal discomfort and pain, flatulence, nausea, loss of appetite, acid reflux, dyspepsia |
| Intestinal inflammation, intestinal malabsorption and maldigestion, nutrient deficiencies, altered nutritional status | |
| Lactose intolerance (in ~50% of patients at diagnosis) and intolerance to other carbohydrates | |
| Vitamin, mineral, and other deficiencies (B vitamins, vitamin D, folate, calcium, zinc, iron, and ferritin) due to malabsorption | |
|
Extraintestinal Manifestations |
Cardiovascular: pericardial effusion, myocarditis, cardiomyopathy |
| Musculoskeletal: osteopenia, osteoporosis, osteomalacia, muscle wasting, myalgia, arthritis | |
| Dermatological: dermatitis herpetiformis, atopic dermatitis, eczema, psoriasis, alopecia areata, chronic urticaria, vitiligo | |
| Reproductive: infertility, recurrent miscarriage, amenorrhea, delayed menarche, early menopause | |
| Hepatological: hepatitis, transaminitis | |
| Neurological: headaches, migraines, peripheral neuropathy, gluten ataxia, epilepsy, cognitive impairment ("brain fog") | |
| Oral: dental enamel defects, aphthous stomatitis | |
| Psychiatric: anxiety, depression, fatigue, dysthymia, behavioral disorders, autism spectrum disorders, autism, ADHD | |
| Other: weight loss, short stature | |
|
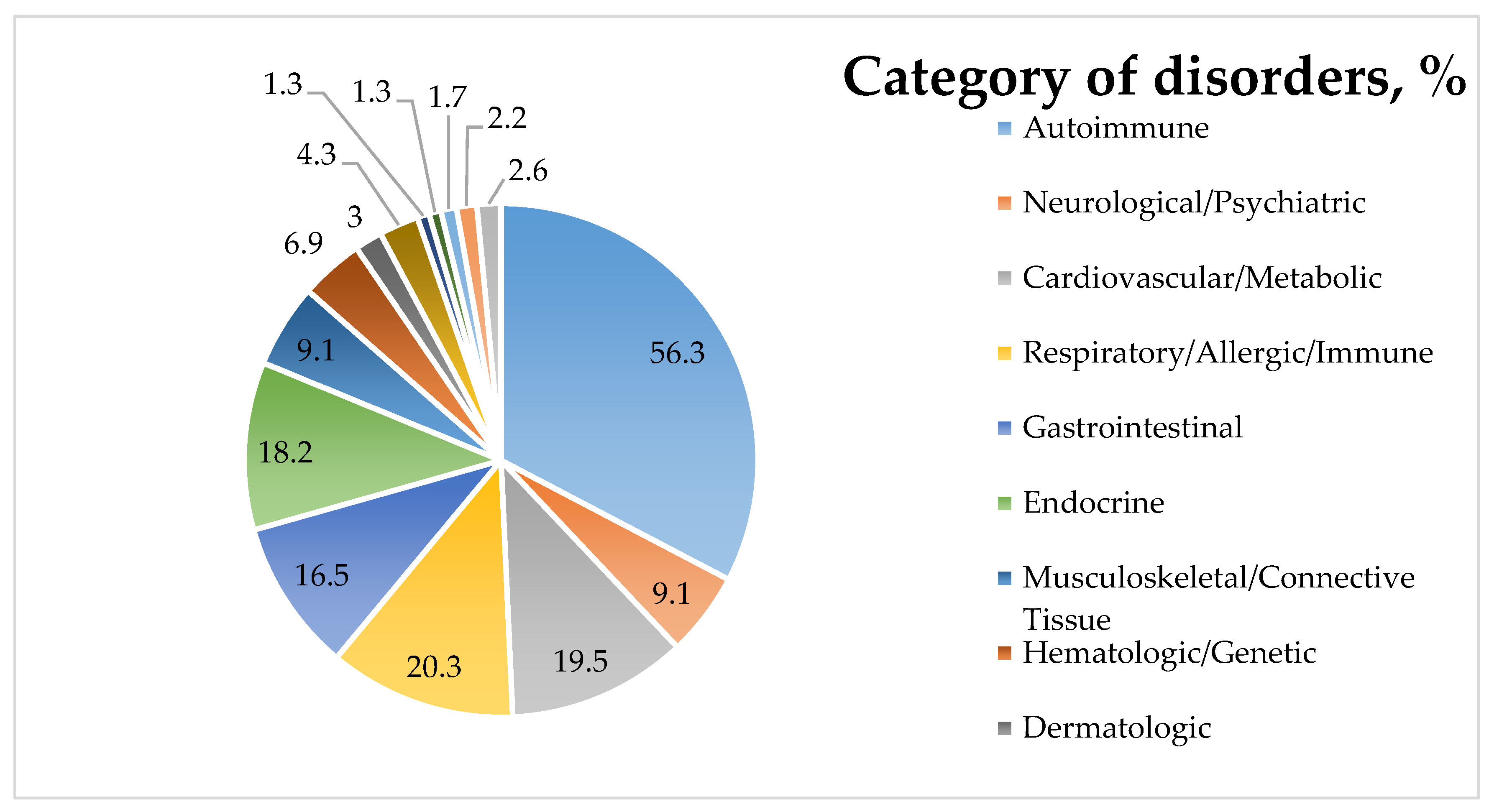
Associated Conditions |
Autoimmune endocrine disorders: thyroid diseases such as autoimmune thyroiditis mainly Hashimoto’s, Graves’ disease; Addison’s disease; type 1 diabetes mellitus |
| Autoimmune dermatological diseases: dermatitis herpetiformis, vitiligo, alopecia areata, dermatomyositis | |
| Autoimmune neurological diseases: gluten ataxia, peripheral neuropathies | |
| Autoimmune liver diseases: autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, Wilson’s disease, Budd–Chiari syndrome | |
| Other rheumatological and connective tissue diseases: Sjögren’s syndrome, systemic lupus erythematosus, systemic sclerosis, rheumatoid arthritis, and idiopathic inflammatory myopathies | |
| Other: iron-deficiency anemia, food intolerances, Down syndrome, dementia, epilepsy, autoimmune pericarditis, psoriasis, pancreatitis |
| Category | Signs and Symptoms |
|---|---|
|
Gastrointestinal Manifestations |
Diarrhea, constipation, nausea, vomiting, bloating, abdominal distension, flatulence, acid reflux, dyspepsia, epigastric pain, abdominal discomfort and pain, pyrosis, aerophagia |
| Intestinal inflammation, nutrient deficiencies, altered nutritional status | |
| Food intolerances (mainly lactose intolerance) | |
| Vitamin and mineral deficiencies (B vitamins, vitamin D, folate, calcium, iron) | |
|
Extraintestinal Manifestations |
Psychiatric: anxiety, depression, behavioral or mood disorders, sleep disorder |
| Neurological: headaches, gluten ataxia, cognitive impairment ("brain fog"), fatigue | |
| Musculoskeletal: myalgia, arthritis, fibromyalgia, limb numbness | |
| Dermatological: dermatitis herpetiformis, atopic dermatitis, eczema, rashes, psoriasis, urticaria | |
| Reproductive: menstrual disorders | |
| Oral: aphthous stomatitis | |
| Other: Weight loss | |
|
Associated Conditions |
Autoimmune disorders (such as autoimmune thyroiditis), allergic rhinitis, food allergies, anemia, IBS, IgE-mediated allergies, food intolerances, rhinitis, asthma, gluten ataxia, eating disorders, dermatitis herpetiformis, atopic dermatitis, psoriasis |
| Hypotheses | Medical and Individual Factors |
|---|---|
| H1 | Patients who have relatives with the same condition as themselves show higher adherence to the GFD. |
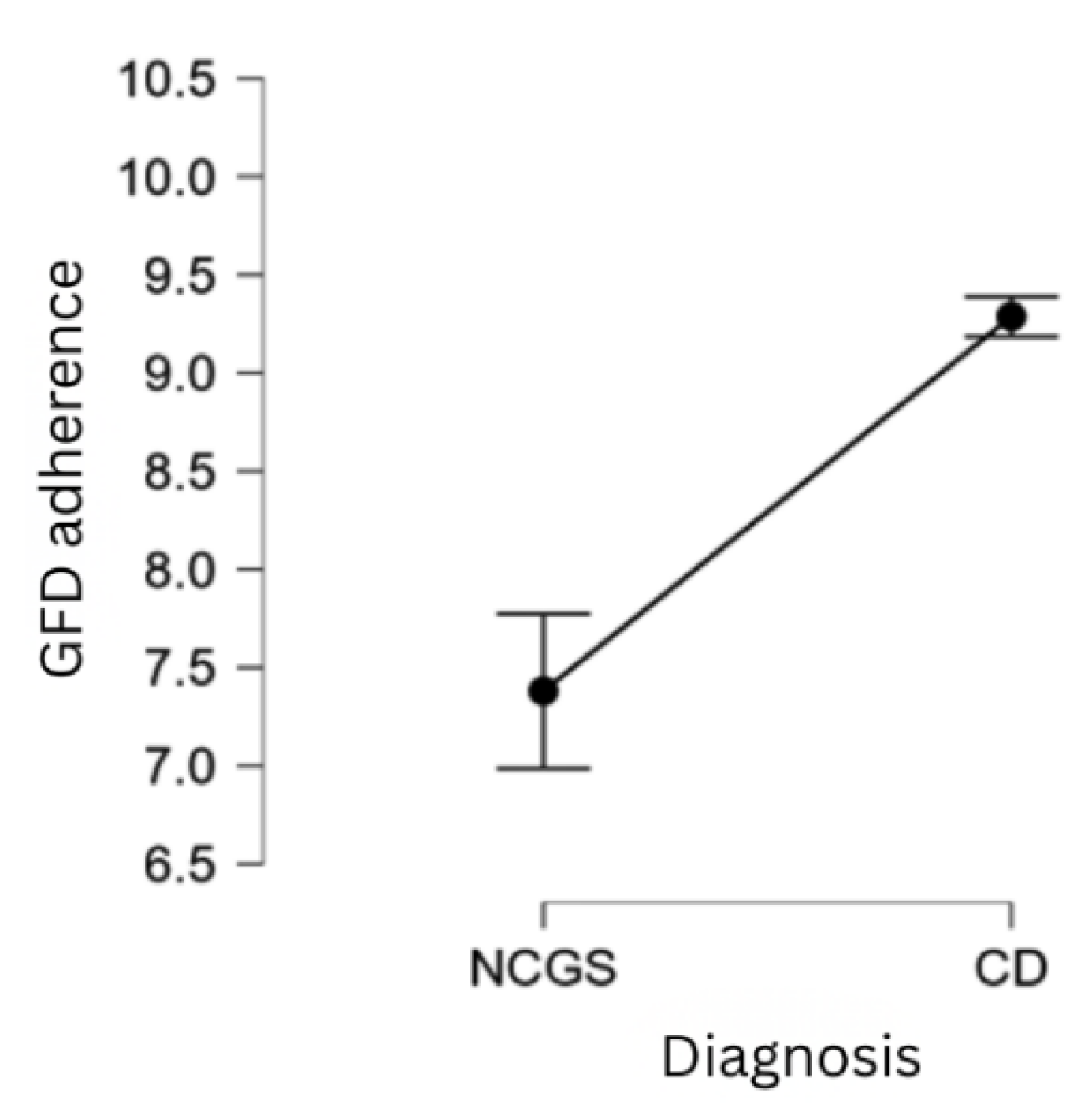
| H2 | Patients with CD exhibit greater adherence to the GFD compared to those with NCGS. |
| H3 | Time since diagnosis is positively correlated with dietary adherence (patients diagnosed longer ago adhere more strictly to the GFD). |
| H4 | Patients with multiple chronic conditions demonstrate higher adherence to the GFD than those without comorbidities. |
| H5 | An official medical diagnosis increases the likelihood of dietary adherence compared to self-diagnosis. |
| H6 | Patients who frequently read/check food labels show greater adherence to the GFD. There is an interaction effect between food label reading and time since diagnosis on dietary adherence. |
| Psychosocial Factors | |
| H7 | Personal acceptance of the diagnosis positively influences adherence to the GFD. |
| H8 | Patients who perceive the GFD as difficult are more likely to be non-adherent. |
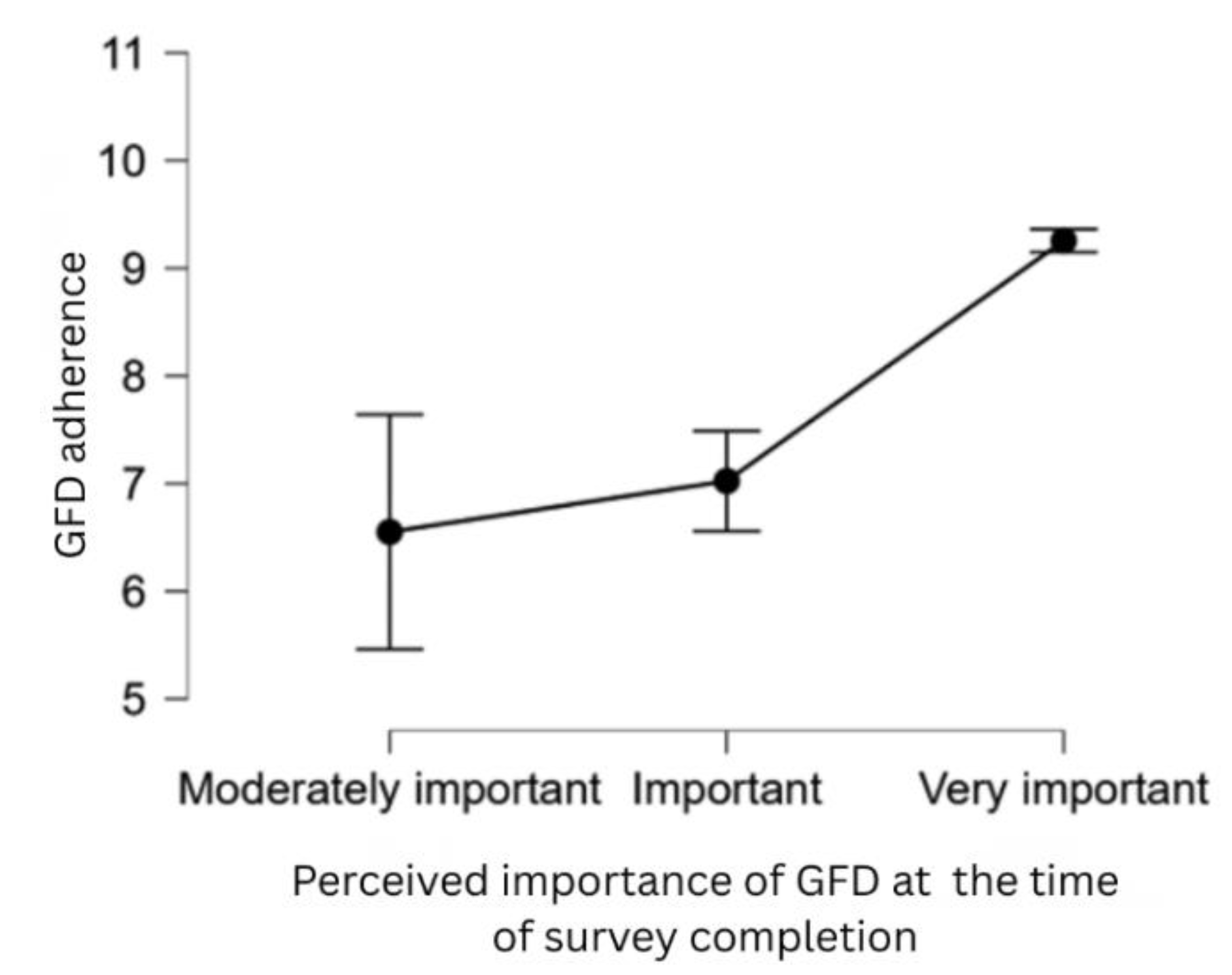
| H9 | Patients who perceive the GFD as more important are more likely to be adherent to it. |
| H10 | Frequent attendance at social events without GF options is associated with lower dietary adherence. Those who are more adherent to the GFD tend to isolate themselves more. |
| H11 | Patients who feel discriminated against or isolated because of their diet have lower adherence. |
| Economic and Accessibility Factors | |
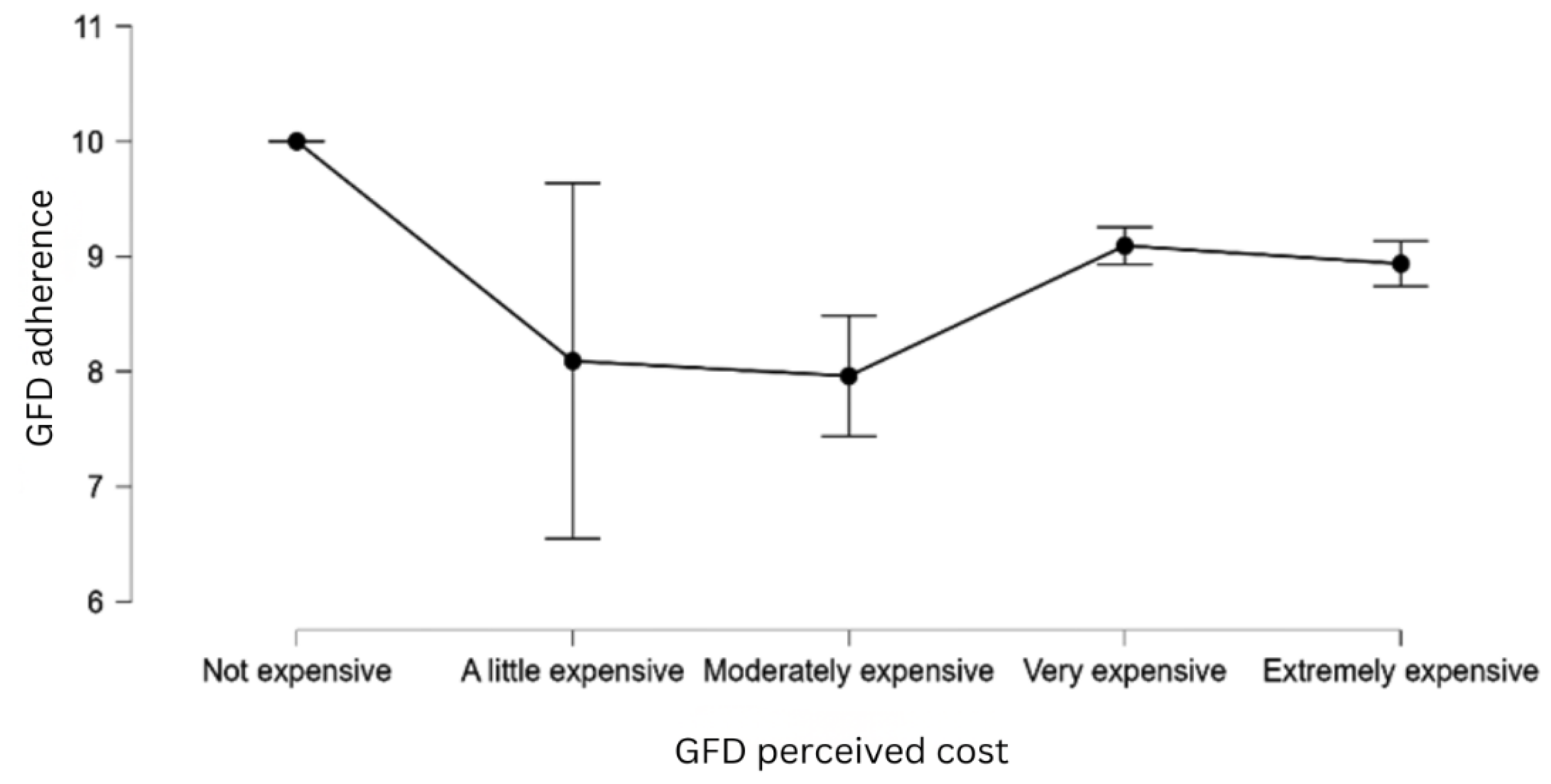
| H12 | Perceiving GF products as expensive is associated with lower dietary adherence. |
| H13 | Greater availability of GF products in stores and restaurants is positively correlated with dietary adherence. |
| H14 | Patients who receive nutritional education from specialists have higher adherence with diet therapy than those who self-educate. |
| № | Auxiliary Analyses and Study Questions |
|---|---|
| 1 | What is the overall rate of self-diagnosis among patients with GRDs? |
| 2 | Which patient group is more prone to self-diagnosis? (We initially hypothesized that those with NCGS would be more prone, as observed in other studies.) |
| 3 | Do GRDs show a hereditary component, such that individuals with CD report more affected relatives than those with NCGS? |
| 4 | Are patients with CD more likely to bring their own food to social events than those with NCGS? |
| 5 | Which patient group checks food labels more frequently — patients with CD or NCGS? |
| 6 | How frequently do individuals with GRDs, adhering to a GFD, experience symptoms after consuming products labeled as certified GF? |
| 7 | Is there an association between weight and GFD adherence, and between weight and time since diagnosis? |
| 8 | Which group finds the GFD to be more challenging — patients diagnosed with CD or those with NCGS? |
| 9 | Do those who feel discriminated against tend to isolate themselves more? Who isolate themselves more — patients with CD or NCGS? |
| 10 | Which group perceives the cost of dietary therapy as being higher — patients with CD or NCGS? |
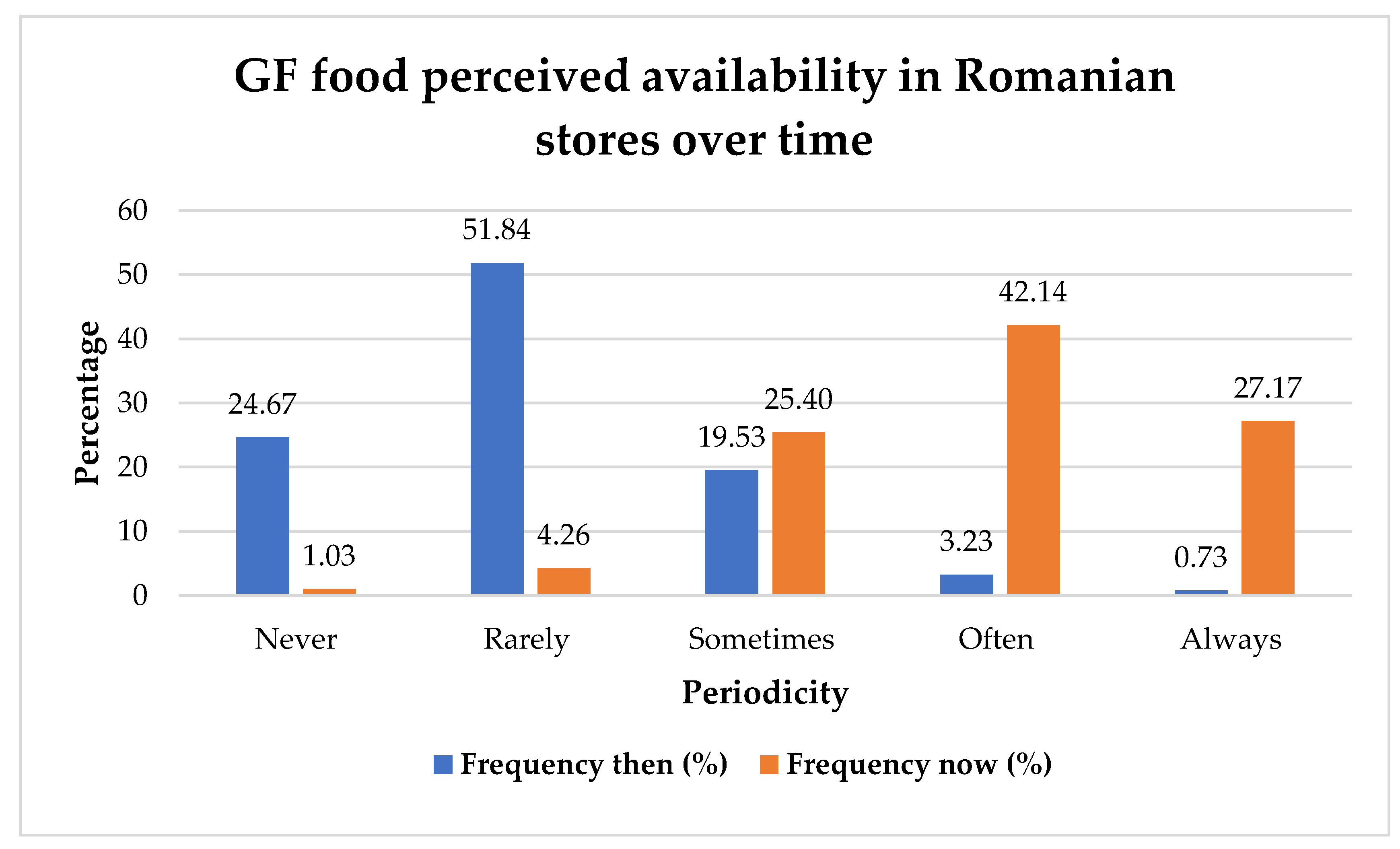
| 11 | How does time impact GF food perceived availability in stores? |
| 12 | Is there a correlation between the duration since diagnosis and the perceived availability of GF food? |
| 13 | From whom do the patients tend to learn more about their diet? |
| 14 | Do those with NCGS tend to learn more about the diet from non-specialists? |
| 15 | Do patients who receive nutritional education from specialists perceive diet therapy as easier to follow than those who self-educate? |
| 16 | Are patients who receive nutritional education from specialists less prone to making unsuitable dietary choices for the GFD than those who self-educate? |
| 17 | How prevalent is LI among GRDs patients? |
| 18 | What are the most commonly associated chronic diseases among GRD patients? |
| 19 | Among those who self-diagnosed: what are the most commonly associated chronic conditions? |
| Gender | n | Missing | Mean | SD | Min. | Max. |
|---|---|---|---|---|---|---|
| Male | 107 | 0 | 6.673 | 0.822 | 3 | 7 |
| Female | 574 | 0 | 6.427 | 1.085 | 2 | 7 |
| Diagnosis | Frequency | Percentage |
|---|---|---|
| CD | 539 | 79.148 |
| NCGS | 142 | 20.852 |
| Total | 681 | 100.000 |
| Diagnosed by | Frequency | Percentage |
|---|---|---|
| Specialist doctor | 606 | 88.987 |
| Self-diagnosis | 75 | 11.013 |
| Total | 681 | 100.000 |
| Model | SS | df | MS | F | p |
|---|---|---|---|---|---|
| Regression | 434.540 | 8 | 54.317 | 18.625 | < .001 |
| Residual | 1538.200 | 672 | 2.051 | ||
| Total | 1972.740 | 680 |
| Variable | Coefficient | Std. Err. | t | p |
|---|---|---|---|---|
| Family history of GRDs (answer: no) | 0.093 | 0.141 | 0.660 | 0.509 |
| Diagnosis (answer: CD) | 1.167 | 0.169 | 9.748 | < .001 |
| Time since diagnosis (answer: 1-2 yrs) | 0.201 | 0.199 | 1.011 | 0.312 |
| Time since diagnosis (answer: 2-5 yrs) | 0.149 | 0.183 | 0.811 | 0.418 |
| Time since diagnosis (answer: 5-10 yrs) | 0.273 | 0.182 | 1.498 | 0.135 |
| Time since diagnosis (answer: >10 yrs) | 0.126 | 0.202 | 0.623 | 0.533 |
| Self-diagnosed | -0.603 | 0.217 | -2.783 | 0.006 |
| Other chronic conditions (answer: no) | 0.084 | 0.124 | 0.675 | 0.500 |
| Model | SS | df | MS | F | p |
|---|---|---|---|---|---|
| Regression | 661.556 | 20 | 33.078 | 16.650 | < .001 |
| Residual | 1311.184 | 660 | 1.987 | ||
| Total | 1972.740 | 680 |
| Variable | Coefficient | Std. Err. | t | p |
|---|---|---|---|---|
| Perceived importance at diagnosis time (a little important) |
-1.850 | 0.657 | -2.817 | 0.005 |
| Perceived importance now (moderately important) |
-3.341 | 1.457 | -2.294 | 0.022 |
| Perceived importance now (important) |
-3.022 | 1.432 | -2.110 | 0.035 |
| Absence from social contexts (Sometimes) | 0.452 | 0.174 | 2.594 | 0.010 |
| Absence from social contexts (Most times) | 0.815 | 0.182 | 4.490 | < .001 |
| Model | Sum of Squares | df | Mean Square | F | p |
|---|---|---|---|---|---|
| Regression | 172.570 | 21 | 8.218 | 3.008 | < .001 |
| Residual | 1800.170 | 659 | 2.732 | ||
| Total | 1972.740 | 680 |
| Variable | Coefficient | Std. Err. | t | p |
|---|---|---|---|---|
| GFD cost (moderately expensive) | -2.790 | 1.286 | -2.168 | .030 |
| GF food alternatives in restaurants (sometimes) | -0.688 | 0.231 | -2.983 | .003 |
| GF food alternatives in restaurants (always) | -2.325 | 0.990 | -2.349 | .018 |
| Sources of information about GFD |
From a specialist doctor |
From a specialist nutritionist- dietitian |
Self-sourced information |
With the help of other people with the same diagnosis |
Total |
|---|---|---|---|---|---|
| CD | 22.82% (n=123) |
5.57% (n=30) |
48.98% (n=264) |
22.63% (n=122) |
100% (n=539) |
| NCGS | 12.68% (n=18) |
9.15% (n=13) |
63.38% (n=90) |
14.79% (n=21) |
100% (n=142) |
| Overall | 20.71% (n=141) | 6.31% (n=43) |
51.98% (n=354) |
21.00% (n=143) |
100% (n=681) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




