Submitted:
16 October 2025
Posted:
17 October 2025
You are already at the latest version
Abstract
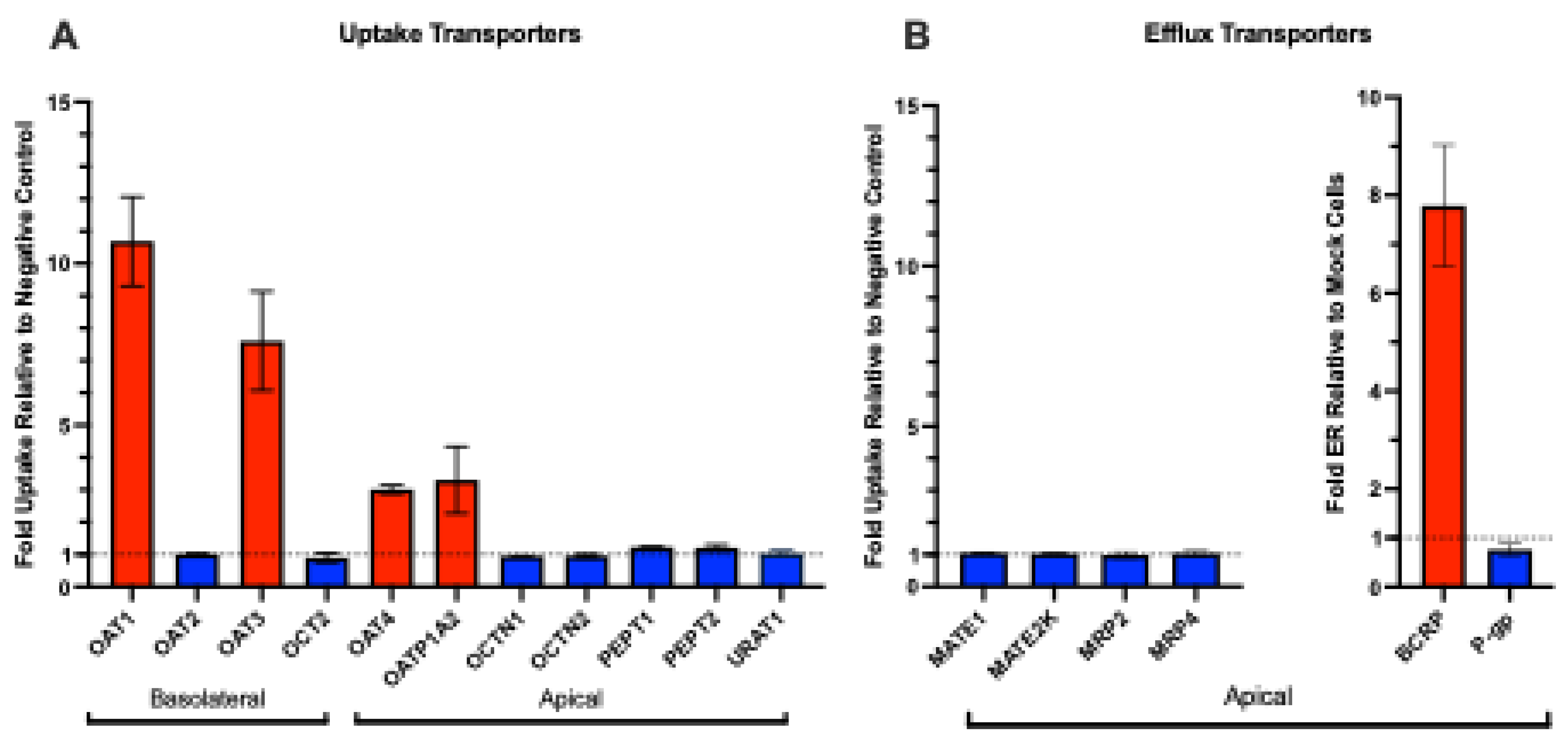
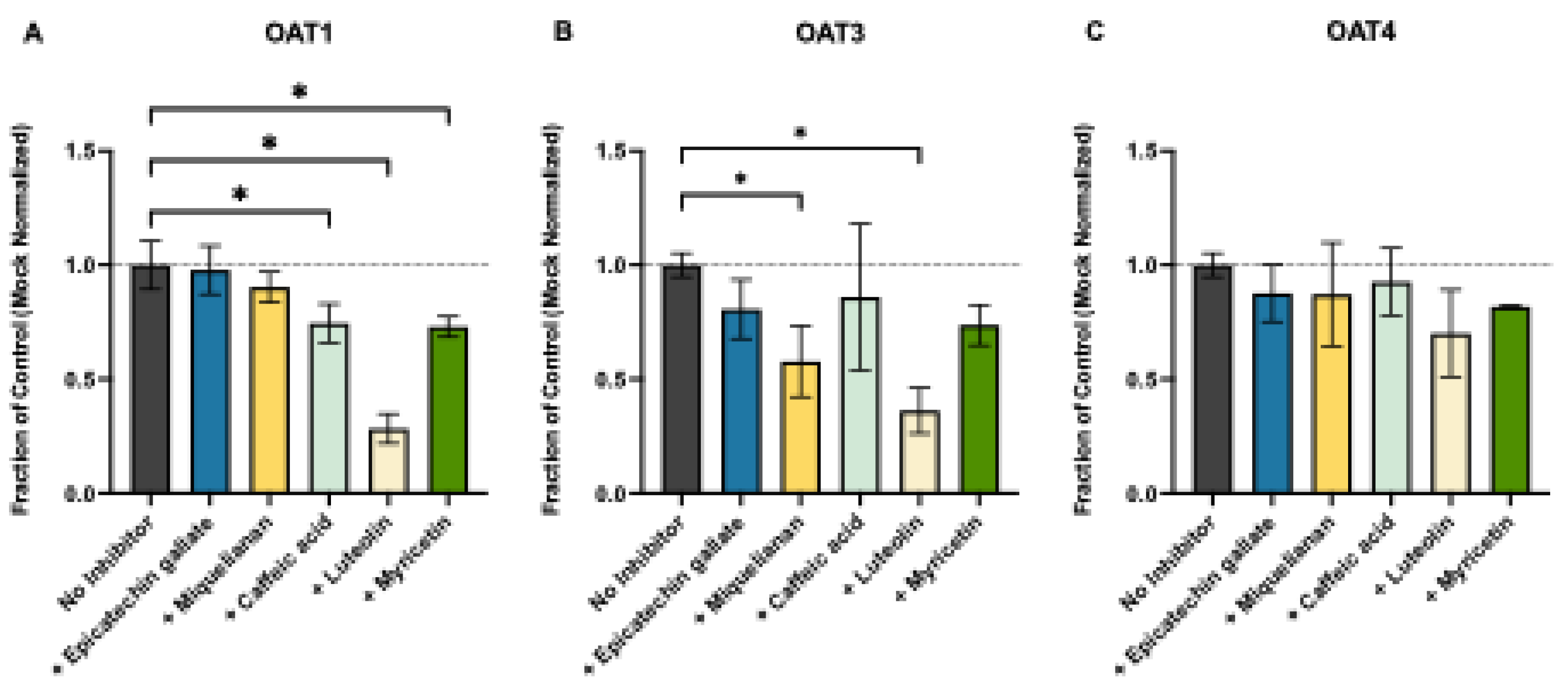
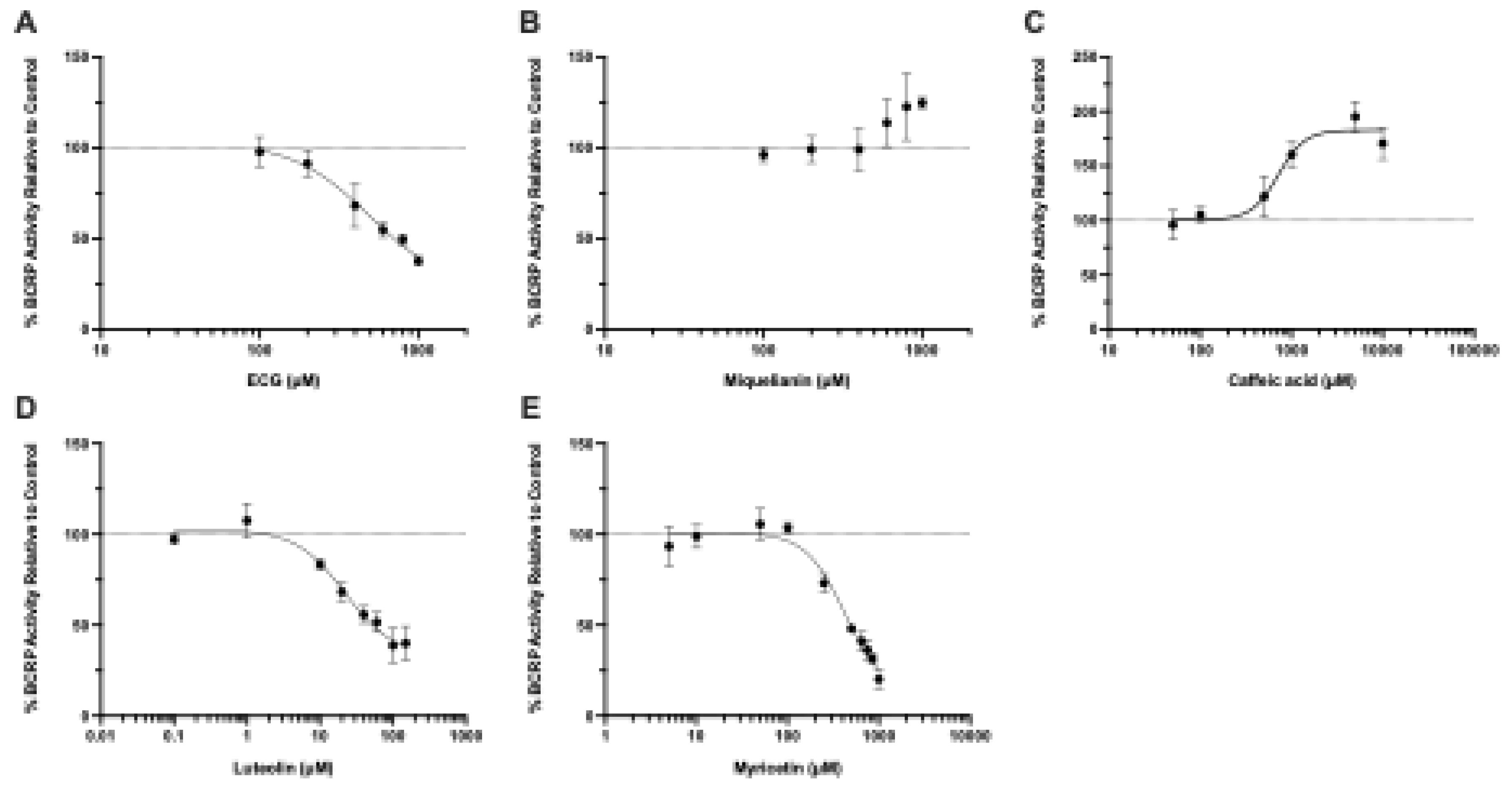
Background/Objectives: Ochratoxin A (OTA) is a widespread foodborne mycotoxin linked to chronic kidney disease of unknown etiology. Despite evidence from animal models showing OTA accumulation in the kidney, the molecular mechanisms underlying its renal disposition in humans remain only partially understood. Here. we identify human renal transporters responsible for OTA kidney accumulation, elimination, and establish Michaelis-Menten kinetics under matched conditions to directly compare transport mechanisms. We also aim to identify inhibition potential of these transport mechanisms with common dietary polyphenols. Methods: Mammalian cells and membrane vesicles overexpressing human renal transporters were used to screen and profile the uptake and efflux of OTA. Miquelianin, (-)-Epicatechin-3-O-gallate, myricetin, luteolin, and caffeic acid were tested as potential concentration-dependent transporter inhibitors. Results: We demonstrate that OTA is a substrate for human organic anion transporter (hOAT) 1 (Km: 2.10 ± 0.50 μM, Vmax: 396.9 ± 27.0 pmol/mg/min), hOAT3 (Km: 2.58 ± 0.83 μM, Vmax: 141.4 ± 30.3 pmol/mg/min), hOAT4 (Km: 6.38 ± 1.45 μM, Vmax: 96.9 ± 18.8 pmol/mg/min), and human organic anion transporting polypeptide (hOATP) 1A2 (Km: 37.3 ± 6.2 μM, Vmax: 801.0 ± 133.9 pmol/mg/min). Among efflux transporters, OTA was transported only by human breast cancer resistance protein (hBCRP), which has minimal renal expression. While none of the uptake transporters were potently inhibited (>90%) by polyphenols at 10 μM, luteolin inhibited hBCRP-mediated transport of OTA with an IC50 of 22 μM and caffeic acid stimulated hBCRP-mediated efflux with an EC50 of 713.8 μM, both of which are physiologically relevant intestinal lumen concentrations. Conclusions: Our results confirm that exposures to OTA will lead to renal accumulation and increased health risks in affected populations, necessitating increased scrutiny of our food sources.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Cell Culture
2.3. Uptake Assays in Transporter-overexpressing Mock HEK293 and CHO cells
2.4. Uptake Assays in MRP2 and MRP4-expressing Sf9 Membrane Vesicles
2.5. Uptake Assays in PEPT1, PEPT2, and URAT1-expressing MDCK-II Cells
2.6. Bidirectional Transport Assays in BCRP and P-gp-expressing MDCK-II Cells
2.7. Transporter-Mediated Natural Product-OTA Interaction Study
2.8. Quantification of OTA by LC-MS/MS
2.9. Data and Statistical Analysis
3. Results
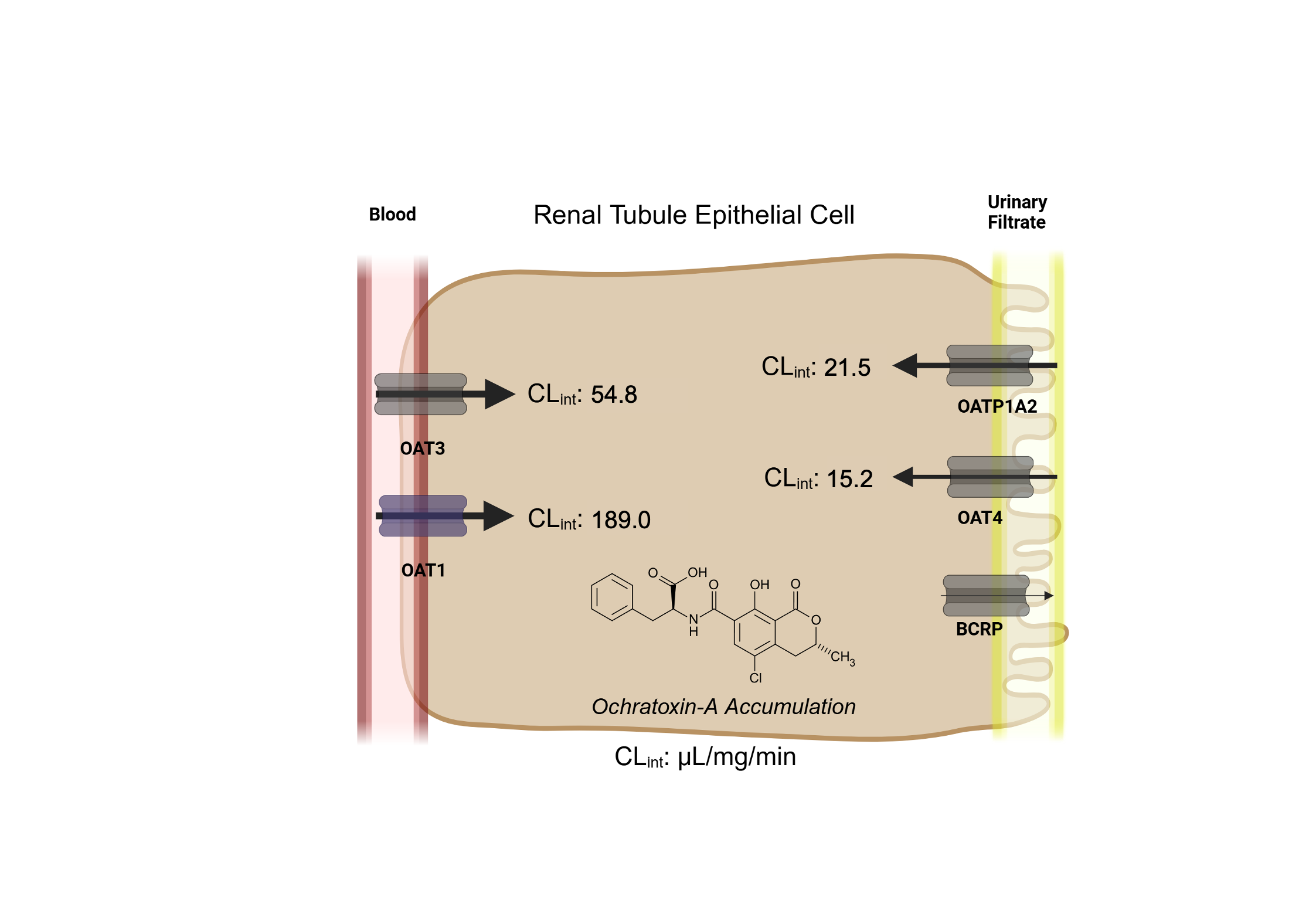
3.1. Human Renal Transport Screen for OTA
3.2. Transporter Kinetics of OTA via OAT1, OAT3, OAT4, OATP1A2, and BCRP
3.3. Natural Product-OTA Interactions at Human OAT1, OAT3, and OAT4
3.4. Natural Product-OTA Interactions at Human BCRP
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BCA | Bicinchoninic Acid |
| BCRP | Breast Cancer Resistance Protein |
| CHO | Chinese Hamster Ovary |
| CKDu | Chronic Kidney Disease of Unknown Etiology |
| DPBS | Dulbecco’s Phosphate-Buffered Saline |
| EC50 | Half-maximal Effective Concentration |
| ECG | (-)-Epicatechin-3-O-gallate |
| FBS | Fetal Bovine Serum |
| HBSS++ | Hank’s balanced salt solution with calcium and magnesium |
| HCl | Hydrochloric Acid |
| HEK293 | Human Embryonic Kidney 293 |
| HSA | Human Serum Albumin |
| IC50 | Half-maximal Inhibitory Concentration |
| MATEs | Multidrug and Toxin Extrusion Proteins |
| MDCK-II | Madin-Darby Canine Kidney II |
| MRPs | Multidrug Resistance-associated Proteins |
| NaOH | Sodium Hydroxide |
| OAT | Organic Anion Transporters |
| OATP1A2 | Organic Anion Transporting Polypeptide 1A2 |
| OCT2 | Organic Cation Transporter 2 |
| OCTNs | Organic Cation/carnitine Transporters |
| OTA | Ochratoxin A |
| P-gp | P-glycoprotein |
| PEPTs | Peptide Transporters |
| Sf9 | Spodoptera Frugiperda 9 |
| SLC | Solute Carriers |
| URAT1 | Urate Transporter 1 |
| BCA | Bicinchoninic Acid |
| BCRP | Breast Cancer Resistance Protein |
| CHO | Chinese Hamster Ovary |
References
- Duarte SC, Pena A, Lino CM. A review on ochratoxin A occurrence and effects of processing of cereal and cereal derived food products. Food Microbiology. 2010;27(2):187-198. [CrossRef]
- Nguyen KTN, Ryu D. Concentration of ochratoxin A in breakfast cereals and snacks consumed in the United States. Food Control. 2014;40:140-144. [CrossRef]
- Khoi CS, Chen JH, Lin TY, Chiang CK, Hung KY. Ochratoxin A-Induced Nephrotoxicity: Up-to-Date Evidence. Int J Mol Sci. 2021;22(20):11237. [CrossRef]
- Wang W, Zhai S, Xia Y, et al. Ochratoxin A induces liver inflammation: involvement of intestinal microbiota. Microbiome. 2019;7(1):151. [CrossRef]
- Mateo E, Tonino RPB, Canto A, et al. The Neurotoxic Effect of Ochratoxin-A on the Hippocampal Neurogenic Niche of Adult Mouse Brain. Toxins (Basel). 2022;14(9):624. [CrossRef]
- Desalegn B, Nanayakkara S, Harada KH, et al. Mycotoxin Detection in Urine Samples from Patients with Chronic Kidney Disease of Uncertain Etiology in Sri Lanka. Bull Environ Contam Toxicol. 2011;87(1):6-10. [CrossRef]
- Maaroufi K, Achour A, Hammami M, et al. Ochratoxin A in human blood in relation to nephropathy in Tunisia. Hum Exp Toxicol. 1995;14(7):609-614. [CrossRef]
- Daniel C, Enghard P, Ratnatunga N, et al. Early Ultrastructural Changes in Biopsies From Patients With Symptomatic CKD of Uncertain Etiology. Kidney International Reports. 2023;8(3):642-657. [CrossRef]
- Vervaet BA, Nast CC, Jayasumana C, et al. Chronic interstitial nephritis in agricultural communities is a toxin-induced proximal tubular nephropathy. Kidney International. 2020;97(2):350-369. [CrossRef]
- Studer-Rohr I, Schlatter J, Dietrich DR. Kinetic parameters and intraindividual fluctuations of ochratoxin A plasma levels in humans. Arch Toxicol. 2000;74(9):499-510. [CrossRef]
- Schwerdt, G. Accumulation of ochratoxin A in rat kidney in vivo and in cultivated renal epithelial cells in vitro. Toxicology. 1996;114(3):177-185. [CrossRef]
- Zepnik H, Völkel W, Dekant W. Toxicokinetics of the mycotoxin ochratoxin A in F 344 rats after oral administration. Toxicology and Applied Pharmacology. 2003;192(1):36-44. [CrossRef]
- Tsuda M, Sekine T, Takeda M, et al. Transport of ochratoxin A by renal multispecific organic anion transporter 1. J Pharmacol Exp Ther. 1999;289(3):1301-1305.
- Jung KY, Takeda M, Kim DK, et al. Characterization of ochratoxin A transport by human organic anion transporters. Life Sciences. 2001;69(18):2123-2135. [CrossRef]
- Babu E, Takeda M, Narikawa S, et al. Role of human organic anion transporter 4 in the transport of ochratoxin A. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 2002;1590(1):64-75. [CrossRef]
- Schrickx J, Lektarau Y, Fink-Gremmels J. Ochratoxin A secretion by ATP-dependent membrane transporters in Caco-2 cells. Arch Toxicol. 2006;80(5):243-249. [CrossRef]
- Salphati L, Chu X, Chen L, et al. Evaluation of Organic Anion Transporting Polypeptide 1B1 and 1B3 Humanized Mice as a Translational Model to Study the Pharmacokinetics of Statins. Drug Metabolism and Disposition. 2014;42(8):1301-1313. [CrossRef]
- Tsang YP, Rodriguez AG, Warren MS, Unadkat JD. Identification of selective substrates and inhibitors of the major human renal uptake transporters. Drug Metabolism and Disposition. 2025;53(3):100046. [CrossRef]
- An G, Wang X, Morris ME. Flavonoids Are Inhibitors of Human Organic Anion Transporter 1 (OAT1)–Mediated Transport. Drug Metab Dispos. 2014;42(9):1357-1366. [CrossRef]
- Uwai Y, Kawasaki T, Nabekura T. Caffeic acid inhibits organic anion transporters OAT1 and OAT3 in rat kidney. Drug Metabol Drug Interact. 2013;28(4):247-250. [CrossRef]
- Kawasaki T, Kondo M, Hiramatsu R, Nabekura T. (−)-Epigallocatechin-3-gallate Inhibits Human and Rat Renal Organic Anion Transporters. ACS Omega. 2021;6(6):4347-4354. [CrossRef]
- Hagelberg S, Hult K, Fuchs R. Toxicokinetics of ochratoxin A in several species and its plasma-binding properties. Journal of Applied Toxicology. 1989;9(2):91-96. [CrossRef]
- Zhang X, Groves CE, Bahn A, et al. Relative contribution of OAT and OCT transporters to organic electrolyte transport in rabbit proximal tubule. American Journal of Physiology-Renal Physiology. 2004;287(5):F999-F1010. [CrossRef]
- Martinez-Guerrero LJ, Zhang X, Wright SH, Cherrington NJ. Characterization of Human Organic Anion Transporter 4 (hOAT4) and Mouse Oat5 (mOat5) As Functional Orthologs for Renal Anion Uptake and Efflux Transport. The Journal of Pharmacology and Experimental Therapeutics. 2024;391(3):378-386. [CrossRef]
- Hagos Y, Stein D, Ugele B, Burckhardt G, Bahn A. Human Renal Organic Anion Transporter 4 Operates as an Asymmetric Urate Transporter. Journal of the American Society of Nephrology. 2007;18(2):430. [CrossRef]
- Kullak-Ublick GA, Hagenbuch B, Stieger B, et al. Molecular and functional characterization of an organic anion transporting polypeptide cloned from human liver. Gastroenterology. 1995;109(4):1274-1282. [CrossRef]
- Badagnani I, Castro RA, Taylor TR, et al. Interaction of Methotrexate with Organic-Anion Transporting Polypeptide 1A2 and Its Genetic Variants. The Journal of Pharmacology and Experimental Therapeutics. 2006;318(2):521-529. [CrossRef]
- Roncal-Jimenez C, García-Trabanino R, Barregard L, et al. Heat Stress Nephropathy From Exercise-Induced Uric Acid Crystalluria: A Perspective on Mesoamerican Nephropathy. American Journal of Kidney Diseases. 2016;67(1):20-30. [CrossRef]
- De Silva PMCS, Ekanayake EMDV, Gunasekara TDKSC, et al. Occupational heat exposure alone does not explain chronic kidney disease of uncertain aetiology (CKDu) in Sri Lanka. The Journal of Climate Change and Health. 2022;8:100143. [CrossRef]
- Schwerdt G, Gekle M, Freudinger R, Mildenberger S, Silbernagl S. Apical-to-basolateral transepithelial transport of Ochratoxin A by two subtypes of Madin-Darby canine kidney cells. Biochim Biophys Acta. 1997;1324(2):191-199. [CrossRef]
- Schwerdt G, Freudinger R, Silbernagl S, Gekle M. Apical uptake of radiolabelled ochratoxin A into Madin-Darby canine kidney cells. Toxicology. 1998;131(2-3):193-202. [CrossRef]
- Qi X, Wagenaar E, Xu W, Huang K, Schinkel AH. Ochratoxin A transport by the human breast cancer resistance protein (BCRP), multidrug resistance protein 2 (MRP2), and organic anion-transporting polypeptides 1A2, 1B1 and 2B1. Toxicology and Applied Pharmacology. 2017;329:18-25. [CrossRef]
- Basit A, Radi Z, Vaidya VS, Karasu M, Prasad B. Kidney Cortical Transporter Expression across Species Using Quantitative Proteomics. Drug Metabolism and Disposition. 2019;47(8):802-808. [CrossRef]
- Huls M, Brown CDA, Windass AS, et al. The breast cancer resistance protein transporter ABCG2 is expressed in the human kidney proximal tubule apical membrane. Kidney International. 2008;73(2):220-225. [CrossRef]
- Oswald S, Müller J, Neugebauer U, et al. Protein Abundance of Clinically Relevant Drug Transporters in The Human Kidneys. International Journal of Molecular Sciences. 2019;20(21):5303. [CrossRef]
- Liu M, Cheng C, Li X, et al. Luteolin alleviates ochratoxin A induced oxidative stress by regulating Nrf2 and HIF-1α pathways in NRK-52E rat kidney cells. Food and Chemical Toxicology. 2020;141:111436. [CrossRef]
- Costa S, Utan A, Cervellati R, Speroni E, Guerra MC. Catechins: natural free-radical scavengers against ochratoxin A-induced cell damage in a pig kidney cell line (LLC-PK1). Food and Chemical Toxicology. 2007;45(10):1910-1917. [CrossRef]
- El-Haleem MRA, Kattaia AAA, El-Baset SAA, Mostafa HES. Alleviative effect of myricetin on ochratoxin A-induced oxidative stress in rat renal cortex: histological and biochemical study. Histol Histopathol. 2016;31(4):441-451. [CrossRef]
- Cariddi LN, Sabini MC, Escobar FM, et al. Polyphenols as possible bioprotectors against cytotoxicity and DNA damage induced by ochratoxin A. Environmental Toxicology and Pharmacology. 2015;39(3):1008-1018. [CrossRef]
- Grgić J, Šelo G, Planinić M, Tišma M, Bucić-Kojić A. Role of the Encapsulation in Bioavailability of Phenolic Compounds. Antioxidants (Basel). 2020;9(10):923. [CrossRef]
- Peters CM, Green RJ, Janle EM, Ferruzzi MG. Formulation with ascorbic acid and sucrose modulates catechin bioavailability from green tea. Food Res Int. 2010;43(1):95-102. [CrossRef]
- Selma MV, Espín JC, Tomás-Barberán FA. Interaction between phenolics and gut microbiota: role in human health. J Agric Food Chem. 2009;57(15):6485-6501. [CrossRef]
- In Vitro Metabolism- and Transporter- Mediated Drug-Drug Interaction Studies Guidance for Industry. Published online October 2017.
- Teng YN, Wang CCN, Liao WC, Lan YH, Hung CC. Caffeic Acid Attenuates Multi-Drug Resistance in Cancer Cells by Inhibiting Efflux Function of Human P-glycoprotein. Molecules. 2020;25(2):247. [CrossRef]
- Tan SPF, Tillmann A, Murby SJ, Rostami-Hodjegan A, Scotcher D, Galetin A. Albumin-Mediated Drug Uptake by Organic Anion Transporter 1/3 Is Real: Implications for the Prediction of Active Renal Secretion Clearance. Mol Pharmaceutics. 2024;21(9):4603-4617. [CrossRef]
- Scherpinski LA, Fromm MF, Maas R, König J. Transport of the uremic toxin symmetric dimethylarginine (SDMA) by renal transport proteins. Amino Acids. 2025;57(1):34. [CrossRef]
- Taghikhani E, Maas R, Fromm MF, König J. The renal transport protein OATP4C1 mediates uptake of the uremic toxin asymmetric dimethylarginine (ADMA) and efflux of cardioprotective L-homoarginine. PLoS One. 2019;14(3):e0213747. [CrossRef]
- Žlender V, Breljak D, Ljubojević M, et al. Low doses of ochratoxin A upregulate the protein expression of organic anion transporters Oat1, Oat2, Oat3 and Oat5 in rat kidney cortex. Toxicology and Applied Pharmacology. 2009;239(3):284-296. [CrossRef]
- Mitchell NJ, Chen C, Palumbo JD, et al. A risk assessment of dietary Ochratoxin a in the United States. Food Chem Toxicol. 2017;100:265-273. [CrossRef]
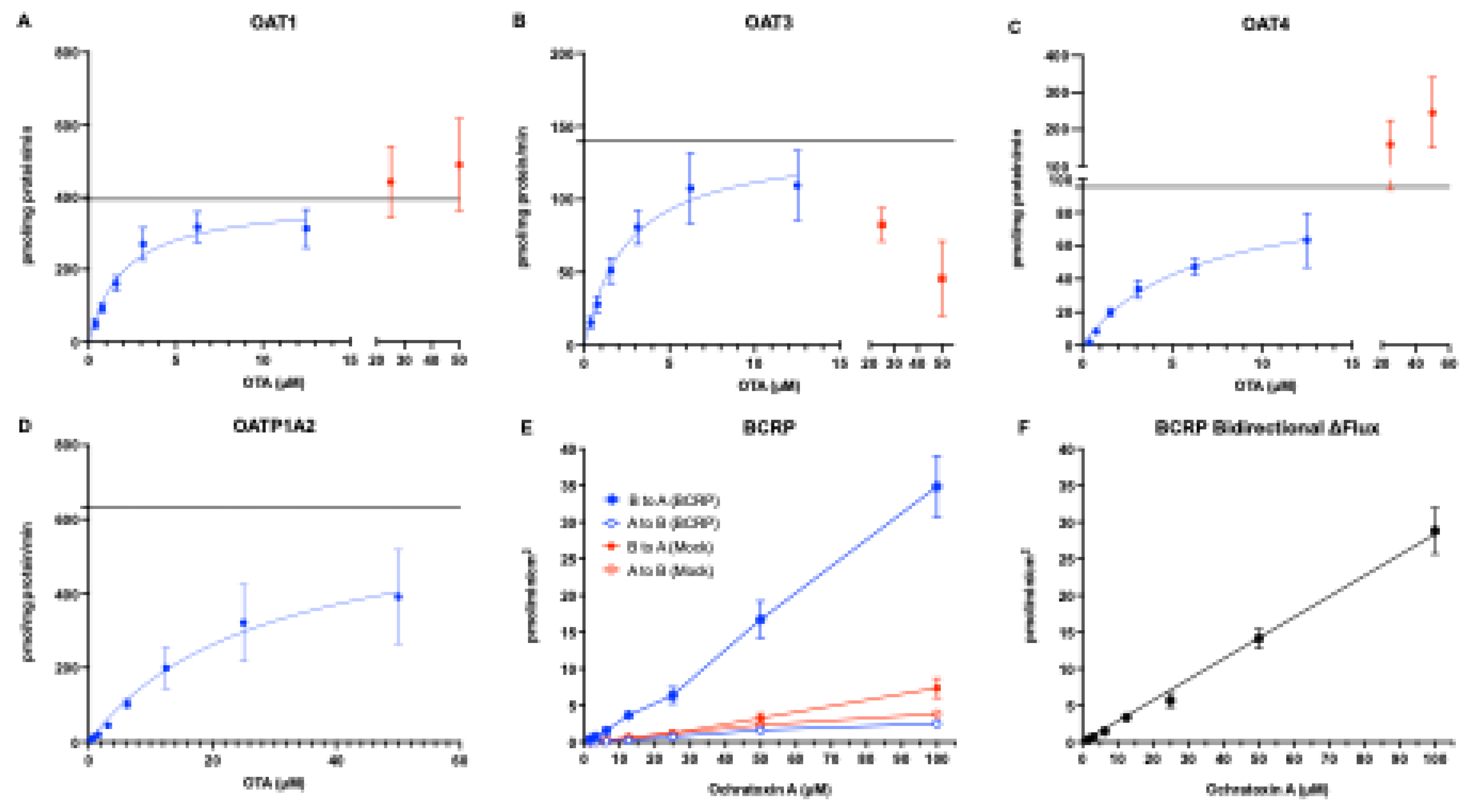
| OAT1 | OAT3 | OAT4 | OATP1A2 | |
|---|---|---|---|---|
| Km (μM) | 2.10 ± 0.50 | 2.58 ± 0.83 | 6.38 ± 1.45 | 37.3 ± 6.2 |
| Vmax (pmol/mg/min) | 396.9 ± 27.0 | 141.4 ± 30.3 | 96.9 ± 18.8 | 801.0 ± 133.9 |
| CLint (μL/mg/min) | 194.4 ± 34.4 | 56.1 ± 6.3 | 15.3 ± 0.8 | 21.6 ± 2.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




