Submitted:
09 October 2025
Posted:
13 October 2025
You are already at the latest version
Abstract
Keywords:
1. Background
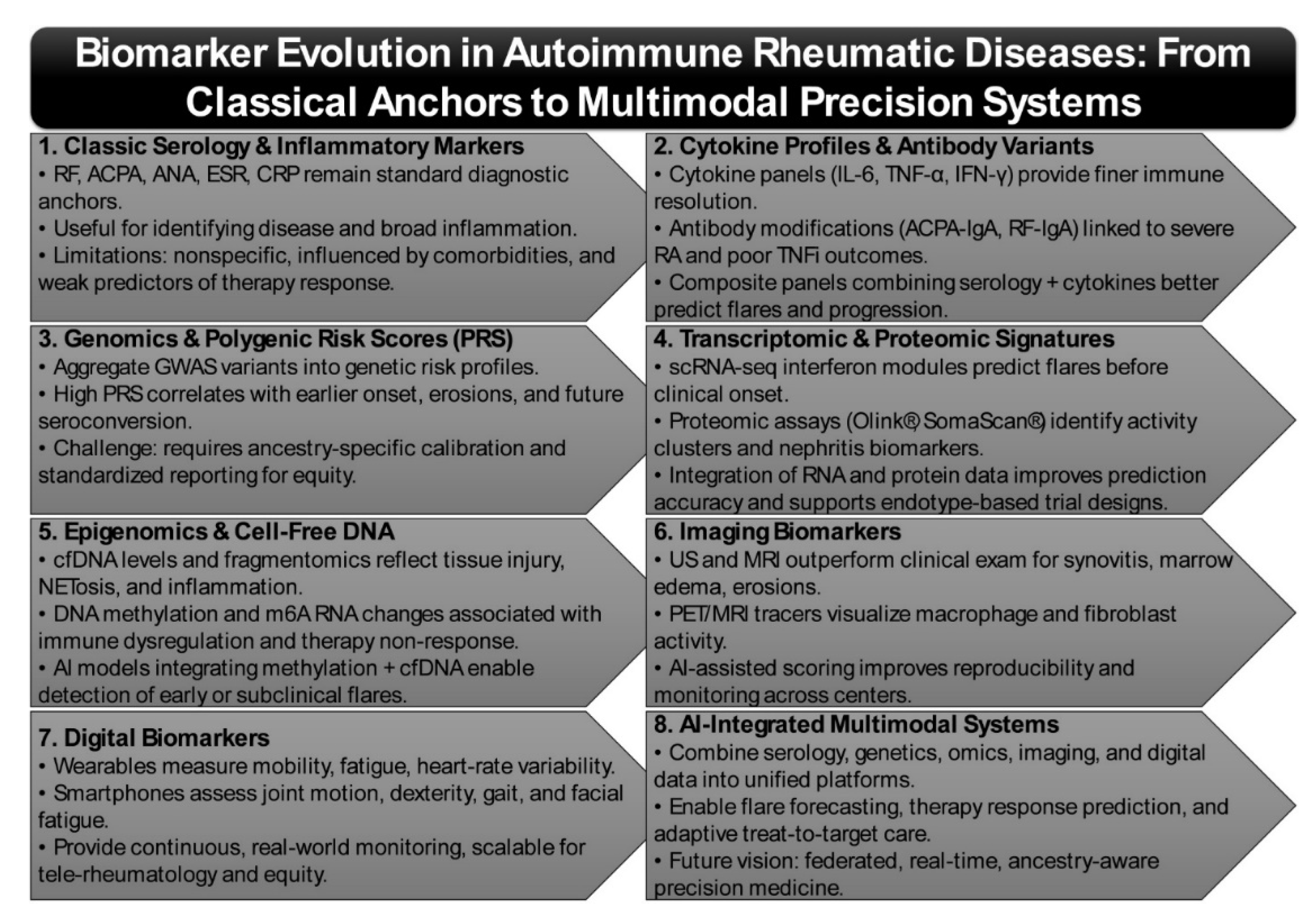
2. Biomarker Evolution in Autoimmune Rheumatic Diseases
2.1. Classic Biomarkers—Autoantibodies and Inflammatory Markers: Limitations and Drift
2.2. Genomics & Polygenic Risk
2.3. Transcriptomic & Proteomic Signatures
2.4. Epigenomic Alterations and Cell-Free DNA/Fragmentomics as Emerging Biomarkers
2.5. Imaging Biomarkers
2.6. Digital Biomarkers (Wearables/Smartphones)
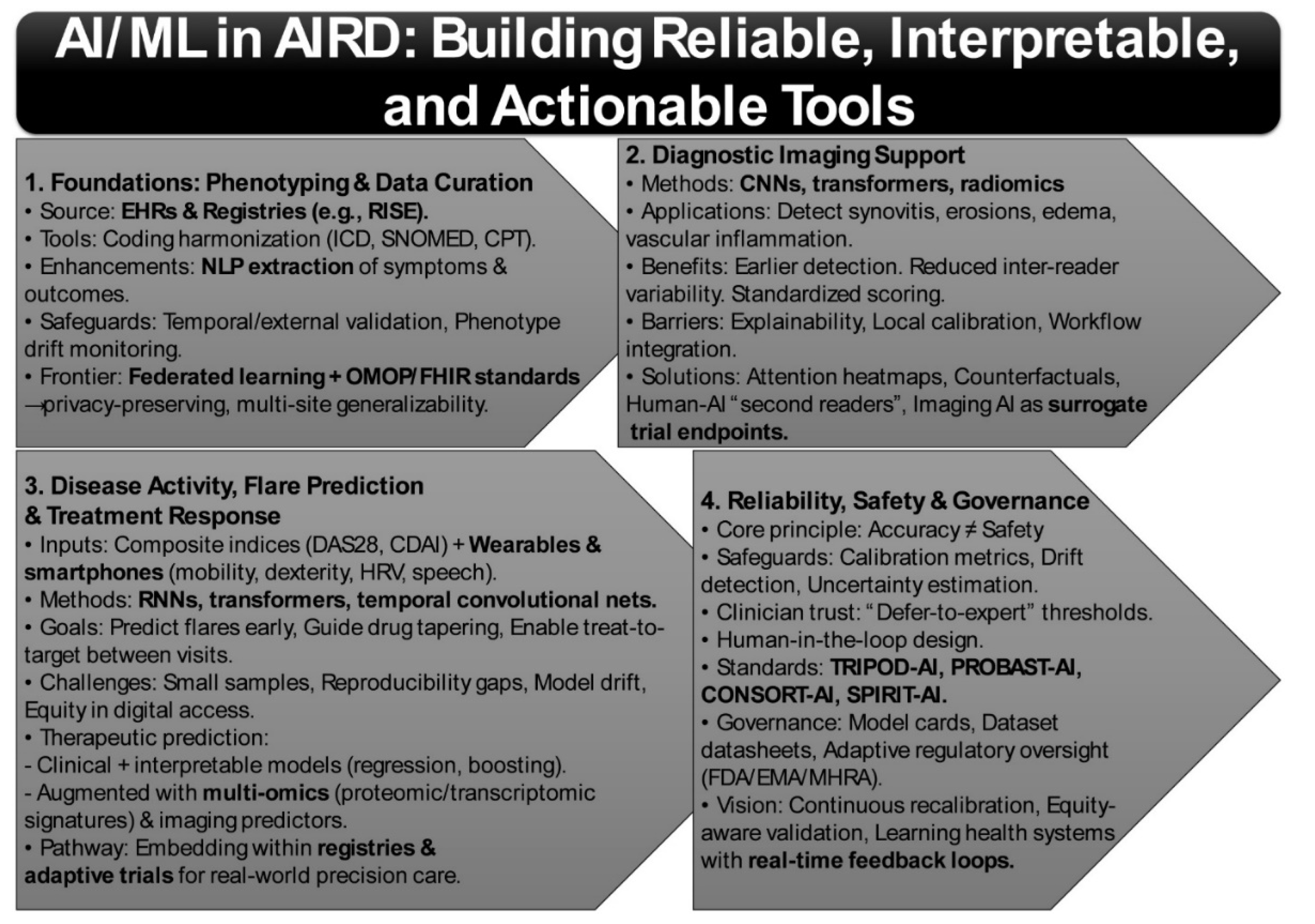
3. Harnessing AI and Machine Learning for Autoimmune Rheumatic Diseases
3.1. Phenotyping and EHR Curation
3.2. Diagnostic Imaging Support
3.3. Disease Activity, Flare Prediction, and Treatment Response
3.4. Reliability, Safety, and Governance
4. Redefining Autoimmune Rheumatic Disease Pathways: From Immune Signatures to AI-Enhanced Precision Medicine
4.1. Rheumatoid Arthritis (RA)
4.2. Systemic Lupus Erythematosus (SLE)
4.2.1. IFN Signature & Targeted Therapy
4.2.2. Digital Measures & Flare Prediction
4.3. Systemic Sclerosis (SSc)
4.4. Spondyloarthritis/Psoriatic Arthritis (SpA/PsA)
4.5. Other Conditions
4.5.1. Sjögren’s Syndrome (SjD)
4.5.2. Idiopathic Inflammatory Myopathies (IIM)
4.5.3. Vasculitides
5. Artificial Intelligence in Rheumatology: From Triage to Therapy Selection
5.1. AI-Enhanced Triage and Access
5.2. Imaging Decision Support
5.3. Predictive Tools for Therapy Selection
6. Data Infrastructures for AI in Rheumatology: Registries, Interoperability, and Federated Collaboration
6.1. Registries and EHR as Foundational Substrates
6.2. Interoperability and Common Data Models
6.3. Privacy-Preserving Collaboration Through Federated Learning
6.4. Pitfalls of Multisite Modeling and Mitigation Strategies
6.5. Case Illustration: Predicting RA Disease Activity Using RISE
7. Standards and Study Designs for AI Prediction Models in Clinical Research
7.1. Core Reporting Standards for Prediction Models
7.2. Study Design Foundations: Reviewer Expectations and Best Practices
8.1. PRS Portability and Ancestry Gaps
8.2. Data Drift, Bias Audits, and Transparent Documentation
8.3. Governance and Safety by Design
9. Future Directions
9.1. Multimodal Fusion (Omics, Imaging, and Digital Phenotypes)
9.2. Mechanism-Aware Machine Learning to Guide Drug Targeting
9.3. Digital Twins, N-of-1 Trials, Adaptive Platforms, and Home Testing
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tanaka, H.; Okada, Y.; Nakayamada, S.; Miyazaki, Y.; Sonehara, K.; Namba, S.; Honda, S.; Shirai, Y.; Yamamoto, K.; Kubo, S.; et al. Extracting immunological and clinical heterogeneity across autoimmune rheumatic diseases by cohort-wide immunophenotyping. Ann Rheum Dis 2024, 83, 242–252. [Google Scholar] [CrossRef]
- Al-Ewaidat, O.A.; Naffaa, M.M. Stroke risk in rheumatoid arthritis patients: exploring connections and implications for patient care. Clin Exp Med 2024, 24, 30. [Google Scholar] [CrossRef]
- Bilgin, E. Current application, possibilities, and challenges of artificial intelligence in the management of rheumatoid arthritis, axial spondyloarthritis, and psoriatic arthritis. Ther Adv Musculoskelet Dis 2025, 17, 1759720X251343579. [Google Scholar] [CrossRef] [PubMed]
- Stafford, I.S.; Kellermann, M.; Mossotto, E.; Beattie, R.M.; MacArthur, B.D.; Ennis, S. A systematic review of the applications of artificial intelligence and machine learning in autoimmune diseases. NPJ Digit Med 2020, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Carotti, M.; Di Carlo, M.; Ceccarelli, L.; Farah, S.; Poliseno, A.C.; Di Matteo, A.; Bandinelli, F.; Giovagnoni, A. Magnetic Resonance Imaging (MRI)-Based Semi-Quantitative Methods for Rheumatoid Arthritis: From Scoring to Measurement. J Clin Med 2024, 13. [Google Scholar] [CrossRef]
- Parodis, I.; Lindblom, J.; Toro-Dominguez, D.; Beretta, L.; Borghi, M.O.; Castillo, J.; Carnero-Montoro, E.; Enman, Y.; Mohan, C.; Alarcon-Riquelme, M.E.; et al. Interferon and B-cell Signatures Inform Precision Medicine in Lupus Nephritis. Kidney Int Rep 2024, 9, 1817–1835. [Google Scholar] [CrossRef] [PubMed]
- Al-Ewaidat, O.A.; Naffaa, M.M. Deciphering Mechanisms, Prevention Strategies, Management Plans, Medications, and Research Techniques for Strokes in Systemic Lupus Erythematosus. Medicines (Basel) 2024, 11. [Google Scholar] [CrossRef]
- Creagh, A.P.; Hamy, V.; Yuan, H.; Mertes, G.; Tomlinson, R.; Chen, W.H.; Williams, R.; Llop, C.; Yee, C.; Duh, M.S.; et al. Digital health technologies and machine learning augment patient reported outcomes to remotely characterise rheumatoid arthritis. NPJ Digit Med 2024, 7, 33. [Google Scholar] [CrossRef]
- Creagh, A.P.; Dondelinger, F.; Lipsmeier, F.; Lindemann, M.; De Vos, M. Longitudinal Trend Monitoring of Multiple Sclerosis Ambulation Using Smartphones. IEEE Open J Eng Med Biol 2022, 3, 202–210. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.; Li, M.; Shang, S.; Cao, Y.; Shen, X.; Huang, C. Artificial intelligence in autoimmune diseases: a bibliometric exploration of the past two decades. Front Immunol 2025, 16, 1525462. [Google Scholar] [CrossRef]
- Sequi-Sabater, J.M.; Benavent, D. Artificial intelligence in rheumatology research: what is it good for? RMD Open 2025, 11. [Google Scholar] [CrossRef]
- Hammam, N.; Izadi, Z.; Li, J.; Evans, M.; Kay, J.; Shiboski, S.; Schmajuk, G.; Yazdany, J. The Relationship Between Electronic Health Record System and Performance on Quality Measures in the American College of Rheumatology's Rheumatology Informatics System for Effectiveness (RISE) Registry: Observational Study. JMIR Med Inform 2021, 9, e31186. [Google Scholar] [CrossRef]
- Yazdany, J.; Bansback, N.; Clowse, M.; Collier, D.; Law, K.; Liao, K.P.; Michaud, K.; Morgan, E.M.; Oates, J.C.; Orozco, C.; et al. Rheumatology Informatics System for Effectiveness: A National Informatics-Enabled Registry for Quality Improvement. Arthritis Care Res (Hoboken) 2016, 68, 1866–1873. [Google Scholar] [CrossRef] [PubMed]
- Prot, V.; Aguilera, H.M.; Skallerud, B.; Persson, R.; Urheim, S. A method for non-invasive estimation of mitral valve annular regional strains. Comput Biol Med 2025, 187, 109773. [Google Scholar] [CrossRef] [PubMed]
- Eden, R.; Chukwudi, I.; Bain, C.; Barbieri, S.; Callaway, L.; de Jersey, S.; George, Y.; Gorse, A.D.; Lawley, M.; Marendy, P.; et al. A scoping review of the governance of federated learning in healthcare. NPJ Digit Med 2025, 8, 427. [Google Scholar] [CrossRef] [PubMed]
- Moons, K.G.M.; Damen, J.A.A.; Kaul, T.; Hooft, L.; Andaur Navarro, C.; Dhiman, P.; Beam, A.L.; Van Calster, B.; Celi, L.A.; Denaxas, S.; et al. PROBAST+AI: an updated quality, risk of bias, and applicability assessment tool for prediction models using regression or artificial intelligence methods. BMJ 2025, 388, e082505. [Google Scholar] [CrossRef]
- Collins, G.S.; Moons, K.G.M.; Dhiman, P.; Riley, R.D.; Beam, A.L.; Van Calster, B.; Ghassemi, M.; Liu, X.; Reitsma, J.B.; van Smeden, M.; et al. TRIPOD+AI statement: updated guidance for reporting clinical prediction models that use regression or machine learning methods. BMJ 2024, 385, e078378. [Google Scholar] [CrossRef]
- Ibrahim, H.; Liu, X.; Rivera, S.C.; Moher, D.; Chan, A.W.; Sydes, M.R.; Calvert, M.J.; Denniston, A.K. Reporting guidelines for clinical trials of artificial intelligence interventions: the SPIRIT-AI and CONSORT-AI guidelines. Trials 2021, 22, 11. [Google Scholar] [CrossRef]
- Ribeiro Junior, H.L.; Nepomuceno, F.; Pessoa, C.D.O. AI in clinical trials is missing from CONSORT and SPIRIT 2025 guidelines. Lancet 2025, 406, 25. [Google Scholar] [CrossRef]
- Hammam, N.; Evans, M.; Morgan, E.; Reimold, A.; Anastasiou, C.; Kay, J.L.; Yazdany, J.; Schmajuk, G. Treatment of Sarcoidosis in US Rheumatology Practices: Data From the American College of Rheumatology's Rheumatology Informatics System for Effectiveness (RISE) Registry. Arthritis Care Res (Hoboken) 2022, 74, 371–376. [Google Scholar] [CrossRef]
- Guthridge, J.M.; Wagner, C.A.; James, J.A. The promise of precision medicine in rheumatology. Nat Med 2022, 28, 1363–1371. [Google Scholar] [CrossRef]
- Birtane, M.; Yavuz, S.; Tastekin, N. Laboratory evaluation in rheumatic diseases. World J Methodol 2017, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Prat, L.; Nissen, M.J.; Lamacchia, C.; Bentow, C.; Cesana, L.; Roux-Lombard, P.; Gabay, C.; Mahler, M. Comparison of Serological Biomarkers in Rheumatoid Arthritis and Their Combination to Improve Diagnostic Performance. Front Immunol 2018, 9, 1113. [Google Scholar] [CrossRef] [PubMed]
- Steiner, G.; Toes, R.E.M. Autoantibodies in rheumatoid arthritis - rheumatoid factor, anticitrullinated protein antibodies and beyond. Curr Opin Rheumatol 2024, 36, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Hansson, M.; Mathsson-Alm, L.; Wijayatunga, P.; Verheul, M.K.; Trouw, L.A.; Holmdahl, R.; Ronnelid, J.; Klareskog, L.; Rantapaa-Dahlqvist, S. Rheumatoid factor isotypes in relation to antibodies against citrullinated peptides and carbamylated proteins before the onset of rheumatoid arthritis. Arthritis Res Ther 2016, 18, 43. [Google Scholar] [CrossRef]
- Perera, J.; Delrosso, C.A.; Nerviani, A.; Pitzalis, C. Clinical Phenotypes, Serological Biomarkers, and Synovial Features Defining Seropositive and Seronegative Rheumatoid Arthritis: A Literature Review. Cells 2024, 13. [Google Scholar] [CrossRef]
- Avouac, J.; Kay, J.; Choy, E. Personalised treatment of rheumatoid arthritis based on cytokine profiles and synovial tissue signatures: potentials and challenges. Semin Arthritis Rheum 2025, 73, 152740. [Google Scholar] [CrossRef]
- Rayner, F.; Hiu, S.; Melville, A.; Bigirumurame, T.; Anderson, A.; Dyke, B.; Kerrigan, S.; McGucken, A.; Prichard, J.; Shahrokhabadi, M.S.; et al. Clinical predictors of flare and drug-free remission in rheumatoid arthritis: preliminary results from the prospective BIO-FLARE experimental medicine study. BMJ Open 2025, 15, e092478. [Google Scholar] [CrossRef]
- Huang, X.; Luu, L.D.W.; Jia, N.; Zhu, J.; Fu, J.; Xiao, F.; Liu, C.; Li, S.; Shu, G.; Hou, J.; et al. Multi-Platform Omics Analysis Reveals Molecular Signatures for Pathogenesis and Activity of Systemic Lupus Erythematosus. Front Immunol 2022, 13, 833699. [Google Scholar] [CrossRef]
- Huang, H.; Sun, X.; Zhang, Q.; Liu, C.; Cao, X.; Zhang, D.; Wang, G.; Pu, C. Combined serum IFN-gamma and IL-22 levels as predictive biomarkers for hepatocellular carcinoma risk: A clinical investigation. Biomed Rep 2025, 23, 149. [Google Scholar] [CrossRef]
- Pisetsky, D.S. Pathogenesis of autoimmune disease. Nat Rev Nephrol 2023, 19, 509–524. [Google Scholar] [CrossRef]
- Kutsuna, Y.J.; Aibara, N.; Hashizume, J.; Kawarabayashi, S.; Tamai, M.; Miyata, J.; Yoshifuji, H.; Miyamoto, H.; Sato, K.; Kodama, Y.; et al. Identification of immune complex antigens that are detected prior to early rheumatoid arthritis symptoms and increase with disease progression: Comprehensive serum immune complexome analysis to identify candidate disease biomarkers in health checkup cohort study. Clin Immunol 2025, 281, 110591. [Google Scholar] [CrossRef] [PubMed]
- Lerga-Jaso, J.; Terpolovsky, A.; Novkovic, B.; Osama, A.; Manson, C.; Bohn, S.; De Marino, A.; Kunitomi, M.; Yazdi, P.G. Optimization of multi-ancestry polygenic risk score disease prediction models. Sci Rep 2025, 15, 17495. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.S.; Chen, Y.J.; Hsiung, C.N.; Mao, C.L.; Wei, C.Y.; Chen, I.C.; Kao, C.M.; Hsiao, T.H.; Huang, W.N.; Chen, Y.H.; et al. Polygenic risk scores of rheumatoid arthritis associated with seropositivity and bone erosions in a Taiwanese population. Sci Rep 2025, 15, 25700. [Google Scholar] [CrossRef] [PubMed]
- Honda, S.; Ikari, K.; Yano, K.; Terao, C.; Tanaka, E.; Harigai, M.; Kochi, Y. Association of Polygenic Risk Scores With Radiographic Progression in Patients With Rheumatoid Arthritis. Arthritis Rheumatol 2022, 74, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Liying Chen, O.D., Ting Fang, Mei Chen, Xvfeng Zhang, Ruichen Cong, Dingqi Lu, Runrun Zhang, Qun Jin, Xinchang Wang. Phenome-wide causal proteomics enhance systemic lupus erythematosus flare prediction: A study in Asian populations. medrxiv 2024. medrxiv.
- Paek, S.J.; Lee, H.S.; Lee, Y.J.; Bang, S.Y.; Kim, D.; Kang, B.K.; Park, D.J.; Joo, Y.B.; Kim, M.; Kim, H.; et al. Tracking clonal dynamics of CD8 T cells and immune dysregulation in progression of systemic lupus erythematosus with nephritis. Exp Mol Med 2025, 57, 1700–1710. [Google Scholar] [CrossRef]
- Zhang, J.; Zhuang, W.; Li, Y.; Deng, C.; Xuan, J.; Sun, Y.; He, Y. Bioinformatic analysis and experimental verification reveal expansion of monocyte subsets with an interferon signature in systemic lupus erythematosus patients. Arthritis Res Ther 2025, 27, 96. [Google Scholar] [CrossRef]
- Shen, M.; Duan, C.; Xie, C.; Wang, H.; Li, Z.; Li, B.; Wang, T. Identification of key interferon-stimulated genes for indicating the condition of patients with systemic lupus erythematosus. Front Immunol 2022, 13, 962393. [Google Scholar] [CrossRef]
- Haslam, D.E.; Li, J.; Dillon, S.T.; Gu, X.; Cao, Y.; Zeleznik, O.A.; Sasamoto, N.; Zhang, X.; Eliassen, A.H.; Liang, L.; et al. Stability and reproducibility of proteomic profiles in epidemiological studies: comparing the Olink and SOMAscan platforms. Proteomics 2022, 22, e2100170. [Google Scholar] [CrossRef]
- Shang, S.; Xia, J.; He, G.; Zheng, Y.; Zhang, J.; Lu, H.; Wang, H.; Li, W.; Li, Q.; Chen, X. Advances in precision medicine for lupus nephritis: biomarker- and AI-driven diagnosis and treatment response prediction and targeted therapies. EBioMedicine 2025, 117, 105785. [Google Scholar] [CrossRef]
- Cervia-Hasler, C.; Bruningk, S.C.; Hoch, T.; Fan, B.; Muzio, G.; Thompson, R.C.; Ceglarek, L.; Meledin, R.; Westermann, P.; Emmenegger, M.; et al. Persistent complement dysregulation with signs of thromboinflammation in active Long Covid. Science 2024, 383, eadg7942. [Google Scholar] [CrossRef]
- Duvvuri, B.; Lood, C. Cell-Free DNA as a Biomarker in Autoimmune Rheumatic Diseases. Front Immunol 2019, 10, 502. [Google Scholar] [CrossRef]
- Dihlmann, S.; Kaduk, C.; Passek, K.H.; Spieler, A.; Bockler, D.; Peters, A.S. Exploring circulating cell-free DNA as a biomarker and as an inducer of AIM2-inflammasome-mediated inflammation in patients with abdominal aortic aneurysm. Sci Rep 2025, 15, 20196. [Google Scholar] [CrossRef] [PubMed]
- Jackson Chornenki, N.L.; Coke, R.; Kwong, A.C.; Dwivedi, D.J.; Xu, M.K.; McDonald, E.; Marshall, J.C.; Fox-Robichaud, A.E.; Charbonney, E.; Liaw, P.C. Comparison of the source and prognostic utility of cfDNA in trauma and sepsis. Intensive Care Med Exp 2019, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Su, Y.; Liu, X.; Zhao, L.; Wu, Z.; Liu, Y.; Zhang, L. Cell-free DNA: a metabolic byproduct with diagnostic and prognostic potential in rheumatic disorders. Front Pharmacol 2025, 16, 1537934. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.; Giaglis, S.; Kyburz, D.; Daoudlarian, D.; Walker, U.A. Plasma mtDNA as a possible contributor to and biomarker of inflammation in rheumatoid arthritis. Arthritis Res Ther 2024, 26, 97. [Google Scholar] [CrossRef]
- Kerachian, M.A.; Azghandi, M.; Mozaffari-Jovin, S.; Thierry, A.R. Guidelines for pre-analytical conditions for assessing the methylation of circulating cell-free DNA. Clin Epigenetics 2021, 13, 193. [Google Scholar] [CrossRef]
- Peng, H.; Pan, M.; Zhou, Z.; Chen, C.; Xing, X.; Cheng, S.; Zhang, S.; Zheng, H.; Qian, K. The impact of preanalytical variables on the analysis of cell-free DNA from blood and urine samples. Front Cell Dev Biol 2024, 12, 1385041. [Google Scholar] [CrossRef]
- Sathyanarayana, S.H.; Spracklin, S.B.; Deharvengt, S.J.; Green, D.C.; Instasi, M.D.; Gallagher, T.L.; Shah, P.S.; Tsongalis, G.J. Standardized Workflow and Analytical Validation of Cell-Free DNA Extraction for Liquid Biopsy Using a Magnetic Bead-Based Cartridge System. Cells 2025, 14. [Google Scholar] [CrossRef]
- Qi, T.; Pan, M.; Shi, H.; Wang, L.; Bai, Y.; Ge, Q. Cell-Free DNA Fragmentomics: The Novel Promising Biomarker. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Fresneda Alarcon, M.; McLaren, Z.; Wright, H.L. Neutrophils in the Pathogenesis of Rheumatoid Arthritis and Systemic Lupus Erythematosus: Same Foe Different M.O. Front Immunol 2021, 12, 649693. [Google Scholar] [CrossRef]
- Huang, Y.; Xue, Q.; Chang, J.; Wang, Y.; Cheng, C.; Xu, S.; Wang, X.; Miao, C. M6A methylation modification in autoimmune diseases, a promising treatment strategy based on epigenetics. Arthritis Res Ther 2023, 25, 189. [Google Scholar] [CrossRef]
- Wu, J.; Deng, L.J.; Xia, Y.R.; Leng, R.X.; Fan, Y.G.; Pan, H.F.; Ye, D.Q. Involvement of N6-methyladenosine modifications of long noncoding RNAs in systemic lupus erythematosus. Mol Immunol 2022, 143, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Liu, J.; Li, S.; Xu, P. Analysis of m6A regulators related immune characteristics in ankylosing spondylitis by integrated bioinformatics and computational strategies. Sci Rep 2024, 14, 2724. [Google Scholar] [CrossRef]
- Cheng, L.; Li, H.; Zhan, H.; Liu, Y.; Li, X.; Huang, Y.; Wang, L.; Zhang, F.; Li, Y. Alterations of m6A RNA methylation regulators contribute to autophagy and immune infiltration in primary Sjogren's syndrome. Front Immunol 2022, 13, 949206. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, Y.; Liu, X. Rheumatoid arthritis: pathogenesis and therapeutic advances. MedComm (2020) 2024, 5, e509. [Google Scholar] [CrossRef] [PubMed]
- Wardowska, A. m6A RNA Methylation in Systemic Autoimmune Diseases-A New Target for Epigenetic-Based Therapy? Pharmaceuticals (Basel) 2021, 14. [Google Scholar] [CrossRef]
- Pengcheng Xu, J.C. , Yulin Gao, Ziqi Rong. MIRACLE: Multi-task Learning based Interpretable Regulation of Autoimmune Diseases through Common Latent Epigenetics. arxiv 2023, 06. [Google Scholar]
- Kim, S.; Zhang, L.; Qin, Y.; Bohn, R.I.C.; Park, H.J. Pathway information on methylation analysis using deep neural network (PROMINENT): An interpretable deep learning method with pathway prior for phenotype prediction using gene-level DNA methylation. Artif Intell Med 2025, 170, 103236. [Google Scholar] [CrossRef]
- Lee, K.; Niku, S.; Koo, S.J.; Belezzuoli, E.; Guma, M. Molecular imaging for evaluation of synovitis associated with osteoarthritis: a narrative review. Arthritis Res Ther 2024, 26, 25. [Google Scholar] [CrossRef]
- Boeren, A.M.P.; Oei, E.H.G.; van der Helm-van Mil, A.H.M. The value of MRI for detecting subclinical joint inflammation in clinically suspect arthralgia. RMD Open 2022, 8. [Google Scholar] [CrossRef]
- So, H.; Cheng, I.; Tam, L.S. The Role of Imaging in Predicting the Development of Rheumatoid Arthritis. Rheumatol Immunol Res 2021, 2, 27–33. [Google Scholar] [CrossRef]
- Ogdie, A.; Coates, L.C.; Mease, P. Measuring Outcomes in Psoriatic Arthritis. Arthritis Care Res (Hoboken) 2020, 72 Suppl 10, 82–109. [Google Scholar] [CrossRef] [PubMed]
- Frenken, M.; Schleich, C.; Brinks, R.; Abrar, D.B.; Goertz, C.; Schneider, M.; Ostendorf, B.; Sewerin, P. The value of the simplified RAMRIS-5 in early RA patients under methotrexate therapy using high-field MRI. Arthritis Res Ther 2019, 21, 21. [Google Scholar] [CrossRef] [PubMed]
- Schleich, C.; Buchbender, C.; Sewerin, P.; Miese, F.; Aissa, J.; Brinks, R.; Schneider, M.; Antoch, G.; Ostendorf, B. Evaluation of a simplified version of the Rheumatoid Arthritis Magnetic Resonance Imaging Score (RAMRIS) comprising 5 joints (RAMRIS5). Clin Exp Rheumatol 2015, 33, 209–215. [Google Scholar]
- Ben-Eltriki, M.; Rafiq, A.; Paul, A.; Prabhu, D.; Afolabi, M.O.S.; Baslhaw, R.; Neilson, C.J.; Driedger, M.; Mahmud, S.M.; Lacaze-Masmonteil, T.; et al. Adaptive designs in clinical trials: a systematic review-part I. BMC Med Res Methodol 2024, 24, 229. [Google Scholar] [CrossRef]
- Kaizer, A.M.; Belli, H.M.; Ma, Z.; Nicklawsky, A.G.; Roberts, S.C.; Wild, J.; Wogu, A.F.; Xiao, M.; Sabo, R.T. Recent innovations in adaptive trial designs: A review of design opportunities in translational research. J Clin Transl Sci 2023, 7, e125. [Google Scholar] [CrossRef]
- Noversa de Sousa, R.; Tascilar, K.; Corte, G.; Atzinger, A.; Minopoulou, I.; Ohrndorf, S.; Waldner, M.; Schmidkonz, C.; Kuwert, T.; Knieling, F.; et al. Metabolic and molecular imaging in inflammatory arthritis. RMD Open 2024, 10. [Google Scholar] [CrossRef]
- MacKay, J.W.; Watkins, L.; Gold, G.; Kogan, F. [(18)F]NaF PET-MRI provides direct in-vivo evidence of the association between bone metabolic activity and adjacent synovitis in knee osteoarthritis: a cross-sectional study. Osteoarthritis Cartilage 2021, 29, 1155–1162. [Google Scholar] [CrossRef]
- MacRitchie, N.; Frleta-Gilchrist, M.; Sugiyama, A.; Lawton, T.; McInnes, I.B.; Maffia, P. Molecular imaging of inflammation - Current and emerging technologies for diagnosis and treatment. Pharmacol Ther 2020, 211, 107550. [Google Scholar] [CrossRef]
- Chandrupatla, D.; Molthoff, C.F.M.; Lammertsma, A.A.; van der Laken, C.J.; Jansen, G. The folate receptor beta as a macrophage-mediated imaging and therapeutic target in rheumatoid arthritis. Drug Deliv Transl Res 2019, 9, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Novruzov, E.; Schmitt, D.; Cardinale, J.; Watabe, T.; Choyke, P.L.; Alavi, A.; Haberkorn, U.; Giesel, F.L. Clinical applications of fibroblast activation protein inhibitor positron emission tomography (FAPI-PET). Npj Imaging 2024, 2, 48. [Google Scholar] [CrossRef] [PubMed]
- Kastelik-Hryniewiecka, A.; Jewula, P.; Bakalorz, K.; Kramer-Marek, G.; Kuznik, N. Targeted PET/MRI Imaging Super Probes: A Critical Review of Opportunities and Challenges. Int J Nanomedicine 2021, 16, 8465–8483. [Google Scholar] [CrossRef] [PubMed]
- Hamy, V.; Llop, C.; Yee, C.W.; Garcia-Gancedo, L.; Maxwell, A.; Chen, W.H.; Tomlinson, R.; Bobbili, P.; Bendelac, J.; Landry, J.; et al. Patient-centric assessment of rheumatoid arthritis using a smartwatch and bespoke mobile app in a clinical setting. Sci Rep 2023, 13, 18311. [Google Scholar] [CrossRef]
- Reed, M.; Rampono, B.; Turner, W.; Harsanyi, A.; Lim, A.; Paramalingam, S.; Massasso, D.; Thakkar, V.; Mundae, M.; Rampono, E. A multicentre validation study of a smartphone application to screen hand arthritis. BMC Musculoskelet Disord 2022, 23, 433. [Google Scholar] [CrossRef]
- Venerito, V.; Manigold, T.; Capodiferro, M.; Markham, D.; Blanchard, M.; Iannone, F.; Hugle, T. Single-camera motion capture of finger joint mobility as a digital biomarker for disease activity in rheumatoid arthritis. Rheumatol Adv Pract 2025, 9, rkae143. [Google Scholar] [CrossRef]
- Guo, L.; Chang, R.; Wang, J.; Narayanan, A.; Qian, P.; Leong, M.C.; Kundu, P.P.; Senthilkumar, S.; Garlapati, S.C.; Yong, E.C.K.; et al. Artificial intelligence-enhanced 3D gait analysis with a single consumer-grade camera. J Biomech 2025, 187, 112738. [Google Scholar] [CrossRef]
- Hadjileontiadis, L.J.; Charisis, V.; Hadjidimitriou, S.; Dias, S.B.; Apostolidis, G.; Dimaridis, G.; Kitsas, I.; Karlas, A.; Fasoula, N.A.; Levi-Schaffer, F.; et al. European advances in digital rheumatology: explainable insights and personalized digital health tools for psoriatic arthritis. EClinicalMedicine 2025, 84, 103243. [Google Scholar] [CrossRef]
- Santosa, A.; Li, J.W.; Tan, T.C. Digital Health for Equitable Rheumatic Care: Integrating Real-World Experiences to Guide Policy Pathways. Healthcare (Basel) 2025, 13. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, Y.; Chen, Y.; Luo, D.; Xu, K.; Zhang, L. Artificial intelligence for predicting treatment responses in autoimmune rheumatic diseases: advancements, challenges, and future perspectives. Front Immunol 2024, 15, 1477130. [Google Scholar] [CrossRef]
- Zhao, J.; Li, L.; Li, J.; Zhang, L. Application of artificial intelligence in rheumatic disease: a bibliometric analysis. Clin Exp Med 2024, 24, 196. [Google Scholar] [CrossRef]
- Nelson, A.E.; Arbeeva, L. Narrative Review of Machine Learning in Rheumatic and Musculoskeletal Diseases for Clinicians and Researchers: Biases, Goals, and Future Directions. J Rheumatol 2022, 49, 1191–1200. [Google Scholar] [CrossRef]
- Knevel, R.; Liao, K.P. From real-world electronic health record data to real-world results using artificial intelligence. Ann Rheum Dis 2023, 82, 306–311. [Google Scholar] [CrossRef]
- Tonner, C.; Schmajuk, G.; Yazdany, J. A new era of quality measurement in rheumatology: electronic clinical quality measures and national registries. Curr Opin Rheumatol 2017, 29, 131–137. [Google Scholar] [CrossRef]
- Francisco, M.; Johansson, T.; Kazi, S. Overview of the American College of Rheumatology's Electronic Health Record-Enabled Registry: The Rheumatology Informatics System for Effectiveness. Clin Exp Rheumatol 2016, 34, S102–S104. [Google Scholar] [PubMed]
- Oatis, C.A.; Konnyu, K.J.; Franklin, P.D. Generating consistent longitudinal real-world data to support research: lessons from physical therapists. ACR Open Rheumatol 2022, 4, 771–774. [Google Scholar] [CrossRef] [PubMed]
- Zara Izadi, G.S., Milena Gianfrancesco, Meera Subash, Michael Evans, Laura Trupin, Jinoos Yazdany. Rheumatology Informatics System for Effectiveness (RISE) Practices See Significant Gains in Rheumatoid Arthritis Quality Measures. Arthritis Care Res (Hoboken) 2021, 74(2), 219–228.
- Tabatabaei Hosseini, S.A.; Kazemzadeh, R.; Foster, B.J.; Arpali, E.; Susal, C. New Tools for Data Harmonization and Their Potential Applications in Organ Transplantation. Transplantation 2024, 108, 2306–2317. [Google Scholar] [CrossRef] [PubMed]
- Carbonaro, A.; Giorgetti, L.; Ridolfi, L.; Pasolini, R.; Pagliarani, A.; Cavallucci, M.; Andalo, A.; Gaudio, L.D.; De Angelis, P.; Vespignani, R.; et al. From raw data to research-ready: A FHIR-based transformation pipeline in a real-world oncology setting. Comput Biol Med 2025, 197, 111051. [Google Scholar] [CrossRef]
- Omar, M.; Naffaa, M.E.; Glicksberg, B.S.; Reuveni, H.; Nadkarni, G.N.; Klang, E. Advancing rheumatology with natural language processing: insights and prospects from a systematic review. Rheumatol Adv Pract 2024, 8, rkae120. [Google Scholar] [CrossRef]
- Benavent, D.; Madrid-Garcia, A. Large language models and rheumatology: are we there yet? Rheumatol Adv Pract 2025, 9, rkae119. [Google Scholar] [CrossRef]
- Humbert-Droz, M.; Izadi, Z.; Schmajuk, G.; Gianfrancesco, M.; Baker, M.C.; Yazdany, J.; Tamang, S. Development of a Natural Language Processing System for Extracting Rheumatoid Arthritis Outcomes From Clinical Notes Using the National Rheumatology Informatics System for Effectiveness Registry. Arthritis Care Res (Hoboken) 2023, 75, 608–615. [Google Scholar] [CrossRef]
- Maghsoudi, A.; Sada, Y.H.; Nowakowski, S.; Guffey, D.; Zhu, H.; Yarlagadda, S.R.; Li, A.; Razjouyan, J. A Multi-Institutional Natural Language Processing Pipeline to Extract Performance Status From Electronic Health Records. Cancer Control 2024, 31, 10732748241279518. [Google Scholar] [CrossRef]
- Zhang, F.; Kreuter, D.; Chen, Y.; Dittmer, S.; Tull, S.; Shadbahr, T.; BloodCounts, c.; Preller, J.; Rudd, J.H.F.; Aston, J.A.D.; et al. Recent methodological advances in federated learning for healthcare. Patterns (N Y) 2024, 5, 101006. [Google Scholar] [CrossRef] [PubMed]
- Austin, J.A.; Lobo, E.H.; Samadbeik, M.; Engstrom, T.; Philip, R.; Pole, J.D.; Sullivan, C.M. Decades in the Making: The Evolution of Digital Health Research Infrastructure Through Synthetic Data, Common Data Models, and Federated Learning. J Med Internet Res 2024, 26, e58637. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Bressem, K.; Adams, L.; Poddubnyy, D.; Proft, F. AI for imaging evaluation in rheumatology: applications of radiomics and computer vision-current status, future prospects and potential challenges. Rheumatol Adv Pract 2025, 9, rkae147. [Google Scholar] [CrossRef] [PubMed]
- Stoel, B. Use of artificial intelligence in imaging in rheumatology - current status and future perspectives. RMD Open 2020, 6. [Google Scholar] [CrossRef]
- Bird, A.; Oakden-Rayner, L.; McMaster, C.; Smith, L.A.; Zeng, M.; Wechalekar, M.D.; Ray, S.; Proudman, S.; Palmer, L.J. Artificial intelligence and the future of radiographic scoring in rheumatoid arthritis: a viewpoint. Arthritis Res Ther 2022, 24, 268. [Google Scholar] [CrossRef]
- Zhang, N.; Yang, S.; Zwagemaker, A.F.; Huo, A.; Li, Y.J.; Zhou, F.; Hilliard, P.; Squire, S.; Bouskill, V.; Mohanta, A.; et al. A semiquantitative color Doppler ultrasound scoring system for evaluation of synovitis in joints of patients with blood-induced arthropathy. Insights Imaging 2021, 12, 132. [Google Scholar] [CrossRef]
- Zwanenburg, A.; Vallieres, M.; Abdalah, M.A.; Aerts, H.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-based Phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef]
- Oettl, F.C.; Zsidai, B.; Oeding, J.F.; Hirschmann, M.T.; Feldt, R.; Fendrich, D.; Kraeutler, M.J.; Winkler, P.W.; Szaro, P.; Samuelsson, K.; et al. Artificial intelligence-assisted analysis of musculoskeletal imaging-A narrative review of the current state of machine learning models. Knee Surg Sports Traumatol Arthrosc 2025, 33, 3032–3038. [Google Scholar] [CrossRef]
- Salvi, M.; Seoni, S.; Campagner, A.; Gertych, A.; Acharya, U.R.; Molinari, F.; Cabitza, F. Explainability and uncertainty: Two sides of the same coin for enhancing the interpretability of deep learning models in healthcare. Int J Med Inform 2025, 197, 105846. [Google Scholar] [CrossRef]
- Chen, M.; Wang, Y.; Wang, Q.; Shi, J.; Wang, H.; Ye, Z.; Xue, P.; Qiao, Y. Impact of human and artificial intelligence collaboration on workload reduction in medical image interpretation. NPJ Digit Med 2024, 7, 349. [Google Scholar] [CrossRef]
- Guermazi, A.; Roemer, F.W.; Crema, M.D.; Jarraya, M.; Mobasheri, A.; Hayashi, D. Strategic application of imaging in DMOAD clinical trials: focus on eligibility, drug delivery, and semiquantitative assessment of structural progression. Ther Adv Musculoskelet Dis 2023, 15, 1759720X231165558. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, M.M.A.; Westgeest, A.A.A.; Schwarting, A.; Jacobs, J.W.G.; Heller, C.; van Laar, J.M.; Lafeber, F.; Tekstra, J.; Triantafyllias, K.; Welsing, P.M.J. Development and Validation of Rheumatoid Arthritis Disease Activity Indices Including HandScan (Optical Spectral Transmission) Scores. Arthritis Care Res (Hoboken) 2022, 74, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- Park, D.J. Importance of Time-integrated Cumulative Parameters for Radiographic Progression Prediction of Rheumatoid Arthritis. J Rheum Dis 2022, 29, 129–131. [Google Scholar] [CrossRef] [PubMed]
- Gumber, L.; Rayner, F.; Bigirumurame, T.; Dyke, B.; Melville, A.; Kerrigan, S.; McGucken, A.; Naamane, N.; Prichard, J.; Buckley, C.D.; et al. Patient-reported outcomes as early warning signs of flare following drug cessation in rheumatoid arthritis. RMD Open 2025, 11. [Google Scholar] [CrossRef]
- Gandrup, J.; Selby, D.A.; van der Veer, S.N.; McBeth, J.; Dixon, W.G. Using patient-reported data from a smartphone app to capture and characterize real-time patient-reported flares in rheumatoid arthritis. Rheumatol Adv Pract 2022, 6, rkac021. [Google Scholar] [CrossRef]
- Momtazmanesh, S.; Nowroozi, A.; Rezaei, N. Artificial Intelligence in Rheumatoid Arthritis: Current Status and Future Perspectives: A State-of-the-Art Review. Rheumatol Ther 2022, 9, 1249–1304. [Google Scholar] [CrossRef]
- Gul, H.; Di Matteo, A.; Anioke, I.; Shuweidhi, F.; Mankia, K.; Ponchel, F.; Emery, P. Predicting Flare in Patients With Rheumatoid Arthritis in Biologic Induced Remission, on Tapering, and on Stable Therapy. ACR Open Rheumatol 2024, 6, 294–303. [Google Scholar] [CrossRef]
- Patharkar, A.; Cai, F.; Al-Hindawi, F.; Wu, T. Predictive modeling of biomedical temporal data in healthcare applications: review and future directions. Front Physiol 2024, 15, 1386760. [Google Scholar] [CrossRef]
- Richardson, S.; Lawrence, K.; Schoenthaler, A.M.; Mann, D. A framework for digital health equity. NPJ Digit Med 2022, 5, 119. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.E.; Dorn, C.; Park, D.J.; Matheny, M.E. Emerging algorithmic bias: fairness drift as the next dimension of model maintenance and sustainability. J Am Med Inform Assoc 2025, 32, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Temmoku, J.; Migita, K.; Yoshida, S.; Matsumoto, H.; Fujita, Y.; Matsuoka, N.; Yashiro-Furuya, M.; Asano, T.; Sato, S.; Suzuki, E.; et al. Real-world comparative effectiveness of bDMARDs and JAK inhibitors in elderly patients with rheumatoid arthritis. Medicine (Baltimore) 2022, 101, e31161. [Google Scholar] [CrossRef] [PubMed]
- Eberhard, A.; Di Giuseppe, D.; Askling, J.; Bergman, S.; Bower, H.; Chatzidionysiou, K.; Forsblad-d'Elia, H.; Kastbom, A.; Olofsson, T.; Frisell, T.; et al. Effectiveness of JAK Inhibitors Compared With Biologic Disease-Modifying Antirheumatic Drugs on Pain Reduction in Rheumatoid Arthritis: Results From a Nationwide Swedish Cohort Study. Arthritis Rheumatol 2025, 77, 253–262. [Google Scholar] [CrossRef]
- Efthimiou, O.; Seo, M.; Chalkou, K.; Debray, T.; Egger, M.; Salanti, G. Developing clinical prediction models: a step-by-step guide. BMJ 2024, 386, e078276. [Google Scholar] [CrossRef]
- Liu, D.; Yu, G.; Yuan, N.; Nie, D. The efficacy and safety of biologic or targeted synthetic DMARDs in rheumatoid arthritis treatment: one year of review 2024. Allergol Immunopathol (Madr) 2025, 53, 140–162. [Google Scholar] [CrossRef]
- Favalli, E.G.; Maioli, G.; Caporali, R. Biologics or Janus Kinase Inhibitors in Rheumatoid Arthritis Patients Who are Insufficient Responders to Conventional Anti-Rheumatic Drugs. Drugs 2024, 84, 877–894. [Google Scholar] [CrossRef]
- Wang, S.S.; Lewis, M.J.; Pitzalis, C. DNA Methylation Signatures of Response to Conventional Synthetic and Biologic Disease-Modifying Antirheumatic Drugs (DMARDs) in Rheumatoid Arthritis. Biomedicines 2023, 11. [Google Scholar] [CrossRef]
- Bhasin, S.; Cheung, P.P. The Role of Power Doppler Ultrasonography as Disease Activity Marker in Rheumatoid Arthritis. Dis Markers 2015, 2015, 325909. [Google Scholar] [CrossRef]
- Subash, M.; Liu, L.H.; DeQuattro, K.; Choden, S.; Jacobsohn, L.; Katz, P.; Bajaj, P.; Barton, J.L.; Bartels, C.; Bermas, B.; et al. The Development of the Rheumatology Informatics System for Effectiveness Learning Collaborative for Improving Patient-Reported Outcome Collection and Patient-Centered Communication in Adult Rheumatology. ACR Open Rheumatol 2021, 3, 690–698. [Google Scholar] [CrossRef]
- Abernethy, A.; Adams, L.; Barrett, M.; Bechtel, C.; Brennan, P.; Butte, A.; Faulkner, J.; Fontaine, E.; Friedhoff, S.; Halamka, J.; et al. The Promise of Digital Health: Then, Now, and the Future. NAM Perspect 2022, 2022. [Google Scholar] [CrossRef] [PubMed]
- Subasri, V.; Krishnan, A.; Kore, A.; Dhalla, A.; Pandya, D.; Wang, B.; Malkin, D.; Razak, F.; Verma, A.A.; Goldenberg, A.; et al. Detecting and Remediating Harmful Data Shifts for the Responsible Deployment of Clinical AI Models. JAMA Netw Open 2025, 8, e2513685. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.J.; Wang, J.J.; Williamson, D.F.K.; Chen, T.Y.; Lipkova, J.; Lu, M.Y.; Sahai, S.; Mahmood, F. Algorithmic fairness in artificial intelligence for medicine and healthcare. Nat Biomed Eng 2023, 7, 719–742. [Google Scholar] [CrossRef] [PubMed]
- Kore, A.; Abbasi Bavil, E.; Subasri, V.; Abdalla, M.; Fine, B.; Dolatabadi, E.; Abdalla, M. Empirical data drift detection experiments on real-world medical imaging data. Nat Commun 2024, 15, 1887. [Google Scholar] [CrossRef]
- Liu, L.H.; Choden, S.; Yazdany, J. Quality improvement initiatives in rheumatology: an integrative review of the last 5 years. Curr Opin Rheumatol 2019, 31, 98–108. [Google Scholar] [CrossRef]
- Liu, T.; Gu, Y.; Chen, H.; Zhang, Y.; Zheng, L.; Huang, X.; Xu, Y.; Wen, C.; Chen, M.; Lin, J.; et al. A foundational triage system for improving accuracy in moderate acuity level emergency classifications. Commun Med (Lond) 2025, 5, 322. [Google Scholar] [CrossRef]
- Portela, A.; Banga, J.R.; Matabuena, M. Conformal prediction for uncertainty quantification in dynamic biological systems. PLoS Comput Biol 2025, 21, e1013098. [Google Scholar] [CrossRef]
- Chen, D.; He, E.; Pace, K.; Chekay, M.; Raman, S. Concordance with SPIRIT-AI guidelines in reporting of randomized controlled trial protocols investigating artificial intelligence in oncology: a systematic review. Oncologist 2025, 30. [Google Scholar] [CrossRef]
- Bodnari, A.; Travis, J. Scaling enterprise AI in healthcare: the role of governance in risk mitigation frameworks. NPJ Digit Med 2025, 8, 272. [Google Scholar] [CrossRef]
- Palaniappan, K.; Lin, E.Y.T.; Vogel, S. Global Regulatory Frameworks for the Use of Artificial Intelligence (AI) in the Healthcare Services Sector. Healthcare (Basel) 2024, 12. [Google Scholar] [CrossRef]
- Martin, A.R.; Kanai, M.; Kamatani, Y.; Okada, Y.; Neale, B.M.; Daly, M.J. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat Genet 2019, 51, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Cope, A.P.; Jasenecova, M.; Vasconcelos, J.C.; Filer, A.; Raza, K.; Qureshi, S.; D'Agostino, M.A.; McInnes, I.B.; Isaacs, J.D.; Pratt, A.G.; et al. Abatacept in individuals at high risk of rheumatoid arthritis (APIPPRA): a randomised, double-blind, multicentre, parallel, placebo-controlled, phase 2b clinical trial. Lancet 2024, 403, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Rech, J.; Tascilar, K.; Hagen, M.; Kleyer, A.; Manger, B.; Schoenau, V.; Hueber, A.J.; Kleinert, S.; Baraliakos, X.; Braun, J.; et al. Abatacept inhibits inflammation and onset of rheumatoid arthritis in individuals at high risk (ARIAA): a randomised, international, multicentre, double-blind, placebo-controlled trial. Lancet 2024, 403, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Zhao, J.; Li, M.; Zeng, X. New insights into the pathogenesis and management of rheumatoid arthritis. Chronic Dis Transl Med 2022, 8, 256–263. [Google Scholar] [CrossRef]
- McDonald, S.M.; Felfeliyan, B.; Hassan, A.; Kupper, J.C.; El-Hajj, R.; Wichuk, S.; Aneja, A.; Kwok, C.; Zhang, C.X.Y.; Jans, L.; et al. Evaluating potential for AI automation of quantitative and semi-quantitative MRI scoring in arthritis, especially at the knee: a systematic literature review. Skeletal Radiol 2025. [Google Scholar] [CrossRef]
- Mao, Y.; Imahori, K.; Fang, W.; Sugimori, H.; Kiuch, S.; Sutherland, K.; Kamishima, T. Artificial Intelligence Quantification of Enhanced Synovium Throughout the Entire Hand in Rheumatoid Arthritis on Dynamic Contrast-Enhanced MRI. J Magn Reson Imaging 2025, 61, 771–783. [Google Scholar] [CrossRef]
- Nicoara, A.I.; Sas, L.M.; Bita, C.E.; Dinescu, S.C.; Vreju, F.A. Implementation of artificial intelligence models in magnetic resonance imaging with focus on diagnosis of rheumatoid arthritis and axial spondyloarthritis: narrative review. Front Med (Lausanne) 2023, 10, 1280266. [Google Scholar] [CrossRef]
- Schlereth, M.; Mutlu, M.Y.; Utz, J.; Bayat, S.; Heimann, T.; Qiu, J.; Ehring, C.; Liu, C.; Uder, M.; Kleyer, A.; et al. Deep learning-based classification of erosion, synovitis and osteitis in hand MRI of patients with inflammatory arthritis. RMD Open 2024, 10. [Google Scholar] [CrossRef]
- Kumar, R.; Sporn, K.; Prabhakar, V.; Alnemri, A.; Khanna, A.; Paladugu, P.; Gowda, C.; Clarkson, L.; Zaman, N.; Tavakkoli, A. Computational and Imaging Approaches for Precision Characterization of Bone, Cartilage, and Synovial Biomolecules. J Pers Med 2025, 15. [Google Scholar] [CrossRef]
- Currie, G.; Rohren, E. The deep radiomic analytics pipeline. Vet Radiol Ultrasound 2022, 63 Suppl 1, 889–896. [Google Scholar] [CrossRef]
- Ou, Y.; Ambalathankandy, P.; Furuya, R.; Kawada, S.; Zeng, T.; An, Y.; Kamishima, T.; Tamura, K.; Ikebe, M. A Sub-Pixel Accurate Quantification of Joint Space Narrowing Progression in Rheumatoid Arthritis. IEEE J Biomed Health Inform 2023, 27, 53–64. [Google Scholar] [CrossRef]
- Ichikawa, S.; Kamishima, T.; Sutherland, K.; Okubo, T.; Katayama, K. Radiographic quantifications of joint space narrowing progression by computer-based approach using temporal subtraction in rheumatoid wrist. Br J Radiol 2016, 89, 20150403. [Google Scholar] [CrossRef]
- Ou, J.; Zhang, J.; Alswadeh, M.; Zhu, Z.; Tang, J.; Sang, H.; Lu, K. Advancing osteoarthritis research: the role of AI in clinical, imaging and omics fields. Bone Res 2025, 13, 48. [Google Scholar] [CrossRef]
- Woelfle, T.; Bourguignon, L.; Lorscheider, J.; Kappos, L.; Naegelin, Y.; Jutzeler, C.R. Wearable Sensor Technologies to Assess Motor Functions in People With Multiple Sclerosis: Systematic Scoping Review and Perspective. J Med Internet Res 2023, 25, e44428. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kang, S.; Eun, Y.; Won, H.H.; Kim, H.; Lee, J.; Koh, E.M.; Cha, H.S. Machine learning-based prediction model for responses of bDMARDs in patients with rheumatoid arthritis and ankylosing spondylitis. Arthritis Res Ther 2021, 23, 254. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Concepcion, A.N.; Vianen, M.; Marijnissen, A.C.A.; Lafeber, F.; Radstake, T.; Pandit, A. Multiomics and Machine Learning Accurately Predict Clinical Response to Adalimumab and Etanercept Therapy in Patients With Rheumatoid Arthritis. Arthritis Rheumatol 2021, 73, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Benavent, D.; Carmona, L.; Garcia Llorente, J.F.; Montoro, M.; Ramirez, S.; Oton, T.; Loza, E.; Gomez-Centeno, A. Artificial intelligence to predict treatment response in rheumatoid arthritis and spondyloarthritis: a scoping review. Rheumatol Int 2025, 45, 91. [Google Scholar] [CrossRef]
- Hameed, M.; Exarchou, S.; Eberhard, A.; Sharma, A.; Bergstrom, U.; Cagnotto, G.; Einarsson, J.T.; Turesson, C. Predictors at diagnosis for start of biologic disease-modifying antirheumatic drugs in patients with early rheumatoid arthritis: a cohort study. BMJ Open 2024, 14, e076131. [Google Scholar] [CrossRef]
- Postal, M.; Vivaldo, J.F.; Fernandez-Ruiz, R.; Paredes, J.L.; Appenzeller, S.; Niewold, T.B. Type I interferon in the pathogenesis of systemic lupus erythematosus. Curr Opin Immunol 2020, 67, 87–94. [Google Scholar] [CrossRef]
- Miyachi, K.; Iwamoto, T.; Kojima, S.; Ida, T.; Suzuki, J.; Yamamoto, T.; Mimura, N.; Sugiyama, T.; Tanaka, S.; Furuta, S.; et al. Relationship of systemic type I interferon activity with clinical phenotypes, disease activity, and damage accrual in systemic lupus erythematosus in treatment-naive patients: a retrospective longitudinal analysis. Arthritis Res Ther 2023, 25, 26. [Google Scholar] [CrossRef]
- Kaan, E.D.; Brunekreef, T.E.; Drylewicz, J.; van den Hoogen, L.L.; van der Linden, M.; Leavis, H.L.; van Laar, J.M.; van der Vlist, M.; Otten, H.G.; Limper, M. Association of autoantibodies with the IFN signature and NETosis in patients with systemic lupus erythematosus. J Transl Autoimmun 2024, 9, 100246. [Google Scholar] [CrossRef]
- Felten, R.; Scher, F.; Sagez, F.; Chasset, F.; Arnaud, L. Spotlight on anifrolumab and its potential for the treatment of moderate-to-severe systemic lupus erythematosus: evidence to date. Drug Des Devel Ther 2019, 13, 1535–1543. [Google Scholar] [CrossRef]
- Furie, R.; Khamashta, M.; Merrill, J.T.; Werth, V.P.; Kalunian, K.; Brohawn, P.; Illei, G.G.; Drappa, J.; Wang, L.; Yoo, S.; et al. Anifrolumab, an Anti-Interferon-alpha Receptor Monoclonal Antibody, in Moderate-to-Severe Systemic Lupus Erythematosus. Arthritis Rheumatol 2017, 69, 376–386. [Google Scholar] [CrossRef]
- Baker, T.; Sharifian, H.; Newcombe, P.J.; Gavin, P.G.; Lazarus, M.N.; Ramaswamy, M.; White, W.I.; Ferrari, N.; Muthas, D.; Tummala, R.; et al. Type I interferon blockade with anifrolumab in patients with systemic lupus erythematosus modulates key immunopathological pathways in a gene expression and proteomic analysis of two phase 3 trials. Ann Rheum Dis 2024, 83, 1018–1027. [Google Scholar] [CrossRef]
- Cleanthous, S.; Strzok, S.; Haier, B.; Cano, S.; Morel, T. The Patient Experience of Fatigue in Systemic Lupus Erythematosus: A Conceptual Model. Rheumatol Ther 2022, 9, 95–108. [Google Scholar] [CrossRef]
- Mai, L.; Asaduzzaman, A.; Noamani, B.; Fortin, P.R.; Gladman, D.D.; Touma, Z.; Urowitz, M.B.; Wither, J. The baseline interferon signature predicts disease severity over the subsequent 5 years in systemic lupus erythematosus. Arthritis Res Ther 2021, 23, 29. [Google Scholar] [CrossRef]
- Ruscitti, P.; Allanore, Y.; Baldini, C.; Barilaro, G.; Bartoloni Bocci, E.; Bearzi, P.; Bellis, E.; Berardicurti, O.; Biaggi, A.; Bombardieri, M.; et al. Tailoring the treatment of inflammatory rheumatic diseases by a better stratification and characterization of the clinical patient heterogeneity. Findings from a systematic literature review and experts' consensus. Autoimmun Rev 2024, 23, 103581. [Google Scholar] [CrossRef]
- Oliveira, J.J.; Karrar, S.; Rainbow, D.B.; Pinder, C.L.; Clarke, P.; Rubio Garcia, A.; Al-Assar, O.; Burling, K.; Morris, S.; Stratton, R.; et al. The plasma biomarker soluble SIGLEC-1 is associated with the type I interferon transcriptional signature, ethnic background and renal disease in systemic lupus erythematosus. Arthritis Res Ther 2018, 20, 152. [Google Scholar] [CrossRef] [PubMed]
- Perng, Y.C.; Lenschow, D.J. ISG15 in antiviral immunity and beyond. Nat Rev Microbiol 2018, 16, 423–439. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, S.; Wang, J. IFN-gamma, should not be ignored in SLE. Front Immunol 2022, 13, 954706. [Google Scholar] [CrossRef]
- Gomez-Banuelos, E.; Goldman, D.W.; Andrade, V.; Darrah, E.; Petri, M.; Andrade, F. Uncoupling interferons and the interferon signature explains clinical and transcriptional subsets in SLE. Cell Rep Med 2024, 5, 101569. [Google Scholar] [CrossRef]
- Jupe, E.R.; Lushington, G.H.; Purushothaman, M.; Pautasso, F.; Armstrong, G.; Sorathia, A.; Crawley, J.; Nadipelli, V.R.; Rubin, B.; Newhardt, R.; et al. Tracking of Systemic Lupus Erythematosus (SLE) Longitudinally Using Biosensor and Patient-Reported Data: A Report on the Fully Decentralized Mobile Study to Measure and Predict Lupus Disease Activity Using Digital Signals-The OASIS Study. BioTech (Basel) 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yao, L.; Lee, Y.A.; Huang, Y.; Merkel, P.A.; Vina, E.; Yeh, Y.Y.; Li, Y.; Allen, J.M.; Bian, J.; et al. A fair machine learning model to predict flares of systemic lupus erythematosus. JAMIA Open 2025, 8, ooaf072. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Chen, Y.; Song, Y.; Wu, K.; Chen, T.; Zhang, Y.; Jia, W.; Zhang, H.T.; Liang, D.D.; Yang, J.; et al. Deep learning model to predict lupus nephritis renal flare based on dynamic multivariable time-series data. BMJ Open 2024, 14, e071821. [Google Scholar] [CrossRef] [PubMed]
- Kamudoni, P.; Lyden, K.; Gunther, O.; Jaitely, V.; Araujo, T.D.; Spies, E.; Park, J.; Thomas, E.; Buie, J.; Blankenship, J.M.; et al. Identifying meaningful aspects of health and concepts of interest for assessment in systemic lupus erythematosus: implications for digital clinical measure development. J Patient Rep Outcomes 2024, 8, 154. [Google Scholar] [CrossRef]
- Chen L, D.O. , Fang T, Chen M, Zhang X, Cong R, Lu D, Zhang R, Jin Q, Wang X. Phenome-wide causal proteomics enhance systemic lupus erythematosus flare prediction: A study in Asian populations. arxiv.
- Brzezinska, O.E.; Rychlicki-Kicior, K.A.; Makowska, J.S. Automatic assessment of nailfold capillaroscopy software: a pilot study. Reumatologia 2024, 62, 346–350. [Google Scholar] [CrossRef]
- Ebadi Jalal, M.; Emam, O.S.; Castillo-Olea, C.; Garcia-Zapirain, B.; Elmaghraby, A. Abnormality detection in nailfold capillary images using deep learning with EfficientNet and cascade transfer learning. Sci Rep 2025, 15, 2068. [Google Scholar] [CrossRef]
- Emam O S, E.J.M., Garcia-Zapirain B, Elmaghraby A S. Artificial Intelligence Algorithms in Nailfold Capillaroscopy Image Analysis: A Systematic Review. medrxiv 2024. [CrossRef]
- Lledo-Ibanez, G.M.; Saez Comet, L.; Freire Dapena, M.; Mesa Navas, M.; Martin Cascon, M.; Guillen Del Castillo, A.; Simeon, C.P.; Martinez Robles, E.; Todoli Parra, J.; Varela, D.C.; et al. CAPI-Detect: machine learning in capillaroscopy reveals new variables influencing diagnosis. Rheumatology (Oxford) 2025, 64, 3667–3675. [Google Scholar] [CrossRef]
- Adams, L.C.; Bressem, K.K.; Poddubnyy, D. Artificial intelligence and machine learning in axial spondyloarthritis. Curr Opin Rheumatol 2024, 36, 267–273. [Google Scholar] [CrossRef]
- Pons, M.; Georgiadis, S.; Hetland, M.L.; Ahmadzay, Z.F.; Rasmussen, S.; Christiansen, S.N.; Di Giuseppe, D.; Wallman, J.K.; Pavelka, K.; Zavada, J.; et al. Predictors of Secukinumab Treatment Response and Continuation in Axial Spondyloarthritis: Results From the EuroSpA Research Collaboration Network. J Rheumatol 2025, 52, 572–582. [Google Scholar] [CrossRef]
- Dalix, E.; Marcelli, C.; Bejan-Angoulvant, T.; Finckh, A.; Rancon, F.; Akrour, M.; De Araujo, L.; Presles, E.; Marotte, H.; group, R.O.-S.s. Rotation or change of biotherapy after TNF blocker treatment failure for axial spondyloarthritis: the ROC-SpA study, a randomised controlled study protocol. BMJ Open 2024, 14, e087872. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, G.; Scagnellato, L.; Lorenzin, M.; Collesei, A.; Oliviero, F.; Damasco, A.; Cosma, C.; Basso, D.; Doria, A.; Ramonda, R. Predictors of response to bDMARDs and tsDMARDs in psoriatic arthritis: a pilot study on the role of musculoskeletal ultrasound. Front Med (Lausanne) 2024, 11, 1482894. [Google Scholar] [CrossRef] [PubMed]
- Tillett, W.; Ogdie, A.; Passey, A.; Gorecki, P. Impact of psoriatic arthritis and comorbidities on ustekinumab outcomes in psoriasis: a retrospective, observational BADBIR cohort study. RMD Open 2023, 9. [Google Scholar] [CrossRef] [PubMed]
- Kunzler, T.; Bamert, M.; Sprott, H. Factors predicting treatment response to biological and targeted synthetic disease-modifying antirheumatic drugs in psoriatic arthritis - a systematic review and meta-analysis. Clin Rheumatol 2024, 43, 3723–3746. [Google Scholar] [CrossRef]
- Guo, H.; Gao, J.; Gong, L.; Wang, Y. Multi-omics analysis reveals novel causal pathways in psoriasis pathogenesis. J Transl Med 2025, 23, 100. [Google Scholar] [CrossRef]
- Shi, Z.; Ding, Y.; Dong, X.; Li, G.; Li, B.; Hou, J.; Xue, L. The diagnostic value and clinical relevance of salivary gland ultrasound in patients with highly suspected Sjogren's Disease: a prospective monocentric study. Arthritis Res Ther 2025, 27, 175. [Google Scholar] [CrossRef]
- Yang, J.; Park, Y.; Lee, J.J.; Kim, W.U.; Park, S.H.; Kwok, S.K. Clinical value of salivary gland ultrasonography in evaluating secretory function, disease activity, and lymphoma risk factors in primary Sjogren's syndrome. Clin Rheumatol 2025, 44, 1643–1652. [Google Scholar] [CrossRef]
- De Vita, S.; Isola, M.; Baldini, C.; Goules, A.V.; Chatzis, L.G.; Quartuccio, L.; Zabotti, A.; Giovannini, I.; Donati, V.; Ferro, F.; et al. Predicting lymphoma in Sjogren's syndrome and the pathogenetic role of parotid microenvironment through precise parotid swelling recording. Rheumatology (Oxford) 2023, 62, 1586–1593. [Google Scholar] [CrossRef]
- Umapathy, V.R.; Natarajan, P.M.; Swamikannu, B. Review Insights on Salivary Proteomics Biomarkers in Oral Cancer Detection and Diagnosis. Molecules 2023, 28. [Google Scholar] [CrossRef]
- Sembler-Moller, M.L.; Belstrom, D.; Locht, H.; Pedersen, A.M.L. Proteomics of saliva, plasma, and salivary gland tissue in Sjogren's syndrome and non-Sjogren patients identify novel biomarker candidates. J Proteomics 2020, 225, 103877. [Google Scholar] [CrossRef]
- Hu, S.; Gao, K.; Pollard, R.; Arellano-Garcia, M.; Zhou, H.; Zhang, L.; Elashoff, D.; Kallenberg, C.G.; Vissink, A.; Wong, D.T. Preclinical validation of salivary biomarkers for primary Sjogren's syndrome. Arthritis Care Res (Hoboken) 2010, 62, 1633–1638. [Google Scholar] [CrossRef] [PubMed]
- Bonroy, C.; Piette, Y.; Allenbach, Y.; Bossuyt, X.; Damoiseaux, J. Positioning of myositis-specific and associated autoantibody (MSA/MAA) testing in disease criteria and routine diagnostic work-up. J Transl Autoimmun 2022, 5, 100148. [Google Scholar] [CrossRef] [PubMed]
- McLeish, E.; Slater, N.; Mastaglia, F.L.; Needham, M.; Coudert, J.D. From data to diagnosis: how machine learning is revolutionizing biomarker discovery in idiopathic inflammatory myopathies. Brief Bioinform 2023, 25. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, X.; Du, Y.; Wang, L.; Wang, Q.; Wu, H.; Liu, L.; Xue, J. Mortality risk in patients with anti-MDA5 dermatomyositis is related to rapidly progressive interstitial lung disease and anti-Ro52 antibody. Arthritis Res Ther 2023, 25, 127. [Google Scholar] [CrossRef]
- Nagawa, K.; Suzuki, M.; Yamamoto, Y.; Inoue, K.; Kozawa, E.; Mimura, T.; Nakamura, K.; Nagata, M.; Niitsu, M. Texture analysis of muscle MRI: machine learning-based classifications in idiopathic inflammatory myopathies. Sci Rep 2021, 11, 9821. [Google Scholar] [CrossRef]
- Wang, F.; Zhou, S.; Hou, B.; Santini, F.; Yuan, L.; Guo, Y.; Zhu, J.; Hilbert, T.; Kober, T.; Zhang, Y.; et al. Assessment of idiopathic inflammatory myopathy using a deep learning method for muscle T2 mapping segmentation. Eur Radiol 2023, 33, 2350–2357. [Google Scholar] [CrossRef]
- Danieli, M.G.; Brunetto, S.; Gammeri, L.; Palmeri, D.; Claudi, I.; Shoenfeld, Y.; Gangemi, S. Machine learning application in autoimmune diseases: State of art and future prospectives. Autoimmun Rev 2024, 23, 103496. [Google Scholar] [CrossRef]
- Moingeon, P. Artificial intelligence-driven drug development against autoimmune diseases. Trends Pharmacol Sci 2023, 44, 411–424. [Google Scholar] [CrossRef]
- Wu, H.; Li, X.; Xu, H.; Li, Z.; Feng, F.; Zhang, J.; Xu, Z.; Ni, H.; Guo, Y.; Li, Y. Malignancy in Idiopathic Inflammatory Myopathies: Recent Insights. Clin Rev Allergy Immunol 2025, 68, 83. [Google Scholar] [CrossRef]
- van der Geest, K.S.M.; Treglia, G.; Glaudemans, A.; Brouwer, E.; Sandovici, M.; Jamar, F.; Gheysens, O.; Slart, R. Diagnostic value of [18F]FDG-PET/CT for treatment monitoring in large vessel vasculitis: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging 2021, 48, 3886–3902. [Google Scholar] [CrossRef]
- Wilk, B.; Wisenberg, G.; Dharmakumar, R.; Thiessen, J.D.; Goldhawk, D.E.; Prato, F.S. Hybrid PET/MR imaging in myocardial inflammation post-myocardial infarction. J Nucl Cardiol 2020, 27, 2083–2099. [Google Scholar] [CrossRef] [PubMed]
- Brilland, B.; Riou, J.; Quemeneur, T.; Vandenbussche, C.; Merillon, N.; Boizard-Moracchini, A.; Roy, M.; Despre, M.; Piccoli, G.B.; Djema, A.; et al. Identification of Renal Transcripts Associated with Kidney Function and Prognosis in ANCA-Associated Vasculitis. J Am Soc Nephrol 2025. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Han, S.; Li, L.; Fu, Y.; Zhou, D. Interferon-Stimulated Genes: Novel Targets in Renal Pathogenesis. Kidney Dis (Basel) 2025, 11, 390–401. [Google Scholar] [CrossRef]
- Omar, M.; Agbareia, R.; Naffaa, M.E.; Watad, A.; Glicksberg, B.S.; Nadkarni, G.N.; Klang, E. Applications of Artificial Intelligence in Vasculitides: A Systematic Review. ACR Open Rheumatol 2025, 7, e70016. [Google Scholar] [CrossRef] [PubMed]
- Maarseveen, T.D.; Glas, H.K.; Veris-van Dieren, J.; van den Akker, E.; Knevel, R. Improving musculoskeletal care with AI enhanced triage through data driven screening of referral letters. NPJ Digit Med 2025, 8, 98. [Google Scholar] [CrossRef]
- Knitza, J.; Janousek, L.; Kluge, F.; von der Decken, C.B.; Kleinert, S.; Vorbruggen, W.; Kleyer, A.; Simon, D.; Hueber, A.J.; Muehlensiepen, F.; et al. Machine learning-based improvement of an online rheumatology referral and triage system. Front Med (Lausanne) 2022, 9, 954056. [Google Scholar] [CrossRef]
- Guo, L.; Wang, J.; Li, J.; Yao, J.; Zhao, H. Biomarkers of rheumatoid arthritis-associated interstitial lung disease: a systematic review and meta-analysis. Front Immunol 2024, 15, 1455346. [Google Scholar] [CrossRef]
- Frederiksen, B.A.; Hammer, H.B.; Terslev, L.; Ammitzboll-Danielsen, M.; Savarimuthu, T.R.; Weber, A.B.H.; Just, S.A. Automated ultrasound system ARTHUR V.2.0 with AI analysis DIANA V.2.0 matches expert rheumatologist in hand joint assessment of rheumatoid arthritis patients. RMD Open 2025, 11. [Google Scholar] [CrossRef]
- Nigro, A. Fast-track capillaroscopic progression in systemic sclerosis: a case-based review of active pattern emerging within 3 months of raynaud's phenomenon onset. Rheumatol Int 2025, 45, 203. [Google Scholar] [CrossRef]
- Giansanti, D. Revolutionizing Medical Imaging: The Transformative Role of Artificial Intelligence in Diagnostics and Treatment. Diagnostics (Basel) 2025, 15. [Google Scholar] [CrossRef]
- Kocak, B.; Ponsiglione, A.; Stanzione, A.; Bluethgen, C.; Santinha, J.; Ugga, L.; Huisman, M.; Klontzas, M.E.; Cannella, R.; Cuocolo, R. Bias in artificial intelligence for medical imaging: fundamentals, detection, avoidance, mitigation, challenges, ethics, and prospects. Diagn Interv Radiol 2025, 31, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Hasanzadeh, F.; Josephson, C.B.; Waters, G.; Adedinsewo, D.; Azizi, Z.; White, J.A. Bias recognition and mitigation strategies in artificial intelligence healthcare applications. NPJ Digit Med 2025, 8, 154. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.S.; Eun, S.; Shin, K.; Yoon, H.; Hong, C.; Kim, D.H.; Hong, S.; Kim, Y.G.; Lee, C.K.; Yoo, B.; et al. Machine learning model for identifying important clinical features for predicting remission in patients with rheumatoid arthritis treated with biologics. Arthritis Res Ther 2021, 23, 178. [Google Scholar] [CrossRef] [PubMed]
- Bellocchi, C.; Favalli, E.G.; Maioli, G.; Agape, E.; Rossato, M.; Paini, M.; Severino, A.; Vigone, B.; Biggioggero, M.; Trombetta, E.; et al. Whole-Blood RNA Sequencing Profiling of Patients With Rheumatoid Arthritis Treated With Tofacitinib. ACR Open Rheumatol 2025, 7, e11761. [Google Scholar] [CrossRef]
- Lee, H.K.; Jung, O.; Hennighausen, L. JAK inhibitors dampen activation of interferon-stimulated transcription of ACE2 isoforms in human airway epithelial cells. Commun Biol 2021, 4, 654. [Google Scholar] [CrossRef]
- Bergero, M.A.; Martinez, P.; Modina, P.; Hosman, R.; Villamil, W.; Gudino, R.; David, C.; Costa, L. Artificial intelligence model for predicting early biochemical recurrence of prostate cancer after robotic-assisted radical prostatectomy. Sci Rep 2025, 15, 30822. [Google Scholar] [CrossRef]
- Chen, M.M.; Rosenkrantz, A.B.; Nicola, G.N.; Silva, E., 3rd; McGinty, G.; Manchikanti, L.; Hirsch, J.A. The Qualified Clinical Data Registry: A Pathway to Success within MACRA. AJNR Am J Neuroradiol 2017, 38, 1292–1296. [Google Scholar] [CrossRef]
- Izadi, Z.; Schmajuk, G.; Gianfrancesco, M.; Subash, M.; Evans, M.; Trupin, L.; Yazdany, J. Significant Gains in Rheumatoid Arthritis Quality Measures Among RISE Registry Practices. Arthritis Care Res (Hoboken) 2022, 74, 219–228. [Google Scholar] [CrossRef]
- E, K.; J, L.; J, A.-M.; J, Y.; S, S.; et al. Association of Qualified Clinical Data Registry Clinician Dashboard Engagement With Performance on Quality-of-Care Measures: Cross-Sectional Analysis. Journal of Medical Internet Research 2025, 27. [Google Scholar]
- Tabari, P.; Costagliola, G.; De Rosa, M.; Boeker, M. State-of-the-Art Fast Healthcare Interoperability Resources (FHIR)-Based Data Model and Structure Implementations: Systematic Scoping Review. JMIR Med Inform 2024, 12, e58445. [Google Scholar] [CrossRef]
- Marfoglia, A.; Nardini, F.; Arcobelli, V.A.; Moscato, S.; Mellone, S.; Carbonaro, A. Towards real-world clinical data standardization: A modular FHIR-driven transformation pipeline to enhance semantic interoperability in healthcare. Comput Biol Med 2025, 187, 109745. [Google Scholar] [CrossRef] [PubMed]
- Rossander, A.; Lindskold, L.; Ranerup, A.; Karlsson, D. A State-of-the Art Review of SNOMED CT Terminology Binding and Recommendations for Practice and Research. Methods Inf Med 2021, 60, e76–e88. [Google Scholar] [CrossRef] [PubMed]
- Bakken, S. Standards and frameworks. J Am Med Inform Assoc 2024, 31, 1629–1630. [Google Scholar] [CrossRef]
- Shen, Y.; Yu, J.; Zhou, J.; Hu, G. Twenty-Five Years of Evolution and Hurdles in Electronic Health Records and Interoperability in Medical Research: Comprehensive Review. J Med Internet Res 2025, 27, e59024. [Google Scholar] [CrossRef] [PubMed]
- Gulden, C.; Macho, P.; Reinecke, I.; Strantz, C.; Prokosch, H.U.; Blasini, R. recruIT: A cloud-native clinical trial recruitment support system based on Health Level 7 Fast Healthcare Interoperability Resources (HL7 FHIR) and the Observational Medical Outcomes Partnership Common Data Model (OMOP CDM). Comput Biol Med 2024, 174, 108411. [Google Scholar] [CrossRef]
- Kim D, O.K.; Lee Y, W.H. Overview of fair federated learning for fairness and privacy preservation. Expert Systems with Applications 2025, 293. [Google Scholar] [CrossRef]
- Ye, H.; Zhang, X.; Liu, K.; Liu, Z.; Chen, W.; Liu, B.; Ngai, E.W.; Hu, Y. A personalized federated learning approach to enhance joint modeling for heterogeneous medical institutions. Digit Health 2025, 11, 20552076251360861. [Google Scholar] [CrossRef]
- Dayan, I.; Roth, H.R.; Zhong, A.; Harouni, A.; Gentili, A.; Abidin, A.Z.; Liu, A.; Costa, A.B.; Wood, B.J.; Tsai, C.S.; et al. Federated learning for predicting clinical outcomes in patients with COVID-19. Nat Med 2021, 27, 1735–1743. [Google Scholar] [CrossRef]
- Solitano, V.; Ahuja, D.; Lee, H.H.; Gaikwad, R.; Yeh, K.H.; Facciorusso, A.; Singh, A.G.; Ma, C.; Ananthakrishnan, A.N.; Yuan, Y.; et al. Comparative Safety of JAK Inhibitors vs TNF Antagonists in Immune-Mediated Inflammatory Diseases: A Systematic Review and Meta-Analysis. JAMA Netw Open 2025, 8, e2531204. [Google Scholar] [CrossRef]
- Li, H.; Zang, C.; Xu, Z.; Pan, W.; Rajendran, S.; Chen, Y.; Wang, F. Federated target trial emulation using distributed observational data for treatment effect estimation. NPJ Digit Med 2025, 8, 387. [Google Scholar] [CrossRef]
- Matta, S.; Lamard, M.; Zhang, P.; Le Guilcher, A.; Borderie, L.; Cochener, B.; Quellec, G. A systematic review of generalization research in medical image classification. Comput Biol Med 2024, 183, 109256. [Google Scholar] [CrossRef]
- Le, J.P.; Shashikumar, S.P.; Malhotra, A.; Nemati, S.; Wardi, G. Making the Improbable Possible: Generalizing Models Designed for a Syndrome-Based, Heterogeneous Patient Landscape. Crit Care Clin 2023, 39, 751–768. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Bai, J.; Li, N.; Li, X.; Liu, D.; Buckeridge, D.L.; Li, Y. FedWeight: mitigating covariate shift of federated learning on electronic health records data through patients re-weighting. NPJ Digit Med 2025, 8, 286. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Reps, J.M.; Li, L.; Zhang, B.; Lu, Y.; Tong, J.; Zhang, D.; Lumley, T.; Brand, M.T.; Van Zandt, M.; et al. COLA-GLM: collaborative one-shot and lossless algorithms of generalized linear models for decentralized observational healthcare data. NPJ Digit Med 2025, 8, 442. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Zhai, D.; Bai, G.; Jiang, J.; Ye, Q.; Ji, X.; Liu, X. Towards fairness-aware and privacy-preserving enhanced collaborative learning for healthcare. Nat Commun 2025, 16, 2852. [Google Scholar] [CrossRef]
- Curtis, J.R.; Su, Y.; Black, S.; Xu, S.; Langholff, W.; Bingham, C.O.; Kafka, S.; Xie, F. Machine Learning Applied to Patient-Reported Outcomes to Classify Physician-Derived Measures of Rheumatoid Arthritis Disease Activity. ACR Open Rheumatol 2022, 4, 995–1003. [Google Scholar] [CrossRef]
- Williams, E.; Kienast, M.; Medawar, E.; Reinelt, J.; Merola, A.; Klopfenstein, S.A.I.; Flint, A.R.; Heeren, P.; Poncette, A.S.; Balzer, F.; et al. A Standardized Clinical Data Harmonization Pipeline for Scalable AI Application Deployment (FHIR-DHP): Validation and Usability Study. JMIR Med Inform 2023, 11, e43847. [Google Scholar] [CrossRef]
- Xiao, G.; Pfaff, E.; Prud'hommeaux, E.; Booth, D.; Sharma, D.K.; Huo, N.; Yu, Y.; Zong, N.; Ruddy, K.J.; Chute, C.G.; et al. FHIR-Ontop-OMOP: Building clinical knowledge graphs in FHIR RDF with the OMOP Common data Model. J Biomed Inform 2022, 134, 104201. [Google Scholar] [CrossRef]
- Collins, G.S.; Moons, K.G.M.; Dhiman, P.; Riley, R.D.; Beam, A.L.; Calster, B.V.; Ghassemi, M.; Liu, X.; Reitsma, J.B.; Smeden, M.V.; et al. TRIPOD+AI statement: updated guidance for reporting clinical prediction models that use regression or machine learning methods: a Korean translation. Ewha Med J 2025, 48, e48. [Google Scholar] [CrossRef]
- Hopewell, S.; Chan, A.W.; Collins, G.S.; Hrobjartsson, A.; Moher, D.; Schulz, K.F.; Tunn, R.; Aggarwal, R.; Berkwits, M.; Berlin, J.A.; et al. CONSORT 2025 explanation and elaboration: updated guideline for reporting randomised trials. BMJ 2025, 389, e081124. [Google Scholar] [CrossRef]
- Collins, G.S.; Dhiman, P.; Ma, J.; Schlussel, M.M.; Archer, L.; Van Calster, B.; Harrell, F.E., Jr.; Martin, G.P.; Moons, K.G.M.; van Smeden, M.; et al. Evaluation of clinical prediction models (part 1): from development to external validation. BMJ 2024, 384, e074819. [Google Scholar] [CrossRef] [PubMed]
- de Hond, A.A.H.; Shah, V.B.; Kant, I.M.J.; Van Calster, B.; Steyerberg, E.W.; Hernandez-Boussard, T. Perspectives on validation of clinical predictive algorithms. NPJ Digit Med 2023, 6, 86. [Google Scholar] [CrossRef] [PubMed]
- Piovani, D.; Sokou, R.; Tsantes, A.G.; Vitello, A.S.; Bonovas, S. Optimizing Clinical Decision Making with Decision Curve Analysis: Insights for Clinical Investigators. Healthcare (Basel) 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Vickers, A.J.; Holland, F. Decision curve analysis to evaluate the clinical benefit of prediction models. Spine J 2021, 21, 1643–1648. [Google Scholar] [CrossRef]
- Kerr, K.F.; Brown, M.D.; Zhu, K.; Janes, H. Assessing the Clinical Impact of Risk Prediction Models With Decision Curves: Guidance for Correct Interpretation and Appropriate Use. J Clin Oncol 2016, 34, 2534–2540. [Google Scholar] [CrossRef]
- El Arab, R.A.; Al Moosa, O.A. Systematic review of cost effectiveness and budget impact of artificial intelligence in healthcare. NPJ Digit Med 2025, 8, 548. [Google Scholar] [CrossRef]
- Tam, T.Y.C.; Sivarajkumar, S.; Kapoor, S.; Stolyar, A.V.; Polanska, K.; McCarthy, K.R.; Osterhoudt, H.; Wu, X.; Visweswaran, S.; Fu, S.; et al. A framework for human evaluation of large language models in healthcare derived from literature review. NPJ Digit Med 2024, 7, 258. [Google Scholar] [CrossRef]
- Venerito, V. Artificial intelligence in rheumatology: days of a future past. Rheumatol Adv Pract 2025, 9, rkaf022. [Google Scholar] [CrossRef]
- Chopra, H.; Annu; Shin, D. K.; Munjal, K.; Priyanka; Dhama, K.; Emran, T.B. Revolutionizing clinical trials: the role of AI in accelerating medical breakthroughs. Int J Surg 2023, 109, 4211–4220. [Google Scholar] [CrossRef]
- Dolin P, L.W. , Dasarathy G, Berisha V. Statistically Valid Post-Deployment Monitoring Should Be Standard for AI-Based Digital Health. arxiv.
- Moreno-Grau, S.; Vernekar, M.; Lopez-Pineda, A.; Mas-Montserrat, D.; Barrabes, M.; Quinto-Cortes, C.D.; Moatamed, B.; Lee, M.T.M.; Yu, Z.; Numakura, K.; et al. Polygenic risk score portability for common diseases across genetically diverse populations. Hum Genomics 2024, 18, 93. [Google Scholar] [CrossRef]
- Miao, J.; Guo, H.; Song, G.; Zhao, Z.; Hou, L.; Lu, Q. Quantifying portable genetic effects and improving cross-ancestry genetic prediction with GWAS summary statistics. Nat Commun 2023, 14, 832. [Google Scholar] [CrossRef]
- Roschewitz, M.; Khara, G.; Yearsley, J.; Sharma, N.; James, J.J.; Ambrozay, E.; Heroux, A.; Kecskemethy, P.; Rijken, T.; Glocker, B. Automatic correction of performance drift under acquisition shift in medical image classification. Nat Commun 2023, 14, 6608. [Google Scholar] [CrossRef]
- Lambert, B.; Forbes, F.; Doyle, S.; Dehaene, H.; Dojat, M. Trustworthy clinical AI solutions: A unified review of uncertainty quantification in Deep Learning models for medical image analysis. Artif Intell Med 2024, 150, 102830. [Google Scholar] [CrossRef]
- Gilbert, S.; Adler, R.; Holoyad, T.; Weicken, E. Could transparent model cards with layered accessible information drive trust and safety in health AI? NPJ Digit Med 2025, 8, 124. [Google Scholar] [CrossRef] [PubMed]
- Chinta, S.V.; Wang, Z.; Palikhe, A.; Zhang, X.; Kashif, A.; Smith, M.A.; Liu, J.; Zhang, W. AI-driven healthcare: Fairness in AI healthcare: A survey. PLOS Digit Health 2025, 4, e0000864. [Google Scholar] [CrossRef] [PubMed]
- Gallifant, J.; Kistler, E.A.; Nakayama, L.F.; Zera, C.; Kripalani, S.; Ntatin, A.; Fernandez, L.; Bates, D.; Dankwa-Mullan, I.; Celi, L.A. Disparity dashboards: an evaluation of the literature and framework for health equity improvement. Lancet Digit Health 2023, 5, e831–e839. [Google Scholar] [CrossRef] [PubMed]
- Alderman, J.E.; Palmer, J.; Laws, E.; McCradden, M.D.; Ordish, J.; Ghassemi, M.; Pfohl, S.R.; Rostamzadeh, N.; Cole-Lewis, H.; Glocker, B.; et al. Tackling algorithmic bias and promoting transparency in health datasets: the STANDING Together consensus recommendations. Lancet Digit Health 2025, 7, e64–e88. [Google Scholar] [CrossRef]
- Teikari P, J.M. , Azh M, Pesola H. The Architecture of Trust: A Framework for AI-Augmented Real Estate Valuation in the Era of Structured Data. arxiv.
- Davis, S.E.; Embi, P.J.; Matheny, M.E. Sustainable deployment of clinical prediction tools-a 360 degrees approach to model maintenance. J Am Med Inform Assoc 2024, 31, 1195–1198. [Google Scholar] [CrossRef]
- Rosenthal, J.T.; Beecy, A.; Sabuncu, M.R. Rethinking clinical trials for medical AI with dynamic deployments of adaptive systems. NPJ Digit Med 2025, 8, 252. [Google Scholar] [CrossRef]
- Zerrouk, N.; Auge, F.; Niarakis, A. Building a modular and multi-cellular virtual twin of the synovial joint in Rheumatoid Arthritis. NPJ Digit Med 2024, 7, 379. [Google Scholar] [CrossRef]

| Disease | Key Biomarkers/Targets | AI/Digital Innovations | Clinical Impact | Key Limitations/Gaps |
|---|---|---|---|---|
| Rheumatoid Arthritis (RA) | Pre-RA prevention with abatacept (APIPPRA, ARIAA); autoantibodies (ACPA, RF); MRI-detected subclinical inflammation | Deep learning for US/MRI synovitis segmentation; sub-pixel JSN quantification; smartphone-based fist closure (MeFISTO) as a functional biomarker; ML models combining multi-omics + imaging | Demonstrates feasibility of disease interception; scalable imaging and digital biomarkers; early steps toward individualized drug response prediction | Long-term durability of prevention unknown; small imaging datasets; lack of external validation; heterogeneity in ML pipelines |
| Systemic Lupus Erythematosus (SLE) | Type I IFN gene signature; SIGLEC-1 expression; proteomic biomarkers (SAA1, B4GALT5, etc.) | IFN-signature guided therapy with anifrolumab; wearables + PROs (OASIS study); EHR-based flare prediction (FLAME); deep learning for lupus nephritis flares; proteomic + ML flare models | Establishes IFN signature as both predictive and monitoring biomarker; digital phenotyping enables early flare detection | Variable organ-specific response; inconsistent LN outcomes; digital tools often under-validated; flare definitions heterogeneous |
| Systemic Sclerosis (SSc) | Microvascular patterns (giant capillaries, hemorrhages, density loss) on nailfold capillaroscopy | AI-assisted NFC classification: ResNet, EfficientNet, CAPI-Detect; large, annotated NFC datasets; pattern staging (early/active/late) | Enhances reproducibility and early diagnosis; potential for risk stratification (e.g., pulmonary hypertension, ulcers) | Few longitudinal outcome studies; lack of standardized acquisition protocols; external validation limited |
| Spondyloarthritis (SpA) | HLA-B27, MRI sacroiliac inflammation, PROs, PK parameters | Registry-based ML models (EuroSpA secukinumab cohort); ROC-SpA trial testing PK-guided prediction | Supports treatment persistence and real-world prediction; PK may inform therapeutic drug monitoring | Heterogeneous endpoints; small sample sizes; lack of standardized composite outcomes |
| Psoriatic Arthritis (PsA) | Disease activity, comorbidities, sonographic inflammation | US-based short-interval predictors (MIJET/2MIJET); early discrimination of JAKi vs TNFi/ILi responses | Demonstrates feasibility of early imaging response markers; pragmatic outcome (drug retention) | Small pilot cohorts; scarce validated molecular predictors; multi-domain disease complicates modeling |
| Sjögren’s Syndrome (SjD) | SGUS scores (OMERACT, Hočevar); salivary/tear proteomics; expanded autoantibodies | Standardized SGUS linked to lymphoma risk; proteomic pipelines integrating saliva, plasma, tissue | Non-invasive early diagnosis and risk stratification; complements biopsy | Need for longitudinal validation; risk of over-screening; proteomic candidates require replication |
| Idiopathic Inflammatory Myopathies (IIM) | Myositis-specific autoantibodies (MSAs); MRI muscle edema; multi-omics panels | ML clustering integrating MSAs + MRI + omics; radiomics-based antibody group prediction | Improved subtype stratification; potential guidance for ILD or therapy selection | Mostly retrospective, single-center; translation to outcomes (e.g., steroid-sparing) unproven |
| Vasculitides | CRP, ANCA patterns, type I IFN signatures; renal 12-gene transcriptomic panel | PET-CT radiomics/ML distinguishing GCA vs atherosclerosis; transcriptomics predicting kidney failure in AAV | Enables precision risk stratification (renal outcomes, vascular inflammation) | Early-stage; need for harmonized endpoints; prospective trials embedding predictors into care are lacking |
| Domain | Data/ Input |
Model Types | Validation Status | Clinical Maturity | Key Challenges | Next Translational Step |
|---|---|---|---|---|---|---|
| Triage & Access | Referral letters (NLP), structured intake | NLP (transformers, boosting) | External-site validation [199] | High – first in line for deployment | Calibration drift, subgroup fairness, governance rules | Registry/workflow embedding, prospective drift monitoring |
| Imaging Decision Support | MRI, US, NVC images | CNNs, end-to-end pipelines | Multicenter feasibility (MRI/US/NVC); reproducibility demonstrated [97,98,172] | Moderate – reader-assist tools maturing | Scanner/vendor heterogeneity, lack of prospective trials | Workflow-embedded prospective evaluation; standardized reporting |
| Therapy Selection | Clinical data, serology, omics, imaging | Gradient boosting, multimodal ML, RNA-seq signatures | Mostly retrospective; limited external/temporal validation [118,149] | Early – promising but not trial-ready | Heterogeneity, lack of harmonized endpoints, no impact trials | Registry pilots; decision-curve analysis; prospective impact studies |
| Risk Stratification (Adjacent: RA-ILD) | Biomarkers (KL-6), imaging, clinical data | XGBoost, ensemble ML | Cohort-level validation [201] | Moderate – translational potential for early screening | Generalizability, integration into care pathway |
| Infrastructure Pillar | Strengths | Limitations | Clinical Applications |
|---|---|---|---|
| Registries & EHR (e.g., RISE) | National-scale QCDR; supports quality improvement and reimbursement; real-world evidence of improved care quality. | Dependent on practice adoption and data quality; disease coverage is still limited (e.g., lupus measures emerging). | Quality benchmarking, CMS Quality Payment Program reporting, registry-based research. |
| Interoperability (OMOP/FHIR) | FHIR enables clinical data exchange; OMOP supports multi-site analytics; hybrid architectures proven feasible. | Standards alone are insufficient; require metadata, governance, and ontology alignment (SNOMED CT, LOINC). | Multi-site analytics, phenotyping, clinical trial recruitment, harmonized real-world evidence generation. |
| Privacy-Preserving Collaboration (Federated Learning) | Allows multi-site model training without centralizing patient data; governance frameworks emerging; feasible in diverse clinical tasks. | Technically complex; potential for bias and fairness issues; resource-intensive implementation. | Comparative effectiveness research (e.g., RA biologics), collaborative risk prediction across sites. |
| Multisite Modeling Pitfalls & Mitigations | Recognition of covariate shift, site bias, and acquisition drift; new methods (FedWeight, COLA-GLMM) improve calibration and cross-site validity. | Residual generalization challenges: continuous monitoring and retraining required. | Development of fairer, more robust models; deployment with embedded recalibration triggers. |
| Case Example: RA Disease Activity Prediction | Demonstrated feasibility using EHR + PRO features; established templates for computable disease-activity endpoints. | Early studies lacked robust external validation; not yet embedded in clinical dashboards. | Risk-stratified dashboards for RA management; integration of prediction models into quality improvement cycles. |
| Domain | Key Challenges | Risks if Unaddressed | Mitigation Strategies | Minimum Reporting Set |
|---|---|---|---|---|
| PRS Portability & Ancestry Gaps | Loss of accuracy across ancestries due to allele-frequency and LD differences; poor calibration across sex, age, and SES strata. | Worsening health disparities; misleading risk predictions; inequitable clinical recommendations. | Multi-ancestry GWAS; ancestry-aware modeling; ancestry/site-specific recalibration; subgroup reporting of performance. | Ancestry-stratified R²/AUC; calibration curves; decision-curve analysis by subgroup. |
| Data Drift & Bias Audits | Model degradation due to covariate, prior-probability, acquisition, and concept drift; hidden biases in datasets. | Silent failure of AI models; unfair treatment allocation; erosion of clinical trust. | Drift detectors with temporal validation; scheduled recalibration; bias audits; integration of bias dashboards in registries. | Performance stratified by sex, age, ancestry, SES proxies, and site; bias dashboard outputs. |
| Governance & Safety by Design | Ensuring continuous safety across lifecycles: bias detection, calibration monitoring, clinician-defer thresholds, and accountability. | Unsafe deployment; lack of transparency; patient harm; regulatory non-compliance. | Pre-deployment bias assessment; live calibration monitoring; safety circuit-breakers; audit trails; periodic re-validation and changelogs. | Intended-use statement; subgroup performance reports; update logs with versioning; governance protocols. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




