Submitted:
05 October 2025
Posted:
06 October 2025
You are already at the latest version
Abstract
Keywords:
Background
Methods
Study Settings
Study Design
Sample Size and Data Collection
Data Collection Tools and Procedures
Data Analysis and Presentation
Ethical Clearance
Results
Profile of Study Setting
Performance Summary
Performance of iSC in Terms of Facility Operations (E’s) and Management (M’s)
Vaccine Arrival (E1)
Temperature Management (E2)
Storage and Transport Capacity (E3)
Facility Infrastructure and Equipment (E4)
Maintenance and Repair (E5)
Stock Management (E6)
Distribution of Vaccines and Dry Goods (E7)
Vaccine Management (E8)
Waste Management (E9)
Annual Need Forecast (M1)
Annual Work Planning (M2)
Supportive Supervision (M3)
iSC Performance Monitoring (M4)
Discussions
Conclusions and Recommendations
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- UNICEF. Immunization accessed https://data.unicef.org/topic/child-health/immunization/. Secondary Immunization accessed https://data.unicef.org/topic/child-health/immunization/ 2024.
- Ota, M.O.; de Moraes, J.C.; Vojtek, I.; et al. Unveiling the contributions of immunization for progressing towards Universal Health Coverage. Human Vaccines & Immunotherapeutics 2022, 18, 2036048. [Google Scholar] [CrossRef]
- WHO. IMMUNIZATION AGENDA 2030 A global strategy to leave no one behind; WHO: Sweizerland, Geneva.
- Huang, X.X.; Guillermet, E.; Le Gargasson, J.-B.; et al. Costing analysis and anthropological assessment of the vaccine supply chain system redesign in the Comé District (Benin). Vaccine 2017, 35, 2183–2188. [Google Scholar] [CrossRef] [PubMed]
- Pambudi, N.A.; Sarifudin, A.; Gandidi, I.M.; Romadhon, R. Vaccine cold chain management and cold storage technology to address the challenges of vaccination programs. Energy Reports 2022, 8, 955–972. [Google Scholar] [CrossRef]
- Cooper, S.; Betsch, C.; Sambala, E.Z.; Mchiza, N.; Wiysonge, C.S. Vaccine hesitancy–a potential threat to the achievements of vaccination programmes in Africa. Human vaccines & immunotherapeutics 2018, 14, 2355–2357. [Google Scholar]
- Tilahun, B.; Mekonnen, Z.; Sharkey, A.; et al. What we know and don’t know about the immunization program of Ethiopia: a scoping review of the literature. BMC Public Health 2020, 20, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Gebremedhin, S.; Shiferie, F.; Tsegaye, D.A.; et al. Perspectives on the Performance of the Ethiopian Vaccine Supply Chain and Logistics System after the Last Mile Delivery Initiative: A Phenomenological Study. The American Journal of Tropical Medicine and Hygiene 2024, 110, 1029. [Google Scholar] [CrossRef] [PubMed]
- Federal Democratic Republic of Ethiopia Pharmaceutical Supply Agency. Immunization supply chain management strategy (iSCM) 2018-2023. Addis Ababa 2018.
- Ministry of Health. National Implementation Guideline for Expanded Program on Immunization. Addis Ababa, Ethiopia 2021.
- WHO, UNICEF. Effective Vaccine Management (EVM) Global Data Analysis 2009-2020. Secondary Effective Vaccine Management (EVM) Global Data Analysis 2009-2020 2021. https://knowledge.unicef.org/resource/effective-vaccine-management-evm-global-data-analysis-2009-2020.
- Sharma, L.; Gupta, G.; Kapuria, B.; et al. Strengthening the immunization supply chain through EVM assessment: Comparing India’s two successive national assessments. Journal of Family Medicine and Primary Care 2022, 11, 5298–5304. [Google Scholar] [CrossRef] [PubMed]
- Prosser, W.; Jaillard, P.; Assy, E.; et al. System redesign of the immunization supply chain: experiences from Benin and Mozambique. Vaccine 2017, 35, 2162–2166. [Google Scholar] [CrossRef] [PubMed]
- Consortium of CBMP Universities. National Immunization Program Evaluation in Ethiopia. Addis Ababa; 2024.
- UNICEF, WHO. EVM Manager Guide (v1.01), 2021.
- Sethy, G.; Chisema, M.N.; Sharma, L.; et al. Key lessons learned from the immunization supply chain of Malawi, an African country using EVM2. 0. Vaccine: X 2022, 12, 100239. [Google Scholar]
- Ateudjieu, J.; Kenfack, B.; Nkontchou, B.W.; Demanou, M. Program on immunization and cold chain monitoring: the status in eight health districts in Cameroon. BMC research notes 2013, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Haidari, L.A.; Connor, D.L.; Wateska, A.R.; et al. Augmenting transport versus increasing cold storage to improve vaccine supply chains. PloS one 2013, 8, e64303. [Google Scholar] [CrossRef]
- Ministry of Health E. Routine immunization catch-up vaccination guidelines, 2022.
- Federal Ministry of Health. ETHIOPIA IMMUNIZATION FULL PORTFOLIO PLAN (FPP): SITUATIONAL ANALYSIS, 2022.
- Federal Ministry of Health. Ethiopia National Expanded Program on Immunization a Comprehensive Multi Year plan 2016 -2020. Addis Ababa, 2015.
- Nigatu, T.; Abraham, L.; Willems, H.; et al. The status of immunization program and challenges in Ethiopia: A mixed method study. SAGE Open Medicine 2024, 12, 20503121241237115. [Google Scholar] [CrossRef] [PubMed]
| Site selection | Assessed | |
|---|---|---|
| Total facilities | 302 | 302 |
| PR locations | 1 | 1 |
| SN locations | 15 | 15 |
| LD locations | 89 | 89 |
| SP locations | 196 | 196 |
| EVM Assessment Years | Primary store | Sub-national centers | Lower distribution | Service provision | Composite score |
|---|---|---|---|---|---|
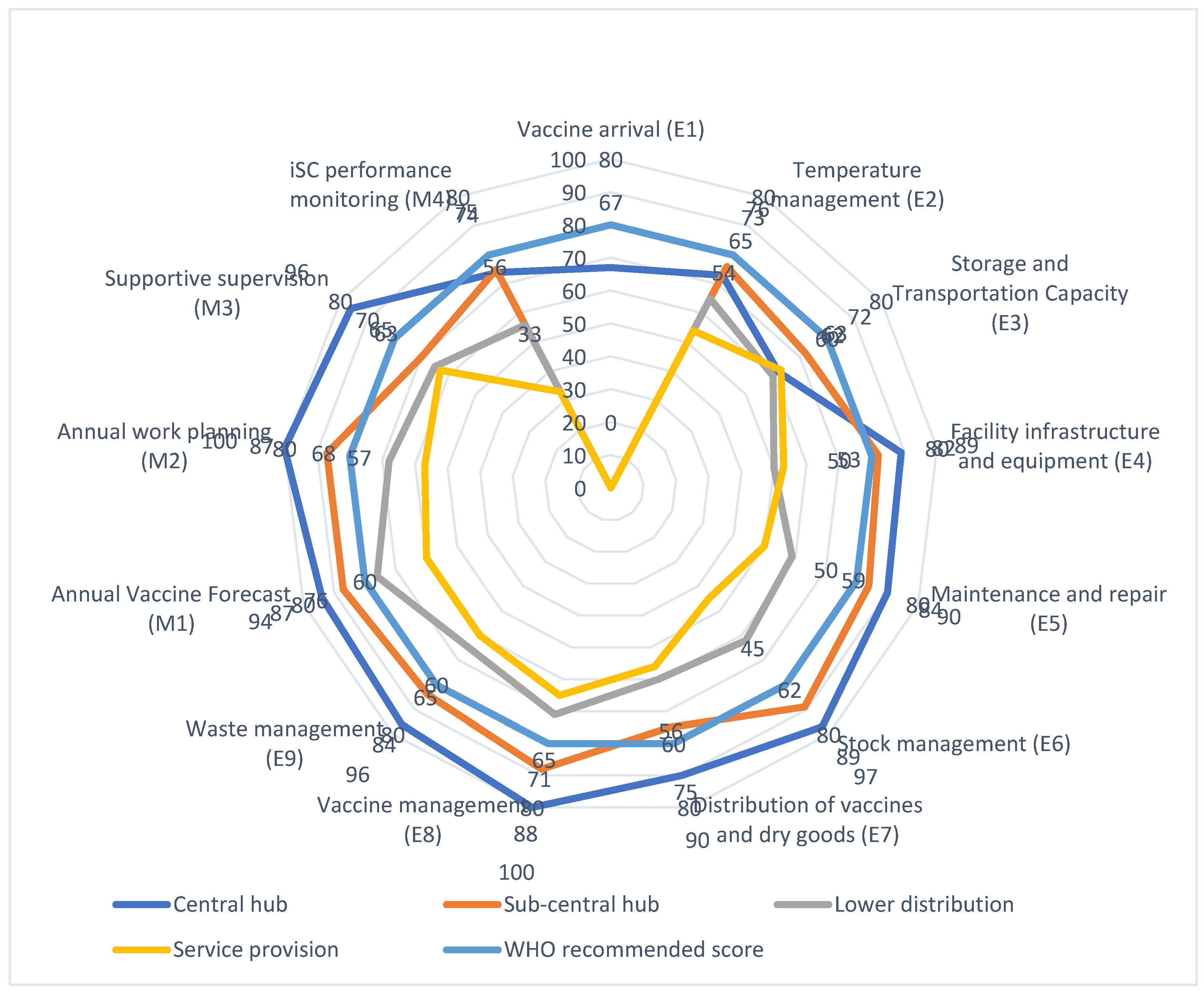
| Vaccine arrival (E1) | 67 | NA | NA | NA | 67 |
| Temperature management (E2) | 73 | 76 | 65 | 54 | 66 |
| Storage and Transportation Capacity (E3) | 62 | 72 | 60 | 63 | 64 |
| Facility infrastructure and equipment (E4) | 89 | 82 | 50 | 53 | 67 |
| Maintenance and repair (E5) | 90 | 84 | 59 | 50 | 69 |
| Stock management (E6) | 97 | 89 | 62 | 45 | 70 |
| Distribution of vaccines and dry goods (E7) | 90 | 75 | 60 | 56 | 69 |
| Vaccine management (E8) | 100 | 88 | 71 | 65 | 80 |
| Waste management (E9) | 96 | 84 | 65 | 60 | 75 |
| Annual Vaccine Forecast (M1) | 94 | 87 | 76 | 60 | 78 |
| Annual work planning (M2) | 100 | 87 | 68 | 57 | 76 |
| Supportive supervision (M3) | 96 | 70 | 65 | 63 | 72 |
| iSC performance monitoring (M4) | 74 | 75 | 56 | 33 | 57 |
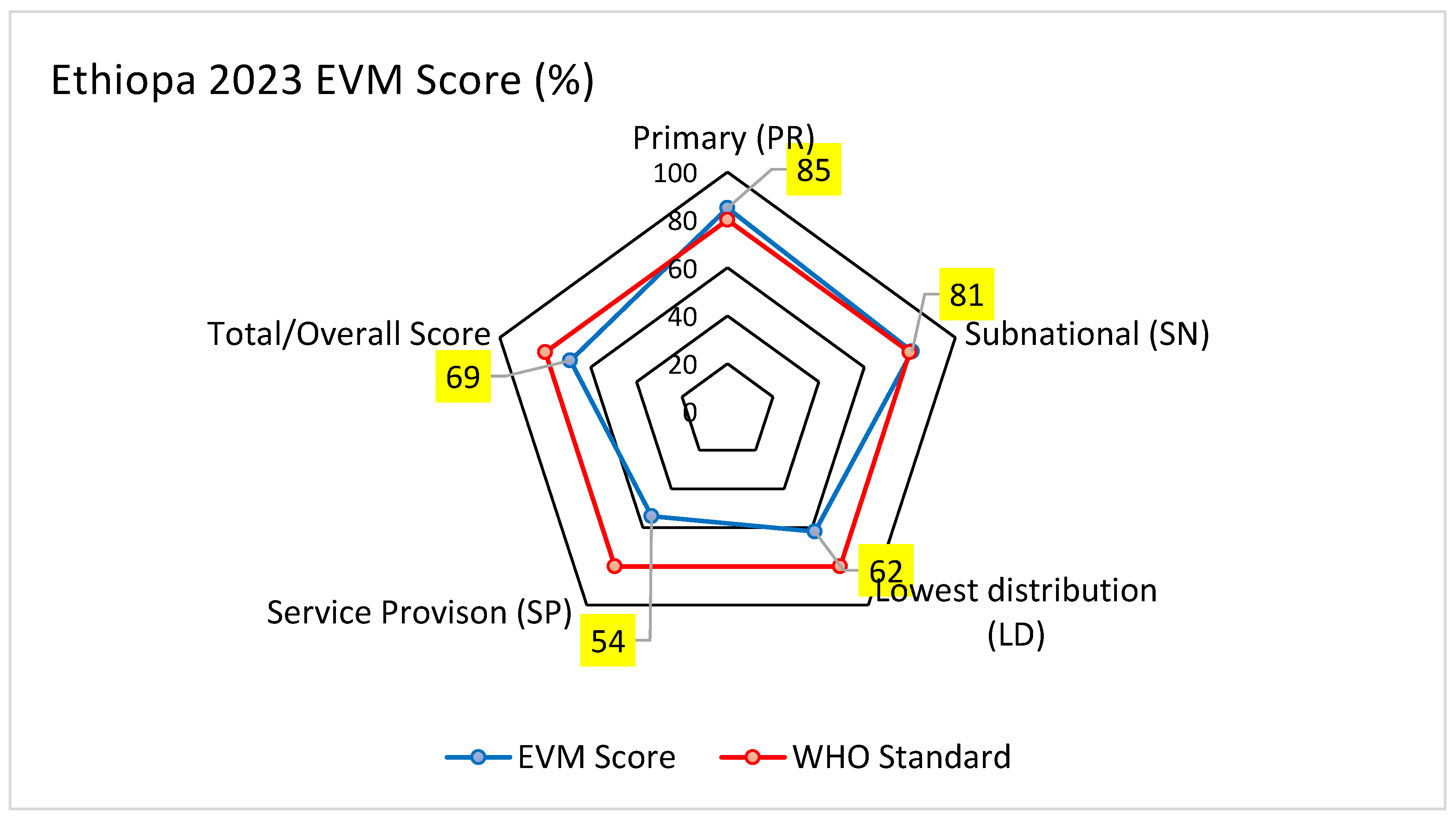
| Total | 85 | 81 | 62 | 54 | 69 |
| Primary store | Sub-national centers | Lower distribution (Woreda Health offices) | Service Provision | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Years | 2013 | 2019 | 2023 | 2013 | 2019 | 2023 | 2013 | 2019 | 2023 | 2013 | 2019 | 2023 |
| Vaccine arrival (E1) | 57 | 74 | 67 | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Temperature management (E2) | 40 | 75 | 73 | 54 | 63 | 76 | 71 | 71 | 65 | 69 | 70 | 54 |
| Storage and Transportation Capacity (E3) | 89 | 71 | 62 | 80 | 90 | 72 | 76 | 76 | 60 | 82 | 76 | 63 |
| Facility infrastructure and equipment (E4) | 91 | 96 | 89 | 80 | 82 | 82 | 69 | 71 | 50 | 76 | 74 | 53 |
| Maintenance and repair (E5) | 65 | 64 | 90 | 68 | 72 | 84 | 71 | 51 | 59 | 67 | 56 | 50 |
| Stock management (E6) | 64 | 77 | 97 | 54 | 80 | 89 | 51 | 57 | 62 | 53 | 50 | 45 |
| Distribution of vaccines and dry goods (E7) | 60 | 81 | 90 | 43 | 77 | 75 | 44 | 65 | 60 | 84 | 59 | 56 |
| Vaccine management (E8) | 50 | 80 | 100 | 68 | 84 | 88 | 67 | 77 | 71 | 80 | 82 | 65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




