Submitted:
03 October 2025
Posted:
04 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Animal Ethics Statement
2.3. Production of IgG-αGalomab: Immunization Protocol and Hybridoma Generation
2.4. Preparation of Bioprosthetic Commercial-Like Tissue Samples
2.5. Preparation of Red Meat Extract
2.6. ELISA Test
2.6.1. Sensitivity Analysis
2.6.2. Cross-Reactivity Assay
2.7α. Gal Detection Via Absorbance in Bovine Pericardium
2.8α. Gal Detection by Western Blot
2.9. α. Gal Detection by Flow Cytometry
2.10. α. Galomab Application in Histological Studies
Immunofluorescence and Immunohistochemistry
2.11. Statistical analysis
3. Results
3.1. Description of Experimental Data
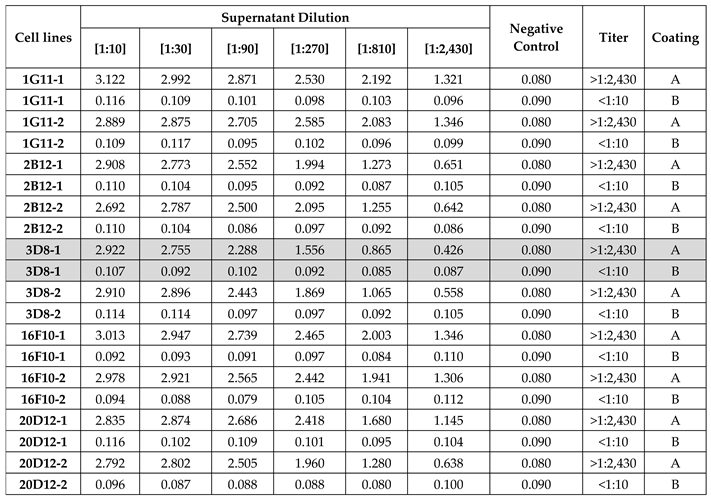
3.1.1. IgG-αGalomab Selection
3.1.2. Sensitivity Assay
3.1.3. Cross-Reactivity Assay
3.1.4.α. Gal detection in Bovine Pericardium
3.1.5. Western Blot
3.1.6. Flow Cytometry
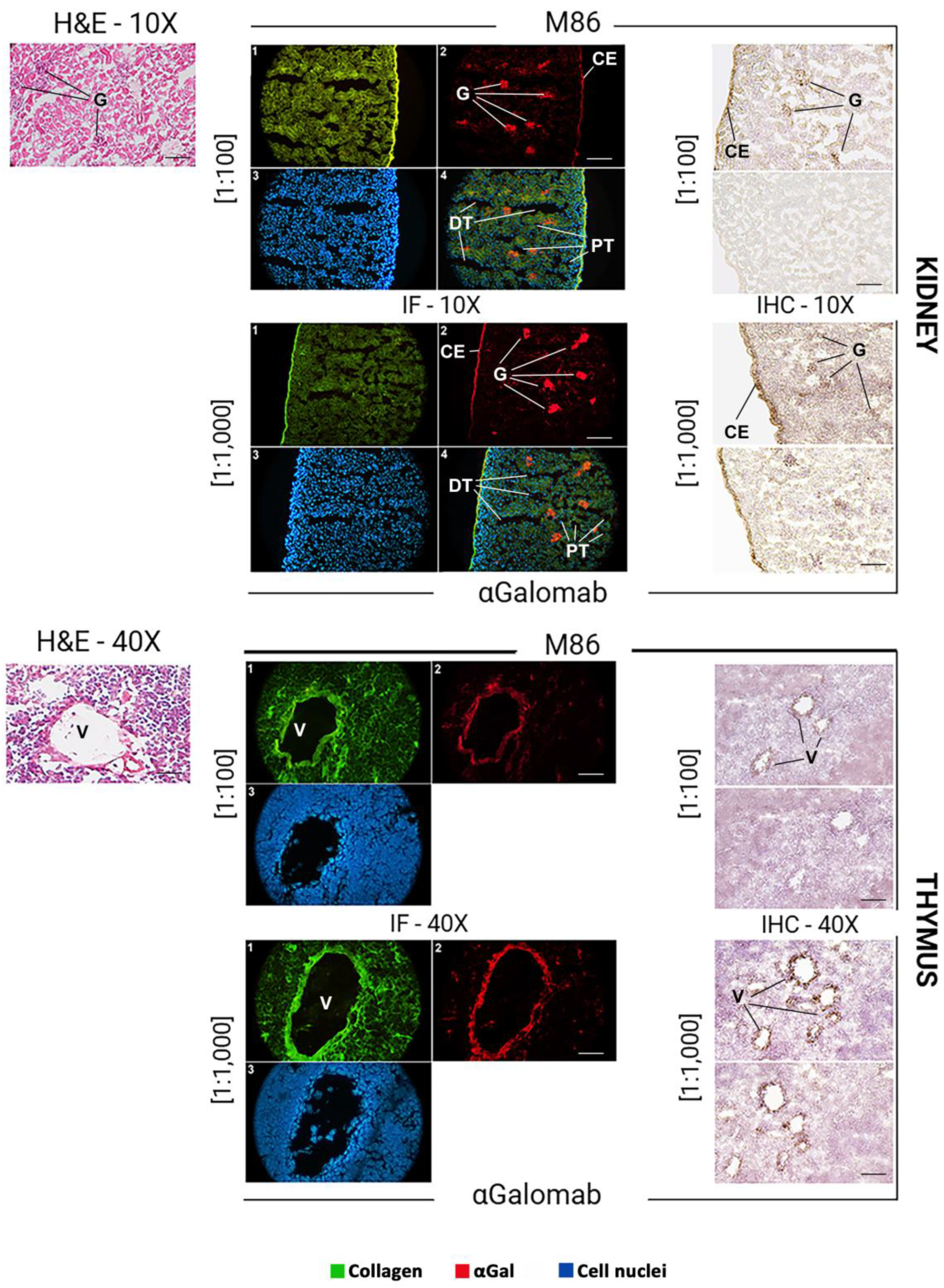
3.1.7. Microscopy
- Immunofluorescence
- Immunohistochemistry
3.2. Tables, figures
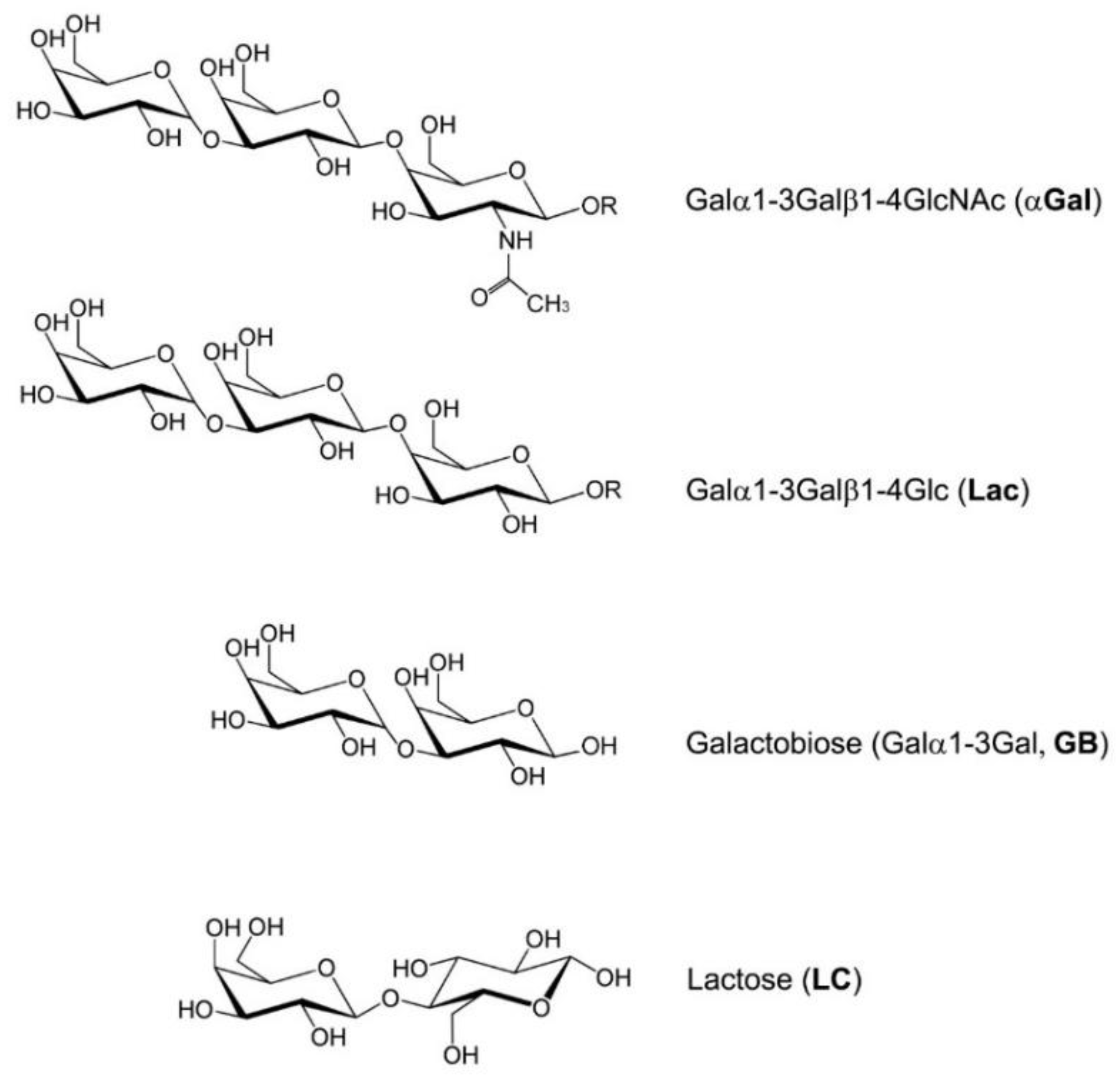
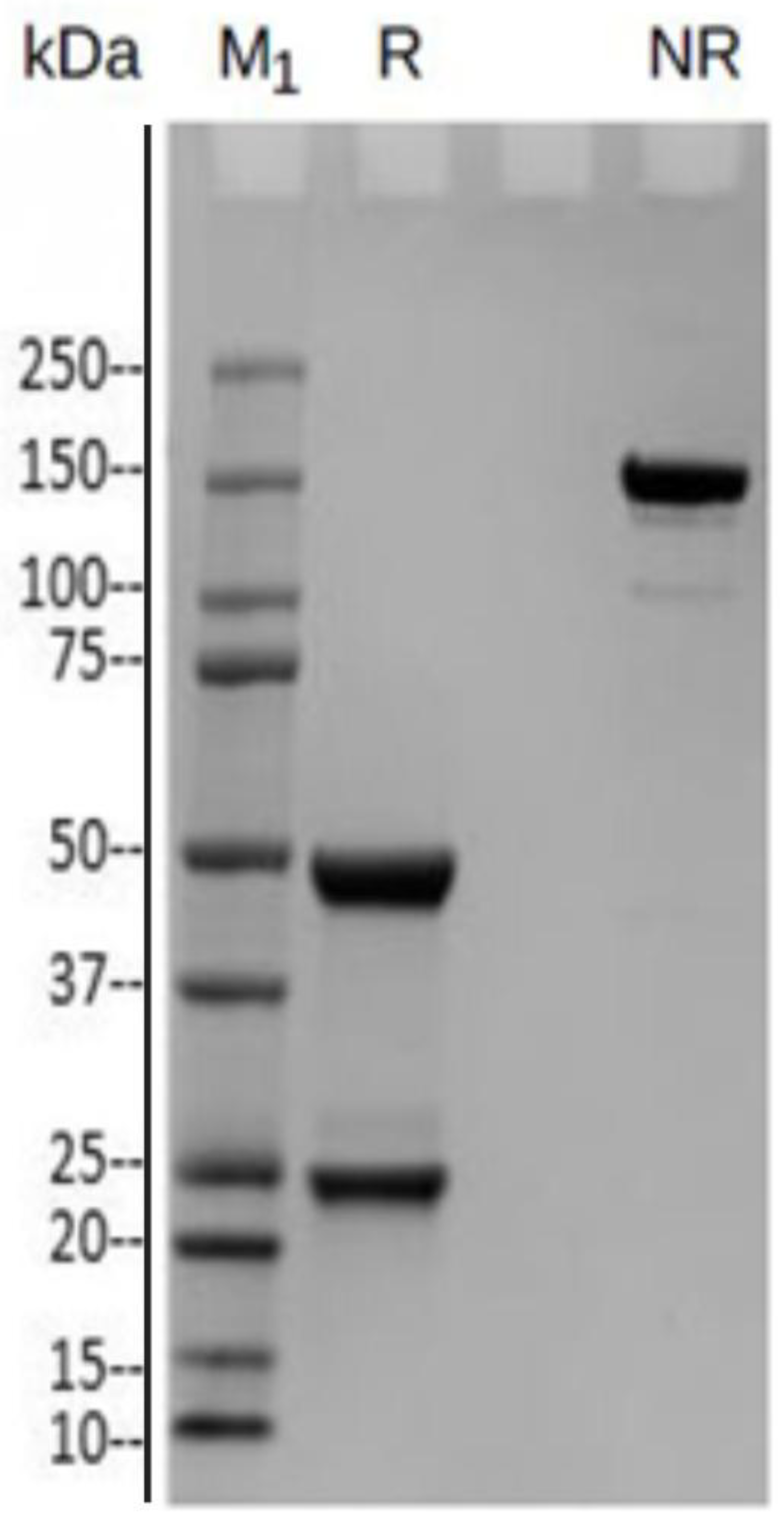
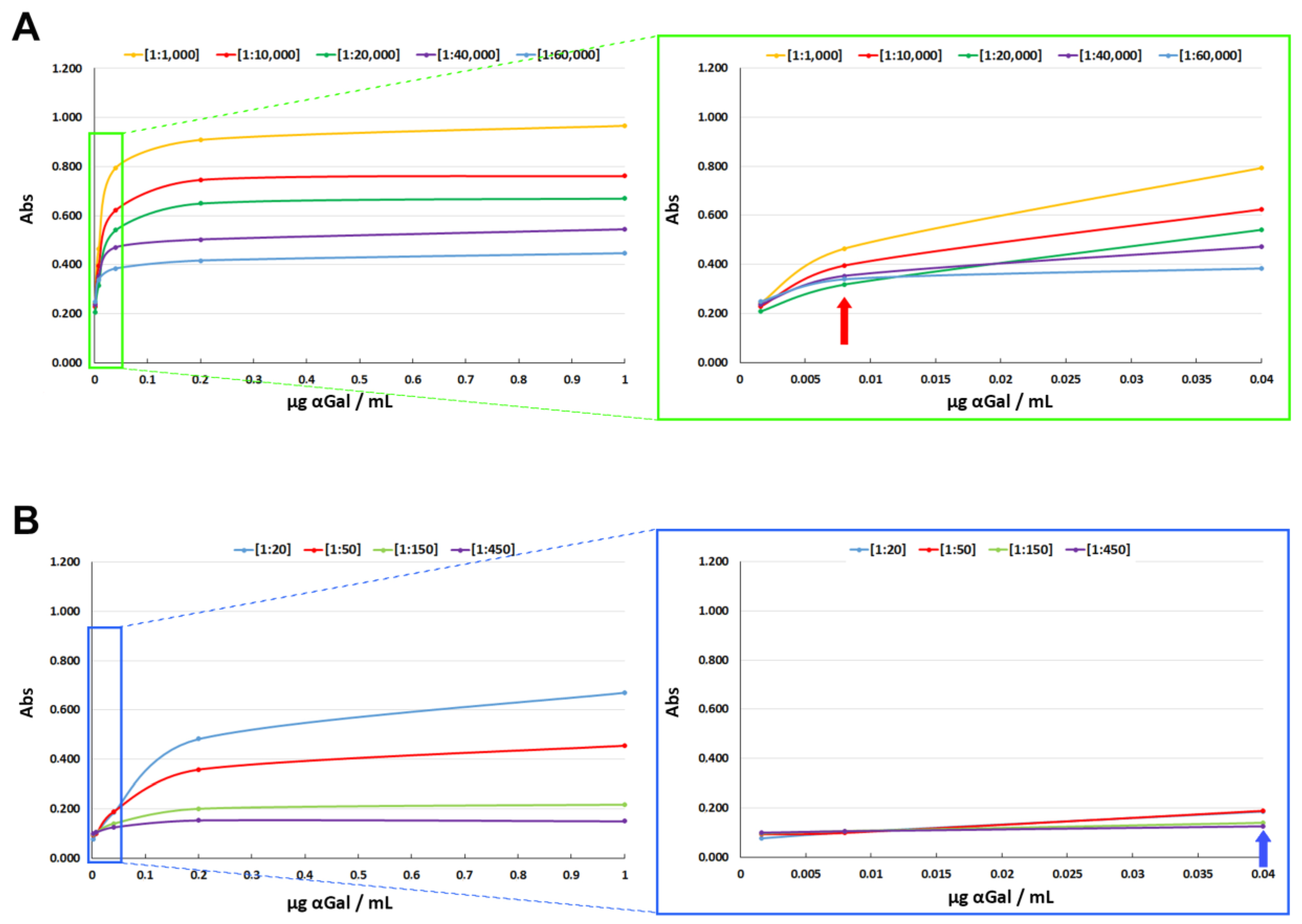
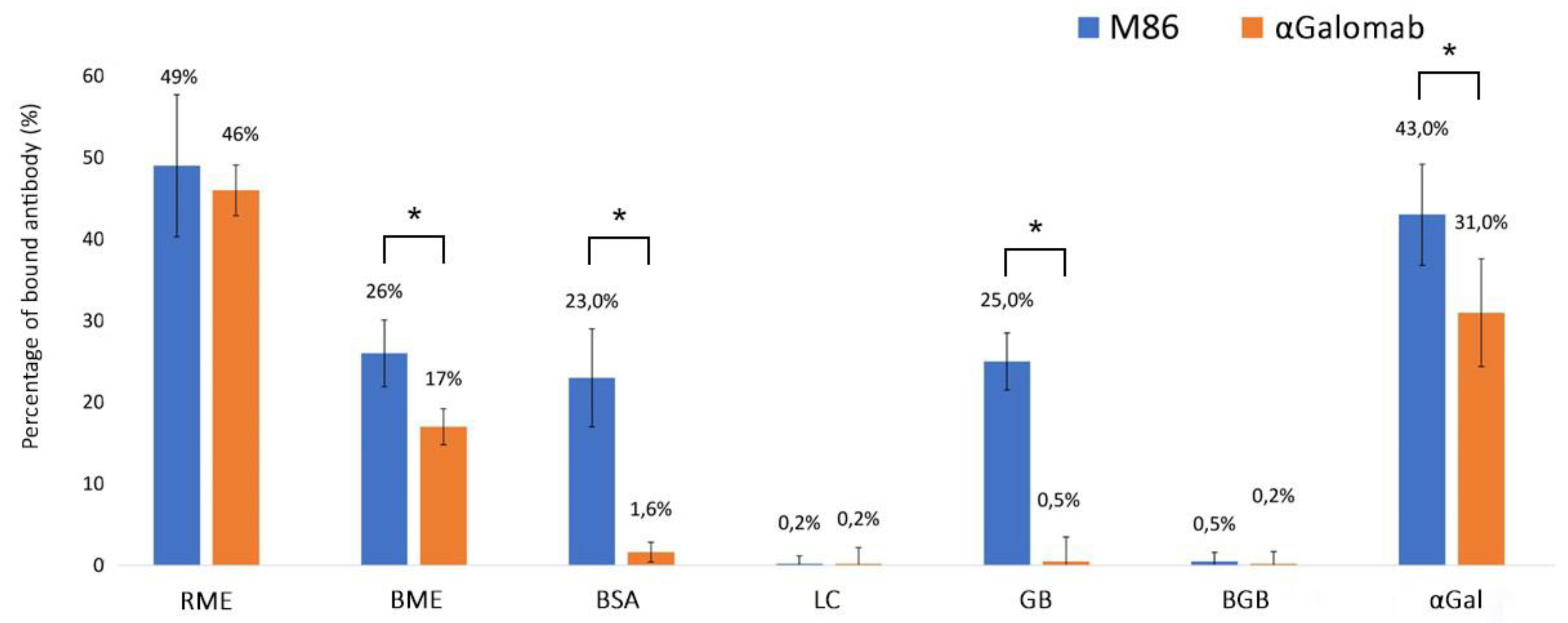
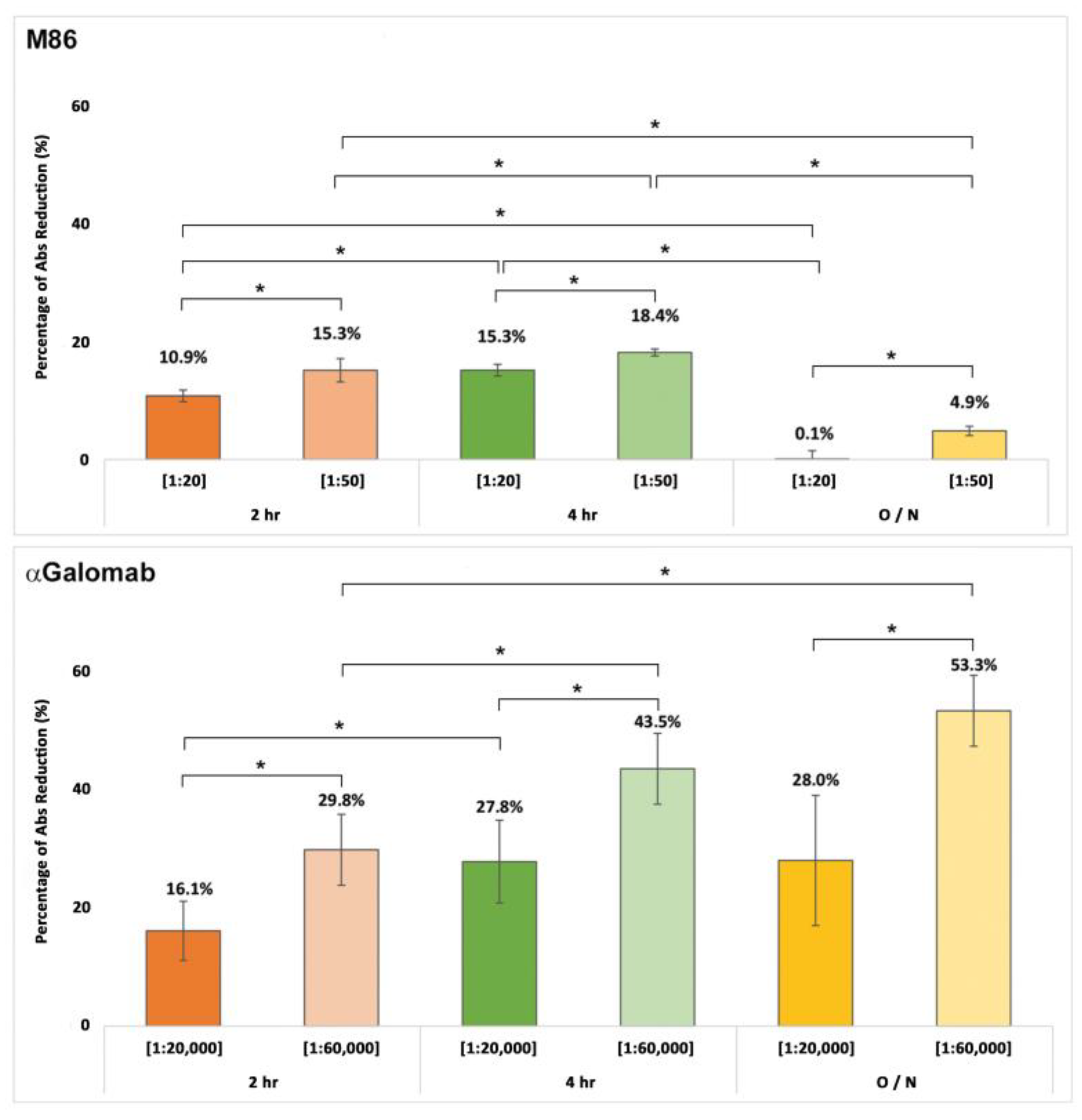
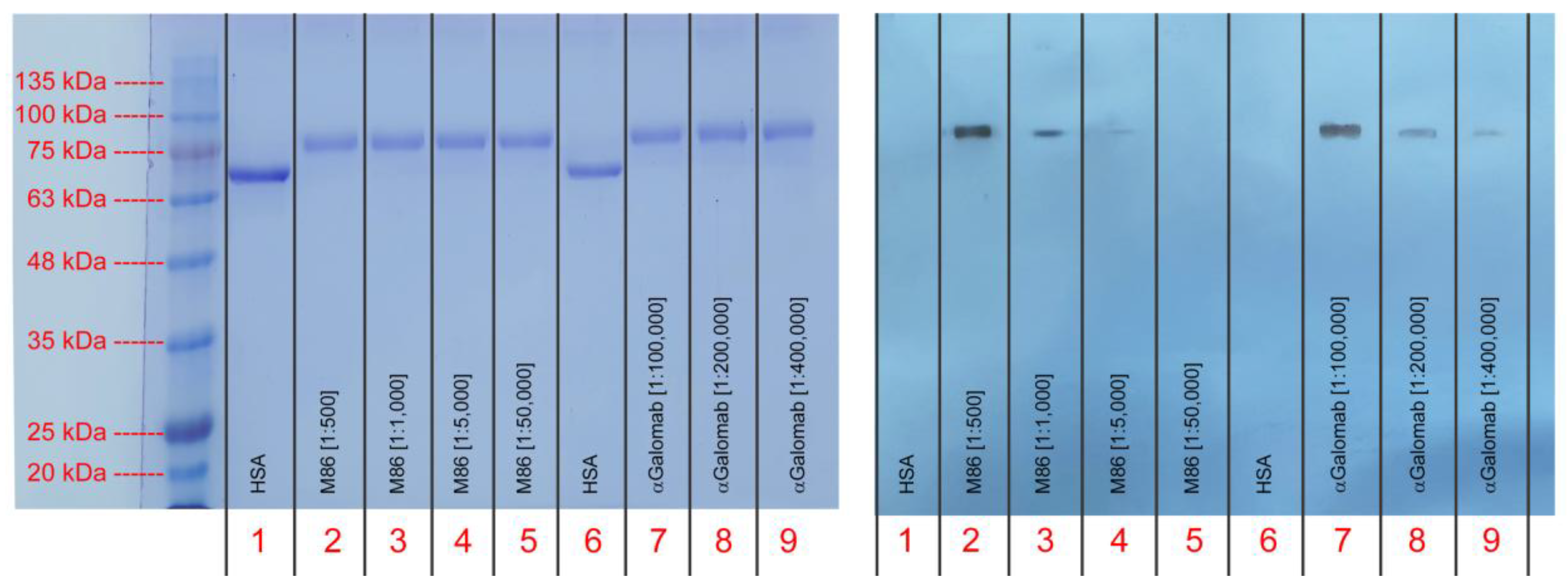
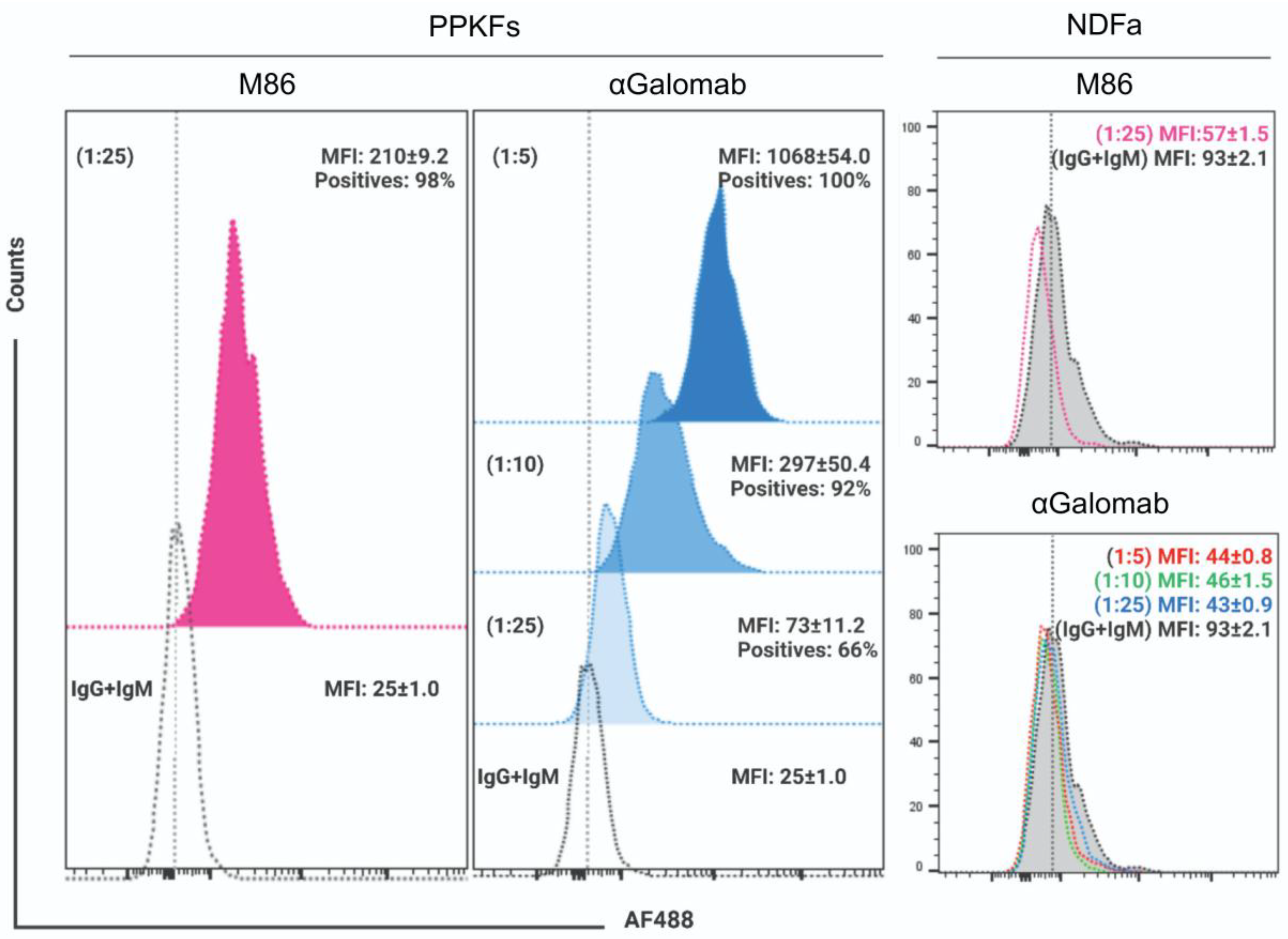
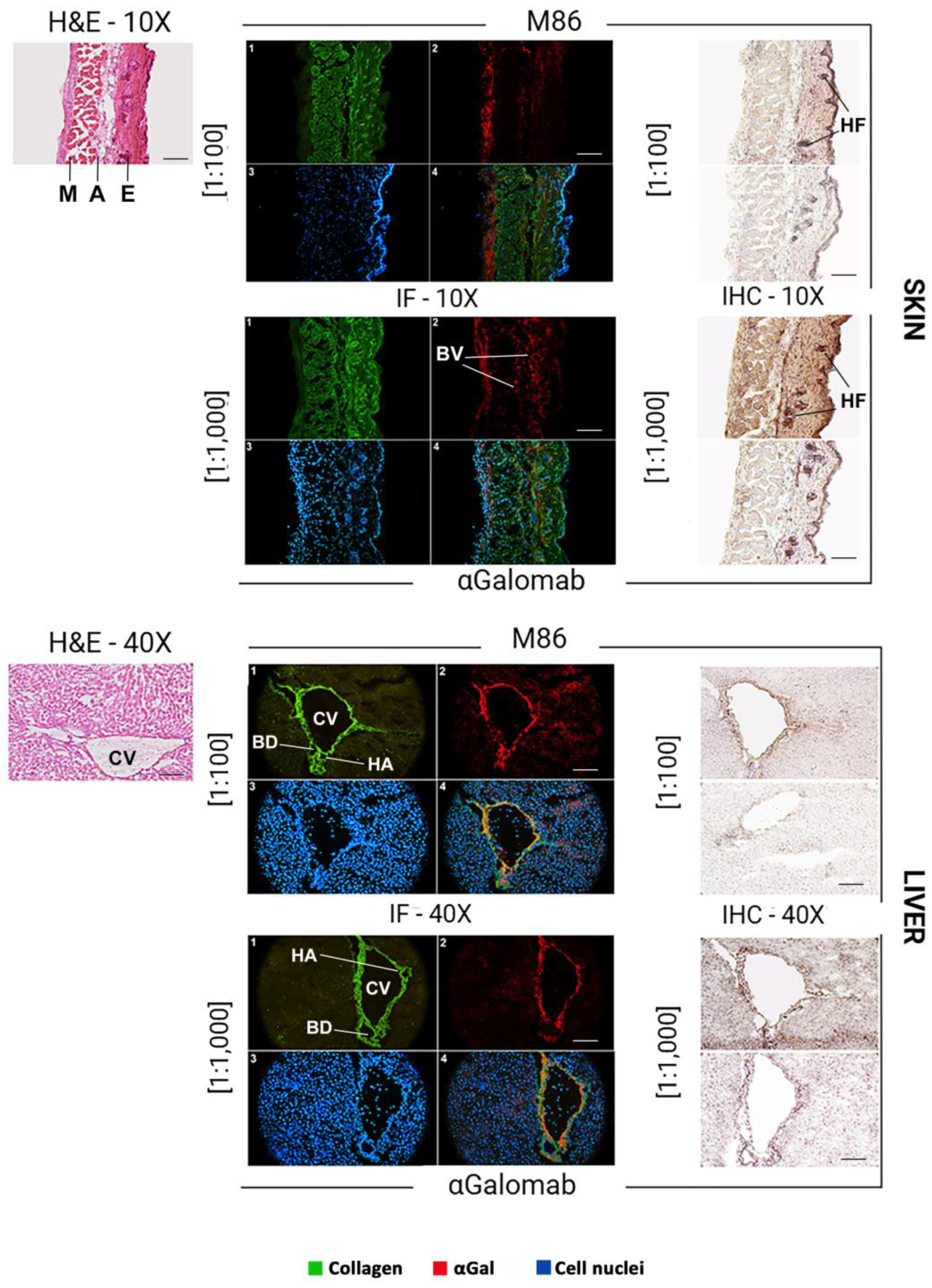
4. Discussion
5. Conclusions
Abbreviations
Author Contributions
Institutional Review Board Statement
Conflicts of Interest
References
- Galili U, Shohet SB, Kobrin E, Stults CLM, Macher BA. Man, apes, and Old World monkeys differ from other mammals in the expression of a-galactosyl epitopes on nucleated cells. J. Biol. Chem. 263, 17755–17762 (1988).
- Hamadeh RM, Galili U, Zhou P, Griffiss, JM. Anti-alpha-galactosyl immunoglobulin A (IgA), IgG, and IgM in human secretions. Clin. Diagn. Lab. Immunol. 2, 125-131 (1995). [CrossRef]
- Montassier E, Al-Ghalith GA, Mathé C, Le Bastard Q, Douillard V, Garnier A et al. Distribution of Bacterial α1,3-Galactosyltransferase Genes in the Human Gut Microbiome. Front. Immunol. 13, 10:3000 (2020). [CrossRef]
- Zhang J, Tao A. Antigenicity, Immunogenicity, Allergenicity. Allergy Bioinformatics. 8, 175–86 (2015). [CrossRef]
- Calafiore AM, Haverich A, Gaudino M, Di Mauro M, Fattouch K, Prapas S et al. Immunoreaction to xenogenic tissue in cardiac surgery: alpha-Gal and beyond. Eur. J. Cardiothorac. Surg. 62, ezac115 (2022). [CrossRef]
- Senage T, Paul A, Le Tourneau T, Fellah-Hebia I, Vadori M, Bashir S, et al. The role of antibody responses against glycans in bioprosthetic heart valve calcification and deterioration. Nat. Med.;28, 283-294 (2022). [CrossRef]
- Veraar C, Koschutnik M, Nitsche C, Laggner M, Polak D, Bohle B, Mangold A, et al. Inflammatory immune response in recipients of transcatheter aortic valves. JTCVS Open. 6, 85-96 (2021). [CrossRef]
- Bozso SJ, El-Andari R, Al-Adra D, Moon MC, Freed DH, Nagendran J, et al. A review of the immune response stimulated by xenogenic tissue heart valves. Scand. J. Immunol. 93, e13018 (2021). [CrossRef]
- Kostyunin AE, Yuzhalin AE, Rezvova MA, Ovcharenko EA, Glushkova TV, Kutikhin AG. Degeneration of Bioprosthetic Heart Valves: Update 2020. J. Am. Heart Assoc. 9, e018506 (2020). [CrossRef]
- Swiontek K, Morisset M, Codreanu-Morel F, Fischer J, Mehlich J, Darsow U, et al. Drugs of porcine origin-A risk for patients with α-gal syndrome? J. Allergy Clin. Immunol. Pract. 7, 1687-1690 (2019). [CrossRef]
- Perusko M, Grundström J, Eldh M, Hamsten C, Apostolovic D, van Hage M. The α-Gal epitope - the cause of a global allergic disease. Front Immunol. 15, 1335911 (2024). [CrossRef]
- Copic D, Bormann D, Direder M, Ankersmit HJ. Alpha-Gal-specific humoral immune response and reported clinical consequence for cardiac valve replacement in patients below 65 years: moving beyond conjecture. Eur. J. Cardiothorac. Surg. 62, ezac227 (2022). [CrossRef]
- https://www.fda.gov/news-events/press-announcements/fda-approves-first-its-kind-intentional-genomic-alteration-line-domestic-pigs-both-human-food.
- Galili U, LaTemple DC, Radic MZ. A sensitive assay for measuring alpha-Gal epitope expression on cells by a monoclonal anti-Gal antibody. Transplantation 65, 1129-1132 (1998). [CrossRef]
- Naso F, Gandaglia A, Iop L, Spina M, Gerosa G. First quantitative assay of alpha-Gal in soft tissues: presence and distribution of the epitope before and after cell removal from xenogeneic heart valves. Acta Biomater. 7, 1728-1734 (2011). [CrossRef]
- Naso F, Gandaglia A, Bottio T, Tarzia V, Nottle MB, d'Apice AJ. First quantification of alpha-Gal epitope in current glutaraldehyde-fixed heart valve bioprostheses. Xenotransplantation. 20, 252-261 (2013). [CrossRef]
- Cunningham S, Starr E, Shaw I, Glavin J, Kane M, Joshi L. Development of a convenient competitive ELISA for the detection of the free and protein-bound nonhuman galactosyl-α-(1,3)-galactose epitope based on highly specific chicken single-chain antibody variable-region fragments. Anal. Chem. 5, 949-955 (2013). [CrossRef]
- Kreft L, Schepers A, Hils M, Swiontek K, Flatley A, Janowski R, et al. A novel monoclonal IgG1 antibody specific for Galactose-alpha-1,3-galactose questions alpha-Gal epitope expression by bacteria. Front. Immunol. 13:958952 (2022). [CrossRef]
- Naso F, Stefanelli U, Buratto E, Lazzari G, Perota A, Galli C, et al. Alpha-Gal Inactivated Heart Valve Bioprostheses Exhibit an Anti-Calcification Propensity Similar to Knockout Tissues<sup/>. Tissue Eng Part A. 2017 Oct;23(19-20):1181-1195. [CrossRef]
- Naso F, Colli A, Zilla P, Calafiore AM, Lotan C, Padalino MA, et al. Correlations between the alpha-Gal antigen, antibody response and calcification of cardiac valve bioprostheses: experimental evidence obtained using an alpha-Gal knockout mouse animal model. Front Immunol. 2023 Jun 21;14:1210098. [CrossRef]
- Naso F, Gandaglia A, Sturaro G, Galli C, Melder RJ. The α-Gal KO Mouse Animal Model is a Reliable and Predictive Tool for the Immune-Mediated Calcification Assessment of Heart Valve Bioprostheses. Front Biosci (Landmark Ed). 2024 May 10;29(5):181. [CrossRef]
- Köhler G, Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 7, 495-497 (1975). [CrossRef]
- Stacchino C, Bona G, Bonetti F, Rinaldi S, Della Ciana L, Grignani A. Detoxification process for glutaraldehyde treated bovine pericardium: biological, chemical and mechanical characterization. J. Heart Valve Dis. 7, 190 (1998).
- Kirley TL, Norman AB. Unfolding of IgG domains detected by non-reducing SDS-PAGE. Biochem. Biophys. Res. Commun. 503, 944-949 (2018). [CrossRef]
- Agostino M, Sandrin MS, Thompson PE, Yuriev E, Ramsland PA. In silico analysis of antibody-carbohydrate interactions and its application to xenoreactive antibodies. Mol. Immunol. 47, 233-246 (2009). [CrossRef]
- Agostino M, Sandrin MS, Thompson PE, Yuriev E, Ramsland PA. Identification of preferred carbohydrate binding modes in xenoreactive antibodies by combining conformational filters and binding site maps. Glycobiology. 20, 724-735 (2010). [CrossRef]
- Strahan KM, Gu F, Preece AF, Gustavsson I, Andersson L, Gustafsson K. cDNA sequence and chromosome localization of pig alpha 1,3 galactosyltransferase. Immunogenetics 41, 101-105 (1995). [CrossRef]
- Milland J, Christiansen D, Lazarus BD, Taylor SG, Xing PX, Sandrin MS. The molecular basis for galalpha(1,3)gal expression in animals with a deletion of the alpha1,3galactosyltransferase gene. J. Immunol. 176, 2448-2454 (2006). [CrossRef]
- Anraku K, Shun S, Nicholas TJ, Lisa ME, Beverly AE, Kim DJ. The design and synthesis of an α-Gal trisaccharide epitope that provides a highly specific anti-Gal immune response. Org. Biomol. Chem. 15, 2979-2992 (2017). [CrossRef]
- Sharon, N. Lectins: carbohydrate-specific reagents and biological recognition molecules. J. Biol. Chem. Feb 2;282, 2753-2764 (2007). [CrossRef]
- Natchiar SK, Srinivas O, Mitra N, Surolia A, Jayaraman N, Vijayan M. Structural studies on peanut lectin complexed with disaccharides involving different linkages: further insights into the structure and interactions of the lectin. Acta Crystallogr. D. Biol. Crystallogr. 62, 1413-1421 (2006). [CrossRef]
- Blanchard B, Nurisso A, Hollville E, Tétaud C, Wiels J, Pokorná M, et. al. Structural basis of the preferential binding for globo-series glycosphingolipids displayed by Pseudomonas aeruginosa lectin I. J. Mol. Biol. 383, 837-853 (2008). [CrossRef]
- Walser PJ, Haebel PW, Künzler M, Sargent D, Kües U, Aebi M. Structure and functional analysis of the fungal galectin CGL2. Structure. 12, 689-702 (2004). [CrossRef]
- Greco A, Ho JG, Lin SJ, Palcic MM, Rupnik M, Ng KK. Carbohydrate recognition by Clostridium difficile toxin A. Nat. Struct. Mol. Biol. 13, 460-461 (2006). [CrossRef]
- Grahn E, Askarieh G, Holmner A, Tateno H, Winter HC, Goldstein IJ, et al. Crystal structure of the Marasmius oreades mushroom lectin in complex with a xenotransplantation epitope. J. Mol. Biol. 369, 710-721 (2007). [CrossRef]
- Keyt BA, Baliga R, Sinclair AM, Carroll SF, Peterson MS. Structure, Function, and Therapeutic Use of IgM Antibodies. Antibodies (Basel). 9, 53 (2020). [CrossRef]
- Schroeder HW Jr, Cavacini L. Structure and function of immunoglobulins. J. Allergy Clin. Immunol. 125, S41-52 (2010). [CrossRef]
- Klaus T, Stalińska K, Czaplicki D, Mak P, Skupien-Rabian B, Kedracka-Krok S, et al. Mouse Antibody of IgM Class is Prone to Non-Enzymatic Cleavage between CH1 and CH2 Domains. Sci Rep. 8, 519 (2018). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




