Submitted:
01 October 2025
Posted:
02 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animal Model
2.2. Study Protocol
2.3. Hemodynamic Measurements and Sampling of Materials
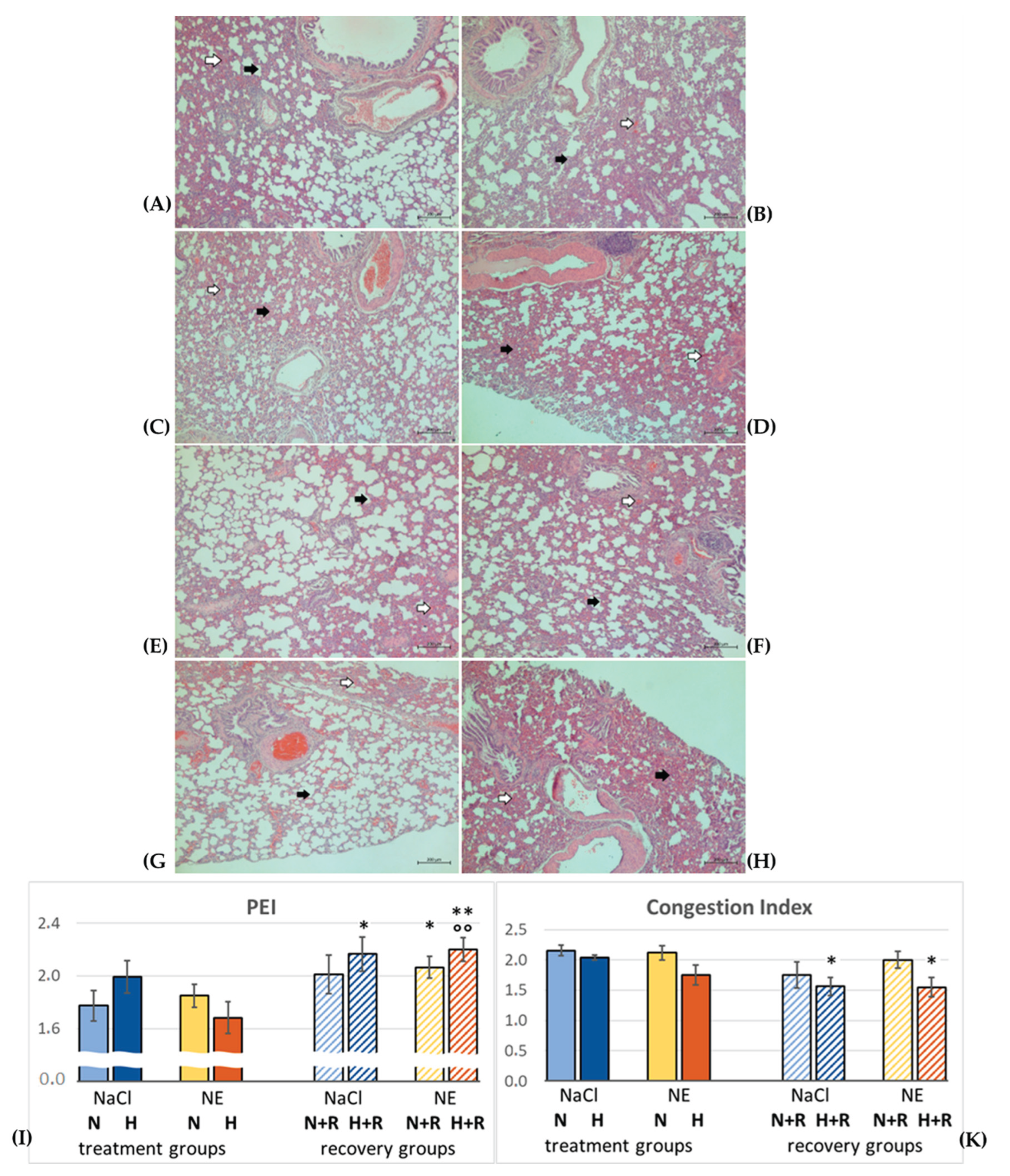
2.4. Lung Histology
- 0, no PE
- 1, mild PE: alveolar septa slightly thickened, alveolar space well defined
- 2, moderate PE: thickness of alveolar septa about twice the normal width, alveolar space narrowed but still defined
- 3, severe PE: alveolar spaces hardly determinable and/or alveolar edema.
- 0, no congestion
- 1, mild congestion, in the interstitium only
- 2, moderate congestion, alveolar walls affected as well
- 3, severe congestion, hemorrhage, erythrocytes in the alveolar spaces.
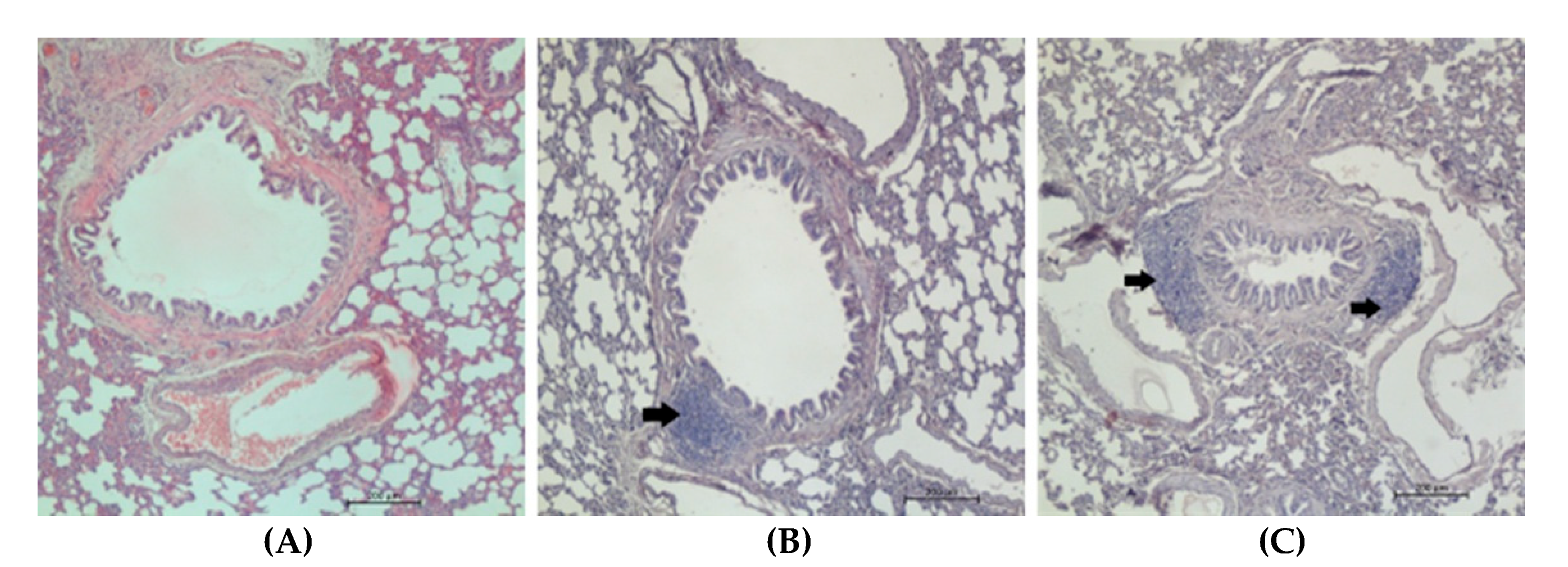
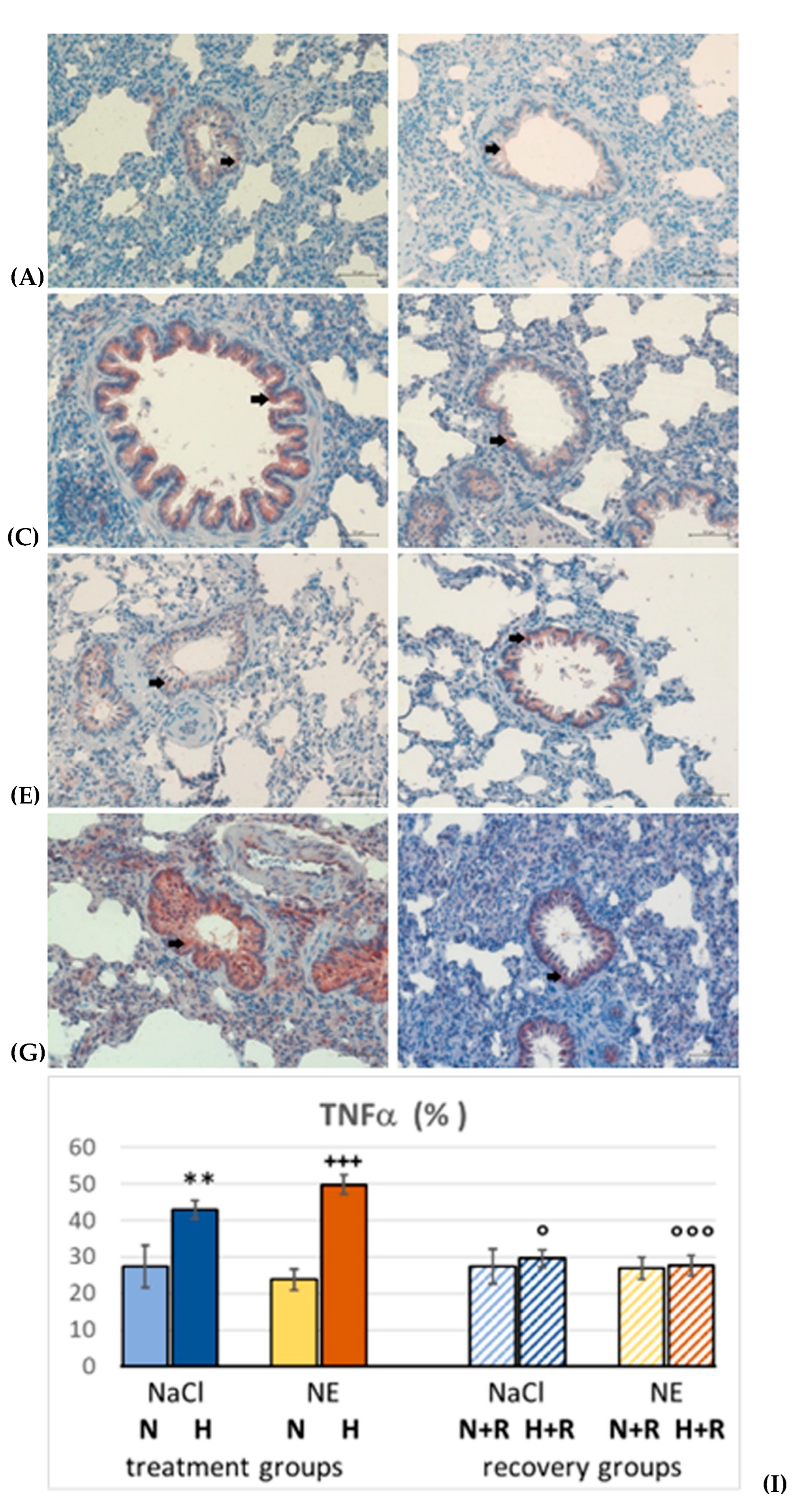
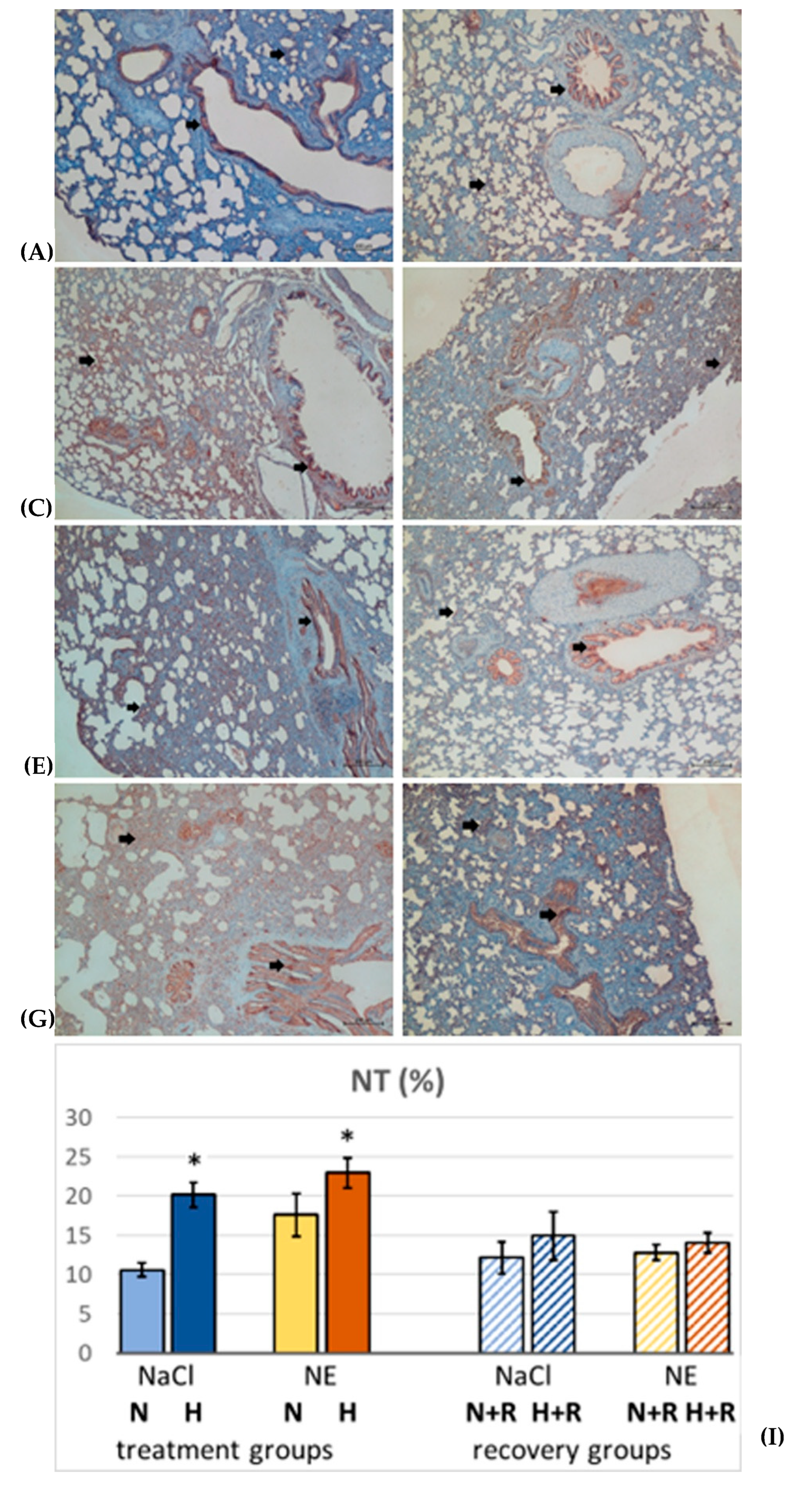
2.5. Immunohistochemistry
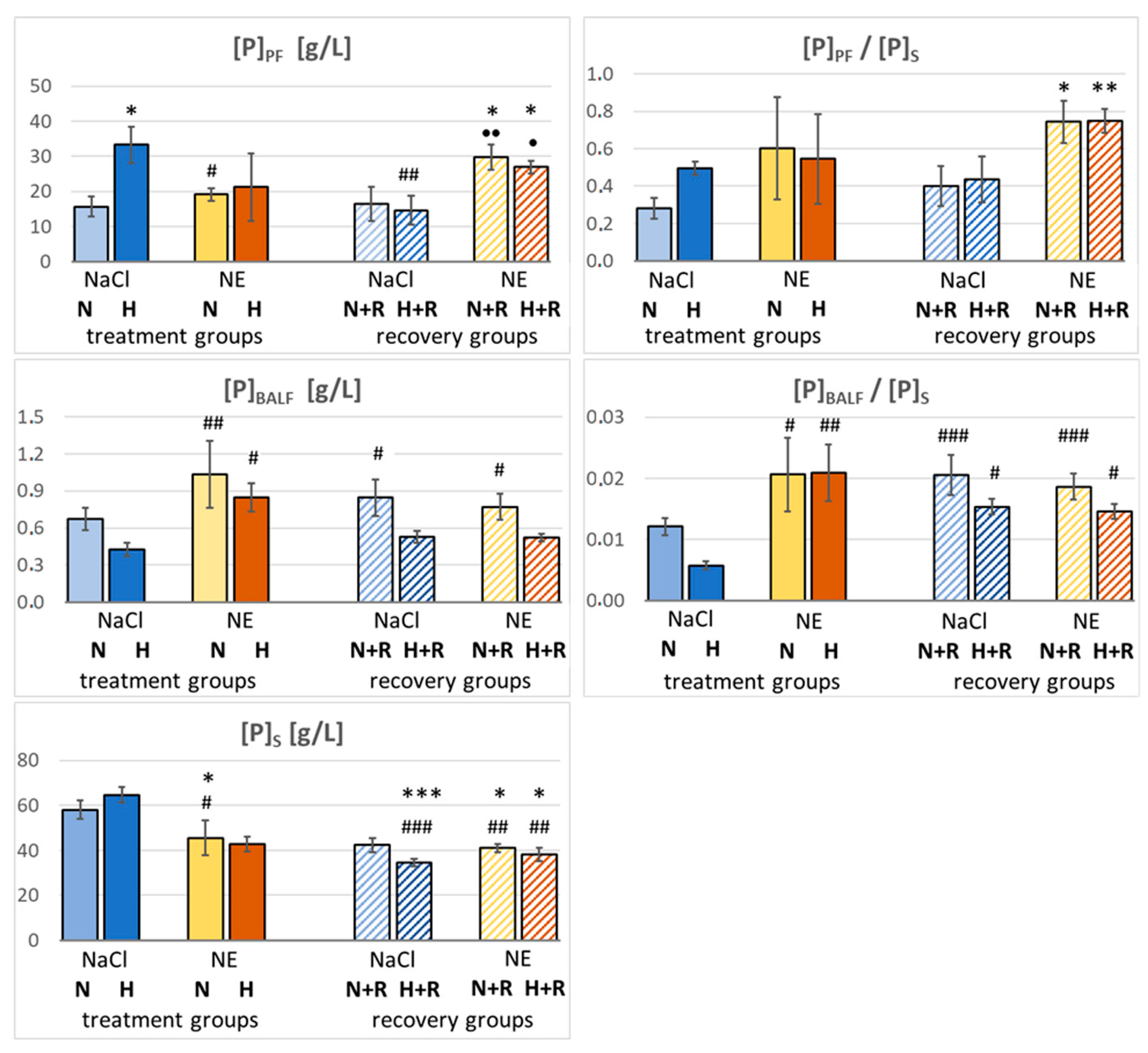
2.6. Protein Concentration in Serum, BALF and PF
2.7. Statistical Analysis
3. Results
3.1. Pulmonary Injury
3.1.1. Pulmonary Edema
3.1.2. Pulmonary Inflammation
3.1.3. Oxidative Stress in the Lung
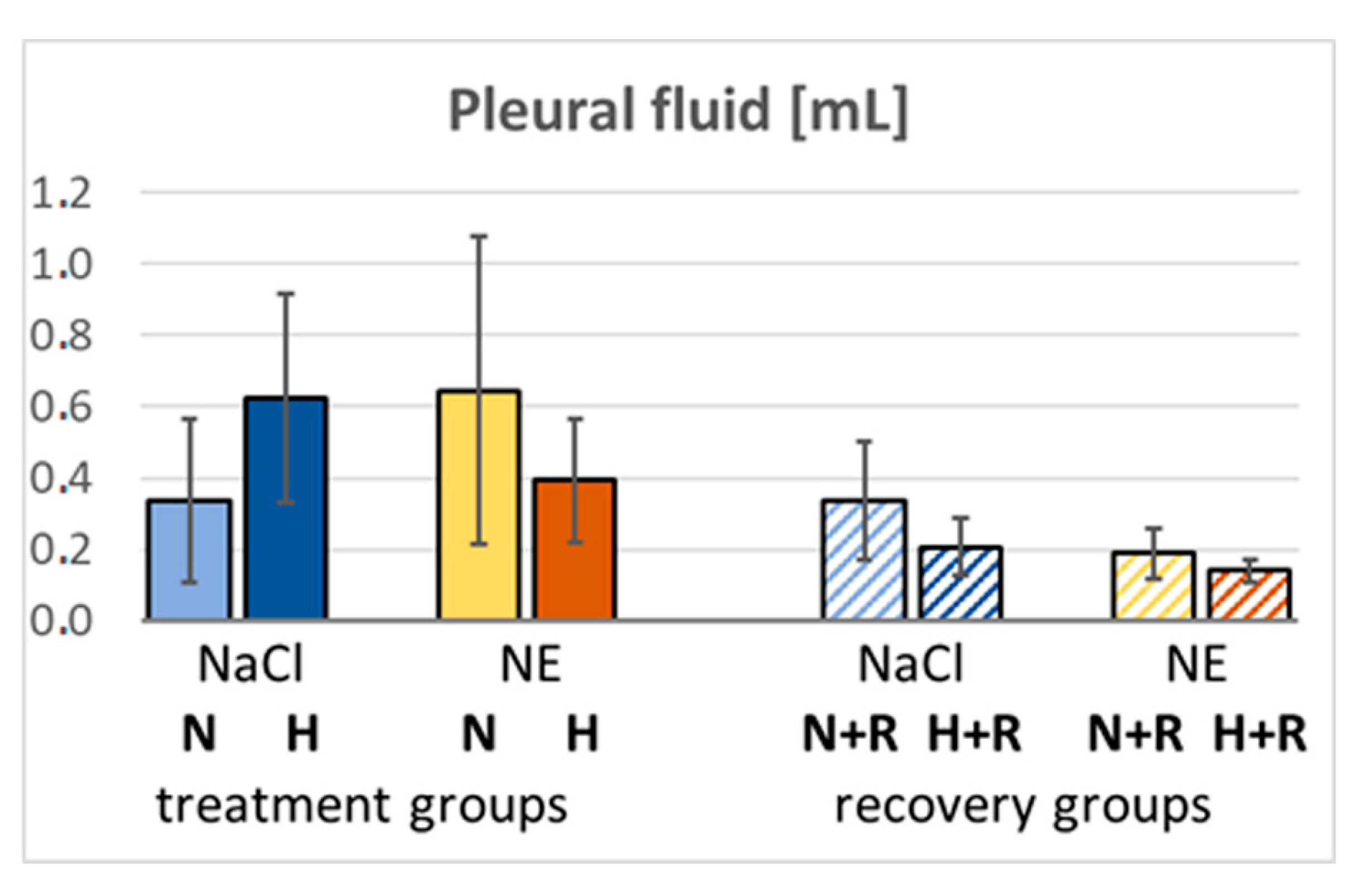
3.2. Pleural Fluid and BALF
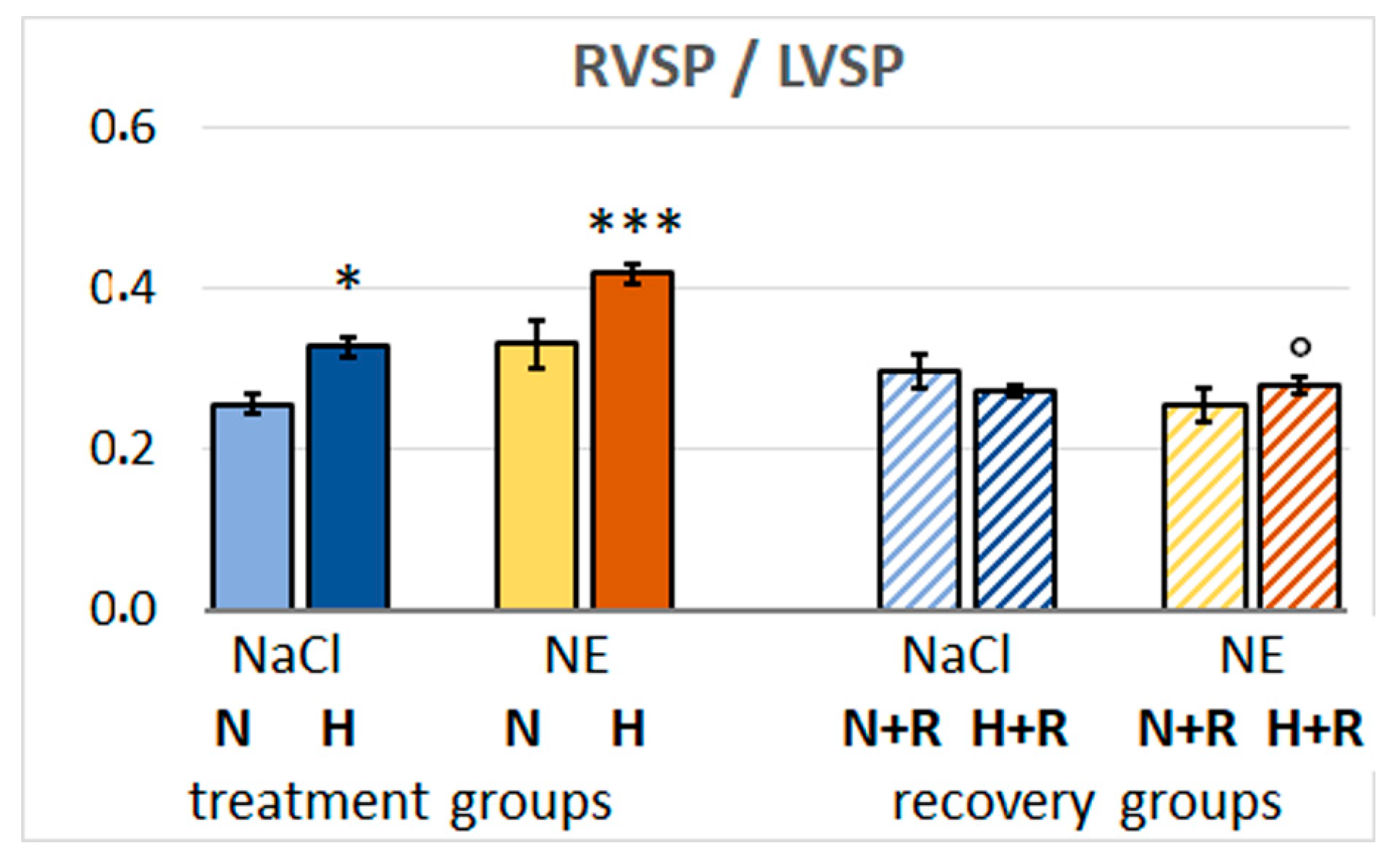
3.3. Hemodynamic Results
4. Discussion
4.1. The Effects of Hemodynamic Changes due to Hypoxia and NE Application
4.2. The Role of Inflammation and Oxidative Stress
4.3. The Protein Content in PF and BALF
4.4. Limitations to the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kubo, K.; Hanaoka, M.; Hayano, T.; Miyahara, T.; Hachiya, T.; Hayasaka, M.; Koizumi T; Fujimoto, K. ; Kobayashi, T.; Honda, T. Inflammatory cytokines in BAL fluid and pulmonary hemodynamics in high-altitude pulmonary edema. Respir. Physiol. 1998, 111, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Madjdpour, C.; Jewell, U.R.; Kneller, S.; Ziegler, U.; Schwendener, R.; Booy, C.; Kläusli, L.; Pasch, T.; Schimmer, R.C.; Beck-Schimmer, B. Decreased alveolar oxygen induces lung inflammation. Am. J. Physiol. Lung Cell Mol. Physiol. 2003, 284, L360–L367. [Google Scholar] [CrossRef] [PubMed]
- Kowalleck, U.; Ahmed, M.A.A.; Koedel, J.; Schierle, K.; Salameh, A.; Rassler, B. Relaxin does not prevent development of hypoxia-induced pulmonary edema in rats. Pflugers Arch. 2022, 474, 1053–1067. [Google Scholar] [CrossRef] [PubMed]
- Riha, I.; Salameh, A.; Hoschke, A.; Raffort, C.; Koedel, J.; Rassler, B. Hypoxia-Induced Pulmonary Injury-Adrenergic Blockade Attenuates Nitrosative Stress, and Proinflammatory Cytokines but Not Pulmonary Edema. J. Cardiovasc. Dev. Dis. 2024, 11, 195. [Google Scholar] [CrossRef]
- Swenson, E.R.; Maggiorini, M.; Mongovin, S.; Gibbs, J.S.; Greve, I.; Mairbäurl, H.; Bärtsch, P. Pathogenesis of high-altitude pulmonary edema: inflammation is not an etiologic factor. JAMA 2002, 287, 2228–2235. [Google Scholar] [CrossRef]
- Berger, M.M.; Hesse, C.; Dehnert, C.; Siedler, H.; Kleinbongard, P.; Bardenheuer, H.J.; Kelm, M.; Bärtsch, P.; Haefeli, W.E. Hypoxia impairs systemic endothelial function in individuals prone to high-altitude pulmonary edema. Am. J. Respir. Crit. Care Med. 2005, 172, 763–767. [Google Scholar] [CrossRef]
- Bärtsch, P.; Gibbs, J.S. Effect of altitude on the heart and the lungs. Circulation 2007, 116, 2191–2202. [Google Scholar] [CrossRef]
- Scherrer, U.; Rexhaj, E.; Jayet, P.Y.; Allemann, Y.; Sartori, C. New insights in the pathogenesis of high-altitude pulmonary edema. Prog. Cardiovasc. Dis. 2010, 52, 485–492. [Google Scholar] [CrossRef]
- Bärtsch, P.; Mairbäurl, H.; Maggiorini, M.; Swenson, E.R. Physiological aspects of high-altitude pulmonary edema. J. Appl. Physiol. (1985) 2005, 98, 1101–1110. [Google Scholar] [CrossRef]
- Broaddus, V.C.; Wiener-Kronish, J.P.; Berthiaume, Y.; Staub, N.C. Removal of pleural liquid and protein by lymphatics in awake sheep. J. Appl. Physiol. (1985) 1988, 64, 384–390. [Google Scholar] [CrossRef]
- Broaddus, V.C.; Wiener-Kronish, J.P.; Staub, N.C. Clearance of lung edema into the pleural space of volume-loaded anesthetized sheep. J. Appl. Physiol. (1985) 1990, 68, 2623–2630. [Google Scholar] [CrossRef]
- Miserocchi, G. Mechanisms controlling the volume of pleural fluid and extravascular lung water. Eur. Respir. Rev. 2009, 18, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Beretta, E.; Romanò, F.; Sancini, G.; Grotberg, J.B.; Nieman, G.F.; Miserocchi, G. Pulmonary Interstitial Matrix and Lung Fluid Balance From Normal to the Acutely Injured Lung. Front. Physiol. 2021, 12, 781874. [Google Scholar] [CrossRef]
- Maggiorini, M. Prevention and treatment of high-altitude pulmonary edema. Prog. Cardiovasc. Dis. 2010, 52, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, E.N.; Ciriolo, M.R.; Ciccarone, F. Hypoxia-Induced Reactive Oxygen Species: Their Role in Cancer Resistance and Emerging Therapies to Overcome It. Antioxidants (Basel) 2025, 14, 94. [Google Scholar] [CrossRef]
- Yuan, G.; Khan, S.A.; Luo, W.; Nanduri, J.; Semenza, G.L.; Prabhakar, N.R. Hypoxia-inducible Factor 1 Mediates Increased Expression of NADPH Oxidase-2 in Response to Intermittent Hypoxia. J. Cell. Physiol. 2011, 226, 2925–2933. [Google Scholar] [CrossRef] [PubMed]
- Knepler, J.L. Jr.; Taher, L.N.; Gupta, M.P.; Patterson, C.; Pavalko, F.; Ober, M.D.; Hart, C.M. Peroxynitrite causes endothelial cell monolayer barrier dysfunction. Am. J. Physiol. Cell Physiol. 2001, 281, C1064–C1075. [Google Scholar] [CrossRef]
- Salameh, A.; Zöbisch, H.; Schröder, B.; Vigelahn, J.; Jahn, M.; Abraham, G.; Seeger, J.; Dähnert, I.; Dhein, S. Effects of Hypoxia and Acidosis on Cardiac Electrophysiology and Hemodynamics. Is NHE-Inhibition by Cariporide Still Advantageous? Front. Physiol. 2020, 11, 224. [Google Scholar] [CrossRef]
- Chelombitko, M.A. Role of Reactive Oxygen Species in Inflammation: A Minireview. Moscow Univ. Biol. Sci. Bull. 2018, 73, 199–202. [Google Scholar] [CrossRef]
- Dai, S.; Xue, Q.; Sun, R.; Wang, S.; Li, C.; Wu, Y.; Si, Q.; Hu, S. Hemodynamic and nonhemodynamic mechanisms of experimental pulmonary edema in rats and the effect of anisodamine and tetramethylpyrazine. Part 1: Survival rate, pulmonary index, pathological change and pulmonary vascular permeability. Chin. Med. Sci. J. 1993, 8, 72–76. [Google Scholar]
- Rassler, B.; Barth, W.; Zimmer, H.G. Transient pleural effusion in norepinephrine-stimulated rats. Basic Res. Cardiol. 2001, 96, 471–477. [Google Scholar] [CrossRef]
- Rassler, B.; Reissig, C.; Briest, W.; Tannapfel, A.; Zimmer, H.G. Pulmonary edema and pleural effusion in norepinephrine-stimulated rats--hemodynamic or inflammatory effect? Mol. Cell. Biochem. 2003, 250, 55–63. [Google Scholar] [CrossRef]
- Simmons, R.L.; Martin, A.M. Jr.; Heisterkamp, C.A. 3rd.; Ducker, T.B. Respiratory insufficiency in combat casualties. II. Pulmonary edema following head injury. Ann. Surg. 1969, 170, 39–44. [Google Scholar] [CrossRef]
- Sedý, J.; Zicha, J.; Nedvídková, J.; Kunes, J. The role of sympathetic nervous system in the development of neurogenic pulmonary edema in spinal cord-injured rats. J. Appl. Physiol. (1985) 2012, 112, 1–8. [Google Scholar] [CrossRef]
- Dai, J.; Chen, S.J.; Yang, B.S.; Lü, S.M.; Zhu, M.; Xu, Y.F.; Chen, J.; Cai, H.W.; Mao, W. Recurrence of non-cardiogenic pulmonary edema and sustained hypotension shock in cystic pheochromocytoma. J. Zhejiang Univ. Sci. B. 2017, 18, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Y-Hassan, S.; Falhammar, H. Cardiovascular Manifestations and Complications of Pheochromocytomas and Paragangliomas. J. Clin. Med. 2020, 9, 2435. [Google Scholar] [CrossRef]
- Teboul, J.L.; Douguet, D.; Mercat, A.; Depret, J.; Richard, C.; Zelter, M. Effects of catecholamines on the pulmonary venous bed in sheep. Crit. Care Med. 1998, 26, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Appelt, P.; Gabriel, P.; Bölter, C.; Fiedler, N.; Schierle, K.; Salameh, A.; Rassler, B. Left ventricular depression and pulmonary edema in rats after short-term normobaric hypoxia: effects of adrenergic blockade and reduced fluid load. Pflugers Arch. 2021, 473, 1723–1735. [Google Scholar] [CrossRef]
- Bambor, C.; Daunheimer, S.; Raffort, C.; Koedel, J.; Salameh, A.; Raßler, B. Effects of a Three-Day vs. Six-Day Exposure to Normobaric Hypoxia on the Cardiopulmonary Function of Rats. Curr. Issues Mol. Biol. 2025, 47, 125. [Google Scholar] [CrossRef]
- Neubert, E.; Rassler, B.; Hoschke, A.; Raffort, C.; Salameh, A. Effects of Normobaric Hypoxia and Adrenergic Blockade over 72 h on Cardiac Function in Rats. Int. J. Mol. Sci. 2023, 24, 11417. [Google Scholar] [CrossRef]
- Abramoff, M.D.; Magalhaes, P.J.; Ram, S.J. Image processing with ImageJ. Biophotonics Int. 2004, 11, 36–42. [Google Scholar]
- Swenson, E.R. Early hours in the development of high-altitude pulmonary edema: time course and mechanisms. J. Appl. Physiol. (1985) 2020, 128, 1539–1546. [Google Scholar] [CrossRef]
- Magnusdottir, S.O.; Simonsen, C.; Karbing, D.S.; Rasmussen, B.S.; Kjaergaard, B. Hypoxia in the pulmonary vein increases pulmonary vascular resistance independently of oxygen in the pulmonary artery. Animal Model Exp. Med. 2024, 7, 156–165. [Google Scholar] [CrossRef]
- Hopkins, S.R.; Garg, J.; Bolar, D.S.; Balouch, J.; Levin, DL. Pulmonary blood flow heterogeneity during hypoxia and high-altitude pulmonary edema. Am. J. Respir. Crit. Care Med. 2005, 171, 83–87. [Google Scholar] [CrossRef]
- West, J.B.; Tsukimoto, K.; Mathieu-Costello, O.; Prediletto, R. Stress failure in pulmonary capillaries. J. Appl. Physiol. (1985) 1991, 70, 1731–1742. [Google Scholar] [CrossRef]
- West, J.B. Invited review: pulmonary capillary stress failure. J. Appl. Physiol. (1985) 2000, 89, 2483–2489. [Google Scholar] [CrossRef]
- Bölter, C.; Gabriel, P.; Appelt, P.; Salameh, A.; Schierle, K.; Rassler, B. Effects of Adrenergic Agonists and Antagonists on Cardiopulmonary Function During Normobaric Hypoxia in Rat. Front. Physiol. 2019, 10, 860. [Google Scholar] [CrossRef]
- Staub, N.C. New concepts about the pathophysiology of pulmonary edema. J. Thorac. Imaging 1988, 3, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Wiener-Kronish, J.P.; Broaddus, V.C.; Albertine, K.H.; Gropper, M.A.; Matthay, M.A.; Staub, N.C. Relationship of pleural effusions to increased permeability pulmonary edema in anesthetized sheep. J. Clin. Invest. 1988, 82, 1422–1429. [Google Scholar] [CrossRef] [PubMed]
- Rassler, B.; Reissig, C.; Briest, W.; Tannapfel, A.; Zimmer, H.G. Catecholamine-induced pulmonary edema and pleural effusion in rats--alpha- and beta-adrenergic effects. Respir. Physiol. Neurobiol. 2003, 135, 25–37. [Google Scholar] [CrossRef]
- Šedý, J.; Kuneš, J.; Zicha, J. Pathogenetic Mechanisms of Neurogenic Pulmonary Edema. J. Neurotrauma 2015, 32, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Rachfalska, N.; Putowski, Z.; Krzych, J. Distant Organ Damage in Acute Brain Injury. Brain Sci. 2020, 10, 1019. [Google Scholar] [CrossRef]
- Ghezzi, P.; Dinarello, C.A.; Bianchi, M.; Rosandich, M.E.; Repine, J.E.; White, C.W. Hypoxia increases production of interleukin-1 and tumor necrosis factor by human mononuclear cells. Cytokine 1991, 3, 189–194. [Google Scholar] [CrossRef]
- Hartmann, G.; Tschöp, M.; Fischer, R.; Bidlingmaier, C.; Riepl, R.; Tschöp, K.; Hautmann, H.; Endres, S.; Toepfer, M. High altitude increases circulating interleukin-6, interleukin-1 receptor antagonist and C-reactive protein. Cytokine 2000, 12, 246–252. [Google Scholar] [CrossRef]
- Sarada, S.; Himadri, P.; Mishra, C.; Geetali, P.; Ram, M.S.; Ilavazhagan, G. Role of oxidative stress and NFκB in hypoxia-induced pulmonary edema. Exp. Biol. Med. (Maywood) 2008, 233, 1088–1098. [Google Scholar] [CrossRef]
- Araneda, O.F.; Tuesta, M. Lung oxidative damage by hypoxia. Oxid. Med. Cell. Longev. 2012, 2012, 856918. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox. Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef]
- Liu, Y.; Xiang, D.; Zhang, H.; Yao, H.; Wang, Y. Hypoxia-Inducible Factor-1: A Potential Target to Treat Acute Lung Injury. Oxid. Med. Cell. Longev. 2020, 2020, 8871476. [Google Scholar] [CrossRef] [PubMed]
- El Alam, S.; Pena, E.; Aguilera, D.; Siques, P.; Brito, J. Inflammation in Pulmonary Hypertension and Edema Induced by Hypobaric Hypoxia Exposure. Int. J. Mol. Sci. 2022, 23, 12656. [Google Scholar] [CrossRef] [PubMed]
- Beckman, D.L.; Mehta, P.; Hanks, V.; Rowan, W.H.; Liu, L. Effects of peroxynitrite on pulmonary edema and the oxidative state. Exp. Lung Res. 2000, 26, 349–359. [Google Scholar] [CrossRef]
- Neumann, P.; Gertzberg, N.; Vaughan, E.; Weisbrot, J.; Woodburn, R.; Lambert, W.; Johnson, A. Peroxynitrite mediates TNF-alpha-induced endothelial barrier dysfunction and nitration of actin. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 290, L674–L684. [Google Scholar] [CrossRef] [PubMed]
- Caporarello, N.; Ligresti, G. Vascular Contribution to Lung Repair and Fibrosis. Am. J. Respir. Cell. Mol. Biol. 2023, 69, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Elliott, A.R.; Fu, Z.; Tsukimoto, K.; Prediletto, R.; Mathieu-Costello, O.; West, J.B. Short-term reversibility of ultrastructural changes in pulmonary capillaries caused by stress failure. J. Appl. Physiol (1985) 1992, 73, 1150–1158. [Google Scholar] [CrossRef]
- Stenmark, K.R.; Fagan, K.A.; Frid, M.G. Hypoxia-induced pulmonary vascular remodeling: cellular and molecular mechanisms. Circ. Res. 2006, 99, 675–691. [Google Scholar] [CrossRef]
- Su, Y.; Lucas, R.; Fulton, D.J.R.; Verin, A.D. Mechanisms of pulmonary endothelial barrier dysfunction in acute lung injury and acute respiratory distress syndrome. Chin. Med. J. Pulm. Crit. Care Med. 2024, 2, 80–87. [Google Scholar] [CrossRef]
- Jay, S.J. Pleural effusions. 1. Preliminary evaluation--recognition of the transudate. Postgrad. Med. 1986, 80, 164–167. [Google Scholar] [CrossRef]
- Light, R.W.; Macgregor, M.I.; Luchsinger, P.C.; Ball, W.C. Jr. Pleural effusions: the diagnostic separation of transudates and exudates. Ann. Intern. Med. 1972, 77, 507–513. [Google Scholar] [CrossRef]
- Schoene, R.B.; Hackett, P.H.; Henderson, W.R.; Sage, E.H.; Chow, M.; Roach, R.C.; Mills, W.J. Jr.; Martin, T.R. High-altitude pulmonary edema. Characteristics of lung lavage fluid. JAMA 1986, 256, 63–69. [Google Scholar] [CrossRef]
- West, J.B.; Mathieu-Costello, O. Structure.; strength.; failure.; and remodeling of the pulmonary blood-gas barrier. Annu. Rev. Physiol. 1999, 61, 543–572. [Google Scholar] [CrossRef]
- Hopkins, S.R.; Schoene, R.B.; Henderson, W.R.; Spragg, R.G.; Martin, T.R.; West, J.B. Intense exercise impairs the integrity of the pulmonary blood-gas barrier in elite athletes. Am. J. Respir. Crit. Care Med. 1997, 155, 1090–1094. [Google Scholar] [CrossRef]
- Eldridge, M.W.; Braun, R.K.; Yoneda, K.Y.; Walby, W.F. Effects of altitude and exercise on pulmonary capillary integrity: evidence for subclinical high-altitude pulmonary edema. J. Appl. Physiol (1985) 2006, 100, 972–980. [Google Scholar] [CrossRef]
- Duplain, H.; Vollenweider, L.; Delabays, A.; Nicod, P.; Bärtsch, P.; Scherrer, U. Augmented sympathetic activation during short-term hypoxia and high-altitude exposure in subjects susceptible to high-altitude pulmonary edema. Circulation 1999, 99, 1713–1718. [Google Scholar] [CrossRef]
- Rassler, B.; Rohling, M.A.; Reissig, C.; Briest, W.; Tannapfel, A.; Zimmer, H.G. Involvement of cytokines and inflammation in catecholamine-induced pulmonary injury in rats. Vasc. Dis. Prev. 2005, 2, 1–9. [Google Scholar] [CrossRef]
- Haditsch, B.; Roessler, A.; Krisper, P.; Frisch, H.; Hinghofer-Szalkay, H.G.; Goswami, N. Volume regulation and renal function at high altitude across gender. PLoS One 2015, 10, e0118730. [Google Scholar] [CrossRef]
- Hildebrandt, W.; Ottenbacher, A.; Schuster, M.; Swenson, E.R.; Bärtsch, P. Diuretic effect of hypoxia, hypocapnia, and hyperpnea in humans: relation to hormones and O(2) chemosensitivity. J. Appl. Physiol (1985) 2000, 88, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Bärtsch, P.; Swenson, E.R.; Paul, A.; Jülg, B.; Hohenhaus, E. Hypoxic ventilatory response, ventilation, gas exchange, and fluid balance in acute mountain sickness. High Alt. Med. Biol. 2002, 3, 361–376. [Google Scholar] [CrossRef]
- Mortola, J.P.; Saiki, C. Ventilatory response to hypoxia in rats: Gender differences. Respir. Physiol. 1996, 106, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Dzhalilova, D.; Makarova, O. Differences in Tolerance to Hypoxia: Physiological, Biochemical, and Molecular-Biological Characteristics. Biomedicines 2020, 8, 428. [Google Scholar] [CrossRef]
- Pichler Hefti, J.; Jean, D.; Rosier, A.J.; Derstine, M.; Hillebrandt, D.; Horakova, L.; Keyes, L.E.; Mateikaite-Pipiriene, K.; Paal, P.; Andjelkovic, M.; Beidlemann, B.A.; Kriemler, S. High-Altitude Pulmonary Edema in Women: A Scoping Review-UIAA Medical Commission Recommendations. High Alt. Med. Biol. 2023, 24, 268–273. [Google Scholar] [CrossRef]
- West, J. B; American College of Physicians; American Physiological Society. The physiologic basis of high-altitude diseases. Ann. Intern. Med. 2004, 141, 789–800. [Google Scholar] [CrossRef]
| --------------Treatment groups -------------- | --------------Recovery groups -------------- | |||||||
|---|---|---|---|---|---|---|---|---|
| N-NaCl | H-NaCl | N-NE | H-NE | N-NaCl +R |
H-NaCl +R |
N-NE +R |
H-NE +R |
|
| LVSP [mmHg] | 123.3 ± 3.4 # |
103.5 ± 3.8 * |
106.7 ± 4.1 * |
92.1 ± 3.4 *#+ |
102.0 ± 5.5 $° |
123.8 ± 3.8 §° |
131.1 ± 3.8 #§° |
120.9 ± 3.3 #§° |
| RVSP [mmHg] | 30.9 ± 0.9 |
34.3 ± 1.5 |
34.5 ± 2.4 |
38.5 ± 1.5 |
30.1 ± 2.7 |
33.9 ± 1.2 |
33.0 ± 1.8 |
33.7 ± 1.1 |
| MAP [mmHg] | 109.6 ± 3.5 # |
91.4 ± 3.6 * |
94.7 ± 4.0 * |
77.9 ± 3.0 *#+ |
91.4 ± 5.6 $° |
111.9 ± 3.2 §° |
117.2 ± 3.5 #§° |
108.0 ± 2.9 #§° |
| SV [µL] | 215.3 ± 7.2 |
185.6 ± 11.6 | 161.6 ± 11.5 * | 131.2 ± 20.0 *#+ |
215.5 ± 13.9 | 214.2 ± 18.5 | 192.3 ± 23.5 §$ | 220.1 ± 7.9 ° |
| HR [min-1] | 441.8 ± 8.1 # |
410.4 ± 8.3 * |
451.0 ± 10.5 | 436.2 ± 8.6 |
431.3 ± 13.0 | 457.0 ± 12.0 ° | 423.4 ± 7.0 $° |
415.8 ± 9.4 $ |
| CI [mL min-1 kg-1] | 397.7 ± 16.7 | 334.9 ± 18.5 | 299.9 ± 19.0 | 223.8 ± 32.5 * | 392.4 ± 24.1 | 388.5 ± 29.1 | 316.5 ± 29.3 | 357.6 ± 14.2 |
| LV edP [mmHg] | 5.93 ± 0.54 |
6.38 ± 0.35 |
4.58 ± 0.38 |
8.08 ± 0.43 + |
5.24 ± 1.74 |
5.20 ± 0.59 |
5.58 ± 0.39 |
5.68 ± 0.86 |
| TPR [mmHg min kg mL-1] | 0.29 ± 0.01 |
0.27 ± 0.02 |
0.33 ± 0.02 |
0.43 ± 0.08 |
0.24 ± 0.02 |
0.30 ± 0.02 |
0.41 ± 0.06 § |
0.31 ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




