Submitted:
27 September 2025
Posted:
02 October 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Correlation-Causation Distinction: Origins and Utility
3. Challenging the Dogma: Do Vaccines Cause Harm?
- (i)
- It protects the vaccine program from reputational harm.
- (ii)
- It delegitimizes anecdotal or post-marketing reports as “anti-vaccine” or “misinformation”.
4. Case Report Analysis Under the Bradford Hill Criteria
I. Temporality
II. Plausibility
III. Coherence
IV. Specificity
V. Biological Gradient (Dose-Response Relationship)
VI. Analogy
VII. Consistency
VIII. Experimental Evidence
IX. Strength of Association
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Watson, O.J.; Barnsley, G.; Toor, J.; Hogan, A.B.; Winskill, P.; Ghani, A.C. Global impact of the first year of COVID-19 vaccination: a mathematical modelling study. Lancet Infect Dis 2022, 22, 1293–1302. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A.; Pezzullo, A.M.; Cristiano, A.; Boccia, S. Global Estimates of Lives and Life-Years Saved by COVID-19 Vaccination During 2020-2024. JAMA Health Forum 2025, 6, e252223. [Google Scholar] [CrossRef]
- Kuhbandner, C.; Reitzner, M. Estimation of excess mortality in Germany during 2020-2022. Cureus 2023, 15. [Google Scholar] [CrossRef]
- Forthun, I.; Madsen, C.; Emilsson, L.; Nilsson, A.; Kepp, K.P.; Björk, J.; Vollset, S.E.; Lallukka, T.; Skrindo Knudsen, A.K. Excess mortality in Denmark, Finland, Norway and Sweden during the COVID-19 pandemic 2020–2022. European Journal of Public Health 2024, 34, 737–743. [Google Scholar] [CrossRef]
- Jang, M.; Kim, S.; Choi, S.; Ryu, B.; Choi, S.Y.; Choi, S.; An, M.; Kim, S.-S. Estimating Excess Mortality During the COVID-19 Pandemic Between 2020–2022 in Korea. J Korean Med Sci 2024, 39, e267. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Jang, H.; Oh, J. Excess mortality during the Coronavirus disease pandemic in Korea. BMC Public Health 2023, 23, 1698. [Google Scholar] [CrossRef] [PubMed]
- Scherb, H.; Hayashi, K. Annual all-cause mortality rate in Germany and Japan (2005 to 2022) with focus on the COVID-19 pandemic: hypotheses and trend analyses. Med Clin Sci 2023, 5, 1–7. [Google Scholar] [CrossRef]
- Okoro, E.O.; Ikoba, N.A.; Okoro, B.E.; Akpila, A.S.; Salihu, M.O. Paradoxical increase in global COVID-19 deaths with vaccination coverage: World Health Organization estimates (2020–2023). International Journal of Risk & Safety in Medicine 2025, 09246479251336610. [Google Scholar] [CrossRef] [PubMed]
- Aarstad, J. Is There a Link between the 2021 COVID-19 Vaccination Uptake in Europe and 2022 Excess All-Cause Mortality? 2023.
- Aarstad, J. Deaths among young people in England increased significantly in 10 of 11 weeks after COVID-19 vaccination and doubled in three. EXCLI journal 2024, 23, 908. [Google Scholar]
- Mostert, S.; Hoogland, M.; Huibers, M.; Kaspers, G. Excess mortality across countries in the Western World since the COVID-19 pandemic:‘Our World in Data’estimates of January 2020 to December 2022. BMJ Public Health 2024, 2. [Google Scholar] [CrossRef]
- Alessandria, M.; Malatesta, G.M.; Berrino, F.; Donzelli, A. A critical analysis of all-cause deaths during COVID-19 vaccination in an Italian Province. Microorganisms 2024, 12, 1343. [Google Scholar] [CrossRef]
- Alessandria, M.; Malatesta, G.; Di Palmo, G.; Cosentino, M.; Donzelli, A. All-cause mortality according to COVID-19 vaccination status: An analysis of the UK office for National statistics public data. F1000Research 2025, 13, 886. [Google Scholar] [CrossRef]
- Barrowman, N. Correlation, causation, and confusion. The New Atlantis 2014, 23–44. [Google Scholar]
- Negri, F. Correlation is not causation, yet… matching and weighting for better counterfactuals. In Causality in Policy Studies: a Pluralist Toolbox; Springer International Publishing Cham, 2023; pp. 71–98. [Google Scholar]
- Kuhn, T.S.; Hacking, I. The structure of scientific revolutions; University of Chicago press Chicago, 1970; Volume 2. [Google Scholar]
- Karamitros, G.; Grant, M.P.; Lamaris, G.A. Associations in Medical Research Can Be Misleading: A Clinician’s Guide to Causal Inference. Journal of Surgical Research 2025, 310, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Hume, D. A treatise of human nature: Volume 1: Texts. 1739.
- Ward, A.C. The role of causal criteria in causal inferences: Bradford Hill’s” aspects of association”. Epidemiologic Perspectives & Innovations 2009, 6, 2. [Google Scholar]
- Li, X.; Mukandavire, C.; Cucunubá, Z.M.; Londono, S.E.; Abbas, K.; Clapham, H.E.; Jit, M.; Johnson, H.L.; Papadopoulos, T.; Vynnycky, E. Estimating the health impact of vaccination against ten pathogens in 98 low-income and middle-income countries from 2000 to 2030: a modelling study. The Lancet 2021, 397, 398–408. [Google Scholar] [CrossRef]
- Viliani, F.; Winkler, M.S. Ensure Healthy Lives and Promote Well-Being for All at All Ages. In Mining, Materials, and the Sustainable Development Goals (SDGs); CRC Press, 2020; pp. 15–28. [Google Scholar]
- Pan, J.; Wang, Y.; Cao, L.; Wang, Y.; Zhao, Q.; Tang, S.; Gong, W.; Guo, L.; Liu, Z.; Wen, Z. Impact of immunization programs on 11 childhood vaccine-preventable diseases in China: 1950–2018. The Innovation 2021, 2. [Google Scholar] [CrossRef]
- Ozawa, S.; Clark, S.; Portnoy, A.; Grewal, S.; Brenzel, L.; Walker, D.G. Return on investment from childhood immunization in low-and middle-income countries, 2011–20. Health Affairs 2016, 35, 199–207. [Google Scholar] [CrossRef]
- Morley, D. Saving children’s lives by vaccination. BMJ: British Medical Journal 1989, 299, 1544. [Google Scholar] [CrossRef]
- Rubio-Casillas, A.; Rodriguez-Quintero, C.M.; Redwan, E.M.; Gupta, M.N.; Uversky, V.N.; Raszek, M. Do vaccines increase or decrease susceptibility to diseases other than those they protect against? Vaccine, 42, 426-440. 2024.
- Aaby, P.; Benn, C.S. Developing the concept of beneficial non-specific effect of live vaccines with epidemiological studies. Clinical Microbiology and Infection 2019, 25, 1459–1467. [Google Scholar] [CrossRef]
- Benn, C.S.; Netea, M.G.; Selin, L.K.; Aaby, P. A small jab–a big effect: nonspecific immunomodulation by vaccines. Trends in immunology 2013, 34, 431–439. [Google Scholar] [CrossRef]
- Benn, C.S.; Fisker, A.B.; Whittle, H.C.; Aaby, P. Revaccination with live attenuated vaccines confer additional beneficial nonspecific effects on overall survival: a review. EBioMedicine 2016, 10, 312–317. [Google Scholar] [CrossRef]
- Benn, C.S.; Fisker, A.B.; Rieckmann, A.; Sørup, S.; Aaby, P. Vaccinology: time to change the paradigm? The Lancet infectious diseases 2020, 20, e274–e283. [Google Scholar] [CrossRef]
- Siebner, A.S.; Habib, M.; Osmani, V.; Adegnika, A.A.; Bogdan, C.; Breloer, M.; Elliott, A.; Fathi, A.; Hendrickx, G.; Nono, J.K. Interdisciplinary symposium on challenges and opportunities for vaccines: A comprehensive approach of current and future vaccine strategies to improve vaccine effectiveness in complex chronic infectious contexts. Vaccine: X 2025, 100615. [Google Scholar] [CrossRef]
- Poland, G.A.; Jacobson, R.M. Vaccine safety: injecting a dose of common sense. In Proceedings of the Mayo Clinic Proceedings; 2000; pp. 135–139. [Google Scholar]
- Feres, M.; Feres, M.F.N. Absence of evidence is not evidence of absence. 2023, 31, ed001.
- Hill, A.B. The environment and disease: association or causation? 1965.
- Imbens, G.W.; Rubin, D.B. Causal inference in statistics, social, and biomedical sciences; Cambridge university press: 2015.
- Phillips, C.V.; Goodman, K.J. The missed lessons of sir Austin Bradford Hill. Epidemiologic Perspectives & Innovations 2004, 1, 3. [Google Scholar] [CrossRef] [PubMed]
- Mörz, M. A case report: multifocal necrotizing encephalitis and myocarditis after BNT162b2 mRNA vaccination against COVID-19. Vaccines 2022, 10, 1651. [Google Scholar] [CrossRef]
- Won, J.H.; Byun, S.J.; Oh, B.-M.; Park, S.J.; Seo, H.G. Risk and mortality of aspiration pneumonia in Parkinson’s disease: a nationwide database study. Scientific reports 2021, 11, 6597. [Google Scholar] [CrossRef]
- Matsumoto, H.; Sengoku, R.; Saito, Y.; Kakuta, Y.; Murayama, S.; Imafuku, I. Sudden death in Parkinson’s disease: a retrospective autopsy study. Journal of the Neurological Sciences 2014, 343, 149–152. [Google Scholar] [PubMed]
- Brogna, C.; Cristoni, S.; Marino, G.; Montano, L.; Viduto, V.; Fabrowski, M.; Lettieri, G.; Piscopo, M. Detection of recombinant Spike protein in the blood of individuals vaccinated against SARS-CoV-2: Possible molecular mechanisms. PROTEOMICS–Clinical Applications 2023, 2300048. [Google Scholar]
- Pons, S.; Fodil, S.; Azoulay, E.; Zafrani, L. The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Critical care 2020, 24, 353. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Zhang, J.; Schiavon, C.R.; He, M.; Chen, L.; Shen, H.; Zhang, Y.; Yin, Q.; Cho, Y.; Andrade, L. SARS-CoV-2 spike protein impairs endothelial function via downregulation of ACE 2. Circulation research 2021, 128, 1323–1326. [Google Scholar] [CrossRef] [PubMed]
- Avolio, E.; Carrabba, M.; Milligan, R.; Kavanagh Williamson, M.; Beltrami, A.P.; Gupta, K.; Elvers, K.T.; Gamez, M.; Foster, R.R.; Gillespie, K. The SARS-CoV-2 Spike protein disrupts human cardiac pericytes function through CD147 receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. Clinical Science 2021, 135, 2667–2689. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, C.S.; Navarro Ramil, L.; Bieligk, J.; Meineke, R.; Rimmelzwaan, G.; Käufer, C.; Richter, F. Intravenous SARS-CoV-2 Spike protein induces neuroinflammation and alpha-Synuclein accumulation in brain regions relevant to Parkinson’s disease. Brain Behav Immun 2025, 129, 102–123. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Fal, A. To aspirate or not to aspirate? Considerations for the COVID-19 vaccines. Pharmacological Reports 2022, 74, 1223–1227. [Google Scholar] [CrossRef]
- Jarius, S.; Bieber, N.; Haas, J.; Wildemann, B. MOG encephalomyelitis after vaccination against severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2): case report and comprehensive review of the literature. Journal of neurology 2022, 269, 5198–5212. [Google Scholar] [CrossRef]
- Ancau, M.; Liesche-Starnecker, F.; Niederschweiberer, J.; Krieg, S.M.; Zimmer, C.; Lingg, C.; Kumpfmüller, D.; Ikenberg, B.; Ploner, M.; Hemmer, B. Case series: acute hemorrhagic encephalomyelitis after SARS-CoV-2 vaccination. Frontiers in neurology 2022, 12, 820049. [Google Scholar] [CrossRef]
- Choi, S.; Lee, S.; Seo, J.-W.; Kim, M.-j.; Jeon, Y.H.; Park, J.H.; Lee, J.K.; Yeo, N.S. Myocarditis-induced sudden death after BNT162b2 mRNA COVID-19 vaccination in Korea: case report focusing on histopathological findings. Journal of Korean medical science 2021, 36. [Google Scholar] [CrossRef]
- Sung, J.G.; Sobieszczyk, P.S.; Bhatt, D.L. Acute myocardial infarction within 24 hours after COVID-19 vaccination. The American Journal of Cardiology 2021, 156, 129–131. [Google Scholar] [CrossRef]
- Baumeier, C.; Aleshcheva, G.; Harms, D.; Gross, U.; Hamm, C.; Assmus, B.; Westenfeld, R.; Kelm, M.; Rammos, S.; Wenzel, P. Intramyocardial inflammation after COVID-19 vaccination: an endomyocardial biopsy-proven case series. International Journal of Molecular Sciences 2022, 23, 6940. [Google Scholar] [CrossRef]
- Walter, A.; Kraemer, M. A neurologist’s rhombencephalitis after comirnaty vaccination. A change of perspective. Neurological Research and Practice 2021, 3, 56. [Google Scholar] [CrossRef]
- Bensaidane, M.R.; Picher-Martel, V.; Émond, F.; De Serres, G.; Dupré, N.; Beauchemin, P. Case report: acute necrotizing encephalopathy following COVID-19 vaccine. Frontiers in Neurology 2022, 13, 872734. [Google Scholar] [CrossRef] [PubMed]
- Pongpitakmetha, T.; Hemachudha, P.; Rattanawong, W.; Thanapornsangsuth, P.; Viswanathan, A.; Hemachudha, T. COVID-19 related acute necrotizing encephalopathy with extremely high interleukin-6 and RANBP2 mutation in a patient with recently immunized inactivated virus vaccine and no pulmonary involvement. BMC Infectious Diseases 2022, 22, 640. [Google Scholar] [CrossRef]
- Rezvani, M.; Mahmoodkhani, M.; Sourani, A.; Sharafi, M.; Foroughi, M.; Mahdavi, S.B.; Sourani, A.; Khah, R.N.; Veisi, S. Treatment refractory acute necrotizing myelitis after COVID-19 vaccine injection: a case report. Annals of Medicine and Surgery 2024, 86, 1185–1190. [Google Scholar] [PubMed]
- Mikami, S.; Ishii, M.; Yano, T.; Hirayama, I.; Hayashi, Y.; Shiomi, T.; Tominaga, Y.; Ishida, T. Multifocal meningoencephalitis after vaccination against COVID-19. Pathology International 2024, 74, 697–703. [Google Scholar] [CrossRef]
- Siriratnam, P.; Buzzard, K.; Yip, G. Acute haemorrhagic necrotizing encephalopathy in the setting of SARS-CoV-2 vaccination: a case report and literature review. Acta neurologica Belgica 2023, 123, 303–305. [Google Scholar] [CrossRef] [PubMed]
- Senda, J.; Ashida, R.; Sugawara, K.; Kawaguchi, K. Acute meningoencephalitis after COVID-19 vaccination in an adult patient with rheumatoid vasculitis. Internal Medicine 2022, 61, 1609–1612. [Google Scholar] [CrossRef]
- Permezel, F.; Borojevic, B.; Lau, S.; de Boer, H.H. Acute disseminated encephalomyelitis (ADEM) following recent Oxford/AstraZeneca COVID-19 vaccination. Forensic Science, Medicine and Pathology 2022, 18, 74–79. [Google Scholar]
- Zanin, L.; Saraceno, G.; Renisi, G.; Signorini, L.; Battaglia, L.; Ferrara, M.; Rasulo, F.A.; Panciani, P.P.; Fontanella, M.M. Delayed onset of fatal encephalitis in a COVID-19 positive patient. International Journal of Neuroscience 2023, 133, 77–80. [Google Scholar]
- Ayuningtyas, T.; Natadidjaja, R.I.; Octaviani, C.; Sahli, F.; Adlani, H. Confirmed severe acute respiratory syndrome coronavirus 2 encephalitis in cerebrospinal fluid: a case report. Journal of Medical Case Reports 2022, 16, 154. [Google Scholar] [CrossRef]
- Benameur, K.; Agarwal, A.; Auld, S.C.; Butters, M.P.; Webster, A.S.; Ozturk, T.; Howell, J.C.; Bassit, L.C.; Velasquez, A.; Schinazi, R.F. Encephalopathy and encephalitis associated with cerebrospinal fluid cytokine alterations and coronavirus disease, Atlanta, Georgia, USA, 2020. Emerging infectious diseases 2020, 26, 2016. [Google Scholar] [CrossRef]
- Bernard-Valnet, R.; Pizzarotti, B.; Anichini, A. Two patients with acute meningo-encephalitis concomitant to SARS-CoV-2 infection. medRxiv. 2020; 20060251. Publisher Full Text.
- Bodro, M.; Compta, Y.; Llansó, L.; Esteller, D.; Doncel-Moriano, A.; Mesa, A.; Rodríguez, A.; Sarto, J.; Martínez-Hernandez, E.; Vlagea, A. Increased CSF levels of IL-1β, IL-6, and ACE in SARS-CoV-2–associated encephalitis. Neurology: Neuroimmunology & Neuroinflammation 2020, 7, e821. [Google Scholar]
- Cao, A.; Rohaut, B.; Le Guennec, L.; Saheb, S.; Marois, C.; Altmayer, V.; Carpentier, V.T.; Nemlaghi, S.; Soulie, M.; Morlon, Q. Severe COVID-19-related encephalitis can respond to immunotherapy. Brain 2020, 143, e102–e102. [Google Scholar] [CrossRef]
- Cheraghali, F.; Tahamtan, A.; Hosseini, S.A.; Gharib, M.H.; Moradi, A.; Razavi Nikoo, H.; Tabarraei, A. Case report: detection of SARS-CoV-2 from cerebrospinal fluid in a 34-month-old child with encephalitis. Frontiers in pediatrics 2021, 9, 565778. [Google Scholar] [CrossRef]
- Dahshan, A.; Abdellatef, A.A. Autoimmune encephalitis as a complication of COVID-19 infection: a case report. The Egyptian Journal of Internal Medicine 2022, 34, 32. [Google Scholar] [CrossRef]
- Dono, F.; Carrarini, C.; Russo, M.; De Angelis, M.V.; Anzellotti, F.; Onofrj, M.; Bonanni, L. New-onset refractory status epilepticus (NORSE) in post SARS-CoV-2 autoimmune encephalitis: a case report. Neurological Sciences 2021, 42, 35–38. [Google Scholar] [CrossRef]
- Elmouhib, A.; Benramdane, H.; zohra Ahsayen, F.; El Haddad, I.A.; El Ghalet, A.; Laaribi, I.; Bkiyar, H.; Nasri, S.; Skiker, I.; Housni, B. A case of limbic encephalitis associated with severely COVID-19 infection. Annals of Medicine and Surgery 2022, 74, 103274. [Google Scholar] [CrossRef] [PubMed]
- Etemadifar, M.; Salari, M.; Murgai, A.A.; Hajiahmadi, S. Fulminant encephalitis as a sole manifestation of COVID-19. Neurological Sciences 2020, 41, 3027–3029. [Google Scholar] [CrossRef]
- Gunawardhana, C.; Nanayakkara, G.; Gamage, D.; Withanage, I.; Bandara, M.; Siriwimala, C.; Senaratne, N.; Chang, T. Delayed presentation of postinfectious encephalitis associated with SARS-CoV-2 infection: a case report. Neurological Sciences 2021, 42, 3527–3530. [Google Scholar] [CrossRef] [PubMed]
- Khoo, A.; McLoughlin, B.; Cheema, S.; Weil, R.S.; Lambert, C.; Manji, H.; Zandi, M.S.; Morrow, J.M. Postinfectious brainstem encephalitis associated with SARS-CoV-2. Journal of Neurology, Neurosurgery & Psychiatry 2020, 91, 1013–1014. [Google Scholar] [CrossRef]
- Koh, S.; Kim, Y.S.; Kim, M.H.; Choi, Y.H.; Choi, J.Y.; Kim, T.-J. Encephalitis with status epilepticus and stroke as complications of non-severe COVID-19 in a young female patient: a case report. BMC neurology 2022, 22, 253. [Google Scholar] [CrossRef]
- Mekheal, E.; Mekheal, M.; Roman, S.; Mikhael, D.; Mekheal, N.; Manickam, R. A case report of autoimmune encephalitis: could post-COVID-19 autoimmunity become a lethal health issue? Cureus 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Meshref, M.; Hewila, I.M.; Mageed, S.A.; Morra, M.E. COVID-19 associated with encephalitis: case report and review of literature. The neurologist 2021, 26, 268–270. [Google Scholar] [CrossRef] [PubMed]
- Pizzanelli, C.; Milano, C.; Canovetti, S.; Tagliaferri, E.; Turco, F.; Verdenelli, S.; Nesti, L.; Franchi, M.; Bonanni, E.; Menichetti, F. Autoimmune limbic encephalitis related to SARS-CoV-2 infection: case-report and review of the literature. Brain, behavior, & immunity-health 2021, 12, 100210. [Google Scholar]
- Sangare, A.; Dong, A.; Valente, M.; Pyatigorskaya, N.; Cao, A.; Altmayer, V.; Zyss, J.; Lambrecq, V.; Roux, D.; Morlon, Q. Neuroprognostication of consciousness recovery in a patient with COVID-19 related encephalitis: preliminary findings from a multimodal approach. Brain Sciences 2020, 10, 845. [Google Scholar] [CrossRef]
- Sarmast, S.T.; Mohamed, A.S.; Amar, Z.; Sarwar, S.; Ahmed, Z. A case of acute encephalitis in COVID-19 patient: a rare complication. Cureus 2021, 13. [Google Scholar] [CrossRef]
- Sattar, S.B.A.; Haider, M.A.; Zia, Z.; Niazi, M.; Iqbal, Q.Z.; Niazi, M.R.K. Clinical, radiological, and molecular findings of acute encephalitis in a COVID-19 patient: a rare case report. Cureus 2020, 12. [Google Scholar] [CrossRef]
- Sharma, R.; Nalleballe, K.; Shah, V.; Haldal, S.; Spradley, T.; Hasan, L.; Mylavarapu, K.; Vyas, K.; Kumar, M.; Onteddu, S. Spectrum of hemorrhagic encephalitis in COVID-19 patients: a case series and review. Diagnostics 2022, 12, 924. [Google Scholar] [CrossRef]
- Sofijanova, A.; Bojadzieva, S.; Duma, F.; Superlishka, E.; Murtezani, A.; Jordanova, O. Severe encephalitis in infant with COVID-19: a case report. Open Access Macedonian Journal of Medical Sciences 2020, 8, 514–517. [Google Scholar] [CrossRef]
- Wettervik, T.S.; Kumlien, E.; Rostami, E.; Howells, T.; von Seth, M.; Velickaite, V.; Lewén, A.; Enblad, P. Intracranial pressure dynamics and cerebral vasomotor reactivity in coronavirus disease 2019 patient with acute encephalitis. Critical care explorations 2020, 2, e0197. [Google Scholar] [CrossRef] [PubMed]
- Tee, T.Y.; Thabit, A.A.M.; Khoo, C.S.; Shahrom, H.M.; Chan, E.Z.; Marzukie, M.M.; Kamaruddin, Z.A.C.; Thayan, R.; Chidambaram, S.K. Acute encephalitis associated with SARS-CoV-2 confirmed in cerebrospinal fluid: First case in Malaysia. Journal of Clinical Neurology (Seoul, Korea) 2021, 17, 490. [Google Scholar]
- Woldie, I.L.; Brown, I.G.; Nwadiaro, N.F.; Patel, A.; Jarrar, M.; Quint, E.; Khokhotva, V.; Hugel, N.; Winger, M.; Briskin, A. Autoimmune hemolytic anemia in a 24-year-old patient with COVID-19 complicated by secondary cryptococcemia and acute necrotizing encephalitis: a case report and review of literature. Journal of medical cases 2020, 11, 362. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Ren, Y.; Lv, T. Encephalitis as a clinical manifestation of COVID-19. Brain, behavior, and immunity 2020, 88, 945. [Google Scholar] [CrossRef] [PubMed]
- Urso, L.; Distefano, M.G.; Cambula, G.; Colomba, A.I.; Nuzzo, D.; Picone, P.; Giacomazza, D.; Sicurella, L. The case of encephalitis in a COVID-19 pediatric patient. Neurological Sciences 2022, 43, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Vandervorst, F.; Guldolf, K.; Peeters, I.; Vanderhasselt, T.; Michiels, K.; Berends, K.J.; Van Laethem, J.; Pipeleers, L.; Vincken, S.; Seynaeve, L. Encephalitis associated with the SARS-CoV-2 virus: a case report. Interdisciplinary Neurosurgery 2020, 22, 100821. [Google Scholar] [CrossRef]
- Zambreanu, L.; Lightbody, S.; Bhandari, M.; Hoskote, C.; Kandil, H.; Houlihan, C.F.; Lunn, M.P. A case of limbic encephalitis associated with asymptomatic COVID-19 infection. Journal of Neurology, Neurosurgery & Psychiatry 2020, 91, 1229–1230. [Google Scholar] [CrossRef]
- Zandifar, S.; Zandifar, Z. Acute viral encephalitis associated with SARS-CoV-2. Ann Clin Case Rep 2020, 5, 1845. [Google Scholar]
- Lersy, F.; Anheim, M.; Willaume, T.; Chammas, A.; Brisset, J.-C.; Cotton, F.; Kremer, S. Cerebral vasculitis of medium-sized vessels as a possible mechanism of brain damage in COVID-19 patients. Journal of Neuroradiology 2021, 48, 141–146. [Google Scholar] [CrossRef]
- Zuhorn, F.; Omaimen, H.; Ruprecht, B.; Stellbrink, C.; Rauch, M.; Rogalewski, A.; Klingebiel, R.; Schäbitz, W.-R. Parainfectious encephalitis in COVID-19:“the claustrum sign”. Journal of neurology 2021, 268, 2031–2034. [Google Scholar]
- Tan, S.; Cui, C.; Song, E.-P.; Shan, Y.; Chang, Y.; Qiu, W.; Lu, Z. Acute haemorrhagic necrotizing encephalopathy and inflammatory demyelinating encephalopathy associated with COVID-19 in adults in Southern China. BMC Infectious Diseases 2025, 25, 1–10. [Google Scholar] [CrossRef]
- Ghosh, R.; Dubey, S.; Finsterer, J.; Chatterjee, S.; Ray, B.K. SARS-CoV-2-associated acute hemorrhagic, necrotizing encephalitis (AHNE) presenting with cognitive impairment in a 44-year-old woman without comorbidities: a case report. The American journal of case reports 2020, 21, e925641–925641. [Google Scholar] [CrossRef]
- Mierzewska-Schmidt, M.; Baranowski, A.; Szymanska, K.; Ciaston, M.; Kuchar, E.; Ploski, R.; Kosinska, J.; Pagowska-Klimek, I. The case of fatal acute hemorrhagic necrotizing encephalitis in a two-month-old boy with Covid-19. International Journal of Infectious Diseases 2022, 116, 151–153. [Google Scholar] [CrossRef]
- Morvan, A.-C.; Kerambrun, H. Fatal necrotizing encephalitis associated with COVID-19. Neurology: Clinical Practice 2021, 11, e214–e215. [Google Scholar] [CrossRef]
- Mullaguri, N.; Sivakumar, S.; Battineni, A.; Anand, S.; Vanderwerf, J. COVID-19 related acute hemorrhagic necrotizing encephalitis: a report of two cases and literature review. Cureus 2021, 13. [Google Scholar] [CrossRef]
- Massari, M.; Spila Alegiani, S.; Morciano, C.; Spuri, M.; Marchione, P.; Felicetti, P.; Belleudi, V.; Poggi, F.R.; Lazzeretti, M.; Ercolanoni, M. Postmarketing active surveillance of myocarditis and pericarditis following vaccination with COVID-19 mRNA vaccines in persons aged 12 to 39 years in Italy: A multi-database, self-controlled case series study. PLoS Medicine 2022, 19, e1004056. [Google Scholar] [CrossRef]
- Ling, R.R.; Ramanathan, K.; Tan, F.L.; Tai, B.C.; Somani, J.; Fisher, D.; MacLaren, G. Myopericarditis following COVID-19 vaccination and non-COVID-19 vaccination: a systematic review and meta-analysis. The Lancet Respiratory Medicine 2022, 10, 679–688. [Google Scholar] [CrossRef]
- Stowe, J.; Miller, E.; Andrews, N.; Whitaker, H.J. Risk of myocarditis and pericarditis after a COVID-19 mRNA vaccine booster and after COVID-19 in those with and without prior SARS-CoV-2 infection: a self-controlled case series analysis in England. PLoS Medicine 2023, 20, e1004245. [Google Scholar]
- Abue, M.; Mochizuki, M.; Shibuya-Takahashi, R.; Ota, K.; Wakui, Y.; Iwai, W.; Kusaka, J.; Saito, M.; Suzuki, S.; Sato, I. Repeated COVID-19 Vaccination as a Poor Prognostic Factor in Pancreatic Cancer: A Retrospective, Single-Center Cohort Study. Cancers 2025, 17, 2006. [Google Scholar] [CrossRef]
- Ota, N.; Itani, M.; Aoki, T.; Sakurai, A.; Fujisawa, T.; Okada, Y.; Noda, K.; Arakawa, Y.; Tokuda, S.; Tanikawa, R. Expression of SARS-CoV-2 spike protein in cerebral Arteries: Implications for hemorrhagic stroke Post-mRNA vaccination. Journal of Clinical Neuroscience 2025, 136, 111223. [Google Scholar] [CrossRef] [PubMed]
- Vogel, C.; Marcotte, E.M. Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nature reviews genetics 2012, 13, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Clausen, L.; Abildgaard, A.B.; Gersing, S.K.; Stein, A.; Lindorff-Larsen, K.; Hartmann-Petersen, R. Protein stability and degradation in health and disease. Advances in protein chemistry and structural biology 2019, 114, 61–83. [Google Scholar] [PubMed]
- Li, W.; Kitsios, G.D.; Bain, W.; Wang, C.; Li, T.; Fanning, K.V.; Deshpande, R.; Qin, X.; Morris, A.; Lee, J.S. Stability of SARS-CoV-2-encoded proteins and their antibody levels correlate with interleukin 6 in COVID-19 patients. Msystems 2022, 7, e00058–00022. [Google Scholar] [CrossRef]
- Moderbacher, C.R.; Ramirez, S.I.; Dan, J.M.; Grifoni, A.; Hastie, K.M.; Weiskopf, D.; Belanger, S.; Abbott, R.K.; Kim, C.; Choi, J. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell 2020, 183, 996–1012. e1019. [Google Scholar] [CrossRef]
- Rong, Z.; Mai, H.; Ebert, G.; Kapoor, S.; Puelles, V.G.; Czogalla, J.; Hu, S.; Su, J.; Prtvar, D.; Singh, I. Persistence of spike protein at the skull-meninges-brain axis may contribute to the neurological sequelae of COVID-19. Cell Host & Microbe 2024. [Google Scholar]
- Olsen, J.V.; Ong, S.-E.; Mann, M. Trypsin cleaves exclusively C-terminal to arginine and lysine residues. Molecular & cellular proteomics 2004, 3, 608–614. [Google Scholar]
- Miller, N.L.; Clark, T.; Raman, R.; Sasisekharan, R. Insights on the mutational landscape of the SARS-CoV-2 Omicron variant. BioRxiv 2021. [Google Scholar]
- Zuo, W.; He, D.; Liang, C.; Du, S.; Hua, Z.; Nie, Q.; Zhou, X.; Yang, M.; Tan, H.; Xu, J. The persistence of SARS-CoV-2 in tissues and its association with long COVID symptoms: a cross-sectional cohort study in China. The Lancet Infectious Diseases 2024, 24, 845–855. [Google Scholar] [CrossRef]
- Gould, A.L. Unified screening for potential elevated adverse event risk and other associations. Statistics in Medicine 2018, 37, 2667–2689. [Google Scholar] [CrossRef] [PubMed]
- Broudic, K.; Laurent, S.; Perkov, V.; Simon, C.; Garinot, M.; Truchot, N.; Latour, J.; Désert, P. Nonclinical safety assessment of an mRNA Covid-19 vaccine candidate following repeated administrations and biodistribution. Journal of Applied Toxicology 2024, 44, 371–390. [Google Scholar] [CrossRef] [PubMed]
- Hassett, K.J.; Benenato, K.E.; Jacquinet, E.; Lee, A.; Woods, A.; Yuzhakov, O.; Himansu, S.; Deterling, J.; Geilich, B.M.; Ketova, T. Optimization of lipid nanoparticles for intramuscular administration of mRNA vaccines. Molecular Therapy-Nucleic Acids 2019, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Reichmuth, A.M.; Oberli, M.A.; Jaklenec, A.; Langer, R.; Blankschtein, D. mRNA vaccine delivery using lipid nanoparticles. Therapeutic delivery 2016, 7, 319–334. [Google Scholar] [CrossRef]
- Röltgen, K.; Nielsen, S.C.; Silva, O.; Younes, S.F.; Zaslavsky, M.; Costales, C.; Yang, F.; Wirz, O.F.; Solis, D.; Hoh, R.A. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell 2022, 185, 1025–1040. e1014. [Google Scholar] [CrossRef]
- Lindsay, K.E.; Bhosle, S.M.; Zurla, C.; Beyersdorf, J.; Rogers, K.A.; Vanover, D.; Xiao, P.; Araínga, M.; Shirreff, L.M.; Pitard, B. Visualization of early events in mRNA vaccine delivery in non-human primates via PET–CT and near-infrared imaging. Nature biomedical engineering 2019, 3, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Krauson, A.J.; Casimero, F.V.C.; Siddiquee, Z.; Stone, J.R. Duration of SARS-CoV-2 mRNA vaccine persistence and factors associated with cardiac involvement in recently vaccinated patients. npj Vaccines 2023, 8, 141. [Google Scholar] [CrossRef] [PubMed]
- Angeli, F.; Spanevello, A.; Reboldi, G.; Visca, D.; Verdecchia, P. SARS-CoV-2 vaccines: Lights and shadows. European journal of internal medicine 2021, 88, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mead, M.N.; Rose, J.; Seneff, S.; Rogers, C.; Craven, B.; Hulscher, N.; Cosgrove, K.; Marik, P.; McCullough, P.A. Compound Adverse Effects of COVID-19 mRNA Vaccination and Coronavirus Infection: A Convergence of Extensive Spike Protein Harms to the Human Body. 2025.
- Patterson, B.K.; Yogendra, R.; Francisco, E.B.; Guevara-Coto, J.; Long, E.; Pise, A.; Osgood, E.; Bream, J.; Kreimer, M.; Jeffers, D. Detection of S1 spike protein in CD16+ monocytes up to 245 days in SARS-CoV-2-negative post-COVID-19 vaccine syndrome (PCVS) individuals. Human Vaccines & Immunotherapeutics 2025, 21, 2494934. [Google Scholar]
- Bhattacharjee, B.; Lu, P.; Monteiro, V.S.; Tabachnikova, A.; Wang, K.; Hooper, W.B.; Bastos, V.; Greene, K.; Sawano, M.; Guirgis, C. Immunological and antigenic signatures associated with chronic illnesses after COVID-19 vaccination. medRxiv 2025, 2025.2002. 2018.25322379.
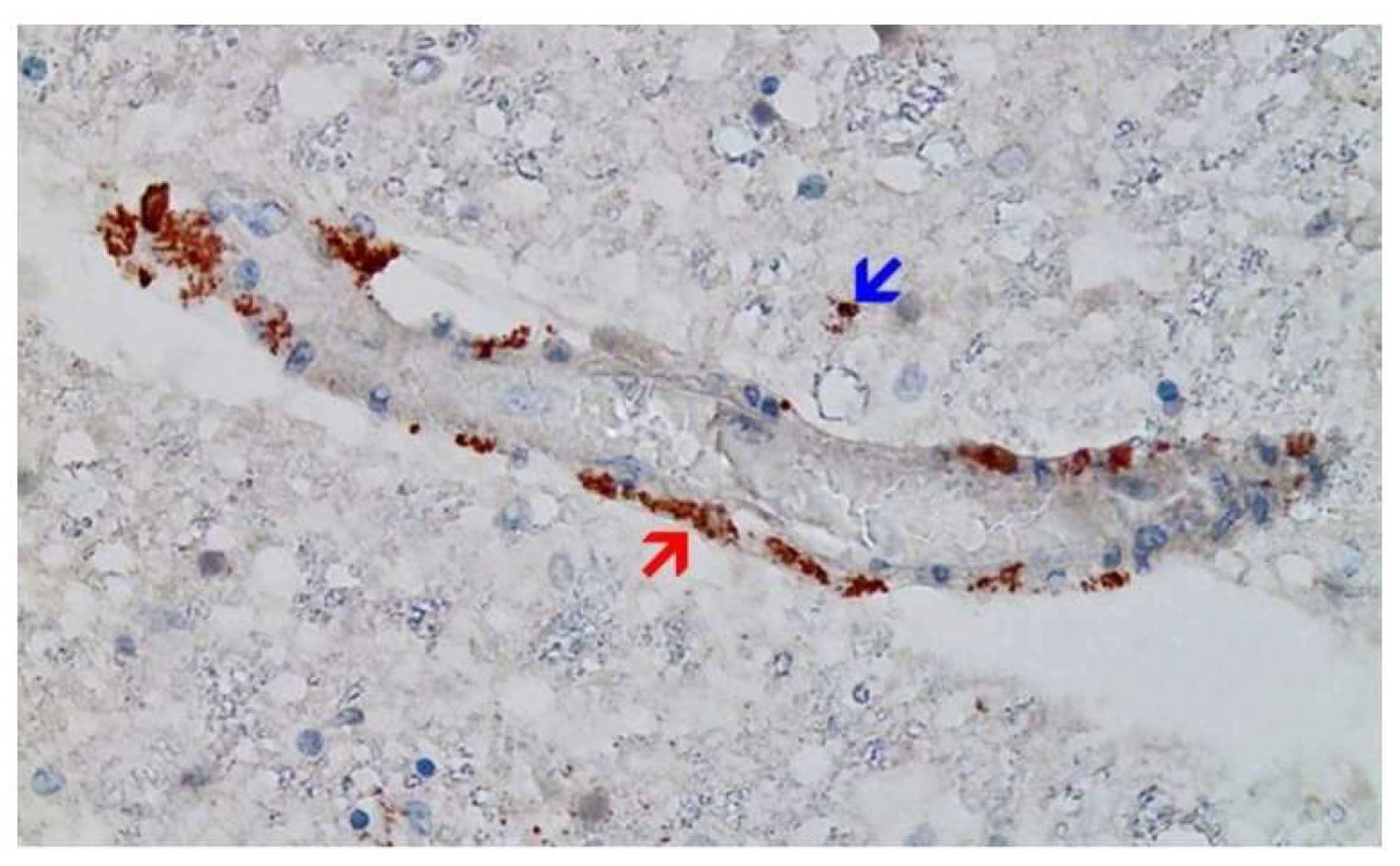
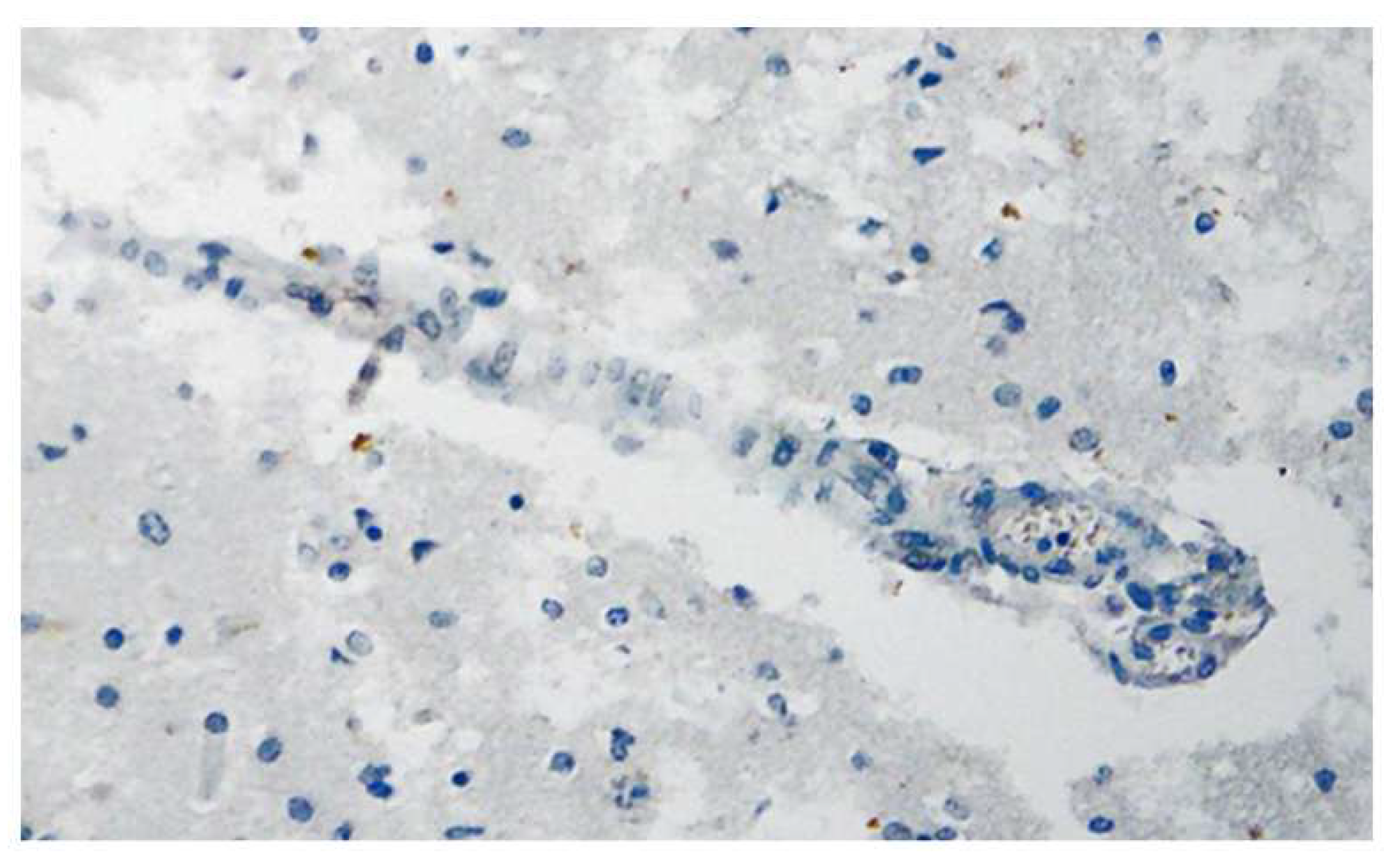
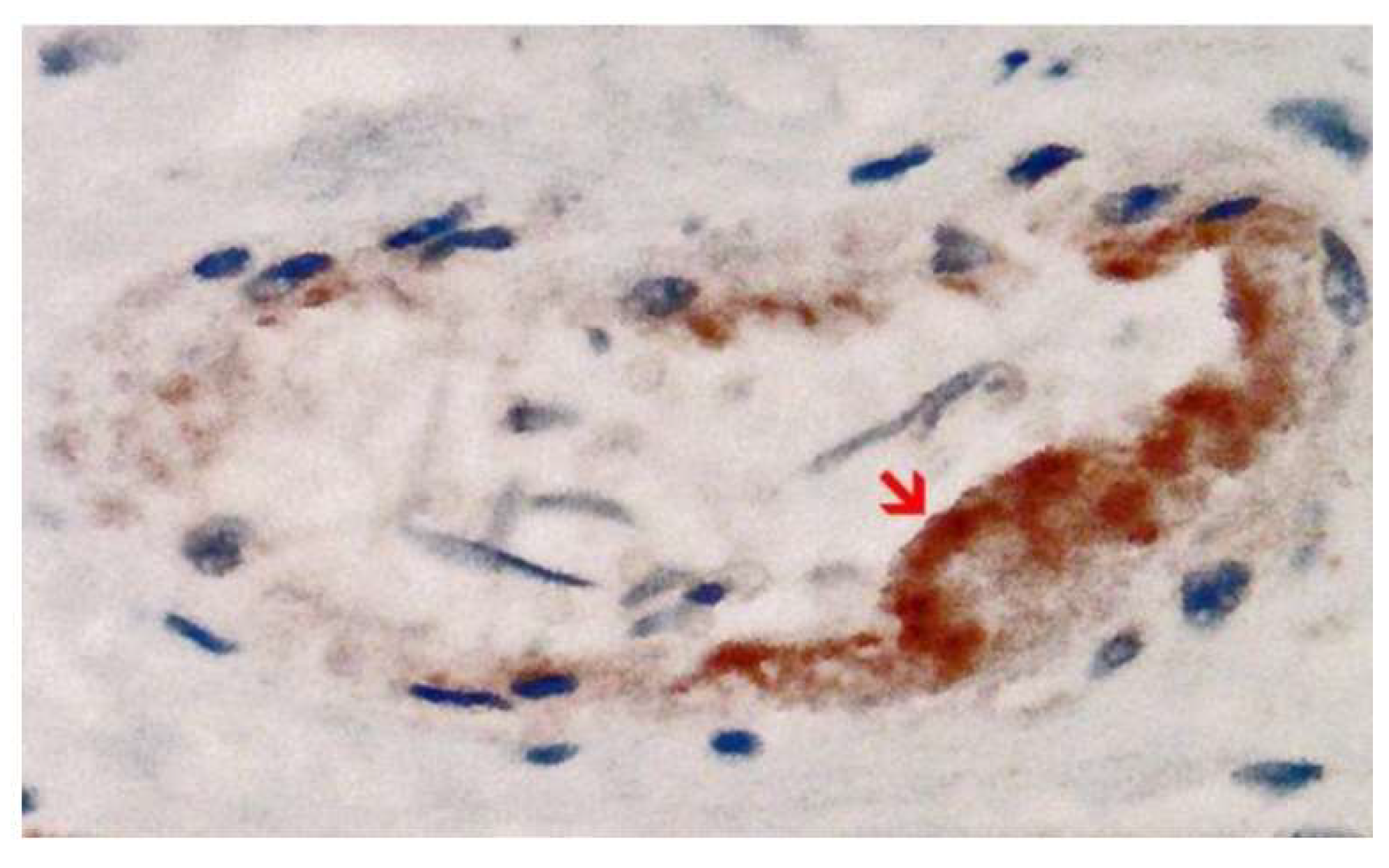
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



