Submitted:
29 September 2025
Posted:
30 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Institutional and Regulatory Framework
Ethical and Regulatory Approval
Standard Operating Procedures (SOPs)
Institutional Resources and Integration
Risk Management and Insurance
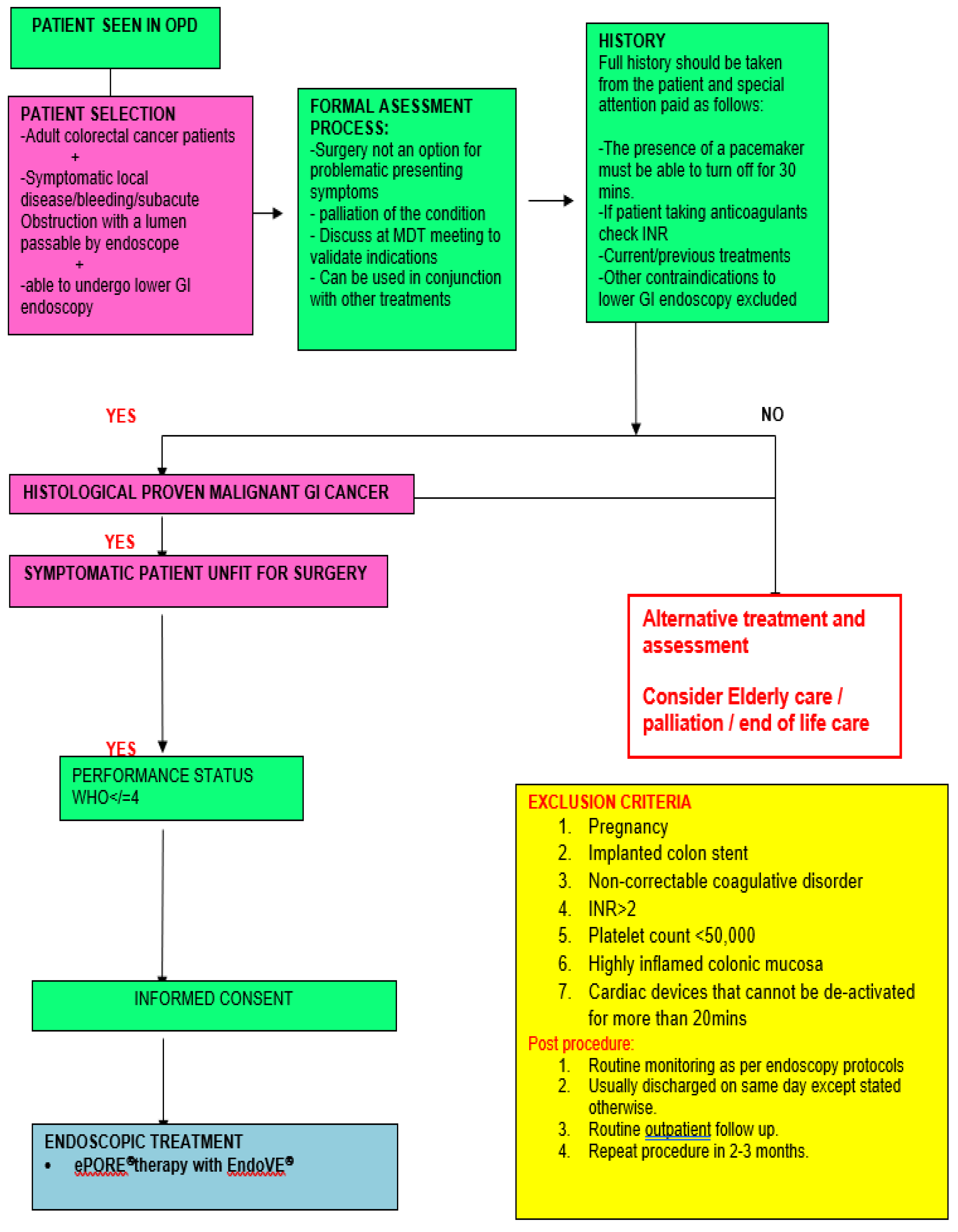
Patient Referral and Clinical Indications for Endoscopic Electroporation
- Inoperable, advanced, or recurrent left colonic or rectal adenocarcinoma presenting with symptoms such as bleeding, pain, or mucous discharge, particularly when not amenable to surgery, chemotherapy, radiation, or stenting.
- Progressive tumours with anticipated worsening of symptoms, where localised therapy may delay deterioration or prevent emergency interventions such as de-functioning stomas.
- Symptomatic lesions (e.g., ulcerated, friable, or bleeding tumours) in patients undergoing systemic therapy, where EE may provide local control and improve quality of life.
- Patient preference for minimally invasive or non-systemic palliative therapy, especially in those declining further chemotherapy or radiotherapy due to prior toxicity or personal values.
- Elderly or frail patients with poor performance status (Eastern Cooperative Oncology Group -ECOG ≥2), for whom conventional surgery or systemic options carry disproportionate risk.
Contraindications
Absolute Contraindications:
- Implanted metallic colon stents, due to the risk of conducting electric pulses through the bowel.
- Pregnancy and lactation: As the safety of IT calcium electroporation during pregnancy has not been established, pregnant or lactating patients should not undergo EE. Potentially fertile patients must use reliable contraception before, during, and after treatment to avoid foetal exposure.
- Perforation risk: Patients with transmural ulceration, extensive tumour necrosis, or full-thickness involvement on imaging may be theoretically at elevated risk for colonic or rectal perforation and should not be considered unless surgical backup is immediately available.
- Inaccessible tumour location: Lesions located beyond the reach of a flexible sigmoidoscope (proximal to the splenic flexure), or those obscured by strictures or impassable narrowing, are not currently suitable for EE.
- Relative Contraindications:
- Severe proctitis or inflammation in the target area (e.g., active IBD, radiation proctopathy) due to risk of unpredictable tissue response or complications.
- Uncorrected coagulopathy: While not a strict contraindication, patients with an international normalised ratio (INR) >2 or platelets <50,000/mm³ require correction or delay of the procedure. Anticoagulation may be managed using standard perioperative bridging protocols [28]. EE has a profile similar to that of low-risk endoscopic procedures, and relevant guidelines from the British Society of Gastroenterology (BSG) for anticoagulants/antiplatelets are applicable [28].
- Pacemakers and implanted defibrillators that cannot be deactivated for >30 minutes: These are not contraindications to EE using bipolar electrodes, even in close anatomical proximity. However, defibrillators should be deactivated before pulse delivery and reactivated immediately thereafter to prevent inappropriate discharges or malfunction. A discussion with cardiologists and electrophysiologists on the impact of the Bipolar EE probe on cardiac devices should be sought.
- Poor performance status (ECOG >3): Although EE is minimally invasive, its application should be restricted to patients likely to tolerate bowel preparation and sedation safely.
- Circumferential tumour involvement >75% or active colorectal obstruction requiring immediate surgery
Pre-Treatment Evaluation
Medical History
Laboratory Investigations
Imaging and Endoscopic Assessment
Special Considerations
Infrastructure and Equipment
Clinical Setting
Core Equipment Requirements
A. Electroporation Device
-
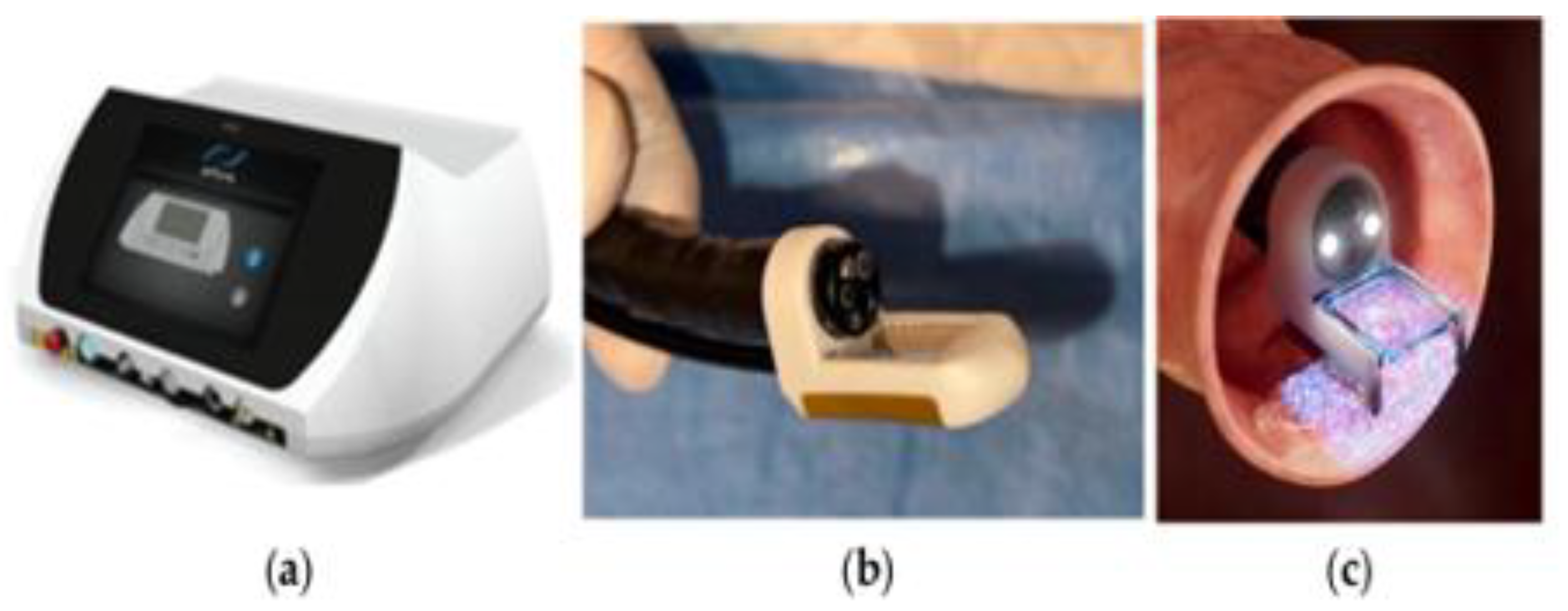
A CE-marked or UKCA-approved pulse generator capable of delivering square-wave, bipolar, high-voltage electric pulses in a controlled and reproducible way. The only delivery system in this field is the CE-marked and MDD-certified (2027/MDD) ePORE electroporation generator, along with the single-use CE-marked EndoVE probe (Mirai Medical, Galway, Ireland).
- ○
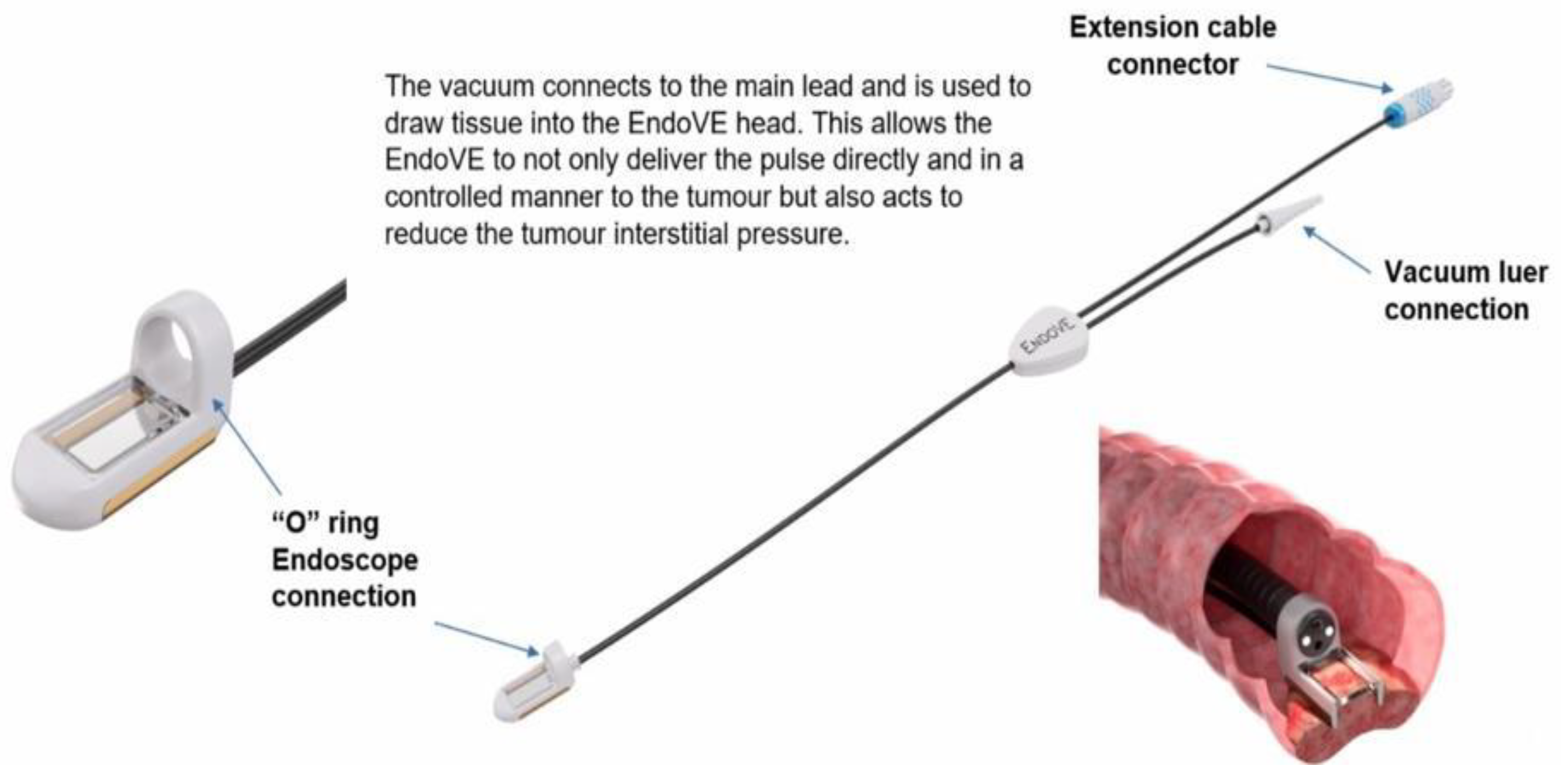
- The EndoVE® platform is a novel, endoscope-compatible device specifically engineered to deliver EE to gastrointestinal mucosal and submucosal tumours. The catheter includes a treatment chamber with two bipolar electrodes, into which tissue is drawn by vacuum, enabling maximum tissue contact. The device is mounted on the endoscope through an “O” ring (Figure 3).
- ○
- The electrode allows for segmental delivery of electrical stimulation following intra-tumour calcium instillation via the endoscope (Ca-EP) or systemic chemotherapy injection, in accordance with pharmacy administration guidelines.
- ○
- A foot pedal or remote switch can be used for controlled pulse delivery
- A vacuum suction device able to deliver pressure up to 400-500 mmHg.
B. Endoscopic Probes and Accessories
- A standard flexible endoscope with a diameter measuring 9.5-10.5mm (e.g., adult gastroscope or paediatric colonoscope) to fit into the EndoVE® probe.
- Standard injection needle catheter for intra-tumoral injection of calcium gluconate or chemotherapy (Bleomycin/cisplatin).
- Carbon dioxide (CO2) insufflation for endoscopic comfort and reduced risk of barotrauma.
C. Injection and Drug Preparation Materials
- Calcium gluconate 10% solution (typically 10-20mls), preservative-free, stored under appropriate conditions and verified before use
- 15,000 IU/m2 for IV bleomycin in ECT
- 10mg of IV chlorpheniramine and 100mg of IV hydrocortisone as premedication for IV bleomycin
- 20 mL syringe for IT injection
- Sterile saline for flushing
D. Monitoring and Support Equipment
- Continuous cardiopulmonary monitoring system for patients under sedation
- Conscious sedation management as per BSG guidelines.
Imaging and Diagnostic Support
Safety Systems and Equipment Maintenance
Personnel and Operational Readiness
Training and Accreditation
Clinical Competency Framework
Clinical and Endoscopic Competence
-
EE should be delivered by accredited colorectal surgeons or therapeutic endoscopists. The minimum skill set consists of the following four T’s: tip control, Torque of the endoscopy shaft, Therapeutic endo-injections, and turning the patient when appropriate. For more complex procedures, advanced endoscopists require the following skill set:
- ○
- experienced in diagnostic and interventional flexible sigmoidoscopy or colonoscopy (minimum of lifetime procedures recommended by BSG or other relevant regulatory bodies)
- ○
- can safely deliver submucosal or IT injections under endoscopic guidance in complex settings
- ○
- trained to manage intra-procedural complications such as bleeding, perforation, or patient instability
- Operators must also be familiar with the selection criteria for EE, the principles of tumour accessibility, and contraindications.
Electroporation-Specific Training
Anaesthetic and Nursing Team Training
Certification and Accreditation
Continuing Professional Development (CPD)
Simulation and Dry Lab Models
Patient Information, Consent, and Preparation
- Treatment intent– palliative, salvage, debulking, or disease control
- Procedure– sedation, IT injection, electrical pulse delivery, and expected duration (~30 minutes in straightforward cases)
- Expected outcomes– tumour shrinkage, local control, symptom relief (e.g., pain, bleeding)
-
Risks and side effects– including:
- ○
- Transient pain or minor self-limiting bleeding
- ○
- Ulceration or mucosal necrosis
- ○
- Risk of perforation (rare)
- ○
- Post-treatment mucoid discharge or tenesmus
- ○
- Pulmonary fibrosis with systemic bleomycin in ECT
Procedural Technique and Treatment Delivery
- Site: Rectal, recto-sigmoid, sigmoid, or descending colon. The distance from the anal verge and involvement of the sphincter complex (for low rectal lesions), as well as accessibility and lumen traversability with the flexible endoscope.
- Single (or multiple) - Number of lesions: Single or multiple; if multiple, contiguous or discontinuous
- Size of each lesion: Maximum diameter (cm) and estimated volume (if spherical or cylindrical) to determine the amount of IT injection.
- Shape (Morphology): Polypoid, flat, ulcerated, infiltrative, annular constricting, exophytic, bulky, or circumferential
- Surface: bleeding, ulcerated, dominant nodule.
- Surrounding: e.g., Radiation proctitis, IBD, Diverticular disease
- Synchronous lesions: e.g., Benign Polyps
- -
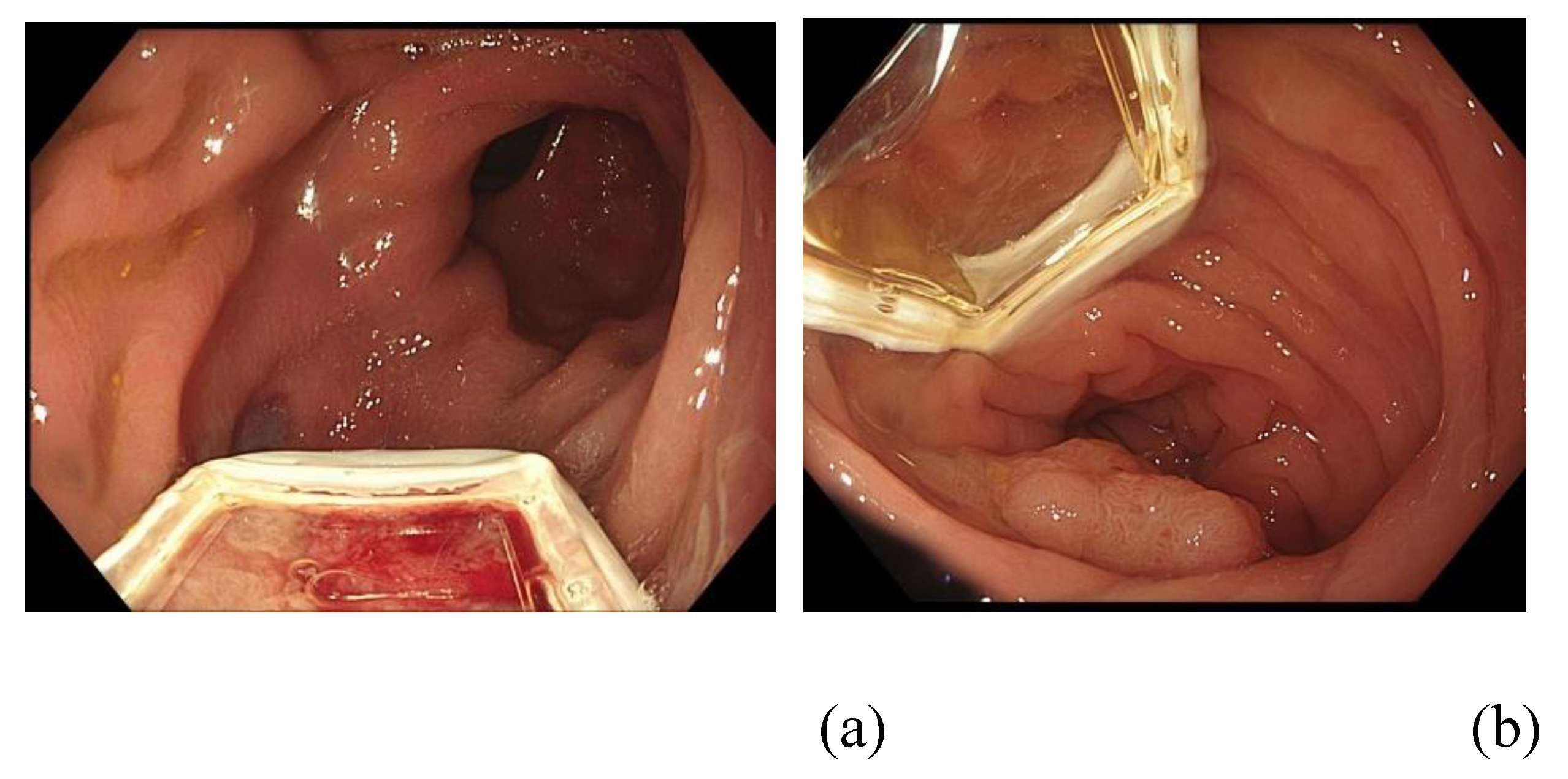
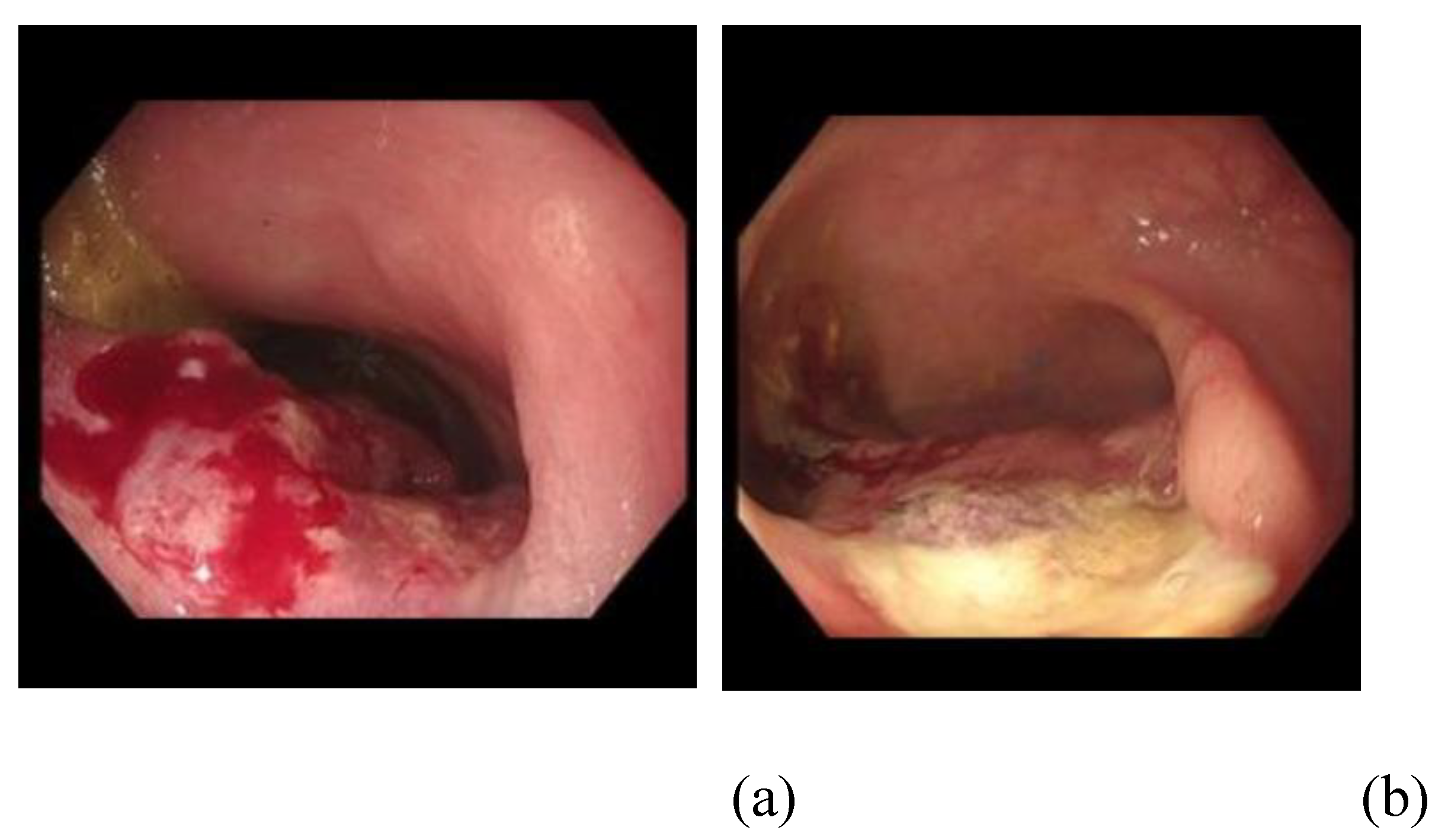
- ‘Thumb print’ sign of contact (demarcation zone of ablation)
- -
- ‘White’ sign of impact (tissue blanching following EE)
- -
- Tumour regression (subjective reduction in size of lesion)
Follow-up, Outcomes Assessment, and Retreatments
Structured Follow-Up Schedule
- Week 1: Initial telephone consultation with a physician or nurse to assess pain, bleeding, discharge, and early mucosal reaction. This can be repeated in week 4 to monitor symptoms and initial quality of life (QoL) using tools like the SF-12 questionnaire [25]. If necessary, a face-to-face clinic appointment can be scheduled in week 4 (or earlier) to address any significant issues.
- Weeks 8-12 (2-3 months): Perform primary response assessment using flexible sigmoidoscopy and targeted biopsy (if applicable); this is the earliest point to confirm a complete clinical response (cCR). Patients may receive retreatment at this visit if clinically indicated, following the initial assessment.
- Weeks 16 and 24 (4 and 6 months): Continue tumour surveillance and assess quality of life. If the patient remains asymptomatic with cCR, further surveillance can be determined by MDT consensus. Patients with symptom control and stable disease, confirmed through endoscopy and imaging, may be moved to a 6- to 12-month surveillance schedule.
- For large, bulky, or circumferential lesions, multiple sessions are typically required. These are usually scheduled every 6 to 12 weeks, though they may occur earlier depending on the circumstances. Our definition of failed treatment is a lack of response or disease progression after 3 to 4 sessions.
Outcomes Assessment
- Complete Clinical Response (cCR): Absence of visible tumour, ulceration, or bleeding; negative targeted biopsy following magnification chromo-endoscopy.
- Partial Response (PR): ≥10% reduction in tumour size and symptoms, but residual visible or histologically positive tumour.
- Stable Disease (SD): No significant change in tumour size or symptoms.
- Progressive Disease (PD): Enlargement or new lesions post-treatment.
Retreatments
- Incomplete response or residual tumour after initial treatment
- For symptomatic relief (e.g. bleeding, subacute obstruction)
- Tumour recurrence within the previously treated field
- New or metachronous lesions elsewhere in the rectum or distal colon
Discharge from the SEE Service
Patient Access to Information
- Written information leaflets outlining expectations, self-care, symptom management, and when to seek help.
- Dedicated hospital website pages with educational videos and Frequently Asked Questions (FAQs) about EE.
- Contact details for the clinical team and CNS for post-procedural support.
Integration into Clinical Practice
- Inoperable CRC
- Recurrent/residual disease after conventional therapy
- Salvage therapy for patients desiring non-operative management
- Palliative care needs with bleeding or obstructing tumours
Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | Ademola Adeyeye |
| AH | Amyn Haji |
| APC | Argon Plasma Coagulation |
| ATP | Adenosine Triphosphate |
| BSG | British Society of Gastroenterology |
| Ca-EP | Calcium Electroporation |
| CAN | Cancer Alliance Network |
| CE | Conformité Européenne |
| cCR | Complete Clinical Response |
| CNS | Clinical Nurse Specialist |
| CPD | Continuing Professional Development |
| CRC | Complex Colorectal Cancer |
| CRM | Circumferential Resection Margin |
| CT | Computed Tomography |
| EAL | Endoscopic Ablation Lesion |
| ECT | Electrochemotherapy |
| EE | Endoscopic Electroporation |
| EMVI | Extramural Vascular Invasion |
| EP | Electroporation |
| ESOPE | European Standard Operating Procedures of Electrochemotherapy |
| FAQ | Frequently Asked Questions |
| GCP | Good Clinical Practice |
| INR | Internationalised Normal Ratio |
| IRB | Institutional Review Board |
| IT | Intratumoral |
| IV | Intravenous |
| KCH | King’s College Hospital |
| MDD | Medical Device Directive |
| MDT | Multi-Disciplinary Team |
| MRI | Magnetic Resonance Imaging |
| NAAP | Non-Anaesthesiology Administered Propofol Sedation |
| NAPS | Nurse Assisted Propofol Sedation |
| NCPC | New Clinical Procedures Committee |
| NHS | National Health Service |
| PET-CT | Positron Emission Tomography – computed tomography |
| R&D | Research and Development |
| REC | Resarch Ethics Committee |
| SEE | Salvage Endoscopic Electroporation |
| SOP | Standard Operating Procedure |
| SPECC | Significant Polyp and Early Colorectal Cancer |
| TIVA | Total Intravenous Anaesthesia |
| TNM | Tumour-Node-Metastasis |
| UK | United Kingdom |
References
- Gehl, J.; Sersa, G.; Matthiessen, L.W.; Muir, T.; Soden, D.; Occhini, A.; Quaglino, P.; Curatolo, P.; Campana, L.G.; Kunte, C.; Clover, A.J.P.; Bertino, G.; Farricha, V.; Odili, J.; Dahlstrom, K.; Benazzo, M.; Mir, L.M. Updated standard operating procedures for electrochemotherapy of cutaneous tumours and skin metastases. Acta Oncol. 2018 Jul;57(7):874-882. [CrossRef] [PubMed]
- Cabula, C.; Campana, L.G.; Grilz, G.; Galuppo, S.; Bussone, R.; De Meo, L.; Bonadies, A.; Curatolo, P.; De Laurentiis, M.; Renne, M.; Valpione, S.; Fabrizio, T.; Solari, N.; Guida, M.; Santoriello, A.; D'Aiuto, M.; Agresti, R. Electrochemotherapy in the Treatment of Cutaneous Metastases from Breast Cancer: A Multicenter Cohort Analysis. Ann Surg Oncol. 2015 Dec;22 Suppl 3:S442-50. [CrossRef] [PubMed]
- Ricotti, F.; Giuliodori, K.; Cataldi, I.; Campanati, A.; Ganzetti, G.; Ricotti, G.; Offidani, A. Electrochemotherapy: an effective local treatment of cutaneous and subcutaneous melanoma metastases. Dermatol Ther. 2014 May-Jun;27(3):148-52. [CrossRef] [PubMed]
- Michel Marty Gregor Sersa Jean Rémi Garbay Julie Gehl Christopher, G. Collins, Marko Snoj, Valérie Billard, Poul F. Geertsen, John O. Larkin, Damijan Miklavcic, Ivan Pavlovic, Snezna M. Paulin-Kosir, Maja Cemazar, Nassim Morsli, Declan M. Soden, Zvonimir Rudolf, Caroline Robert, Gerald C. O’Sullivan, Lluis M. Mir, Electrochemotherapy – An easy, highly effective and safe treatment of cutaneous and subcutaneous metastases: Results of ESOPE (European Standard Operating Procedures of Electrochemotherapy) study, European Journal of Cancer Supplements. 2006; 4(11): 3-13. [CrossRef]
- Campana, L.G.; Clover, A.J.; Valpione, S.; Quaglino, P.; Gehl, J.; Kunte, C.; Snoj, M.; Cemazar, M.; Rossi, C.R.; Miklavcic, D.; Sersa, G. Recommendations for improving the quality of reporting clinical electrochemotherapy studies based on qualitative systematic review. Radiol Oncol. 2016 Feb 16;50(1):1-13. [CrossRef] [PubMed]
- Bertino, G.; Sersa, G.; De Terlizzi, F.; Occhini, A.; Plaschke, C.C.; Groselj, A.; Langdon, C.; Grau, J.J.; McCaul, J.A.; Heuveling, D.; Cemazar, M.; Strojan, P.; de Bree, R.; Leemans, C.R.; Wessel, I.; Gehl, J.; Benazzo, M. European Research on Electrochemotherapy in Head and Neck Cancer (EURECA) project: Results of the treatment of skin cancer. Eur J Cancer. 2016 Aug;63:41-52. [CrossRef] [PubMed]
- Mir, L.M.; Belehradek, M.; Domenge, C.; et al. [Electrochemotherapy, a new antitumor treatment: first clinical trial] CR Acad Sci III. 1991;313: 613–618.
- Calvet, C.Y.; Mir, L.M. The promising alliance of anti-cancer electrochemotherapy with immunotherapy. Cancer Metastasis Rev. 2016 Jun;35(2):165-77. [CrossRef] [PubMed]
- Sersa, G.; Miklavcic, D.; Cemazar, M.; Rudolf, Z.; Pucihar, G.; Snoj, M. Electrochemotherapy in treatment of tumours. Eur J Surg Oncol. 2008;34:232–240. [CrossRef]
- Miklavcic, D.; Corovic, S.; Pucihar, G.; Pavselj, N. Importance of tumour coverage by sufficiently high local electric field for effective electrochemotherapy. Eur J Cancer Suppl. 2006;4:45–51.
- Miklavcic, D.; Semrov, D.; Mekid, H.; Mir, L.M. A validated model of in vivo electric field distribution in tissues for electrochemotherapy and for DNA electrotransfer for gene therapy. Biochim Biophys Acta. 2000;1523:73–83. [CrossRef]
- Sersa, G.; Cufer, T.; Paulin, S.M.; Cemazar, M.; Snoj, M. Electrochemotherapy of chest wall breast cancer recurrence. Cancer Treat Rev. 2012;38:379–386. [CrossRef]
- Lyons, P,; Polini, D; Russell-Ryan, K,; Clover, A.J.P High-Frequency Electroporation and Chemotherapy for the Treatmetn of Cutaneous Malignancies: Evaluation of Early Clinical Response. Cancers 2023, 15, 3212. [CrossRef]
- Frandsen, S.K.; Vissing, M.; Gehl, J. A Comprehensive Review of Calcium Electroporation -A Novel Cancer Treatment Modality. Cancers (Basel). 2020 Jan 25;12(2):290. [CrossRef] [PubMed]
- Falk, H.; Matthiessen, L.W.; Wooler, G.; Gehl, J. Calcium electroporation for treatment of cutaneous metastases; a randomized double-blinded phase II study, comparing the effect of calcium electroporation with electrochemotherapy. Acta Oncol. 2018 Mar;57(3):311-319. [CrossRef] [PubMed]
- SK Frandsen, L. Gibot, M. Madi, J. Gehl, M.P. Rols. Calcium electroporation: evidence for differential effects in normal and malignant cell lines, evaluated in a 3D spheroid model PLoS One, 10 (12) (2015), pp. 1-11. [CrossRef]
- Anna Szewczyk, Dagmara Baczyńska, Anna Choromańska, Zofia Łapińska, Agnieszka Chwiłkowska, Jolanta Saczko, Julita Kulbacka, Advancing cancer therapy: Mechanisms, efficacy, and limitations of calcium electroporation, Biochimica et Biophysica Acta (BBA) - Reviews on Cancer, Volume 1880, Issue 3,2025. [CrossRef]
- Egeland, C.; Baeksgaard, L.; Gehl, J.; Gögenur, I.; Achiam, M.P. Palliative Treatment of Esophageal Cancer Using Calcium Electroporation. Cancers (Basel). 2022 Oct 27;14(21):5283. [CrossRef] [PubMed]
- Broholm, M.; Vogelsang, R.; Bulut, M.; Stigaard, T.; Falk, H.; Frandsen, S.; Pedersen, D.L.; Perner, T.; Fiehn, A.K.; Mølholm, I.; Bzorek, M.; Rosen, A.W.; Andersen, C.S.A.; Pallisgaard, N.; Gögenur, I.; Gehl, J. Endoscopic calcium electroporation for colorectal cancer: a phase I study. Endosc Int Open. 2023 May 9;11(5):E451-E459. [CrossRef] [PubMed]
- Szewczyk, A.; Rembiałkowska, N.; Saczko, J. et al. Calcium electroporation induces stress response through upregulation of HSP27, HSP70, aspartate β-hydroxylase, and CD133 in human colon cancer cells. Biol Res 58, 10 (2025). [CrossRef]
- Pellegrino, R.; Nacca, V.; Paragliola, F.; Martinelli, E.; Federico, A.; Gravina, A.G. Endoscopic calcium electroporation for unfit-for-surgery bleeding colorectal cancer: the dawn of a new treatment? Minerva Med. 2024 Apr 10. [CrossRef] [PubMed]
- Broholm, M.; Vogelsang, R.; Bulut, M.; Gögenur, M.; Stigaard, T.; Orhan, A.; Schefte, X.; Fiehn, A.M.K.; Gehl, J.; Gögenur, I. Neoadjuvant calcium electroporation for potentially curable colorectal cancer. Surg. Endosc. 2024, 38, 697–705.
- Morgan, E.; Arnold, M.; Gini, A.; Lorenzoni, V.; Cabasag, C.J.; Laversanne, M.; Vignat, J.; Ferlay, J.; Murphy, N.; Bray, F. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023 Feb;72(2):338-344. [CrossRef] [PubMed]
- Young, A.; Rea, D. ABC of colorectal cancer: treatment of advanced disease. BMJ. 2000 Nov 18;321(7271):1278-81. [CrossRef] [PubMed]
- Adeyeye, A.; Olabintan, O.; Ayubi, H.; Gao, H.; Saini, A.; Emmanuel, A.; Hayee, B.; Haji, A. Palliative Luminal Treatment of Colorectal Cancer Using Endoscopic Calcium-Electroporation: First Case Series from United Kingdom. J Clin Med. 2025 Jun 11;14(12):4138. [CrossRef] [PubMed]
- Roe, F.R. How to NAPS: A Discussion of the Safety and Implementation of a Nurse Administered Propofol Sedation (NAPS) Program. Gastroenterol Nurs. 2024 Nov-Dec 01;47(6):467-475. [CrossRef] [PubMed]
- Mandarino, F.V.; Fanti, L.; Barchi, A.; Sinagra, E.; Massimino, L.; Azzolini, F.; Viale, E.; Napolitano, M.; Salmeri, N.; Agostoni, M.; Danese, S. Safety and tolerability outcomes of nonanesthesiologist-administered propofol using target-controlled infusion in routine GI endoscopy. Gastrointest Endosc. 2024 Jun;99(6):914-923. [CrossRef] [PubMed]
- Veitch, A.M.; Radaelli FAlikhan, R.; et al. Endoscopy in patients on antiplatelet or anticoagulant therapy: British Society of Gastroenterology (BSG) and European Society of Gastrointestinal (ESGE) guideline update. Gut 2021; 70:1611-1628.
- https://www.bsg.org.uk/getattachment/33800384-054e-4109-a015-788a09402bf7/BSG-Decontamination-guidance-2020-update.pdf?lang=en-US (accessed 15th August 2025).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



