Submitted:
22 September 2025
Posted:
23 September 2025
You are already at the latest version
Abstract
Keywords:
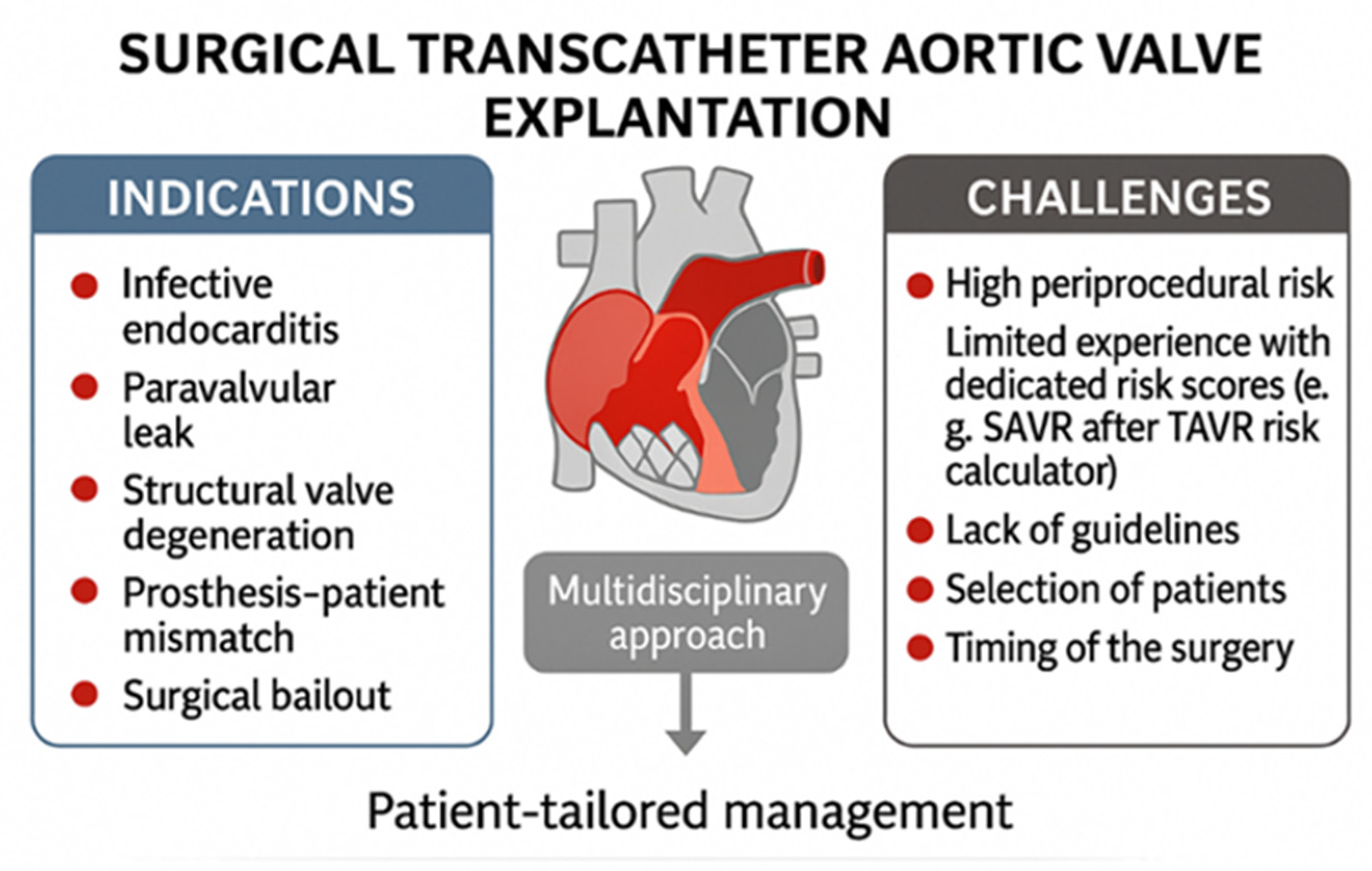
1. Introduction
2. Incidence and Indications for TAVI Explant Surgery
3. EXPLANT-TAVR Registry: Comparison of Self-Expanding and Balloon-Expandable Valves
4. Technical Challenges and Expert Insights for TAVR Explantation
5. The SURPLUS Hybrid Procedure
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Thourani VH, Leon MB, Makkar R, Ascione G, Szeto WY, Madhavan MV, Kodali SK, Hahn RT, Pibarot P, Malaisrie SC, Kapadia SR, Russo MJ, Herrmann HC, Babaliaros V, Guyton R, Genereux P, Cohen DJ, Park B, Clarke S, Gunnarsson M, Szerlip M, Ternacle J, Leipsic J, Blanke P, Webb JG, Smith CR, Mack MJ. Five- Year Outcomes in Low-Risk Patients Undergoing Surgery in the PARTNER 3 Trial. Ann Thorac Surg. 2025 Mar;119(3):555-566.
- Forrest JK, Yakubov SJ, Deeb GM, Gada H, Mumtaz MA, Ramlawi B, Bajwa T, Crouch J, Merhi W, Wai Sang SL, Kleiman NS, Petrossian G, Robinson NB, Sorajja P, Iskander A, Berthoumieu P, Tchétché D, Feindel C, Horlick EM, Saito S, Oh JK, Jung Y, Reardon MJ; Low Risk Trial Investigators. 5-Year Outcomes After Transcatheter or Surgical Aortic Valve Replacement in Low-Risk Patients with Aortic Stenosis.J Am Coll Cardiol. 2025 Apr 22;85(15):1523-1532. [CrossRef]
- Kaneko T, Bapat VN, Alakhtar AM, Zaid S, George I, Grubb KJ, Harrington K, Pirelli L, Atkins M, Desai ND et al.: Transcatheter heart valve explantation for transcatheter aortic valve replacement failure: A Heart Valve Collaboratory expert consensus document on operative techniques. J Thorac Cardiovasc Surg 2025, 169(3):878-889.
- Durand E, Eltchaninoff H, Tchetche D, Levesque T, Garmendia C, Iung B, Benamer H, Cayla G, Van Belle E, Commeau P, Le Breton H, Cuisset T, Akodad M, Verhoye JP, Beurtheret S, du Chayla F, Leclere M, Gilard M; STOP-AS and France TAVI Investigators; Predictors of Outcomes of Reintervention After Transcatheter Aortic Valve Replacement: FRANCE 2 and FRANCE TAVI Registries N. J Am Coll Cardiol. 2025 Mar 11;85(9):896-907. [CrossRef]
- Zaid S, Kleiman NS, Goel SS et al. Impact of transcatheter valve type on out-comes of surgical explantation after failed transcatheter aortic valve replacement: the EXPLANT-TAVR international registry. EuroIntervention 2024;20: e146–57.
- Bapat VN, Zaid S, Fukuhara S, Saha S, Vitanova K, Kiefer P, Squiers JJ, Voisine P, Pi-relli L, von Ballmoos MW, Chu MWA, Rodés-Cabau J, DiMaio JM, Borger MA, Lange R, Hagl C, Denti P, Modine T, Kaneko T, Tang GHL; EXPLANT-TAVR Investigators. Surgi-cal Explantation After TAVR Failure: Mid-Term Outcomes From the EXPLANT-TAVR International Registry. JACC Cardiovasc Interv. 2021 Sep 27;14(18):1978-1991.
- Pineda AM, Harrison JK, Kleiman NS, Rihal CS, Kodali SK, Kirtane AJ, Leon MB, Sherwood MW, Manandhar P, Vemulapalli S, Beohar N. Incidence and Outcomes of Surgical Bailout During TAVR: Insights From the STS/ACC TVT Registry.JACC Cardi-ovasc Interv. 2019 Sep 23;12(18):1751-1764.
- Malvindi PG, Luthra S, Sarvananthan S, Zingale A, Olevano C, Ohri S. Surgical treatment of transcatheter aortic valve infective endocarditis. Neth Heart J. 2021 Feb;29(2):71-77.
- Calcaterra D, Harris K, Goessl M, Dasari G, Kaur N, Chavez I. Findings of prosthetic valve endocarditis in the balloon-expandable trans-catheter aortic valve: review of the literature and tips of management.J Cardiothorac Surg. 2021 Aug 16;16(1):234.
- Pisani A, Hounat F, Brega C, Borghese O, Braham W, Alkhoder S. Infective endocarditis following transcatheter aortic valve implantation. Ann Cardiol Angeiol (Paris). 2020 Oct;69(4):204-206.
- Fukuhara S, Suzuki T, Deeb GM, Ailawadi G, Patel HJ, Yang B. Clinical outcomes of TAVR explant stratified by original risk profile: insights from 110 TAVR explants. Ann Cardiothorac Surg. 2025 Mar 31;14(2):122-130.
- Τhe Society of Thoracic Surgeons. SAVR after TAVR Risk Calculator. Accessed August 19, 2025. https://www.sts.org/resources/savr-after-tavr-risk-calculator and https://savr-after-tavrriskcalc.research.sts.org/.
- Sakakura R, Fukuzawa M, Sugiyama H, Tani K, Yoshida T, Murakami A, Terai H, Ueyama K. Early surgical explantation of a TAVI valve for severe hemolytic outcomes caused by mild paravalvular leak. Gen Thorac Cardiovasc Surg Cases. 2025 Jul 28;4(1):35.
- Fukuhara, S. Safe late explantation of transcatheter aortic bioprosthesis. Ann Thorac Surg. 2020; 110: e555-e558.
- Kurosaka K, Iino K, Yamamoto Y, Takemura H. Recapture technique for the surgical explantation of an infected self-expanding prosthesis after transcatheter aortic valve re-placement prior to surgical aortic valve replacement. Interdiscip Cardiovasc Thorac Surg. 2025 Jun 4;40(6): ivaf135.
- Ogami T, Ridgley J, Serna-Gallegos D, Kliner DE, Toma C, Sanon S, Brown JA, Yousef S, Sultan I. Outcomes of Surgical Aortic Valve Replacement After Transcatheter Aortic Valve Implantation. Am J Cardiol. 2022 Nov 1; 182:63-68.
- Fukuhara S, Kim KM, Yang B, Romano M, Ailawadi G, Patel HJ, Deeb GM. Reoperation following transcatheter aortic valve replacement: Insights from 10 years’ experience. J Thorac Cardiovasc Surg. 2024 Aug;168(2):488-497.e3.
- Fukuhara, S. TAVR reintervention strategies: unveiling trends and outcomes of redo TAVR and TAVR explant., New York Valves 2025, New York, USA, 25/06/2025.
| Indication | SEV (%) | BEV (%) | p-value |
|---|---|---|---|
| Endocarditis | 36.0 | 55.4 | <0.001 |
| Paravalvular leak | 21.2 | 11.9 | 0.014 |
| Structural valve deterioration | 30.2 | 21.8 | 0.065 |
| Prosthesis-patient mismatch | 8.5 | 10.4 | 0.61 |
| Group | Urgent/Emergency Surgeries (%) | p-value (Urgent/Emergency) | Root Replacement (%) | p-value (Root Replacement) |
|---|---|---|---|---|
| SEV | 52.0 | 0.057 | 15.3 | 0.016 |
| BEV | 62.3 | 0.057 | 7.4 | 0.016 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




