Submitted:
19 September 2025
Posted:
23 September 2025
You are already at the latest version
Abstract
Keywords:
Outline
- Intro
- The pathophysiology & importance of congestion
- The Importance of the Inferior Vena Cava
-
Clinical Assessment of Fluid Tolerance of Key Organ Systems
- The respiratory system
- The cardiovascular system
- The kidneys
- The abdomen
- The central nervous system
- The skin and soft tissues
- Discussion
- Conclusion
- References
1. Introduction
1.1. The Pathophysiological Consequences of Fluid Overload and Venous Congestion
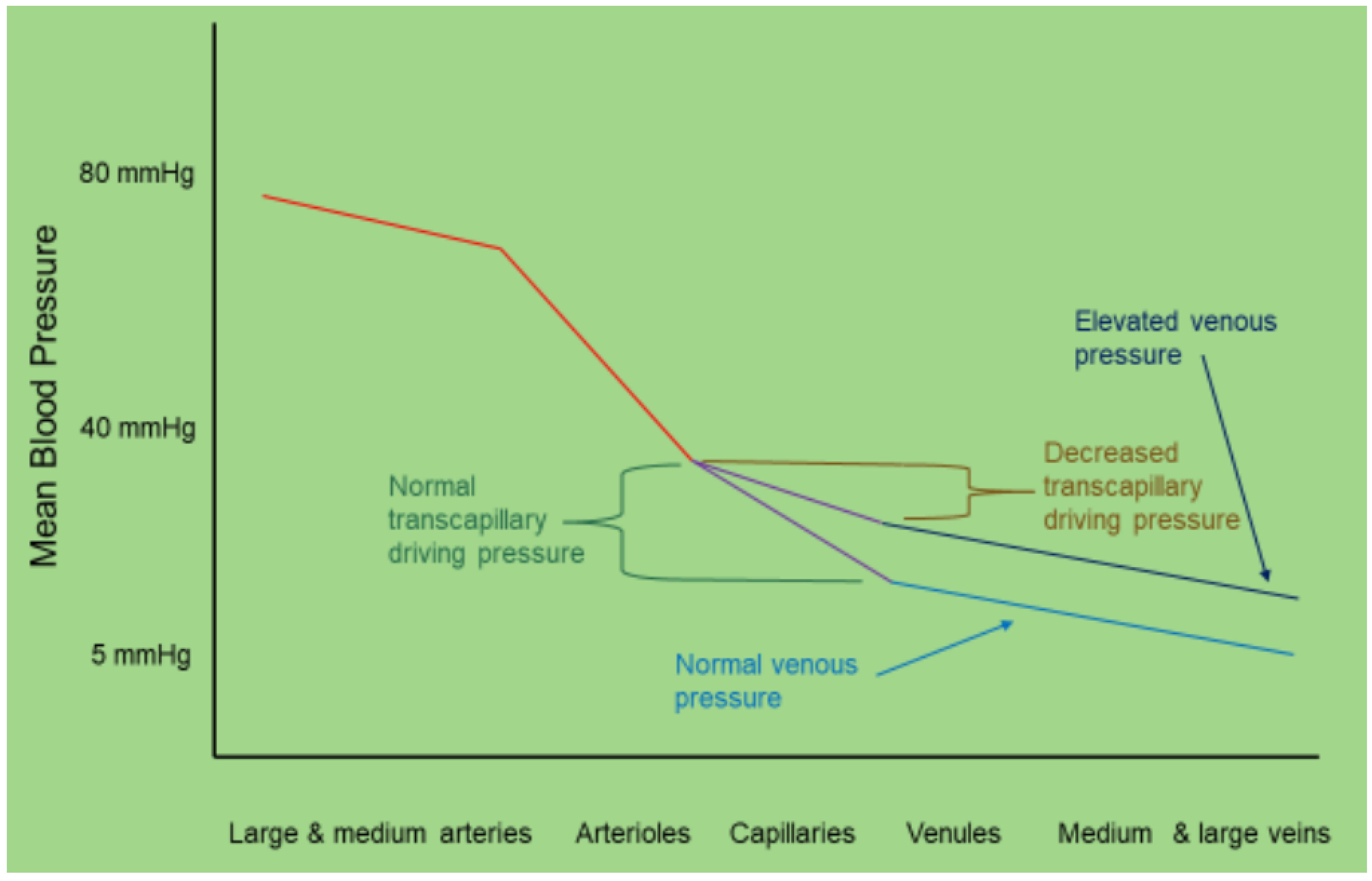
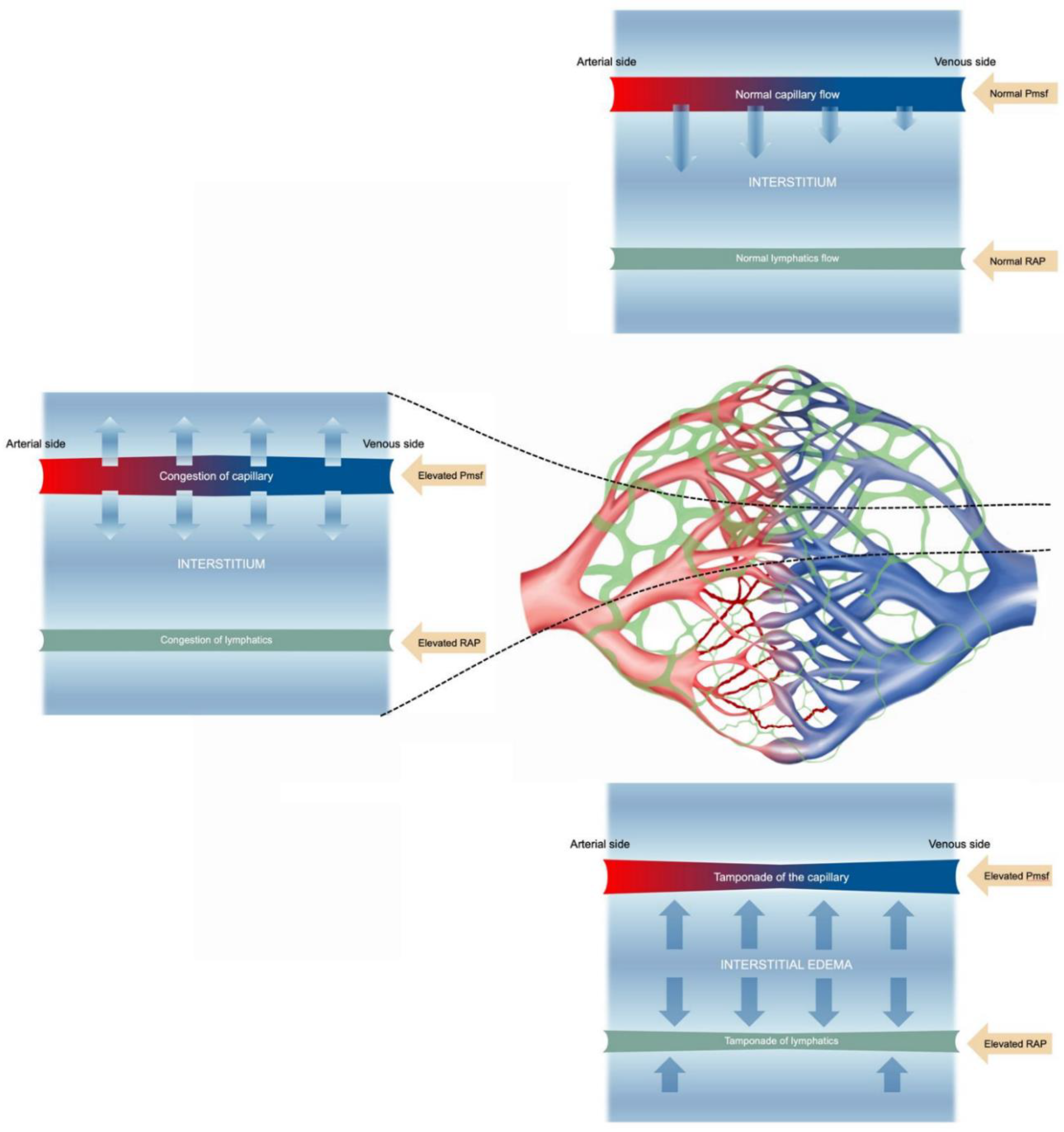
1.2. In defense of the Central Venous Pressure and Inferior Vena Cava
1.3. Clinical Assessment of Fluid Tolerance: An Organ Based Approach
1.3.1. Device-Based Assessments
Noninvasive Monitoring
Invasive Monitoring
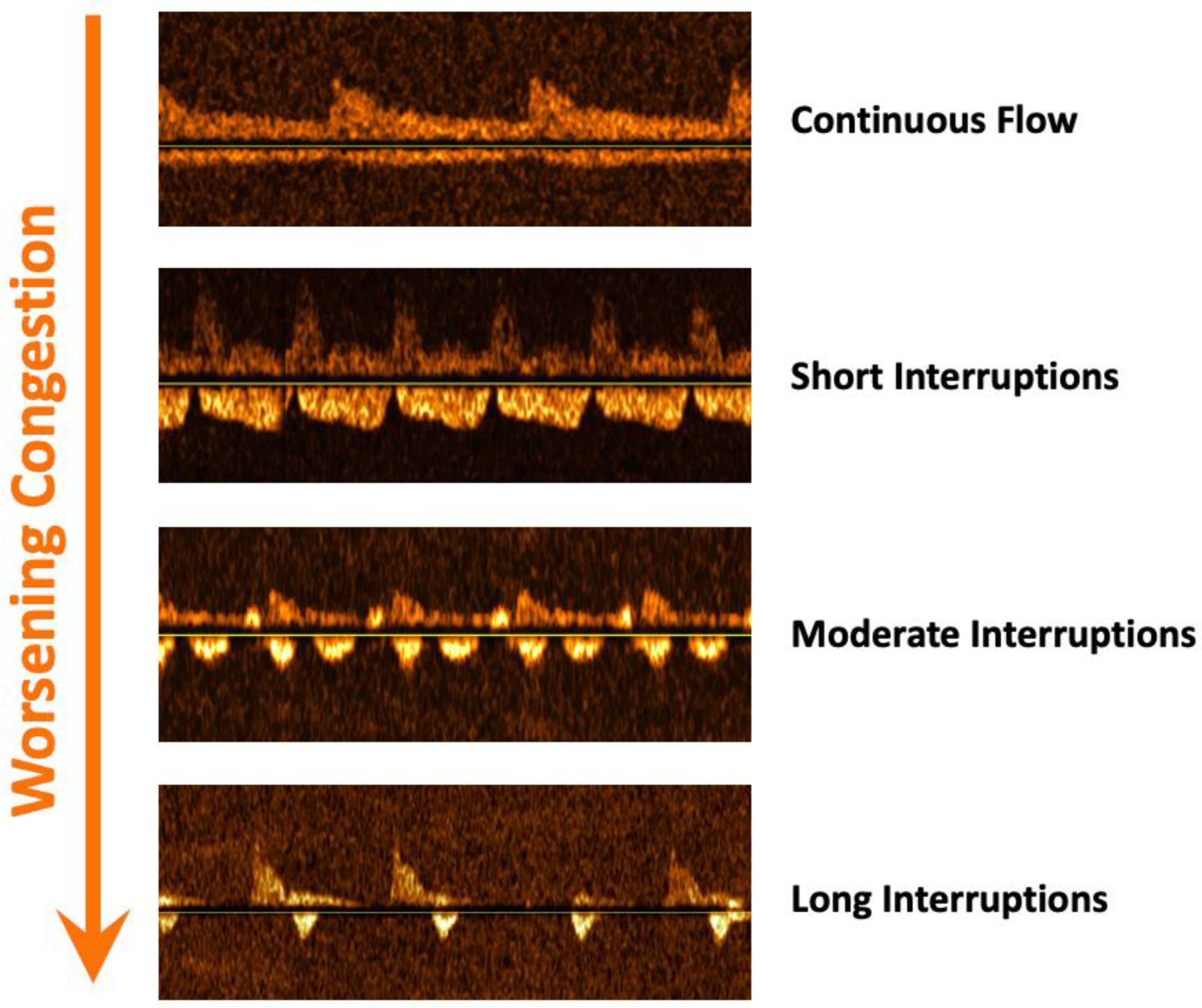
1.3.2. Clinical Assessment
At the Bedside
At the Bedside
1.4. Fluid Intolerance - Putting it All Together
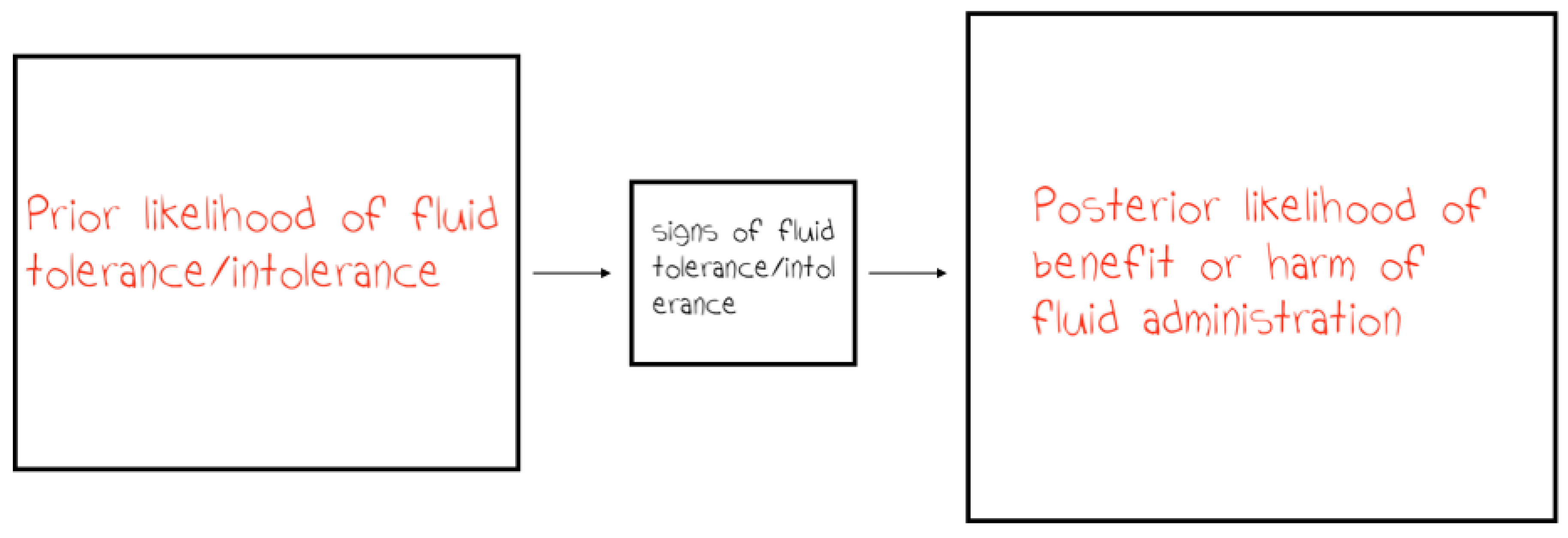
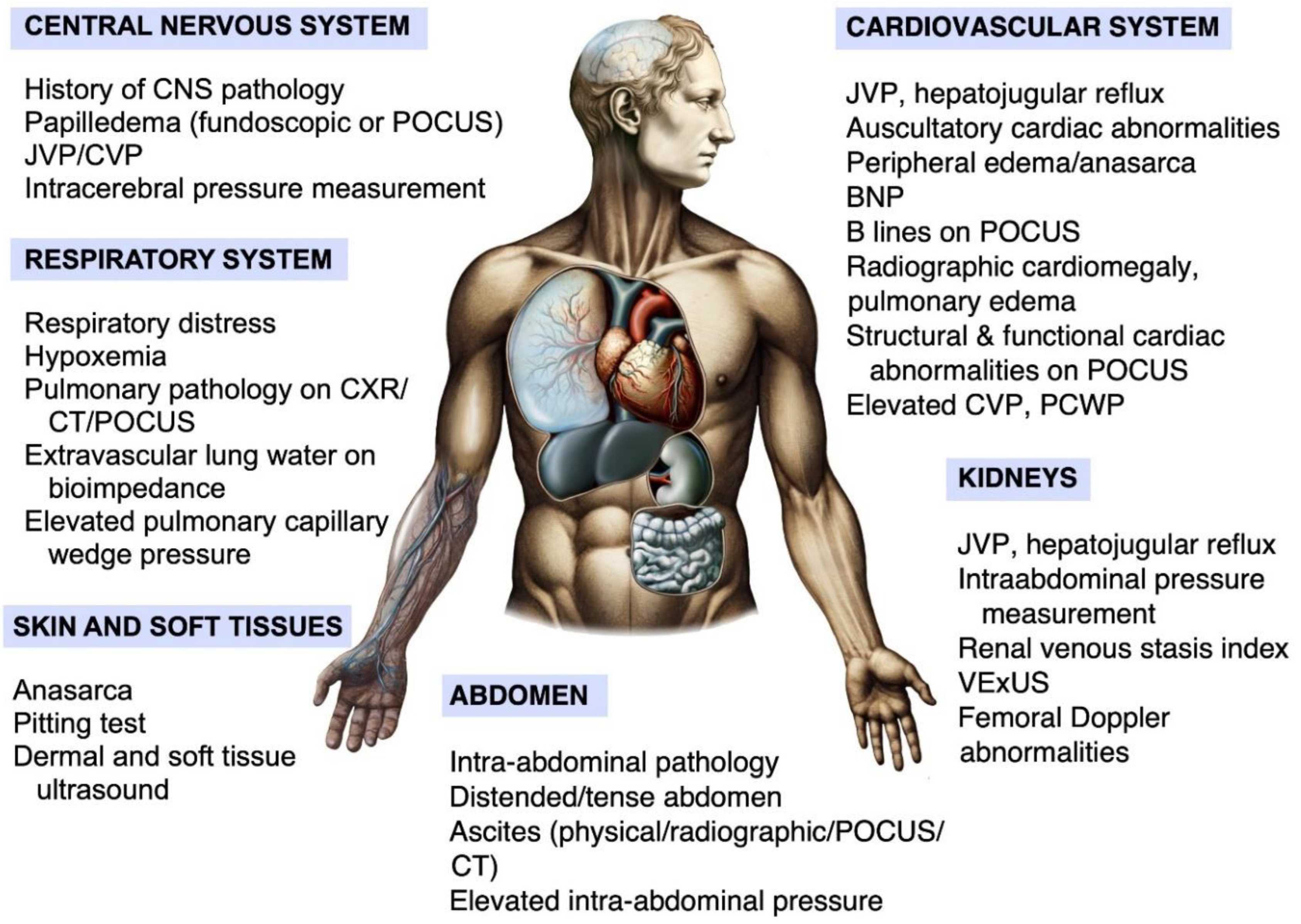
2. Summary
References
- Bakker J, Kattan E, Annane D, Castro R, Cecconi M, de Backer D, et al. Current practice and evolving concepts in septic shock resuscitation. Intensive Care Med 2022;48:148–63. [CrossRef]
- Bentzer P, Griesdale DE, Boyd J, MacLean K, Sirounis D, Ayas NT. Will this hemodynamically unstable patient respond to a bolus of intravenous fluids? JAMA - J Am Med Assoc 2016;316:1298–309. [CrossRef]
- Boyd JH, Frcp C, Forbes J, Nakada T, Walley KR, Frcp C, et al. Fluid resuscitation in septic shock: A positive fluid balance and elevated central venous pressure are associated with increased mortality* 2011;39:259–65. [CrossRef]
- Bagshaw SM, Brophy PD, Cruz D, Ronco C. Fluid balance as a biomarker : impact of fluid overload on outcome in critically ill patients with acute kidney injury. Crit Care 2008;12:1–3. [CrossRef]
- Monnet X, Shi R, Teboul J-L. Prediction of fluid responsiveness. What’s new? Ann Intensive Care 2022;12:46. [CrossRef]
- Kattan E, Ospina-Tascón GA, Teboul J-L, Castro R, Cecconi M, Ferri G, et al. Systematic assessment of fluid responsiveness during early septic shock resuscitation: Secondary analysis of the ANDROMEDA-SHOCK trial. Crit Care 2020;24. [CrossRef]
- Muñoz F, Born P, Bruna M, Ulloa R, González C, Philp V, et al. Coexistence of a fluid responsive state and venous congestion signals in critically ill patients: a multicenter observational proof-of-concept study. Crit Care 2024;28:52. [CrossRef]
- Aya HD, Ster IC, Fletcher N, Grounds RM, Rhodes A, Cecconi M. Pharmacodynamic analysis of a fluid challenge. Crit Care Med 2016;44:880–91. [CrossRef]
- Kattan E, Castro R, Miralles-Aguiar F, Hernandez G RP. The emerging concept of Fluid Tolerance : a position paper. J Crit Care 2022;71. [CrossRef]
- Winton, FR. The influence of venous pressure on the isolated mammalian kidney. J Physiol 1931;72:49. [CrossRef]
- Pinsky MR, García MIM, Dubin A. Significance of critical closing pressures (starling resistors) in arterial circulation. Crit Care 2024;28:1–3. [CrossRef]
- Maas JJ, De Wilde RB, Aarts LP, Pinsky MR, Jansen JR. Determination of Vascular Waterfall Phenomenon by Bedside Measurement of Mean Systemic Filling Pressure and Critical Closing Pressure in the Intensive Care Unit. Anesth Analg 2012;114:803. [CrossRef]
- Andrei S, Bar S, Nguyen M, Bouhemad B, Guinot PG. Effect of norepinephrine on the vascular waterfall and tissue perfusion in vasoplegic hypotensive patients: a prospective, observational, applied physiology study in cardiac surgery. Intensive Care Med Exp 2023;11:1–11. [CrossRef]
- Kara A, Akin S, Ince C. Monitoring microcirculation in critical illness. Curr Opin Crit Care 2016;22:444–52. [CrossRef]
- Ince, C. Hemodynamic coherence and the rationale for monitoring the microcirculation. Crit Care 2015;19:S8. [CrossRef]
- Vellinga NAR, Ince C, Boerma EC. Elevated central venous pressure is associated with impairment of microcirculatory blood flow in sepsis: A hypothesis generating post hoc analysis. BMC Anesthesiol 2013;13. [CrossRef]
- Berlin DA, Bakker J. Starling curves and central venous pressure. Crit Care 2014;19. [CrossRef]
- Ruste M, Reskot R, Schweizer R, Mayet V, Fellahi JL, Lagrèze MJ. Changes in portal pulsatility index induced by a fluid challenge in patients with haemodynamic instability and systemic venous congestion : a prospective cohort study. Ann Intensive Care 2024. [CrossRef]
- Woodcock TE, Woodcock TM. Revised Starling equation and the glycocalyx model of transvascular fluid exchange: An improved paradigm for prescribing intravenous fluid therapy. Br J Anaesth 2012;108:384–94. [CrossRef]
- Viana-Rojas JA, Argaiz E, Robles-Ledesma M, Arias-Mendoza A, Nájera-Rojas NA, Alonso-Bringas AP, et al. Venous excess ultrasound score and acute kidney injury in patients with acute coronary syndrome. Eur Hear J Acute Cardiovasc Care 2023;12:413–9. [CrossRef]
- Longino A, Martin K, Leyba K, Siegel G, Gill E, Douglas IS, et al. Correlation between the VExUS score and right atrial pressure: a pilot prospective observational study. Crit Care 2023;27:1–5. [CrossRef]
- Beaubien-Souligny W, Rola P, Haycock K, Bouchard J, Lamarche Y, Spiegel R, et al. Quantifying systemic congestion with Point-Of-Care ultrasound: development of the venous excess ultrasound grading system. Ultrasound J 2020;12. [CrossRef]
- De Backer D, Vincent JL. Should we measure the central venous pressure to guide fluid management? Ten answers to 10 questions. Crit Care 2018;22:1–6. [CrossRef]
- Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, et al. Early-goal directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368–77.
- Rola P, Haycock K, Spiegel R. What every intensivist should know about the IVC. J Crit Care 2024;80. [CrossRef]
- Persichini R, Lai C, Teboul JL, Adda I, Guérin L, Monnet X. Venous return and mean systemic filling pressure: physiology and clinical applications. Crit Care 2022;26:1–11. [CrossRef]
- Malbrain MLNG, Marik PE, Witters I, Cordemans C, Kirkpatrick AW, Roberts DJ, et al. Fluid overload, de-resuscitation, and outcomes in critically ill or injured patients: A systematic review with suggestions for clinical practice. Anaesthesiol Intensive Ther 2014;46:361–80. [CrossRef]
- Finfer S, Myburgh J, Bellomo R. Intravenous fluid therapy in critically ill adults. Nat Rev Nephrol 2018;14:541–57. [CrossRef]
- Mojoli F, Bouhemad B, Mongodi S, Lichtenstein D. Lung ultrasound for critically ill patients. Am J Respir Crit Care Med 2019;199:701–14. [CrossRef]
- Lichtenstein, DA. Lung ultrasound in the critically ill. Ann Intensive Care 2014;4:1. [CrossRef]
- Koratala A, Ronco C, Kazory A. Diagnosis of Fluid Overload: From Conventional to Contemporary Concepts. Cardiorenal Med 2022;12:141–54. [CrossRef]
- Peacock WF, Albert NM, White RD, Emerman CL. Bioimpedance monitoring: Better than chest x-ray for predicting abnormal pulmonary fluid? Congest Hear Fail 2000;6:86–9. [CrossRef]
- Hammad Y, Hasanin A, Elsakka A, Refaie A, Abdelfattah D, Rahman SA, et al. Thoracic fluid content: a novel parameter for detection of pulmonary edema in parturients with preeclampsia. J Clin Monit Comput 2019;33:413–8. [CrossRef]
- Ware LB, Matthay MA. Clinical practice. Acute pulmonary edema. N Engl J Med 2005;353:2788–96. [CrossRef]
- Jozwiak M, Teboul JL, Monnet X. Extravascular lung water in critical care: recent advances and clinical applications. Ann Intensive Care 2015;5:1–13. [CrossRef]
- López-Candales A, Edelman K. Shape of the right ventricular outflow Doppler envelope and severity of pulmonary hypertension. Eur J Echocardiogr 2012;13:309–16. [CrossRef]
- Vieillard-Baron A, Millington SJ, Sanfilippo F, Chew M, Diaz-Gomez J, McLean A, et al. A decade of progress in critical care echocardiography: a narrative review. Intensive Care Med 2019;45:770–88. [CrossRef]
- Nagueh SF, Smiseth OA, Appleton CP, Byrd BF, Dokainish H, Edvardsen T, et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 2016;29:277–314. [CrossRef]
- Orde S, Slama M, Hilton A, Yastrebov K, McLean A. Pearls and pitfalls in comprehensive critical care echocardiography. Crit Care 2017;21. [CrossRef]
- Brault C, Marc J, Mercado P, Diouf M, Tribouilloy C, Zerbib Y, et al. Estimation of Pulmonary Artery Occlusion Pressure Using Doppler Echocardiography in Mechanically Ventilated Patients. Crit Care Med 2020;48:E943–50. [CrossRef]
- Magder, S. Right Atrial Pressure in the Critically Ill: How to Measure, What Is the Value, What Are the Limitations? Chest 2017;151:908–16. [CrossRef]
- Rako ZA, Yogeswaran A, Yildiz S, Weidemann P, Zedler D, da Rocha BB, et al. Liver stiffness is associated with right heart dysfunction, cardiohepatic syndrome, and prognosis in pulmonary hypertension. J Heart Lung Transplant 2024;43:1105. [CrossRef]
- Beaubien-Souligny W, Benkreira A, Robillard P, Bouabdallaoui N, Chassé M, Desjardins G, et al. Alterations in Portal Vein Flow and Intrarenal Venous Flow Are Associated With Acute Kidney Injury After Cardiac Surgery: A Prospective Observational Cohort Study. J Am Heart Assoc 2018;7. [CrossRef]
- Gómez-Rodríguez C, Tadeo-Espinoza H, Solis-Huerta F, Leal-Villarreal MA de J, Guerrero-Cabrera P, Cruz N, et al. Hemodynamic Evaluation of Right-Sided Congestion With Doppler Ultrasonography in Pulmonary Hypertension. Am J Cardiol 2023;203:459–62. [CrossRef]
- Husain-Syed F, Birk HW, Ronco C, Schörmann T, Tello K, Richter MJ, et al. Doppler-Derived Renal Venous Stasis Index in the Prognosis of Right Heart Failure. J Am Heart Assoc 2019;8. [CrossRef]
- Fujii K, Nakayama I, Izawa J, Iida N, Seo Y, Yamamoto M, et al. Association between intrarenal venous flow from Doppler ultrasonography and acute kidney injury in patients with sepsis in critical care: a prospective, exploratory observational study. Crit Care 2023;27:278. [CrossRef]
- Spiegel R, Teeter W, Sullivan S, Tupchong K, Mohammed N, Sutherland M, et al. The use of venous Doppler to predict adverse kidney events in a general ICU cohort. Crit Care 2020;24. [CrossRef]
- Argaiz ER, Rola P, Haycock KH, Verbrugge FH. Fluid management in acute kidney injury: from evaluating fluid responsiveness towards assessment of fluid tolerance. Eur Hear Journal Acute Cardiovasc Care 2022;11:786–93. [CrossRef]
- Argaiz, ER. VExUS Nexus: Bedside Assessment of Venous Congestion. Adv Chronic Kidney Dis 2021;28:252–61. [CrossRef]
- Prager R, Arntfield R, Wong MYS, Ball I, Lewis K, Rochwerg B, et al. Venous congestion in septic shock quantified with point-of-care ultrasound: a pilot prospective multicentre cohort study. Can J Anaesth 2024. [CrossRef]
- De Laet IE, Malbrain MLNG, De Waele JJ. A Clinician’s Guide to Management of Intra-Abdominal Hypertension and Abdominal Compartment Syndrome in Critically Ill Patients. Crit Care 2020;24:1–9. [CrossRef]
- Kirkpatrick AW, Roberts DJ, De Waele J, Jaeschke R, Malbrain MLNG, De Keulenaer B, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: Updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 2013;39:1190–206. [CrossRef]
- Perez-Calatayud AA, Carrillo-Esper R, Anica-Malagon ED, Briones-Garduño JC, Arch-Tirado E, Wise R, et al. Point-of-care gastrointestinal and urinary tract sonography in daily evaluation of gastrointestinal dysfunction in critically ill patients (GUTS Protocol). Anaesthesiol Intensive Ther 2018;50:40–8. [CrossRef]
- Wong A, Vieillard-Baron A, Malbrain MLNG. Emergency bedside ultrasound: benefits as well as caution - part 1. General. Curr Opin Crit Care 2019;25:613–21. [CrossRef]
- Malbrain MLNG, De Tavernier B, Haverals S, Slama M, Vieillard-Baron A, Wong A, et al. Executive summary on the use of ultrasound in the critically ill: consensus report from the 3rd Course on Acute Care Ultrasound (CACU). Anaesthesiol Intensive Ther 2017;49:393–411. [CrossRef]
- Severgnini P, Inzigneri G, Olvera C, Fugazzola C, Mangini M, Padalino P, et al. New and old tools for abdominal imaging in critically ill patients. Acta Clin Belg 2007;62:173–82. [CrossRef]
- Malbrain MLNG, Chiumello D, Pelosi P, Bihari D, Innes R, Ranieri VM, et al. Incidence and prognosis of intraabdominal hypertension in a mixed population of critically ill patients: A multiple-center epidemiological study*. Crit Care Med 2005;33:315–22.
- Aldabal L, Bahammam AS. Cheyne-stokes respiration in patients with heart failure. Lung 2010;188:5–14. [CrossRef]
- Benkreira A, Beaubien-Souligny W, Mailhot T, Bouabdallaoui N, Robillard P, Desjardins G, et al. Portal Hypertension Is Associated With Congestive Encephalopathy and Delirium After Cardiac Surgery. Can J Cardiol 2019;35:1134–41. [CrossRef]
- Ameloot K, Genbrugge C, Meex I, Eertmans W, Jans F, De Deyne C, et al. Is venous congestion associated with reduced cerebral oxygenation and worse neurological outcome after cardiac arrest? Crit Care 2016;20:1–8. [CrossRef]
- Minato S, Ookawara S, Ito K, Hayasaka H, Kofuji M, Uchida T, et al. Continuous monitoring of changes in cerebral oxygenation during hemodialysis in a patient with acute congestive heart failure. J Artif Organs 2020;23:292–5. [CrossRef]
- Selhorst JB, Gudeman SK, Butterworth JF, Harbison JW, Miller JD, Becker DP. Papilledema after acute head injury. Neurosurgery 1985;16:357–63. [CrossRef]
- Stead GA, Cresswell F V., Jjunju S, Oanh PKN, Thwaites GE, Donovan J. The role of optic nerve sheath diameter ultrasound in brain infection. ENeurologicalSci 2021;23. [CrossRef]
- Canac N, Jalaleddini K, Thorpe SG, Thibeault CM, Hamilton RB. Review: pathophysiology of intracranial hypertension and noninvasive intracranial pressure monitoring. Fluids Barriers CNS 2020;17. [CrossRef]
- Danziger J, Chen K, Cavender S, Lee J, Feng M, Mark RG, et al. Admission Peripheral Edema, Central Venous Pressure, and Survival in Critically Ill Patients. Ann Am Thorac Soc 2016;13:705–11. [CrossRef]
- Chen C, Lee J, Johnson AE, Mark RG, Celi LA, Danziger J. Right Ventricular Function, Peripheral Edema, and Acute Kidney Injury in Critical Illness. Kidney Int Reports 2017;2:1059. [CrossRef]
- Zhang W, Gu Y, Zhao Y, Lian J, Zeng Q, Wang X, et al. Focused liquid ultrasonography in dropsy protocol for quantitative assessment of subcutaneous edema. Crit Care 2023;27:1–12. [CrossRef]
- De Backer D, Aissaoui N, Cecconi M, Chew MS, Denault A, Hajjar L, et al. Intensive Care Med 2022. [CrossRef]
- De Backer D, Cecconi M, Chew MS, Hajjar L, Monnet X, Ospina-Tascón GA, et al. A plea for personalization of the hemodynamic management of septic shock. Crit Care 2022;26:1–13. [CrossRef]
- Pfortmueller CA, Dabrowski W, Wise R, van Regenmortel N, Malbrain MLNG. Fluid accumulation syndrome in sepsis and septic shock: pathophysiology, relevance and treatment—a comprehensive review. Ann Intensive Care 2024;14. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




