Submitted:
20 September 2025
Posted:
22 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Role of Emotion Regulation Networks in MDD
2.1. Role of Limbic System Disruptions in MDD
2.2. The Role of PFC Disruptions in Depression
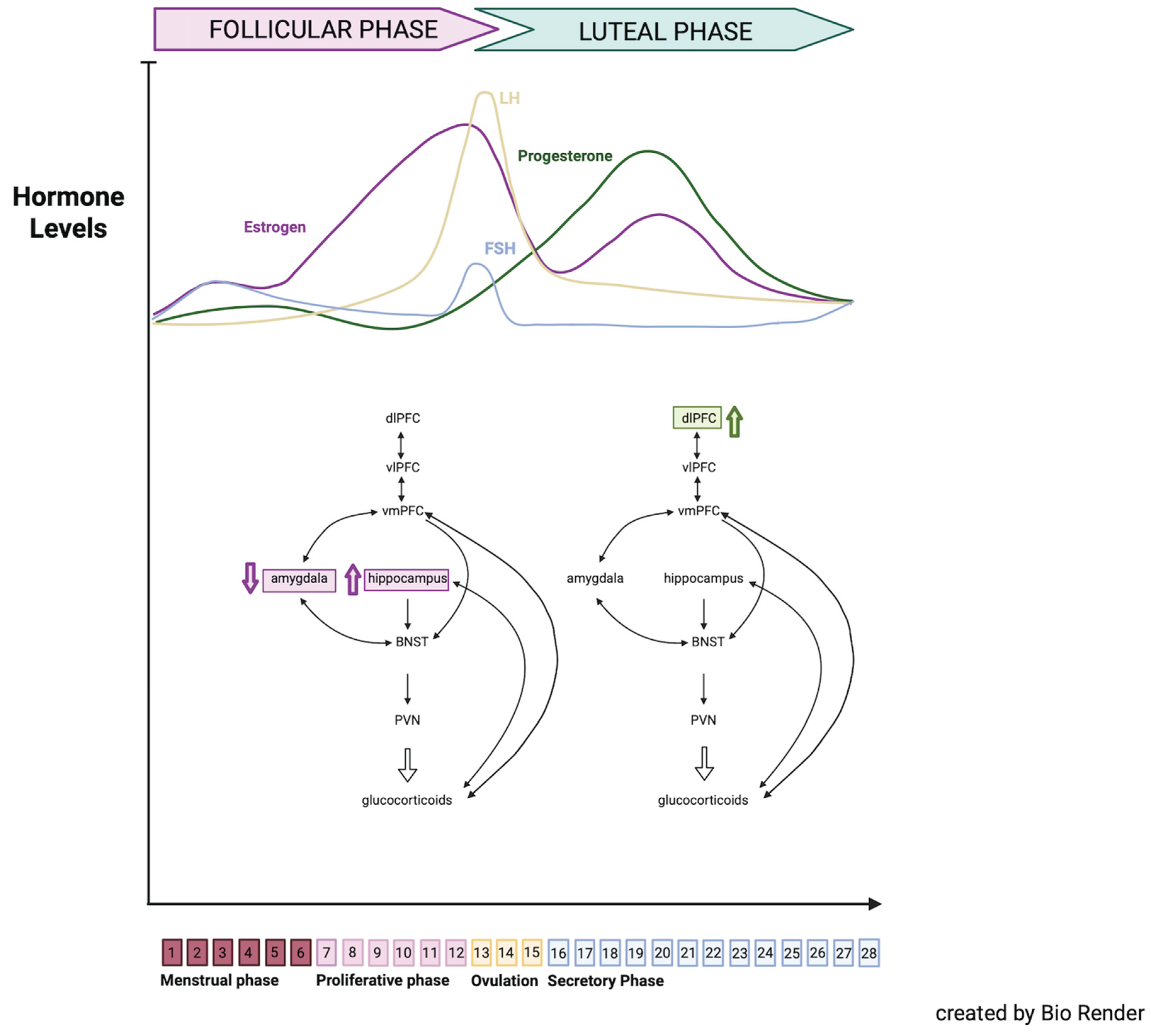
3. The Influence of Fluctuations in Female Gonadal Hormones on Emotion Regulation Networks
3.1. Hormonal Fluctuations Along the Menstrual Cycle
3.2. Influence of Hormonal Fluctuations Along the Menstrual Cycle on Hippocampal Structure and Function
3.3. Influence of Hormonal Fluctuations Along the Menstrual Cycle on Amygdala Structure and Function
3.4. Influence of Hormonal Fluctuations Along the Menstrual Cycle on dlPFC Structure and Function
3.5. Synergistic Effects of Hormonal Fluctuations and Stress Reactivity on Psychiatric Symptoms
4. HPA Axis
4.1. HPA Axis Regulation Through Harmful Feedback Mechanisms
4.2. HPA Axis and Female Gonadal Hormones
5. GABAergic System
5.1. GABAergic System and Female Gonadal Hormones
6. Perineuronal Nets
6.1. Modulation of PNNs by Exposure to Female Gonadal Hormones
6.2. The Role of Microglia in PNN Remodeling
7. Microglia
7.1. Microglial Involvement in Neuroplasticity
7.2. Microglial Modulation Through Hormonal Exposure
7.3. Microglial Modulation by Glucocorticoids
7.4. Microglial Role in the Shaping of Interneuronal Microcircuits
8. Conclusion and Future Perspectives
Abbreviation List
| GABA | gamma-aminobutyric acid |
| ACC | anterior cingulate cortex |
| ACTH | adrenocorticotropic hormones |
| AVP | arginine vasopressin |
| BDNF | brain-derived neurotrophic factor |
| BLA | basolateral amygdala |
| BNST | bed nucleus of the stria terminalis |
| C3/C3aR | complement component 3/complement component 3a receptor |
| CD11b | cluster of differentiation 11b |
| CeA | central nucleus of the amygdala |
| Cl- | chloride ion |
| CNS | central nervous system |
| CRH | corticotropin-releasing hormone |
| CSPGs | chondroitin sulphate proteoglycans |
| dlPFC | dorsolateral PFC |
| dmPFC | dorsomedial PFC |
| DSM-5 | Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition |
| ERα | estrogen receptors alpha |
| ERβ | estrogen receptors beta |
| fMRI | functional magnetic resonance imaging |
| GAD | glutamic acid decarboxylase |
| GPER | G protein–coupled estrogen receptor |
| GR | glucocorticoid receptors |
| HPA | hypothalamic pituitary adrenal axis |
| Iba1 | ionized calcium-binding adapter molecule 1 |
| IL-12p40 | interleukin 12p40 |
| IL-12p40 | interleukin-12 subunit beta |
| IL-1β | interleukin-1 beta |
| IL-6 | interleukin-6 |
| iNOS | NO synthase |
| LH | luteinizing hormone |
| LPS | lipopolysaccharide |
| MDD | major depressive disorder |
| MMP-9 | metalloproteinase-9 |
| mPFC | medial prefrontal cortex |
| mPOA | medial preoptic area |
| MR | mineralocorticoid receptors |
| MRI | magnetic resonance imaging |
| MRS | magnetic resonance spectroscopy |
| NLRP3 | NOD-like receptor family pyrin domain containing 3 |
| NO | nitric oxide |
| P75^NTR | p75 neurotrophin receptor |
| PAM | positive allosteric modulator |
| PANAS | Positive and Negative Affect Schedule |
| PET | positron emission tomography |
| PFC | prefrontal cortex |
| PGE(2) | prostaglandin-E(2) |
| PMS | premenstrual syndrome |
| PNN | perineuronal nets |
| PV | parvalbumin |
| PVN | paraventricular nucleus |
| RCT | randomized control trials |
| rs-fMRI | resting state functional magnetic imaging |
| rsFC | resting state functional connectivity |
| rsfMRI | resting state functional magnetic resonance imaging |
| spDCM | spectral dynamic causal modelling |
| SSRIs | selective serotonin reuptake inhibitors |
| SST | somatostatin |
| TMS | transcranial magnetic stimulation |
| TNFα | tumor necrosis factor alpha |
| TrkB | tropomyosin receptor kinase B |
| TSPO | 18 kDa translocator protein |
| TSST | Trier Social Stress Test |
| vlPFC | ventrolateral prefrontal cortex |
| vmPFC | ventromedial prefrontal cortex |
| WHO | World Health Organization |
References
- Zhang Y, Jia X, Yang Y, Sun N, Shi S, Wang W. Change in the global burden of depression from 1990-2019 and its prediction for 2030. J Psychiatr Res. 2024;178:16-22. [CrossRef]
- Lépine JP, Briley M. The increasing burden of depression. Neuropsychiatr Dis Treat. 2011;7(Suppl 1):3-7. [CrossRef]
- Uher R, Payne JL, Pavlova B, Perlis RH. Major Depressive Disorder in Dsm-5: Implications for Clinical Practice and Research of Changes from Dsm-Iv. Depress Anxiety. 2014;31(6):459-471. [CrossRef]
- Tolentino JC, Schmidt SL. DSM-5 Criteria and Depression Severity: Implications for Clinical Practice. Front Psychiatry. 2018;9:450. [CrossRef]
- Gonda X, Pompili M, Serafini G, Carvalho AF, Rihmer Z, Dome P. The role of cognitive dysfunction in the symptoms and remission from depression. Ann Gen Psychiatry. 2015;14(1):27. [CrossRef]
- Medina A, Seasholtz AF, Sharma V, et al. Glucocorticoid and mineralocorticoid receptor expression in the human hippocampus in major depressive disorder. J Psychiatr Res. 2013;47(3):307-314. [CrossRef]
- Wiersma JE, Hovens JGFM, van Oppen P, et al. The importance of childhood trauma and childhood life events for chronicity of depression in adults. J Clin Psychiatry. 2009;70(7):983-989. [CrossRef]
- Bisht K, Sharma K, Tremblay MÈ. Chronic stress as a risk factor for Alzheimer’s disease: Roles of microglia-mediated synaptic remodeling, inflammation, and oxidative stress. Neurobiol Stress. 2018;9:9-21. [CrossRef]
- Hellwig S, Brioschi S, Dieni S, et al. Altered microglia morphology and higher resilience to stress-induced depression-like behavior in CX3CR1-deficient mice. Brain Behav Immun. 2016;55:126-137. [CrossRef]
- Köhler CA, Freitas TH, Maes M, et al. Peripheral cytokine and chemokine alterations in depression: a meta-analysis of 82 studies. Acta Psychiatr Scand. 2017;135(5):373-387. [CrossRef]
- Kopelman J, Keller TA, Panny B, et al. Rapid neuroplasticity changes and response to intravenous ketamine: a randomized controlled trial in treatment-resistant depression. Transl Psychiatry. 2023;13(1):1-9. [CrossRef]
- Hirschfeld RMA. Efficacy of SSRIs and Newer Antidepressants in Severe Depression: Comparison With TCAs. J Clin Psychiatry. Published online 1999.
- Tynan RJ, Weidenhofer J, Hinwood M, Cairns MJ, Day TA, Walker FR. A comparative examination of the anti-inflammatory effects of SSRI and SNRI antidepressants on LPS stimulated microglia. Brain Behav Immun. 2012;26(3):469-479. [CrossRef]
- Lepack AE, Fuchikami M, Dwyer JM, Banasr M, Duman RS. BDNF Release Is Required for the Behavioral Actions of Ketamine. Int J Neuropsychopharmacol. 2015;18(1):pyu033. [CrossRef]
- Saelens J, Gramser A, Watzal V, Zarate CA, Lanzenberger R, Kraus C. Relative effectiveness of antidepressant treatments in treatment-resistant depression: a systematic review and network meta-analysis of randomized controlled trials. Neuropsychopharmacology. 2025;50(6):913-919. [CrossRef]
- Lynall ME, McIntosh AM. The Heterogeneity of Depression. Am J Psychiatry. 2023;180(10):703-704. [CrossRef]
- Garcia-Sifuentes Y, Maney DL. Reporting and misreporting of sex differences in the biological sciences. Allison DB, Zaidi M, Vorland CJ, Kahathuduwa C, eds. eLife. 2021;10:e70817. [CrossRef]
- Sloan DM, Sandt AR. Gender Differences in Depression. Womens Health. 2006;2(3):425-434. [CrossRef]
- Genuchi MC, Mitsunaga LK. Sex Differences in Masculine Depression: Externalizing Symptoms as a Primary Feature of Depression in Men. J Men’s Stud. 2015;23(3):243-251. [CrossRef]
- Rice SM, Fallon BJ, Aucote HM, Möller-Leimkühler A, Treeby MS, Amminger GP. Longitudinal sex differences of externalising and internalising depression symptom trajectories: Implications for assessment of depression in men from an online study. Int J Soc Psychiatry. 2015;61(3):236-240. [CrossRef]
- Cavanagh A, Wilson CJ, Kavanagh DJ, Caputi P. Differences in the Expression of Symptoms in Men Versus Women with Depression: A Systematic Review and Meta-analysis. Harv Rev Psychiatry. 2017;25(1):29. [CrossRef]
- Tremblay MÈ, Lowery RL, Majewska AK. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 2010;8(11):e1000527. [CrossRef]
- Baker AE, Brautigam VM, Watters JJ. Estrogen Modulates Microglial Inflammatory Mediator Production via Interactions with Estrogen Receptor β. Endocrinology. 2004;145(11):5021-5032. [CrossRef]
- Picard K, Bisht K, Poggini S, et al. Microglial-glucocorticoid receptor depletion alters the response of hippocampal microglia and neurons in a chronic unpredictable mild stress paradigm in female mice. Brain Behav Immun. 2021;97:423-439. [CrossRef]
- Berboth S, Morawetz C. Amygdala-prefrontal connectivity during emotion regulation: A meta-analysis of psychophysiological interactions. Neuropsychologia. 2021;153:107767. [CrossRef]
- Damborská A, Honzírková E, Barteček R, et al. Altered directed functional connectivity of the right amygdala in depression: high-density EEG study. Sci Rep. 2020;10(1):4398. [CrossRef]
- Helm K, Viol K, Weiger TM, et al. Neuronal connectivity in major depressive disorder: a systematic review. Neuropsychiatr Dis Treat. 2018;14:2715-2737. [CrossRef]
- Zotev V, Phillips R, Young KD, Drevets WC, Bodurka J. Prefrontal Control of the Amygdala during Real-Time fMRI Neurofeedback Training of Emotion Regulation. PLOS ONE. 2013;8(11):e79184. [CrossRef]
- Zhang JX, Bo K, Wager TD, Gross JJ. The brain bases of emotion generation and emotion regulation. Trends Cogn Sci. Published online May 29, 2025. [CrossRef]
- Siegle GJ, Thompson W, Carter CS, Steinhauer SR, Thase ME. Increased Amygdala and Decreased Dorsolateral Prefrontal BOLD Responses in Unipolar Depression: Related and Independent Features. Biol Psychiatry. 2007;61(2):198-209. [CrossRef]
- Jamieson AJ, Leonards CA, Davey CG, Harrison BJ. Major depressive disorder associated alterations in the effective connectivity of the face processing network: a systematic review. Transl Psychiatry. 2024;14(1):62. [CrossRef]
- Izquierdo A, Murray EA. Combined Unilateral Lesions of the Amygdala and Orbital Prefrontal Cortex Impair Affective Processing in Rhesus Monkeys. J Neurophysiol. 2004;91(5):2023-2039. [CrossRef]
- Costafreda SG, Brammer MJ, David AS, Fu CHY. Predictors of amygdala activation during the processing of emotional stimuli: A meta-analysis of 385 PET and fMRI studies. Brain Res Rev. 2008;58(1):57-70. [CrossRef]
- Qasim SE, Mohan UR, Stein JM, Jacobs J. Neuronal activity in the human amygdala and hippocampus enhances emotional memory encoding. Nat Hum Behav. 2023;7(5):754-764. [CrossRef]
- Vandekerckhove M, Cluydts R. The emotional brain and sleep: An intimate relationship. Sleep Med Rev. 2010;14(4):219-226. [CrossRef]
- Treadway MT, Grant MM, Ding Z, Hollon SD, Gore JC, Shelton RC. Early Adverse Events, HPA Activity and Rostral Anterior Cingulate Volume in MDD. PLOS ONE. 2009;4(3):e4887. [CrossRef]
- Herman JP, Ostrander MM, Mueller NK, Figueiredo H. Limbic system mechanisms of stress regulation: hypothalamo-pituitary-adrenocortical axis. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29(8):1201-1213. [CrossRef]
- Dedovic K, Duchesne A, Andrews J, Engert V, Pruessner JC. The brain and the stress axis: the neural correlates of cortisol regulation in response to stress. NeuroImage. 2009;47(3):864-871. [CrossRef]
- Suzuki Y, Tanaka SC. Functions of the ventromedial prefrontal cortex in emotion regulation under stress. Sci Rep. 2021;11(1):18225. [CrossRef]
- He Z, Li S, Mo L, et al. The VLPFC-Engaged Voluntary Emotion Regulation: Combined TMS-fMRI Evidence for the Neural Circuit of Cognitive Reappraisal. J Neurosci. 2023;43(34):6046-6060. [CrossRef]
- Keller M, Mendoza-Quiñones R, Cabrera Muñoz A, et al. Transdiagnostic alterations in neural emotion regulation circuits – neural substrates of cognitive reappraisal in patients with depression and post-traumatic stress disorder. BMC Psychiatry. 2022;22(1):173. [CrossRef]
- Cieslik EC, Zilles K, Caspers S, et al. Is There “One” DLPFC in Cognitive Action Control? Evidence for Heterogeneity From Co-Activation-Based Parcellation. Cereb Cortex. 2013;23(11):2677-2689. [CrossRef]
- Morawetz C, Bode S, Baudewig J, Kirilina E, Heekeren HR. Changes in Effective Connectivity Between Dorsal and Ventral Prefrontal Regions Moderate Emotion Regulation. Cereb Cortex. 2016;26(5):1923-1937. [CrossRef]
- Pletzer B, Harris TA, Scheuringer A, Hidalgo-Lopez E. The cycling brain: menstrual cycle related fluctuations in hippocampal and fronto-striatal activation and connectivity during cognitive tasks. Neuropsychopharmacology. 2019;44(11):1867-1875. [CrossRef]
- Heller C, Güllmar D, Koeppel CJ, et al. Hippocampal volume and affect in response to fluctuating estrogens in menstrual cycle irregularity: a longitudinal single-subject study. Npj Womens Health. 2024;2(1):19. [CrossRef]
- Holesh JE, Bass AN, Lord M. Physiology, Ovulation. In: StatPearls. StatPearls Publishing; 2025. Accessed August 26, 2025. http://www.ncbi.nlm.nih.gov/books/NBK441996/.
- Ko CH, Wong TH, Suen JL, Lin PC, Long CY, Yen JY. Estrogen, progesterone, cortisol, brain-derived neurotrophic factor, and vascular endothelial growth factor during the luteal phase of the menstrual cycle in women with premenstrual dysphoric disorder. J Psychiatr Res. 2024;169:307-317. [CrossRef]
- Baerwald AR, Adams GP, Pierson RA. Form and function of the corpus luteum during the human menstrual cycle. Ultrasound Obstet Gynecol. 2005;25(5):498-507. [CrossRef]
- Lisofsky N, Mårtensson J, Eckert A, Lindenberger U, Gallinat J, Kühn S. Hippocampal volume and functional connectivity changes during the female menstrual cycle. NeuroImage. 2015;118:154-162. [CrossRef]
- Ossewaarde L, van Wingen GA, Rijpkema M, Bäckström T, Hermans EJ, Fernández G. Menstrual cycle-related changes in amygdala morphology are associated with changes in stress sensitivity. Hum Brain Mapp. 2013;34(5):1187-1193. [CrossRef]
- Goldstein JM, Jerram M, Poldrack R, et al. Hormonal cycle modulates arousal circuitry in women using functional magnetic resonance imaging. J Neurosci Off J Soc Neurosci. 2005;25(40):9309-9316. [CrossRef]
- Chung YS, Poppe A, Novotny S, et al. A preliminary study of association between adolescent estradiol level and dorsolateral prefrontal cortex activity during emotion regulation. Psychoneuroendocrinology. 2019;109:104398. [CrossRef]
- Handy AB, Greenfield SF, Yonkers KA, Payne LA. Psychiatric Symptoms Across the Menstrual Cycle in Adult Women: A Comprehensive Review. Harv Rev Psychiatry. 2022;30(2):100. [CrossRef]
- Ko MY, Darouian N. The Importance of an Interdisciplinary Approach in the Transition of At-Risk Patients From Hospital to Skilled Nursing Facilities: A Case Report. Cureus. 2024;16(10):e71629. [CrossRef]
- Beddig T, Reinhard I, Kuehner C. Stress, mood, and cortisol during daily life in women with Premenstrual Dysphoric Disorder (PMDD). Psychoneuroendocrinology. 2019;109:104372. [CrossRef]
- Huang Y, Zhou R, Wu M, Wang Q, Zhao Y. Premenstrual syndrome is associated with blunted cortisol reactivity to the TSST. Stress Amst Neth. 2015;18(2):160-168. [CrossRef]
- Kendler KS, Karkowski LM, Prescott CA. Causal Relationship Between Stressful Life Events and the Onset of Major Depression. Am J Psychiatry. 1999;156(6):837-841. [CrossRef]
- Bhagwagar Z, Hafizi S, Cowen PJ. Increased salivary cortisol after waking in depression. Psychopharmacology (Berl). 2005;182(1):54-57. [CrossRef]
- Hinkelmann K, Moritz S, Botzenhardt J, et al. Cognitive Impairment in Major Depression: Association with Salivary Cortisol. Biol Psychiatry. 2009;66(9):879-885. [CrossRef]
- Peeters F, Nicolson NA, Berkhof J. Levels and variability of daily life cortisol secretion in major depression. Psychiatry Res. 2004;126(1):1-13. [CrossRef]
- Green MR, McCormick CM. Sex and stress steroids in adolescence: Gonadal regulation of the hypothalamic-pituitary-adrenal axis in the rat. Gen Comp Endocrinol. 2016;234:110-116. [CrossRef]
- Goel N, Workman JL, Lee TT, Innala L, Viau V. Sex Differences in the HPA Axis. In: Terjung R, ed. Comprehensive Physiology. 1st ed. Wiley; 2014:1121-1155. [CrossRef]
- Gray TS, Carney ME, Magnuson DJ. Direct Projections from the Central Amygdaloid Nucleus to the Hypothalamic Paraventricular Nucleus: Possible Role in Stress-Induced Adrenocorticotropin Release. Neuroendocrinology. 2008;50(4):433-446. [CrossRef]
- Holmes MC, Antoni FA, Aguilera G, Catt KJ. Magnocellular axons in passage through the median eminence release vasopressin. Nature. 1986;319(6051):326-329. [CrossRef]
- Szabó K, Csányi K. The vascular architecture of the developing pituitary-median eminence complex in the rat. Cell Tissue Res. 1982;224(3):563-577. [CrossRef]
- Wiegand SJ, Price JL. Cells of origin of the afferent fibers to the median eminence in the rat. J Comp Neurol. 1980;192(1):1-19. [CrossRef]
- Luo X, Kiss A, Makara G, Lolait SJ, Aguilera G. Stress-Specific Regulation of Corticotropin Releasing Hormone Receptor Expression in the Paraventricular and Supraoptic Nuclei of the Hypothalamus in the Rat. J Neuroendocrinol. 1994;6(6):689-696. [CrossRef]
- Tse A, Lee AK, Tse FW. Ca2+ signaling and exocytosis in pituitary corticotropes. Cell Calcium. 2012;51(3):253-259. [CrossRef]
- McCartney CR, Marshall JC. Chapter 1 - Neuroendocrinology of Reproduction. In: Strauss JF, Barbieri RL, eds. Yen and Jaffe’s Reproductive Endocrinology (Eighth Edition). Elsevier; 2019:1-24.e8. [CrossRef]
- NEWMAN CB, WARDLAW SL, VAN VUGT DA, FERIN M, FRANTZ AG. Adrenocorticotropin Immunoactivity in Monkey Hypophyseal Portal Blood*. J Clin Endocrinol Metab. 1984;59(1):108-112. [CrossRef]
- Gjerstad JK, Lightman SL, Spiga F. Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress. 2018;21(5):403-416. [CrossRef]
- Herrero MT, Estrada C, Maatouk L, Vyas S. Inflammation in Parkinson’s disease: role of glucocorticoids. Front Neuroanat. 2015;9. [CrossRef]
- Tang S, Zhang Z, Oakley RH, et al. Intestinal epithelial glucocorticoid receptor promotes chronic inflammation–associated colorectal cancer. JCI Insight. 2021;6(24). [CrossRef]
- Pujades-Rodriguez M, Morgan AW, Cubbon RM, Wu J. Dose-dependent oral glucocorticoid cardiovascular risks in people with immune-mediated inflammatory diseases: A population-based cohort study. PLOS Med. 2020;17(12):e1003432. [CrossRef]
- Sapolsky RM. Glucocorticoids and Hippocampal Atrophy in Neuropsychiatric Disorders. Arch Gen Psychiatry. 2000;57(10):925-935. [CrossRef]
- Wang Q, Van Heerikhuize J, Aronica E, et al. Glucocorticoid receptor protein expression in human hippocampus; stability with age. Neurobiol Aging. 2013;34(6):1662-1673. [CrossRef]
- McKlveen JM, Myers B, Flak JN, et al. Role of Prefrontal Cortex Glucocorticoid Receptors in Stress and Emotion. Biol Psychiatry. 2013;74(9):672-679. [CrossRef]
- Kim EJ, Pellman B, Kim JJ. Stress effects on the hippocampus: a critical review. Learn Mem. 2015;22(9):411-416. [CrossRef]
- McEwen BS, Nasca C, Gray JD. Stress Effects on Neuronal Structure: Hippocampus, Amygdala, and Prefrontal Cortex. Neuropsychopharmacology. 2016;41(1):3-23. [CrossRef]
- Dogra P, Vijayashankar NP. Dexamethasone Suppression Test. In: StatPearls [Internet]. StatPearls Publishing; 2024. Accessed April 14, 2025. https://www.ncbi.nlm.nih.gov/sites/books/NBK542317/.
- Almeida FB, Pinna G, Barros HMT. The Role of HPA Axis and Allopregnanolone on the Neurobiology of Major Depressive Disorders and PTSD. Int J Mol Sci. 2021;22(11):5495. [CrossRef]
- Daskalakis NP, Meijer OC, de Kloet ER. Mineralocorticoid receptor and glucocorticoid receptor work alone and together in cell-type-specific manner: Implications for resilience prediction and targeted therapy. Neurobiol Stress. 2022;18:100455. [CrossRef]
- Heuser I, Yassouridis A, Holsboer F. The combined dexamethasone/CRH test: A refined laboratory test for psychiatric disorders. J Psychiatr Res. 1994;28(4):341-356. [CrossRef]
- Dong HW, Petrovich GD, Watts AG, Swanson LW. Basic organization of projections from the oval and fusiform nuclei of the bed nuclei of the stria terminalis in adult rat brain. J Comp Neurol. 2001;436(4):430-455. [CrossRef]
- Radley JJ, Gosselink KL, Sawchenko PE. A Discrete GABAergic Relay Mediates Medial Prefrontal Cortical Inhibition of the Neuroendocrine Stress Response. J Neurosci. 2009;29(22):7330-7340. [CrossRef]
- Herman JP, Cullinan WE, Ziegler DR, Tasker JG. Role of the paraventricular nucleus microenvironment in stress integration. Eur J Neurosci. 2002;16(3):381-385. [CrossRef]
- Cole AB, Montgomery K, Bale TL, Thompson SM. What the hippocampus tells the HPA axis: Hippocampal output attenuates acute stress responses via disynaptic inhibition of CRF+ PVN neurons. Neurobiol Stress. 2022;20:100473. [CrossRef]
- Gungor NZ, Yamamoto R, Paré D. Optogenetic study of the projections from the bed nucleus of the stria terminalis to the central amygdala. J Neurophysiol. 2015;114(5):2903-2911. [CrossRef]
- Lowery-Gionta EG, Crowley NA, Bukalo O, Silverstein S, Holmes A, Kash TL. Chronic stress dysregulates amygdalar output to the prefrontal cortex. Neuropharmacology. 2018;139:68-75. [CrossRef]
- Han RW, Zhang ZY, Jiao C, Hu ZY, Pan BX. Synergism between two BLA-to-BNST pathways for appropriate expression of anxiety-like behaviors in male mice. Nat Commun. 2024;15(1):3455. [CrossRef]
- Bartos M, Vida I, Jonas P. Synaptic mechanisms of synchronized gamma oscillations in inhibitory interneuron networks. Nat Rev Neurosci. 2007;8(1):45-56. [CrossRef]
- Ferguson BR, Gao WJ. PV Interneurons: Critical Regulators of E/I Balance for Prefrontal Cortex-Dependent Behavior and Psychiatric Disorders. Front Neural Circuits. 2018;12:37. [CrossRef]
- Hájos N, Paulsen O. Network mechanisms of gamma oscillations in the CA3 region of the hippocampus. Neural Netw Off J Int Neural Netw Soc. 2009;22(8):1113-1119. [CrossRef]
- Antonoudiou P, Tan YL, Kontou G, Upton AL, Mann EO. Parvalbumin and Somatostatin Interneurons Contribute to the Generation of Hippocampal Gamma Oscillations. J Neurosci. 2020;40(40):7668-7687. [CrossRef]
- McKlveen JM, Moloney RD, Scheimann JR, Myers B, Herman JP. “Braking” the Prefrontal Cortex: The Role of Glucocorticoids and Interneurons in Stress Adaptation and Pathology. Biol Psychiatry. 2019;86(9):669-681. [CrossRef]
- Cottam JCH, Smith SL, Häusser M. Target-Specific Effects of Somatostatin-Expressing Interneurons on Neocortical Visual Processing. J Neurosci. 2013;33(50):19567-19578. [CrossRef]
- Yizhar O, Fenno LE, Prigge M, et al. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011;477(7363):171-178. [CrossRef]
- Katona L, Micklem B, Borhegyi Z, et al. Behavior-dependent activity patterns of GABAergic long-range projecting neurons in the rat hippocampus. Hippocampus. 2017;27(4):359-377. [CrossRef]
- Jie F, Yin G, Yang W, et al. Stress in Regulation of GABA Amygdala System and Relevance to Neuropsychiatric Diseases. Front Neurosci. 2018;12. [CrossRef]
- Biggio G, Cristina Mostallino M, Follesa P, Concas A, Sanna E. GABA(A) receptor function and gene expression during pregnancy and postpartum. Int Rev Neurobiol. 2009;85:73-94. [CrossRef]
- Shen Q, Lal R, Luellen BA, Earnheart JC, Andrews AM, Luscher B. GABAA receptor deficits cause HPA axis hyperactivity and antidepressant drug sensitivity reminiscent of melancholic forms of depression. Biol Psychiatry. 2010;68(6):512-520. [CrossRef]
- Tongta S, Daendee S, Kalandakanond-Thongsong S. Effects of estrogen receptor β or G protein-coupled receptor 30 activation on anxiety-like behaviors in relation to GABAergic transmission in stress-ovariectomized rats. Neurosci Lett. 2022;789:136885. [CrossRef]
- Maguire JL, Mennerick S. Neurosteroids: mechanistic considerations and clinical prospects. Neuropsychopharmacology. 2024;49(1):73-82. [CrossRef]
- Rodríguez-Palma EJ, Islas-Espinoza AM, Ramos-Rodríguez II, et al. Estradiol modulates the role of the spinal α6-subunit containing GABAA receptors in female rats with neuropathic pain. Eur J Pharmacol. 2024;974:176616. [CrossRef]
- Camille Melón L, Maguire J. GABAergic regulation of the HPA and HPG axes and the impact of stress on reproductive function. J Steroid Biochem Mol Biol. 2016;160:196-203. [CrossRef]
- Glykys J, Mann EO, Mody I. Which GABA(A) receptor subunits are necessary for tonic inhibition in the hippocampus? J Neurosci Off J Soc Neurosci. 2008;28(6):1421-1426. [CrossRef]
- Sabaliauskas N, Shen H, Molla J, et al. Neurosteroid effects at α4βδ GABAA receptors alter spatial learning and synaptic plasticity in CA1 hippocampus across the estrous cycle of the mouse. Brain Res. 2015;1621:170-186. [CrossRef]
- Zhang L, Gao YZ, Zhao CJ, Xia JY, Yang JJ, Ji MH. Reduced inhibitory and excitatory input onto parvalbumin interneurons mediated by perineuronal net might contribute to cognitive impairments in a mouse model of sepsis-associated encephalopathy. Neuropharmacology. 2023;225:109382. [CrossRef]
- Locci A, Porcu P, Talani G, et al. Neonatal estradiol exposure to female rats changes GABAA receptor expression and function, and spatial learning during adulthood. Horm Behav. 2017;87:35-46. [CrossRef]
- Pytel M, Wójtowicz T, Mercik K, et al. 17 β-estradiol modulates GABAergic synaptic transmission and tonic currents during development in vitro. Neuropharmacology. 2007;52(6):1342-1353. [CrossRef]
- De Bondt T, De Belder F, Vanhevel F, Jacquemyn Y, Parizel PM. Prefrontal GABA concentration changes in women—Influence of menstrual cycle phase, hormonal contraceptive use, and correlation with premenstrual symptoms. Brain Res. 2015;1597:129-138. [CrossRef]
- De Bondt T, De Belder F, Vanhevel F, Jacquemyn Y, Parizel PM. Prefrontal GABA concentration changes in women-Influence of menstrual cycle phase, hormonal contraceptive use, and correlation with premenstrual symptoms. Brain Res. 2015;1597:129-138. [CrossRef]
- Wójtowicz T, Lebida K, Mozrzymas JW. 17beta-estradiol affects GABAergic transmission in developing hippocampus. Brain Res. 2008;1241:7-17. [CrossRef]
- Derntl B, Eber CH, Kogler L, Rehbein E, Sundstöm-Poromaa I, Morawetz C. Estradiol modulates changes in effective connectivity in emotion regulation networks. Psychoneuroendocrinology. 2024;167:107103. [CrossRef]
- Derntl B, Eber CH, Kogler L, Rehbein E, Sundstöm-Poromaa I, Morawetz C. Estradiol modulates changes in effective connectivity in emotion regulation networks. Psychoneuroendocrinology. 2024;167:107103. [CrossRef]
- Carceller H, Guirado R, Ripolles-Campos E, Teruel-Marti V, Nacher J. Perineuronal Nets Regulate the Inhibitory Perisomatic Input onto Parvalbumin Interneurons and γ Activity in the Prefrontal Cortex. J Neurosci. 2020;40(26):5008-5018. [CrossRef]
- Dityatev A, Brückner G, Dityateva G, Grosche J, Kleene R, Schachner M. Activity-dependent formation and functions of chondroitin sulfate-rich extracellular matrix of perineuronal nets. Dev Neurobiol. 2007;67(5):570-588. [CrossRef]
- Ye Q, Miao QL. Experience-dependent development of perineuronal nets and chondroitin sulfate proteoglycan receptors in mouse visual cortex. Matrix Biol J Int Soc Matrix Biol. 2013;32(6):352-363. [CrossRef]
- Venturino A, Schulz R, Jesús-Cortés HD, et al. Microglia enable mature perineuronal nets disassembly upon anesthetic ketamine exposure or 60-Hz light entrainment in the healthy brain. Cell Rep. 2021;36(1). [CrossRef]
- Cabungcal JH, Steullet P, Morishita H, et al. Perineuronal nets protect fast-spiking interneurons against oxidative stress. Proc Natl Acad Sci. 2013;110(22):9130-9135. [CrossRef]
- Gottschling C, Wegrzyn D, Denecke B, Faissner A. Elimination of the four extracellular matrix molecules tenascin-C, tenascin-R, brevican and neurocan alters the ratio of excitatory and inhibitory synapses. Sci Rep. 2019;9(1):13939. [CrossRef]
- Bischop DP, Orduz D, Lambot L, Schiffmann SN, Gall D. Control of Neuronal Excitability by Calcium Binding Proteins: A New Mathematical Model for Striatal Fast-Spiking Interneurons. Front Mol Neurosci. 2012;5:78. [CrossRef]
- Orduz D, Bischop DP, Schwaller B, Schiffmann SN, Gall D. Parvalbumin tunes spike-timing and efferent short-term plasticity in striatal fast spiking interneurons. J Physiol. 2013;591(Pt 13):3215-3232. [CrossRef]
- Wang AS, Wan X, Storch DS, et al. Cross-species conservation in the regulation of parvalbumin by perineuronal nets. Front Neural Circuits. 2023;17. [CrossRef]
- Drzewiecki CM, Willing J, Juraska JM. Influences of age and pubertal status on number and intensity of perineuronal nets in the rat medial prefrontal cortex. Brain Struct Funct. 2020;225(8):2495-2507. [CrossRef]
- Uriarte N, Ferreño M, Méndez D, Nogueira J. Reorganization of perineuronal nets in the medial Preoptic Area during the reproductive cycle in female rats. Sci Rep. 2020;10(1):5479. [CrossRef]
- Hammond BP, Manek R, Kerr BJ, Macauley MS, Plemel JR. Regulation of microglia population dynamics throughout development, health, and disease. Glia. 2021;69(12):2771-2797. [CrossRef]
- Jäntti H, Sitnikova V, Ishchenko Y, et al. Microglial amyloid beta clearance is driven by PIEZO1 channels. J Neuroinflammation. 2022;19(1):147. [CrossRef]
- Stence N, Waite M, Dailey ME. Dynamics of microglial activation: A confocal time-lapse analysis in hippocampal slices. Glia. 2001;33(3):256-266. [CrossRef]
- St-Pierre MK, Carrier M, González Ibáñez F, et al. Ultrastructural characterization of dark microglia during aging in a mouse model of Alzheimer’s disease pathology and in human post-mortem brain samples. J Neuroinflammation. 2022;19(1):235. [CrossRef]
- Chagas L da S, Serfaty CA. The Influence of Microglia on Neuroplasticity and Long-Term Cognitive Sequelae in Long COVID: Impacts on Brain Development and Beyond. Int J Mol Sci. 2024;25(7):3819. [CrossRef]
- Ji K, Akgul G, Wollmuth LP, Tsirka SE. Microglia Actively Regulate the Number of Functional Synapses. PLOS ONE. 2013;8(2):e56293. [CrossRef]
- Ostrycharz E, Hukowska-Szematowicz B. New Insights into the Role of the Complement System in Human Viral Diseases. Biomolecules. 2022;12(2):226. [CrossRef]
- Soteros BM, Sia GM. Complement and microglia dependent synapse elimination in brain development. WIREs Mech Dis. 2022;14(3):e1545. [CrossRef]
- Huang L, Jin J, Chen K, et al. BDNF produced by cerebral microglia promotes cortical plasticity and pain hypersensitivity after peripheral nerve injury. PLOS Biol. 2021;19(7):e3001337. [CrossRef]
- Parkhurst CN, Yang G, Ninan I, et al. Microglia Promote Learning-Dependent Synapse Formation through Brain-Derived Neurotrophic Factor. Cell. 2013;155(7):1596-1609. [CrossRef]
- Smith JA, Das A, Butler JT, Ray SK, Banik NL. Estrogen or Estrogen Receptor Agonist Inhibits Lipopolysaccharide Induced Microglial Activation and Death. Neurochem Res. 2011;36(9):1587-1593. [CrossRef]
- Loiola RA, Wickstead ES, Solito E, McArthur S. Estrogen Promotes Pro-resolving Microglial Behavior and Phagocytic Cell Clearance Through the Actions of Annexin A1. Front Endocrinol. 2019;10:420. [CrossRef]
- Vegeto E, Bonincontro C, Pollio G, et al. Estrogen prevents the lipopolysaccharide-induced inflammatory response in microglia. J Neurosci Off J Soc Neurosci. 2001;21(6):1809-1818. [CrossRef]
- Herman JP, McKlveen JM, Ghosal S, et al. Regulation of the hypothalamic-pituitary-adrenocortical stress response. Compr Physiol. 2016;6(2):603-621. [CrossRef]
- Tanaka J, Fujita H, Matsuda S, Toku K, Sakanaka M, Maeda N. Glucocorticoid- and mineralocorticoid receptors in microglial cells: The two receptors mediate differential effects of corticosteroids. Glia. 1997;20(1):23-37. [CrossRef]
- Chen K, Qi X, Zhu L lin, Li M li, Cong B, Li Y min. Quantitative analysis of microglia morphological changes in the hypothalamus of chronically stressed rats. Brain Res Bull. 2024;206:110861. [CrossRef]
- Frank MG, Thompson BM, Watkins LR, Maier SF. Glucocorticoids mediate stress-induced priming of microglial pro-inflammatory responses. Brain Behav Immun. 2012;26(2):337-345. [CrossRef]
- Nair A, Bonneau RH. Stress-induced elevation of glucocorticoids increases microglia proliferation through NMDA receptor activation. J Neuroimmunol. 2006;171(1-2):72-85. [CrossRef]
- Bollinger JL, Collins KE, Patel R, Wellman CL. Behavioral stress alters corticolimbic microglia in a sex- and brain region-specific manner. PLoS ONE. 2017;12(12):e0187631. [CrossRef]
- Bollinger JL, Bergeon Burns CM, Wellman CL. Differential effects of stress on microglial cell activation in male and female medial prefrontal cortex. Brain Behav Immun. 2016;52:88-97. [CrossRef]
- Gaspar R, Soares-Cunha C, Domingues AV, et al. The Duration of Stress Determines Sex Specificities in the Vulnerability to Depression and in the Morphologic Remodeling of Neurons and Microglia. Front Behav Neurosci. 2022;16. [CrossRef]
- Yu D, Jain S, Wangzhou A, et al. Microglia regulate GABAergic neurogenesis in prenatal human brain through IGF1. bioRxiv. Preprint posted online October 20, 2024:2024.10.19.619180. [CrossRef]
- Gervais É, Iloun P, Martianova E, Gonçalves Bessa AC, Rivest S, Topolnik L. Structural analysis of the microglia-interneuron interactions in the CA1 hippocampal area of the APP/PS1 mouse model of Alzheimer’s disease. J Comp Neurol. 2022;530(9):1423-1437. [CrossRef]
- Wu J, Zhang J, Chen X, et al. Microglial over-pruning of synapses during development in autism-associated SCN2A-deficient mice and human cerebral organoids. Mol Psychiatry. 2024;29(8):2424-2437. [CrossRef]
- Favuzzi E, Huang S, Saldi GA, et al. GABA-receptive microglia selectively sculpt developing inhibitory circuits. Cell. 2021;184(15):4048-4063.e32. [CrossRef]
- Chen JQA, McNamara NB, Engelenburg HJ, et al. Distinct transcriptional changes distinguish efficient and poor remyelination in multiple sclerosis. Brain J Neurol. 2025;148(6):2201-2217. [CrossRef]
- Zhang J, He H, Qiao Y, et al. Priming of microglia with IFN-γ impairs adult hippocampal neurogenesis and leads to depression-like behaviors and cognitive defects. Glia. 2020;68(12):2674-2692. [CrossRef]
- Cramer T, Gill R, Thirouin ZS, et al. Cross-talk between GABAergic postsynapse and microglia regulate synapse loss after brain ischemia. Sci Adv. 2022;8(9):eabj0112. [CrossRef]
- Paolicelli RC, Sierra A, Stevens B, et al. Microglia states and nomenclature: A field at its crossroads. Neuron. 2022;110(21):3458-3483. [CrossRef]
- Kuhn SA, van Landeghem FKH, Zacharias R, et al. Microglia express GABAB receptors to modulate interleukin release. Mol Cell Neurosci. 2004;25(2):312-322. [CrossRef]
- Rossano SM, Johnson AS, Smith A, et al. Microglia measured by TSPO PET are associated with Alzheimer’s disease pathology and mediate key steps in a disease progression model. Alzheimers Dement J Alzheimers Assoc. 2024;20(4):2397-2407. [CrossRef]
- Canário N, Jorge L, Martins R, Santana I, Castelo-Branco M. Dual PET-fMRI reveals a link between neuroinflammation, amyloid binding and compensatory task-related brain activity in Alzheimer’s disease. Commun Biol. 2022;5(1):804. [CrossRef]
- Da Silva T, Hafizi S, Rusjan PM, et al. GABA levels and TSPO expression in people at clinical high risk for psychosis and healthy volunteers: a PET-MRS study. J Psychiatry Neurosci JPN. 2019;44(2):111-119. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



