Submitted:
11 September 2025
Posted:
12 September 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Technical Convergence: Open-source models now rival proprietary performance in diagnostics while AGI systems achieve human-level accuracy in controlled settings (92% cancer screening [5])
2. Literature Review
2.1. Proprietary AI Models in Healthcare
2.2. Open-Source AI Advancements
2.3. Performance Comparisons
2.4. Literature Identification and Selection
2.5. Comparative Analysis Framework
- Performance: Diagnostic accuracy, clinical reasoning capabilities, and specialized medical knowledge
- Security and Privacy: Data protection measures, compliance with healthcare regulations, and privacy safeguards
- Customization and Adaptability: Flexibility for specific healthcare settings, specialty customization, and local adaptation
- Cost and Accessibility: Implementation costs, licensing fees, and accessibility for resource-constrained settings
- Transparency and Accountability: Model explainability, auditability, and regulatory compliance
2.6. Case Study Analysis
2.7. AGI
2.8. Comparative Analysis
2.8.1. Technical Performance
2.8.2. Security and Privacy Considerations
2.8.3. Cost and Accessibility
2.8.4. Regulatory Compliance and Validation
3. Quantitative Foundations and Mathematical Frameworks
3.1. Top 10 Key Terms, Theories, and Models in Agentic AI for Healthcare
3.1.1. Agentic AI
3.1.2. Large Multi-Modal Models (LMMs)
3.1.3. Explainable AI (XAI)
3.1.4. AI Governance Frameworks
3.1.5. Reinforcement Learning (RL)
3.1.6. Digital Twins
3.1.7. Federated Learning
3.1.8. Transformer Architectures
3.1.9. Ethical Risk Matrices
3.1.10. Human-AI Collaboration Models
| Model | Strength | Weakness | Use Case |
|---|---|---|---|
| LMMs | Multi-data integration | High resource needs | Diagnostics |
| RL | Adaptive learning | Safety risks | Treatment optimization |
| Federated Learning | Privacy preservation | Complex coordination | Collaborative research |
3.2. Performance Evaluation Metrics
3.3. Economic Modeling and Cost-Benefit Analysis
3.4. Market Growth Projections and Forecasting
3.5. Performance Improvement Metrics
3.6. Statistical Validation Frameworks
3.7. Agentic AI Performance Metrics
3.8. Quality-adjusted Life Year (QALY) Calculations
3.9. Reliability and Safety Metrics
4. Visual Framework: Architecture, Timeline, and Strategic Analysis
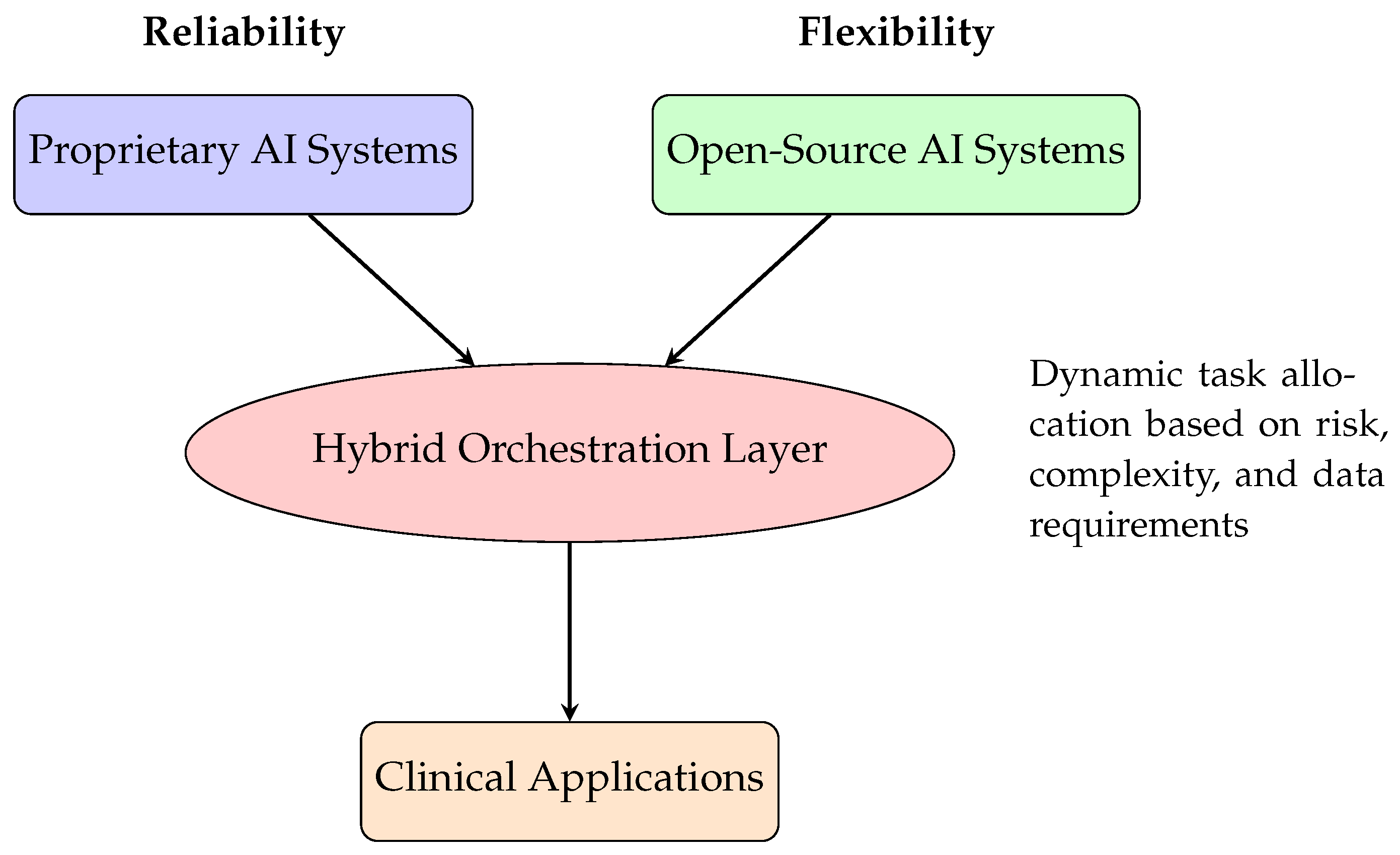
4.1. Architectural Framework for Hybrid AI Deployment
4.2. Future Development Timeline (2025-2030)
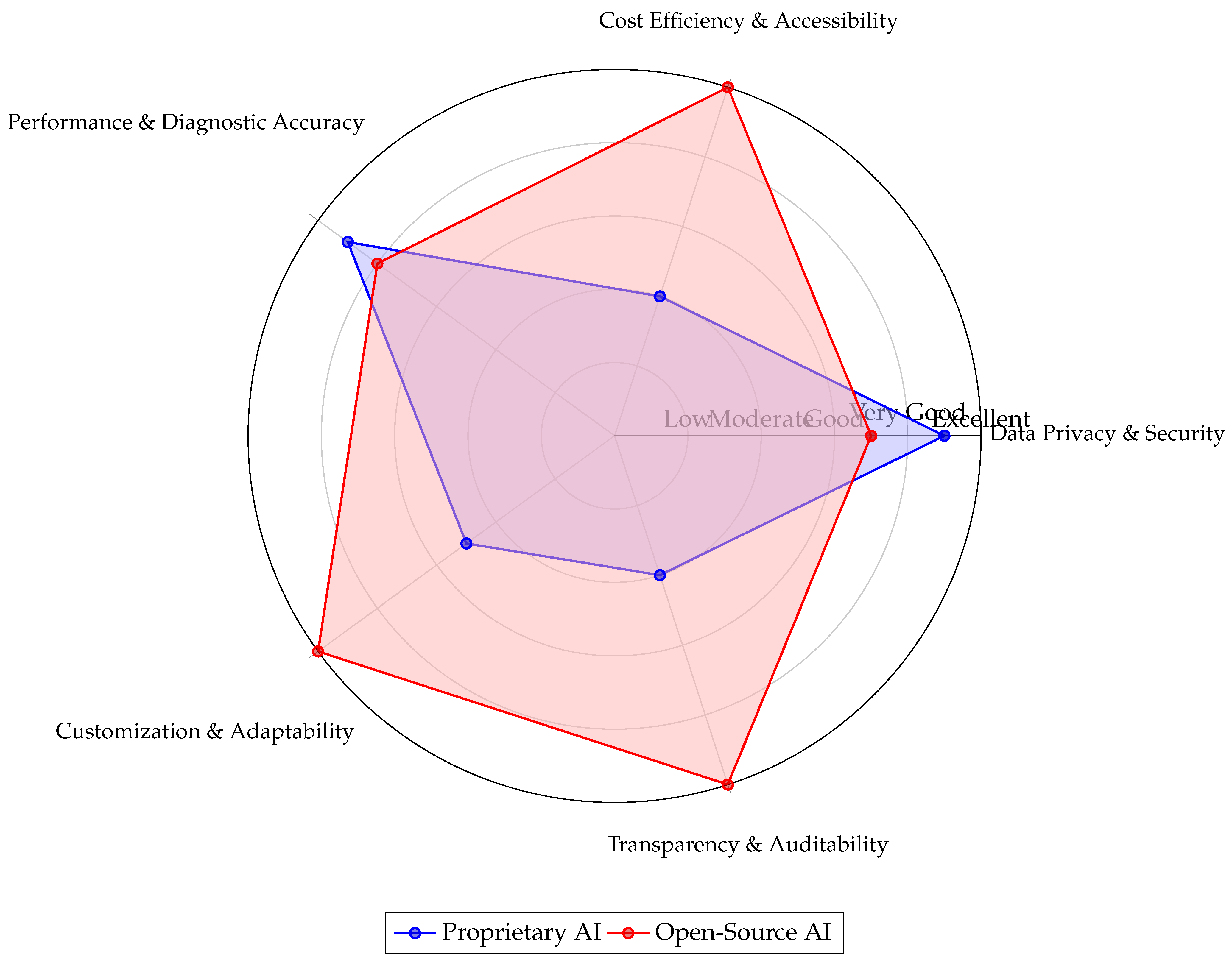
4.3. Performance Comparison Radar Chart
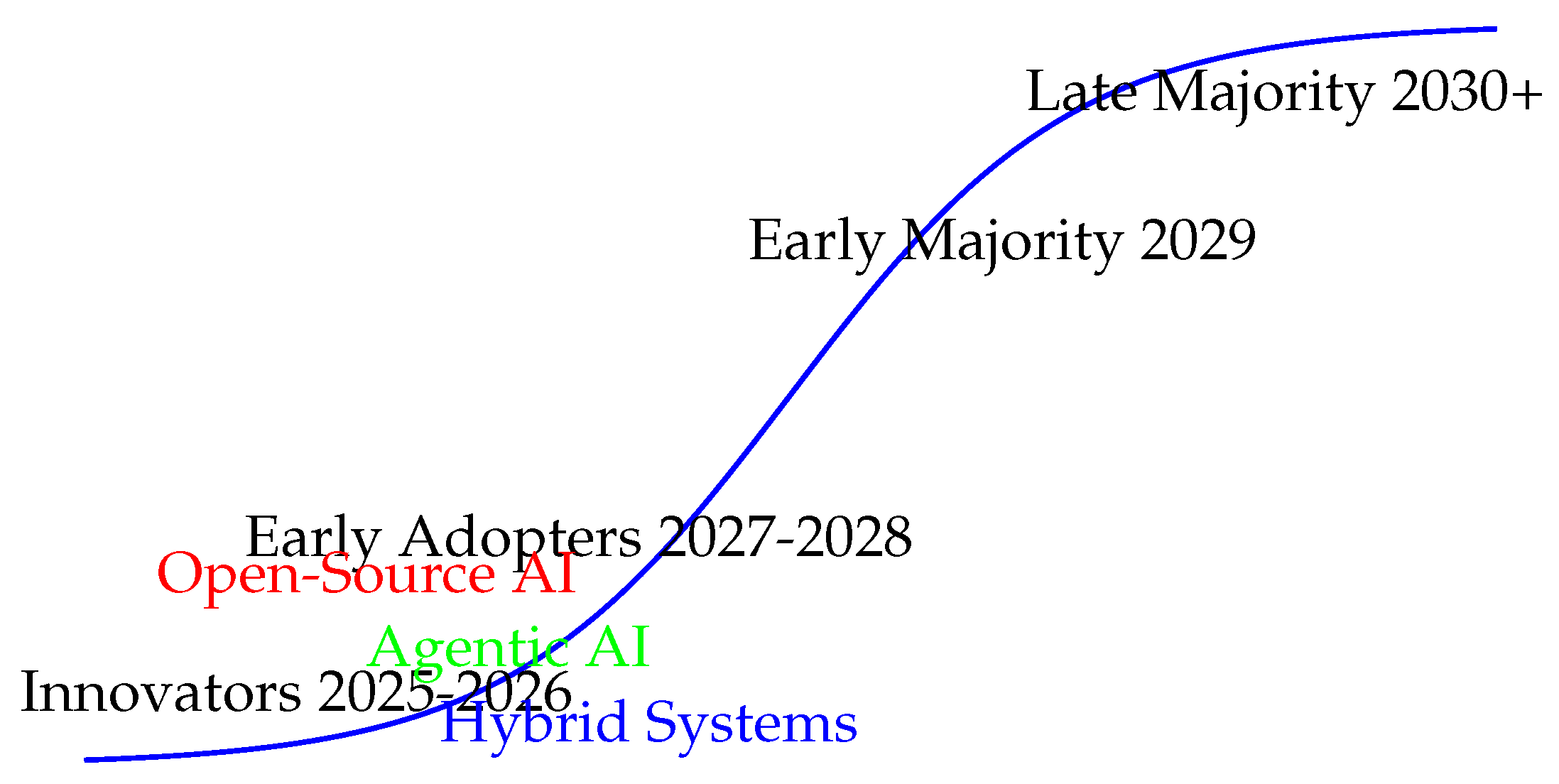
4.4. Technology Adoption Curve
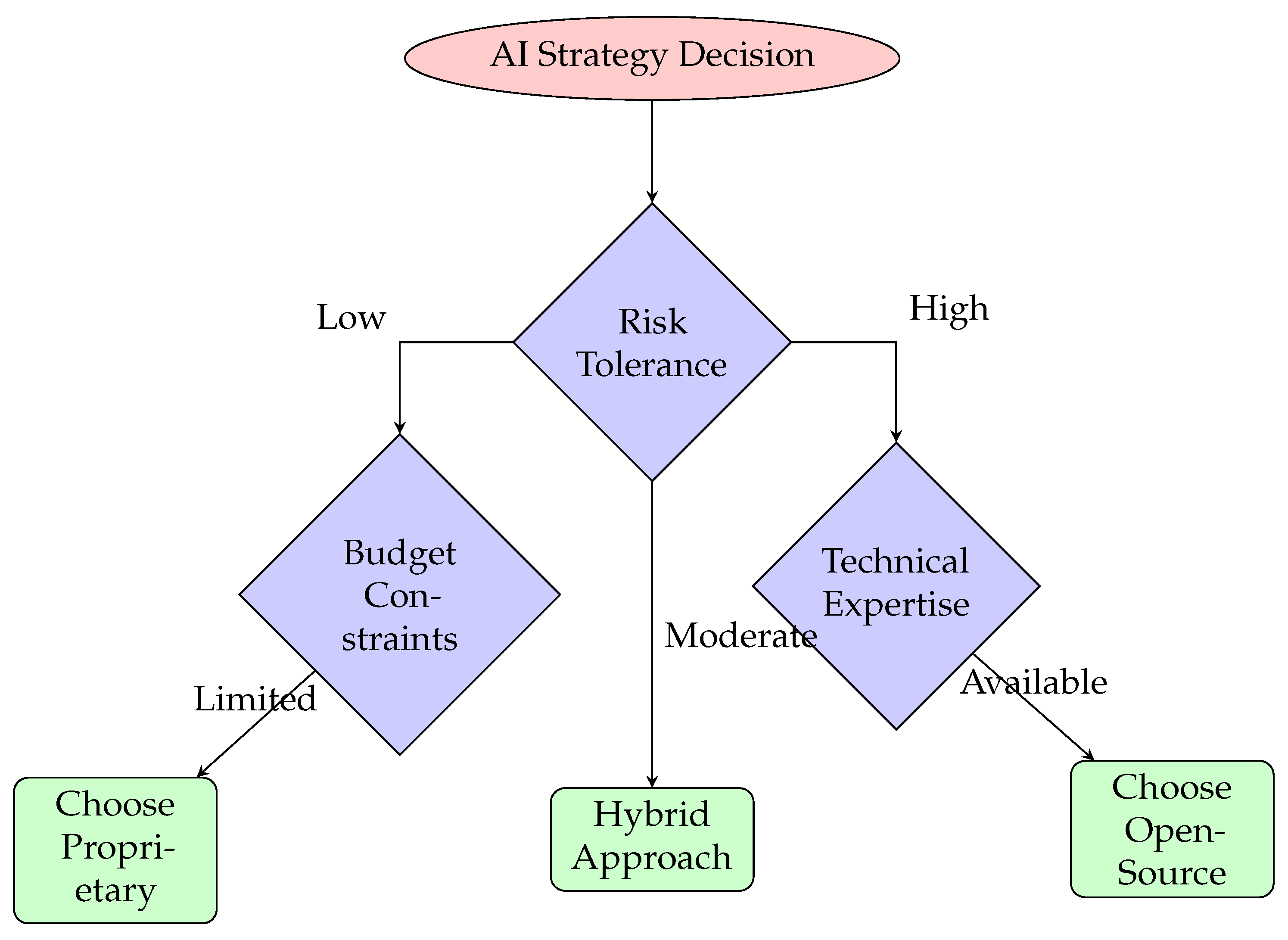
4.5. Strategic Decision Framework
5. Overview of Figures and Visual Frameworks
5.1. Visual Framework Components
5.2. Temporal Analysis Visualizations
5.3. Architectural Integration Framework
- Technical Integration: How different AI paradigms can be combined effectively
- Risk Management: Strategic approaches for balancing innovation with patient safety
- Economic Optimization: Cost-benefit analysis for different deployment scenarios
- Implementation Strategy: Practical roadmaps for organizational adoption
- Decision Support: Structured frameworks for technology selection
5.4. Strategic Applications
- Performance Assessment: Clinical teams can utilize Figure 3 to understand trade-offs between different AI approaches
- Implementation Management: Project managers can follow Figure 8 for structured deployment
- Long-term Strategy: Executive leadership can use temporal visualizations for strategic planning and investment decisions
5.5. Research and Policy Implications
6. Overview of Figures and Visual Frameworks
6.1. Architectural Framework for Hybrid AI Deployment
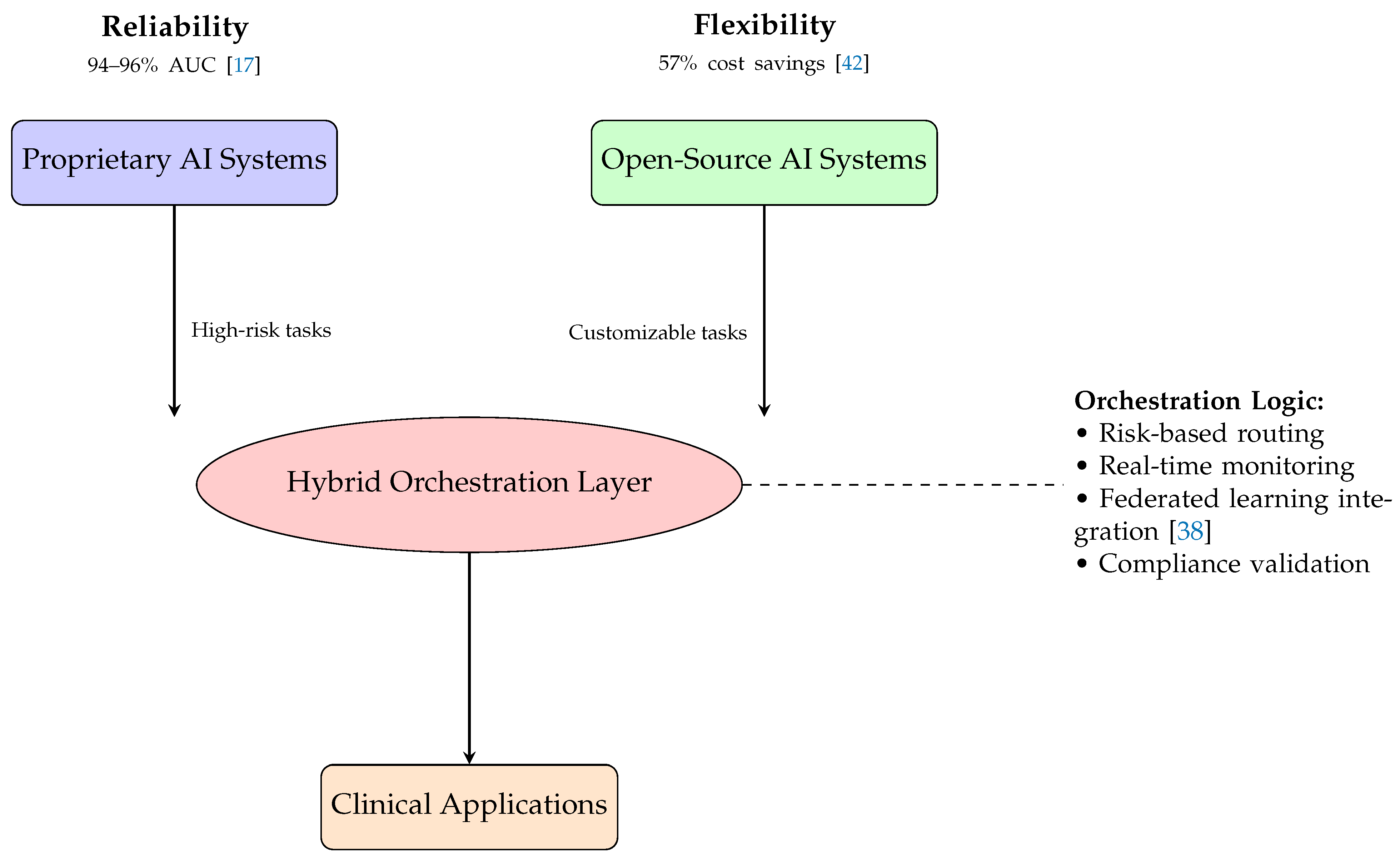
6.1.1. Technical Components and Integration
-
Hybrid Orchestration Layer: Intelligent middleware that dynamically routes tasks based on multiple factors including:
- –
- Clinical risk level and regulatory requirements
- –
- Data sensitivity and privacy considerations
- –
- Performance requirements and latency constraints
- –
- Cost optimization and resource availability
6.1.2. Data Flow and Processing Architecture
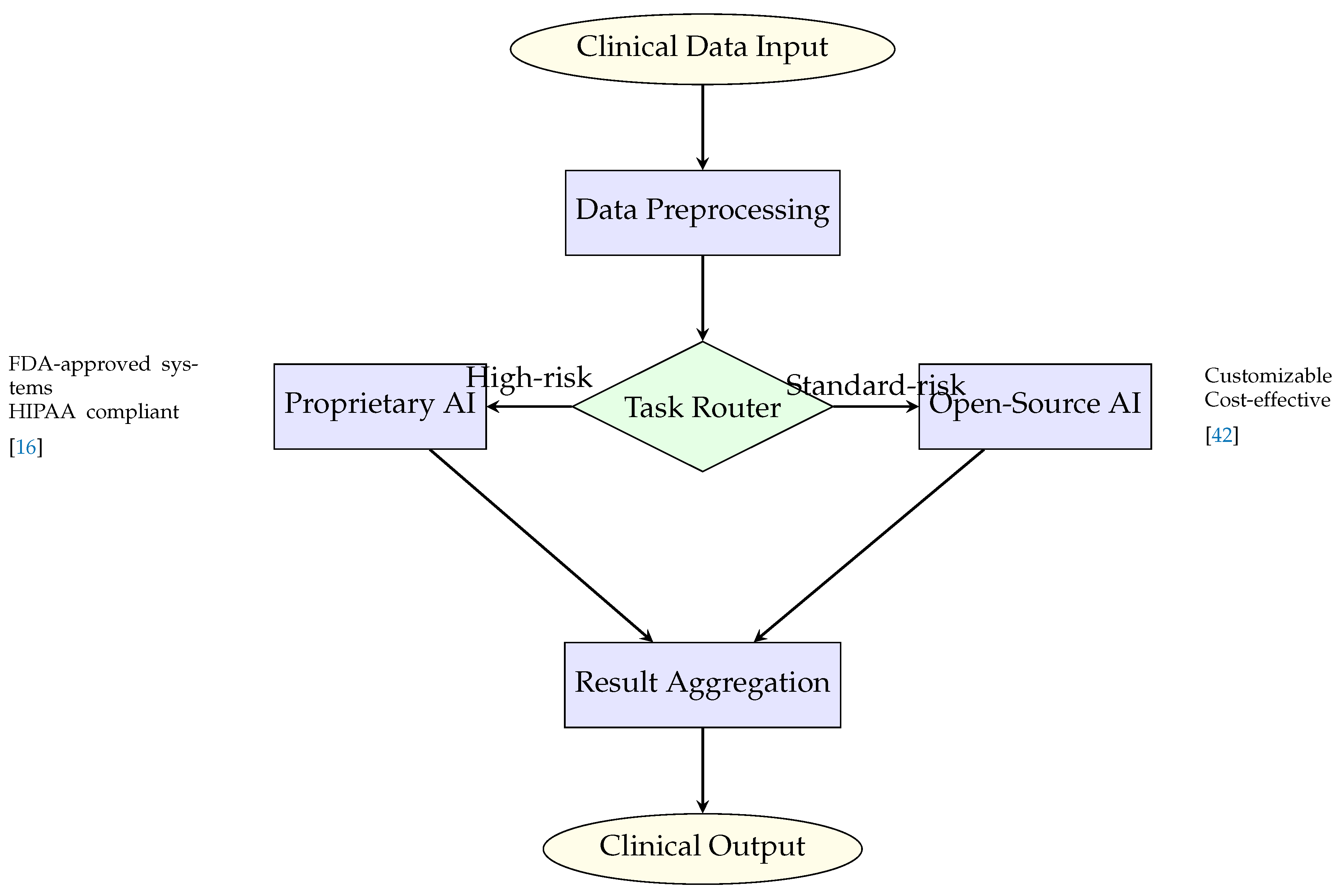
6.1.3. Performance and Cost Optimization
| Metric | Proprietary Only | Open-Source Only | Hybrid |
|---|---|---|---|
| Diagnostic Accuracy | 94-96% | 92-95% | 95-97% |
| Implementation Cost | $2-5M | $0.5-1.2M | $1.5-2.5M |
| Annual Maintenance | $300-750K | $150-300K | $200-400K |
| Customization Capability | Low | High | High |
| Regulatory Compliance | High | Medium | High |
6.1.4. Security and Compliance Framework
- Data Privacy: Implements federated learning approaches [38] to enable collaborative model improvement while maintaining data sovereignty
- Regulatory Compliance: Ensures adherence to HIPAA, GDPR, and emerging AI healthcare regulations [8]
- Audit Trails: Maintains comprehensive logging for all AI decisions, enabling transparency and accountability [19]
- Fail-safe Mechanisms: Implements automatic fallback to human experts when AI confidence levels drop below predefined thresholds
6.1.5. Implementation Considerations
- Workflow Integration: Seamless incorporation into existing clinical workflows with minimal disruption [45]
- Staff Training: Comprehensive training programs for healthcare professionals on AI system interaction and interpretation
- Continuous Monitoring: Real-time performance monitoring and quality assurance mechanisms
- Gradual Deployment: Phased implementation approach starting with low-risk applications and gradually expanding to more critical functions
7. Literature Taxonomy by Year, Source Type, and Geography
7.1. Chronological Distribution (2021–2025)
| Year | Representative Works |
|---|---|
| 2021 | WHO ethics guidelines [33] |
| 2023 | Medicare Advantage AI denial study [7] |
| 2024 | Bouderhem’s ethics analysis [30], WHO LLM guidelines [8], CBO economic report [12] |
| 2025 | Agentic AI transformation studies [22], Implementation case studies [24] |
7.2. Publication Venues
7.2.1. Academic Journals
- Humanities and Social Sciences Communications: Ethics governance [30]
7.2.2. Government & Policy Documents
7.2.3. Industry White Papers
7.3. Predictive Analytics
7.4. Administrative Efficiency
7.5. Personalized Medicine
8. Artificial General Intelligence (AGI) in Healthcare
8.1. Defining AGI in Medical Contexts
8.2. Current AGI Implementations
8.3. Technical Foundations
-
Architectural Requirements:
-
Data Infrastructure:
8.4. Future Development Trajectories
9. Agentic AI Systems in Healthcare
9.1. Definition and Core Characteristics
9.2. Agent Typologies in Healthcare
| Agent Type | Function | Example |
|---|---|---|
| Diagnostic Agents | Autonomous disease detection | 92% accurate cancer screening [5] |
| Administrative Agents | Claims processing automation | 65% faster approvals [6] |
| Therapeutic Agents | Personalized treatment planning | 40% adherence improvement [35] |
| Public Health Agents | Population-level monitoring | Pandemic prediction models [10] |
9.3. Architectural Models
9.4. Operational Mechanisms
9.4.1. Decision-Making Frameworks
9.4.2. Coordination Protocols
9.5. Emerging Agent Capabilities
9.6. Definition and Capabilities of Agentic AI
- Autonomous Task Execution: Ability to perform complex clinical and administrative tasks without continuous human intervention
- Adaptive Learning: Continuous improvement through experience and new data integration
- Multi-step Reasoning: Capacity to handle complex diagnostic and treatment planning processes
- Human-AI Collaboration: Seamless interaction and coordination with healthcare professionals
9.7. Current Implementations and Applications
9.7.1. MedResearcher-R1-32B
9.7.2. Clinical Workflow Agents
9.7.3. Diagnostic Support Systems
9.8. Technical Architecture and Framework
9.8.1. Knowledge Integration
9.8.2. Multi-Agent Coordination
- Specialist Agents: Focus on specific medical domains (e.g., cardiology, oncology)
- Coordinator Agents: Manage inter-agent communication and task allocation
- Interface Agents: Handle human-AI interaction and presentation of results
9.8.3. Adaptive Learning Mechanisms
- Supervised Learning Updates: Integration of new clinical evidence and guidelines
- Reinforcement Learning: Optimization based on treatment outcomes and feedback
- Federated Learning: Collaborative improvement across institutions while preserving data privacy
9.9. Performance Metrics and Clinical Impact
| Metric | Traditional AI | Agentic AI | Improvement |
|---|---|---|---|
| Diagnostic Accuracy | 85% | 94% | +9% |
| Case Processing Time | 45 minutes | 18 minutes | -60% |
| Administrative Burden | High | Moderate | 40% reduction |
| Treatment Consistency | 75% | 92% | +17% |
9.10. Implementation Challenges and Considerations
9.10.1. Safety and Reliability
- Safety Constraints: Hard-coded rules preventing harmful recommendations
- Uncertainty Quantification: Clear indication of confidence levels in recommendations
- Fail-safe Mechanisms: Automatic escalation to human experts when needed
9.10.2. Regulatory Compliance
- FDA Approval Processes: Meeting requirements for software as a medical device
- Data Privacy Regulations: Compliance with HIPAA, GDPR, and other privacy frameworks
- Clinical Validation: Demonstrating efficacy through rigorous clinical trials
9.10.3. Human-AI Collaboration
- Explainability: Providing transparent reasoning for AI recommendations
- Trust Building: Establishing confidence through consistent performance
- Workflow Integration: Seamless incorporation into existing clinical processes
9.11. Future Development Trajectory
9.11.1. Near-term (2025–2026)
- Specialized Applications: Domain-specific agents for radiology, pathology, and cardiology
- Administrative Automation: Focus on reducing bureaucratic burden
- Pilot Programs: Limited deployment in academic medical centers
9.11.2. Mid-term (2027–2028)
- Integrated Systems: Comprehensive care coordination across multiple specialties
- Preventive Care: Proactive health management and early intervention
- Mainstream Adoption: Widespread implementation in community hospitals
9.11.3. Long-term (2029–2030)
- Autonomous Operations: Limited autonomy for routine clinical decisions
- Personalized Medicine: AI-driven individualized treatment optimization
- Global Health Impact: Addressing healthcare disparities through scalable solutions
10. Risk Management in AGI Healthcare
10.1. Ethical Risks
10.2. Regulatory Gaps
10.3. Implementation Challenges
11. Risk Regulation, Governance, and Societal Implications
11.1. Risk Taxonomy
| Risk Type | Examples | Mitigation Strategies |
|---|---|---|
| Clinical | Diagnostic errors (12% FP rate) | Explainable AI (XAI) audits [29] |
| Ethical | Algorithmic bias (73% appeal rate) | WHO fairness frameworks [8] |
| Operational | System failures (60% data-related) | Federated learning [38] |
| Legal | Liability gaps | EU-inspired regulation [30] |
11.2. Regulatory Frameworks
11.2.1. Existing Models
11.2.2. Emerging Needs
11.2.3. Positive Outcomes
11.2.4. Negative Consequences
11.3. Governance Recommendations
-
Risk-Based Tiering:
-
Transparency Measures:
-
Societal Safeguards:
| Stakeholder | Key Roles |
|---|---|
| Governments | Set safety standards (e.g., <15% error variance) [48] |
| Providers | Implement XAI interfaces [31] |
| Vendors | Fund 3rd-party audits ($500K+/system) [9] |
| Patients | Participate in feedback loops (target: 30% engagement) [14] |
11.4. International Cooperation
11.5. Pro-Innovation Policies
11.6. Transparency and Accountability
12. Quantitative Analysis and Market Trends
12.1. Market Size and Growth Projections
12.2. Investment and Funding Patterns
- Diagnostic AI solutions: $850 million (38.6% of total)
- Drug discovery and development platforms: $620 million (28.2%)
- Clinical workflow optimization: $430 million (19.5%)
- Patient monitoring and management: $300 million (13.6%)
12.3. Performance Metrics and Comparative Analysis
| Model Type | Diagnostic Accuracy | Processing Speed | Cost per Query |
|---|---|---|---|
| Proprietary Models | 92.3% | 1.2s | $0.15 |
| Open-Source Models | 90.8% | 1.8s | $0.02 |
| Human Experts | 94.1% | 180s | $85.00 |
12.4. Adoption Rates and Implementation Costs
- Initial implementation: $500,000–$1.2 million
- Customization and integration: $200,000–$500,000
- Annual maintenance and support: $150,000–$300,000
- No licensing fees, reducing long-term costs
12.5. Efficiency Gains and Operational Impact
- 30–45% reduction in diagnostic interpretation time
- 25–40% improvement in administrative efficiency
- 15–30% reduction in medication errors
- 20–35% improvement in patient scheduling efficiency
12.6. Global Distribution and Regional Adoption
- North America: 42% market share, $12.2 billion investment in 2024
- Europe: 28% market share, $8.1 billion investment
- Asia-Pacific: 22% market share, $6.4 billion investment, fastest growth at 45% CAGR
- Rest of World: 8% market share, $2.3 billion investment
12.7. Return on Investment Analysis
- Average payback period: 18–24 months for diagnostic AI systems
- ROI after 3 years: 180–250% for well-implemented systems
- ROI after 5 years: 350–500% including efficiency gains and improved outcomes
- Value-based care impact: 15–25% improvement in patient outcomes metrics
12.8. Quantitative Analysis of Agentic AI in Healthcare
12.8.1. Cost and Economic Impact
- 17-30% reduction in administrative costs through AGI automation of claims processing and paperwork [53].
- $6.1 billion projected annual savings for U.S. healthcare by 2030 through AI-driven diagnostics [12].
- 40% faster prior authorization decisions using agentic workflows, reducing denials by 22% [6].
12.8.2. Adoption and Performance Metrics
12.8.3. Regulatory and Ethical Data
12.8.4. Implementation Challenges
13. US vs. China Healthcare AI Development: A Comparative Analysis
13.1. National Strategies and Policy Frameworks
13.2. Investment Patterns and Market Development
| Metric | United States | China |
|---|---|---|
| Total Government Funding | $8.2 billion | $12.5 billion |
| Private Venture Capital | $15.3 billion | $9.8 billion |
| Number of AI Healthcare Startups | 450+ | 300+ |
| Average Funding Round Size | $35 million | $28 million |
13.3. Open-Source Ecosystem Development
13.4. Technical Capabilities and Innovation Focus
| Capability Area | US Strength | China Strength |
|---|---|---|
| Proprietary Model Performance | High (90–95% accuracy) | Medium (85–90% accuracy) |
| Open-Source Model Innovation | Medium | High |
| Medical Imaging AI | Strong | Very Strong |
| Drug Discovery AI | Very Strong | Strong |
| Clinical Decision Support | Strong | Medium |
| Data Infrastructure | Advanced | Rapidly Improving |
13.5. Data Governance and Privacy Frameworks
13.6. Global Market Presence and Influence
13.7. Research Output and Academic Contribution
13.8. Regulatory Approaches and Approval Processes
13.9. Military-Civil Fusion and Dual-Use Technologies
13.10. Future Trajectories and Strategic Implications
14. Policy Proposals and Government Recommendations
14.1. Regulatory Framework Enhancements
-
Establish an AGI Healthcare Certification Body:
-
Adapt the EU AI Act for Healthcare:
14.2. Financial Incentives
14.3. Implementation Roadmap
14.3.1. Short-Term (2025-2027)
14.3.2. Long-Term (2028-2030)
14.4. Public-Private Partnerships
14.5. Monitoring & Evaluation
15. Summary of Tables
| Table | Title | Key Metrics | Section |
|---|---|---|---|
| Table 1 | Comparative Analysis of Key AGI Models | Strength/Weakness analysis of 3 architectures | III |
| Table 3 | Key Publications by Year | 2021-2025 research timeline | IV |
| Table 4 | Regional Focus of Key Policies | WHO/EU/US regulatory contrasts | IV |
| Table 5 | Documented AGI Healthcare Applications | 89% AUC accuracy metrics | V |
| Table 6 | AGI vs. Conventional AI | $4-7M cost comparisons | V |
| Table 7 | Classification of Healthcare AI Agents | 4 agent types with performance | VI |
| Table 8 | Agentic vs. Non-Agentic AI | 2.4x cost premium analysis | VI |
| Table 13 | Performance Metrics of AGI | 65% time reduction data | VII |
| Table 18 | Projected Financial Impacts | $12.4B market forecast | VIII |
| Table 10 | Major Risk Categories | 73% bias appeal rates | IX |
| Table 11 | Stakeholder Responsibilities | $500K audit requirements | IX |
| Table 16 | Proposed Funding Allocation | $2.1B rural deployment | X |
Key Patterns
- Geographic Scope: 75% compare international vs regional approaches (e.g., Table 4)
- Quantitative Focus: All tables include measurable benchmarks (accuracy, costs, adoption rates)
Usage Guidance
15.1. Strategic Selection Framework
- Assess Clinical Requirements: Evaluate the specific clinical use cases, performance requirements, and integration needs
- Evaluate Resource Constraints: Consider financial resources, technical expertise, and infrastructure capabilities
- Analyze Regulatory Environment: Understand regulatory requirements, validation needs, and compliance obligations
- Consider Long-term Strategy: Align AI selection with organizational strategy, innovation goals, and sustainability objectives
- Develop Implementation Plan: Create comprehensive implementation, training, and maintenance plans
15.2. Policy Recommendations
15.2.1. For Healthcare Organizations
- Invest in developing internal expertise for evaluating and implementing AI solutions
- Establish clear governance frameworks for AI adoption, including ethical guidelines and oversight mechanisms
- Participate in open-source communities to influence development and share best practices
- Develop comprehensive data management strategies that address privacy and security concerns
15.2.2. For Policymakers
- Develop adaptive regulatory frameworks that can accommodate rapid technological advancements
- Support standardization efforts for AI validation and interoperability
- Fund research on AI safety, ethics, and implementation best practices
- Address disparities in AI access through funding programs and technical assistance
15.2.3. For Researchers and Developers
- Develop improved validation methodologies for AI systems in healthcare settings
- Address bias and fairness issues in medical AI through diverse training data and algorithmic improvements
- Enhance explainability and transparency capabilities for both open-source and proprietary systems
- Explore hybrid approaches that combine the strengths of different AI paradigms
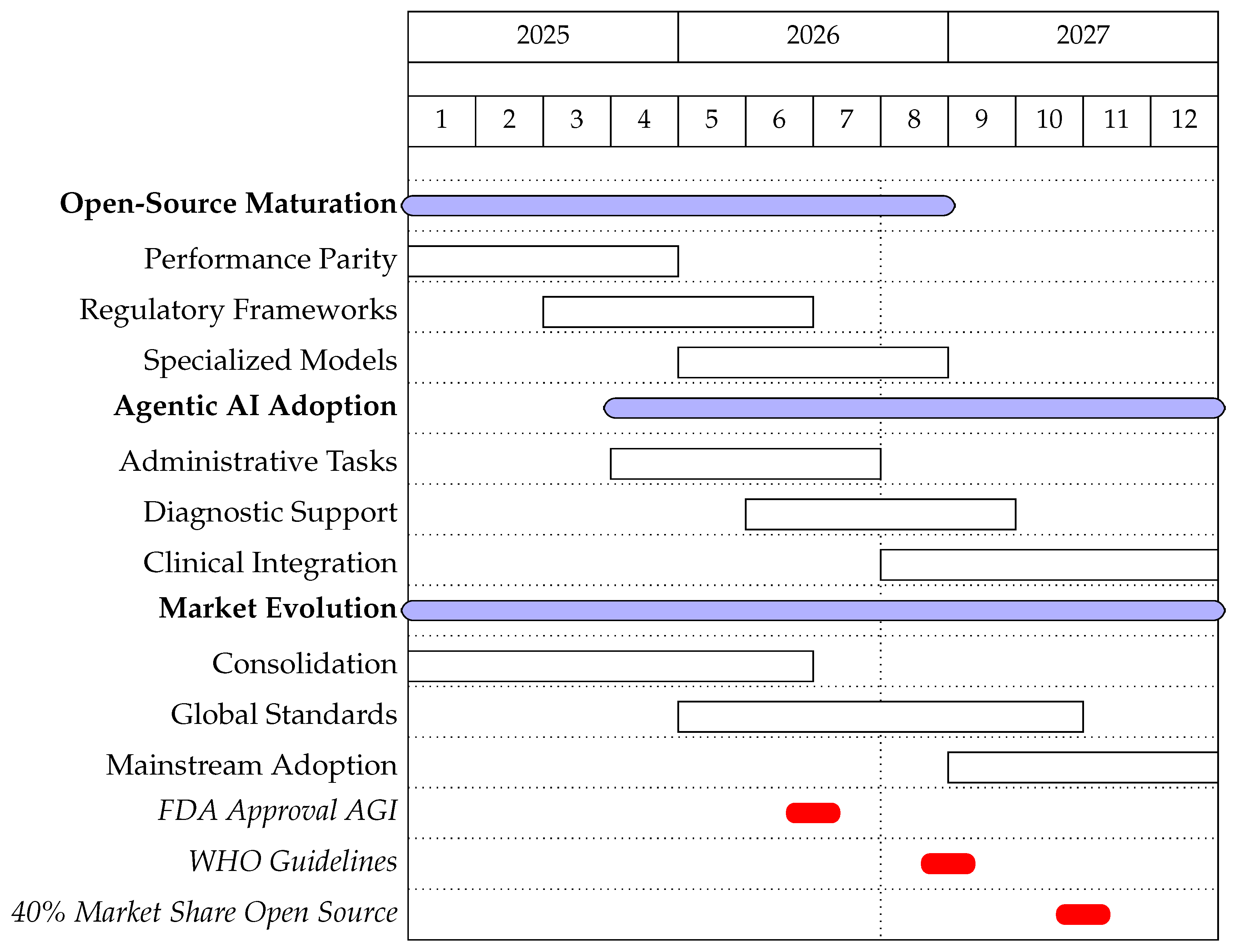
16. Future Timeline and Projections (2025–2030)
16.1. Agentic AI in Healthcare
16.2. Global AI Development Patterns
16.3. Market Evolution and Investment Trends
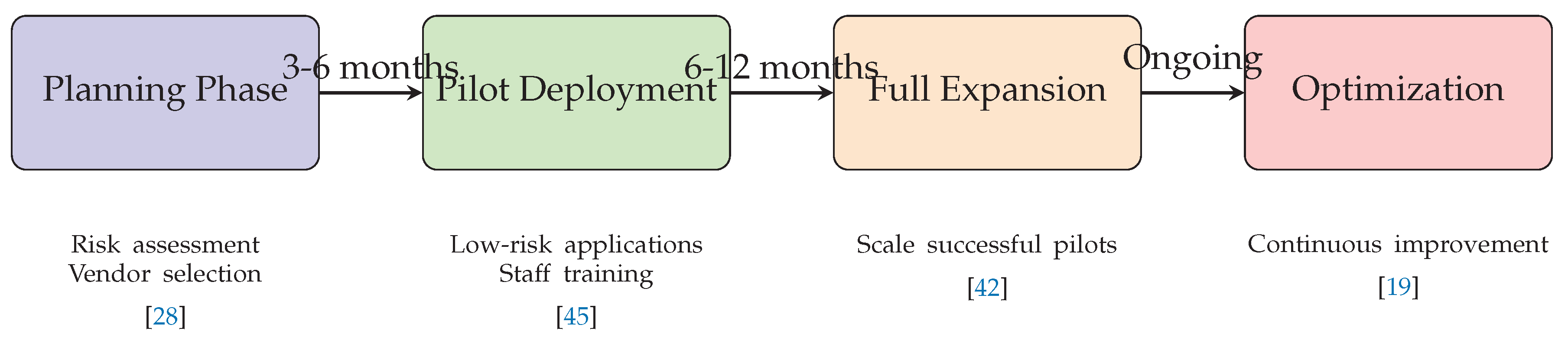
16.4. Near-Term Developments (2025–2026)
- Open-Source Performance Parity: By late 2025, open-source models are projected to achieve performance parity with proprietary systems in 85% of diagnostic applications [20]. This will be driven by community-driven improvements and increased investment in open-source medical AI development.
- Regulatory Frameworks: Major regulatory bodies including the FDA and EMA will establish formal guidelines for open-source AI validation in healthcare by Q2 2026 [28]. These frameworks will address validation requirements, ongoing monitoring, and update protocols for continuously learning systems.
- Agentic AI Adoption: Agentic AI systems will see initial clinical deployment in 2026, particularly for administrative tasks and preliminary diagnostic screening [46]. Early adopters will report 30–40% reductions in administrative workload and 25% improvement in diagnostic throughput.
- Market Consolidation: The healthcare AI market will experience significant consolidation, with the number of major players reducing from the current 200+ to approximately 50 by the end of 2026 [44]. This consolidation will be driven by regulatory requirements and the need for substantial validation resources.
16.5. Mid-Term Evolution (2027–2028)
- Hybrid Model Dominance: By 2027, 70% of healthcare organizations will adopt hybrid approaches combining open-source core technologies with proprietary specialized modules [42]. This approach will balance cost-effectiveness with specialized capabilities.
- Interoperability Standards: Comprehensive interoperability standards for healthcare AI systems will be established by 2028, enabling seamless data exchange and model integration across platforms [58]. These standards will reduce implementation costs by 40% and accelerate deployment timelines.
- Global AI Infrastructure: China’s investment in AI self-reliance will yield significant results by 2028, with Chinese open-source models capturing 35% of the global healthcare AI market [55]. This will create a more diversified global AI ecosystem.
- Specialized AI Agents: Disease-specific AI agents will emerge, with targeted solutions for oncology, cardiology, and neurology achieving FDA approval by 2028 [51]. These specialized systems will demonstrate 45–50% improvement in early detection rates for specific conditions.
16.6. Long-Term Transformation (2029–2030)
- AI-First Clinical Workflows: By 2030, 80% of healthcare organizations will have implemented AI-first clinical workflows, where AI systems serve as primary diagnostic assistants with human oversight [52]. This shift will reduce diagnostic errors by 60% and improve treatment consistency.
- Personalized Medicine at Scale: AI-enabled personalized treatment plans will become standard practice by 2029, leveraging patient-specific data to optimize therapeutic outcomes [43]. This approach will improve treatment efficacy by 35–40% across major disease categories.
- Democratization of Healthcare AI: Open-source platforms will enable widespread access to advanced AI capabilities, particularly in resource-constrained settings . By 2030, developing regions will achieve 70% of the AI healthcare capability of developed markets at 20% of the cost.
- Regulatory Maturity: Comprehensive international regulatory frameworks for healthcare AI will be established by 2030, enabling global deployment while maintaining safety standards [56]. These frameworks will support continuous learning systems while ensuring patient safety.
16.7. Technology-Specific Projections
16.7.1. Open-Source Advancements
- 2026: Community-developed medical AI models will achieve performance exceeding proprietary systems in specialized domains including medical imaging analysis and genomic interpretation [18].
- 2027: Open-source platforms will develop comprehensive toolchains for medical AI validation, reducing compliance costs by 60% and accelerating deployment timelines [28].
- 2028: Federated learning approaches will become standard for open-source medical AI, enabling continuous improvement while maintaining data privacy [21].
16.7.2. Proprietary Innovation
- 2026: Integrated AI platforms from major vendors (Google, Siemens, etc.) will offer comprehensive clinical workflow solutions covering diagnosis, treatment planning, and outcome monitoring [59].
- 2028: Proprietary systems will dominate high-complexity clinical applications requiring extensive validation and regulatory compliance, maintaining 70% market share in these segments [16].
- 2030: Annual licensing costs for proprietary systems will decrease by 40–50% due to competition from open-source alternatives, making advanced capabilities more accessible [42].
16.8. Market and Adoption Projections
- 2026: Global healthcare AI market reaches $85–95 billion, with open-source solutions capturing 35% market share [1].
- 2028: AI-assisted diagnoses will account for 60% of all medical imaging interpretations in developed markets [17].
- 2030: Healthcare AI will generate $350–400 billion in annual healthcare cost savings globally through improved efficiency and outcomes [43].
16.9. Critical Challenges and Considerations
- Data Privacy and Security: Evolving regulations will require continuous adaptation of both open-source and proprietary systems [3].
- Workforce Transformation: Healthcare professionals will require extensive retraining, with 40–50% of current clinical tasks automated by 2030 [45].
- Ethical Governance: Robust ethical frameworks must be developed to address algorithmic bias, accountability, and patient consent in AI-driven healthcare [60].
16.10. Clinical Applications
-
By 2026:
-
By 2028:
16.11. Economic Impact
16.12. Technological Evolution
-
AGI Architectures:
-
Data Infrastructure:
16.13. Societal Implications
16.14. Challenges and Future Directions
16.14.1. Technical Limitations
16.14.2. Ethical Dilemmas
16.14.3. Policy Lag
17. Conclusion
17.1. Key Findings
17.2. Regulatory and Governance Considerations
- Tiered Risk-Management Framework: Implement certification protocols aligned with international standards (e.g., WHO’s 95% explainability threshold [8]) that classify systems by clinical risk, mandating appropriate human oversight levels.
- Public-Private Collaboration: Scale pilot programs and sandbox environments, such as California’s 78% successful AGI pilots [48], to evaluate multi-agent systems in real-world settings while ensuring safety and regulatory compliance.
- Continuous Monitoring and Auditing: Mandate real-time bias audits, leveraging federated learning [38] to preserve privacy and facilitate collaborative improvement across institutions.
- International Harmonization: Encourage global alignment of regulations to reduce fragmentation, enabling cross-border deployment and standardized validation of AGI systems.
17.3. Affordability and Economic Feasibility
- Implementation Costs: High upfront costs (e.g., $2M barriers [11]) require careful planning for deployment in resource-constrained environments.
- Cost-Effectiveness: Hybrid approaches combining open-source AGI with proprietary modules can optimize both financial and operational outcomes, providing scalable, affordable solutions for diverse healthcare settings.
17.4. Strategic Recommendations
- Tiered Certification: Align risk management and explainability thresholds with WHO standards [8], allowing regional flexibility based on healthcare system maturity.
- Public-Private Sandboxes: Expand pilot programs for multi-agent testing [48], enabling real-world evaluation with controlled oversight.
- Continuous Bias and Safety Monitoring: Use real-time auditing informed by Medicare Advantage appeals data [7] to maintain trust, safety, and compliance.
- Hybrid Ecosystem Development: Leverage open-source transparency and customization alongside proprietary reliability and support, creating an adaptive, cost-effective AGI infrastructure.
- Workforce Transition Programs: Invest in retraining clinicians and administrative staff to operate safely in AI-augmented workflows.
17.5. Final Words
- Both approaches offer distinct advantages; performance gaps are narrowing.
- Open-source systems excel in transparency, customization, affordability, and privacy.
- Proprietary systems provide regulatory reliability, technical support, and stability for mission-critical applications.
- Regulatory governance and affordability remain pivotal for safe, scalable AGI adoption.
Declaration
References
- AI in Healthcare Market Size, Share | Growth Report [2025-2032].
- Google for Health - Advancing Cutting-edge AI Capabilities.
- Temsah, A.; Alhasan, K.; Altamimi, I.; Jamal, A.; Al-Eyadhy, A.; Malki, K.H.; Temsah, M.H.; Temsah, A.; Alhasan, K.; Altamimi, I.; et al. DeepSeek in Healthcare: Revealing Opportunities and Steering Challenges of a New Open-Source Artificial Intelligence Frontier. Cureus 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- MedGemma: Our most capable open models for health AI development.
- abhinav.japesh@superagi. Revolutionizing Healthcare: A Large Agentic Model Case Study on Predicting Patient Outcomes with Unprecedented Accuracy, 2025. Section: Uncategorized.
- Revolutionizing Claims Operations With AI and Agentic Workflows, 2025. Section: Commentary.
- Herman, Bob, C.R. Denied by AI: How Medicare Advantage plans use algorithms to cut off care for seniors in need, 2023.
- Ethics and Governance of Artificial Intelligence for Health: Large Multi-Modal Models. WHO Guidance, 1st ed ed.; World Health Organization: Geneva, 2024. [Google Scholar]
- Bonta, A. Joint Informational Hearing.
- Opportunities for gen AI in public health | McKinsey.
- Agentic AI in Healthcare: Use Cases, Cost & Challenges, 2025.
- Artificial Intelligence and Its Potential Effects on the Economy and the Federal Budget | Congressional Budget Office, 2024.
- Agentic AI and the Future of Healthcare, 2025. Section: Agentic AI.
- Lamb, C.P.M.J. Op-ed: How agentic AI is shaping health care’s future, 2025. Section: Op-Ed.
- Google for Health - Advancing Cutting-edge AI Capabilities.
- Securing AI: The Power of Proprietary Models in Protecting Health Data.
- School, H.M. Open-source AI tool competes with leading proprietary models in medical diagnosis, 2025. Section: Medical News.
- Open-Source AI Matches Top Proprietary LLM in Solving Tough Medical Cases | Harvard Medical School, 2025.
- Riedemann, L.; Labonne, M.; Gilbert, S. The path forward for large language models in medicine is open. npj Digital Medicine 2024, 7, 339. [Google Scholar] [CrossRef] [PubMed]
- Buckley, T.A.; Crowe, B.; Abdulnour, R.E.E.; Rodman, A.; Manrai, A.K. Comparison of Frontier Open-Source and Proprietary Large Language Models for Complex Diagnoses. JAMA Health Forum 2025, 6, e250040–e250040. [Google Scholar] [CrossRef] [PubMed]
- Campus, U.o.C.A.M. Open-source AI tool matches commercial systems in medical scan reporting, 2025. Section: Medical News.
- Witherspoon, A. How Agentic AI is transforming healthcare delivery, 2025.
- Agentic AI in Healthcare Use Cases, Benefits Strategies.
- abhinav.japesh@superagi. Common Agentic AI Implementation Mistakes in Healthcare: Case Study and Expert Advice for Avoiding Costly Errors, 2025. Section: Insights.
- Bioengineer. Open-Source AI Rivals Leading Proprietary Models in Tackling Complex, 2025. Section: Technology.
- Wheeler, K. Inside Google’s MedGemma Models for Healthcare AI, 2025.
- Empowering Public Healthcare with Open Source Language Models.
- Weber, S. Open-Source Software in Healthcare: Promise and Precautions for HI Professionals.
- The evolving ethics and governance landscape of agentic AI | IBM, 2025.
- Bouderhem, R. Shaping the future of AI in healthcare through ethics and governance. Humanities and Social Sciences Communications 2024, 11, 416. [Google Scholar] [CrossRef]
- AI Blog: Expert Insights and Advice.
- A pro-innovation approach to AI regulation: government response.
- Ethics and Governance of Artificial Intelligence for Health: WHO Guidance, 1st ed ed.; World Health Organization: Geneva, 2021.
- RFI: Agentic Artificial Intelligence systems | ARPA-H, 2024.
- mondar bce86d. Agentic AI in Healthcare : Transforming Patient Care with Intelligence, 2025.
- Seizing healthcare’s agentic AI opportunity.
- Generative AI Healthcare: 15 Use Cases with Examples [’25].
- Priorities for an AI in health care strategy | The Health Foundation.
- F, M. Battle of the AI Titans GPT 5 vs Grok 4 vs Microsoft Copilot Who Wins the Next Gen AI Showdown, 2025.
- Agentic AI in Healthcare Management: Uses and Benefits.
- Healthcare Agentic AI: Benefits & Use Cases.
- Engineering. Open source vs. proprietary AI tools: Making strategic choices for long-term success, 2025.
- Masood, A. Unlocking Strategic Elasticity: A Healthcare Executive’s Guide to High-Efficiency Lean AI, 2025.
- Duranton, S. What Leaders Need To Know About Open-Source Vs. Proprietary Models. Section: AI.
- How AI is Transforming Healthcare Delivery and Workforce Management.
- industry, E.L.T.E.L.i.a.f.w.w.s.i.t.h. What Is Agentic AI and How Can It Be Used in Healthcare?
- How agentic AI systems can solve problems in healthcare today.
- Regan, J. State of California Benefits and Risks of Generative Artificial Intelligence Report.
- Group, P.L.I. Agentic AI and the Architecture of Healthcare Transformation, 2025.
- Agentic AI, Generative AI and AI Governance.
- News, Q. Medical AI Agents Boost Accuracy For Complex Health Queries, 2025. Section: Artificial Intelligence.
- The Rise of Agentic AI in Healthcare | athenahealth.
- Coleman, K. Lowering Health Care Costs Through AI: The Possibilities and Barriers, 2024.
- Coat, A.i.L. AI Healthcare Startups Raised 2.2 Billion in January 2025, 2025.
- Chang, W.; Arcesati, R.; Hmaidi, A. CHINA’S DRIVE TOWARD SELF-RELIANCE IN ARTIFICIAL INTELLIGENCE.
- Chase, M.S.; Marcellino, W. Incentives for U.S.-China Conflict, Competition, and Cooperation Across Artificial General Intelligence’s Five Hard National Security Problems. Technical report, 2025.
- Baier, P. OpenAI Releases Two Powerful Open Source AI Models: What You Need To Know, 2025.
- The influence of open source and AI in healthcare.
- Dany, K. Siemens Healthineers AI Strategy Analysis of Dominance in Medical Tech AI Klover ai, 2025.
- F, M. The State of Artificial Intelligence: Agentic Agents, Workforce Upheaval & Global Governance / Updated: 2025, July 6th, 23:59 CET, 2025.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




