Submitted:
05 September 2025
Posted:
05 September 2025
You are already at the latest version
Abstract
Keywords:
INTRODUCTION
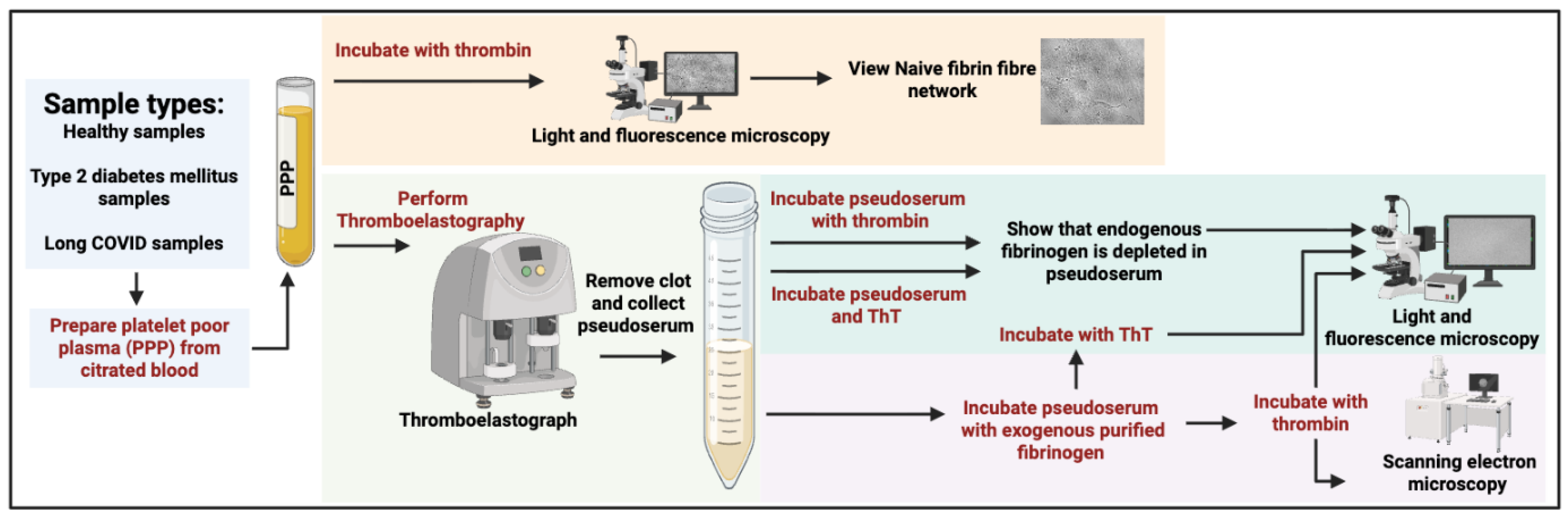
METHODS
Ethical clearance
Sample Cohorts and Collection
Fibrin Network Formation of PPP Samples
Thromboelastography (TEG®) of PPP Samples, and Obtainment of Pseudoserum
Fibrin Networks of Purified Fibrinogen Exposed to Pseudoserum: Light Microscopy
Thioflavin T Staining of Pseudoserum and Fibrin Networks
Fibrin Networks of Purified Fibrinogen Exposed to Pseudoserum: Scanning Electron Microscopy
Statistical Analysis
RESULTS
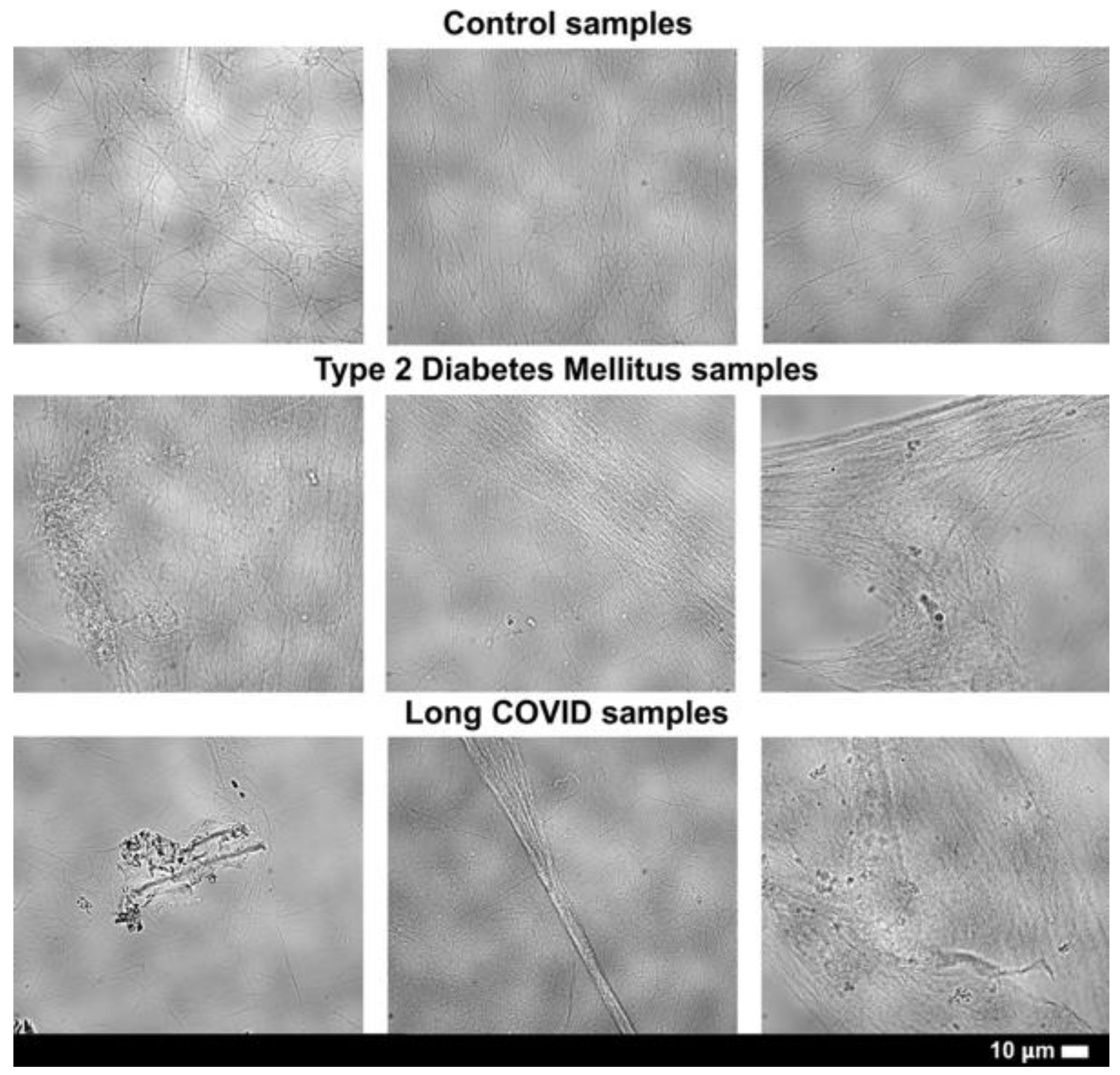
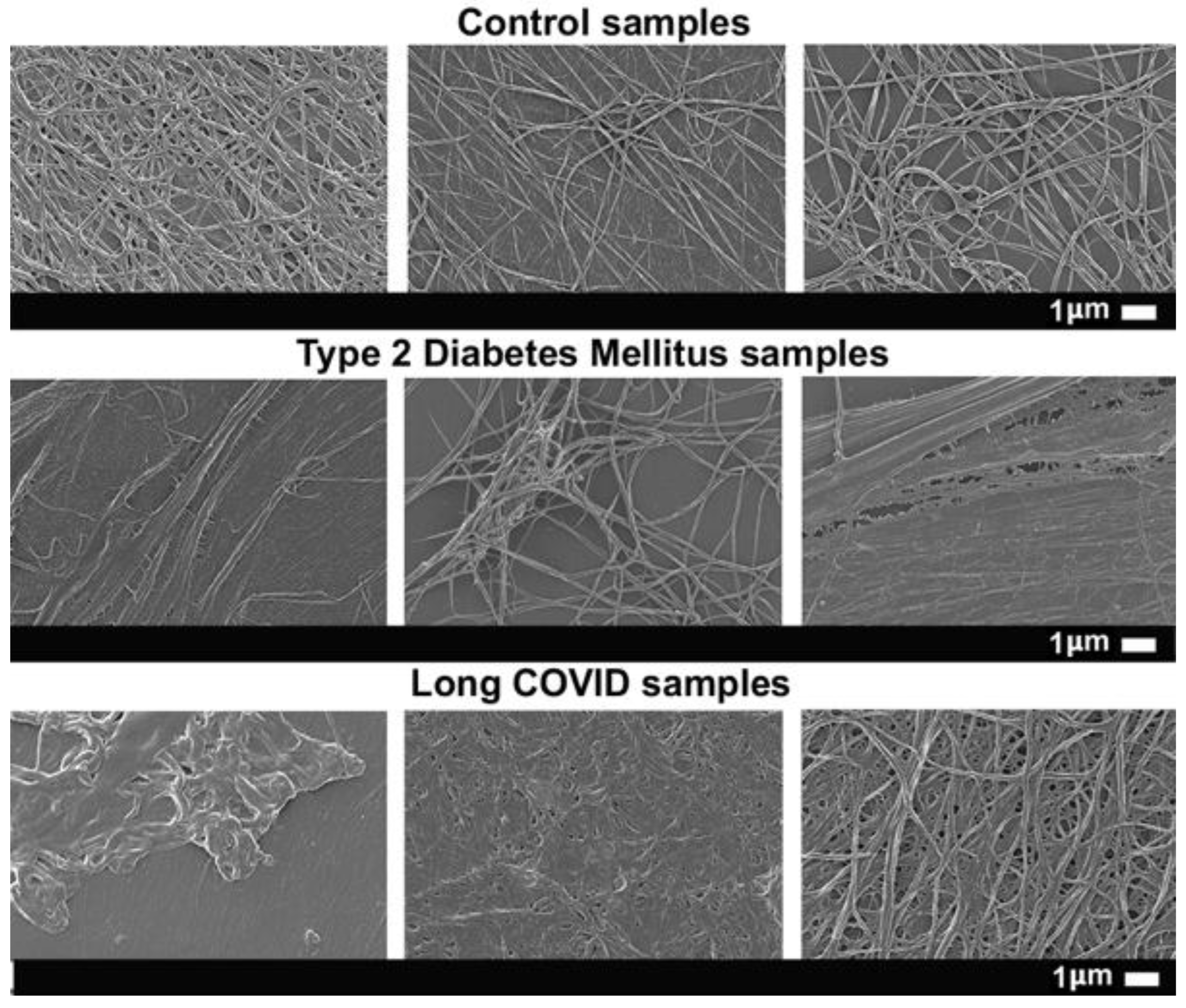
Thrombin-Induced PPP Fibrin Networks
Thromboelastography (TEG®)
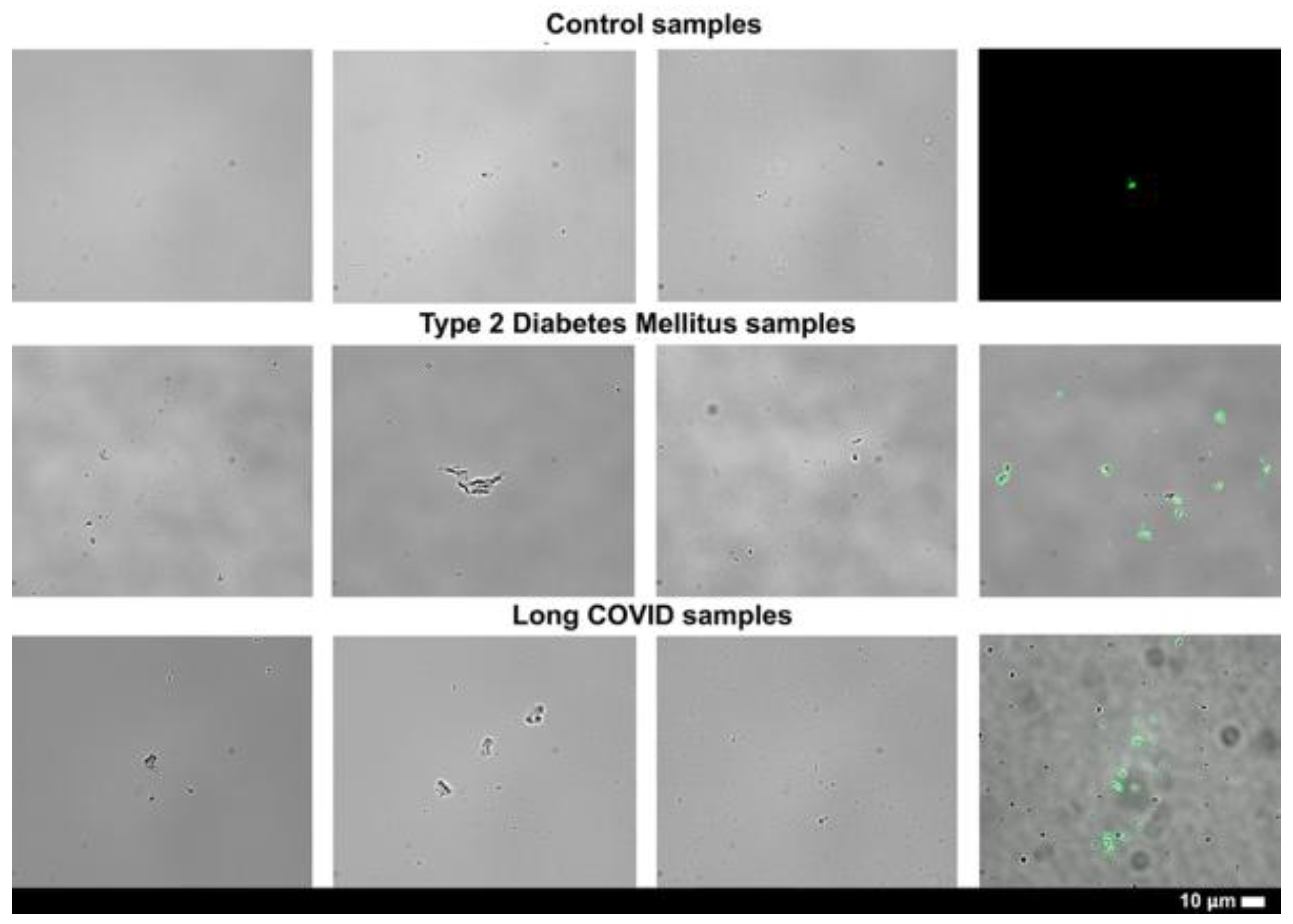
Pseudoserum Exposed to Thrombin
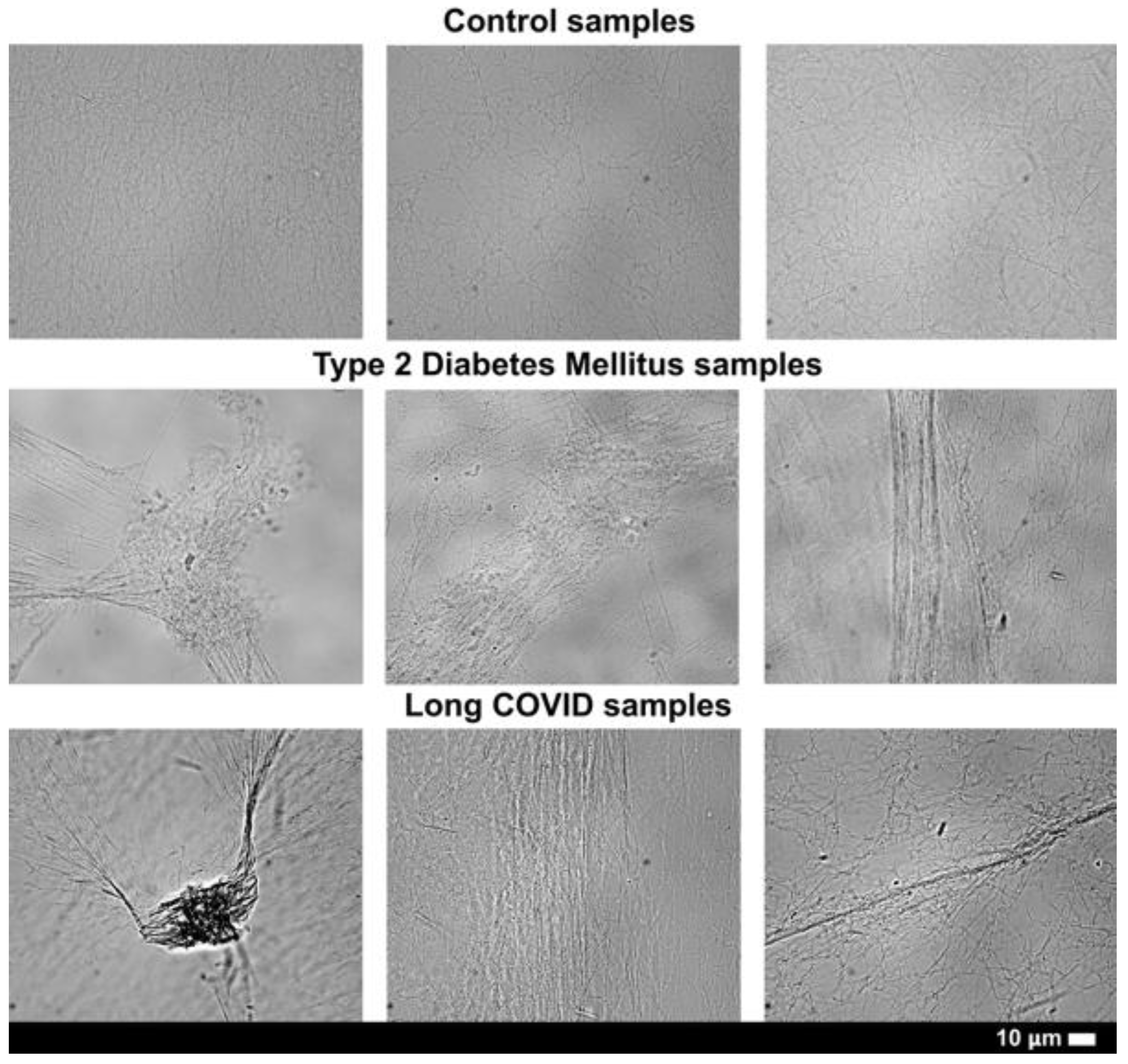
Fibrin Networks of Purified Fibrinogen Exposed to Pseudoserum
DISCUSSION
CONCLUSION AND FUTURE DIRECTIONS
Author Contributions
Acknowledgements and Funding
Conflicts of Interest
References
- S. Palta, R. Saroa, A. Palta, Overview of the coagulation system, Indian Journal of Anaesthesia 58(5) (2014).
- S. Kattula, J.R. Byrnes, A.S. Wolberg, Fibrinogen and fibrin in hemostasis and thrombosis, Arteriosclerosis, thrombosis, and vascular biology 37(3) (2017) e13-e21. [CrossRef]
- R.I. Litvinov, M. Pieters, Z. de Lange-Loots, J.W. Weisel, Fibrinogen and Fibrin, in: J.R. Harris, J. Marles-Wright (Eds.), Macromolecular Protein Complexes III: Structure and Function, Springer International Publishing, Cham, 2021, pp. 471-501.
- C. Longstaff, K. Kolev, Basic mechanisms and regulation of fibrinolysis, Journal of Thrombosis and Haemostasis 13(S1) (2015) S98-S105. [CrossRef]
- S.T. Lord, Molecular mechanisms affecting fibrin structure and stability, Arteriosclerosis, thrombosis, and vascular biology 31(3) (2011) 494-499. [CrossRef]
- J.W. Weisel, R.I. Litvinov, Fibrin formation, structure and properties, Fibrous proteins: structures and mechanisms (2017) 405-456.
- M. Ząbczyk, R.A.S. Ariëns, A. Undas, Fibrin clot properties in cardiovascular disease: from basic mechanisms to clinical practice, Cardiovascular Research 119(1) (2023) 94-111. [CrossRef]
- S. Bloemen, D. Huskens, J. Konings, R.M. Kremers, A. Miszta, B. de Laat, H. Kelchtermans, Interindividual Variability and Normal Ranges of Whole Blood and Plasma Thrombin Generation, The Journal of Applied Laboratory Medicine 2(2) (2017) 150-164. [CrossRef]
- W.B. Dunn, D. Broadhurst, P. Begley, E. Zelena, S. Francis-McIntyre, N. Anderson, M. Brown, J.D. Knowles, A. Halsall, J.N. Haselden, A.W. Nicholls, I.D. Wilson, D.B. Kell, R. Goodacre, Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry, Nat Protoc 6(7) (2011) 1060-83. [CrossRef]
- W.B. Dunn, W. Lin, D. Broadhurst, P. Begley, M. Brown, E. Zelena, A.A. Vaughan, A. Halsall, N. Harding, J.D. Knowles, S. Francis-McIntyre, A. Tseng, D.I. Ellis, S. O'Hagan, G. Aarons, B. Benjamin, S. Chew-Graham, C. Moseley, P. Potter, C.L. Winder, C. Potts, P. Thornton, C. McWhirter, M. Zubair, M. Pan, A. Burns, J.K. Cruickshank, G.C. Jayson, N. Purandare, F.C. Wu, J.D. Finn, J.N. Haselden, A.W. Nicholls, I.D. Wilson, R. Goodacre, D.B. Kell, Molecular phenotyping of a UK population: defining the human serum metabolome, Metabolomics 11(1) (2015) 9-26. [CrossRef]
- M. Levi, Inflammation and Coagulation, Inflammation2017, pp. 833-860.
- M.J. Page, G.J. Thomson, J.M. Nunes, A.-M. Engelbrecht, T.A. Nell, W.J. De Villiers, M.C. De Beer, L. Engelbrecht, D.B. Kell, E. Pretorius, Serum amyloid A binds to fibrin (ogen), promoting fibrin amyloid formation, Scientific reports 9(1) (2019) 3102. [CrossRef]
- J.J. de Vries, C.J.M. Snoek, D.C. Rijken, M.P.M. de Maat, Effects of Post-Translational Modifications of Fibrinogen on Clot Formation, Clot Structure, and Fibrinolysis, Arteriosclerosis, Thrombosis, and Vascular Biology 40(3) (2020) 554-569.
- Z. Zhang, M. Rodriguez, Z. Zheng, Clot or Not? Reviewing the Reciprocal Regulation Between Lipids and Blood Clotting, Arteriosclerosis, Thrombosis, and Vascular Biology 44(3) (2024) 533-544. [CrossRef]
- J. Bester, C. Matshailwe, E. Pretorius, Simultaneous presence of hypercoagulation and increased clot lysis time due to IL-1β, IL-6 and IL-8, Cytokine 110 (2018) 237-242. [CrossRef]
- J.A. Fernández, H. Deguchi, D.J. Elias, J.H. Griffin, Serum amyloid A4 is a procoagulant apolipoprotein that it is elevated in venous thrombosis patients, Research and Practice in Thrombosis and Haemostasis 4(2) (2020) 217-223. [CrossRef]
- A. Sahni, M. Guo, S.K. Sahni, C.W. Francis, Interleukin-1β but not IL-1α binds to fibrinogen and fibrin and has enhanced activity in the bound form, Blood 104(2) (2004) 409-414. [CrossRef]
- M. Becatti, R. Marcucci, G. Bruschi, N. Taddei, D. Bani, A.M. Gori, B. Giusti, G.F. Gensini, R. Abbate, C. Fiorillo, Oxidative Modification of Fibrinogen Is Associated With Altered Function and Structure in the Subacute Phase of Myocardial Infarction, Arteriosclerosis, Thrombosis, and Vascular Biology 34(7) (2014) 1355-1361. [CrossRef]
- J.R. Coleman, E.E. Moore, L. Schmitt, K. Hansen, N. Dow, K. Freeman, M.J. Cohen, C.C. Silliman, Estradiol provokes hypercoagulability and affects fibrin biology: A mechanistic exploration of sex dimorphisms in coagulation, Journal of Trauma and Acute Care Surgery 94(2) (2023). [CrossRef]
- A.C. Swanepoel, A. Visagie, Z. de Lange, O. Emmerson, V.G. Nielsen, E. Pretorius, The clinical relevance of altered fibrinogen packaging in the presence of 17β-estradiol and progesterone, Thrombosis Research 146 (2016) 23-34. [CrossRef]
- F. Nencini, A. Bettiol, F.R. Argento, S. Borghi, E. Giurranna, G. Emmi, D. Prisco, N. Taddei, C. Fiorillo, M. Becatti, Post-translational modifications of fibrinogen: implications for clotting, fibrin structure and degradation, Molecular Biomedicine 5(1) (2024) 45. [CrossRef]
- G. Targher, I. Pichiri, G. Zoppini, E. Bonora, M. Chonchol, Hemostatic and fibrinolytic abnormalities in endocrine diseases: a narrative review, Seminars in thrombosis and hemostasis, © Thieme Medical Publishers, 2009, pp. 605-612. [CrossRef]
- J.J. de Vries, T. Hoppenbrouwers, C. Martinez-Torres, R. Majied, B. Özcan, M. van Hoek, F.W.G. Leebeek, D.C. Rijken, G.H. Koenderink, M.P.M. de Maat, Effects of Diabetes Mellitus on Fibrin Clot Structure and Mechanics in a Model of Acute Neutrophil Extracellular Traps (NETs) Formation, International Journal of Molecular Sciences 21(19) (2020) 7107. [CrossRef]
- P.E. Makris, E. Pithara, Clinical evaluation of new global clotting assay for monitoring of LMWH treatment: pilot study, Int Angiol 17(2) (1998) 69-79.
- B. Hu, L. Zhang, Z.-S. Zhao, Y.-C. Qi, W. Zhou, Y.-L. Ma, The predictive value of Thromboelastography (TEG) parameters in vascular complications in patients with type 2 diabetes mellitus, Journal of Diabetes and its Complications 36(8) (2022) 108236. [CrossRef]
- L. Pretorius, G.J.A. Thomson, R.C.M. Adams, T.A. Nell, W.A. Laubscher, E. Pretorius, Platelet activity and hypercoagulation in type 2 diabetes, Cardiovascular Diabetology 17(1) (2018) 141. [CrossRef]
- S. Turner, G.J. Laubscher, M.A. Khan, D.B. Kell, E. Pretorius, Accelerating discovery: A novel flow cytometric method for detecting fibrin (ogen) amyloid microclots using long COVID as a model, Heliyon 9(9) (2023). [CrossRef]
- C.F. Dalton, M.I. de Oliveira, P. Stafford, N. Peake, B. Kane, A. Higham, D. Singh, N. Jackson, H.E. Davies, D.A. Price, Increased fibrinaloid microclot counts in platelet-poor plasma are associated with Long COVID, medRxiv (2024) 2024.04. 04.24305318.
- A. Kruger, M. Vlok, S. Turner, C. Venter, G.J. Laubscher, D.B. Kell, E. Pretorius, Proteomics of fibrin amyloid microclots in long COVID/post-acute sequelae of COVID-19 (PASC) shows many entrapped pro-inflammatory molecules that may also contribute to a failed fibrinolytic system, Cardiovascular Diabetology 21(1) (2022) 190. [CrossRef]
- E. Pretorius, C. Venter, G.J. Laubscher, P.J. Lourens, J. Steenkamp, D.B. Kell, Prevalence of readily detected amyloid blood clots in ‘unclotted’Type 2 diabetes mellitus and COVID-19 plasma: a preliminary report, Cardiovascular Diabetology 19 (2020) 1-8. [CrossRef]
- J. Štikarová, R. Kotlín, T. Riedel, J. Suttnar, K. Pimková, L. Chrastinová, J.E. Dyr, The Effect of Reagents Mimicking Oxidative Stress on Fibrinogen Function, The Scientific World Journal 2013(1) (2013) 359621. [CrossRef]
- F. Nencini, E. Giurranna, S. Borghi, N. Taddei, C. Fiorillo, M. Becatti, Fibrinogen Oxidation and Thrombosis: Shaping Structure and Function, Antioxidants 14(4) (2025) 390. [CrossRef]
- M.F. Ruberto, S. Marongiu, T. Congiu, L. Barberini, M. Conti, C. Porcu, D.M. Ntoukas, G. Faa, F. Marongiu, D. Barcellona, The Hypercoagulable State: A Study of Clot Waveform Analysis, Thrombin Generation, and Clot Scanning Electron Microscopy, Archives of Pathology & Laboratory Medicine (2025). [CrossRef]
- O.M. Mereuta, S. Fitzgerald, T.A. Christensen, A.L. Jaspersen, D. Dai, M. Abbasi, T. Puttappa, R. Kadirvel, D.F. Kallmes, K.M. Doyle, W. Brinjikji, High-resolution scanning electron microscopy for the analysis of three-dimensional ultrastructure of clots in acute ischemic stroke, Journal of NeuroInterventional Surgery 13(10) (2021) 906-911. [CrossRef]
- L. Wolff-Trombini, A. Ceripa, J. Moreau, H. Galinat, C. James, N. Westbrook, J.-M. Allain, Microrheology and structural quantification of hypercoagulable clots, Biomed. Opt. Express 14(8) (2023) 4179-4189. [CrossRef]
- E. Pretorius, A.C. Swanepoel, S. DeVilliers, J. Bester, Blood clot parameters: Thromboelastography and scanning electron microscopy in research and clinical practice, Thrombosis research 154 (2017) 59-63. [CrossRef]
- D.A. Fell, Metabolic control analysis: a survey of its theoretical and experimental development, Biochemical Journal 286(2) (1992) 313-330. [CrossRef]
- H. Kacser, J.A. Burns, The control of flux, Symp Soc Exp Biol 27 (1973) 65-104.
- D.B. Kell, H.V. Westerhoff, Metabolic control theory: its role in microbiology and biotechnology, FEMS Microbiology Reviews 2(4) (1986) 305-320.
- J. Bester, E. Pretorius, Effects of IL-1β, IL-6 and IL-8 on erythrocytes, platelets and clot viscoelasticity, Scientific Reports 6(1) (2016) 32188. [CrossRef]
- S.Y. Wang, P. Adejumo, C. See, O.K. Onuma, E.J. Miller, E.S. Spatz, Characteristics of patients referred to a cardiovascular disease clinic for post-acute sequelae of SARS-CoV-2 infection, American Heart Journal Plus: Cardiology Research and Practice 18 (2022) 100176. [CrossRef]
- E. Pretorius, J. Bester, N. Vermeulen, S. Alummoottil, P. Soma, A.V. Buys, D.B. Kell, Poorly controlled type 2 diabetes is accompanied by significant morphological and ultrastructural changes in both erythrocytes and in thrombin-generated fibrin: implications for diagnostics, Cardiovascular Diabetology 14(1) (2015) 30. [CrossRef]
- E. Pretorius, H.M. Oberholzer, W.J. van der Spuy, A.C. Swanepoel, P. Soma, Scanning electron microscopy of fibrin networks in rheumatoid arthritis: a qualitative analysis, Rheumatology international 32(6) (2012) 1611-1615. [CrossRef]
- G.M. De Waal, L. Engelbrecht, T. Davis, W.J. De Villiers, D.B. Kell, E. Pretorius, Correlative Light-Electron Microscopy detects lipopolysaccharide and its association with fibrin fibres in Parkinson’s Disease, Alzheimer’s Disease and Type 2 Diabetes Mellitus, Scientific reports 8(1) (2018) 16798. [CrossRef]
- E. Pretorius, J. Bester, M.J. Page, D.B. Kell, The potential of LPS-binding protein to reverse amyloid formation in plasma fibrin of individuals with Alzheimer-type dementia, Frontiers in aging neuroscience 10 (2018) 257. [CrossRef]
- E. Pretorius, M.J. Page, L. Hendricks, N.B. Nkosi, S.R. Benson, D.B. Kell, Both lipopolysaccharide and lipoteichoic acids potently induce anomalous fibrin amyloid formation: assessment with novel Amytracker™ stains, Journal of The Royal Society Interface 15(139) (2018) 20170941. [CrossRef]
- L.M. Grobbelaar, C. Venter, M. Vlok, M. Ngoepe, G.J. Laubscher, P.J. Lourens, J. Steenkamp, D.B. Kell, E. Pretorius, SARS-CoV-2 spike protein S1 induces fibrin (ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19, Bioscience reports 41(8) (2021) BSR20210611. [CrossRef]

| Demographics | ||||||
|---|---|---|---|---|---|---|
| Control | Long COVID | T2DM | p-value (C vs LC) | p-value (C vs T2DM) | p-value (LC vs T2DM) | |
| Age | 40.55 ± 17.26 | 42.32 ± 12.94 | 52.60 ± 9.66 | 0.99 | 0.17 | 0.11 |
| Sex | 8 Females, 3 Males | 8 Females, 5 Males | 5 Females, 5 Males | Not Applicable | Not Applicable | Not Applicable |
| Comorbidities | ||||||
| T2DM | ||||||
| Hypercholesterolaemia | 80% (8/10) | Polycythemia | 10% (1/10) | |||
| Hypertension | 70% (7/10) | Colon Cancer | 10% (1/10) | |||
| Ischaemic Heart Disease | 30% (3/10) | Glaucoma | 10% (1/10) | |||
| Depression | 30% (3/10) | Hypothyroidism | 10% (1/10) | |||
| Anxiety | 20% (2/10) | Chronic Obstructive Pulmonary Disease | 10% (1/10) | |||
| Asthma | 20% (2/10) | Gout | 10% (1/10) | |||
| Long COVID | ||||||
| Hypercholesterolaemia | 15% (2/13) | Epilepsy | 8% (1/13) | |||
| Hypertension | 8% (1/13) | Rheumatoid Arthritis | 8% (1/13) | |||
| Parameter | Control | Long COVID | T2DM | p-value (C vs LC) | p-value (C vs T2DM) | p-value (LC vs T2DM) |
|---|---|---|---|---|---|---|
| R | 19.10 [24.40] | 11.70 [13.60] | 13.40 [11.30] | 0.01* | 0.03* | >0.99 |
| α-Angle | 30.88 ± 16.48 | 43.53 ± 10.57 | 43.41 ± 14.57 | 0.12 | 0.22 | >0.99 |
| MA | 33.30 [32.80] | 23.90 [30.40] | 30.65 [19.90] | 0.07 | >0.99 | 0.19 |
| MRTG | 5.00 [10.92] | 5.01 [7.93] | 7.40 [9.76] | >0.99 | 0.36 | 0.98 |
| TMRTG | 21.00 [29.34] | 13.00 [14.75] | 15.25 [13.09] | 0.01* | 0.03* | >0.99 |
| TTG | 399.30 [394.70] | 286.00 [365.10] | 367.80 [238.80] | 0.07 | >0.99 | 0.20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.a |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




