Submitted:
18 August 2025
Posted:
19 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Shipment and Receiving Samples at Public Health Authority
2.3. Experimental Design
2.4. Statistical Considerations
2.5. Ethical Consideration
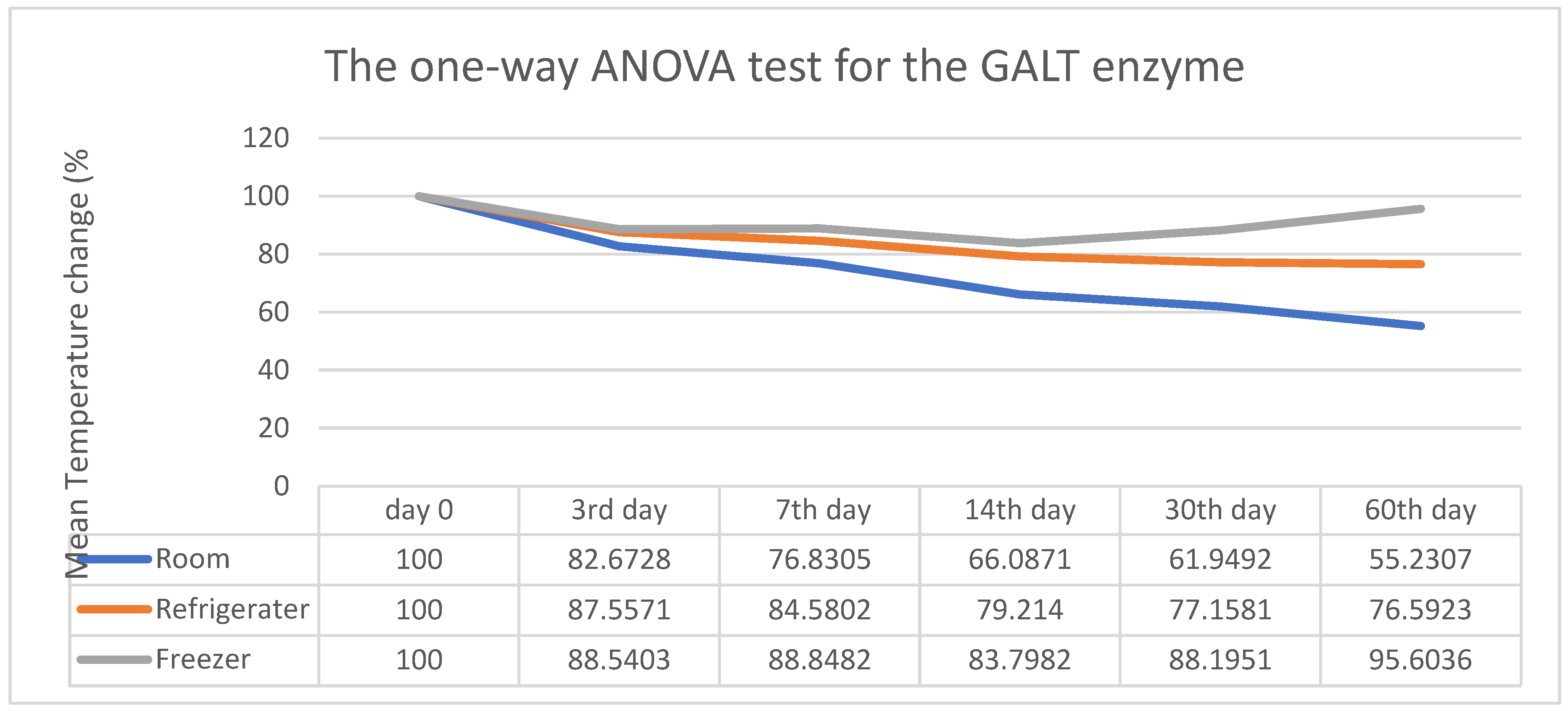
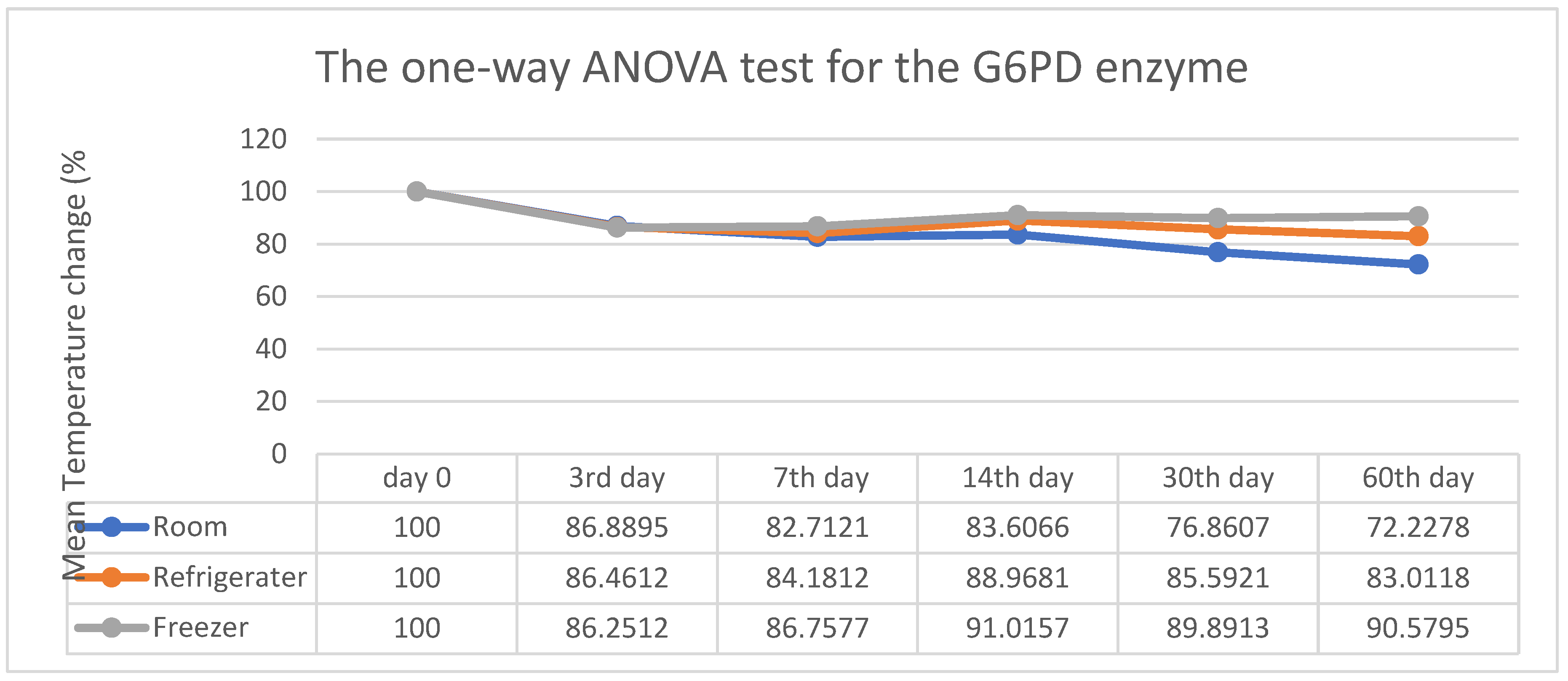
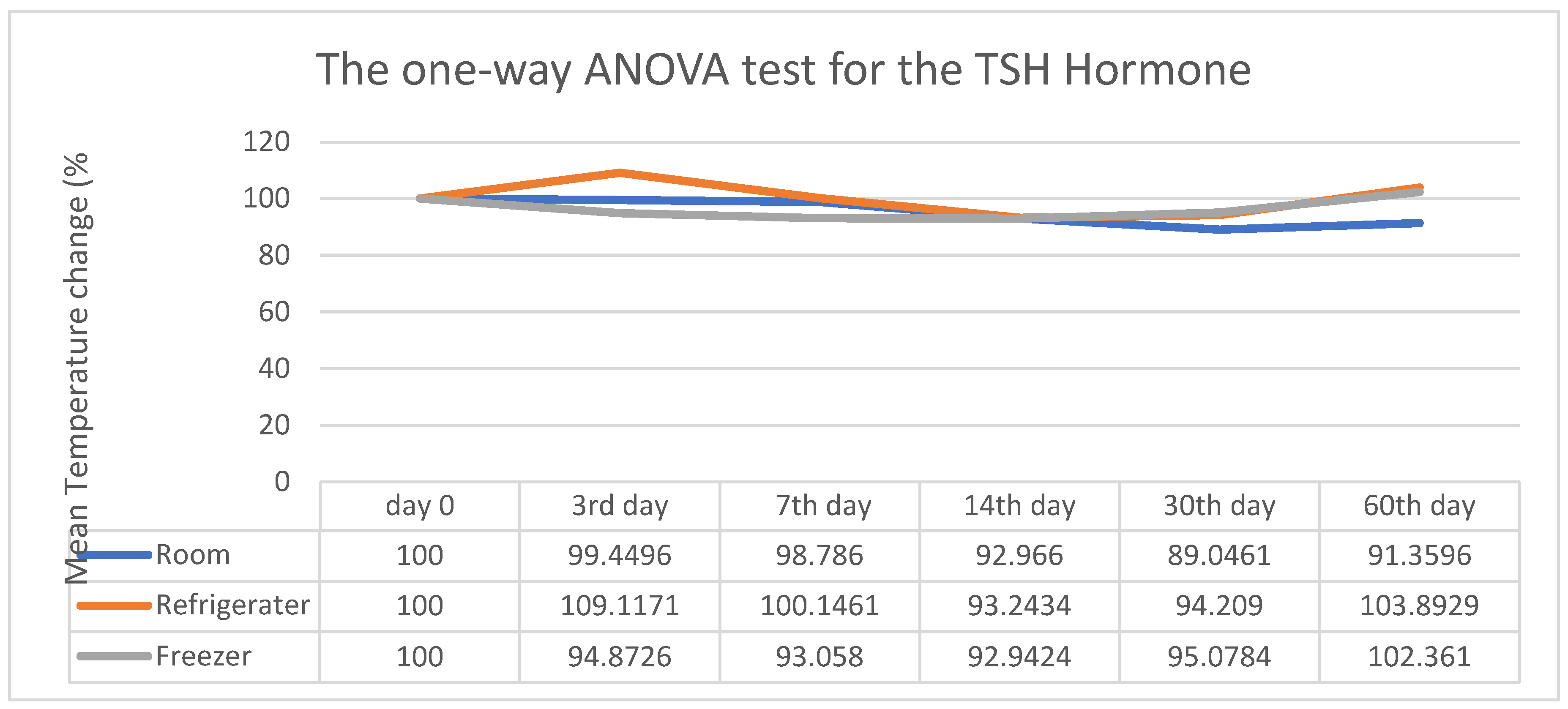
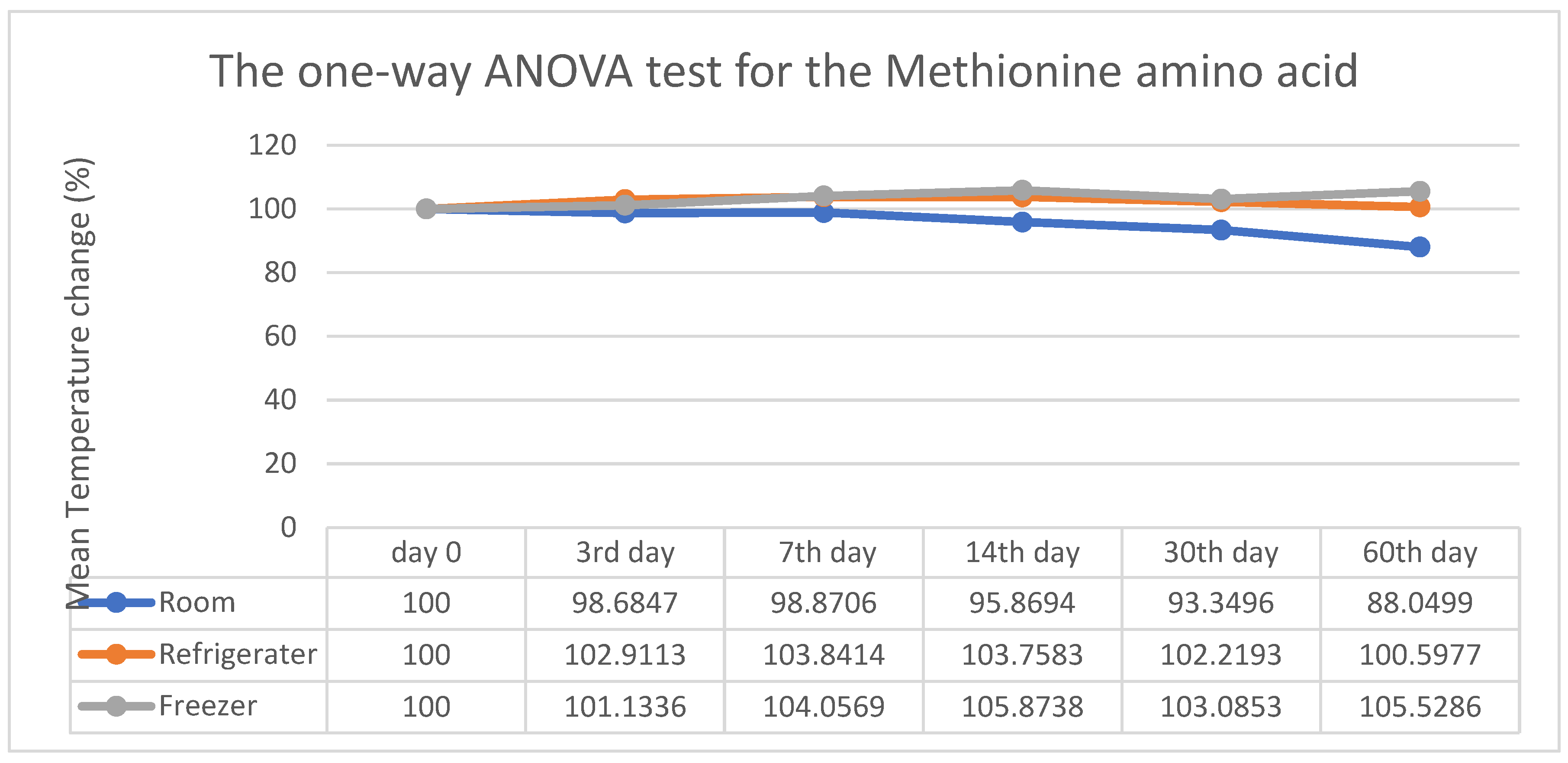
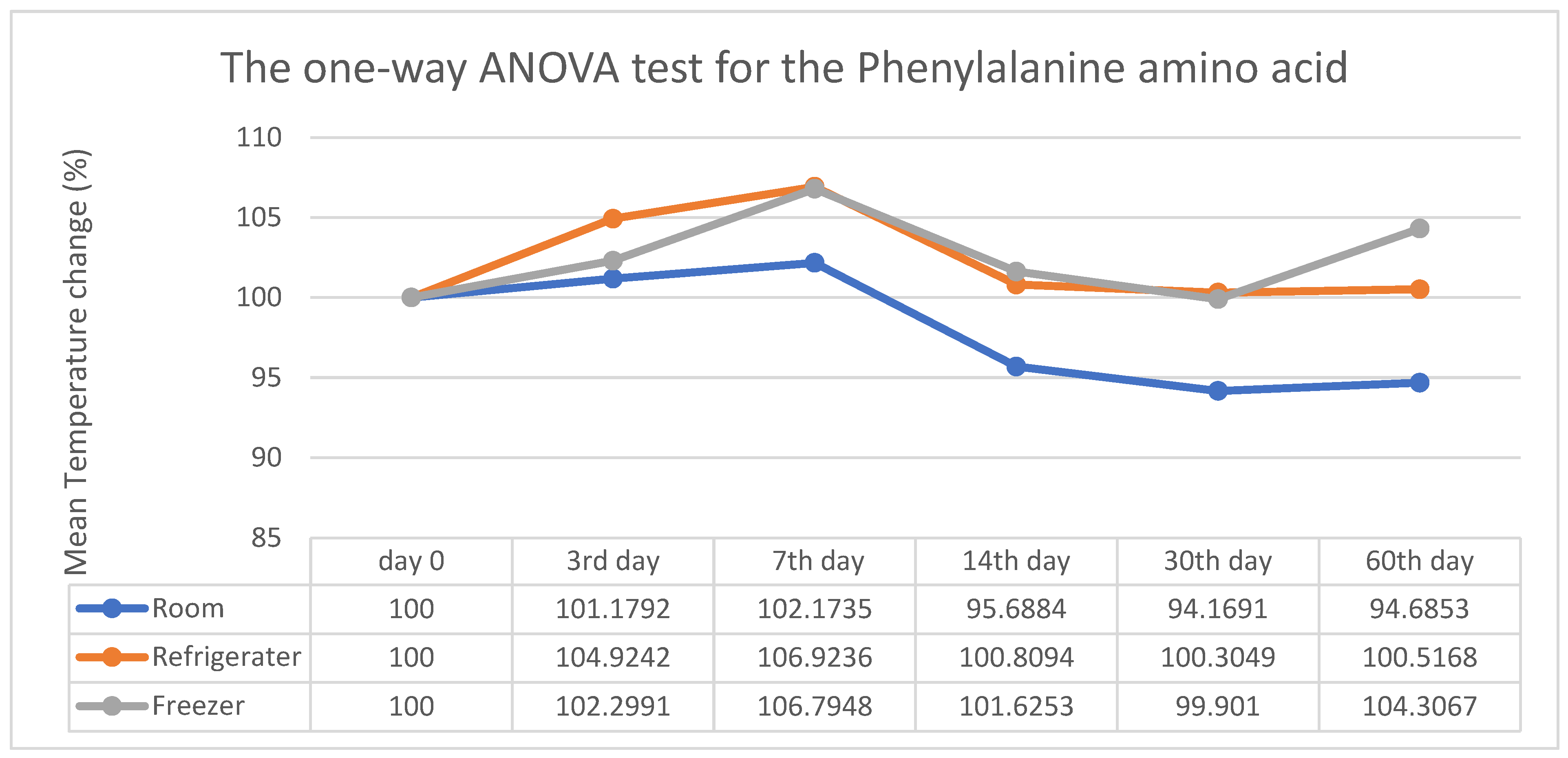
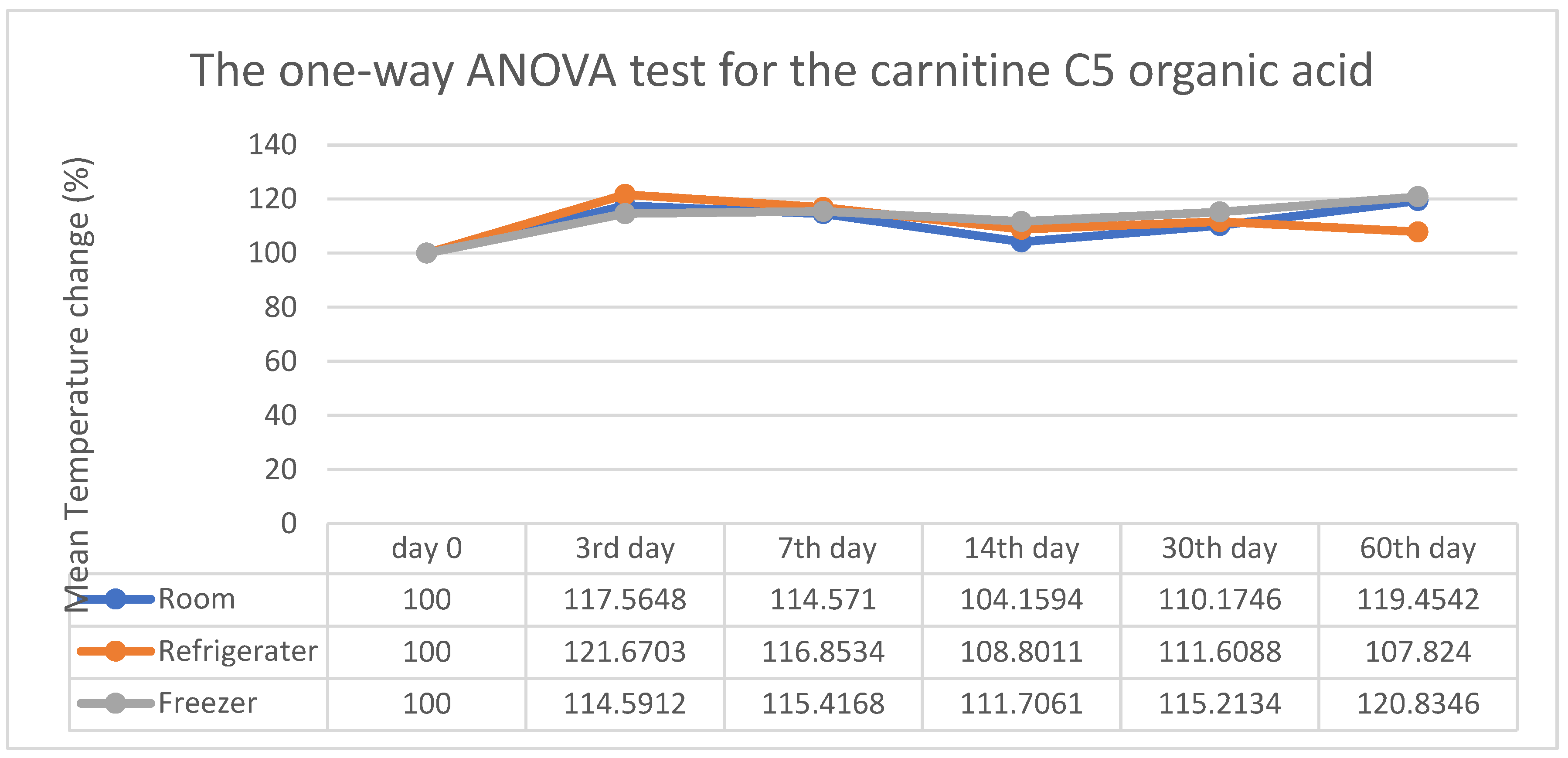
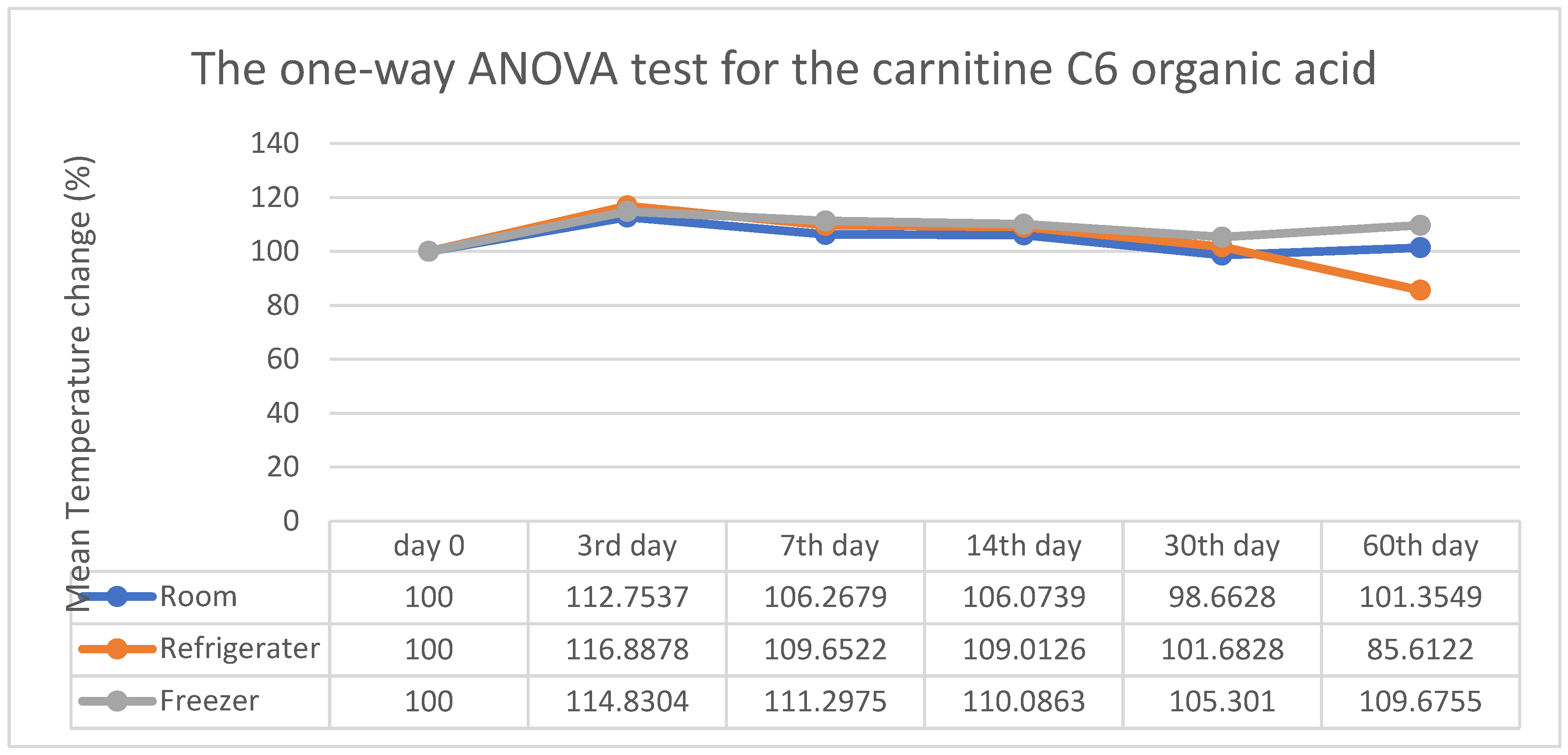
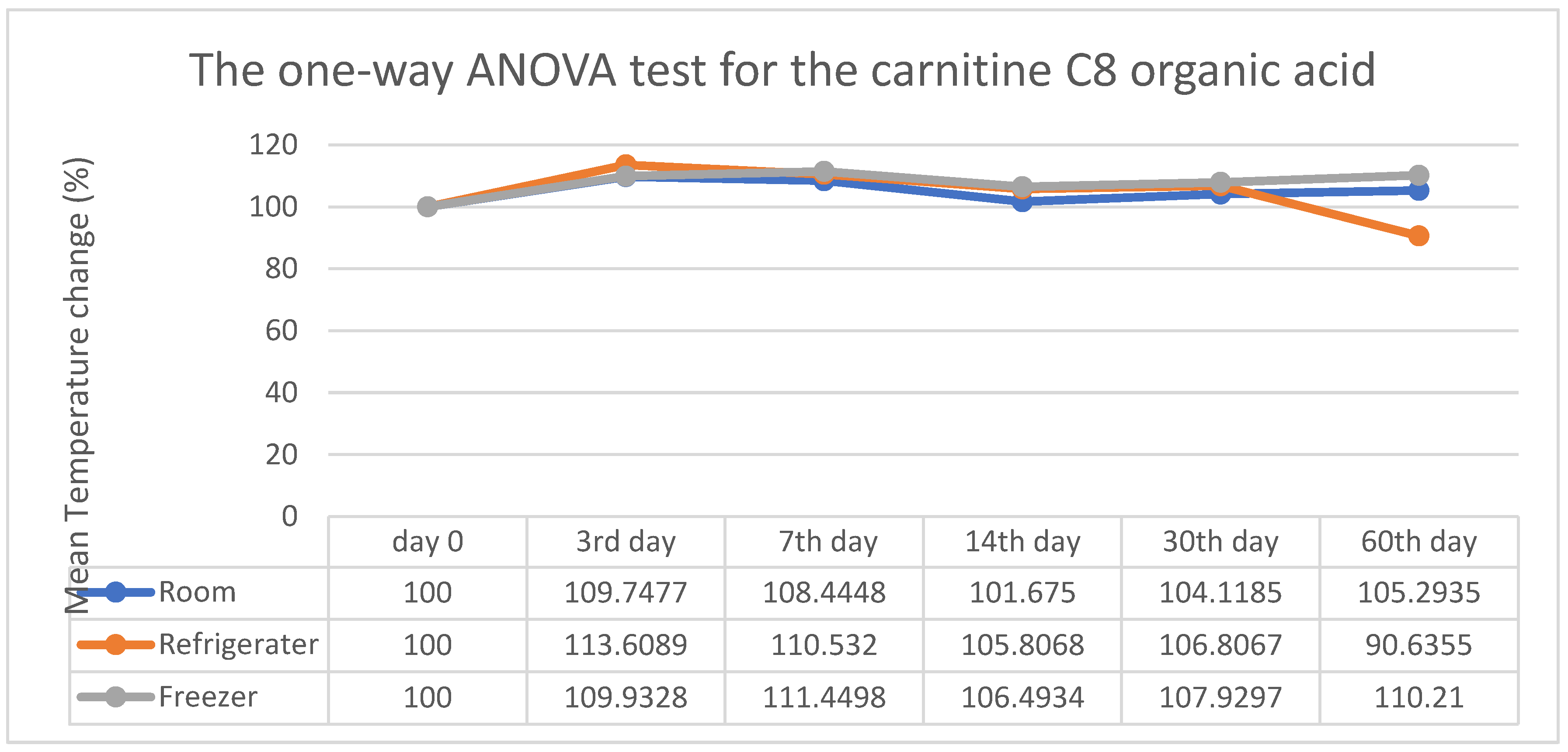
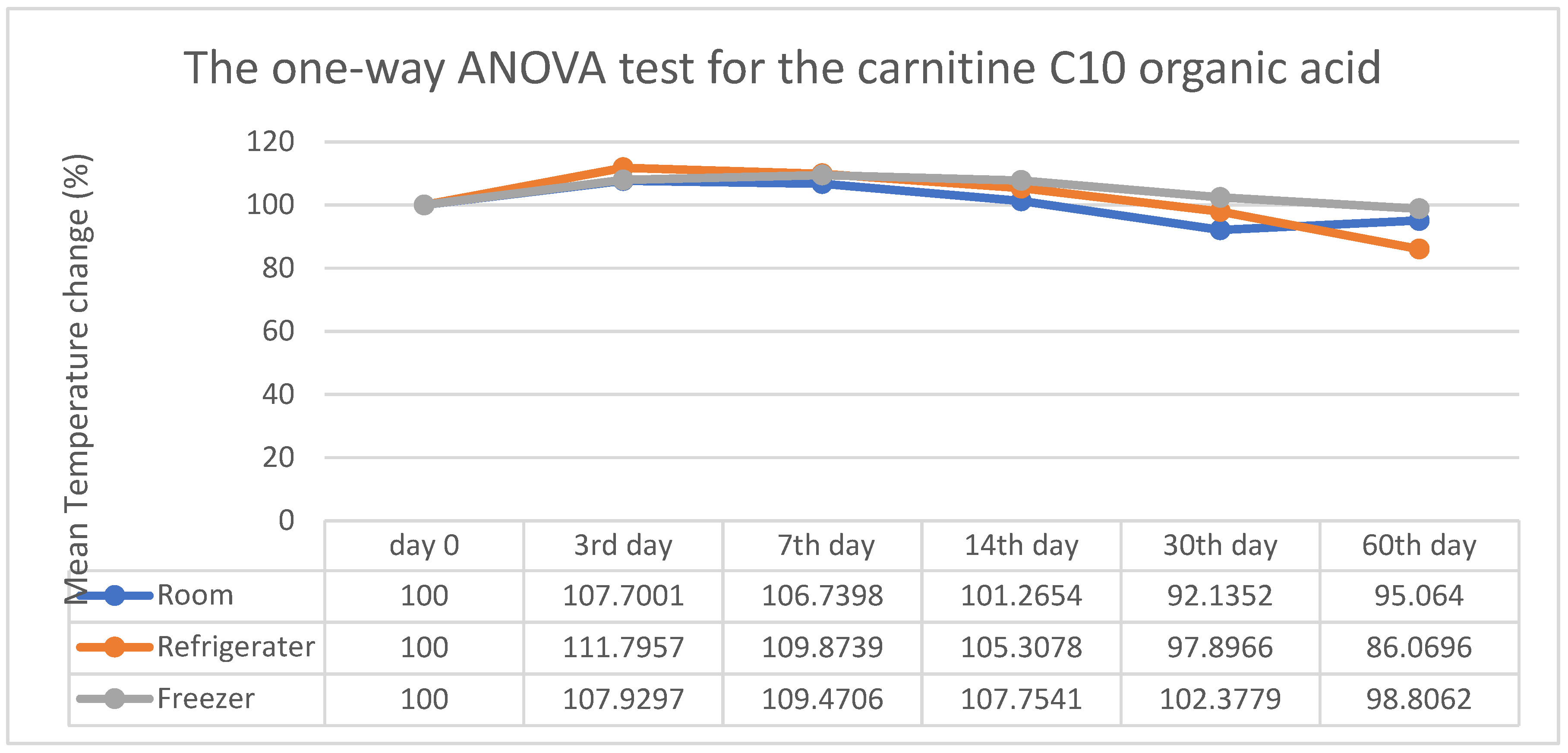
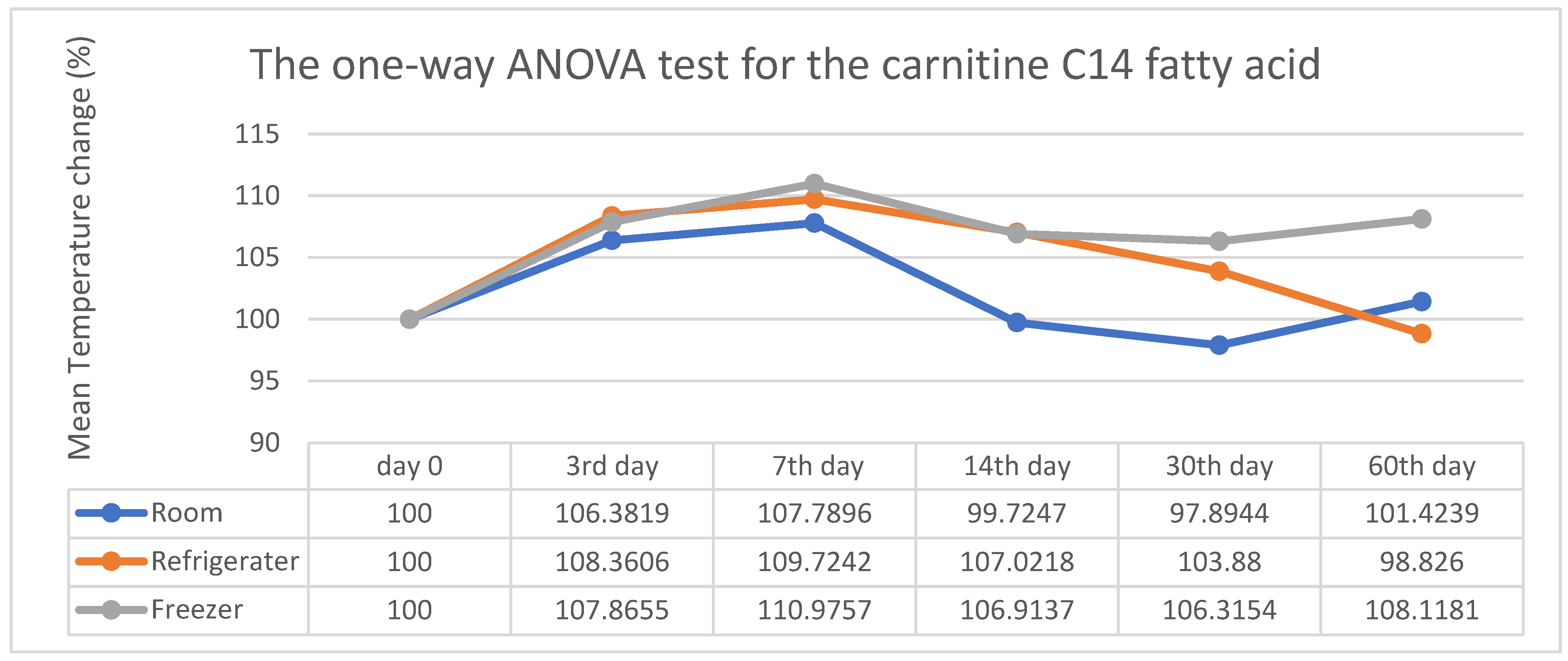
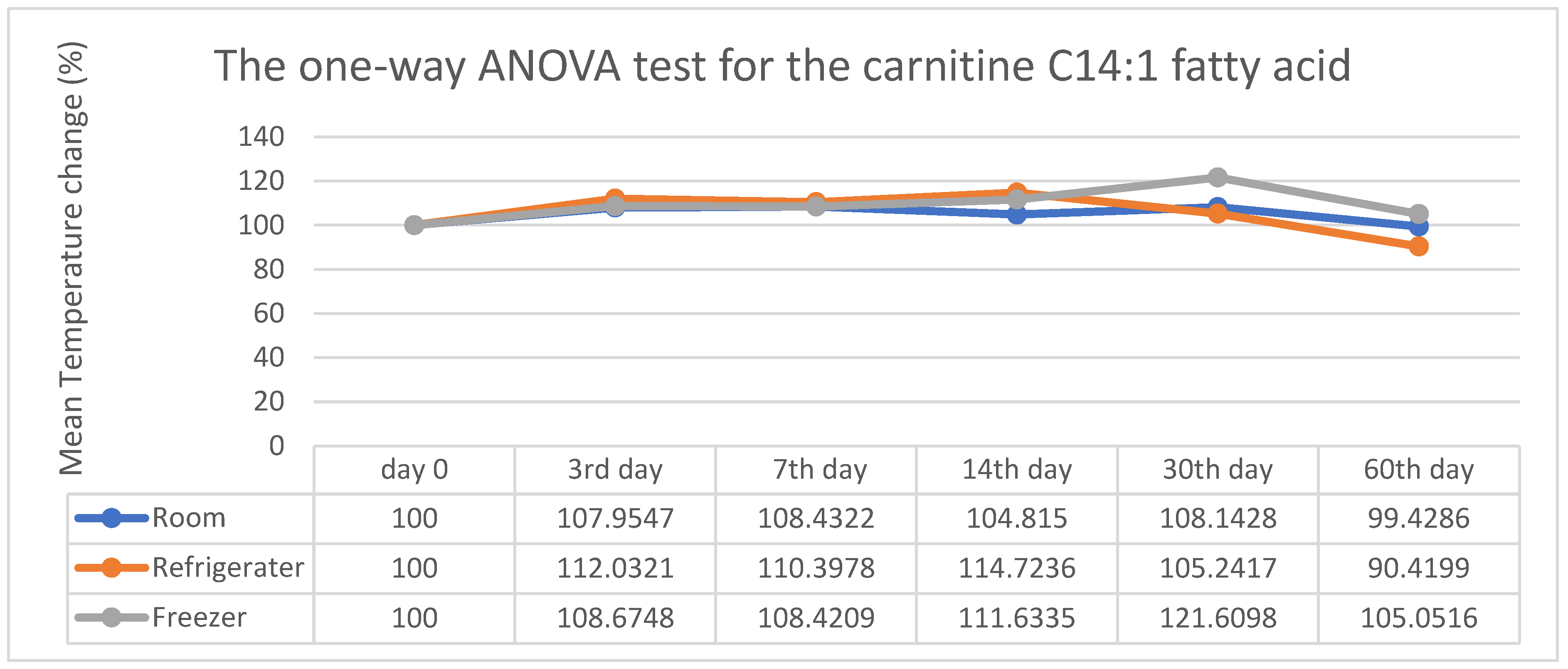
3. Results
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Afifi, A.M.; A Abdul-Jabbar, M. Saudi newborn screening. A national public health program: needs, costs, and challenges. 2007, 28, 1167–70.
- Mohamed, S.; Elsheikh, W.; Al-Aqeel, A.I.; Alhashem, A.M.; Alodaib, A.; Alahaideb, L.; Almashary, M.; Alharbi, F.; AlMalawi, H.; Ammari, A.; et al. Incidence of newborn screening disorders among 56632 infants in Central Saudi Arabia. Saudi Med J. 2020, 41, 703–708, . [CrossRef]
- Chace, D.H.; De Jesús, V.R.; Spitzer, A.R. Clinical Chemistry and Dried Blood Spots: Increasing Laboratory Utilization by Improved Understanding of Quantitative Challenges. Bioanalysis 2014, 6, 2791–2794, . [CrossRef]
- Li W, Lee MS, editors. Dried Blood Spots: Applications and Techniques [Internet]. 1st ed. Wiley; 2014 [cited 2024 Dec 24]. Available from: https://onlinelibrary.wiley.com/doi/book/10.1002/9781118890837.
- Zakaria, R.; Allen, K.J.; Koplin, J.J.; Roche, P.; Greaves, R.F. Advantages and Challenges of Dried Blood Spot Analysis by Mass Spectrometry Across the Total Testing Process. 2016, 27, 288–317.
- Sontag, M.K.; Miller, J.I.; McKasson, S.; Sheller, R.; Edelman, S.; Yusuf, C.; Singh, S.; Sarkar, D.; Bocchini, J.; Scott, J.; et al. Newborn screening timeliness quality improvement initiative: Impact of national recommendations and data repository. PLOS ONE 2020, 15, e0231050, . [CrossRef]
- Golbahar, J.; Altayab, D.; Carreon, E. Short-Term Stability of Amino acids and Acylcarnitines in the Dried Blood Spots Used to Screen Newborns for Metabolic Disorders. J. Med Screen. 2014, 21, 5–9, . [CrossRef]
- World Health Organization. Laboratory quality management system : handbook. Système de gestion de la qualité au laboratoire : manuel. Version 1.1. 2011;247.
- Klak, A.; Pauwels, S.; Vermeersch, P. Preanalytical considerations in therapeutic drug monitoring of immunosuppressants with dried blood spots. Diagnosis 2018, 6, 57–68, . [CrossRef]
- Massingale SP, Williams AM, Rollin DM. Newborn Screening Collection Guidelines [Internet]. ALABAMA DEPARTMENT OF PUBLIC HEALTH BUREAU OF CLINICAL LABORATORIES; 2019. 16 p. Available from: https://www.alabamapublichealth.gov/newbornscreening/assets/newbornscreeningbloodcollectionguidelines.pdf.
- Lehmann S, Delaby C, Vialaret J, Ducos J, Hirtz C. Current and future use of “dried blood spot” analyses in clinical chemistry. Clin Chem Lab Med. 2013 Oct;51(10):1897–909.
- Grüner N, Stambouli O, Ross RS. Dried Blood Spots - Preparing and Processing for Use in Immunoassays and in Molecular Techniques. JoVE. 2015 Mar 13;(97):52619.
- Supriya, M.; De, T.; Christopher, R. Effect of temperature on lysosomal enzyme activity during preparation and storage of dried blood spots. J. Clin. Lab. Anal. 2017, 32, . [CrossRef]
- Adam, B.; Hall, E.; Sternberg, M.; Lim, T.; Flores, S.; O'BRien, S.; Simms, D.; Li, L.; De Jesus, V.; Hannon, W. The stability of markers in dried-blood spots for recommended newborn screening disorders in the United States. Clin. Biochem. 2011, 44, 1445–1450, . [CrossRef]
- Borrajo GJC, Doña VV. How Long are Residual Newborn Screening Specimens Useful for Retesting when Stored in Suboptimal and Uncontrolled Conditions of Temperature and Humidity? J inborn errors metab screen. 2021;9:e20210010.
- Waite, K.V.; Maberly, G.F.; Eastman, C.J. Storage conditions and stability of thyrotropin and thyroid hormones on filter paper.. Clin. Chem. 1987, 33, 853–855, . [CrossRef]
- Coombes, E.J.; Gamlen, T.R.; Batstone, G.F. Effect of Temperature on the Stability of Thyroid Stimulating Hormone in Dried Blood Spots. Ann. Clin. Biochem. Int. J. Biochem. Lab. Med. 1983, 20, 252–253, . [CrossRef]
- Magalhães, P.K.R.; Miranda, C.H.; Vilar, F.C.; Schmidt, A.; Bittar, R.R.; Paixão, G.A.C.d.C.; Martinez, E.Z.; Maciel, L.M.Z. Effects of drying and storage conditions on the stability of TSH in blood spots. Arq. Bras. de Endocrinol. Metabol. 2018, 62, 201–204, . [CrossRef]
- Miller, M.J.; Cusmano-Ozog, K.; Oglesbee, D.; Young, S. Laboratory analysis of acylcarnitines, 2020 update: a technical standard of the American College of Medical Genetics and Genomics (ACMG). Anesthesia Analg. 2020, 23, 249–258, . [CrossRef]
- Fingerhut R, Ensenauer R, Röschinger W, Arnecke R, Olgemöller B, Roscher AA. Stability of acylcarnitines and free carnitine in dried blood samples: implications for retrospective diagnosis of inborn errors of metabolism and neonatal screening for carnitine transporter deficiency. Anal Chem. 2009 May 1;81(9):3571–5.
- van Rijt WJ, Schielen PCJI, Özer Y, Bijsterveld K, van der Sluijs FH, Derks TGJ, et al. Instability of Acylcarnitines in Stored Dried Blood Spots: The Impact on Retrospective Analysis of Biomarkers for Inborn Errors of Metabolism. Int J Neonatal Screen. 2020 Nov 2;6(4):83.

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



