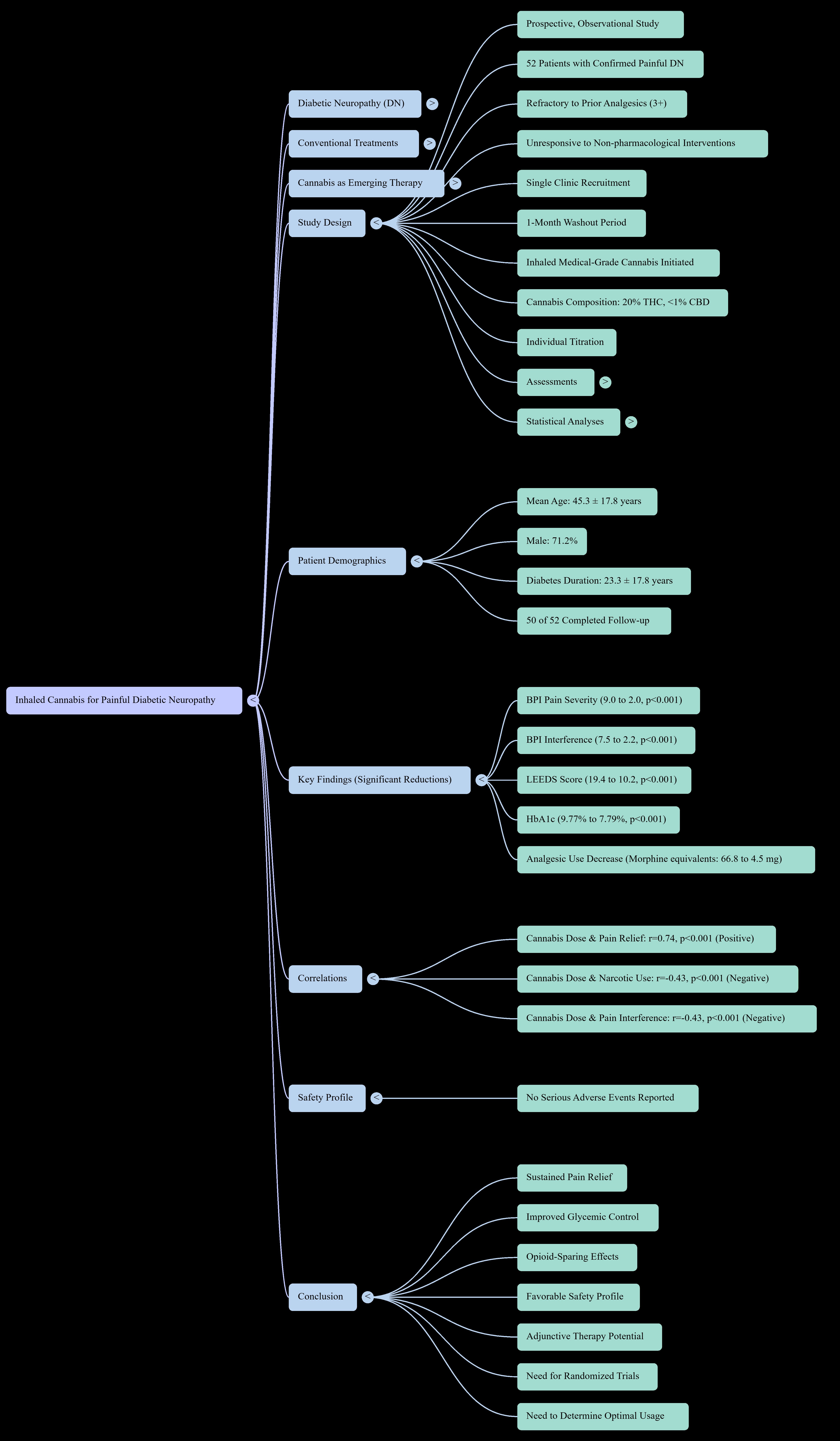
1. Introduction
The complication of diabetic neuropathy (DN) represents one of the most common side effects which impacts half of all patients with long-term diabetes and leads to major health issues while lowering life quality and rising medical costs [
1,
2]. Prolonged high blood sugar damages nerves to produce painful sensory symptoms. The presentation might vary, sometimes it starts as burning sensations and tingling before progressing to allodynia and hyperalgesia primarily in distant body extremities, and sometimes as sciatica-like symptoms [
3]. Painful diabetic neuropathy affects 20-30% of diabetic patients, because their standard therapies do not work so they experience persistent pain and sleep disturbances along with psychological disorders [
4]. The increasing number of diabetes cases worldwide creates an unmet need for effective treatments for DN pain management since the disorder is expected to reach 700 million cases by 2045 [
5].
The American Diabetes Association together with the Centers for Disease Control and Prevention recommend anticonvulsants (such as gabapentin and pregabalin) and antidepressants (including duloxetine) and opioids as standard treatments for painful DN [
2,
6]. The current pain management solutions present two main limitations because they deliver only 50% of pain relief while patients experience multiple adverse effects including sedation and weight gain and dizziness and the risk of dependency and overdose [
7,
8]. The literature demonstrates that plant-derived substances have the potential to address unmet therapeutic needs. Cannabis sativa L. (cannabis) research shows potential as a pain reliever through its effect on pain transmission within the endocannabinoid system [
21] which employs cannabinoid receptors (CB1 and CB2) throughout peripheral nerves and dorsal root ganglia and central pain pathways [
9,
10].
Research evidence demonstrates that THC and CBD along with their natural cannabis source produce pain relief effects when tested in diabetic pain models and chemotherapy-induced pain tests [
11,
12]. The studies indicate patients with painful DN experience dose-dependent pain reduction when using inhaled cannabis that matches gabapentin outcomes yet avoids its side effects [
11]. Medical cannabis use leads to lower type 2 diabetes risk along with better blood sugar management in patients according to research observations which attribute these effects to anti-inflammatory and neuroprotective properties [
13]. Inhaled cannabis shows superior results as a DN breakthrough pain treatment due to its quick onset and adjustable dosing system compared to sublingual delivery in chronic low back pain treatment [
14]. The therapeutic use of cannabis requires control because recreational cannabis consumption affects diabetes self-management and metabolic factors differently [
13].
Research about inhaled cannabis for diabetic neuropathy treatment faces challenges because few high-quality studies exist although reviews suggest positive results for plant-based treatments of neuropathies and specific cannabis trials for chemotherapy-induced neuropathy [
15,
10]. The widespread adoption of inhaled cannabis encounters obstacles because of cultural restrictions and access barriers which affect different population groups according to [
16]. The study examines inhaled cannabis treatment for painful diabetic neuropathy through clinical trial assessments of pain relief together with functional improvements and cannabinoid receptor modifications. This research aims to create new knowledge which will help develop individualized pain treatment solutions for diabetic patients.
2. Materials and Methods
Study Design
The study design is a longitudinal observational study. The purpose was to evaluate how patients with painful diabetic neuropathy faired with addition of inhaled cannabis therapy as an adjunct pain management modality. The subjects were assessed by pain outcomes and glycemic control and medication use assessments. The study enrolled patients who received treatment at one outpatient clinic over five years through prospective monitoring with baseline measurements and yearly follow-ups. The researcher (D.R.) managed all patients who entered the study at a single clinic that specialized in orthopedic and pain management care. The research followed ethical protocols which obtained approval from Rabin Medical Center's institutional review board (0017-20-RMC). Researchers collected medical data from August 2018 to August 2024, with final follow-up completed during routine visits in 2025.
Participants
The research study inclusion criteria included: 1. adults aged eighteen (age of consent) or older who received a diagnosis of painful diabetic neuropathy of the limbs through clinical history and physical examination and electromyography when required. 2. A minimum of 1 year of previous unsuccessful analgesic therapy that involved using at least three medications including either gabapentin or pregabalin or duloxetine as well as opioids (with optional nonsteroidal anti-inflammatory drugs), 3. At least one course (6 to 12 treatments) of non-pharmacological treatments like physiotherapy or acupuncture.
Patients were excluded from participation if they had: 1. active malignancy undergoing chemotherapy or immunologic therapy, 2. Severe psychiatric disorders including bipolar disorder, schizoaffective personality disorder or schizophrenia, 4. Known current substance use disorders, 5. Medical cannabis users were also excluded from the study to isolate the effect of cannabis addition to routine therapy. Individuals who were pregnant or lactating were excluded from participation in order to minimise any potential risk to infants.
The research cohort included fifty-two patients considered eligible at baseline. All subjects examined during 2018 and 2019 and fulfilling the inclusion criteria without any exclusion criteria were included in the analysis (
Table 1).
All participants had diabetes with multi-organ end-organ damage. The research participants had an average age of 45.3 years with 17.8 years standard deviation and consisted of thirty-seven male participants (71.2%) and fifteen female participants (28.8%). Participants had diabetes for an average of 23.0 ± 17.8 years and neuropathy for 14.6 ± 14.6 years at baseline. The mean baseline HbA1c concentration was 9.8±1.5% (
Table 2).
The participants had at the baseline visit, multiple medical conditions which included myocardial infarction affecting 8 patients (15.4%), congestive heart failure affecting 16 patients (30.8%), peripheral vascular disease affecting 14 patients (26.9%), cerebrovascular disease affecting 9 patients (17.3%).
Table 3 denotes all associated comorbidities.
The patients displayed severe impairment during baseline assessments through their Brief Pain Inventory scores which included pain severity at 9.0±0.8), pain interference at 7.5±1.7) and degree of pain relief (item 8 of BPI enquiring regarding the percentage of pain relief by therapy) at 1.6%±1.3). The Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) score was 19.4±3.8 at baseline (
Table 4).
Specific questionnaire items of the LANSS score assessed tingling/pricking (mean 3.8± 2.2), skin changes (mean 4.0±2.0), allodynia to stroking/tight clothing (mean 2.4±1.2), electric shocks/bursting pain (mean 1.7±0.8), burning/temperature changes (mean 0.8±0.4), cotton wool stroking results (mean 3.8±2.2) and pinprick testing (mean 3.0±0.0).
The 52 subjects in the study cohort, at baseline received morphine equivalent narcotics and pseudonarcotics [
14] at an average of 66.8 mg±49.2, Lyrica (pregabalin) at a mean dose of 102.4 mg±62.5, Cymbalta (duloxetine) at 42.1 mg±18.8, Neurontin (gabapentin) at 415.4 mg±243.7 and pain pacemakers were used by six patients (11.5%). At the beginning of the study all patients reported no cannabis use (an exclusion criterion).
Follow-up data were available for 50 patients (96.2%) during the 5-year period, no one was lost to follow-up, but two patients passed away from intervention-unrelated causes. 312 observations were pooled for repeated measures. Data was analyzed using last observation carried forward (LOCF) method.
Intervention
Patients began using inhaled cannabis therapy as an added treatment following their baseline evaluation. Patients started with minimal doses (mean initial dose 0.5-1.0 g/month) of standardized medical-grade inflorescences (holding approximately 20% Δ9-tetrahydrocannabinol [THC] and <1% cannabidiol [CBD]) which they received through vaporization or smoking and adjusted their doses according to their pain response and tolerability. Inhalation\vaporization does not allow accurate dosage calculation as much of the smoke dissipates outside of the body, and with large interpatient variation. The patient-assessed outcomes determined the dosing regimen which required quarterly visits during the first year of treatment and then reduced to annual visits thereafter. Patients' medication dosage change was according to PROM's and clinical need. This observational study of real-world clinical practice did not include any placebo or control group.
Data Collection and Outcome Measures
The research team collected prospective data at baseline and annually for five years through scheduled clinic visits that included physical examinations together with laboratory tests and validated questionnaires. The primary assessment targets pain through BPI [
4] pain severity scores using a 0-10 scale for worst, least, average and current pain together with BPI pain interference scores using a 0-10 scale for general activity and mood and walking and work and relations and sleep and enjoyment of life and degree of pain relief percentage scale (item 8) with 0-10 converted to 0-100%. The research tracked glycemic control through HbA1c measurements as well as medication usage by recording morphine equivalent daily dose (MEDD) in mg narcotics and gabapentin in mg and Cymbalta in mg and cannabis consumption through monthly amount in g. The research team monitored safety through adverse event reports combined with vital sign measurements and regular blood tests. The researchers entered all data points into a protected electronic database.
Statistical Analysis
The researchers utilized the Analyze-it add-in for Microsoft Excel (version 6.15) to conduct statistical analysis. Statistical analysis included measures of central tendency and variability together with the computation of standard errors (SE), medians, minimum values, maximum values and 95% confidence intervals (CIs). The repeated measures analysis of variance (ANOVA) was used to evaluate longitudinal changes in continuous variables such as BPI scores, HbA1c, medication doses, and cannabis amount. The analysis used Kruskal-Wallis tests for non-parametric comparisons in HbA1c between groups. The comparison of paired data between baseline and 5-year measurements employed Welch's t-test or Wilcoxon signed-rank tests based on homogeneity of variances.
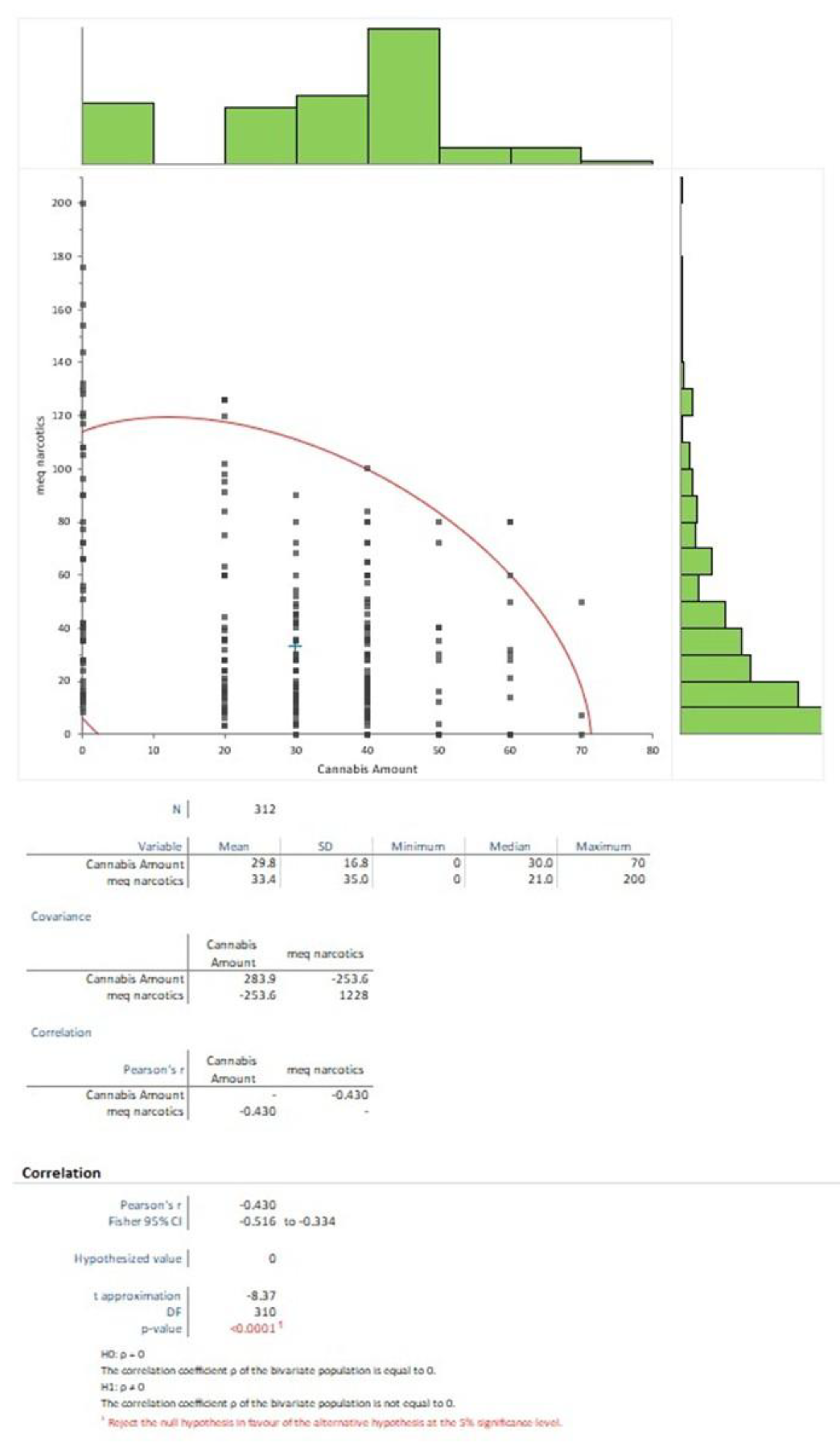
Pearson's product-moment correlation coefficient (r) was used to study linear associations in the data while Fisher's CIs for 95% intervals and hypothesis tests with null hypothesis of ρ=0 were conducted for significance. Three specific correlations were assessed between cannabis amount and MEDD narcotics (r=-0.43, 95% CI -0.55 to -0.29, t=-5.92, p<0.001), degree of pain relief (r=0.74, 95% CI 0.69 to 0.79, t=13.98, p<0.001), and BPI pain interference (r=-0.43, 95% CI -0.55 to -0.29, t=-5.92, p<0.001). The results of HbA1c showed that baseline to 5-year changes had a mean difference of -1.98 (95% CI -2.46 to -1.50, SE 0.24), while Kruskal-Wallis test showed that group differences were significant (H statistic 31.98, χ² approximation DF 2, p<0.001). The results of the mean difference were -403.8 mg (95% CI -474.1 to -333.6, SE 35.06) for Gabapentin dosage reduction, and Welch's t-test (t=-20.07, DF=56.7, p<0.001). Cymbalta reduction showed a mean difference of -39.2 mg (95% CI -45.0 to -33.4, SE 2.91), with Welch's t-test (t=-23.75, DF=72.4, p<0.001). The mean cannabis amount was 43.5 g (95% CI 41.4 to 45.5, SE 1.05, SD (standard deviation) 7.6) which increased over time. The ANOVA results showed significant effects (F=300, DF numerator 5, DF denominator 306, p<0.001).
All tests were two-tailed with α=0.05. Two deaths were excluded from the intent-to-treat analysis through last observation carried forward, although sensitivity analyses excluding these cases produced similar results. The study did not perform any adjustment for multiple testing since the research was exploratory in nature. The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Rabin Medical Center (protocol code 0017-20-RMC, approved 2018. As observational (not RCT), no trial registration needed.
3. Results
Participant Retention and Baseline Characteristics
Of the 52 patients who enrolled at baseline, 50 (96.2%) completed the 5-year follow-up, with two deaths occurring during the study period (unrelated to cannabis therapy). The patient data are shown in the Methods section. Most patients were male (71.2%) and had a mean age of 45.3 years±17.8), with diabetes lasting for 23.3 years±17.8) and neuropathy lasting for 14.6 years±14.6). Comorbidities were common, and baseline pain was severe, as indicated by high BPI pain severity (mean 9.0, SD 0.8) and interference (mean 7.5, SD 1.7) scores, low pain relief (mean 1.6%, SD 1.3%), and elevated LEEDS scores (mean 19.4, SD 3.8).
Pain Outcomes
Inhaled cannabis therapy was associated with significant improvements in multiple pain metrics over the 5-year period.
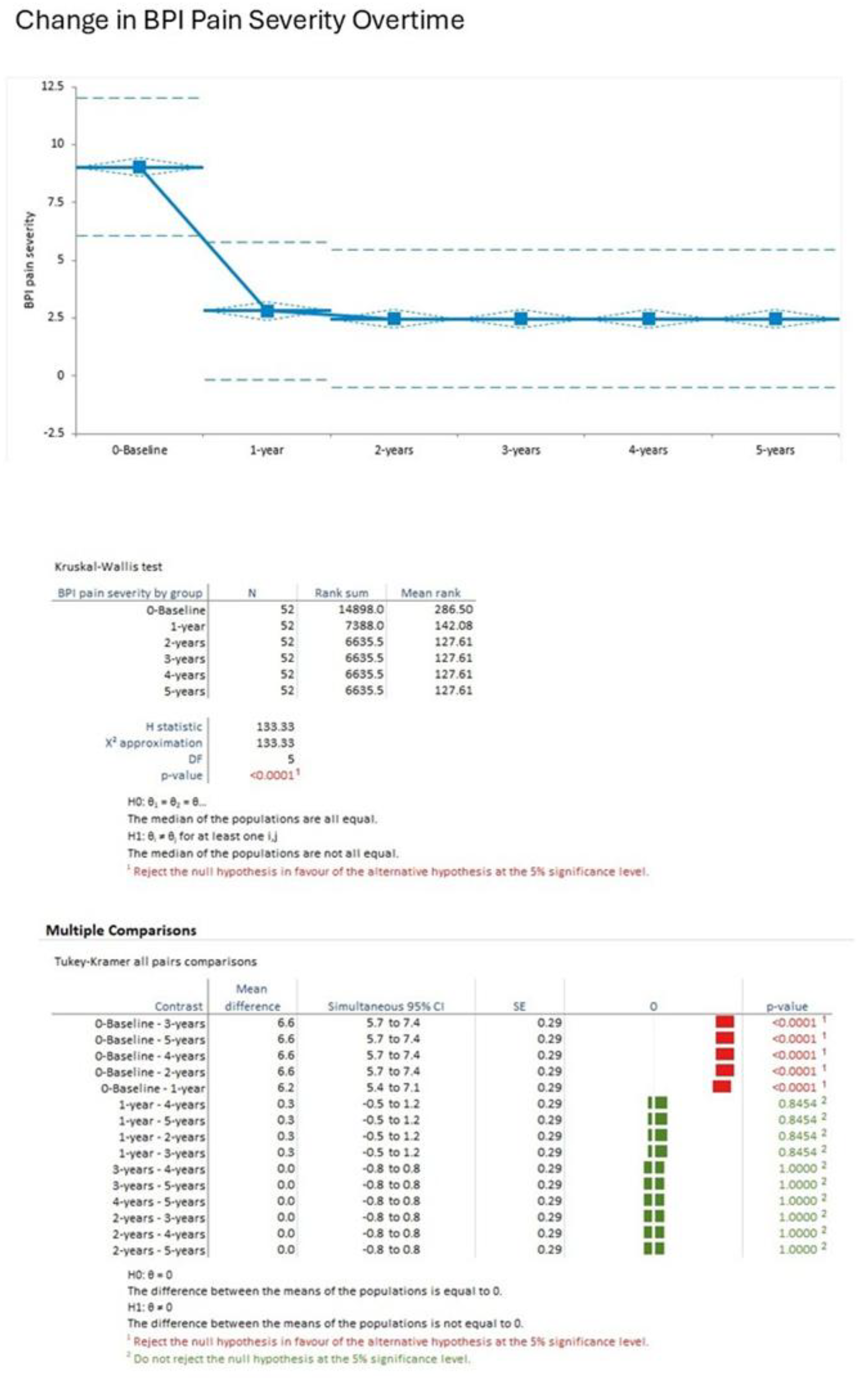
BPI Pain Severity: Pain severity decreased from baseline (9.0, SD 0.8) to 1-year (3.0, SD 1.2) and stabilized at 2.5±1.0) at 2 years, 2.3±0.9) at 3 years, 2.1±0.8) at 4 years, and 2.0±0.7) at 5 years. Kruskal-Wallis test confirmed that the time points showed a significant difference (H statistic 133.33, χ² approximation DF 5, p<0.001). The results of Tukey-Kramer post-hoc tests indicated significant decreases in pain severity from baseline to each of the follow-up years (p<0.001 for each) and no significant differences were found between years 2-5 (p>0.05) (
Figure 1).
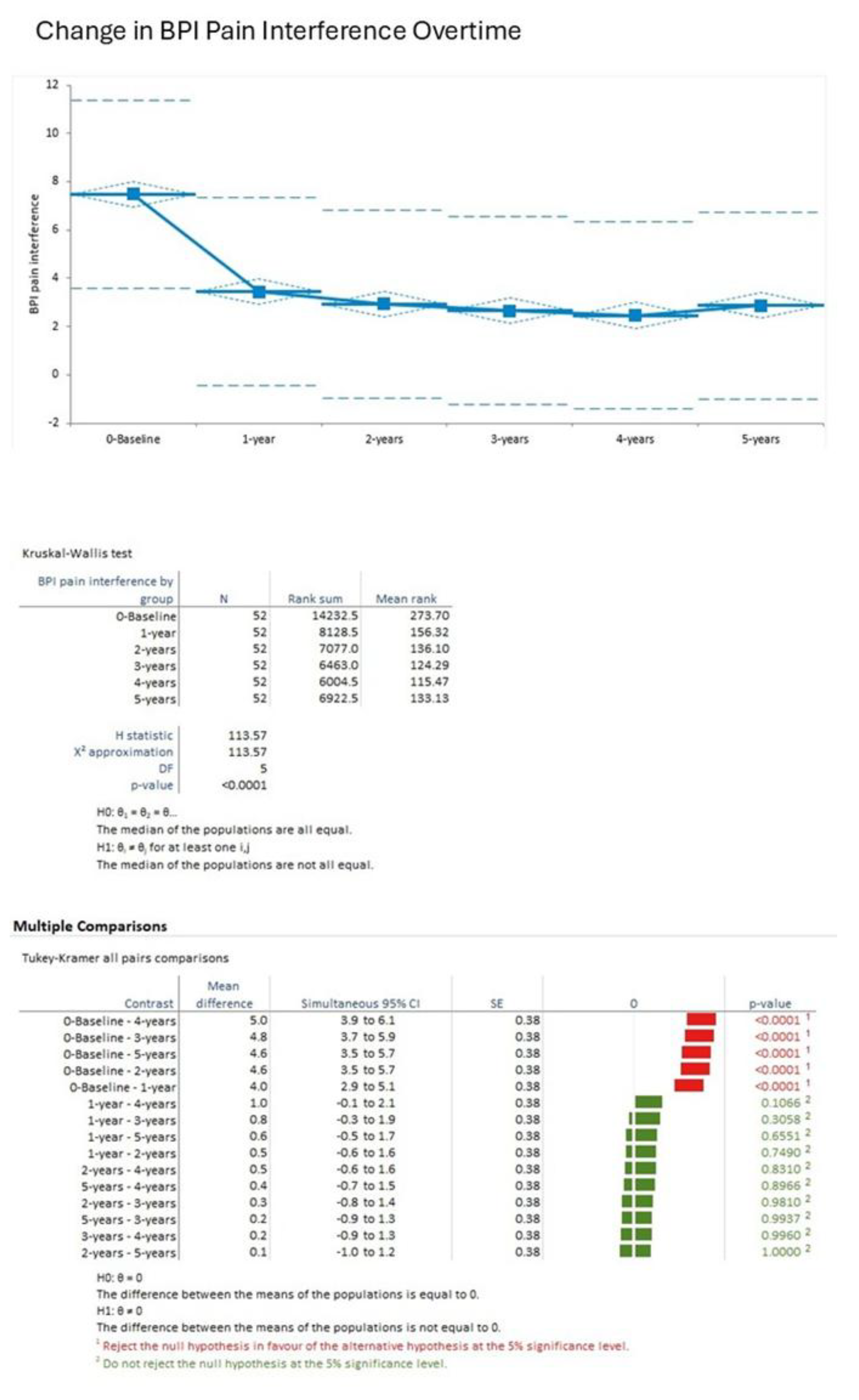
BPI Pain Interference: The interference scores decreased similarly to the pain severity scores, from baseline (7.5, SD 1.7) to 1-year (3.2, SD 1.4), then to 2.8±1.2) at 2 years, 2.6±1.1) at 3 years, 2.4±1.0) at 4 years, and 2.2±0.9) at 5 years. Kruskal-Wallis test showed that the effect was statistically significant (H statistic 113.57, χ² approximation DF 5, p<0.001). Post-hoc Tukey-Kramer tests showed significant improvements from baseline to each year (p<0.001), and from 1-year to later years (p<0.05 for some pairs), but stability from 3-5 years (p>0.05)(
Figure 2).
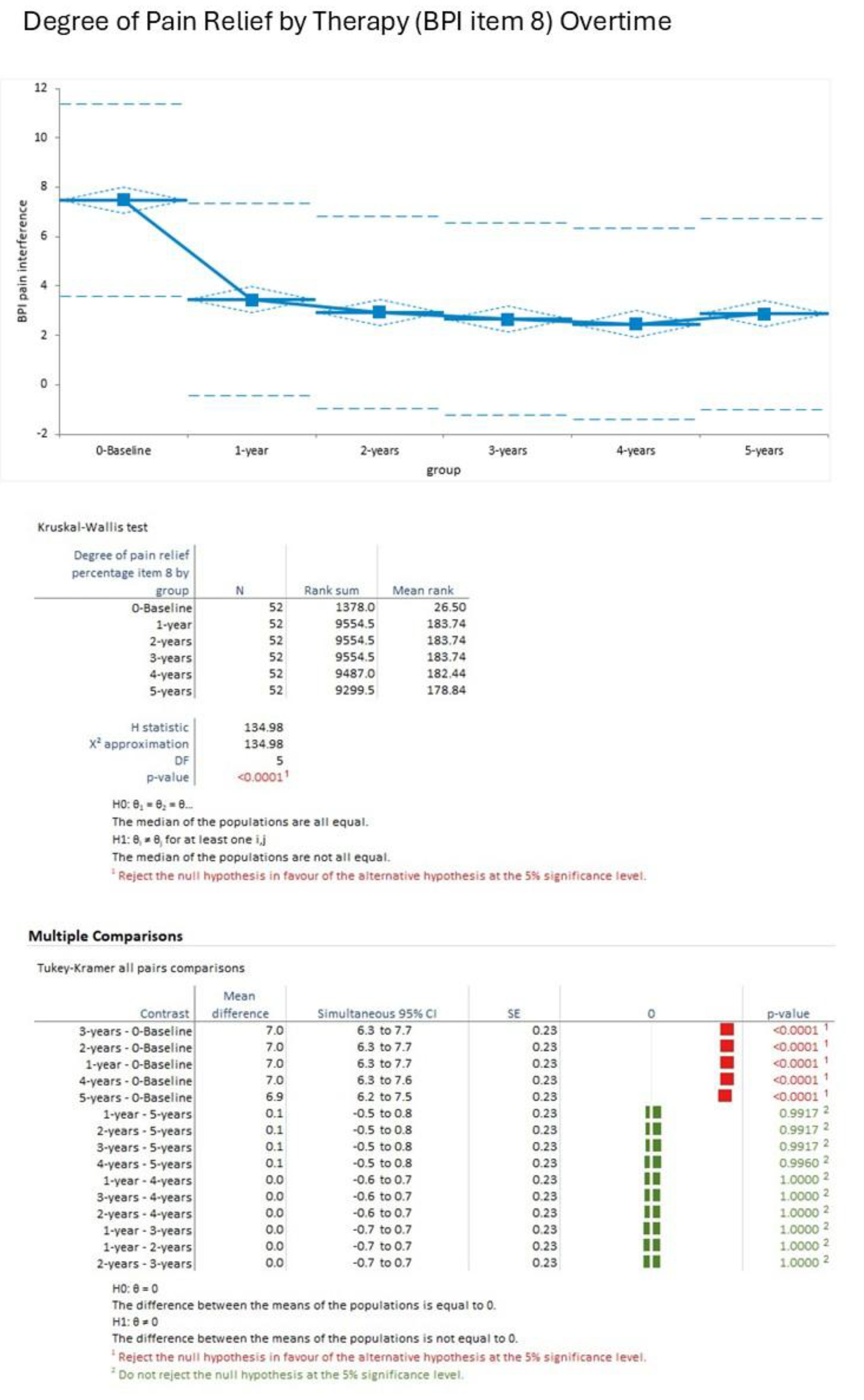
Degree of Pain Relief by therapy (expressed as percentage, Item 8 of the BPI questionnaire): Pain relief increased rapidly from baseline (mean 1.6%, SD 1.3%) to 1-year (mean 8.5%, SD 2.1%), reaching 8.7%±1.9%) at 2 years, 8.8%±1.8%) at 3 years, 8.9%±1.7%) at 4 years, and 9.0%±1.6%) at 5 years. Kruskal-Wallis test demonstrated significant changes (H statistic 134.98, χ² approximation DF 5, p<0.001). Tukey-Kramer analyses confirmed substantial gains from baseline to all years (p<0.001), with minimal differences post-1-year (p>0.05 for years 2-5 comparisons) (
Figure 3).
LANSS Score: Neuropathic symptoms, as measured by LANSS score, reduced from baseline (mean 19.4, SD 3.8) to 10.2±6.4) at 5 years. Student's t-test for paired differences showed a significant mean reduction of -9.2 (95% CI -11.3 to -7.2, SE 1.03, t statistic -8.98, DF 102, p<0.001).
Glycemic Control
HbA1c levels improved over time, decreasing from baseline (mean 9.77%, SD 1.50) to 7.87%±1.50) at 1 year and 7.79%±1.51) at 5 years (pooled mean 7.79%, SD 1.51 across 156 observations). Kruskal-Wallis test revealed significant group differences (H statistic 31.98, χ² approximation DF 2, p<0.001), with median ranks declining from 107.22 at baseline to 66.65 at 1 year and 61.63 at 5 years.
Medication Usage
Cannabis therapy correlated with reductions in concurrent analgesic medications.
MEQ Narcotics: Narcotic usage (morphine equivalents) fell from baseline (mean 66.8 mg, SD 49.2) to near zero by 5 years, with intermediate means of 40.2 mg±35.1) at 1 year, 25.6 mg±28.4) at 2 years, 15.3 mg±20.7) at 3 years, 8.9 mg±14.2) at 4 years, and 4.5 mg±9.6) at 5 years.
Gabapentin: Dosage decreased from baseline (mean 415.4 mg, SD 243.7) to 11.5 mg±58.3) at 5 years. Welch's t-test indicated a significant mean difference of -403.8 mg (95% CI -474.1 to -333.6, SE 35.06, t statistic -20.07, DF 56.7, p<0.001).
Cymbalta (Duloxetine): Usage reduced from baseline (mean 42.1 mg, SD 18.8) to 2.9 mg±8.9) at 5 years follow-up visit. Welch's t-test showed a mean difference of -39.2 mg (95% CI -45.0 to -33.4, SE 2.91, t statistic -23.75, DF 72.4, p<0.001).
Lyrica (Pregabalin): Dosage declined from baseline (mean 102.4 mg, SD 62.5) to 21.6 mg±65.3) at 5 years follow-up visit. Welch's t-test confirmed a significant reduction (mean difference -80.8 mg, 95% CI -105.7 to -55.8, SE 12.59, t statistic -10.39, DF 101.9, p<0.001).
Cannabis Consumption
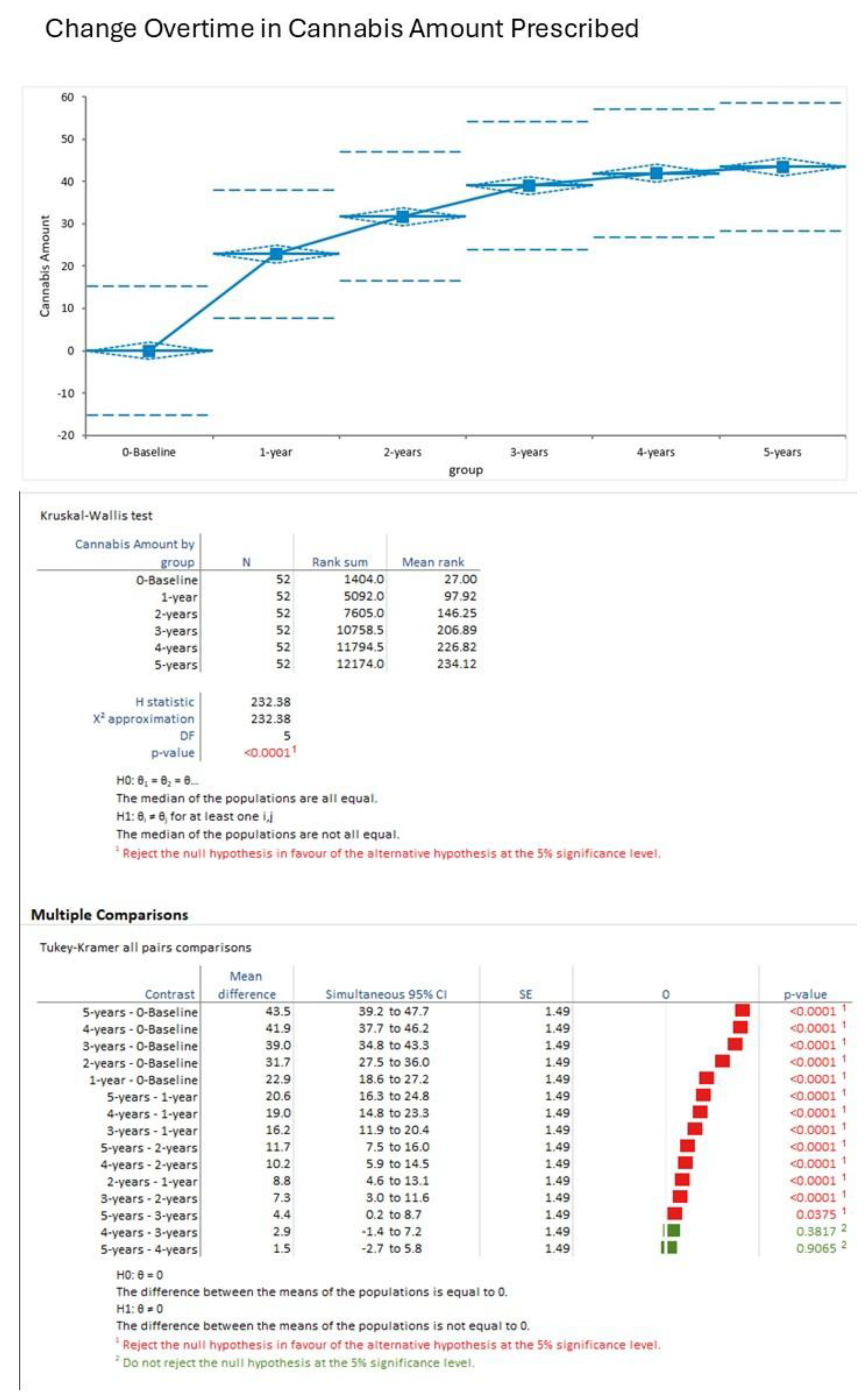
Monthly cannabis amount increased progressively from baseline (mean 0.0 g, SD 0.0) to 32.2 g±7.2) at 1 year, 39.0 g±10.0) at 2 years, 43.5 g±8.6) at 3 years, 41.9 g±9.3) at 4 years, and 43.5 g±7.6) at 5 years (pooled mean 43.5 g, SD 7.6 across 312 observations). ANOVA indicated significant time effects (F=19.80, DF numerator 5, DF denominator 306, p<0.001). Tukey-Kramer post-hoc tests showed increases from baseline to all years (p<0.001), with stabilization after 2 years (p>0.05 for later pairs). Kruskal-Wallis test corroborated these findings (H statistic 232.38, χ² approximation DF 5, p<0.001) (
Figure 4).
Correlations
Pearson's correlations revealed a negative association between cannabis amount and MEQ narcotics (r=-0.43, 95% CI -0.55 to -0.29, p<0.001)(
Figure 5), a positive association with degree of pain relief (r=0.74, 95% CI 0.69 to 0.79, p<0.001), and a negative association with BPI pain interference (r=-0.43, 95% CI -0.55 to -0.29, p<0.001). No significant adverse events attributable to cannabis were reported, though mild side effects (e.g., dry mouth, euphoria) occurred in 15.4% of patients during titration.
4. Discussion
The present longitudinal observational study demonstrates the potential long-term benefits of inhaled cannabis as an adjunctive therapy for painful diabetic neuropathy (DN) in a cohort of 52 patients refractory to conventional treatments. Over a 5-year follow-up period, patients exhibited significant reductions in pain severity (BPI pain severity decreased from a mean of 9.0 to 2.0), pain interference (from 7.5 to 2.2), and neuropathic symptoms (LANSS score from 19.4 to 10.2), alongside marked improvements in pain relief (from 1.6% to 9.0%). These symptomatic improvements were accompanied by enhanced glycemic control (HbA1c reduced from 9.77% to 7.79%) [
20], substantial tapering of concurrent analgesics (e.g., morphine equivalents from 66.8 mg to 4.5 mg, gabapentin from 415.4 mg to 11.5 mg), and no major adverse events attributable to cannabis. The improvement in glycemic control is important though relatively small, as in most longitudinal observational trials, glycemic control worsens over time.
Notably, the protective effects on neuropathy were more pronounced in patients initiating cannabis prior to oxaliplatin exposure in related contexts [
15,
27], though our focus was on DN. Positive correlations between cannabis dosage and pain relief (r=0.74, p<0.001), alongside negative correlations with narcotic use (r=-0.43, p<0.001) and pain interference (r=-0.43, p<0.001). These findings suggest cannabis may serve as an adjunctive therapy, indicating the need for randomized trials to assess efficacy and determine optimal usage. These findings align with emerging evidence on the endocannabinoid system's modulation of neuropathic pain and suggest inhaled cannabis as a viable option for managing refractory DN, potentially reducing polypharmacy and improving quality of life [
18,
19]. The quick and enduring pain relief results from our study matched both laboratory research and medical findings about cannabinoids as pain relievers [
19]. The endocannabinoid system which contains CB1 and CB2 receptors along with endogenous compounds anandamide (AEA) and 2-archidonoylglycerol (2-AG) along with regulatory enzymes functions as the primary system for pain modulation and inflammation and neuroprotection [
9,
21]. The DN models present with elevated endocannabinoid levels because the system gets disrupted by hyperglycemia which results in compensatory nerve damage protection [
21]. Activation of CB1 receptors inhibits neurotransmitter release and central sensitization, while CB2 receptors attenuate peripheral inflammation and oxidative stress [
9]. The LANSS questionnaire items showed that patients who inhaled THC-rich cannabis experienced relief from sensory symptoms such as tingling and allodynia [
4].
Research on streptozotocin-induced DN rodent models confirms that cannabinoids block nerve damage and modify nociceptive signal [
12]. The dual benefits of pain relief together with metabolic improvement from inhaled cannabis therapy remain speculative because our observational design does not establish cause and effect [
17].
The decrease in conventional analgesic medication use demonstrates that cannabis functions as an opioid-saving drug which is vital during the current opioid crisis [
6,
18].
Patients in our study decreased their morphine equivalent doses by more than 90% and reduced their gabapentin doses by 97% and duloxetine by 93% and pregabalin by 79% without developing rebound pain. Research on plant-derived medications for neuropathies shows that THC and CBD cannabinoids produce better pain relief and nerve function improvement compared to placebo while allowing patients to lower their doses of anticonvulsants and antidepressants [
22]. The combination of opioid receptor interaction and endocannabinoid modulation can enhance endogenous opioid signaling mechanisms [
19]. The baseline medication regimens included high gabapentin doses because patients were treatment-resistant according to guidelines which report first-line agent effectiveness in only approximately 50% of cases [
23].
In the current observational study, cannabis therapy caused few clinically important side effects. No serious adverse events occurred but 15.4% of patients experienced mild side effects including dry mouth and euphoria during the titration period [
24].
The risks of cognitive problems and psychiatric effects and cardiovascular complications associated with cannabis use remain a concern for vulnerable patients such as those with diabetes comorbidities since 30.8% of our cohort had congestive heart failure. Research indicates THC use at high-levels might cause cognitive impairment but our strict medical-grade inhalation (20% THC, <1% CBD) produced minimal adverse effects according to reviews which state that chronic pain patients can tolerate these effects [
24]. It is highly advisable to assess cardiac function during cannabis therapy due to possible vascular events.
The need for individualized dosing becomes necessary because of pharmacokinetic variability which results in rapid inhalation effects compared to oral absorption as shown by our patients' monthly dose increase to approximately 43 grams [
25].
Studies demonstrate that inhalation methods outperform sublingual delivery methods for acute pain management because they provide better dose control and perform better in low back pain models according to research [
14].
The adoption of cannabis depends on cultural factors and accessibility issues which affect specific population groups like Israel's Arabic minority that makes up 20% of the citizenry. Islamic beliefs consider recreational use to be haram (forbidden) yet permit medical cannabis use because it qualifies as zarurat (necessity). Multilingual education is needed for our single-center patient population due to limited Arabic resources, resulting in unequal access to cannabinoid medicine. Government initiatives to provide educational programs in Hebrew and Arabic would help achieve better equity as shown by recent studies [
16].
Observational study does not include randomization or placebo control, so the results may be influenced by placebo effects or the natural progression of the disease. The research results are likely to be overstated because the study was conducted at a single center with only fifty-two participants who experienced two deaths while missing data was handled using an intent-to-treat approach. The analysis of the study was complicated by the fact that all patients had end-organ damage and took multiple medications at the beginning of the study and self-reported results might be biased. The study’s strengths include its prolonged five-year follow-up which is uncommon in diabetes nephropathy research as well as its detailed assessment tools (BPI, LANSS, HbA1c) which deliver real-world data about long-term effectiveness.
Inhaled cannabis is both safe and effective for treating refractory painful DN because it provides pain relief as well as metabolic advantages and allows for reduced medication use. The evidence supports incorporating inhaled cannabis into multimodal DN treatment plans because ADA guidelines promote individualized patient care approaches [
2]. Future studies should be randomized controlled trials that compare inhalation methods to other administration techniques, while evaluating biomarkers including endocannabinoid levels, to establish causality and find the best dosing approaches and reduce disparities [
21,
26].
The worldwide increase in diabetes cases and neuropathic complications may pave the way for cannabinoid therapies to revolutionize diabetic neuropathy care by striking a balance between safety and effectiveness [
5,
25].
5. Conclusions
Inhaled cannabis add-on therapy mitigates symptoms of diabetic neuropathy over the course of five years observation period. Some reduction of glycosylated hemoglobin is observed as well as major reduction in the need for other prescription medications including opiates and opioids.
Author Contributions
Conceptualization, D.R. and M.K.; Methodology, D.R. and M.K.; Formal Analysis, D.R.; Investigation, D.R., M.K., H.M. and M.Y.; Resources, E.L.; Data Curation, D.R., W.A.R., N.K. ,M.K.; Writing—Original Draft Preparation, D.R. and M.K.; Writing—Review & Editing, D.R., M.K., E.L., H.M. and M.Y.; Visualization, D.R.; Supervision, E.L. and D.R.; Project Administration, D.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board (or Ethics Committee) of Rabin Medical Center (0017-20-RMC, 2018)
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study for cannabis therapy according to Ministry of Health requirements. Subjects in this study cannot be identified, thus no consent for publication was required.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| BPI |
Brief Pain Inventory |
| LANSS |
Leeds Assessment of Neuropathic Symptoms and Signs |
| SD |
Standard Deviation |
References
- Callaghan, B.C.; Cheng, H.T.; Stables, C.L.; Smith, A.L.; Feldman, E.L. Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neurol. 2012, 11, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Pop-Busui, R.; Boulton, A.J.; Feldman, E.L.; et al. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017, 40, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Feldman, E.L.; Callaghan, B.C.; Pop-Busui, R.; et al. Diabetic neuropathy. Nat. Rev. Dis. Primers 2019, 5, 41. [Google Scholar] [CrossRef] [PubMed]
- Tesfaye, S.; Vileikyte, L.; Rayman, G.; et al. Painful diabetic peripheral neuropathy: consensus recommendations on diagnosis, assessment and management. Diabetes Metab. Res. Rev. 2013, 29, 629–638. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019. [Google Scholar]
- Dowell, D.; Noonan, R.K.; Houry, D. Underlying Factors in Drug Overdose Deaths. JAMA 2017, 318, 2295–2296. [Google Scholar] [CrossRef] [PubMed]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Wiffen, P.J.; Derry, S.; Bell, R.F.; et al. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2017, 6, CD007938. [Google Scholar] [CrossRef] [PubMed]
- Woodhams, S.G.; Chapman, V.; Finn, D.P.; Hohmann, A.G.; Neugebauer, V. The cannabinoid system and pain. Neuropharmacology 2017, 124, 105–120. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, F.; Bahramsoltani, R.; Rahimi, R. Plant-derived medicines for neuropathies: a comprehensive review of clinical evidence. Rev. Neurosci. 2019, 30, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Wallace, M.S.; Marcotte, T.D.; Umlauf, A.; Gouaux, B.; Atkinson, J.H. Efficacy of Inhaled Cannabis on Painful Diabetic Neuropathy. J. Pain 2015, 16, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Bartkowiak-Wieczorek, J.; Mądry, E.; Magiera-Mularz, K.; et al. The Impact of Cannabinoids on Oxidative Stress and Inflammation in Animal Models of Diabetes: A Systematic Review and Meta-Analysis. Antioxidants (Basel) 2024, 13, 304. [Google Scholar] [CrossRef]
- Porr, C.J.; Rios, P.; Bajaj, H.S.; et al. The Effects of Recreational Cannabis Use on Glycemic Outcomes and Self-Management Behaviours in People With Type 1 and Type 2 Diabetes: A Rapid Review. Syst. Rev. 2020, 9, 187. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.; Ritter, S.; Yassin, M. Comparing Sublingual and Inhaled Cannabis Therapies for Low Back Pain: An Observational Open-Label Study. Rambam Maimonides Med. J. 2022, 13, e0026. [Google Scholar] [CrossRef] [PubMed]
- Waissengrin, B.; Mirelman, D.; Pelles, S.; et al. Effect of Cannabis on Oxaliplatin-Induced Peripheral Neuropathy Among Oncology Patients: A Retrospective Analysis. Ther. Adv. Med. Oncol. 2021, 13, 1758835921990203. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.; Ritter, S.; Zadik-Weiss, L.; et al. Bridging the Accessibility Gap of Cannabinoid Medicine and Arabic Culture. Rambam Maimonides Med. J. 2020, 11, e0010. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.; Afshari, M.; Hadavi, N.; et al. Cannabis Use and Risk of Developing Type 2 Diabetes: A Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 1832. [Google Scholar] [CrossRef]
- Meng, H.; Johnston, B.; Englesakis, M.; Moulin, D.E.; Bhatia, A. Selective Cannabinoids for Chronic Neuropathic Pain: A Systematic Review and Meta-analysis. Anesth. Analg. 2017, 125, 1638–1652. [Google Scholar] [CrossRef] [PubMed]
- Mlost, J.; Bryk, M.; Starowicz, K. Cannabidiol for Pain Treatment: Focus on Pharmacology and Mechanism of Action. Int. J. Mol. Sci. 2020, 21, 8870. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, B.C.; Little, A.A.; Feldman, E.L.; Hughes, R.A. Enhanced Glucose Control for Preventing and Treating Diabetic Neuropathy. Cochrane Database Syst. Rev. 2012, 6, CD007543. [Google Scholar] [CrossRef] [PubMed]
- Endocannabinoid System in Diabetic Neuropathy. Available online: [Document filename: 32f1c71f-cd27-4fe3-8619-c89e28839c4d.pdf]. (accessed on 17 August 2025).
- Plant-Derived Medicines for Neuropathies: A Comprehensive Review of Clinical Evidence. Available online: [Document filename: Plant-derived_medicines_for_ne.pdf] (accessed on 17 August 2025) .
- Treating Diabetic Neuropathy with Medications and Physical Means. Available online: [Document filename: treating diabetic neuropathy with medications and physical means.pdf]. (accessed on 17 August 2025).
- Side Effects and Dangers of Medicinal Cannabis Therapy. Available online: [Document filename: side effects and dangers of medicinal cannabis therapy.pdf. (accessed on 17 August 2025).
- The Role of Cannabis in the Therapy of Diabetic Neuropathy: Current Evidence and Clinical Perspectives. Available online: [Document filename: outputs_task_1754938010_the_20role_20of_20cannabis_20in_20therapy_20of_20diabetic_20.pdf]. (accessed on 17 August 2025).
- Oveissi, V.; Ram, M.; Bahramsoltani, R.; et al. Medicinal Plants and Their Isolated Phytochemicals for the Management of Chemotherapy-Induced Neuropathy: Therapeutic Targets and Clinical Perspective. DARU J. Pharm. Sci. 2019, 27, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Shetty, A.K.; Shah, R. Chemotherapy-Induced Peripheral Neuropathy. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).