Submitted:
04 August 2025
Posted:
05 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Presentation
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NBS | Newborn screening |
| SCID | Severe combined immunodeficiency |
| PNP | Purine nucleoside phosphorylase |
| Guo | Guanosine |
| dGuo | Deoxyguanosine |
| Ino | Inosine |
| dIno | Deoxyinosine |
| dGTP | Deoxyguanosine triphosphate |
| HSCT | Hematopoietic stem cell transplantation |
| TREC | T-cell receptor excision circles |
| KREC | kappa-deleting recombination excision circles |
| DBS | Dried blood spot |
| FIA | Flow injection analysis |
| MS/MS | Tandem mass spectrometry |
| LC | Liquid chromatography |
| BCG | Bacillus Calmette-Guérin |
| WES | Whole exome sequencing |
| Allo | Allogenic |
| Ado | Adenosine |
| dAdo | 2-deoxyadenosine |
| ADA | Adenosine deaminase |
References
- Tangye, S.G.; Al-Herz, W.; Bousfiha, A.; Cunningham-Rundles, C.; Franco, J.L.; Holland, S.M.; Klein, C.; Morio, T.; Oksenhendler, E.; Picard, C.; Puel, A.; Puck, J.; Seppänen, M.R.J.; Somech, R.; Su, H.C.; Sullivan, K.E.; Torgerson, T.R.; Meyts, I. Human Inborn Errors of Immunity: 2022 Update on the Classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol 2022, 42, 1473–1507. [Google Scholar] [CrossRef] [PubMed]
- Hirschhorn, R.; Grunebaum, E.; Roifman, C.; Candotti, F. Immunodeficiency Due to Defects of Purine Metabolism: Territorial Administration under Attack in Orleans and Washington. In Primary Immunodeficiency Diseases: A Molecular and Genetic Approach, 3rd ed.; Ochs, H.D.; Smith, C.I.E.; Puck, J.M. Eds; Oxford Academic: 2013.
- Bardou, M.L.D.; Henriques, M.T.; Grumach, A.S. Inborn errors of immunity associated with characteristic phenotypes. J Pediatr (Rio J) 2021, 97 (Suppl. 1), S75–S83. [Google Scholar] [CrossRef] [PubMed]
- Brodszki, N.; Svensson, M.; van Kuilenburg, A.B.; Meijer, J.; Zoetekouw, L.; Truedsson, L.; Toporski, J. Novel Genetic Mutations in the First Swedish Patient with Purine Nucleoside Phosphorylase Deficiency and Clinical Outcome After Hematopoietic Stem Cell Transplantation with HLA-Matched Unrelated Donor. JIMD Rep 2015, 24, 83–89. [Google Scholar] [PubMed]
- Kütükçüler, N.; Bölük, E.; Tökmeci, N.; Karaca, N.E.; Azarsız, E.; Aksu, G.; Aykut, A. Recurrent infections, neurologic signs, low serum uric acid levels, and lymphopenia in childhood: Purine nucleoside phosphorylase deficiency, an emergency for infants. Turk Pediatri Ars 2020, 55, 320–327. [Google Scholar] [PubMed]
- Alasmari, B.G.; Ibrahim, F.; Wafa, S.; Alshehri, A.; Saeed, M. Purine Nucleoside Phosphorylase Deficiency: A Case Report of an Extremely Rare Disorder. Cureus 2024, 16, e76220. [Google Scholar] [CrossRef] [PubMed]
- Grunebaum, E.; Cohen, A.; Roifman, C.M. Recent advances in understanding and managing adenosine deaminase and purine nucleoside phosphorylase deficiencies. Curr Opin Allergy Clin Immunol 2013, 13, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Martín-Nalda, A.; Rivière, J.G.; Català-Besa, M.; García-Prat, M.; Parra-Martínez, A.; Martínez-Gallo, M.; Colobran, R.; Argudo-Ramírez, A.; Marín-Soria, J.L.; García-Villoria, J.; Alonso, L.; Arranz-Amo, J.A.; la Marca, G.; Soler-Palacín, P. Early Diagnosis and Treatment of Purine Nucleoside Phosphorylase (PNP) Deficiency through TREC-Based Newborn Screening. Int J Neonatal Screen 2021, 7, 62. [Google Scholar] [CrossRef] [PubMed]
- Blom, M.; Soomann, M.; Soler-Palacín, P.; Šedivá, A.; Stray-Pedersen, A.; Zetterström, R.; Speckmann, C.; Gennery, A.R.; van der Burg, M. Newborn screening for SCID and severe T lymphocytopenia in Europe. J Allergy Clin Immunol 2025, 155, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Boyarchuk, O.; Yarema, N.; Kravets, V.; Shulhai, O.; Shymanska, I.; Chornomydz, I.; Hariyan, T.; Volianska, L.; Kinash, M.; Makukh, H. Newborn screening for severe combined immunodeficiency: The results of the first pilot TREC and KREC study in Ukraine with involving of 10,350 neonates. Front Immunol 2022, 13, 999664. [Google Scholar] [CrossRef] [PubMed]
- la Marca, G.; Canessa, C.; Giocaliere, E.; Romano, F.; Malvagia, S.; Funghini, S.; Moriondo, M.; Valleriani, C.; Lippi, F.; Ombrone, D.; Della Bona, M.L.; Speckmann, C.; Borte, S.; Brodszki, N.; Gennery, A.R.; Weinacht, K.; Celmeli, F.; Pagel, J.; de Martino, M.; Guerrini, R.; Wittkowski, H.; Santisteban, I.; Bali, P.; Ikinciogullari, A.; Hershfield, M.; Notarangelo, L.D.; Resti, M.; Azzari, C. Diagnosis of immunodeficiency caused by a purine nucleoside phosphorylase defect by using tandem mass spectrometry on dried blood spots. J Allergy Clin Immunol 2014, 134, 155–159. [Google Scholar] [CrossRef] [PubMed]
- la Marca, G.; Giocaliere, E.; Malvagia, S.; Villanelli, F.; Funghini, S.; Ombrone, D.; Della Bona, M.L.; Forni, G.; Canessa, C.; Ricci, S.; Romano, F.; Guerrini, R.; Resti, M.; Azzari, C. Development and validation of a 2nd tier test for identification of purine nucleoside phosphorylase deficiency patients during expanded newborn screening by liquid chromatography-tandem mass spectrometry. Clin Chem Lab Med 2016, 54, 627–632. [Google Scholar] [CrossRef] [PubMed]
- la Marca, G.; Canessa, C.; Giocaliere, E.; Romano, F.; Duse, M.; Malvagia, S.; Lippi, F.; Funghini, S.; Bianchi, L.; Della Bona, M.L.; Valleriani, C.; Ombrone, D.; Moriondo, M.; Villanelli, F.; Speckmann, C.; Adams, S.; Gaspar, B.H.; Hershfield, M.; Santisteban, I.; Fairbanks, L.; Ragusa, G.; Resti, M.; de Martino, M.; Guerrini, R.; Azzari, C. Tandem mass spectrometry, but not T-cell receptor excision circle analysis, identifies newborns with late-onset adenosine deaminase deficiency. J Allergy Clin Immunol 2013, 131, 1604–1610. [Google Scholar] [CrossRef] [PubMed]
- Azzari, C.; la Marca, G.; Resti, M. Neonatal screening for severe combined immunodeficiency caused by an adenosine deaminase defect: a reliable and inexpensive method using tandem mass spectrometry. J Allergy Clin Immunol 2011, 127, 1394–1399. [Google Scholar] [CrossRef] [PubMed]
- Cazzorla, C.; Gragnaniello, V.; Gaiga, G.; Gueraldi, D.; Puma, A.; Loro, C.; Benetti, G.; Schiavo, R.; Porcù, E.; Burlina, A.P.; Burlina, A.B. Newborn Screening for Gaucher Disease: Parental Stress and Psychological Burden. Int J Neonatal Screen 2025, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Bush, L.W.; Levy, H.L. Past as Prologue: Predicting Potential Psychosocial-Ethical Burdens of Positive Newborn Screens as Conditions Propagate. Int J Neonatal Screen 2024, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.; Jukes, T.; Goobie, S.; DiRaimo, J.; Moran, G.; Potter, B.K.; Chakraborty, P.; Rupar, C.A.; Gannavarapu, S.; Prasad, C. Psychosocial impact on mothers receiving expanded newborn screening results. Eur J Hum Genet 2018, 26, 477–484. [Google Scholar] [CrossRef] [PubMed]
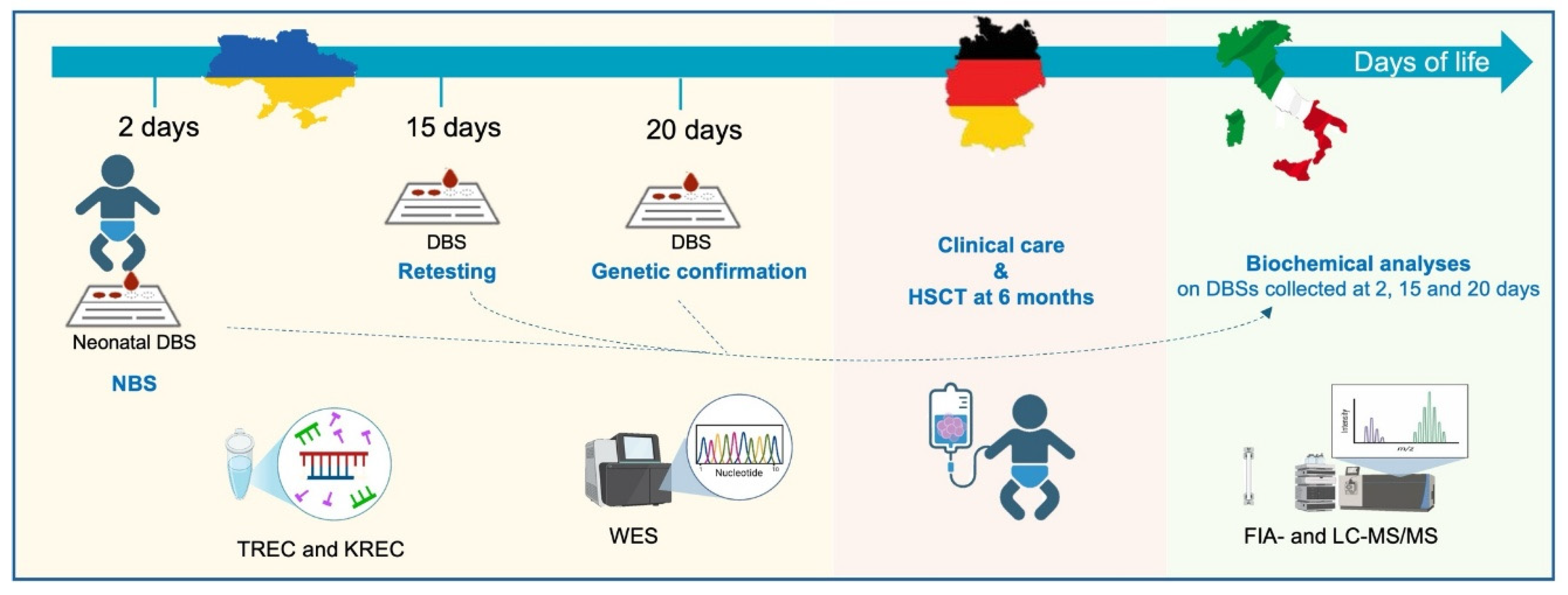
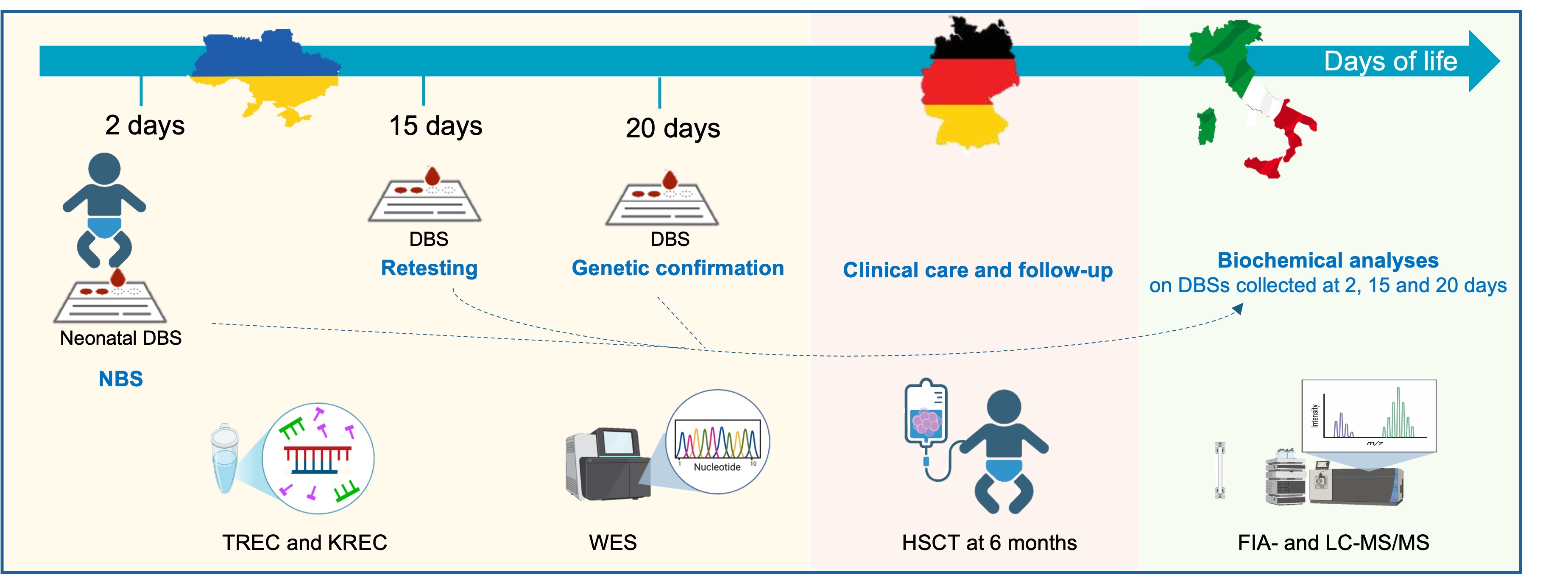
| Neonatal DBS (2 days) |
1st Retest (15 days) |
2nd Retest (20 days) |
|
|---|---|---|---|
| TREC § (n.v. > 2000 copies) | 62 | 398 | - |
| KREC § (n.v. > 2000 copies) | 23 | 17 | - |
| Whole exome sequencing (WES) § | - | - | c.751delA (p.Ser251Alafs*11)+/+ |
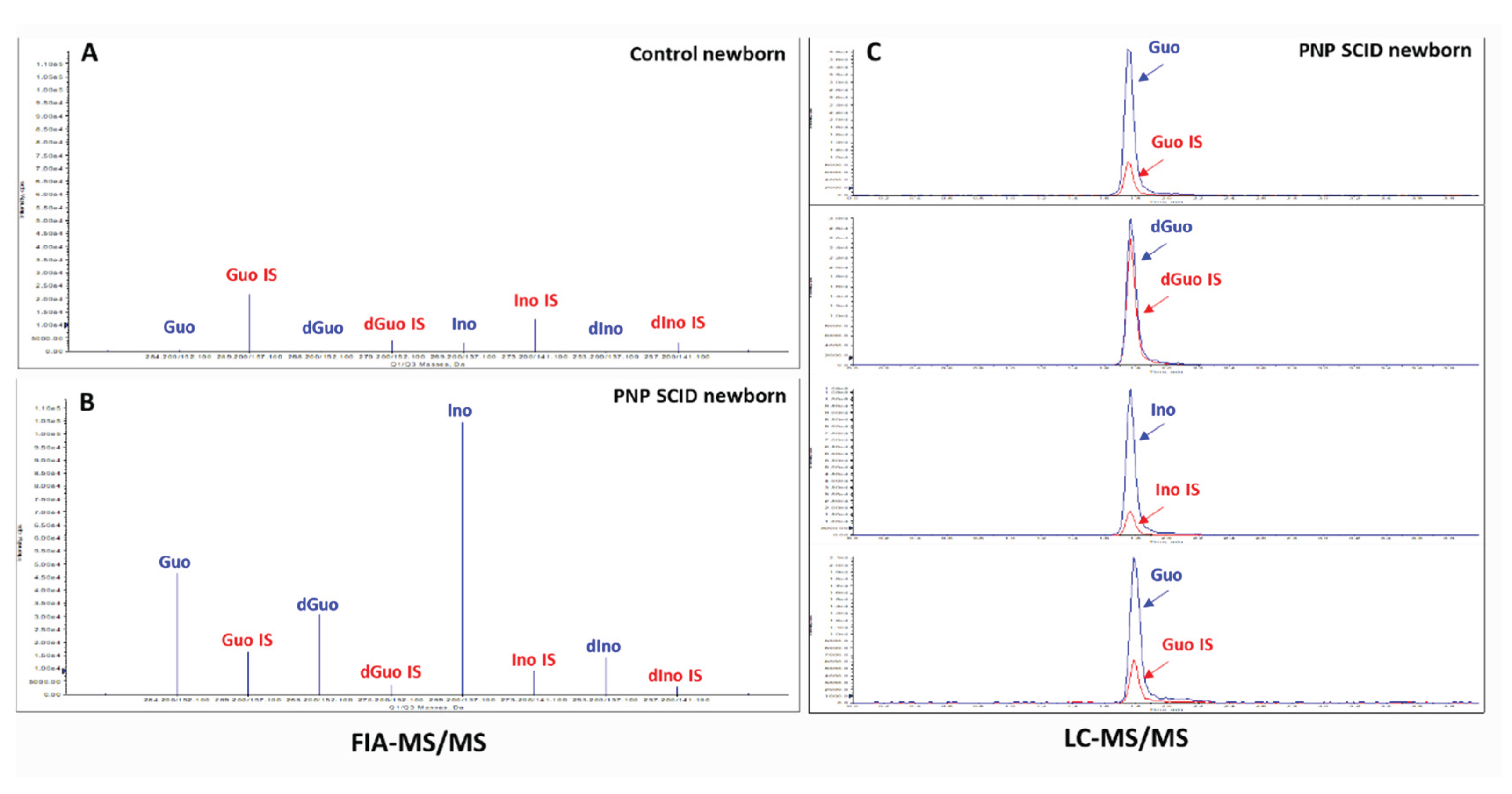
| First tiest test by FIA-MS/MS * | |||
| Guo (n.v. < 4.9 μmol/L) | 16.9 | 8.8 | 3.3 |
| dGuo (n.v. < 0.6 μmol/L) | 14.1 | 6.1 | 4.7 |
| Ino (n.v. < 61.5 μmol/L) | 67.9 | 66.8 | 97.7 |
| dIno (n.v. < 7.7 μmol/L) | 29.0 | 97.7 | 8.9 |
| Second tiest test by LC-MS/MS * | |||
| Guo (n.v. < 1.1 μmol/L) | 46.4 | 33.8 | 10.2 |
| dGuo (n.v. 0 μmol/L) | 12.5 | 6.7 | 4.3 |
| Ino (n.v. < 16.8 μmol/L) | 58.8 | 61.0 | 91.8 |
| dIno (n.v. < 0.1 μmol/L) | 36.5 | 14.5 | 10.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




